Formation and Yield of Multi-Walled Carbon Nanotubes Synthesized via Chemical Vapour Deposition Routes Using Different Metal-Based Catalysts of FeCoNiAl, CoNiAl and FeNiAl-LDH
Abstract
:1. Introduction
2. Results and Discussion
2.1. Carbon Yield
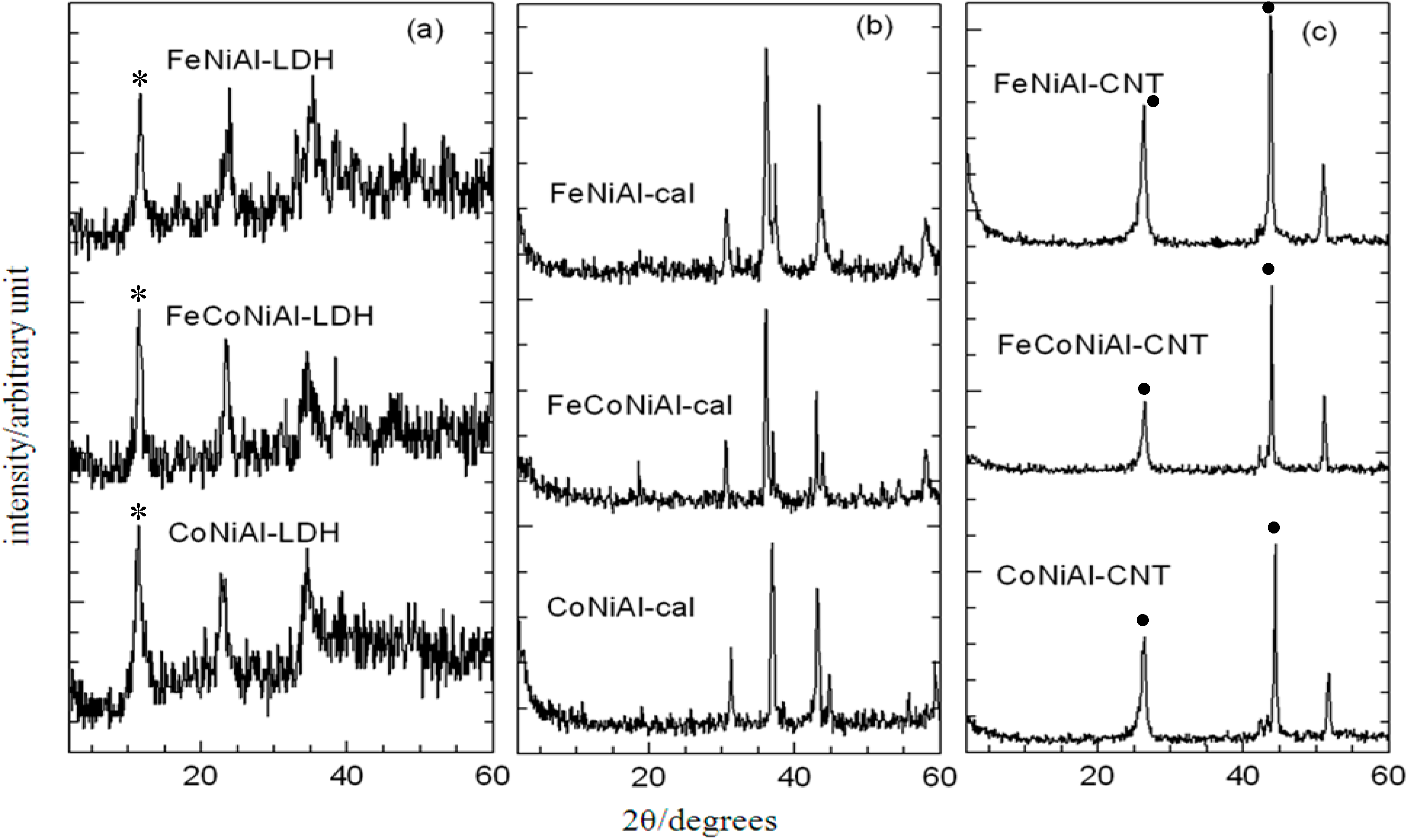
2.2. Powder X-ray Diffraction
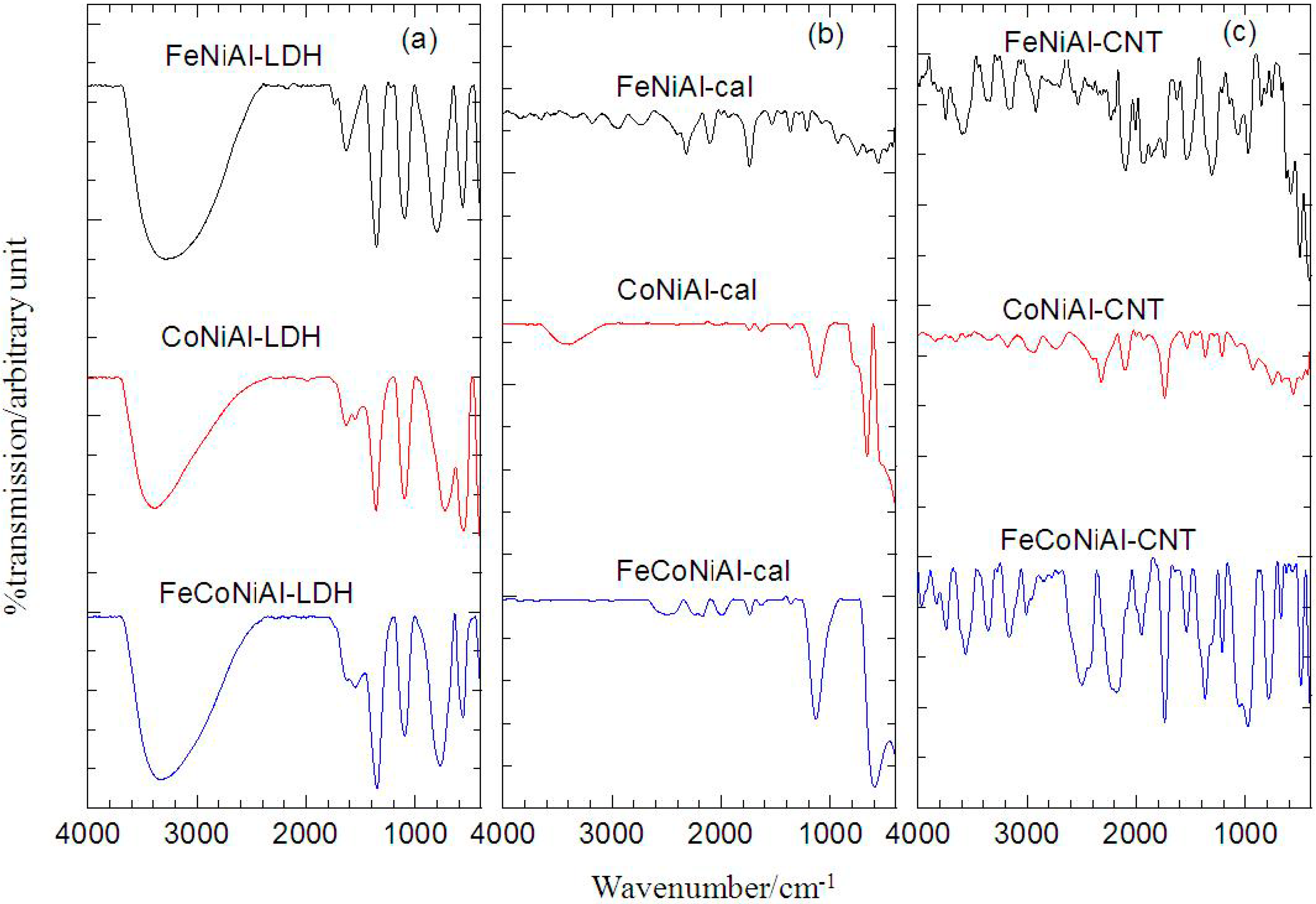
2.3. Fourier Transform Infrared
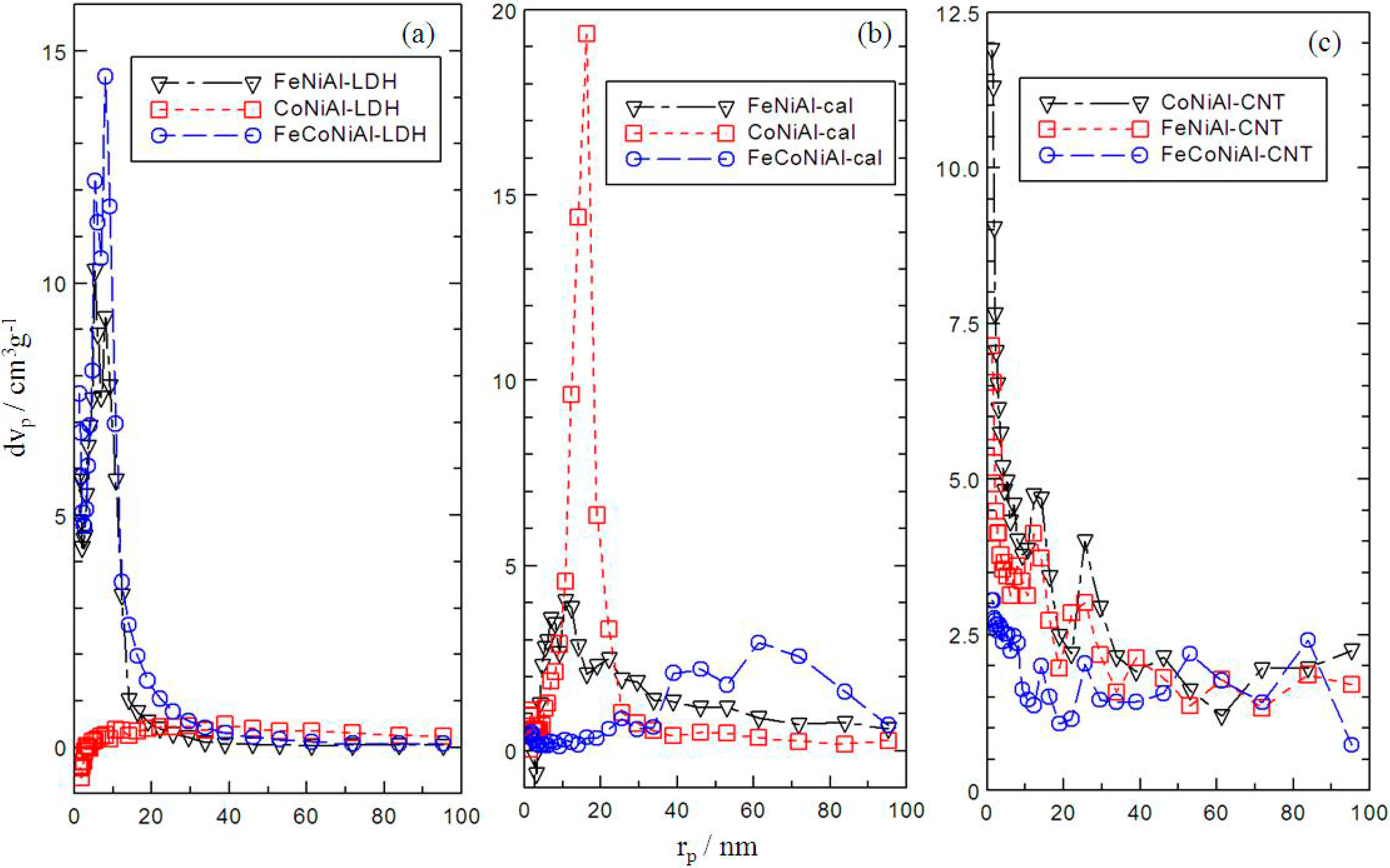
2.4. Surface Area Analysis

| Material | BET Surface Area (m2·g−1) | BJH Desorption Average Pore Diameter (nm) | |
|---|---|---|---|
| LDH | FeNiAl | 35.1 | 5.3 |
| FeCoNiAl | 32.5 | 8.0 | |
| CoNiAl | 1.0 | 39.0 | |
| Calcined LDH | FeNiAl | 6.4 | 10.7 |
| FeCoNiAl | 6.7 | 8.9 | |
| CoNiAl | 22.3 | 1.6 | |
| CNT | FeNiAl | 33.7 | 1.2 |
| FeCoNiAl | 19.8 | 1.6 | |
| CoNiAl | 47.6 | 0.98 | |
2.5. Field Emission Scanning Electron Microscope
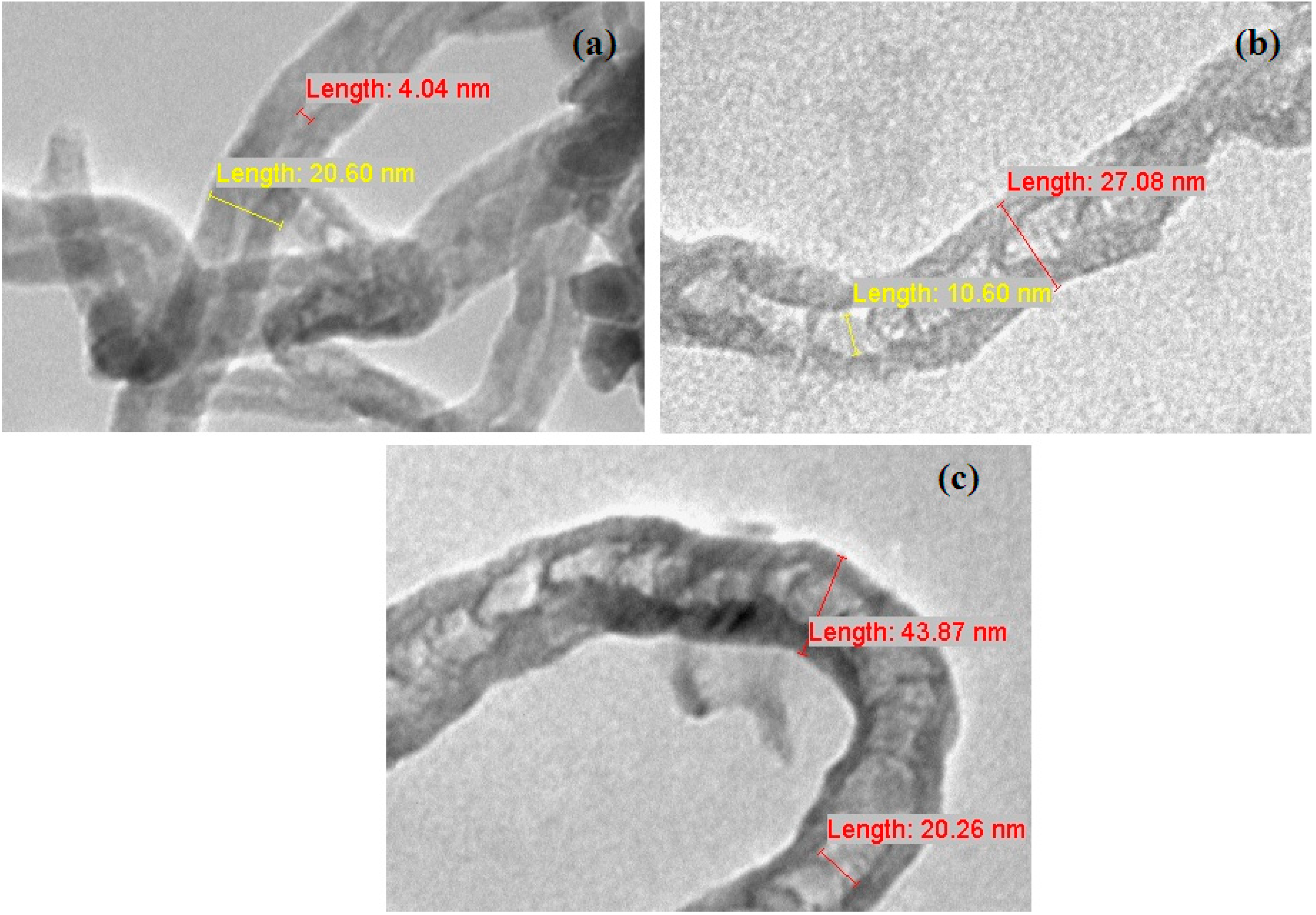
2.6. Transmission Electron Microscope

3. Experimental Section
3.1. Preparation of LDH Precursors
3.2. Growth of CNTs
3.3. Characterisation
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cavani, F.; Trifiro, F.; Vaccari, A. Hydrotalcite-type anionic clays:preparation, properties and applications. Catal. Today 1991, 11, 173–301. [Google Scholar] [CrossRef]
- Iijima, S. Helical microtubules of graphitic carbon. Nature 1991, 354, 56–58. [Google Scholar] [CrossRef]
- An, K.H.; Kim, W.S.; Park, Y.S.; Choi, Y.C.; Lee, S.M.; Chung, D.C.; Bae, D.J.; Lim, S.C.; Lee, Y.H. Supercapacitors using single-walled carbon nanotube electrodes. Adv. Mater. 2001, 13, 497–500. [Google Scholar] [CrossRef]
- Bachtold, A.; Hadley, P.; Nakanishi, T.; Dekker, C. Logic circuits with carbon nanotube transistors. Science 2001, 294, 1317–1320. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.W.; Xu, C.L.; Wu, D.H.; Wei, B.Q.; Vajtai, R.; Ajayan, P.M. Direct synthesis of long single-walled carbon nanotube strands. Science 2002, 296, 884–886. [Google Scholar] [CrossRef] [PubMed]
- Zanello, L.P.; Zhao, B.; Hu, H.; Haddon, R.C. Bone cell proliferation on carbon nanotubes. Nano Lett. 2006, 6, 562–567. [Google Scholar] [CrossRef] [PubMed]
- Queipo, P.; Nasibulin, A.G.; Shandakov, S.D.; Jiang, H.; Gonzalez, D.; Kauppinen, E.I. CVD synthesis and radial deformations of large diameter single-walled CNTs. Curr. Appl. Phys. 2009, 9, 301–305. [Google Scholar] [CrossRef]
- Kamal, K.K.; Rahaman, A.; Sathiyamoorthy, D.; Agnihotri, P. Synthesis of carbon nanotubes on the surface of carbon fiber/fabric by catalytic chemical vapor deposition and their characterization fullerenes. Nanotub. Carbon Nanostruct. 2009, 17, 209–229. [Google Scholar] [CrossRef]
- Flahaut, E.; Bacsa, R.; Peigney, A.; Laurent, C. Gram-scale CCVD synthesis of double-walled carbon nanotubes. Chem. Commun. 2003, 12, 1442–1443. [Google Scholar]
- Chen, Y.; Ciuparu, D.; Lim, S.; Yang, Y.; Haller, G.L.; Pfefferle, L. Synthesis of uniform diameter single-wall carbon nanotubes in Co-MCM-41: Effects of the catalyst prereduction and nanotube growth temperatures. J. Catal. 2004, 225, 453–465. [Google Scholar] [CrossRef]
- Gulino, G.; Vieira, R.; Amadou, J.; Nguyen, P.; Ledoux, M.J.; Galvagno, S.; Centi, G.; Pham-Huu, C. C2H6 as an active carbon source for a large scale synthesis of carbon nanotubes by chemical vapour deposition. Appl. Catal. A Gen. 2005, 279, 89–97. [Google Scholar] [CrossRef]
- Chai, S.P.; Zein, S.H.S.; Mohamed, A.R. The effect of reduction temperature on Co-Mo/Al2O3 catalysts for carbon nanotubes formation. Appl. Catal. A Gen. 2007, 326, 173–179. [Google Scholar] [CrossRef]
- Tran, K.Y.; Heinrichs, B.; Colomer, J.-F.; Pirard, J.-P.; Lambert, S. Carbon nanotubes synthesis by the ethylene chemical catalytic vapour deposition (CCVD) process on Fe, Co, and Fe-Co/Al2O3 sol-gel catalysts. Appl. Catal. A Gen. 2007, 318, 63–69. [Google Scholar] [CrossRef]
- Zhang, L.; Li, F.; Xiang, X.; Wei, M.; Evans, D.G. Ni-based supported catalysts from layered double hydroxides: Tunable microstructure and controlled property for the synthesis of carbon nanotubes. Chem. Eng. J. 2009. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Everett, D.H.; Haul, R.A.W.; Moscou, L.; Pierotti, R.A.; Rouquerol, J.; Siemieniewska, J. Reporting Physisorption data for gas/solid systems with special reference on the determination of surface area and porosity. Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Alvarez, W.E.; Kitiyanan, B.; Borgna, A.; Resasco, D.E. Synergism of Co and Mo in the catalytic production of single-wall carbon nanotubes by decomposition of CO. Carbon 2001, 39, 547–558. [Google Scholar] [CrossRef]
- Liu, J.; Li, F.; Evans, D.G.; Duan, X. Stoichiometric synthesis of a pure ferrite from a tailored layered double hydroxide (hydrotalcite-like) precursor. Chem. Commun. 2003, 542–543. [Google Scholar] [CrossRef]
- Li, F.; Tan, Q.; Evans, D.G.; Duan, X. Synthesis of carbon nanotubes using a novel catalyst derived from hydrotalcite-like Co–Al layered double hydroxide precursor. Catal. Lett. 2005, 99, 151–156. [Google Scholar] [CrossRef]
- Kuzmany, H.; Burger, B.; Thess, A.; Smalley, R.E. Vibrational spectra of single wall carbon nanotubes. Carbon 1998, 36, 709–712. [Google Scholar] [CrossRef]
- Li, F.; Zhang, L.H.; Evans, D.G.; Duan, X. Structure and surface chemistry of manganese-doped copper-based mixed metal oxides derived from layered double hydroxides. Colloids Surf. A: Physicochem. Eng. Aspects 2004, 244, 169–177. [Google Scholar] [CrossRef]
- Dillon, C.; Gennett, T.; Alleman, J.L.; Jones, K.M. Carbon Nanotube Materials for Hydrogen Storage. In Proceedings of the 2000 DOE/NREL Hydrogen Program Review, Denver, CO, USA, 8–10 May 2000.
- Kastner, J.; Pichler, T.; Kuzmany, H.; Curran, S.; Blau, W.; Weldon, D.N.; Delamesiere, M.; Draper, S.; Zandbergen, H. Resonance Raman and infrared spectroscopy of carbon nanotubes. Chem. Phys. Lett. 1994, 221, 53–58. [Google Scholar] [CrossRef]
- Eklund, P.; Holden, J.M.; Jishi, R.A. Vibrational modes of carbon nanotubes; Spectroscopy and theory. Carbon 1995, 33, 959–972. [Google Scholar] [CrossRef]
- Chen, L.; Liu, H.; Yang, K.; Wang, J.; Wang, X. The effect of reaction temperature on the diameter distribution of carbon nanotubes grown from ethylene decomposition over a Co-La-O catalyst. Mater. Chem. Phys. 2008, 112, 407–411. [Google Scholar] [CrossRef]
- Rives, V.; Ulibarri, M.A. Layered double hydroxides (LDH) intercalated with metal coordination compounds and oxometalates. Coord. Chem. Rev. 1999, 181, 61–120. [Google Scholar] [CrossRef]
- Moreno, M.J.H.; Ulibarri, M.A.; Rendon, J.L.; Serna, C.J. IR characteristics of hydrotalcite-like compounds. Phys. Chem. Miner. 1985, 12, 34–38. [Google Scholar]
- Herrera, J.E.; Balzano, L.; Borgna, A.; Alvarez, W.E.; Resasco, D.E. Relationship between the structure/composition of Co-Mo catalysts and their ability to produce single-walled carbon nanotubes by CO disproportionation. J. Catal. 2001, 204, 129–145. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hussein, M.Z.; Jaafar, A.M.; Yahaya, A.H.; Masarudin, M.J.; Zainal, Z. Formation and Yield of Multi-Walled Carbon Nanotubes Synthesized via Chemical Vapour Deposition Routes Using Different Metal-Based Catalysts of FeCoNiAl, CoNiAl and FeNiAl-LDH. Int. J. Mol. Sci. 2014, 15, 20254-20265. https://doi.org/10.3390/ijms151120254
Hussein MZ, Jaafar AM, Yahaya AH, Masarudin MJ, Zainal Z. Formation and Yield of Multi-Walled Carbon Nanotubes Synthesized via Chemical Vapour Deposition Routes Using Different Metal-Based Catalysts of FeCoNiAl, CoNiAl and FeNiAl-LDH. International Journal of Molecular Sciences. 2014; 15(11):20254-20265. https://doi.org/10.3390/ijms151120254
Chicago/Turabian StyleHussein, Mohd Zobir, Adila Mohamad Jaafar, Asmah Hj. Yahaya, Mas Jaffri Masarudin, and Zulkarnain Zainal. 2014. "Formation and Yield of Multi-Walled Carbon Nanotubes Synthesized via Chemical Vapour Deposition Routes Using Different Metal-Based Catalysts of FeCoNiAl, CoNiAl and FeNiAl-LDH" International Journal of Molecular Sciences 15, no. 11: 20254-20265. https://doi.org/10.3390/ijms151120254
APA StyleHussein, M. Z., Jaafar, A. M., Yahaya, A. H., Masarudin, M. J., & Zainal, Z. (2014). Formation and Yield of Multi-Walled Carbon Nanotubes Synthesized via Chemical Vapour Deposition Routes Using Different Metal-Based Catalysts of FeCoNiAl, CoNiAl and FeNiAl-LDH. International Journal of Molecular Sciences, 15(11), 20254-20265. https://doi.org/10.3390/ijms151120254






