Depletion of C3orf1/TIMMDC1 Inhibits Migration and Proliferation in 95D Lung Carcinoma Cells
Abstract
:1. Introduction
2. Results
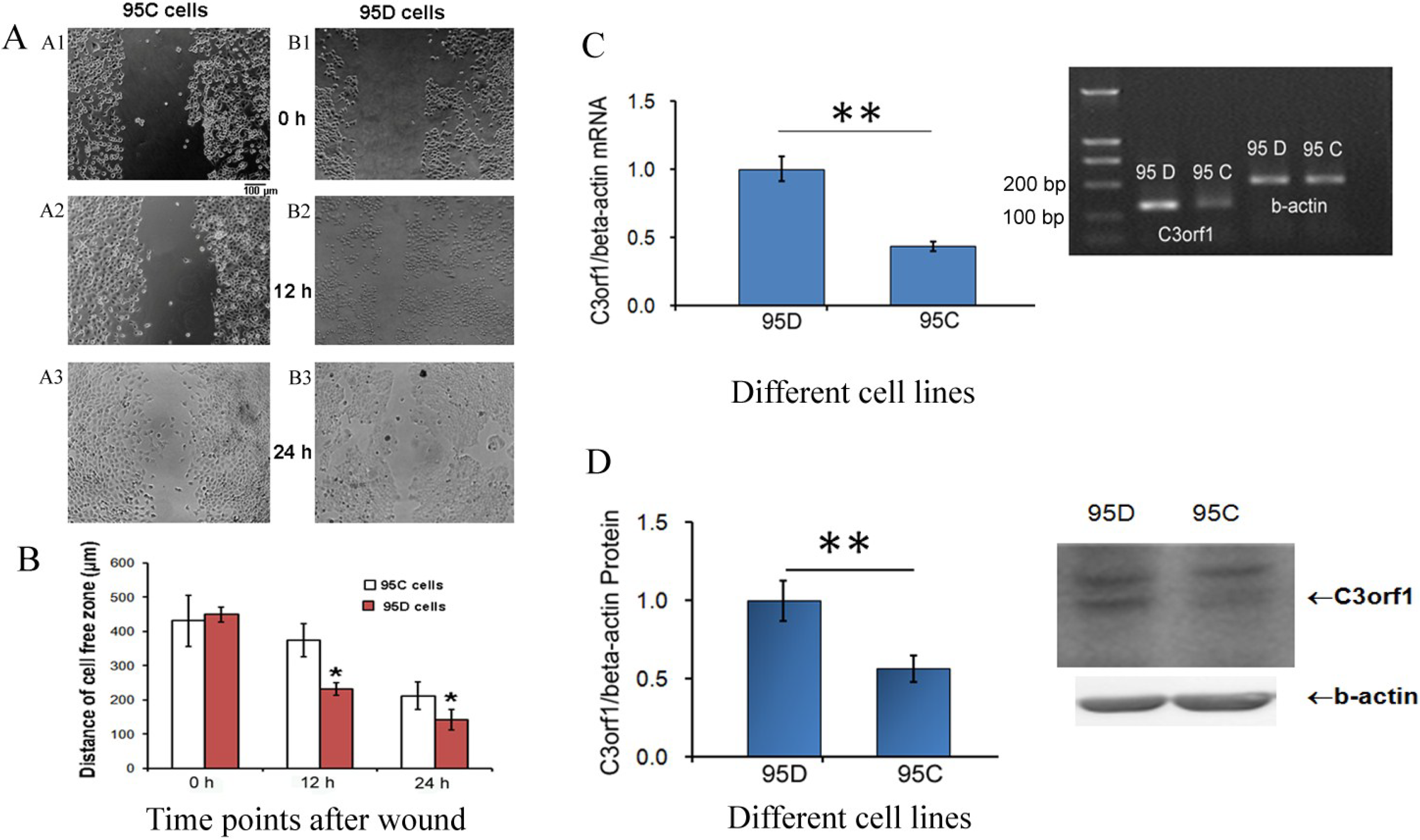
2.1. C3orf1 Gene Expression Is Higherin 95D Cells than in 95C Lung Carcinoma Cells
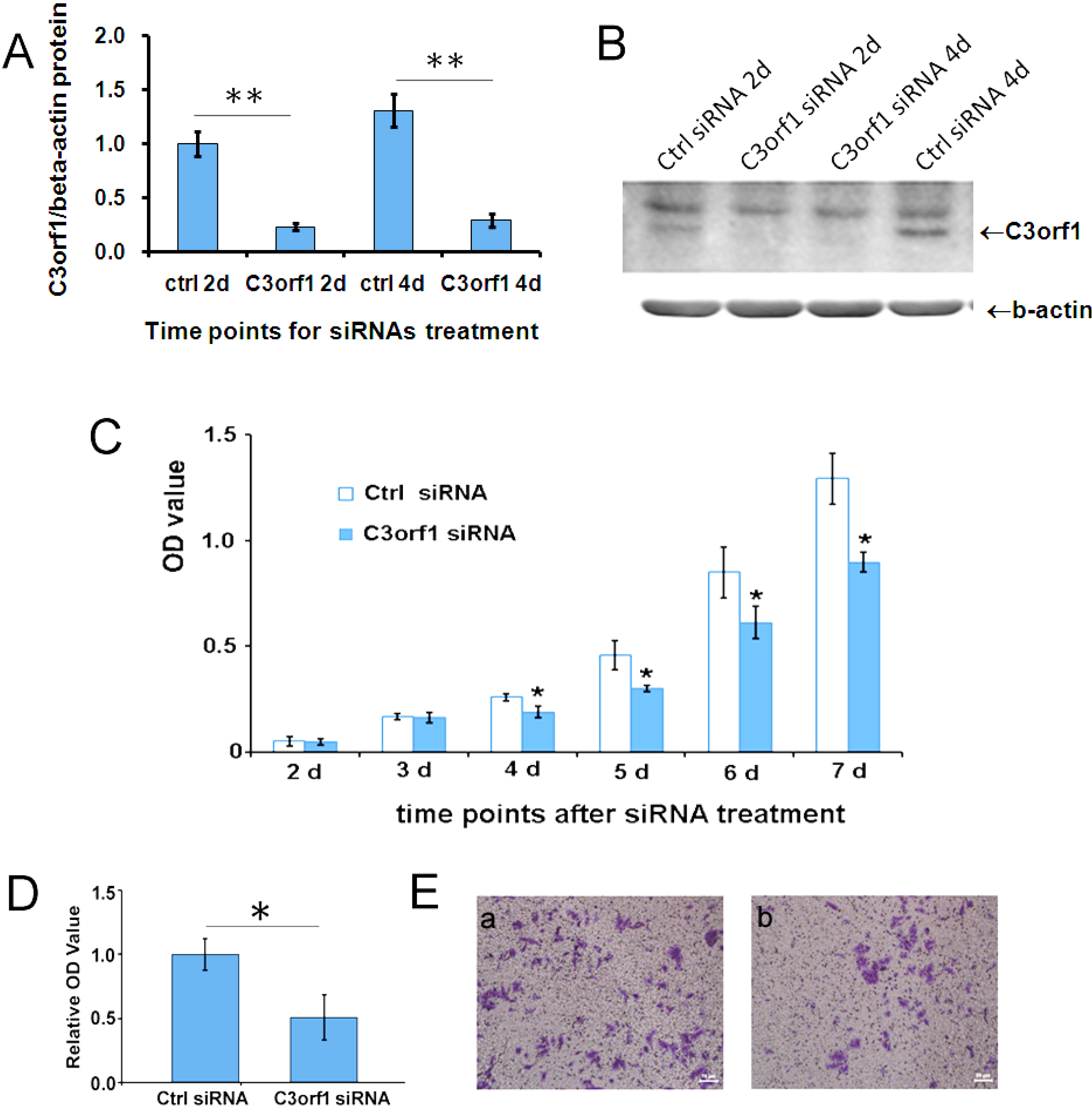
2.2. Depletion of c3orf1 Inhibits Cell Proliferation and Migration in 95D Cells
2.3. C3orf1 Localizes to the Inner Mitochondrial Membrane of 95D Cells and Exhibits Mitochondria-Related Functions
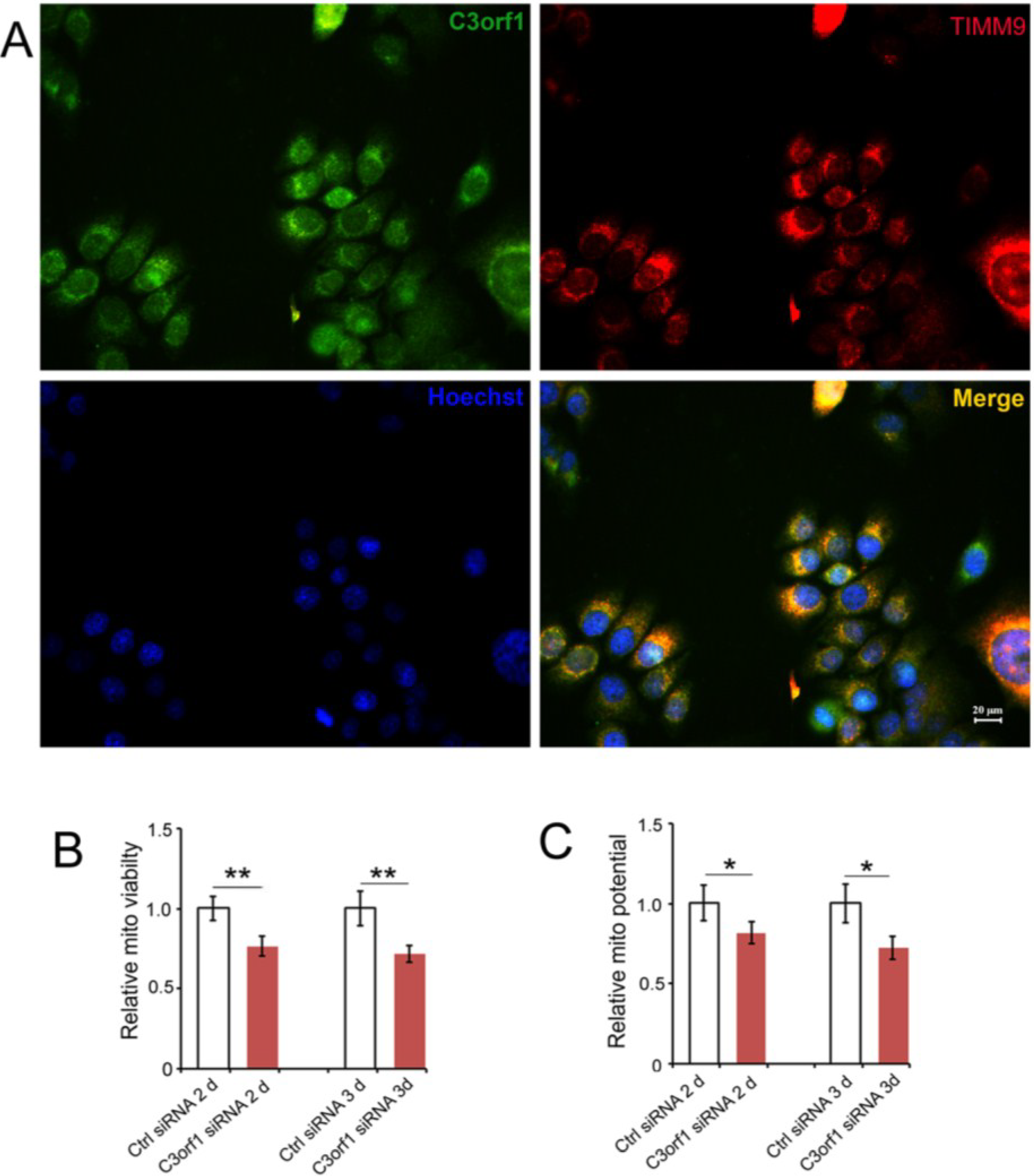
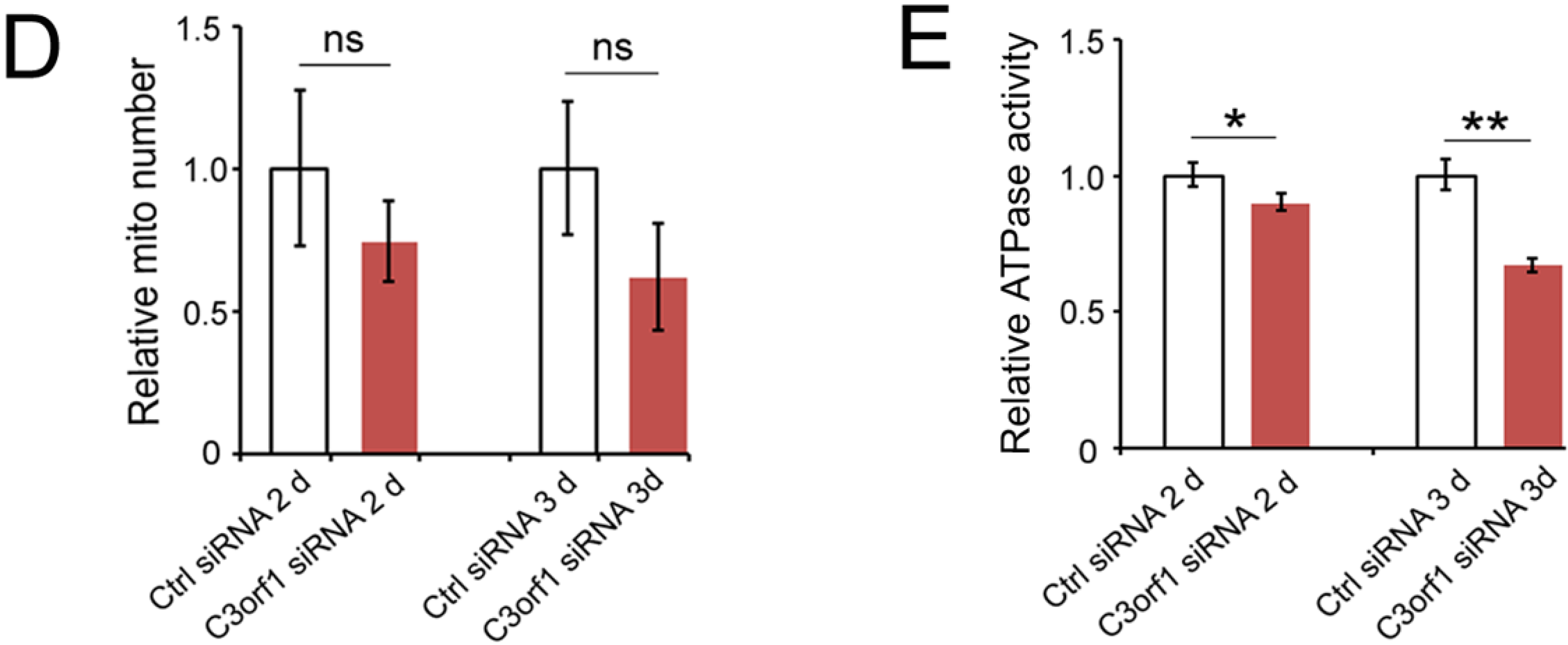
2.4. C3orf1 Depletion Resulted in Alteration of Gene Expression Profiles in 95D Cells
| GO TERM_BP_FAT | Count | % | p Value |
|---|---|---|---|
| 0042981~regulation of apoptosis | 44 | 11.028 | 3.47 × 10−7 |
| 0043067~regulation of programmed cell death | 44 | 11.028 | 4.54 × 10−7 |
| 0010941~regulation of cell death | 44 | 11.028 | 4.97 × 10−7 |
| 0016125~sterol metabolic process | 13 | 3.258 | 4.24 × 10−6 |
| 0043065~positive regulation of apoptosis | 26 | 6.516 | 2.98 × 10−5 |
| 0043068~positive regulation of programmed cell death | 26 | 6.516 | 3.34 × 10−5 |
| 0016044~membrane organization | 24 | 6.015 | 3.46 × 10−5 |
| 0010942~positive regulation of cell death | 26 | 6.516 | 3.60 × 10−5 |
| 0008203~cholesterol metabolic process | 11 | 2.757 | 5.86 × 10−5 |
| 0010627~regulation of protein kinase cascade | 18 | 4.511 | 8.52 × 10−5 |
| 0010033~response to organic substance | 35 | 8.772 | 8.58 × 10−5 |
| 0008202~steroid metabolic process | 16 | 4.010 | 8.70 × 10−5 |
| 0010035~response to inorganic substance | 16 | 4.010 | 1.02 × 10−4 |
| 0016126~sterol biosynthetic process | 7 | 1.754 | 1.47 × 10−4 |
| 0008219~cell death | 34 | 8.521 | 1.80 × 10−4 |
| 0016265~death | 34 | 8.521 | 2.02 × 10−4 |
| 0001666~response to hypoxia | 12 | 3.008 | 3.21 × 10−4 |
| 0009628~response to abiotic stimulus | 21 | 5.263 | 4.51 × 10−4 |
| 0043405~regulation of MAP kinase activity | 12 | 3.008 | 4.96 × 10−4 |
| 0070482~response to oxygen levels | 12 | 3.008 | 4.96 × 10−4 |
| 0048545~response to steroid hormone stimulus | 14 | 3.509 | 6.19 × 10−4 |
| 0043066~negative regulation of apoptosis | 20 | 5.013 | 7.27 × 10−4 |
| 0031667~response to nutrient levels | 14 | 3.509 | 7.88 × 10−4 |
| 0001932~regulation of protein amino acid phosphorylation | 13 | 3.258 | 8.03 × 10−4 |
| 0032268~regulation of cellular protein metabolic process | 24 | 6.015 | 8.47 × 10−4 |
| 0043069~negative regulation of programmed cell death | 20 | 5.013 | 8.57 × 10−4 |
| 0060548~negative regulation of cell death | 20 | 5.013 | 8.89 × 10−4 |
| 0006915~apoptosis | 27 | 6.767 | 0.00210 |
| 0009991~response to extracellular stimulus | 14 | 3.509 | 0.00214 |
| 0030334~regulation of cell migration | 12 | 3.008 | 0.00218 |
| 0005996~monosaccharide metabolic process | 14 | 3.509 | 0.00233 |
| 0012501~programmed cell death | 27 | 6.767 | 0.00261 |
| 0007050~cell-cycle arrest | 9 | 2.256 | 0.00293 |
| 0006006~glucose metabolic process | 11 | 2.757 | 0.00332 |
| 0045859~regulation of protein kinase activity | 18 | 4.511 | 0.00334 |
| 0043549~regulation of kinase activity | 18 | 4.511 | 0.00470 |
| 0001568~blood vessel development | 14 | 3.509 | 0.00534 |
| 0040012~regulation of locomotion | 12 | 3.008 | 0.00578 |
| 0051270~regulation of cell motion | 12 | 3.008 | 0.00598 |
| 0051896~regulation of protein kinase B signaling cascade | 4 | 1.003 | 0.00685 |
| 0002685~regulation of leukocyte migration | 4 | 1.003 | 0.01090 |
| 0030198~extracellular matrix organization | 8 | 2.005 | 0.01131 |
| 0051726~regulation of cell cycle | 16 | 4.010 | 0.01188 |
| 0000187~activation of MAPK activity | 7 | 1.754 | 0.01265 |
| 0022402~cell-cycle process | 23 | 5.764 | 0.01430 |
| 0051241~negative regulation of multicellular organismal process | 10 | 2.506 | 0.01550 |
| 0010629~negative regulation of gene expression | 21 | 5.263 | 0.01579 |
| 0045860~positive regulation of protein kinase activity | 12 | 3.008 | 0.01652 |
| 0051347~positive regulation of transferase activity | 12 | 3.008 | 0.02669 |
| 0051247~positive regulation of protein metabolic process | 12 | 3.008 | 0.02892 |
| 0051674~localization of cell | 14 | 3.509 | 0.02983 |
| 0048870~cell motility | 14 | 3.509 | 0.02983 |
| 0016477~cell migration | 13 | 3.258 | 0.03028 |
| 0042127~regulation of cell proliferation | 28 | 7.018 | 0.03137 |
| 0006928~cell motion | 19 | 4.762 | 0.03161 |
| 0044265~cellular macromolecule catabolic process | 26 | 6.516 | 0.03624 |
| 0042060~wound healing | 10 | 2.506 | 0.03677 |
| 0008629~induction of apoptosis by intracellular signals | 5 | 1.253 | 0.03780 |
| 0006917~induction of apoptosis | 14 | 3.509 | 0.03945 |
| 0012502~induction of programmed cell death | 14 | 3.509 | 0.04030 |
| 0009611~response to wounding | 20 | 5.013 | 0.04407 |
| 0045787~positive regulation of cell cycle | 5 | 1.253 | 0.04474 |
| 0007568~aging | 7 | 1.754 | 0.04522 |
| 0008285~negative regulation of cell proliferation | 15 | 3.759 | 0.04650 |
| Term | % | p Value | Genes |
|---|---|---|---|
| 04144:Endocytosis | 3.01 | 0.016 | VTA1, HSPA1A, EEA1, HSPA1B, CLTC, CHMP2B, TFRC, HSPA2, CXCR4, NEDD4, MDM2, HSPA8, AP2M1 |
| 04510:Focal adhesion | 3.01 | 0.029 | CAV1, ROCK2, ITGB8, ITGAV, COL3A1, PPP1R12A, BIRC3, THBS1, PPP1CB, PTEN, COL5A1, FN1 |
| 04512:ECM-receptor interaction | 1.75 | 0.032 | ITGB8, ITGAV, COL3A1, THBS1, SV2C, COL5A1, FN1 |
| 04910:Insulin signaling pathway | 2.26 | 0.039 | PPP1R3C, IRS2, PPP1R3B, PRKAG2, HK2, RPS6KB1, RPS6, PCK2, PPP1CB |
| 04920:Adipocytokine signaling pathway | 1.50 | 0.042 | IRS2, PRKAG2, SLC2A1, NFKBIA, ACSL4, PCK2 |
| 04115:p53 signaling pathway | 1.50 | 0.045 | MDM2, CCNG1, THBS1, IGFBP3, CCNG2, PTEN |
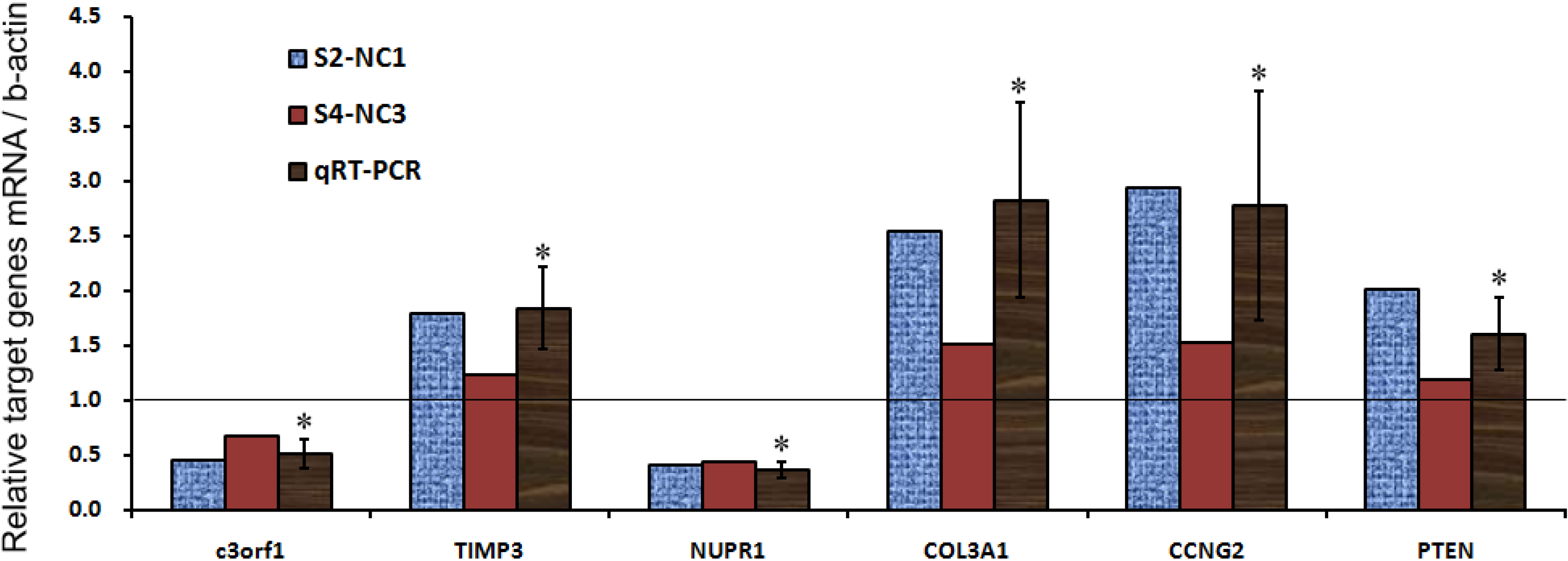
| Genes/Ctrl | c3orf1 | TIMP3 | NUPR1 | COL3A1 | CCNG2 | PTEN |
|---|---|---|---|---|---|---|
| S2-NC1 | 0.4499 | 1.7926 | 0.4058 | 2.5374 | 2.9360 | 2.0069 |
| S4-NC3 | 0.6704 | 1.2319 | 0.4367 | 1.5181 | 1.5311 | 1.1950 |
| qRT-PCR mean | 0.5099 | 1.8332 | 0.3613 | 2.8237 | 2.7743 | 1.6045 |
| SD | 0.1263 | 0.3748 | 0.0722 | 0.8942 | 1.0438 | 0.3368 |
| p value | 0.0052 | 0.0183 | 0.0001 | 0.0242 | 0.0349 | 0.0359 |
3. Discussion
4. Experimental Section
4.1. Cell Culture
4.2. RNA Extraction and Real-Time PCR Analysis
| Gene Name | Sequence (5'→3') |
|---|---|
| c3orf1 sense | 5'-GAGACCTTCAACACCCCAGCC-3' |
| c3orf1 antisense | 5'-CCTGAACAGTCTCACCACTACTTACTTC-3' |
| beta-actin sense | 5'-GACTACCTCATGAAGATCCTCACC-3' |
| beta-actin antisense | 5'-TCTCCTTAATGTCACGCACGATT-3' |
| ctrl siRNA sense | 5'-UUCUCCGAACGUGUCACGUTT-3' |
| ctrl siRNA antisense | 5'-ACGUGACACGUUCGGAGAATT-3' |
| c3orf1 siRNA sense | 5'-UGUAGAGCAUUGUGCCUAUTT-3' |
| c3orf1 siRNA antisense | 5'-AUAGGCACAAUGCUCUACATT-3' |
| CCNG2 sense | 5'-TTTGGATCGTTTCAAGGCGC-3' |
| CCNG2 antisense | 5'-TTGATCACTGGGAGGAGAGC-3' |
| PTEN sense | 5'-TGCAGTATAGAGCGTGCAGA-3' |
| PTEN antisense | 5'-CTGGATTTGACGGCTCCTCT-3' |
| COL3A1 sense | 5'-GGCAAAGATGGAACCAGTGG-3' |
| COL3A1 antisense | 5'-TCACCTCCAATCCCAGCAAT-3' |
| TIMP3 sense | 5'-CTGTGCAACTTCGTGGAGAG-3' |
| TIMP3 antisense | 5'-AGTGTTTGGACTGGTAGCCA-3' |
| NUPR1 sense | 5'-CTGACCTCTATAGCCTGGCC-3' |
| NUPR1 antisense | 5'-GGTCACCAGTTTCCTCTCGT-3' |
4.3. Wound-Healing Assay
4.4. siRNA Treatment and Western Blotting
4.5. Determination of Cell Proliferation Activity and Migration Assay
4.6. Immunocytochemistry
4.7. Measurements of Mitochondrial Viability, Number, Membrane Potential, and ATPase Activity
4.8. Microarray Analysis
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Siegel, R.; Ma, J.M.; Zou, Z.H.; Jemal, A. Cancer Statistics, 2014. CA Cancer J. Clin. 2014, 64, 9–29. [Google Scholar]
- Chen, W.Q.; Zheng, R.S.; Zhang, S.W.; Zhao, P.; Zeng, H.M.; Zou, X.N.; He, J. Annual report on status of cancer in China, 2010. Chin. J. Cancer Res. 2014, 26, 48–58. [Google Scholar]
- Gong, H.C.; Wang, S.; Mayer, G.; Chen, G.; Leesman, G.; Singh, S.; Beer, D.G. Signatures of drug sensitivity in nonsmall cell lung cancer. Int. J. Proteomics 2011, 2011, 215496. [Google Scholar] [CrossRef]
- Ellis, P.M.; Al-Saleh, K. Multitargeted anti-angiogenic agents and NSCLC: Clinical update and future directions. Crit. Rev. Oncol. Hematol. 2012, 84, 47–58. [Google Scholar]
- Shepherd, F.A.; Douillard, J.Y.; Blumenschein, G.R., Jr. Immunotherapy for non-small cell lung cancer: Novel approaches to improve patient outcome. J. Thorac. Oncol. 2011, 6, 1763–1773. [Google Scholar]
- Ha, S.Y.; Lee, J.; Kang, S.Y.; Do, I.G.; Ahn, S.; Park, J.O.; Kang, W.K.; Choi, M.G.; Sohn, T.S.; Bae, J.M.; et al. MET overexpression assessed by new interpretation method predicts gene amplification and poor survival in advanced gastric carcinomas. Mod. Pathol. 2013, 26, 1632–1641. [Google Scholar]
- Stahel, R.; Peters, S.; Baas, P.; Brambilla, E.; Cappuzzo, F.; de Ruysscher, D.; Eberhardt, W.E.; Felip, E.; Fennell, D.; Marchetti, A.; et al. Strategies for improving outcomes in NSCLC: A look to the future. Lung Cancer 2013, 82, 375–382. [Google Scholar]
- Rosato, A.; Menin, C.; Boldrin, D.; Santa, S.D.; Bonaldi, L.; Scaini, M.C.; del Bianco, P.; Zardo, D.; Fassan, M.; Cappellesso, R.; et al. Survivin expression impacts prognostically on NSCLC but not SCLC. Lung Cancer 2013, 79, 180–186. [Google Scholar]
- Wen, Y.; Wang, W.; Xu, W.; Zhu, W. Analysis of c3orf1 gene over-expressed in human lung cancer cell line 95D with high metastatic potential. Chin. J. Clin. Lab. Sci. 2007, 25, 117–119. [Google Scholar]
- Andrews, B.; Carroll, J.; Ding, S.; Fearnley, I.M.; Walker, J.E. Assembly factors for the membrane arm of human complex I. Proc. Natl. Acad. Sci. USA 2013, 110, 18934–18939. [Google Scholar]
- Guarani, V.; Paulo, J.; Zhai, B.; Huttlin, E.L.; Gygi, S.P.; Harper, J.W. TIMMDC1/C3orf1 functions as a membrane-embedded mitochondrial complex I assembly factor through association with the MCIA complex. Mol. Cell. Biol. 2014, 34, 847–861. [Google Scholar]
- Kurt, Y.G.; Cayci, T.; Akgul, E.O.; Kurt, B. MtDNA variations other than point mutations may also have a role in carcinogenesis of lung cancer. Biochem. Biophys. Res. Commun. 2012, 420, 698. [Google Scholar] [CrossRef]
- Dasgupta, S.; Soudry, E.; Mukhopadhyay, N.; Shao, C.; Yee, J.; Lam, S.; Lam, W.; Zhang, W.; Gazdar, A.F.; Fisher, P.B.; et al. Mitochondrial DNA mutations in respiratory complex-I in never-smoker lung cancer patients contribute to lung cancer progression and associated with EGFR gene mutation. J. Cell. Physiol. 2012, 227, 2451–2460. [Google Scholar]
- Choi, S.J.; Kim, S.H.; Kang, H.Y.; Lee, J.; Bhak, J.H.; Sohn, I.; Jung, S.H.; Choi, Y.S.; Kim, H.K.; Han, J.; et al. Mutational hotspots in the mitochondrial genome of lung cancer. Biochem. Biophys. Res. Commun. 2011, 407, 23–27. [Google Scholar]
- Kachadourian, R.; Leitner, H.M.; Day, B.J. Selected flavonoids potentiate the toxicity of cisplatin in human lung adenocarcinoma cells: A role for glutathione depletion. Int. J. Oncol. 2007, 31, 161–168. [Google Scholar]
- Huang, Y.; Yang, X.; Zhao, F.; Shen, Q.; Wang, Z.; Lv, X.; Hu, B.; Yu, B.; Fan, J.; Qin, W. Overexpression of Dickkopf-1 predicts poor prognosis for patients with hepatocellular carcinoma after orthotopic liver transplantation by promoting cancer metastasis and recurrence. Med. Oncol. 2014, 31, 966. [Google Scholar] [CrossRef]
- Zimmermann, M.; Arachchige-Don, A.S.; Donaldson, M.S.; Dallapiazza, R.F.; Cowan, C.E.; Horne, M.C. Elevated cyclin G2 expression intersects with DNA damage checkpoint signaling and is required for a potent G2/M checkpoint arrest response to doxorubicin. J. Biol. Chem. 2012, 287, 22838–22853. [Google Scholar]
- Ferraro, B.; Bepler, G.; Sharma, S.; Cantor, A.; Haura, E.B. EGR1 predicts PTEN and survival in patients with non-small-cell lung cancer. J. Clin. Oncol. 2005, 23, 1921–1926. [Google Scholar]
- Lin, H.; Zhang, Y.; Wang, H.; Xu, D.; Meng, X.; Shao, Y.; Lin, C.; Ye, Y.; Qian, H.; Wang, S. Tissue inhibitor of metalloproteinases-3 transfer suppresses malignant behaviors of colorectal cancer cells. Cancer Gene Ther. 2012, 19, 845–851. [Google Scholar]
- Luo, R.; Jeong, S.J.; Jin, Z.; Strokes, N.; Li, S.; Piao, X. G protein-coupled receptor 56 and collagen III, a receptor-ligand pair, regulates cortical development and lamination. Proc. Natl. Acad. Sci. USA 2011, 108, 12925–12930. [Google Scholar]
- Kim, K.S.; Jin, D.I.; Yoon, S.; Baek, S.Y.; Kim, B.S.; Oh, S.O. Expression and roles of NUPR1 in cholangiocarcinoma cells. Anat. Cell Biol. 2012, 45, 17–25. [Google Scholar]
- Hirst, J. Mitochondrial complex I. Annu. Rev. Biochem. 2013, 82, 551–575. [Google Scholar]
- Murphy, M.P. How mitochondria produce reactive oxygen species. Biochem. J. 2009, 417, 1–13. [Google Scholar]
- Kim, S.H.; Yoo, Y.H.; Lee, J.H.; Park, J.W. Mitochondrial NADP-dependent isocitrate dehydrogenase knockdown inhibits tumorigenicity of melanoma cells. Biochem. Biophys. Res. Commun. 2014, 451, 246–251. [Google Scholar]
- Harrington, L.S.; Lucas, R.; McMaster, S.K.; Moreno, L.; Scadding, G.; Warner, T.D.; Mitchell, J.A. COX-1, and not COX-2 activity, regulates airway function: relevance to aspirin-sensitive asthma. FASEB J. 2008, 22, 4005–4010. [Google Scholar]
- Sotgia, F.; Whitaker-Menezes, D.; Martinez-Outschoorn, U.E.; Salem, A.F.; Tsirigos, A.; Lamb, R.; Sneddon, S.; Hulit, J.; Howell, A.; Lisanti, M.P. Mitochondria “fuel” breast cancer metabolism: fifteen markers of mitochondrial biogenesis label epithelial cancer cells, but are excluded from adjacent stromal cells. Cell Cycle 2012, 11, 4390–4401. [Google Scholar]
- Li, J.; Wu, R.; Chen, H.; Zhou, Y.; Li, Y.; Wang, Y.; Liu, Y.; Liu, M. The cloning and characterization of the Enolase2 gene of Gekko japonicus and its polyclonal antibody preparation. Int. J. Mol. Sci. 2013, 14, 8787–8800. [Google Scholar]
- Yu, B.; Qian, T.; Wang, Y.; Zhou, S.; Ding, G.; Ding, F.; Gu, X. miR-182 inhibits Schwann cell proliferation and migration by targeting FGF9 and NTM, respectively at an early stage following sciatic nerve injury. Nucleic Acids Res. 2012, 40, 10356–10365. [Google Scholar]
- Liu, M.; Wu, R.; Yang, F.; Wang, T.; Zhang, P.; Gu, J.; Wan, D.; Yang, S. Identification of FN1BP1 as a novel cell cycle regulator through modulating G1 checkpoint in human hepatocarcinoma Hep3B cells. PLoS One 2013, 8, e57574. [Google Scholar]
- Xu, Y.; Zhang, Q.; Yu, S.; Yang, Y.; Ding, F. The protective effects of chitooligosaccharides against glucose deprivation-induced cell apoptosis in cultured cortical neurons through activation of PI3K/Akt and MEK/ERK1/2 pathways. Brain Res. 2011, 1375, 49–58. [Google Scholar]
- Mi, H.; Muruganujan, A.; Thomas, P.D. PANTHER in 2013: Modeling the evolution of gene function, and other gene attributes, in the context of phylogenetic trees. Nucleic Acids Res. 2013, 41, D377–D386. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, H.; Wang, W.; Xu, H. Depletion of C3orf1/TIMMDC1 Inhibits Migration and Proliferation in 95D Lung Carcinoma Cells. Int. J. Mol. Sci. 2014, 15, 20555-20571. https://doi.org/10.3390/ijms151120555
Wu H, Wang W, Xu H. Depletion of C3orf1/TIMMDC1 Inhibits Migration and Proliferation in 95D Lung Carcinoma Cells. International Journal of Molecular Sciences. 2014; 15(11):20555-20571. https://doi.org/10.3390/ijms151120555
Chicago/Turabian StyleWu, Huiling, Wenbing Wang, and Huaxi Xu. 2014. "Depletion of C3orf1/TIMMDC1 Inhibits Migration and Proliferation in 95D Lung Carcinoma Cells" International Journal of Molecular Sciences 15, no. 11: 20555-20571. https://doi.org/10.3390/ijms151120555
APA StyleWu, H., Wang, W., & Xu, H. (2014). Depletion of C3orf1/TIMMDC1 Inhibits Migration and Proliferation in 95D Lung Carcinoma Cells. International Journal of Molecular Sciences, 15(11), 20555-20571. https://doi.org/10.3390/ijms151120555




