Materials Characterization of Feraheme/Ferumoxytol and Preliminary Evaluation of Its Potential for Magnetic Fluid Hyperthermia
Abstract
:1. Introduction
2. Results and Discussions
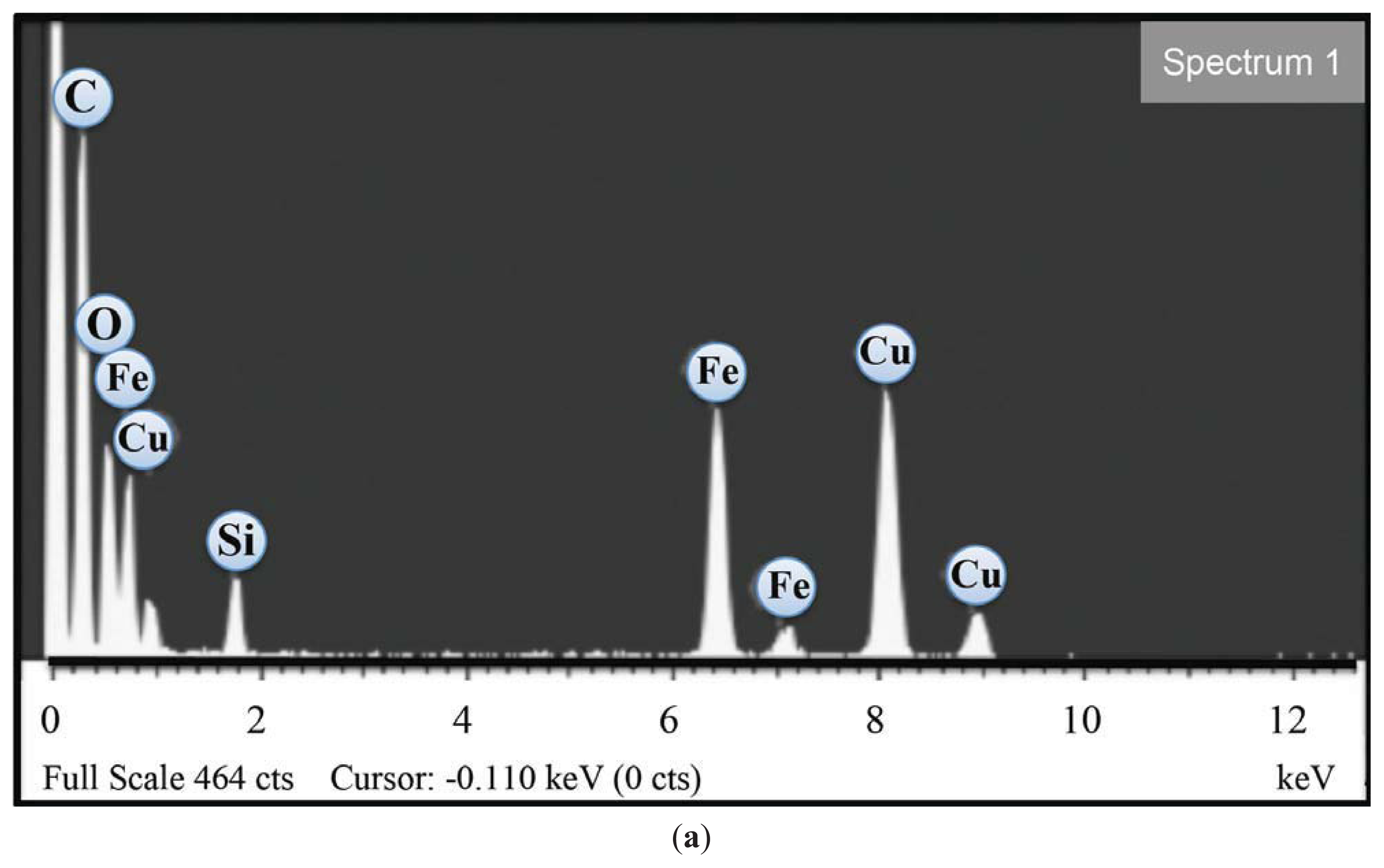
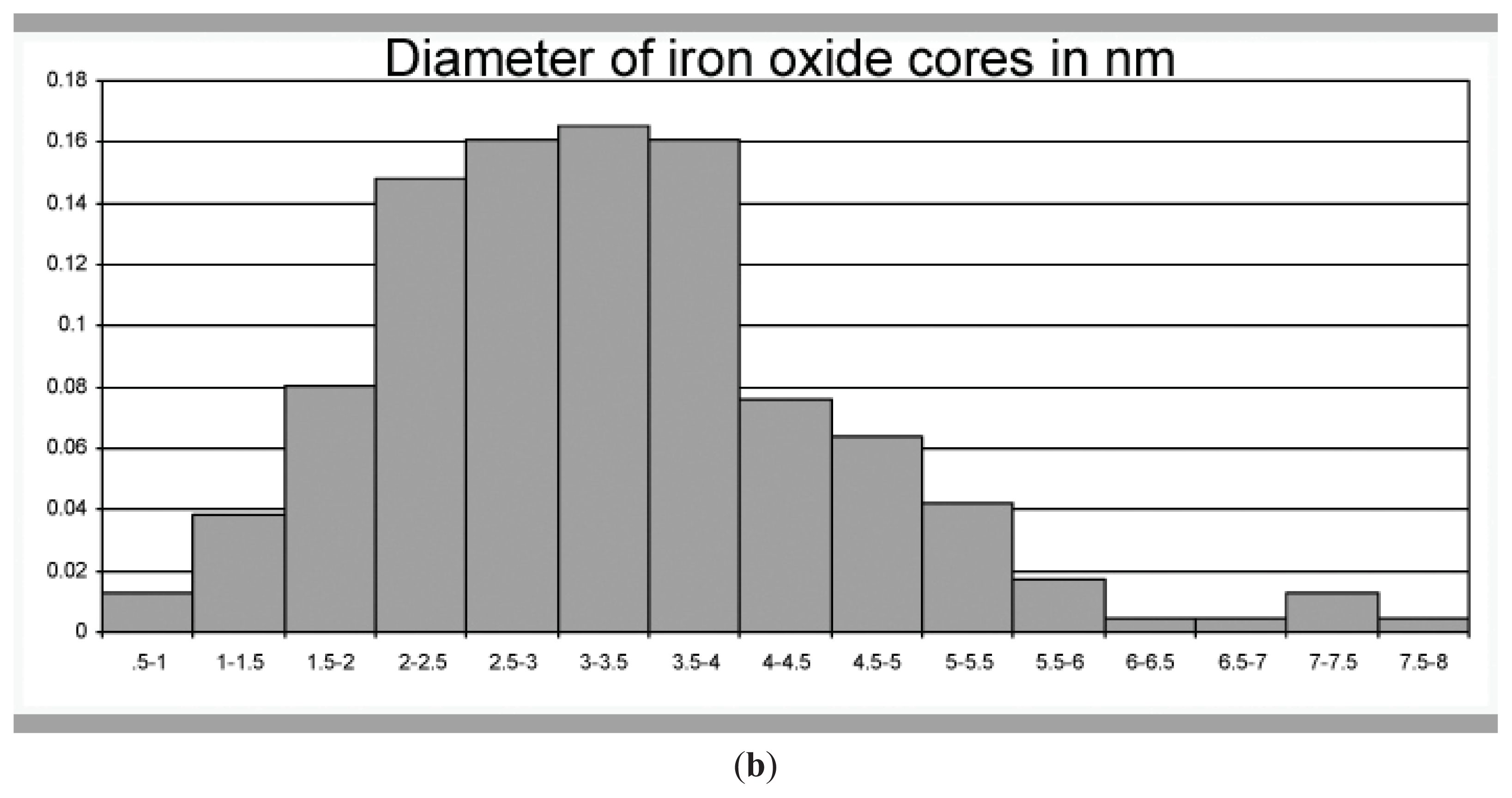
2.1. Transmission Electron Microscopy
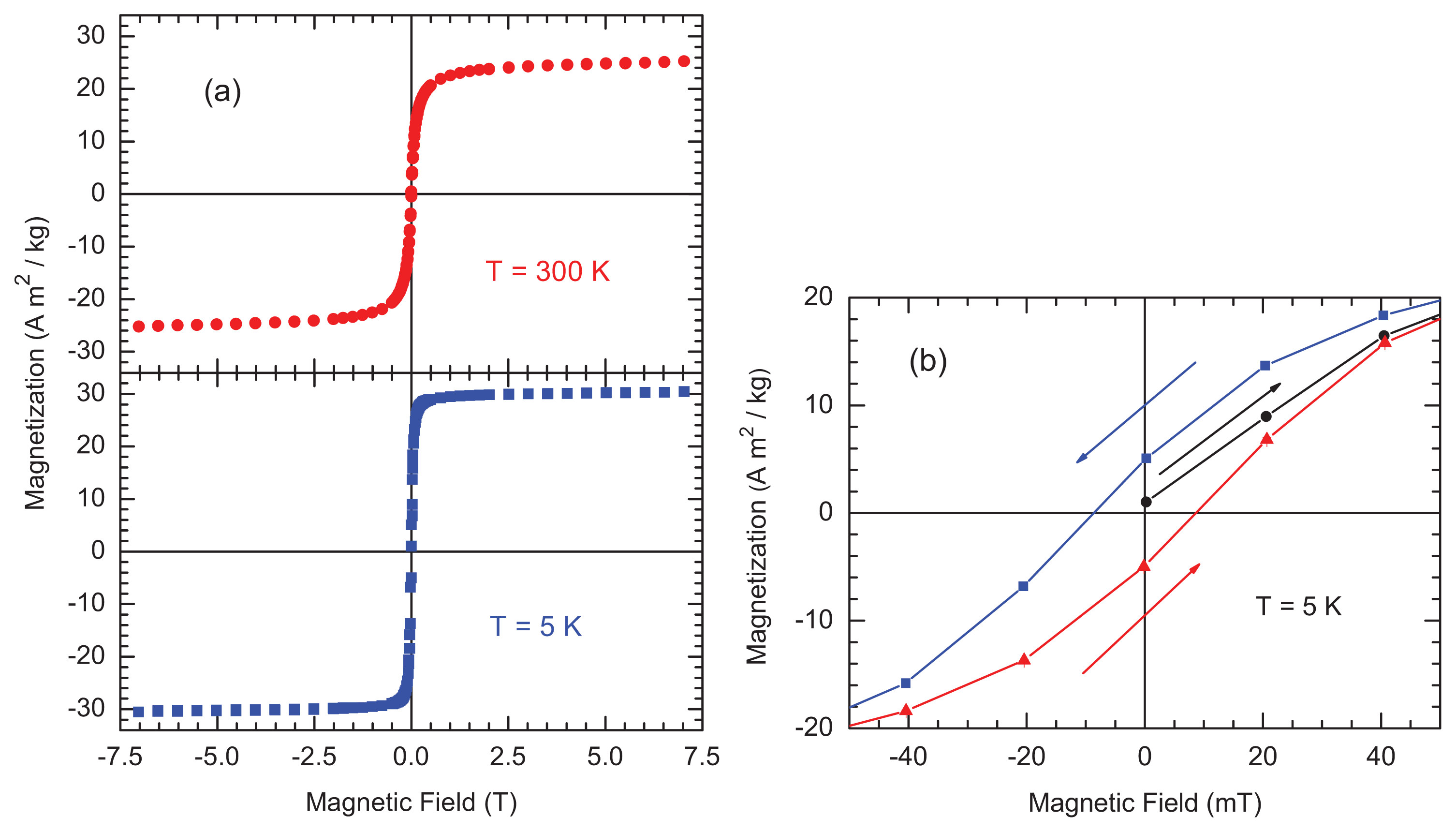
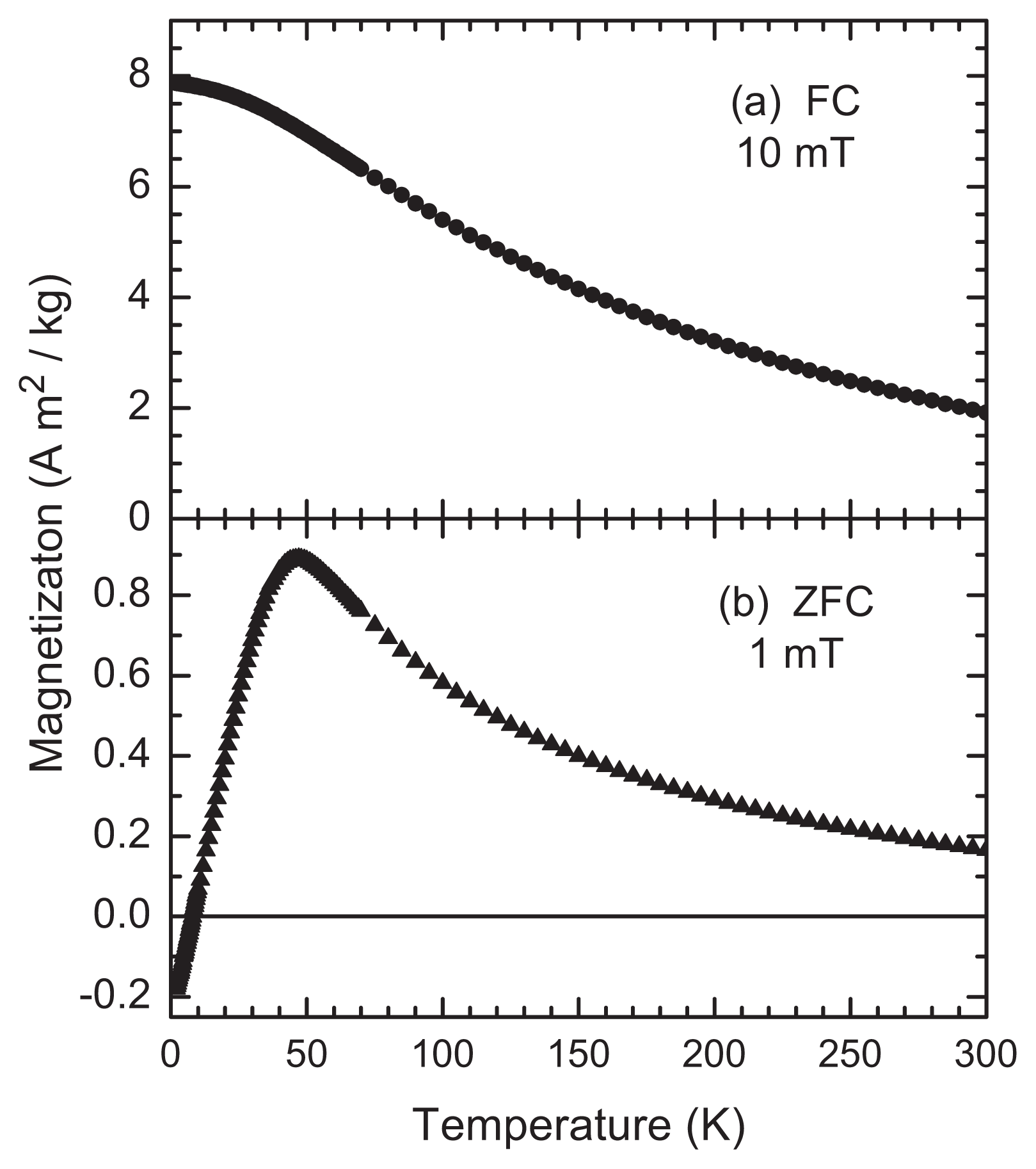
2.2. Magnetic Measurements
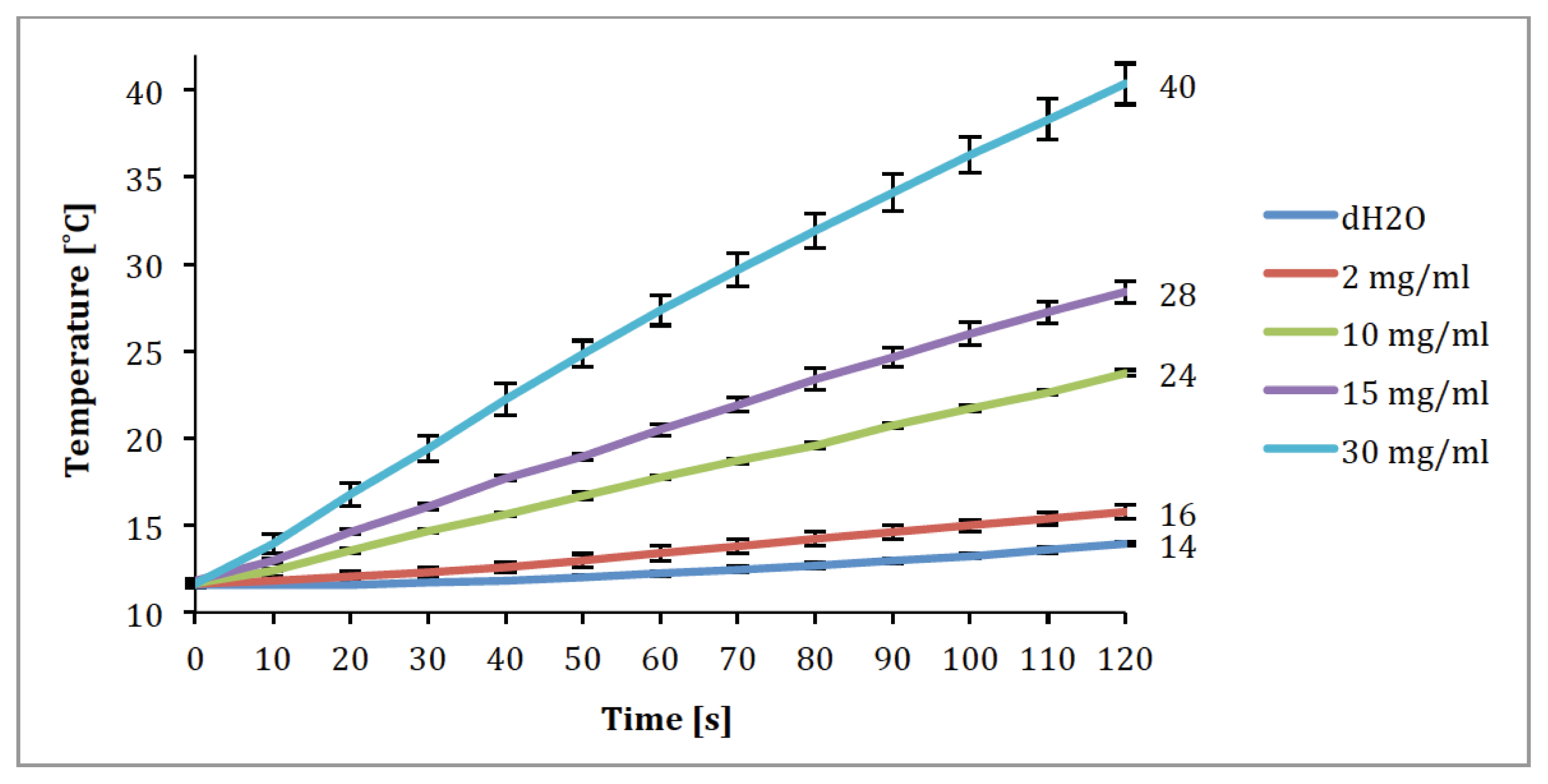
2.3. Magnetic Fluid Hyperthermia
3. Experimental Section
3.1. Feraheme Samples
3.2. High Resolution Transmission Electron Microscopy (HRTEM)
3.3. Superconducting Quantum Interference Device (SQUID) Magnetometry
3.4. Magnetic Fluid Hyperthermia (MFH)
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Pankhurst, Q.A.; Thanh, N.K.T.; Jones, S.K.; Dobson, J. Progress in applications of magnetic nanoparticles in biomedicine. J. Phys. D 2009, 42, 224001, :1–224001:15.. [Google Scholar]
- Liu, F.J.; Laurent, S.; Fattahi, H.; Elst, L.V.; Muller, R.N. Superparamagnetic nanosystems based on iron oxide nanoparticles for biomedical imaging. Nanomedicine 2011, 6, 19–528. [Google Scholar]
- Stephen, Z.R.; Kievit, F.M.; Zhang, M.Q. Magnetite nanoparticles for medical MR imaging. Mat. Today 2011, 14, 330–338. [Google Scholar]
- Pankhurst, Q.A.; Connoly, J.; Jones, S.K.; Dobson, J. Applications of magnetic nanoparticles in biomedicine. J. Phys. D 2003, 36, R167–R181. [Google Scholar]
- Neuwelt, E.A.; Varallyay, P.; Bago, A.G.; Muldoon, L.L.; Nesbit, G.; Nixon, R. Imaging of iron oxide nanoparticles by MR and light microscopy in patients with malignant brain tumours. Neuropath. Appl. Neurobiol 2004, 30, 456–471. [Google Scholar]
- Kumar, M.; Medarova, Z.; Pantazopoulos, P.; Dai, G.P.; Moore, A. Novel membrane-permeable contrast agent for brain tumor detection by MRI. Magn. Res. Med 2010, 63, 617–624. [Google Scholar]
- Berman, S.M.C.; Walczak, P.; Bulte, J.W.M. Tracking stem cells using magnetic nanoparticles. Wiley Interdiscip. Rev. Nanomed. Nanobiotech 2011, 3, 343–355. [Google Scholar]
- Herynek, V.; Berkova, Z.; Horak, D.; Babic, M.; Jirak, D.; Saudek, F.; Hajek, M. Alternative labels for visualization of pancreatic islets. Proc. Intl. Soc. Mag. Reson. Med 2010, 18, 4200. [Google Scholar]
- Provenzano, R.; Schiller, B.; Rao, M.; Coyne, D.; Brenner, L.; Pereira, B.J. Ferumoxytol as an intravenous iron replacement therapy in hemodialysis patients. CJASN 2009, 4, 386–393. [Google Scholar]
- Chen, S. Polymer-Coated Iron Oxide Nanoparticles for Medical Imaging. In Ph.D. Thesis; Massachusetts Institute of Technology: Cambridge, UK, 2010; p. 158. [Google Scholar]
- Balakrishnan, V.S.; Rao, M.; Kasuz, A.T.; Brenner, L.; Pereira, B.J.; Frigo, T.B.; Lewis, J.M. Physicochemical properties of ferumoxytol, a new intravenous iron preparation. Eur. J. Clin. Invest 2009, 39, 489–496. [Google Scholar]
- Barick, K.C.; Aslam, M.; Lin, Y.P.; Bahdur, D.; Prasad, P.V.; Dravid, V.P. Novel and efficient MRactiveaqueous colloidal Fe3O4 nanoparticle assemblies. J. Mater. Chem 2009, 19, 7023–7029. [Google Scholar]
- Dobson, J. Remote control of cellular behavior with magnetic nanoparticles. Nat. Nanotechnol 2008, 3, 139–143. [Google Scholar]
- Ku, S.K.; Lin, J.; Dravid, V.P.; Joshi, H.M. Biological applications of size-controlled iron oxide magnetic nanostructures. Nanoscape 2010, 7, 38–43. [Google Scholar]
- Ge, Q.; Su, J.; Chung, T.-S.; Amy, G. Hydrophilic superparamagnetic nanoparticles: Synthesis, characterization and performance in forward osmosis process. Ind. Eng. Chem. Res 2011, 50, 382–388. [Google Scholar]
- Stjerndahl, M.; Andersson, M.; Hall, H.E.; Pajerowski, D.M.; Meisel, M.W.; Duran, R.S. Superparamagnetic Fe3O4/SiO2 nanocomposites: Enabling the tuning of both the iron oxide load and the size of the nanoparticles. Langmuir 2008, 24, 3532–3536. [Google Scholar]
- Demortiere, A.; Panissod, P.; Pichon, B.P.; Pourroy, G.; Guillon, D.; Donnio, B.; Begin-Colin, S. Size-dependent properties of magnetic iron oxide nanocrystals. Nanoscale 2011, 3, 225–232. [Google Scholar]
- Creixell, M.; Bohorquez, A.C.; Torres-Lugo, M.; Rinaldi, C. EGFR-targeted magnetic nanoparticle heaters can kill cancer cells without a perceptible temperature rise. ACS Nano 2011, 5, 7124–7129. [Google Scholar]

© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bullivant, J.P.; Zhao, S.; Willenberg, B.J.; Kozissnik, B.; Batich, C.D.; Dobson, J. Materials Characterization of Feraheme/Ferumoxytol and Preliminary Evaluation of Its Potential for Magnetic Fluid Hyperthermia. Int. J. Mol. Sci. 2013, 14, 17501-17510. https://doi.org/10.3390/ijms140917501
Bullivant JP, Zhao S, Willenberg BJ, Kozissnik B, Batich CD, Dobson J. Materials Characterization of Feraheme/Ferumoxytol and Preliminary Evaluation of Its Potential for Magnetic Fluid Hyperthermia. International Journal of Molecular Sciences. 2013; 14(9):17501-17510. https://doi.org/10.3390/ijms140917501
Chicago/Turabian StyleBullivant, John P., Shan Zhao, Brad J. Willenberg, Bettina Kozissnik, Christopher D. Batich, and Jon Dobson. 2013. "Materials Characterization of Feraheme/Ferumoxytol and Preliminary Evaluation of Its Potential for Magnetic Fluid Hyperthermia" International Journal of Molecular Sciences 14, no. 9: 17501-17510. https://doi.org/10.3390/ijms140917501
APA StyleBullivant, J. P., Zhao, S., Willenberg, B. J., Kozissnik, B., Batich, C. D., & Dobson, J. (2013). Materials Characterization of Feraheme/Ferumoxytol and Preliminary Evaluation of Its Potential for Magnetic Fluid Hyperthermia. International Journal of Molecular Sciences, 14(9), 17501-17510. https://doi.org/10.3390/ijms140917501




