OPN Induces FoxM1 Expression and Localization through ERK 1/2, AKT, and p38 Signaling Pathway in HEC-1A Cells
Abstract
:1. Introduction
2. Results
2.1. Expression of Forkhead Box M1 (FoxM1) in Human Endometrial Tissues
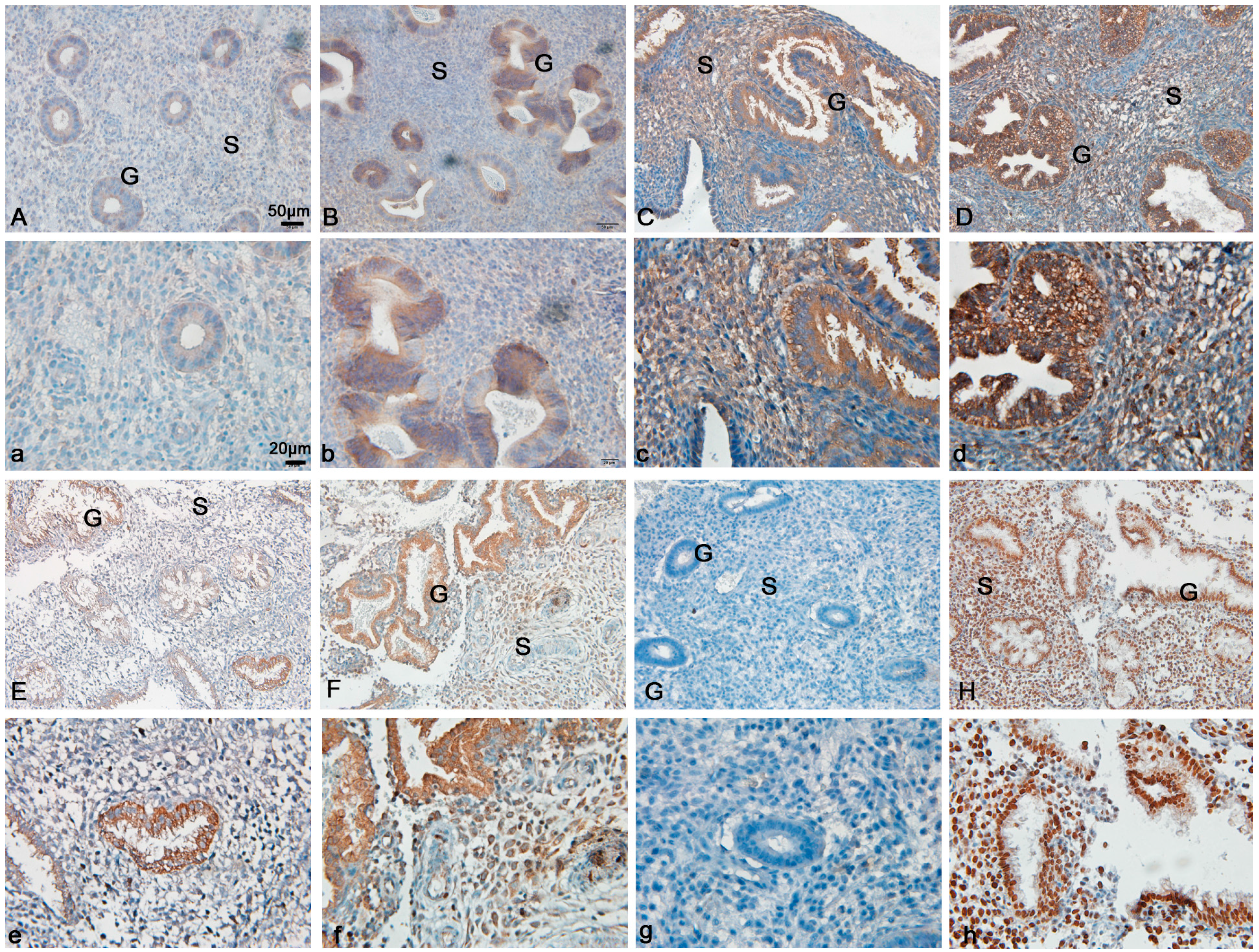
| MC | EP | MP | LP | ES | MS | LS |
|---|---|---|---|---|---|---|
| H-score | 0.12 ± 0.02 | 0.89 ± 0.09 | 1.8 ± 0.16 | 2.5 ± 0.22 | 1.4 ± 0.16 | 1.8 ± 0.20 |
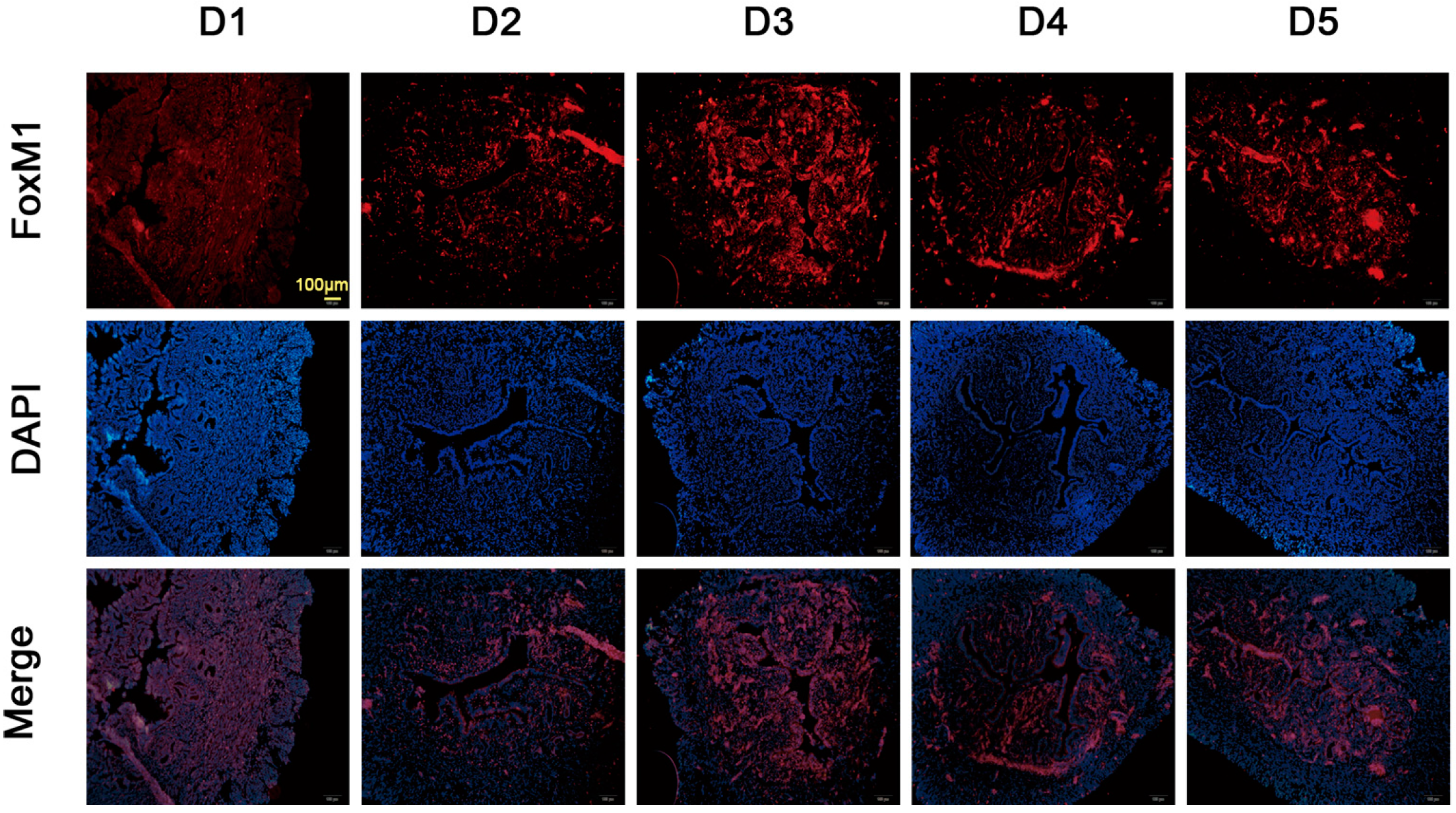
2.2. Expression of FoxM1 in Mouse Uterus during Early Pregnancy
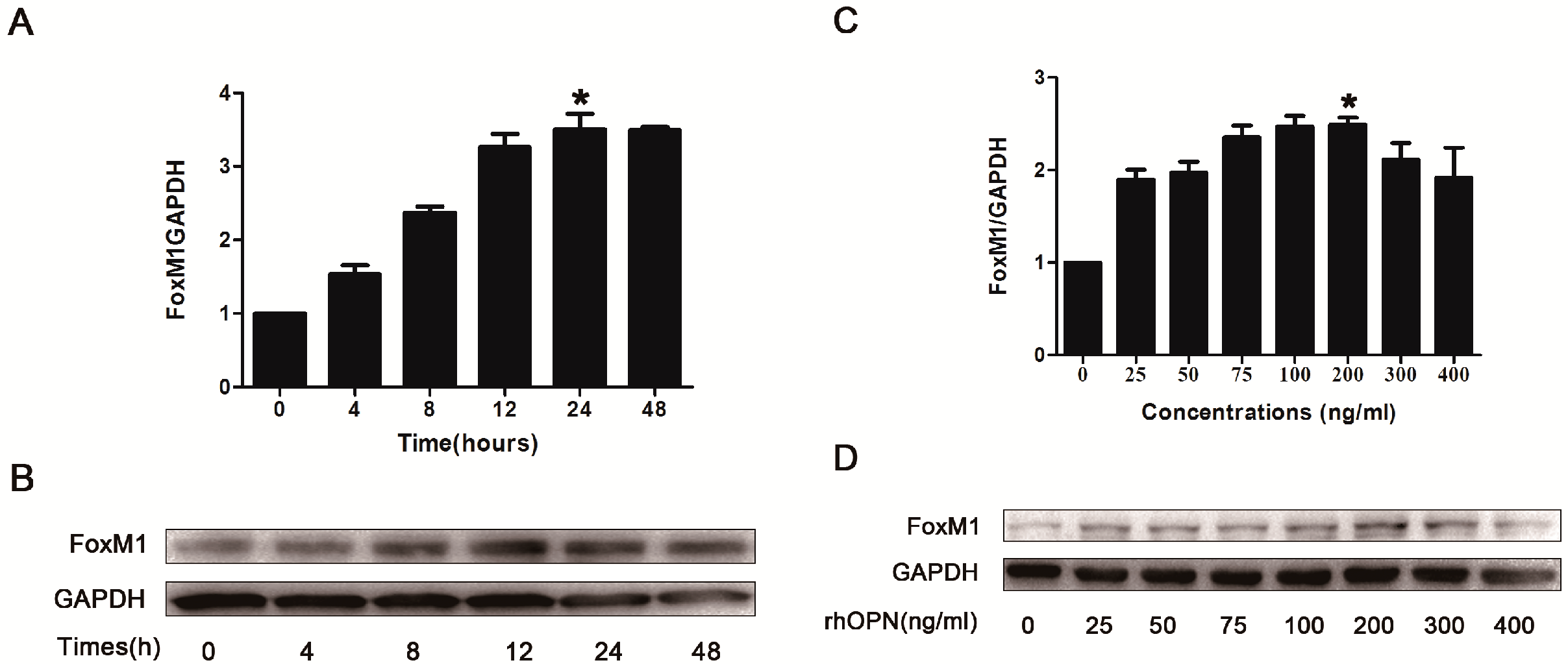
2.3. Osteopontin (OPN) Induces FoxM1 Expression in HEC-1A Cells
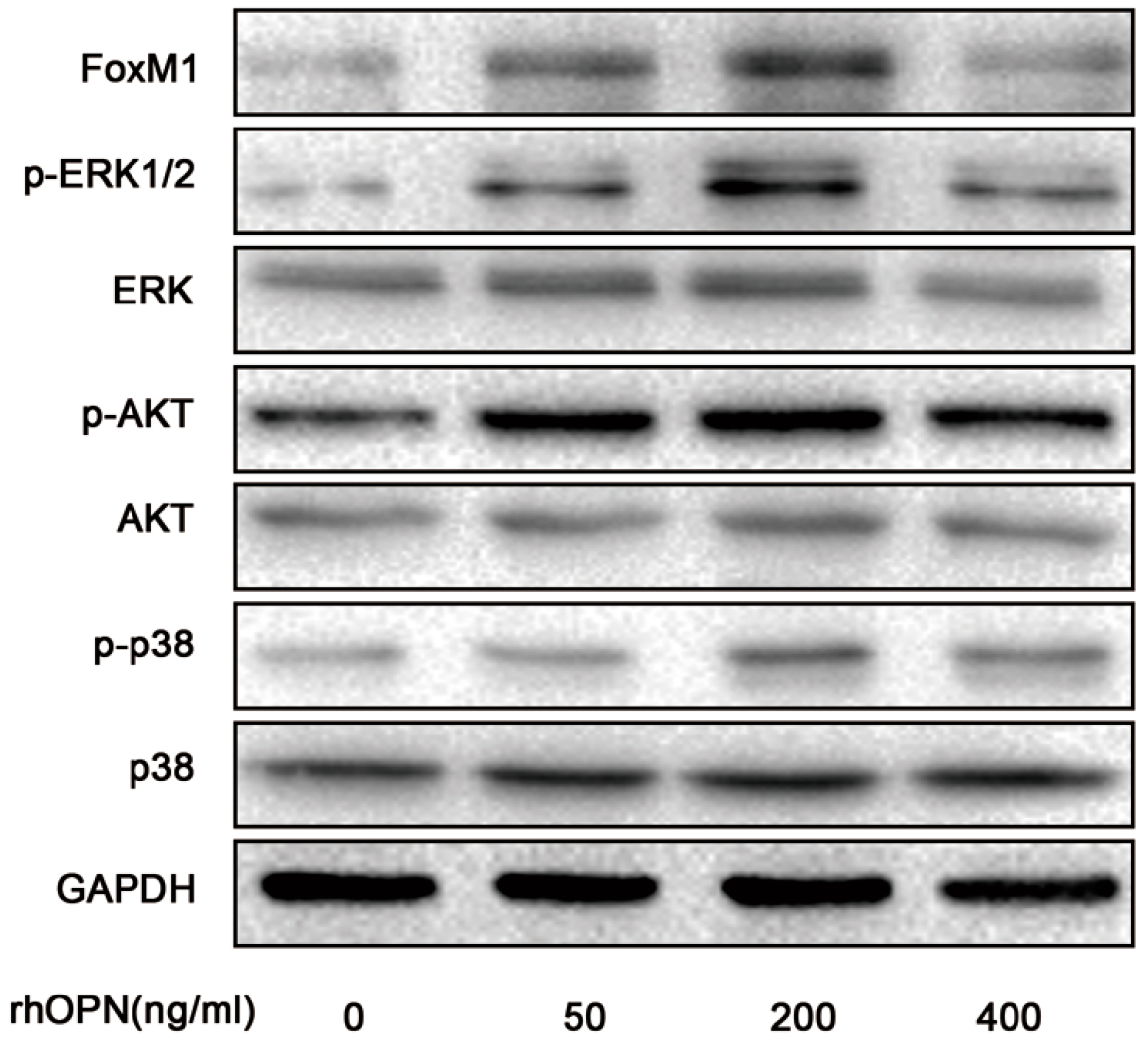
2.4. OPN Induces FoxM1 Protein Expression through Extracellular Regulated Protein Kinases (ERK 1/2), Protein Kinase B (PKB, AKT), and the p38 Mitogen Activated Protein Kinases (p38MAPK, p38) Signaling Pathway
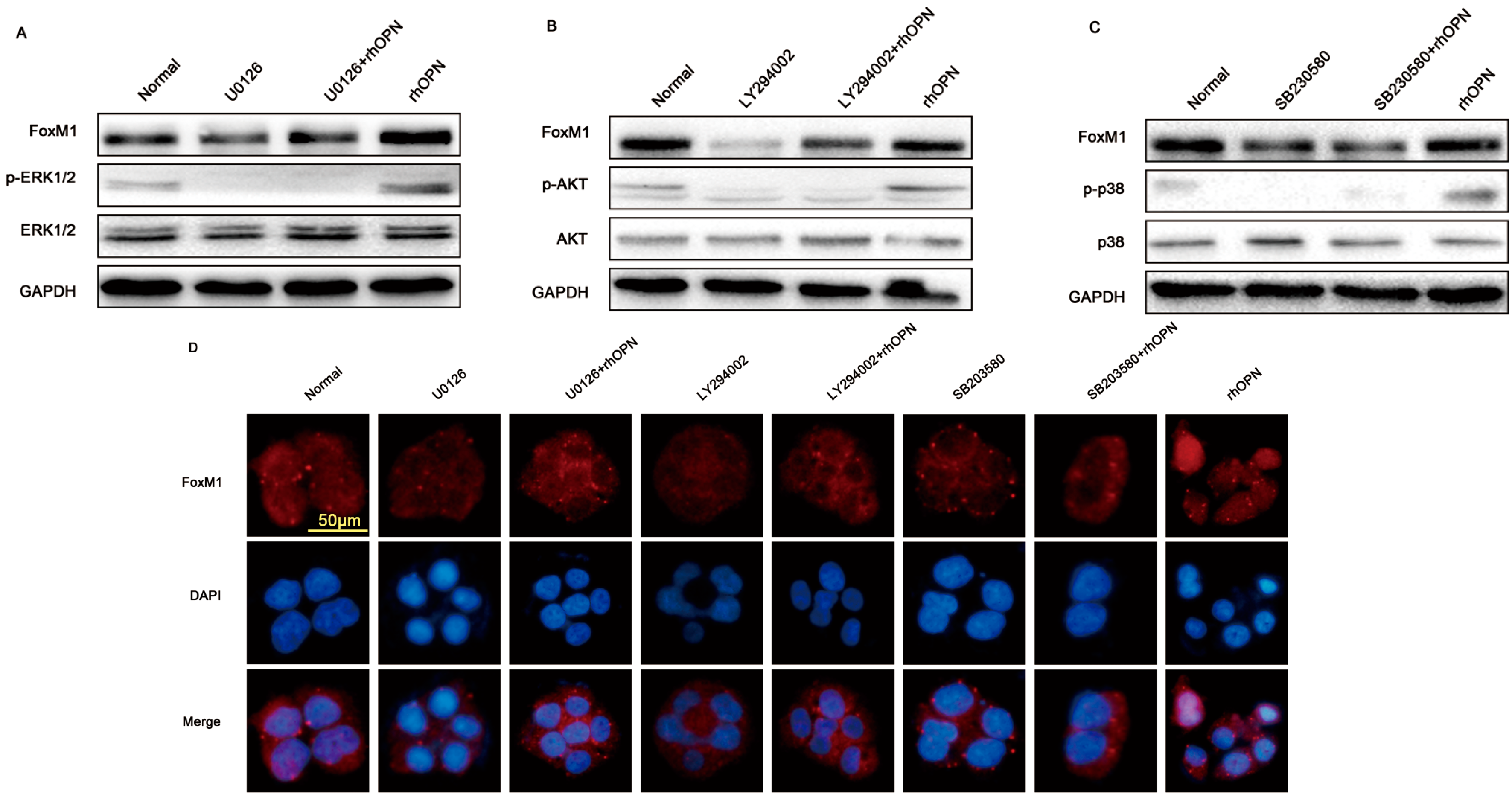
2.5. FoxM1 Regulates Cell Proliferation in HEC-1A Cells
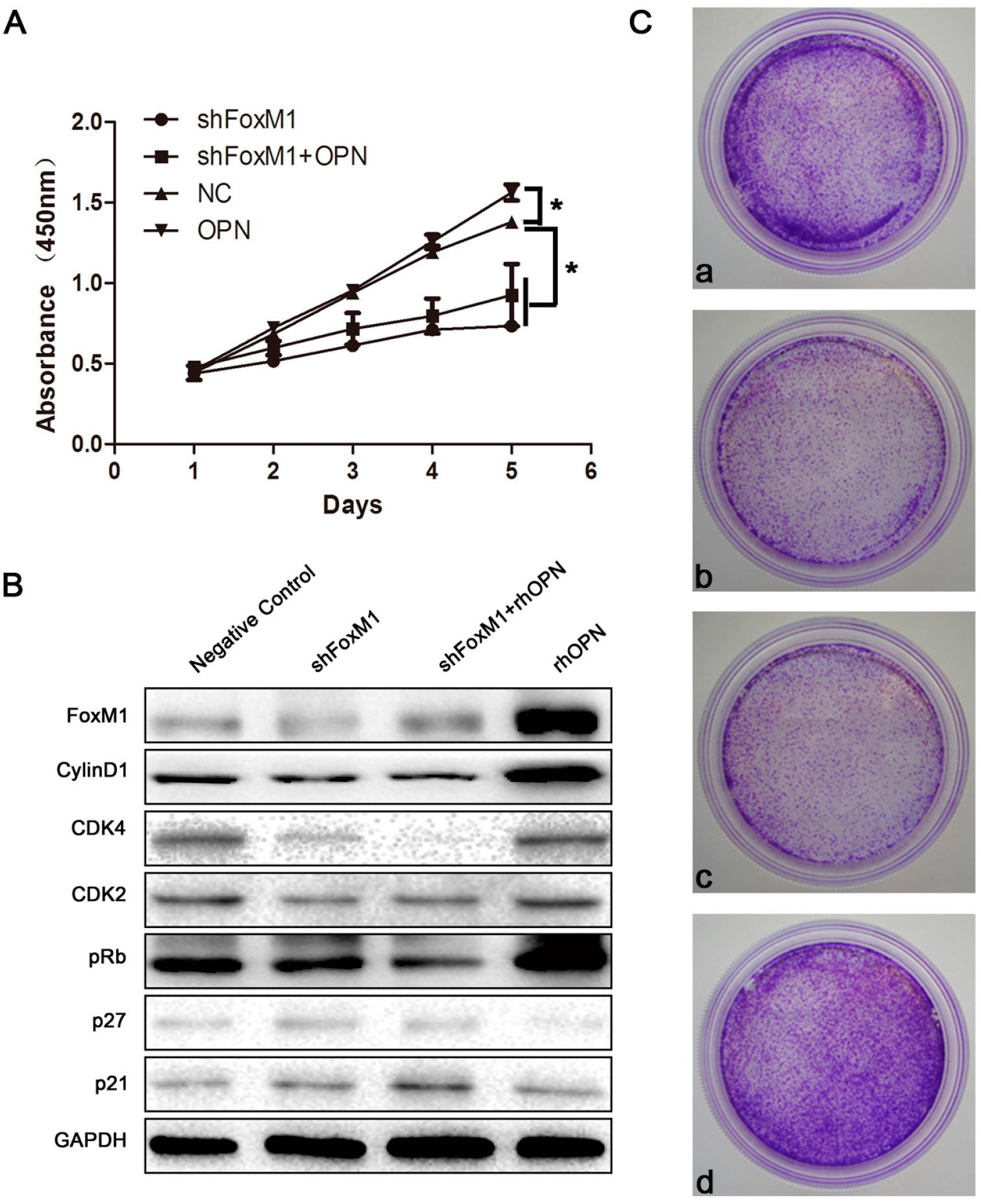
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Tissue Collection
4.3. Animals
4.4. Cell Culture
4.5. Transient Transfection
4.6. Immunohistochemistry
4.7. RNA Isolation and Real-Time PCR
4.8. Western Blot
4.9. Indirect Immunofluorescence Staining
4.10. Cell Proliferation Assay
4.11. Colony Formation Assay
4.12. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Koot, Y.E.; Teklenburg, G.; Salker, M.S.; Brosens, J.J.; Macklon, N.S. Molecular aspects of implantation failure. Biochim. Biophys. Acta 2012, 1822, 1943–1950. [Google Scholar] [CrossRef] [PubMed]
- Van Mourik, M.S.; Macklon, N.S.; Heijnen, C.J. Embryonic implantation: Cytokines, adhesion molecules, and immune cells in establishing an implantation environment. J. Leukoc. Biol. 2009, 85, 4–19. [Google Scholar]
- Johnson, G.A.; Burghardt, R.C.; Spencer, T.E.; Newton, G.R.; Ott, T.L.; Bazer, F.W. Ovine osteopontin: II. Osteopontin and αvβ3 integrin expression in the uterus and conceptus during the periimplantation period. Biol. Reprod. 1999, 61, 892–899. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Zhou, C.Q.; Chellaiah, M.A. Osteopontin and MMP9: Associations with VEGF expression/secretion and angiogenesis in PC3 prostate cancer cells. Cancers 2013, 5, 617–638. [Google Scholar] [CrossRef] [PubMed]
- Wai, P.Y.; Kuo, P.C. The role of osteopontin in tumor metastasis. J. Surg. Res. 2004, 121, 228–241. [Google Scholar] [CrossRef] [PubMed]
- DuQuesnay, R.; Wright, C.; Aziz, A.A.; Stamp, G.W.; Trew, G.H.; Margara, R.A.; White, J.O. Infertile women with isolated polycystic ovaries are deficient in endometrial expression of osteopontin but not alphavbeta3 integrin during the implantation window. Fertil. Steril. 2009, 91, 489–499. [Google Scholar] [CrossRef]
- White, F.J.; Burghardt, R.C.; Hu, J.; Joyce, M.M.; Spencer, T.E.; Johnson, G.A. Secreted phosphoprotein 1 (osteopontin) is expressed by stromal macrophages in cyclic and pregnant endometrium of mice, but is induced by estrogen in luminal epithelium during conceptus attachment for implantation. Reproduction 2006, 132, 919–929. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Li, T.; Xia, E.; Yang, X.; Sun, X.; Zhou, Y. Expression of integrin β3 and osteopontin in the eutopic endometrium of adenomyosis during the implantation window. Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 170, 419–422. [Google Scholar] [CrossRef] [PubMed]
- Johnson, G.A.; Burghardt, R.C.; Bazer, F.W.; Spencer, T.E. Osteopontin: Roles in implantation and placentation. Biol. Reprod. 2003, 69, 1458–1471. [Google Scholar] [CrossRef] [PubMed]
- Wei, Q.; St, C.J.; Fu, T.; Stratton, P.; Nieman, L.K. Reduced expression of biomarkers associated with the implantation window in women with endometriosis. Fertil. Steril. 2009, 91, 1686–1691. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Ahmad, A.; Li, Y.; Banerjee, S.; Kong, D.; Sarkar, F.H. Forkhead box M1 transcription factor: A novel target for cancer therapy. Cancer Treat. Rev. 2010, 36, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Katoh, M.; Katoh, M. Human FOX gene family (Review). Int. J. Oncol. 2004, 25, 1495–1500. [Google Scholar] [PubMed]
- Priller, M.; Poschl, J.; Abrao, L.; von Bueren, A.O.; Cho, Y.J.; Rutkowski, S.; Kretzschmar, H.A.; Schüller, U. Expression of FoxM1 is required for the proliferation of medulloblastoma cells and indicates worse survival of patients. Clin. Cancer Res. 2011, 17, 6791–6801. [Google Scholar] [CrossRef]
- Uddin, S.; Ahmed, M.; Hussain, A.; Abubaker, J.; Al-Sanea, N.; AbdulJabbar, A.; Ashari, L.H.; Alhomoud, S.; Al-Dayel, F.; Jehan, Z.; et al. Genome-wide expression analysis of Middle Eastern colorectal cancer reveals FOXM1 as a novel target for cancer therapy. Am. J. Pathol. 2011, 178, 537–547. [Google Scholar] [CrossRef]
- Xia, L.; Huang, W.; Tian, D.; Zhu, H.; Zhang, Y.; Hu, H.; Fan, D.; Nie, Y.; Wu, K. Upregulated FoxM1 expression induced by hepatitis B virus X protein promotes tumor metastasis and indicates poor prognosis in hepatitis B virus-related hepatocellular carcinoma. J. Hepatol. 2012, 57, 600–612. [Google Scholar] [CrossRef]
- Sanders, D.A.; Ross-Innes, C.S.; Beraldi, D.; Carroll, J.S.; Balasubramanian, S. Genome-wide mapping of FOXM1 binding reveals co-binding with estrogen receptor alpha in breast cancer cells. Genome Biol. 2013, 14, R6. [Google Scholar] [CrossRef] [PubMed]
- Xu, N.; Jia, D.; Chen, W.; Wang, H.; Liu, F.; Ge, H.; Zhu, X.; Song, Y.; Zhang, X.; Zhang, D.; et al. FoxM1 is associated with poor prognosis of non-small cell lung cancer patients through promoting tumor metastasis. PLoS One 2013, 8, e59412. [Google Scholar] [CrossRef]
- Perry, J.K.; Lins, R.J.; Lobie, P.E.; Mitchell, M.D. Regulation of invasive growth: Similar epigenetic mechanisms underpin tumour progression and implantation in human pregnancy. Clin. Sci. 2010, 118, 451–457. [Google Scholar] [CrossRef]
- Xie, Y.; Cui, D.; Kong, Y. FoxM1 influences embryo implantation and is regulated by 17 beta-estradiol and progesterone in mouse uteri and endometrium cells. Int. J. Clin. Exp. Pathol. 2014, 7, 6585–6595. [Google Scholar] [PubMed]
- Wang, H.; Dey, S.K. Roadmap to embryo implantation: clues from mouse models. Nat. Rev. Genet. 2006, 7, 185–199. [Google Scholar] [CrossRef] [PubMed]
- Dey, S.K.; Lim, H.; Das, S.K.; Reese, J.; Paria, B.C.; Daikoku, T.; Wang, H. Molecular cues to implantation. Endocr. Rev. 2004, 25, 341–373. [Google Scholar] [CrossRef]
- Das, S.K.; Wang, X.N.; Paria, B.C.; Damm, D.; Abraham, J.A.; Klagsbrun, M.; Andrews, G.K.; Dey, S.K. Heparin-binding EGF-like growth factor gene is induced in the mouse uterus temporally by the blastocyst solely at the site of its apposition: A possible ligand for interaction with blastocyst EGF-receptor in implantation. Development 1994, 120, 1071–1083. [Google Scholar] [PubMed]
- Wierstra, I.; Alves, J. FOXM1, a typical proliferation-associated transcription factor. Biol. Chem. 2007, 388, 1257–1274. [Google Scholar] [PubMed]
- Brar, A.K.; Frank, G.R.; Kessler, C.A.; Cedars, M.I.; Handwerger, S. Progesterone-dependent decidualization of the human endometrium is mediated by cAMP. Endocrine 1997, 6, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Carson, D.D.; Bagchi, I.; Dey, S.K.; Enders, A.C.; Fazleabas, A.T.; Lessey, B.A.; Yoshinaga, K. Embryo implantation. Dev. Biol. 2000, 223, 217–237. [Google Scholar] [CrossRef] [PubMed]
- Karadedou, C.T. Regulation of the FOXM1 transcription factor by the estrogen receptor alpha at the protein level, in breast cancer. Hippokratia 2006, 10, 128–132. [Google Scholar] [PubMed]
- Goncalves, R.F.; Chapman, D.A.; Bertolla, R.P.; Eder, I.; Killian, G.J. Pre-treatment of cattle semen or oocytes with purified milk osteopontin affects in vitro fertilization and embryo development. Anim. Reprod. Sci. 2008, 108, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Kaloglu, C.; Onarlioglu, B. Extracellular matrix remodelling in rat endometrium during early pregnancy: The role of fibronectin and laminin. Tissue Cell 2010, 42, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Chegini, N.; Rhoton-Vlasak, A.; Williams, R.S. Expression of matrix metalloproteinase-26 and tissue inhibitor of matrix metalloproteinase-3 and -4 in endometrium throughout the normal menstrual cycle and alteration in users of levonorgestrel implants who experience irregular uterine bleeding. Fertil. Steril. 2003, 80, 564–570. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Cao, Y.J.; Zhao, Y.G.; Sang, Q.X.; Duan, E.K. Expression of matrix metalloproteinase-26 and tissue inhibitor of metalloproteinase-4 in human normal cytotrophoblast cells and a choriocarcinoma cell line, JEG-3. Mol. Hum. Reprod. 2002, 8, 659–666. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Banerjee, S.; Kong, D.; Li, Y.; Sarkar, F.H. Down-regulation of Forkhead Box M1 transcription factor leads to the inhibition of invasion and angiogenesis of pancreatic cancer cells. Cancer Res. 2007, 67, 8293–8300. [Google Scholar] [CrossRef] [PubMed]
- Soufla, G.; Sifakis, S.; Baritaki, S.; Zafiropoulos, A.; Koumantakis, E.; Spandidos, D.A. VEGF, FGF2, TGFB1 and TGFBR1 mRNA expression levels correlate with the malignant transformation of the uterine cervix. Cancer Lett. 2005, 221, 105–118. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, N. Vascular endothelial growth factor. Eur. J. Cancer 1996, 32A, 2413–2422. [Google Scholar] [CrossRef]
- Halder, J.B.; Zhao, X.; Soker, S.; Paria, B.C.; Klagsbrun, M.; Das, S.K.; Dey, S.K. Differential expression of VEGF isoforms and VEGF164-specific receptor neuropilin-1 in the mouse uterus suggests a role for VEGF164 in vascular permeability and angiogenesis during implantation. Genesis 2000, 26, 213–224. [Google Scholar]
- Daikoku, T.; Matsumoto, H.; Gupta, R.A.; Das, S.K.; Gassmann, M.; DuBois, R.N.; Dey, S.K. Expression of hypoxia-inducible factors in the peri-implantation mouse uterus is regulated in a cell-specific and ovarian steroid hormone-dependent manner. Evidence for differential function of HIFs during early pregnancy. J. Biol. Chem. 2003, 278, 7683–7691. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, N.; Jia, Z.; Le, X.; Dai, B.; Wei, D.; Huang, S.; Tan, D.; Xie, K. Critical role and regulation of transcription factor FoxM1 in human gastric cancer angiogenesis and progression. Cancer Res. 2009, 69, 3501–3509. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, N.; Dai, B.; Liu, M.; Sawaya, R.; Xie, K.; Huang, S. FoxM1B transcriptionally regulates vascular endothelial growth factor expression and promotes the angiogenesis and growth of glioma cells. Cancer Res. 2008, 68, 8733–8742. [Google Scholar] [CrossRef]
- Zhang, H.; Guo, M.; Chen, J.H.; Wang, Z.; Du, X.-F.; Liu, P.-X.; Li, W.-H. Osteopontin knockdown inhibits αVβ3 integrin-induced cell migration and invasion and promotes apoptosis of breast cancer cells by inducing autophagy and inactivating the PI3K/Akt/mTOR pathway. Cell. Physiol. Biochem. 2014, 33, 991–1002. [Google Scholar] [CrossRef]
- Kale, S.; Raja, R.; Thorat, D.; Soundararajan, G.; Patil, T.V.; Kundu, G.C. Osteopontin signaling upregulates cyclooxygenase-2 expression in tumor-associated macrophages leading to enhanced angiogenesis and melanoma growth via α9β1 integrin. Oncogene 2014, 33, 2295–2306. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xie, Y.; Li, Y.; Kong, Y. OPN Induces FoxM1 Expression and Localization through ERK 1/2, AKT, and p38 Signaling Pathway in HEC-1A Cells. Int. J. Mol. Sci. 2014, 15, 23345-23358. https://doi.org/10.3390/ijms151223345
Xie Y, Li Y, Kong Y. OPN Induces FoxM1 Expression and Localization through ERK 1/2, AKT, and p38 Signaling Pathway in HEC-1A Cells. International Journal of Molecular Sciences. 2014; 15(12):23345-23358. https://doi.org/10.3390/ijms151223345
Chicago/Turabian StyleXie, Yunpeng, Yinghua Li, and Ying Kong. 2014. "OPN Induces FoxM1 Expression and Localization through ERK 1/2, AKT, and p38 Signaling Pathway in HEC-1A Cells" International Journal of Molecular Sciences 15, no. 12: 23345-23358. https://doi.org/10.3390/ijms151223345
APA StyleXie, Y., Li, Y., & Kong, Y. (2014). OPN Induces FoxM1 Expression and Localization through ERK 1/2, AKT, and p38 Signaling Pathway in HEC-1A Cells. International Journal of Molecular Sciences, 15(12), 23345-23358. https://doi.org/10.3390/ijms151223345




