Discordance Rate of HER2 Status in Primary Gastric Carcinomas and Synchronous Lymph Node Metastases: A Multicenter Retrospective Analysis
Abstract
:1. Introduction
2. Results
| Parameter | Number | HER2 | p | |
|---|---|---|---|---|
| Amplified | Not Amplified | |||
| Gender | ||||
| Male | 67 | 13 | 54 | 0.4781 (NS) |
| Female | 41 | 5 | 36 | |
| Site | ||||
| Lower | 52 | 10 | 42 | 0.7471 (NS) |
| Middle | 38 | 5 | 33 | |
| Upper | 18 | 3 | 15 | |
| Lauren Histotype | ||||
| Intestinal | 62 | 15 | 47 | 0.0496 |
| Diffuse | 35 | 2 | 33 | |
| Mixed | 11 | 1 | 10 | |
| WHO histotype | ||||
| Tubular | 62 | 15 | 47 | 0.0496 |
| Poorly cohesive | 35 | 2 | 33 | |
| Mixed | 11 | 1 | 10 | |
| Grade | ||||
| Low | 55 | 10 | 45 | 0.8633 (NS) |
| High | 53 | 8 | 45 | |
| Stage | ||||
| I–II | 40 | 7 | 33 | 0.929 (NS) |
| III–IV | 68 | 11 | 57 | |
| T | ||||
| 1–2 | 15 | 5 | 10 | 0.1354 (NS) |
| 3–4 | 93 | 13 | 80 | |
| N | ||||
| 1 | 39 | 5 | 34 | 0.5909 (NS) |
| 2–3 | 69 | 13 | 56 | |
| Parameter | Discordant GC | Concordant GC | p |
|---|---|---|---|
| Gender | |||
| Male | 9 | 58 | 0.1162 (NS) |
| Female | 1 | 40 | |
| Site | |||
| Lower | 5 | 47 | 0.1281 (NS) |
| Middle | 1 | 34 | |
| Upper | 4 | 17 | |
| Lauren Histotype | |||
| Intestinal | 5 | 57 | 0.5578 (NS) |
| Diffuse | 3 | 32 | |
| Mixed | 2 | 9 | |
| WHO histotype | |||
| Tubular | 5 | 57 | 0.5578 (NS) |
| Poorly cohesive | 3 | 32 | |
| Mixed | 2 | 9 | |
| Grade | |||
| Low | 6 | 49 | 0.7867 (NS) |
| High | 4 | 49 | |
| Stage | |||
| I–II | 4 | 36 | 0.8886 (NS) |
| III–IV | 6 | 62 | |
| T | |||
| 1–2 | 3 | 12 | 0.2862 (NS) |
| 3–4 | 7 | 86 | |
| N | |||
| 1 | 4 | 35 | 0.9388 (NS) |
| 2–3 | 6 | 63 | |
| Sex | Stage | pT | pN | Histotype | Grade | Primary GC | Metastatic LN |
|---|---|---|---|---|---|---|---|
| M | III | 3 | 3 | Mixed | High | 3+ | 0 |
| F | III | 2 | 3 | Intestinal | Low | 3+ | 0 |
| M | II | 2 | 1 | Intestinal | Low | 3+ | 1+ |
| M | III | 2 | 3 | Intestinal | Low | 3+ | 0 |
| M | II | 3 | 1 | Mixed | Low | 3+ | 0 |
| M | III | 3 | 3 | Intestinal | High | 3+ | 0 |
| M | II | 3 | 1 | Mixed | Low | 0 | 3+ |
| M | II | 3 | 1 | Diffuse | Low | 2+ * | 3+ |
| F | III | 3 | 3 | Intestinal | High | 0 | 3+ |
| F | IV | 3 | 3 | Diffuse | High | 0 | 3+ |
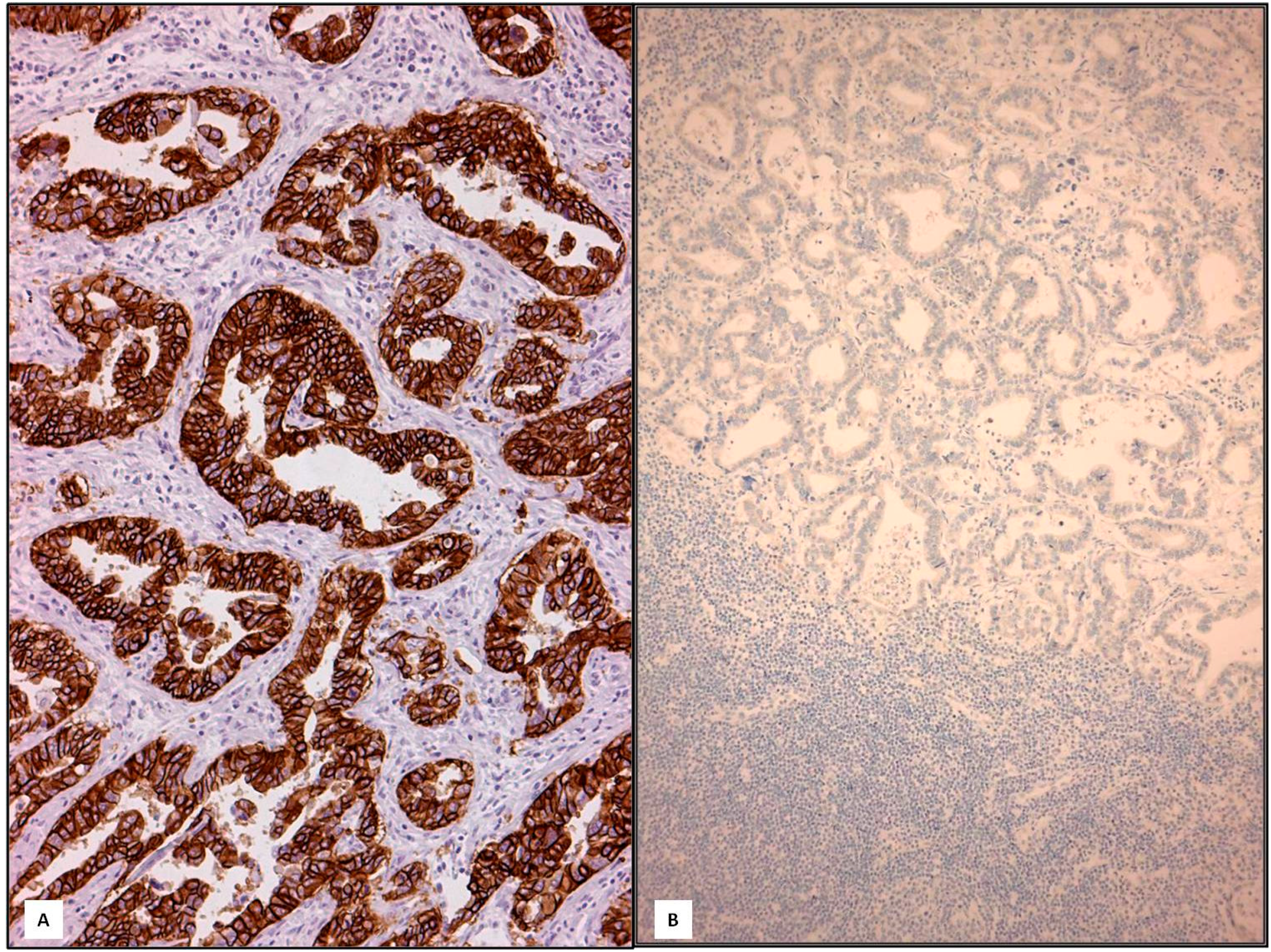
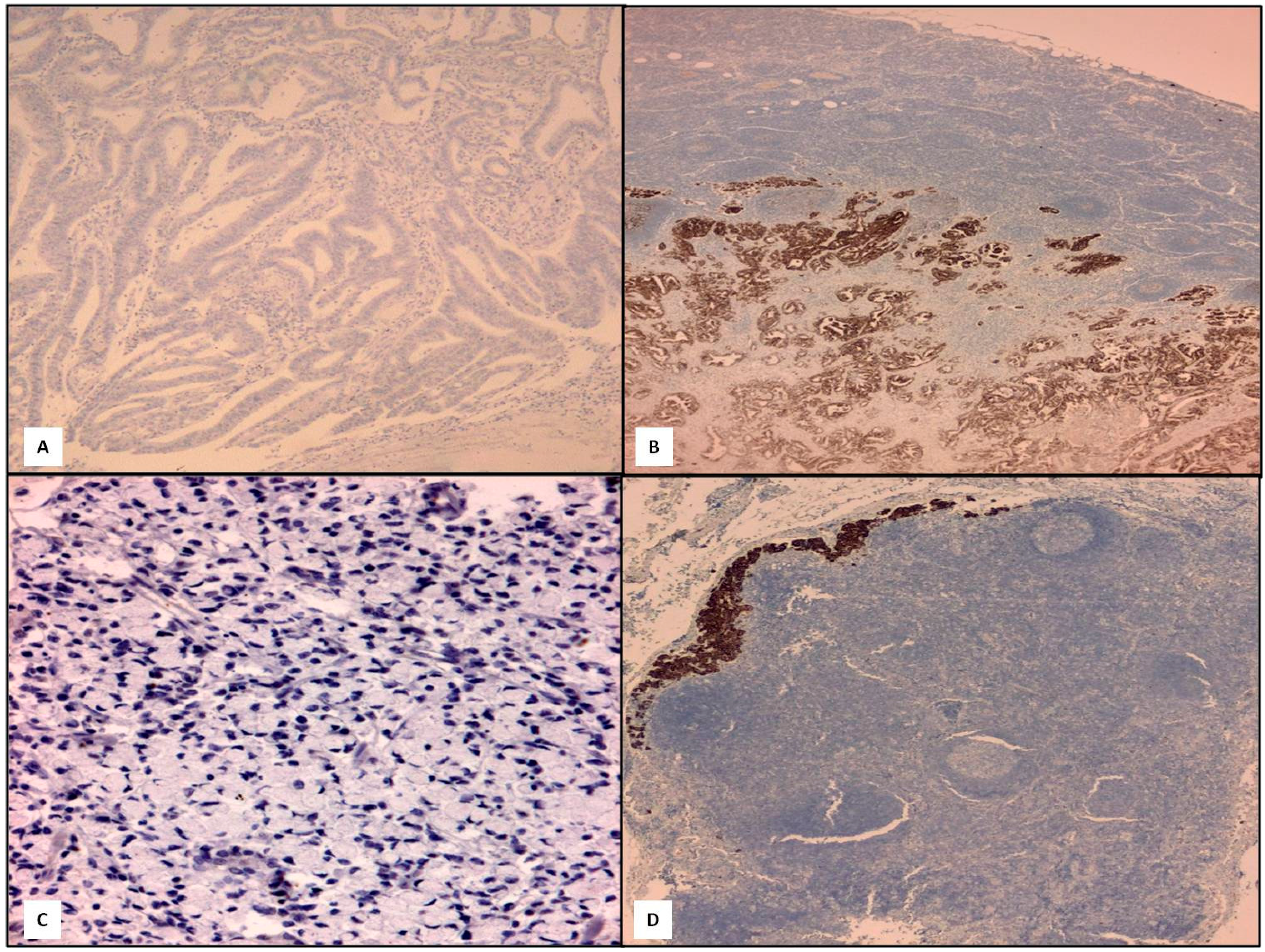
3. Discussion
4. Materials and Methods
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Suzuki, M.; Tarin, D. Gene expression profiling of human lymph node metastases and matched primary breast carcinomas: Clinical implications. Mol. Oncol. 2007, 1, 172–180. [Google Scholar]
- Ki, D.H.; Jeung, H.C.; Park, C.H.; Kang, S.H.; Lee, G.Y.; Lee, W.S.; Kim, N.K.; Chung, H.C.; Rha, S.Y. Whole genome analysis for liver metastasis gene signatures in colorectal cancer. Int. J. Cancer 2007, 121, 2005–2012. [Google Scholar]
- Feng, Y.; Sun, B.; Li, X.; Zhang, L.; Niu, Y.; Xiao, C.; Ning, L.; Fang, Z.; Wang, Y.; Zhang, L.; et al. Differentially expressed genes between primary cancer and paired lymph node metastases predict clinical outcome of node-positive breast cancer patients. Breast Cancer Res. Treat. 2007, 103, 125–127. [Google Scholar]
- Vecchi, M.; Confalonieri, S.; Nuciforo, P.; Viganò, M.A.; Capra, M.; Bianchi, M.; Nicosia, D.; Bianchi, F.; Galimberti, V.; Viale, G.; et al. Breast cancer metastases are molecularly distinct from their primary tumors. Oncogene 2008, 27, 2148–2158. [Google Scholar]
- Stoecklein, N.H.; Klein, C.A. Genetic disparity between primary tumours, disseminated tumour cells, and manifest metastasis. Int. J. Cancer 2010, 126, 5895–5898. [Google Scholar]
- Santinelli, A.; Pisa, E.; Stramazzotti, D.; Fabris, G. HER2 status discrepancy between primary breast cancer and metastatic sites. Impact on target therapy. Int. J. Cancer 2008, 122, 999–1004. [Google Scholar]
- Aitken, S.J.; Thomas, J.S.; Langdon, S.P.; Harrison, D.J.; Faratian, D. Quantitative analysis of changes in ER, PR and HER2 expression in primary breast cancer and paired nodal metastases. Ann. Oncol. 2010, 21, 1254–1261. [Google Scholar]
- Strien, L.; Leidenius, M.; von Smitten, K.; Heikkilä, P. Concordance between HER2 and steroid hormone receptor expression between primary breast cancer, sentinel node metastases, and isolated tumor cells. Pathol. Res. Pract. 2010, 206, 253–258. [Google Scholar]
- Chan, A.; Morey, A.; Brown, B.; Hastrich, D.; Willsher, P.; Ingram, D. A retrospective study investigating the rate of HER2 discordance between primary breast carcinoma and locoregional or metastatic disease. BMC Cancer 2012, 12, 555. [Google Scholar]
- Dieci, M.V.; Barbieri, E.; Piacentini, F.; Ficarra, G.; Bettelli, S.; Dominici, M.; Conte, P.F.; Guarneri, V. Discordance in receptor status between primary and recurrent breast cancer has a prognostic impact: A single-institution analysis. Ann. Oncol. 2013, 24, 101–108. [Google Scholar]
- Aurilio, G.; Disalvatore, D.; Pruneri, G.; Bagnardi, V.; Viale, G.; Curigliano, G.; Adamoli, L.; Munzone, E.; Sciandivasci, A.; de Vita, F.; et al. A meta-analysis of oestrogen receptor, progesterone receptor and human epidermal growth factor receptor 2 discordance between primary breast cancer and metastases. Eur. J. Cancer 2014, 50, 277–289. [Google Scholar]
- Jensen, J.D.; Laenkholm, A.V.; Knoop, A.; Ewertz, M.; Bandaru, R.; Liu, W.; Hackl, W.; Barrett, J.C.; Gardner, H. PIK3CA mutations may be discordant between primary and corresponding metastatic disease in breast cancer. Clin. Cancer Res. 2011, 17, 667–677. [Google Scholar]
- Ataseven, B.; Gologan, D.; Gunesch, A.; Kehl, V.; Hoegel, B.; Beer, M.; Eiermann, W. HER2/neu, topoisomerase 2a, estrogen and progesterone receptors: Discordance between primary breast cancer and metastatic axillary lymph node in expression and amplification characteristics. Breast Care 2012, 7, 465–470. [Google Scholar]
- Falck, A.K.; Fernö, M.; Bendahl, P.O.; Rydén, L. St Gallen molecular subtypes in primary breast cancer and matched lymph node metastases—Aspects on distribution and prognosis for patients with luminal A tumours: Results from a prospective randomised trial. BMC Cancer 2013, 13, 558. [Google Scholar]
- Ieni, A.; Barresi, V.; Giuffrè, G.; Caltabiano, R.; Cascone, A.; del Sordo, R.; Cabibi, D.; Zeppa, P.; Lanzafame, S.; Sidoni, A.; et al. A meta-analysis of oestrogen receptor, progesterone receptor and human epidermal growth factor receptor 2 discordance between primary breast cancer and metastases. Eur. J. Cancer 2014, 50, 1035–1037. [Google Scholar]
- Yao, Z.X.; Lu, L.J.; Wang, R.J.; Jin, L.B.; Liu, S.C.; Li, H.Y.; Ren, G.S.; Wu, K.N.; Wang, D.L.; Kong, L.Q. Discordance and clinical significance of ER, PR, and HER2 status between primary breast cancer and synchronous axillary lymph node metastasis. Med. Oncol. 2014, 31, 798. [Google Scholar]
- Kim, J.H.; Kim, M.A.; Lee, H.S.; Kim, W.H. Comparative analysis of protein expressions in primary and metastatic gastric carcinomas. Hum. Pathol. 2009, 40, 314–322. [Google Scholar]
- Rüschoff, J.; Dietel, M.; Baretton, G.; Arbogast, S.; Walch, A.; Monges, G.; Chenard, M.P.; Penault-Llorca, F.; Nagelmeier, I.; Schlake, W.; et al. HER2 diagnostics in gastric cancer-guideline validation and development of standardized immunohistochemical testing. Virchows Arch. 2010, 457, 299–307. [Google Scholar]
- Kim, M.A.; Lee, H.J.; Yang, H.K.; Bang, Y.J.; Kim, W.H. Heterogeneous amplification of ERBB2 in primary lesions is responsible for the discordant ERBB2 status of primary and metastatic lesions in gastric carcinoma. Histopathology 2011, 59, 822–831. [Google Scholar]
- Rüschoff, J.; Hanna, W.; Bilous, M.; Hofmann, M.; Osamura, R.Y.; Penault-Llorca, F.; van de Vijver, M.; Viale, G. HER2 testing in gastric cancer: A practical approach. Mod. Pathol. 2012, 25, 637–650. [Google Scholar]
- Giuffrè, G.; Ieni, A.; Barresi, V.; Caruso, R.A.; Tuccari, G. HER2 status in unusual histological variants of gastric adenocarcinomas. J. Clin. Pathol. 2012, 65, 237–241. [Google Scholar]
- Barresi, V.; Giuffrè, G.; Caruso, R.A.; Tuccari, G. HER2 status in rarer histologic types of gastric adenocarcinomas. Arch. Pathol. Lab. Med. 2013, 137, 741. [Google Scholar]
- Marx, A.H.; Tharun, L.; Muth, J.; Dancau, A.M.; Simon, R.; Yekebas, E.; Kaifi, J.T.; Mirlacher, M.; Brümmendorf, T.H.; Bokemeyer, C.; et al. HER2 amplification is highly homogenous in gastric cancer. Hum. Pathol. 2009, 40, 769–777. [Google Scholar]
- Bozzetti, C.; Negri, F.V.; Lagrasta, C.A.; Crafa, P.; Bassano, C.; Tamagnini, I.; Gardini, G.; Nizzoli, R.; Leonardi, F.; Gasparro, D.; et al. Comparison of HER2 status in primary and paired metastatic sites of gastric carcinoma. Br. J. Cancer 2011, 104, 1372–1376. [Google Scholar]
- Kochi, M.; Fujii, M.; Masuda, S.; Kanamori, N.; Mihara, Y.; Funada, T.; Tamegai, H.; Watanabe, M.; Suda, H.; Takayama, T. Differing deregulation of HER2 in primary gastric cancer and synchronous related metastatic lymph nodes. Diagn. Pathol. 2013, 8, 191. [Google Scholar]
- Hofmann, M.; Stoss, O.; Shi, D.; Büttner, R.; van de Vijver, M.; Kim, W.; Ochiai, A.; Rüschoff, J.; Henkel, T. Assessment of a HER2 scoring system for gastric cancer: Results from a validation study. Histopathology 2008, 52, 797–805. [Google Scholar]
- Jouret-Mourin, A.; Hoorens, A.; de Hertogh, G.; Vanderveken, J.; Demetter, P.; van Cutsem, E. Analysis of HER2 expression and gene amplification in adenocarcinoma of the stomach and the gastro-oesophageal junction: Rationale for the Belgian way of working. Acta Gastro-Enterol. Belg. 2012, 75, 9–13. (In Belgium) [Google Scholar]
- Ieni, A.; Barresi, V.; Giuffrè, G.; Caruso, R.A.; Lanzafame, S.; Villari, L.; Salomone, E.; Roz, E.; Cabibi, D.; Franco, V.; et al. HER2 status in advanced gastric carcinoma: A retrospective multicentric analysis from Sicily. Oncol. Lett. 2013, 6, 1591–1594. [Google Scholar]
- Pérez-Fidalgo, J.A.; Eroles, P.; Ferrer, J.; Bosch, A.; Burgués, O.; Martínez, F.; Bermejo, B.; Lluch, A.; González-Angulo, A.M. An evaluation of the impact of technical bias on the concordance rate between primary and recurrent tumors in breast cancer. Breast 2013, 22, 974–979. [Google Scholar]
- Turner, N.H.; di Leo, A. HER2 discordance between primary and metastatic breast cancer: Assessing the clinical impact. Cancer Treat. Rev. 2013, 39, 947–957. [Google Scholar]
- Ieni, A.; Barresi, V.; Caltabiano, R.; Cascone, A.M.; del Sordo, R.; Cabibi, D.; Zeppa, P.; Lanzafame, S.; Sidoni, A.; Franco, V.; et al. Discordance rate of HER2 status in primary breast carcinomas versus synchronous axillary lymph node metastases: A multicenter retrospective investigation. OncoTargets Ther. 2014, 7, 1–6. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ieni, A.; Barresi, V.; Caltabiano, R.; Caleo, A.; Bonetti, L.R.; Lanzafame, S.; Zeppa, P.; Caruso, R.A.; Tuccari, G. Discordance Rate of HER2 Status in Primary Gastric Carcinomas and Synchronous Lymph Node Metastases: A Multicenter Retrospective Analysis. Int. J. Mol. Sci. 2014, 15, 22331-22341. https://doi.org/10.3390/ijms151222331
Ieni A, Barresi V, Caltabiano R, Caleo A, Bonetti LR, Lanzafame S, Zeppa P, Caruso RA, Tuccari G. Discordance Rate of HER2 Status in Primary Gastric Carcinomas and Synchronous Lymph Node Metastases: A Multicenter Retrospective Analysis. International Journal of Molecular Sciences. 2014; 15(12):22331-22341. https://doi.org/10.3390/ijms151222331
Chicago/Turabian StyleIeni, Antonio, Valeria Barresi, Rosario Caltabiano, Alessia Caleo, Luca Reggiani Bonetti, Salvatore Lanzafame, Pio Zeppa, Rosario Alberto Caruso, and Giovanni Tuccari. 2014. "Discordance Rate of HER2 Status in Primary Gastric Carcinomas and Synchronous Lymph Node Metastases: A Multicenter Retrospective Analysis" International Journal of Molecular Sciences 15, no. 12: 22331-22341. https://doi.org/10.3390/ijms151222331
APA StyleIeni, A., Barresi, V., Caltabiano, R., Caleo, A., Bonetti, L. R., Lanzafame, S., Zeppa, P., Caruso, R. A., & Tuccari, G. (2014). Discordance Rate of HER2 Status in Primary Gastric Carcinomas and Synchronous Lymph Node Metastases: A Multicenter Retrospective Analysis. International Journal of Molecular Sciences, 15(12), 22331-22341. https://doi.org/10.3390/ijms151222331







