The Effects of Nanomaterials as Endocrine Disruptors
Abstract
:1. Introduction
2. Impact of Endocrine Disrupting NPs on Reproductive Health
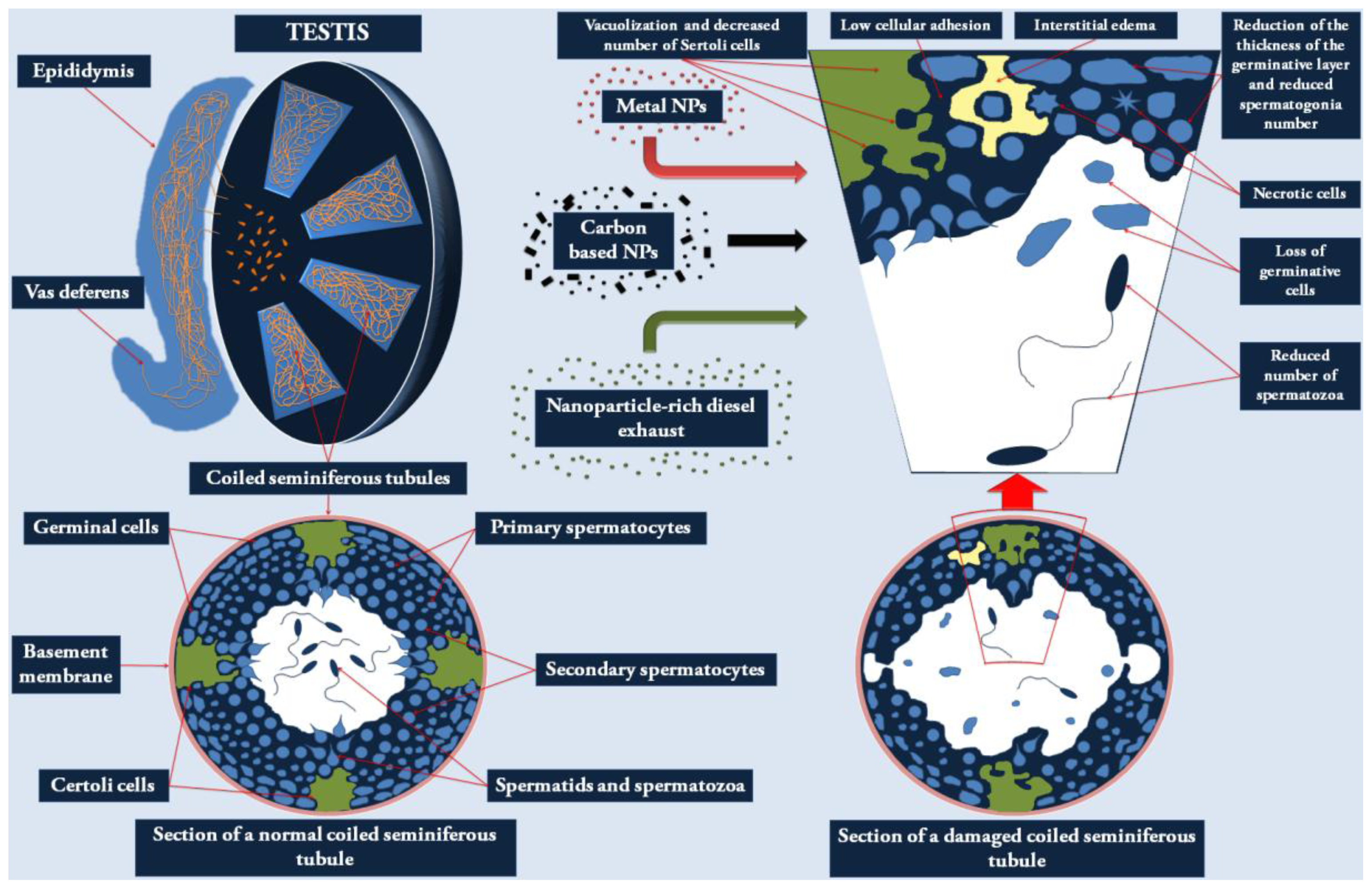
2.1. Effects of NPs on the Male Reproductive System
2.2. Effects of NPs on the Female Reproductive System
2.3. Estrogenic Effects of NPs
3. Impact of Endocrine Disrupting NPs on Thyroid Function
4. Impact of Endocrine Disrupting NPs on Insulin Action and Metabolism
5. Impact of Endocrine Disrupting NPs on the Neuroendocrine System
6. Other Effects of Endocrine Disrupting NPs
7. Impact of Endocrine Disrupting NPs on Invertebrate Species
8. Discussion and Conclusions
Conflicts of Interest
Abbreviations
| 8-OHdG | 8-hydroxy-2-deoxyguanosine |
| ACTH | AdrenoCorticoTropic Hormone |
| ADP | Adenosine Diphosphate |
| Ag-HC-NPs | Hydrocarbon-coated silver Nanoparticles |
| Ag-NPs | Silver Nanoparticles |
| Ag-PS-NPs | Polysaccharide-coated silver Nanoparticles |
| Al-NPs | Aluminum Nanoparticles |
| Al2O3-NPs | Aluminium Oxide Nanoparticles |
| ATP | Adenosine Triphosphate |
| Au-NPs | Gold Nanoparticles |
| C60HyFn | hydrated C60 fullerene |
| CB | Carbon Black |
| CB-NPs | Carbon Black Nanoparticles |
| CdS-QDs | Cadmium Sulfide Quantum Dots |
| CdS/CdTe capped-QDs | Cadmium Sulfide/Cadmium Tellurium capped-Quantum Dots |
| CdSe-core-QDs | Cadmium Selenium core Quantum Dots |
| CdTe-QDs | Cadmium Tellurium Quantum Dots |
| CdTe/ZnTe QDs | Cadmium Tellurium/Zinc Tellurium Quantum Dots |
| CeO2-NPs | Cerium Oxide Nanoparticles |
| Chg | Choriogenin |
| CHO | Chinese Hamster Ovary cells |
| CNTs | Carbon Nanotubes |
| COX-2 | Cyclooxygenase-2 |
| CrCl3-NPs | Chromium Chloride Nanoparticles |
| Cr-NPs | Chromium Nanoparticles |
| Cu-NPs | Copper nanoparticles |
| DA | Dopamine |
| DE-NPs | Diesel Exhaust Nanoparticles |
| DOPAC | dihydroxyphenylacetic acid |
| DSP | Daily Sperm Production |
| EDCs | Endocrine Disrupting Chemicals |
| EDPL | Endocrine Disruptor Priority List |
| ER | Estrogenic Receptor |
| ERK | extracellular signal-regulated kinase |
| EuOH3-NPs | Europium Hydroxied Nanoparticles |
| Eu2O3-NPs | Europium Oxide Nanoparticles |
| Fe3O4-NPs | Magnetic Iron Oxide Nanoparticles |
| FSH | Follicle Stimulating Hormone |
| FT3 | free triiodothyronine |
| FT4 | free thyroxine |
| GH | Growth Hormone |
| GnRH | Gonadotropin Releasing Hormone |
| HMG-CoA | 3-hydroxy-3-methylglutaryl-coenzyme A |
| HVA | HomoVanillic Acid |
| IGF-I | Insulin-like Growth Factor I |
| IR | Insulin Resistance |
| LDL-R | Low Density Lipoprotein Receptor |
| LH | Luteinizing Hormone |
| MAPKs | Mitogen-Activated Protein Kinases |
| MARCO | Macrophage Receptor with Collagenous structure |
| MN | Micro Nuclei |
| MnO-NPs | Manganese Oxide Nanoparticles |
| MoO3-NPs | Molybdenum Trioxide Nanoparticles |
| mPEG@Au-NP | ω-methoxy poly(ethylene glycol) capped gold-NPs |
| MTT | 3-(4,5-dimethyl-2-thiazol)-2,5-diphenyl- 2H-tetrazolium bromide |
| MWCNTs | Multi Walled Carbon Nanotubes |
| NPs | NanoParticles |
| NRDE-NPs | Nanoparticle-Rich Diesel Exhaust Nanoparticles |
| nSP | amorphous nanosilica particles |
| Ntera2, NT2 | human testicular embryonic carcinoma cell line |
| P450 17α | Cytochrome P450 17α |
| P450 17β-HSD | Cytochrome P450 17β-HydroxySteroid Dehydrogenase |
| P450arom | Aromatase P450 |
| P450scc | Cytochrome P450 side-chain cleavage |
| PBR | Peripheral-type Benzodiazepine Receptor |
| PEG | PolyEthilen-Glycol |
| PEG-NH2@Au-NP | ω-aminoethyl poly(ethylene glycol) capped gold-NPs |
| POP | Persistent Organic Pollutants |
| QDs | Quantum Dots |
| RLKI | Rana Larval Keratin Type I |
| ROS | Reactive Oxygen Species |
| SCE | Sister Chromatid Exchanges |
| SiCNWs | Silicon Carbide Nanowires |
| s-MWCNTs-PEG | synthesized functionalized MWCNTs with polyethylene glycol |
| SR-B1 | Scavenger Receptor class B type 1 |
| SSC | Spermatogonial Stem Cells |
| StAR | Steroidogenic Acute Regulatory protein |
| T2DM | Type 2 Diabetes Mellitus |
| TH | Thyroid Hormone |
| TiO2 | Titanium Dioxide |
| TiO2-NPs | Titanium Dioxide nanoparticles |
| TRβ | Thyroid hormone Receptor β |
| TSH | Thyrotropic-Stimulating Hormone |
| UV | Ultra Violet |
| VTG | Vitellogenin |
| ZnO-NPs | Zinc Oxide Nanoparticles |
| ZnS | Zinc Sulfide |
| ZnS-QDs | Zinc Sulfide Quantum Dots |
References
- Iavicoli, I.; Fontana, L.; Bergamaschi, A. The effects of metals as endocrine disruptors. J. Toxicol. Environ. Health B 2009, 12, 206–223. [Google Scholar]
- De Coster, S.; van Larebeke, N. Endocrine-disrupting chemicals: Associated disorders and mechanisms of action. J. Environ. Public Health 2012. [Google Scholar] [CrossRef]
- World Health Organization (WHO), State of the Science of Endocrine Disrupting Chemicals–2012; World Health Organization: Geneva, Switzerland, 2012; pp. 23–237.
- Wormley, D.D.; Ramesh, A.; Hood, D.B. Environmental contaminant-mixture effects on CNS development, plasticity and behavior. Toxicol. Appl. Pharmacol 2004, 197, 49–65. [Google Scholar]
- Diamanti-Kandarakis, E.; Bourguignon, J.P.; Giudice, L.C.; Hauser, R.; Prins, G.S.; Soto, A.M.; Zoeller, R.T.; Gore, A.C. Endocrine-disrupting chemicals: An Endocrine Society scientific statement. Endocr. Rev 2009, 30, 293–342. [Google Scholar]
- Soto, A.M.; Sonnenschein, C. Environmental causes of cancer: Endocrine disruptors as carcinogens. Nat. Rev. Endocrinol 2010, 6, 363–370. [Google Scholar]
- Frye, C.A.; Bo, E.; Calamandrei, G.; Calzà, L.; Dessì-Fulgheri, F.; Fernández, M.; Fusani, L.; Kah, O.; Kajta, M.; Le Page, Y.; et al. Endocrine disrupters: A review of some sources, effects, and mechanisms of actions on behaviour and neuroendocrine systems. J. Neuroendocrinol 2012, 24, 144–159. [Google Scholar]
- Karoutsou, E.; Polymeris, A. Environmental endocrine disruptors and obesity. Endocr. Regul 2012, 46, 37–46. [Google Scholar]
- Meeker, J.D. Exposure to environmental endocrine disruptors and child development. Arch. Pediatr. Adolesc. Med 2012, 166, 952–958. [Google Scholar]
- Rogers, J.A.; Metz, L.; Yong, V.W. Endocrine disrupting chemicals and immune responses: A focus on bisphenol-A and its potential mechanisms. Mol. Immunol 2013, 53, 421–430. [Google Scholar]
- World Health Organization (WHO), Global Assessment of the State-of-the-Science of Endocrine Disruptors; World Health Organization: Geneva, Switzerland, 2002; pp. 1–3.
- Casals-Casas, C.; Desvergne, B. Endocrine disruptors: From endocrine to metabolic disruption. Annu. Rev. Physiol 2011, 73, 135–162. [Google Scholar]
- Baccarelli, A.; Pesatori, A.C.; Bertazzi, P.A. Occupational and environmental agents as endocrine disruptors: Experimental and human evidence. J. Endocrinol. Invest 2000, 23, 771–781. [Google Scholar]
- Ying, G.G. Endocrine Disrupting Chemicals. What? Where? In Analysis of Endocrine Disrupting Compounds in Food; Nollet, L.M.L., Ed.; Wiley-Blackwell: Oxford, UK, 2011; pp. 3–17. [Google Scholar]
- Groshart, C.; Okkerman, P.C. Towards the Establishment of a Priority List of Substances for Further Evaluation of Their Role in Endocrine Disruption—Preparation of a Candidate List of Substances as a Basis for Priority Setting; BKH Consulting Engineers for European Commission Directorate-General for the Environment: Delft, The Netherlands, 2000; pp. 1–29. [Google Scholar]
- Johnson, I.; Harvey, P. Study on the Scientific Evaluation of 12 Substances in the Context of Endocrine Disrupter Priority List of Actions; WRc-NSF for European Commission Directorate-General for the Environment: Medmenham Marlow, UK; p. 2002.
- Okkerman, P.C.; van der Putte, I. Endocrine Disrupters: Study on Gathering Information on 435 Substances with Insufficient Data; RPS BKH Consulting Engineers for European Commission Directorate-General for the Environment: Delft, The Netherlands, 2002; pp. 1–131. [Google Scholar]
- Petersen, G.; Rasmussen, D.; Gustavson, K. Study on Enhancing the Endocrine Disrupter Priority List with a Focus on Low Production Volume Chemical; DHI water & environment for European Commission Directorate-General for the Environment: Hørsholm, Denmark, 2007; pp. 1–249. [Google Scholar]
- International Organization for Standardization (ISO), Nanotechnologies—Terminology and Definitions for Nano-objects—Nanoparticle, Nanofibre and Nanoplate; International Organization for Standardization: Geneva, Switzerland, 2008; pp. 1–7.
- National Institute for Occupational Safety and Health (NIOSH), Approaches to Safe Nanotechnology: Managing the Health and Safety Concerns Associated with Engineered Nanomaterials; National Institute for Occupational Safety and Health, Department of Health and Human Services, Centers for Disease Control and Prevention: Atlanta, GA, USA, 2009; pp. 1–86.
- Nel, A.; Xia, T.; Madler, L.; Li, N. Toxic potential of materials at the nanolevel. Science 2006, 311, 622–627. [Google Scholar]
- Canton, I.; Battaglia, G. Endocytosis at the nanoscale. Chem. Soc. Rev 2012, 41, 2718–2739. [Google Scholar]
- Zhao, F.; Zhao, Y.; Liu, Y.; Chang, X.; Chen, C.; Zhao, Y. Cellular uptake, intracellular trafficking, and cytotoxicity of nanomaterials. Small 2011, 7, 1322–1337. [Google Scholar]
- Iavicoli, I.; Calabrese, E.J.; Nascarella, M.A. Exposure to nanoparticles and hormesis. Dose Response 2010, 8, 501–517. [Google Scholar]
- Italian Workers’ Compensation Authority (INAIL), White Book, Exposure to Engineered Nanomaterials and Occupational Health and Safety Effects; Italian Workers’ Compensation Authority, Department of Occupational Medicine, formerly ISPESL: Rome, Italy, 2011; pp. 1–211.
- Landsiedel, R.; Fabian, E.; Ma-Hock, L.; van Ravenzwaay, B.; Wohlleben, W.; Wiench, K.; Oesch, F. Toxico-/biokinetics of nanomaterials. Arch. Toxicol 2012, 86, 1021–1060. [Google Scholar]
- National Institute for Occupational Safety and Health (NIOSH), Filling the Knowledge Gaps for Safe Nanotechnology in the Workplace. A Progress Report from the NIOSH Nanotechnology Research Center 2004–2011; National Institute for Occupational Safety and Health, Department of Health and Human Services, Centers for Disease Control and Prevention: Atlanta, GA, USA, 2012; pp. 1–395.
- Knez, J. Endocrine-disrupting chemicals and male reproductive health. Reprod. Biomed. Online 2013, 26, 440–448. [Google Scholar]
- Fowler, P.A.; Bellingham, M.; Sinclair, K.D.; Evans, N.P.; Pocar, P.; Fischer, B.; Schaedlich, K.; Schmidt, J.S.; Amezaga, M.R.; Bhattacharya, S.; et al. Impact of endocrine-disrupting compounds (EDCs) on female reproductive health. Mol. Cell. Endocrinol 2012, 355, 231–239. [Google Scholar]
- Komatsu, T.; Tabata, M.; Kubo-Irie, M.; Shimizu, T.; Suzuki, K.; Nihei, Y.; Takeda, K. The effects of nanoparticles on mouse testis Leydig cells in vitro. Toxicol. In Vitro 2008, 22, 1825–1831. [Google Scholar]
- Vevers, W.F.; Jha, A.N. Genotoxic and cytotoxic potential of titanium dioxide (TiO2) nanoparticles on fish cells in vitro. Ecotoxicology 2008, 17, 410–420. [Google Scholar]
- Rosenkranz, P.; Fernández-Cruz, M.L.; Conde, E.; Ramírez-Fernández, M.B.; Flores, J.C.; Fernández, M.; Navas, J.M. Effects of cerium oxide nanoparticles to fish and mammalian cell lines: An assessment of cytotoxicity and methodology. Toxicol. In Vitro 2012, 26, 888–896. [Google Scholar]
- Li, C.; Taneda, S.; Taya, K.; Watanabe, G.; Li, X.; Fujitani, Y.; Ito, Y.; Nakajima, T.; Suzuki, A.K. Effects of inhaled nanoparticle-rich diesel exhaust on regulation of testicular function in adult male rats. Inhal. Toxicol 2009, 21, 803–811. [Google Scholar]
- Li, C.; Taneda, S.; Taya, K.; Watanabe, G.; Li, X.; Fujitani, Y.; Nakajima, T.; Suzuki, A.K. Effects of in utero exposure to nanoparticle-rich diesel exhaust on testicular function in immature male rats. Toxicol. Lett 2009, 185, 1–8. [Google Scholar]
- Takeda, K.; Suzuki, K.I.; Ishihara, A.; Kubo-Irie, M.; Fujimoto, R.; Tabata, M.; Oshio, S.; Nihei, Y.; Ihara, T.; Sugamata, M. Nanoparticles transferred from pregnant mice to their offspring can damage the genital and cranial nerve systems. J. Health Sci 2009, 55, 95–102. [Google Scholar]
- Yoshida, S.; Hiyoshi, K.; Ichinose, T.; Takano, H.; Oshio, S.; Sugawara, I.; Takeda, K.; Shibamoto, T. Effect of nanoparticles on the male reproductive system of mice. Int. J. Androl 2009, 32, 337–342. [Google Scholar]
- Bai, Y.; Zhang, Y.; Zhang, J.; Mu, Q.; Zhang, W.; Butch, E.R.; Snyder, S.E.; Yan, B. Repeated administrations of carbon nanotubes in male mice cause reversible testis damage without affecting fertility. Nat. Nanotechnol 2010, 5, 683–689. [Google Scholar]
- Yoshida, S.; Hiyoshi, K.; Oshio, S.; Takano, H.; Takeda, K.; Ichinose, T. Effects of fetal exposure to carbon nanoparticles on reproductive function in male offspring. Fertil. Steril 2010, 93, 1695–1699. [Google Scholar]
- Li, W.Q.; Wang, F.; Liu, Z.M.; Wang, Y.C.; Wang, J.; Sun, F. Gold nanoparticles elevate plasma testosterone levels in male mice without affecting fertility. Small 2013, 27, 1708–1714. [Google Scholar]
- Morishita, Y.; Yoshioka, Y.; Satoh, H.; Nojiri, N.; Nagano, K.; Abe, Y.; Kamada, H.; Tsunoda, S.; Nabeshi, H.; Yoshikawa, T.; et al. Distribution and histologic effects of intravenously administered amorphous nanosilica particles in the testes of mice. Biochem. Biophys. Res. Commun 2012, 420, 297–301. [Google Scholar]
- Murugan, M.A.; Gangadharan, B.; Mathur, P.P. Antioxidative effect of fullerenol on goat epididymal spermatozoa. Asian J. Androl 2002, 4, 149–152. [Google Scholar]
- Braydich-Stolle, L.; Hussain, S.; Schlager, J.J.; Hofmann, M.C. In vitro cytotoxicity of nanoparticles in mammalian germline stem cells. Toxicol. Sci 2005, 88, 412–419. [Google Scholar]
- Makhluf, S.B.D.; Qasem, R.; Rubinstein, S.; Gedanken, A.; Breibart, H. Loading magnetic nanoparticles into sperm cells does not affect their functionality. Langmuir 2006, 22, 9480–9482. [Google Scholar]
- Makhluf, S.B.D.; Arnon Patra, C.R.; Mukhopadhyay, D.; Gedanken, A.; Mukherjee, P.; Breitbart, H. Labeling of sperm cells via the spontaneous penetration of Eu3+ ions as nanoparticles complexed with PVA or PVP. J. Phys. Chem. C 2008, 112, 12801–12807. [Google Scholar]
- Gopalan, R.; Osman, I.; Amani, A.; de Matas, M.; Anderson, D. The effect of zinc oxide and titanium dioxide nanoparticles in the comet assay with UVA photoactivation of human sperm and lymphocytes. Nanotoxicology 2009, 3, 33–39. [Google Scholar]
- Wiwanitkit, V.; Sereemaspun, A.; Rojanathanes, R. Effect of gold nanoparticles on spermatozoa: The first world report. Fertil. Steril 2009, 91, e7–e8. [Google Scholar]
- Braydich-Stolle, L.K.; Lucas, B.; Schrand, A.; Murdock, R.C.; Lee, T.; Schlager, J.; Hussain, S.; Hofmann, M.C. Silver nanoparticles disrupt GDNF/Fyn kinase signalling in spermatogonial stem cells. Toxicol. Sci 2010, 116, 577–589. [Google Scholar]
- Taylor, U.; Petersen, S.; Barchanski, A.; Mittag, A.; Barcikowski, S.; Rath, D. Influence of gold nanoparticles on vitality parameters of bovine spermatozoa. Reprod. Domest. Anim 2010, 45, 60. [Google Scholar]
- Zakhidov, S.T.; Marshak, T.L.; Malolina, E.A.; Kulibin, A.Y.; Zelenina, I.A.; Pavluchenkova, S.M.; Rudoy, V.M.; Dementeva, O.V.; Skuridin, S.G.; Evdokimov, Y.M. Gold nanoparticles disturb nuclear chromatin decondensation in mouse sperm in vitro. Biol. Membr 2010, 4, 349–353. [Google Scholar]
- Terzuoli, G.; Iacoponi, F.; Moretti, E.; Renieri, T.; Baldi, G.; Collodel, G. In vitro effect of silver engineered nanoparticles on human spermatozoa. J. Siena Acad. Sci 2011, 3, 27–29. [Google Scholar]
- Asare, N.; Instanes, C.; Sandberg, W.J.; Refsnes, M.; Schwarze, P.; Kruszewski, M.; Brunborg, G. Cytotoxic and genotoxic effects of silver nanoparticles in testicular cells. Toxicology 2012, 291, 65–72. [Google Scholar]
- Guo, L.L.; Liu, X.H.; Qin, D.X.; Gao, L.; Zhang, H.M.; Liu, J.Y.; Cui, Y.G. Effects of nanosized titanium dioxide on the reproductive system of male mice. Zhonghua Nan Ke Xue 2009, 15, 517–522. [Google Scholar]
- Bal, R.; Türk, G.; Tuzcu, M.; Yilmaz, O.; Ozercan, I.; Kuloglu, T.; Gür, S.; Nedzvetsky, V.S.; Tykhomyrov, A.A.; Andrievsky, G.V.; et al. Protective effects of nanostructures of hydrated C(60) fullerene on reproductive function in streptozotocin-diabetic male rats. Toxicology 2011, 282, 69–81. [Google Scholar]
- Noori, A.; Parivar, K.; Modaresi, M.; Messripour, M.; Yousefi, M.H.; Amiri, G.R. Effect of magnetic iron oxide nanoparticles on pregnancy and testicular development of mice. Afr. J. Biotechnol 2011, 10, 1221–1227. [Google Scholar]
- Sycheva, L.P.; Zhurkov, V.S.; Iurchenko, V.V.; Daugel-Dauge, N.O.; Kovalenko, M.A.; Krivtsova, E.K.; Durnev, A.D. Investigation of genotoxic and cytotoxic effects of micro- and nanosized titanium dioxide in six organs of mice in vivo. Mutat. Res 2011, 726, 8–14. [Google Scholar]
- Gromadzka-Ostrowska, J.; Dziendzikowska, K.; Lankoff, A.; Dobrzyńska, M.; Instanes, C.; Brunborg, G.; Gajowik, A.; Radzikowska, J.; Wojewódzka, M.; Kruszewski, M. Silver nanoparticles effects on epididymal sperm in rats. Toxicol. Lett 2012, 214, 251–258. [Google Scholar]
- Tang, S.; Tang, Y.; Zhong, L.; Murat, K.; Asan, G.; Yu, J.; Jian, R.; Wang, C.; Zhou, P. Short- and long-term toxicities of multi-walled carbon nanotubes in vivo and in vitro. J. Appl. Toxicol 2012, 32, 900–912. [Google Scholar]
- Kyjovska, Z.O.; Boisen, A.M.; Jackson, P.; Wallin, H.; Vogel, U.; Hougaard, K.S. Daily sperm production: Application in studies of prenatal exposure to nanoparticles in mice. Reprod. Toxicol 2013, 36, 88–97. [Google Scholar]
- Li, C.; Li, X.; Jigami, J.; Hasegawa, C.; Suzuki, A.K.; Zhang, Y.; Fujitani, Y.; Nagaoka, K.; Watanabe, G.; Taya, K. Effect of nanoparticle-rich diesel exhaust on testosterone biosynthesis in adult male mice. Inhal. Toxicol 2012, 24, 599–608. [Google Scholar]
- Ramdhan, D.H.; Ito, Y.; Yanagiba, Y.; Yamagishi, N.; Hayashi, Y.; Li, C.; Taneda, S.; Suzuki, A.K.; Watanabe, G.; Taya, K.; et al. Nanoparticle-rich diesel exhaust may disrupt testosterone biosynthesis and metabolism via growth hormone. Toxicol. Lett 2009, 191, 103–108. [Google Scholar]
- Yamagishi, N.; Ito, Y.; Ramdhan, D.H.; Yanagiba, Y.; Hayashi, Y.; Wang, D.; Li, C.M.; Taneda, S.; Suzuki, A.K.; Taya, K.; et al. Effect of nanoparticle-rich diesel exhaust on testicular and hippocampus steroidogenesis in male rats. Inhal. Toxicol 2012, 24, 459–467. [Google Scholar]
- Cho, W.S.; Duffin, R.; Poland, C.A.; Howie, S.E.; MacNee, W.; Bradley, M.; Megson, I.L.; Donaldson, K. Metal oxide nanoparticles induce unique inflammatory footprints in the lung: Important implications for nanoparticle testing. Environ. Health Perspect 2010, 118, 1699–1706. [Google Scholar]
- Rafeeqi, T.; Kaul, G. Carbon nanotubes as a scaffold for spermatogonial cell maintenance. J. Biomed. Nanotechnol 2010, 6, 710–717. [Google Scholar]
- Zakhidov, S.T.; Pavliuchenkova, S.M.; Marshak, T.L.; Rudoĭ, V.M.; Dementeva, O.V.; Zelenina, I.A.; Skuridin, S.G.; Makarov, A.A.; Khokhlov, A.N.; Evdokimov, I.M. Effect of gold nanoparticles on mouse spermatogenesis. Izv. Akad. Nauk. Ser. Biol 2012, 3, 279–287. [Google Scholar]
- Taylor, U.; Barchanski, A.; Garrels, W.; Klein, S.; Kues, W.; Barcikowski, S.; Rath, D. Toxicity of gold nanoparticles on somatic and reproductive cells. Adv. Exp. Med. Biol 2012, 733, 125–133. [Google Scholar]
- West, L.A.; Horvat, R.D.; Roess, D.A.; Barisas, B.G.; Juengel, J.L.; Niswender, G.D. Steroidogenic acute regulatory protein and peripheral-type benzodiazepine receptor associate at the mitochondrial membrane. Endocrinology 2001, 142, 502–555. [Google Scholar]
- Payne, A.H. Hormonal regulation of cytochrome P450 enzymes, cholesterol side-chain cleavage and 17 alpha-hydroxylase/C17–20 lyase in Leydig cells. Biol. Reprod 1990, 42, 399–404. [Google Scholar]
- Uchino, T.; Tokunaga, H.; Ando, M.; Utsumi, H. Quantitative determination of OH radical generation and its cytotoxicity induced by TiO(2)-UVA treatment. Toxicol. In Vitro 2002, 16, 629–635. [Google Scholar]
- Theogaraj, E.; Riley, S.; Hughes, L.; Maier, M.; Kirkland, D. An investigation of the photo-clastogenic potential of ultrafine titanium dioxide particles. Mutat. Res 2007, 634, 205–219. [Google Scholar]
- Warheit, D.B.; Hoke, R.A.; Finlay, C.; Donner, E.M.; Reed, K.L.; Sayes, C.M. Development of a base set of toxicity tests using ultrafine TiO2 particles as a component of nanoparticle risk management. Toxicol. Lett 2007, 171, 99–110. [Google Scholar]
- Zhu, R.R.; Wang, S.L.; Chao, J.; Shi, D.L.; Zhang, R.; Sun, X.Y.; Yao, S.D. Bio-effects of nano-TiO2 on DNA and cellular ultrastructure with different polymorph and size. Mater. Sci. Eng. C 2009, 29, 691–696. [Google Scholar]
- Di Virgilio, A.L.; Reigosa, M.; Arnal, P.M.; Fernández Lorenzo de Mele, M. Comparative study of the cytotoxic and genotoxic effects of titanium oxide and aluminium oxide nanoparticles in Chinese hamster ovary (CHO-K1) cells. J. Hazard. Mater 2010, 177, 711–718. [Google Scholar]
- Jiang, J.; Wang, J.; Zhang, X.; Huo, K.; Wong, H.M.; Yeung, K.W.; Zhang, W.; Hu, T.; Chu, P.K. Activation of mitogen-activated protein kinases cellular signal transduction pathway in mammalian cells induced by silicon carbide nanowires. Biomaterials 2010, 31, 7856–7862. [Google Scholar]
- Liu, X.; Qin, D.; Cui, Y.; Chen, L.; Li, H.; Chen, Z.; Gao, L.; Li, Y.; Liu, J. The effect of calcium phosphate nanoparticles on hormone production and apoptosis in human granulosa cells. Reprod. Biol. Endocrinol 2010, 8, 32–39. [Google Scholar]
- Abdullah, C.A.; Asanithi, P.; Brunner, E.W.; Jurewicz, I.; Bo, C.; Azad, C.L.; Ovalle-Robles, R.; Fang, S.; Lima, M.D.; Lepro, X.; et al. Aligned, isotropic and patterned carbon nanotube substrates that control the growth and alignment of Chinese hamster ovary cells. Nanotechnology 2011, 22, 205102, :1–205102:11.. [Google Scholar]
- Wang, S.; Hunter, L.A.; Arslan, Z.; Wilkerson, M.G.; Wickliffe, J.K. Chronic exposure to nanosized, anatase titanium dioxide is not cyto- or genotoxic to Chinese hamster ovary cells. Environ. Mol. Mutagen 2011, 52, 614–622. [Google Scholar]
- Wang, S.; Yu, H.; Wickliffe, J.K. Limitation of the MTT and XTT assays for measuring cell viability due to superoxide formation induced by nano-scale TiO2. Toxicol. In Vitro 2011, 25, 2147–2151. [Google Scholar]
- Ekkapongpisit, M.; Giovia, A.; Follo, C.; Caputo, G.; Isidoro, C. Biocompatibility, endocytosis, and intracellular trafficking of mesoporous silica and polystyrene nanoparticles in ovarian cancer cells: Effects of size and surface charge groups. Int. J. Nanomed 2012, 7, 4147–4158. [Google Scholar]
- Hirano, S.; Fujitani, Y.; Furuyama, A.; Kanno, S. Macrophage receptor with collagenous structure (MARCO) is a dynamic adhesive molecule that enhances uptake of carbon nanotubes by CHO-K1 cells. Toxicol. Appl. Pharmacol 2012, 259, 96–103. [Google Scholar]
- Hou, J.; Wan, X.Y.; Wang, F.; Xu, G.F.; Liu, Z. Effects of titanium dioxide nanoparticles on development and maturation of rat preantral follicle in vitro. Acad. J. Second Mil. Med. Univ 2009, 29, 869–873. [Google Scholar]
- Hsieh, M.S.; Shiao, N.H.; Chan, W.H. Cytotoxic effects of CdSe quantum dots on maturation of mouse oocytes, fertilization, and fetal development. Int. J. Mol. Sci 2009, 10, 2122–2135. [Google Scholar]
- Xu, G.; Lin, X.; Yong, K.T.; Roy, I.; Qu, J.; Wang, X. Visualization of Reproduction Toxicity of QDs for in vitro Oocytes Maturation. In Progress in biomedical optics and imaging; Proceedings of SPIE 7519 Eighth International Conference on Photonics and Imaging in Biology and Medicine, Wuhan, China, 8–10 August 2009, Luo, Q., Wang, L.V., Tuchin, V.V., Li, P., Fu, L., Eds.; SPIE: Bellingham,WA, USA, 2009; 7519 1W; pp. 7519–7583. [Google Scholar]
- Wang, X.M.; Yong, K.T.; Xu, G.X.; Lin, X.T.; Zhou, X.Q.; Qu, J.L.; Chen, S.; Hanben, C. Invasion of CdSe/CdS/ZnS Quantum Dots for oocytes in vitro maturation. Chin. Laser 2010, 37, 2730–2734. [Google Scholar]
- Xu, G.; Lin, S.; Law, W.C.; Roy, I.; Lin, X.; Mei, S.; Ma, H.; Chen, S.; Niu, H.; Wang, X. The invasion and reproductive toxicity of QDs-transferrin bioconjugates on preantral follicle in vitro. Theranostics 2012, 2, 734–745. [Google Scholar]
- Wang, J.; Zhu, X.; Zhang, X.; Zhao, Z.; Liu, H.; George, R.; Wilson-Rawls, J.; Chang, Y.; Chen, Y. Disruption of zebrafish (Danio rerio) reproduction upon chronic exposure to TiO2 nanoparticles. Chemosphere 2011, 83, 461–467. [Google Scholar]
- Gao, G.; Ze, Y.; Li, B.; Zhao, X.; Zhang, T.; Sheng, L.; Hu, R.; Gui, S.; Sang, X.; Sun, Q.; et al. Ovarian dysfunction and gene-expressed characteristics of female mice caused by long-term exposure to titanium dioxide nanoparticles. J. Hazard. Mater 2012, 243, 19–27. [Google Scholar]
- Griffitt, R.J.; Brown-Peterson, N.J.; Savin, D.A.; Manning, C.S.; Boube, I.; Ryan, R.A.; Brouwer, M. Effects of chronic nanoparticulate silver exposure to adult and juvenile sheepshead minnows (Cyprinodon variegatus). Environ. Toxicol. Chem 2012, 31, 160–167. [Google Scholar]
- Ramsden, C.S.; Henry, T.B.; Handy, R.D. Sub-lethal effects of titanium dioxide nanoparticles on the physiology and reproduction of zebrafish. Aquat. Toxicol 2013, 126, 404–413. [Google Scholar]
- Stelzer, R.; Hutz, R.J. Gold nanoparticles enter rat ovarian granulosa cells and subcellular organelles, and alter in-vitro estrogen accumulation. J. Reprod. Dev 2009, 55, 685–690. [Google Scholar]
- Li, C.; Li, X.; Suzuki, A.K.; Zhang, Y.; Fujitani, Y.; Nagaoka, K.; Watanabe, G.; Taya, K. Effects of exposure to nanoparticle-rich diesel exhaust on pregnancy in rats. J. Reprod. Dev 2013, 59, 145–150. [Google Scholar]
- Esmaeillou, M.; Moharamnejad, M.; Hsankhani, R.; Tehrani, A.A.; Maadi, H. Toxicity of ZnO nanoparticles in healthy adult mice. Environ. Toxicol. Pharmacol 2013, 35, 67–71. [Google Scholar]
- Pham, C.H.; Yi, J.; Gu, M.B. Biomarker gene response in male Medaka (Oryzias latipes) chronically exposed to silver nanoparticle. Ecotoxicol. Environ. Saf 2012, 78, 239–245. [Google Scholar]
- Gagné, F.; André, C.; Skirrow, R.; Gélinas, M.; Auclair, J.; van Aggelen, G.; Turcotte, P.; Gagnon, C. Toxicity of silver nanoparticles to rainbow trout: A toxicogenomic approach. Chemosphere 2012, 89, 615–622. [Google Scholar]
- Gagné, F.; Fortier, M.; Yu, L.; Osachoff, H.L.; Skirrow, R.C.; van Aggelen, G.; Gagnon, C.; Fournier, M. Immunocompetence and alterations in hepatic gene expression in rainbow trout exposed to CdS/CdTe quantum dots. J. Environ. Monit 2010, 12, 1556–1565. [Google Scholar]
- Sanders, M.B.; Sebire, M.; Sturve, J.; Christian, P.; Katsiadaki, I.; Lyons, B.P.; Sheahan, D.; Weeks, J.M.; Feist, S.W. Exposure of sticklebacks (Gasterosteus aculeatus) to cadmium sulfide nanoparticles: Biological effects and the importance of experimental design. Mar. Environ. Res 2008, 66, 161–163. [Google Scholar]
- Li, C.; Li, X.; Suzuki, A.K.; Fujitani, Y.; Jigami, J.; Nagaoka, K.; Watanabe, G.; Taya, K. Effects of exposure to nanoparticle-rich diesel exhaust on adrenocortical function in adult male mice. Toxicol. Lett 2012, 209, 277–281. [Google Scholar]
- Park, J.W.; Henry, T.B.; Menn, F.M.; Compton, R.N.; Sayler, G. No bioavailability of 17α-ethinylestradiol when associated with nC60 aggregates during dietary exposure in adult male zebrafish (Danio rerio). Chemosphere 2010, 81, 1227–1232. [Google Scholar]
- Park, J.W.; Henry, T.B.; Ard, S.; Menn, F.M.; Compton, R.N.; Sayler, G.S. The association between nC60 and 17α-ethinylestradiol (EE2) decreases EE2 bioavailability in zebrafish and alters nanoaggregate characteristics. Nanotoxicology 2011, 5, 406–416. [Google Scholar]
- Jain, M.P.; Vaisheva, F.; Maysinger, D. Metalloestrogenic effects of quantum dots. Nanomedicine 2012, 7, 23–37. [Google Scholar]
- Jackson, P.; Vogel, U.; Wallin, H.; Hougaard, K.S. Prenatal exposure to carbon black (printex 90): Effects on sexual development and neurofunction. Basic Clin. Pharmacol. Toxicol 2011, 109, 434–437. [Google Scholar]
- Lavado-Autric, R.; Auso, E.; Garcia-Velasco, J.V.; Arufe, M.C.; Escobar, D.R.; Berbel, P.; Morreale, D.E. Early maternal hypothyroxinemia alters histogenesis and cerebral cortex cytoarchitecture of the progeny. J. Clin. Invest 2003, 111, 1073–1082. [Google Scholar]
- Auso, E.; Lavado-Autric, R.; Cuevas, E.; del Rey, F.E.; Morreale, D.E.; Berbel, P. A moderate and transient deficiency of maternal thyroid function at the beginning of fetal neocorticogenesis alters neuronal migration. Endocrinology 2004, 145, 4037–4047. [Google Scholar]
- Berbel, P.; Mestre, J.L.; Santamaria, A.; Palazon, I.; Franco, A.; Graells, M.; Gonzalez-Torga, A.; de Escobar, G.M. Delayed neurobehavioral development in children born to pregnant women with mild hypothyroxinemia during the first month of gestation: The importance of early iodine supplementation. Thyroid 2009, 19, 511–519. [Google Scholar]
- Boas, M.; Feldt-Rasmussen, U.; Main, K.M. Thyroid effects of endocrine disrupting chemicals. Mol. Cell. Endocrinol 2012, 355, 240–248. [Google Scholar]
- Hinther, A.; Vawda, S.; Skirrow, R.C.; Veldhoen, N.; Collins, P.; Cullen, J.T.; van Aggelen, G.; Helbing, C.C. Nanometals induce stress and alter thyroid hormone action in amphibia at or below North American water quality guidelines. Environ. Sci. Technol 2010, 44, 8314–8321. [Google Scholar]
- Zha, L.; Zeng, J.; Sun, S.; Deng, H.; Luo, H.; Li, W. Chromium(III) nanoparticles affect hormone and immune responses in heat-stressed rats. Biol. Trace Elem. Res 2009, 129, 157–169. [Google Scholar]
- Hatch, E.E.; Nelson, J.W.; Stahlhut, R.W.; Webster, T.F. Association of endocrine disruptors and obesity: Perspectives from epidemiological studies. Int. J. Androl 2010, 33, 324–332. [Google Scholar]
- Newbold, R.R. Impact of environmental endocrine disrupting chemicals on the development of obesity. Hormones 2010, 9, 206–217. [Google Scholar]
- Ben-Jonathan, N.; Hugo, E.R.; Brandebourg, T.D. Effects of bisphenol A on adipokine release from human adipose tissue: Implications for the metabolic syndrome. Mol. Cell. Endocrinol 2009, 304, 49–54. [Google Scholar]
- Gurevitch, D.; Shuster-Meiseles, T.; Nov, O.; Zick, Y.; Rudich, A.; Rudich, Y. TiO2 nanoparticles induce insulin resistance in liver-derived cells both directly and via macrophage activation. Nanotoxicology 2012, 6, 804–812. [Google Scholar]
- Pourkhalili, N.; Hosseini, A.; Nili-Ahmadabadi, A.; Rahimifard, M.; Navaei-Nigjeh, M.; Hassani, S.; Baeeri, M.; Abdollahi, M. Improvement of isolated rat pancreatic islets function by combination of cerium oxide nanoparticles/sodium selenite through reduction of oxidative stress. Toxicol. Mech. Methods 2012, 22, 476–482. [Google Scholar]
- Wang, M.Q.; Xu, Z.R.; Zha, L.Y.; Lindemann, M.D. Effects of chromium nanocomposite supplementation on blood metabolites, endocrine parameters and immune traits in finishing pigs. Anim. Feed. Sci. Technol 2007, 139, 69–80. [Google Scholar]
- Zha, L.Y.; Xu, Z.R.; Wang, M.Q.; Gu, L.Y. Effects of chromium nanoparticle dosage on growth, body composition, serum hormones and tissue chromium in Sprague-Dawley rats. J. Zhejiang Univ. Sci. B 2007, 8, 323–330. [Google Scholar]
- Crouzier, D.; Follot, S.; Gentilhomme, E.; Flahaut, E.; Arnaud, R.; Dabouis, V.; Castellarin, C.; Debouzy, J.C. Carbon nanotubes induce inflammation but decrease the production of reactive oxygen species in lung. Toxicology 2010, 272, 39–45. [Google Scholar] [Green Version]
- Pourkhalili, N.; Hosseini, A.; Nili-Ahmadabadi, A.; Hassani, S.; Pakzad, M.; Baeeri, M.; Mohammadirad, A.; Abdollahi, M. Biochemical and cellular evidence of the benefit of a combination of cerium oxide nanoparticles and selenium to diabetic rats. World J. Diabetes 2011, 2, 204–210. [Google Scholar]
- Corona-Morales, A.A.; Castell, A.; Escobar, A.; Drucker-Colín, R.; Zhang, L. Fullerene C60 and ascorbic acid protect cultured chromaffin cells against levodopa toxicity. J. Neurosci. Res 2003, 71, 121–126. [Google Scholar]
- Hussain, S.M.; Javorina, A.K.; Schrand, A.M.; Duhart, H.M.; Ali, S.F.; Schlager, J.J. The interaction of manganese nanoparticles with PC-12 cells induces dopamine depletion. Toxicol. Sci 2006, 92, 456–463. [Google Scholar]
- Wang, J.; Rahman, M.F.; Duhart, H.M.; Newport, G.D.; Patterson, T.A.; Murdock, R.C.; Hussain, S.M.; Schlager, J.J.; Ali, S.F. Expression changes of dopaminergic system-related genes in PC12 cells induced by manganese, silver, or copper nanoparticles. Neurotoxicology 2009, 30, 926–933. [Google Scholar]
- Love, S.A.; Haynes, C.L. Assessment of functional changes in nanoparticle-exposed neuroendocrine cells with amperometry: Exploring the generalizability of nanoparticle-vesicle matrix interactions. Anal. Bioanal. Chem 2010, 398, 677–688. [Google Scholar]
- Gosso, S.; Gavello, D.; Giachello, C.N.; Franchino, C.; Carbone, E.; Carabelli, V. The effect of CdSe-ZnS quantum dots on calcium currents and catecholamine secretion in mouse chromaffin cells. Biomaterials 2011, 32, 9040–9050. [Google Scholar]
- Powers, C.M.; Badireddy, A.R.; Ryde, I.T.; Seidler, F.J.; Slotkin, T.A. Silver nanoparticles compromise neurodevelopment in PC12 cells: Critical contributions of silver ion, particle size, coating, and composition. Environ. Health Perspect 2011, 119, 37–44. [Google Scholar]
- Wu, J.; Wang, C.; Sun, J.; Xue, Y. Neurotoxicity of silica nanoparticles: Brain localization and dopaminergic neurons damage pathways. ACS Nano 2011, 5, 4476–4489. [Google Scholar]
- Love, S.A.; Liu, Z.; Haynes, C.L. Examining changes in cellular communication in neuroendocrine cells after noble metal nanoparticle exposure. Analyst 2012, 137, 3004–3010. [Google Scholar]
- Wang, M.Q.; Xu, Z.R.; Li, W.F.; Jiang, Z.G. Effect of chromium nanocomposite supplementation on growth hormone pulsatile secretion and mRNA expression in finishing pigs. J. Anim. Physiol. Anim. Nutr 2009, 93, 520–525. [Google Scholar]
- Vincent, J.B. The biochemistry of chromium. J. Nutr 2001, 130, 715–718. [Google Scholar]
- Douglas, S.A.; Sreenivasan, D.; Carman, F.H.; Bunn, S.J. Cytokine interactions with adrenal medullary chromaffin cells. Cell. Mol. Neurobiol 2010, 30, 1467–1475. [Google Scholar]
- Guérineau, N.C.; Desarménien, M.G.; Carabelli, V.; Carbone, E. Functional chromaffin cell plasticity in response to stress: Focus on nicotinic, gap junction, and voltage-gated Ca(2+) channels. J. Mol. Neurosci 2012, 48, 368–386. [Google Scholar]
- Carabelli, V.; Marcantoni, A.; Comunanza, V.; de Luca, A.; Díaz, J.; Borges, R.; Carbone, E. Chronic hypoxia up-regulates alpha1H T-type channels and low-threshold catecholamine secretion in rat chromaffin cells. J. Physiol 2007, 584, 149–165. [Google Scholar]
- Pérez-Alvarez, A.; Hernández-Vivanco, A.; Albillos, A. Past, present and future of human chromaffin cells: Role in physiology and therapeutics. Cell. Mol. Neurobiol 2010, 30, 1407–1415. [Google Scholar]
- Gavello, D.; Vandael, D.H.; Cesa, R.; Premoselli, F.; Marcantoni, A.; Cesano, F.; Scarano, D.; Fubini, B.; Carbone, E.; Fenoglio, I.; Carabelli, V. Altered excitability of cultured chromaffin cells following exposure to multi-walled carbon nanotubes. Nanotoxicology 2012, 6, 47–60. [Google Scholar]
- Soin, T.; Verslycke, T.; Janssen, C.; Smagghe, G. Ecdysteroids and Their Importance in Endocrine Disruption Research. In Ecdysone: Structures and Functions; Smagghe, G., Ed.; Springer Science + Business Media B.V.: Dordrecht, The Netherlands, 2009; Volume Chapter 22, pp. 539–549. [Google Scholar]
- Wiench, K.; Wohlleben, W.; Hisgen, V.; Radke, K.; Salinas, E.; Zok, S.; Landsiedel, R. Acute and chronic effects of nano- and non-nano-scale TiO(2) and ZnO particles on mobility and reproduction of the freshwater invertebrate Daphnia magna. Chemosphere 2009, 76, 1356–1365. [Google Scholar]
- Zhu, X.; Chang, Y.; Chen, Y. Toxicity and bioaccumulation of TiO2 nanoparticle aggregates in Daphnia magna. Chemosphere 2010, 78, 209–215. [Google Scholar]
- Seitz, F.; Bundschuh, M.; Rosenfeldt, R.R.; Schulz, R. Nanoparticle toxicity in Daphnia magna reproduction studies: The importance of test design. Aquat. Toxicol 2013, 126, 163–168. [Google Scholar]
- Zhao, C.M.; Wang, W.X. Comparison of acute and chronic toxicity of silver nanoparticles and silver nitrate to Daphnia magna. Environ. Toxicol. Chem 2011, 30, 885–892. [Google Scholar]
- Oberdörster, E.; Zhu, S.; Blickley, T.M.; McClellan-Green, P.; Haasch, M.L. Ecotoxicology of carbon-based engineered nanoparticles: Effects of fullerene (C60) on aquatic organisms. Carbon 2006, 44, 1112–1120. [Google Scholar]
- Tao, X.; Fortner, J.D.; Zhang, B.; He, Y.; Chen, Y.; Hughes, J.B. Effects of aqueous stable fullerene nanocrystals (nC60) on Daphnia magna: Evaluation of sub-lethal reproductive responses and accumulation. Chemosphere 2009, 77, 1482–1487. [Google Scholar]
- Mendonça, E.; Diniz, M.; Silva, L.; Peres, I.; Castro, L.; Correia, J.B.; Picado, A. Effects of diamond nanoparticle exposure on the internal structure and reproduction of Daphnia magna. J. Hazard. Mater 2011, 186, 265–271. [Google Scholar]
- Kim, J.; Park, Y.; Yoon, T.H.; Yoon, C.S.; Choi, K. Phototoxicity of CdSe/ZnSe quantum dots with surface coatings of 3-mercaptopropionic acid or tri-n-octylphosphine oxide/gum arabic in Daphnia magna under environmentally relevant UV-B light. Aquat. Toxicol 2010, 97, 116–124. [Google Scholar]
- Fabrega, J.; Tantra, R.; Amer, A.; Stolpe, B.; Tomkins, J.; Fry, T.; Lead, J.R.; Tyler, C.R.; Galloway, T.S. Sequestration of zinc from zinc oxide nanoparticles and life cycle effects in the sediment dweller amphipod Corophium volutator. Environ. Sci. Technol 2012, 46, 1128–1135. [Google Scholar]
- Nair, P.M.; Park, S.Y.; Lee, S.W.; Choi, J. Differential expression of ribosomal protein gene, gonadotrophin releasing hormone gene and Balbiani ring protein gene in silver nanoparticles exposed Chironomus riparius. Aquat. Toxicol 2011, 101, 31–37. [Google Scholar]
- Nair, P.M.; Choi, J. Modulation in the mRNA expression of ecdysone receptor gene in aquatic midge, Chironomus riparius upon exposure to nonylphenol and silver nanoparticles. Environ. Toxicol. Pharmacol 2012, 33, 98–106. [Google Scholar]
- Vecchio, G.; Galeone, A.; Brunetti, V.; Maiorano, G.; Sabella, S.; Cingolani, R.; Pompa, P.P. Concentration-dependent, size-independent toxicity of citrate capped AuNPs in Drosophila melanogaster. PLoS One 2012, 7, e29980. [Google Scholar]
- Wang, H.; Wick, R.L.; Xing, B. Toxicity of nanoparticulate and bulk ZnO, Al2O3 and TiO2 to the nematode Caernorhabditis elegans. Environ. Poll 2009, 157, 1171–1177. [Google Scholar]
- Menard, A.; Drobne, D.; Jemec, A. Ecotoxicity of nanosized TiO2. Review of in vivo data. Environ. Pollut 2011, 159, 677–684. [Google Scholar]
- Roh, J.Y.; Park, Y.K.; Park, K.; Choi, J. Ecotoxicological investigation of CeO(2) and TiO(2) nanoparticles on the soil nematode Caenorhabditis elegans using gene expression, growth, fertility, and survival as endpoints. Environ. Toxicol. Pharmacol 2010, 29, 167–172. [Google Scholar]
- Roh, J.Y.; Sim, S.J.; Yi, J.; Park, K.; Chung, K.H.; Ryu, D.Y.; Choi, J. Ecotoxicity of silver nanoparticles on the soil nematode Caenorhabditis elegans using functional ecotoxicogenomics. Environ. Sci. Technol 2009, 43, 3933–3940. [Google Scholar]
- Ma, H.; Bertsch, P.M.; Glenn, T.C.; Kabengi, N.J.; Williams, P.L. Toxicity of manufactured zinc oxide nanoparticles in the nematode Caenorhabditis elegans. Environ. Toxicol. Chem 2009, 28, 1324–1330. [Google Scholar]
- Lim, D.; Roh, J.Y.; Eom, H.J.; Choi, J.Y.; Hyun, J.; Choi, J. Oxidative stress-related PMK-1 P38 MAPK activation as a mechanism for toxicity of silver nanoparticles to reproduction in the nematode Caenorhabditis elegans. Environ. Toxicol. Chem 2012, 31, 585–592. [Google Scholar]
- Qu, Y.; Li, W.; Zhou, Y.; Liu, X.; Zhang, L.; Wang, L.; Li, Y.F.; Iida, A.; Tang, Z.; Zhao, Y.; Chai, Z.; Chen, C. Full assessment of fate and physiological behavior of quantum dots utilizing Caenorhabditis elegans as a model organism. Nano Lett 2011, 11, 3174–3183. [Google Scholar]
- Cha, Y.J.; Lee, J.; Choi, S.S. Apoptosis-mediated in vivo toxicity of hydroxylated fullerene nanoparticles in soil nematode Caenorhabditis elegans. Chemosphere 2012, 87, 49–54. [Google Scholar]
- Pluskota, A.; Horzowski, E.; Bossinger, O.; von Mikecz, A. In Caenorhabditis elegans nanoparticle-bio-interactions become transparent: Silica-nanoparticles induce reproductive senescence. PLoS One 2009, 4, e6622. [Google Scholar]
- Unrine, J.M.; Hunyadi, S.E.; Tsyusko, O.V.; Rao, W.; Shoults-Wilson, W.A.; Bertsch, P.M. Evidence for bioavailability of Au nanoparticles from soil and biodistribution within earthworms (Eisenia fetida). Environ. Sci. Technol 2010, 44, 8308–8313. [Google Scholar]
- Schoults-Wilson, W.A.; Reinsch, B.C.; Tsyusko, O.V.; Bertsch, P.M.; Lowry, G.V.; Unrine, J.M. Role of particle size and soil type in toxicity of silver nanoparticles to earthworms. Soil Sci. Soc. Am. J 2011, 75, 365–377. [Google Scholar]
- Coleman, J.G.; Johnson, D.R.; Stanley, J.K.; Bednar, A.J.; Weiss, C.A., Jr; Boyd, R.E.; Steevens, J.A. Assessing the fate and effects of nano aluminum oxide in the terrestrial earthworm Eisenia fetida. Environ. Toxicol. Chem. 2010, 29, 1575–1580. [Google Scholar]
- Cañas, J.E.; Qi, B.; Li, S.; Maul, J.D.; Cox, S.B.; Das, S.; Green, M.J. Acute and reproductive toxicity of nano-sized metal oxides (ZnO and TiO2) to earthworms (Eisenia fetida). J. Environ. Monit 2011, 13, 3351–3357. [Google Scholar]
- Hooper, H.L.; Jurkschat, K.; Morgan, A.J.; Bailey, J.; Lawlor, A.J.; Spurgeon, D.J.; Svendsen, C. Comparative chronic toxicity of nanoparticulate and ionic zinc to the earthworm Eisenia veneta in a soil matrix. Environ. Int 2011, 37, 1111–1117. [Google Scholar]
- Unrine, J.M.; Tsyusko, O.V.; Hunyadi, S.E.; Judy, J.D.; Bertsch, P.M. Effects of particle size on chemical speciation and bioavailability of copper to earthworms (Eisenia fetida) exposed to copper nanoparticles. J. Environ. Qual 2010, 39, 1942–1953. [Google Scholar]
- Scott-Fordsmand, J.J.; Krogh, P.H.; Schaefer, M.; Johansen, A. The toxicity testing of double-walled nanotubes-contaminated food to Eisenia veneta earthworms. Ecotoxicol. Environ. Saf 2008, 71, 616–619. [Google Scholar]
- Li, D.; Alvarez, P.J. Avoidance, weight loss, and cocoon production assessment for Eisenia fetida exposed to C60 in soil. Environ. Toxicol. Chem 2011, 30, 2542–2545. [Google Scholar]
- Van der Ploeg, M.J.; Baveco, J.M.; van der Hout, A.; Bakker, R.; Rietjens, I.M.; van den Brink, N.W. Effects of C60 nanoparticle exposure on earthworms (Lumbricus rubellus) and implications for population dynamics. Environ. Pollut 2011, 159, 198–203. [Google Scholar]
- Pakarinen, K.; Petersen, E.J.; Leppänen, M.T.; Akkanen, J.; Kukkonen, J.V. Adverse effects of fullerenes (nC60) spiked to sediments on Lumbriculus variegatus (Oligochaeta). Environ. Pollut 2011, 159, 3750–3756. [Google Scholar]
- Lynch, I.; Dawson, K.A.; Linse, S. Detecting cryptic epitopes created by nanoparticles. Sci STKE 2006, 327. [Google Scholar] [CrossRef]
- Monopoli, M.P.; Aberg, C.; Salvati, A.; Dawson, K.A. Biomolecular coronas provide the biological identity of nanosized materials. Nat. Nanotechnol 2012, 7, 779–786. [Google Scholar]
- Walkey, C.D.; Chan, W.C. Understanding and controlling the interaction of nanomaterials with proteins in a physiological environment. Chem. Soc. Rev 2012, 41, 2780–2799. [Google Scholar]
- Monopoli, M.P.; Walczyk, D.; Campbell, A.; Elia, G.; Lynch, I.; Bombelli, F.B.; Dawson, K.A. Physical-chemical aspects of protein corona: Relevance to in vitro and in vivo biological impacts of nanoparticles. J. Am. Chem. Soc 2011, 133, 2525–2534. [Google Scholar]
- De Roe, C.; Courtoy, P.J.; Baudhuin, P. A model of protein-colloidal gold interactions. J. Histochem. Cytochem 1987, 35, 1191–1198. [Google Scholar]
- Casals, E.; Gonzalez, E.; Puntes, V.F. Reactivity of inorganic nanoparticles in biological environments: Insights into nanotoxicity mechanisms. J. Phys. D 2012, 45, 443001, :1–443001:15.. [Google Scholar]
- Auffan, M.; Rose, J.; Wiesner, M.R.; Bottero, J.Y. Chemical stability of metallic nanoparticles: A parameter controlling their potential cellular toxicity in vitro. Environ. Pollut 2009, 157, 1127–1133. [Google Scholar]
- Franklin, N.M.; Rogers, N.J.; Apte, S.C.; Batley, G.E.; Gadd, G.E.; Casey, P.S. Comparative toxicity of nanoparticulate ZnO, bulk ZnO, and ZnCl2 to a freshwater microalga (Pseudokirchneriella subcapitata): The importance of particle solubility. Environ. Sci. Technol 2007, 41, 8484–8490. [Google Scholar]
- Zheng, X.; Wu, R.; Chen, Y. Effects of ZnO nanoparticles on wastewater biological nitrogen and phosphorus removal. Environ. Sci. Technol 2011, 45, 2826–2832. [Google Scholar]
- Dibrov, P.; Dzioba, J.; Gosink, K.K.; Häse, C.C. Chemiosmotic mechanism of antimicrobial activity of Ag(+) in vibrio cholerae. Antimicrob. Agents Chemother 2002, 46, 2668–2670. [Google Scholar]
- Morones, J.R.; Elechiguerra, J.L.; Camacho, A.; Holt, K.; Kouri, J.B.; Ramírez, J.T.; Yacaman, M.J. The bactericidal effect of silver nanoparticles. Nanotechnology 2005, 16, 2346–2353. [Google Scholar]
- Nowack, B.; Krug, H.F.; Height, M. 120 years of nanosilver history: Implications for policy makers. Environ. Sci. Technol 2011, 45, 1177–1183. [Google Scholar]
- Derfus, A.M.; Chan, W.C.W.; Bhatia, S.N. Probing the cytotoxicity of semiconductor quantum dots. Nano Lett 2004, 4, 11–18. [Google Scholar]
- Chen, N.; He, Y.; Su, Y.; Li, X.; Huang, Q.; Wang, H.; Zhang, X.; Tai, R.; Fan, C. The cytotoxicity of cadmium-based quantum dots. Biomaterials 2012, 33, 1238–1244. [Google Scholar]
- Oberdörster, G. Safety assessment for nanotechnology and nanomedicine: Concepts of nanotoxicology. J. Intern. Med 2010, 267, 89–105. [Google Scholar]
- Iavicoli, I.; Leso, V.; Fontana, L.; Cottica, D.; Bergamaschi, A. Characterization of inhalable, thoracic, and respirable fractions and ultrafine particle exposure during grinding, brazing, and welding activities in a mechanical engineering factory. J. Occup. Environ. Med 2013, 55, 430–445. [Google Scholar]
- Nascarella, M.A.; Calabrese, E.J. A method to evaluate hormesis in nanoparticle dose-responses. Dose Response 2012, 10, 344–354. [Google Scholar]

| Type of category | Category 1 | Category 2 | Category 3a | Category 3b | References |
| Criteria of classification | At least one in vivo study providing clear evidence of endocrine disruption in an intact organism | Potential for endocrine disruption. In vitro data indicating potential for endocrine disruption in intact organisms. Also includes effects in vivo that may, or may not be endocrine disruption-mediated. | No scientific basis for inclusion in list. Endocrine disruption studies available but no indications of endocrine disruption effects. | Substances with no or insufficient data gathered | |
| Number of chemical substances in each category | 194 | 125 | 23 | 86 | [15–18] |
| Type of study | Type and physico-chemical properties of NPs | Experimental protocol | Cell line/laboratory animals | Results | References |
|---|---|---|---|---|---|
| Alterations and impairment of testicular structure | |||||
| In vitro | TiO2-NPs (25–70 nm); CB-NPs (14 nm); DE-NPs | Treatment with 0–1000 μg/mL for 24 and 48 h | Mouse testis Leydig cell line TM3 |
| [30] |
| In vitro | 75% rutile and 25% anatase TiO2-NPs (24.5 nm) | Treatment with 0.5–50 μg/mL for 4, 24 and 48 h | RTG-2 cells |
| [31] |
| In vitro | CeO2-NPs (10 nm and 20–25 nm) | Treatment with 0–100 μg/mL for 24 and 72 h | RTG-2 cells, derived from rainbow trout (Oncorhynchus mykiss) gonadal tissue |
| [32] |
| In vivo | NRDE-NPs—Average of mode diameter and number concentrations for low, middle and high doses were 22.48, 26.13 and 27.06 nm; 2.27 × 105, 5.11 × 105 and 1.36 × 106, respectively | Inhalation exposure to low (15.37), middle (36.35) and high (168.84) μg/m3 doses for 4, 8, or 12 weeks (5 h/day, 5 days/week) | Fischer F344 rats |
| [33] |
| In vivo | NRDE-NPs—Average of mode diameter and number concentrations were 26.81 nm and 1.83 × 106, respectively | Inhalation exposure to 148.86 μg/m3 for 5 h daily for 19 gestational days | Pregnant Fischer F344 rats |
| [34] |
| In vivo | TiO2-NPs (anatase form, particle size of 25–70 nm, surface area 20–25 m2/g) | Subcutaneous injections of 100 μL of 1mg/mL solution on 4 gestational days | Pregnant Slc:ICR mice |
| [35] |
| In vivo | CB-NPs (14, 56 and 95 nm with a surface area of 300, 45 and 20 m2/g, respectively) | Intratracheal administration of 0.1 mg/kg body weight for 10 times every week | ICR mice |
| [36] |
| In vivo | Carboxylate-functionalized (diameter of 20–30 nm, length 0.5–2.0 μm) and Amine-functionalized (diameter of 20–30 nm, length 0.5–2.0 μm) MWCNTs | Mice were randomly divided into 12 groups (15-day single dose, 15-day multi-dose, 60-day multi-dose, and 90-day multi-dose groups). Mice were given injections (5 mg/kg) via the tail vein once (single dose) or every 3 days for 5 times | BALB/c mice |
| [37] |
| In vivo | CB-NPs (14 nm) | Intratracheal administration of 0.2 mg/kg body weight on days 7 and 14 of gestation | Pregnant ICR mice |
| [38] |
| In vivo | mPEG@Au-NP and PEG-NH2@Au-NP (14 nm) | Intravenous injection of 45 and 225 mg/kg of mPEG@Au-NP and 45 mg/kg of PEG-NH2@ Au-NP at 48 h intervals for 5 days | ICR mice |
| [39] |
| In vivo | Amorphous silica particles (nSP70, 70-nm diameter; nSP300, 300-nm diameter) | Intravenous injection of 0.4 and 0.8 mg of nSP70 | BALB/c mice |
| [40] |
| Alterations and impairment of spermatogenesis | |||||
| In vitro | Fullerenol | Treatment with fullerenol (1, 10 and 100 μmol) for 3 h. | Epididymal sperm samples collected from the fresh epididymis of adult goats |
| [41] |
| In vitro | Silver (15 nm), molybdenum (30 nm), and aluminum (30 nm) | Treatment with 5, 10, 25, 50, and 100 μg/mL culture medium for 48 h | C18-4 spermatogonial stem cell line |
| [42] |
| In vitro | Magnetic iron oxide NPs (Fe3O4-NPs) coated with poly(vinyl alcohol) | Treatment with 7.35 mM for 80 min and 4 h | Bovine sperm cells |
| [43] |
| In vitro | Eu2O3-NPs (30 ± 10 nm); EuOH3-NPs conjugated with polyvinyl alcohol or polyvinyl piyrolidone (15.4 ± 3 nm and 9.3 ± 3 nm, respectively) | 1 mL of washed sperm cells was incubated for 24 h at 39 °C with 2.5 mg/mL of Eu2O3-NPs or of EuOH3-NPs | Bovine sperm cells |
| [44] |
| In vitro | TiO2-NPs and ZnO-NPs (40–70 nm) | Treatment with 3.73–59.7 μg/mL of TiO2-NPs and 11.5–93.2 μg/mL of ZnO-NPs | Human spermatozoa |
| [45] |
| In vitro | Au-NPs (9 nm) | Analyses were conducted on a mixture of 500 μL of Au-NPs (44 ppm) solution and semen | Human spermatozoa |
| [46] |
| In vitro | Hydrocarbon-coated silver (Ag-HC) nanoparticles of 15, 25, and 80 nm diameters and Polysaccharide-coated silver (Ag-PS) nanoparticles of 10, 25–30, and 80 nm diameters | Treatment with 5, 10, 25, 50, and 100 μg/mL culture medium for 24 h | C18-4 spermatogonial stem cell line |
| [47] |
| In vitro | Au-NPs (5–65 nm) | Treatment with 0.5–50 μM for 2 h | Bovine spermatozoa |
| [48] |
| In vitro | Au-NPs (~2.5 nm in diameter) | Treatment with concentrations of 0.5 × 1015 or 1 × 1015 particles/mL for 20 and 40 min | Epididymal sperm samples collected from the epididymis of hybrid mice CBA × C57B1/6 |
| [49] |
| In vitro | Ag-NPs (65 nm) | Aliquots of total semen were incubated at 37 °C for 60 min and 120 min at the concentration of 125, 250 and 500 μM | Semen samples obtained from 10 healthy donors |
| [50] |
| In vitro | Ag-NPs (20 nm) and TiO2-NPs (21 nm) | Cells were exposed for 24, 48 and 72 h to 10, 50 and 100 μg/mL, equivalent to 7.8, 15.6 and 31 μg/cm2, respectively | Ntera2 (NT2, human testicular embryonic carcinoma cell line) and primary testicular cells from C57BL6 mice of wild type (WT) and 8- oxoguanine DNA glycosylase knock-out (KO, mOgg1−/−) genotype |
| [51] |
| In vivo | TiO2-NPs | Intraperitoneal injection of 200 and 500 μg/kg every other day for five times | ICR mice |
| [52] |
| In vivo | TiO2-NPs (anatase form, particle size of 25–70 nm, surface area 20–25 m2/g) | Subcutaneous injections of 100 μL of 1 mg/mL solution | Pregnant Slc: ICR mice |
| [35] |
| In vivo | CB-NPs (14, 56 and 95 nm with a surface area of 300, 45 and 20 m2/g, respectively) | Intratracheal administration of 0.1 mg/kg body weight every week for 10 times | ICR mice |
| [36] |
| In vivo | Carboxylate-functionalized (diameter of 20–30 nm, length 0.5–2.0 μm) and Amine-functionalized (diameter of 20–30 nm, length 0.5–2.0 μm) MWCNTs | Mice were randomly divided into 12 groups (15-day single dose, 15-day multi-dose, 60-day multi-dose, and 90-day multi-dose groups). Mice were given injections (5 mg/kg) via the tail vein once (single dose) or every 3 days for 5 times | BALB/c mice |
| [37] |
| In vivo | CB-NPs (14 nm) | Intratracheal administration of 0.2 mg/kg body weight of CB-NPs on days 7 and 14 of gestation | Pregnant female ICR mice |
| [38] |
| In vivo | C60 fullerene (3–36 nm) | Rats received distilled water containing aqueous solutions of C60HyFn at concentration of 4~μg/kg daily for 5 weeks | Healthy and streptozotocin-induced diabetic male Wistar albino rats |
| [53] |
| In vivo | Dimercaptosuccinic acid coated Fe3O4-NPs (3–9 nm) | Intraperitoneal injection in a single dose of 50, 100, 200 and 300 mg/kg | Female pregnant Balb/C mice |
| [54] |
| In vivo | TiO2-NPs (33.2 ± 16.7 nm) | Administration by oral gavage of 40, 200 and 1000 mg/kg with volume of suspension 10 mL/kg of mouse weight | Male CBAxB6 mice |
| [55] |
| In vivo | Ag-NPs with a nominal diameter of 20 ± 5 nm | Intravenous injection with a single dose (5 mg/kg or 10 mg/kg) of Ag-NPs | Wistar rats |
| [56] |
| In vivo | mPEG@Au-NP and PEG-NH2@Au-NP (14 nm) | Intravenous injection of 45 and 225 mg/kg of mPEG@Au-NP and 45 mg/kg of PEG-NH2@Au-NP at 48 h intervals for 5 days | ICR mice |
| [39] |
| In vivo | Nude short MWCNTs (50–200 nm) and synthesized functionalized MWCNTs with polyethylene glycol (PEG) (s-MWCNTs-PEG). | Intravenous administration with a single dose of 100 μg/kg body weight | Kunming mice |
| [57] |
| In vivo | Powder of nanoparticulate TiO2 (primary particle size was 20.6 nm, in the exposure atmosphere the particle number concentration was 1.7 × 106 particles/cm3 and the major particle size mode was 97 nm); nanosized CB (particle size was 14 nm) | Mice were exposed by whole body inhalation, 1 h/day from gestation day 8 to 18, to 42 mg/m3 of aerosolized powder of nanoparticulate TiO2; mice were intratracheally instilled four times during gestation, (days 7, 10, 15 and 18) with 67 μg/animal of nanosized CB | Pregnant female C57BL/6J mice |
| [58] |
| Disruption of normal levels of sex hormones | |||||
| In vitro | TiO2-NPs (25–70 nm); CB-NPs (14 nm); DE-NPs | Treatment with 0–1000 μg/mL for 24 and 48 h | Mouse testis Leydig cell line TM3 |
| [30] |
| In vitro | NRDE-NPs | Mice were exposed to 152.01 μg/m3 of NRDE-NPs for 8 weeks | Interstitial testicular cells, dissected from male C57BL/Jcl mice |
| [59] |
| In vivo | NRDE-NPs—Averages of mode diameter and number concentrations for low, middle and high doses were 22.48, 26.13 and 27.06 nm; 2.27 × 105, 5.11 × 105 and 1.36 × 106, respectively | Inhalation exposure to low (15.37), middle (36.35) and high (168.84) μg/m3 doses for 4, 8, or 12 weeks (5 h/day, 5 days/week) | Fischer F344 rats |
| [33] |
| In vivo | NRDE-NPs—Averages of mode diameter and number concentrations were 26.81 nm and 1.83 × 106, respectively | Inhalation exposure to 148.86 μg/m3 for 5 h daily for 19 gestational days | Pregnant Fischer F344 rats and male offspring |
| [34] |
| In vivo | NRDE-NPs—Averages of mode diameter and number concentrations for low, middle and high doses were 22.48, 26.13 and 27.06 nm; 2.27 × 105, 5.11 × 105 and 1.36 × 106, respectively | Inhalation exposure to low (15.37), middle (36.35) and high (168.84) μg/m3 doses for 4, 8, or 12 weeks (5 h/day, 5 days/week) | Fischer F344 rats |
| [60] |
| In vivo | CB-NPs (14, 56 and 95 nm with a surface area of 300, 45 and 20 m2/g, respectively) | Intratracheal administration of 0.1 mg/kg body weight for 10 times every week | ICR mice |
| [36] |
| In vivo | Carboxylate-functionalized (diameter of 20–30 nm, length 0.5–2.0 μm) and Amine-functionalized (diameter of 20–30 nm, length 0.5–2.0 μm) MWCNTs | Mice were randomly divided into 12 groups (15-day single dose, 15-day multi-dose, 60-day multi-dose, and 90-day multi-dose groups). Mice were given injections (5 mg/kg) via the tail vein once (single dose) or every 3 days for 5 times | BALB/c mice |
| [37] |
| In vivo | CB-NPs (14 nm) | Intratracheal administration of 200 μg of CB-NPs on days 7 and 14 of gestation | Pregnant female ICR mice |
| [38] |
| In vivo | NRDE-NPs | Mice were exposed to 152.01 μg/m3 of NRDE-NPs for 8 weeks | C57BL/Jcl mice |
| [59] |
| In vivo | mPEG@Au-NP and PEG-NH2@Au-NP (14 nm) | Intravenous injection of 45 and 225 mg/kg of mPEG@Au-NP and 45 mg/kg of PEG-NH2@ Au-NP at 48 h intervals for 5 days | ICR mice |
| [39] |
| In vivo | NRDE-NPs | Inhalation exposure to low (38 ± 3 μg/m3) and high (149 ± 8 μg/m3) NRDE-NP concentrations | Fischer F344 rats |
| [61] |
| Type of study | Type and physico-chemical properties of NPs | Experimental protocol | Cell line/laboratory animals | Results | References |
|---|---|---|---|---|---|
| Effects on ovarian cells | |||||
| In vitro | Anatase TiO2-NPs (30 nm) | Treatment with increasing concentrations of TiO2-NPs (0–100 μg/mL) for 24 h | CHO-K1 |
| [68] |
| In vitro | TiO2-NPs (anatase-80% rutile-20% with organic coating, ~21 nm; anatase-80% rutile-20% doped with di-iron trioxide, ~21 nm; anatase-80% rutile-20%, ~21 nm; rutile 100% with inorganic and organic coating, 14 nm; anatase 100% with inorganic coating, 60 nm; rutile 100% with inorganic and organic coating, 20 nm; rutile 100% with inorganic and organic coating, 15 nm; rutile 100% with inorganic coating, 20–22 nm) | Exposure for 3 h to different concentrations of TiO2-NPs (800, 1950, 3000, 5000 μg/mL) | CHO-WBL |
| [69] |
| In vitro | TiO2-NPs (rutile-79% anatase-21%, particle size: 140 ± 44 nm, surface area: 38.5 m2/g) | Cytogenetic evaluations were conducted exposing cells to 750, 1250, and 2500 μg/mL for the 4 h non-activated test condition, to 62.5, 125, and 250 μg/mL, for the 4 h activated test condition, and to 25, 50, and 100 μg/mL for the 20 h non-activated test condition. | CHO |
| [70] |
| In vitro | Anatase TiO2-NPs (10–20, 50–60 nm) and rutile TiO2-NPs (50–60 nm) | Treatment with increasing concentrations of TiO2-NPs (25–325 μg/mL) for 24 h | CHO |
| [71] |
| In vitro | TiO2-NPs (shape: complex; average particle sizes: 20 ± 7 nm; specific surface area: 142 m2/g) and Al2O3-NPs (shape: spherical; average particle sizes: 28 ± 19 nm; specific surface area: 39 m2/g) | Treatment with increasing concentrations of NPs (0.5, 1, 5, 10, 25, 50 and 100 μg/mL) for 24 h | CHO-K1 |
| [72] |
| In vitro | SiCNW (diameter of 80 nm; chemical composition: Si to C ratio close to 1:1) | Exposure to different concentrations (0.5, 1.0, 5.0 and 10.0 μg/mL) of SiCNWs for 1, 3, and 5 days | CHO |
| [73] |
| In vitro | Calcium phosphate-NPs (20–30 nm, less than 10% particles were greater than 100 nm) | Cells were divided into different groups of exposure: control (treated with 4-androstene-3,17-dione), group II (treated with 10 μM of Calcium phosphate-NPs and 4-androstene-3, 17-dione) and group III (treated with 100 μM of Calcium phosphate-NPs and 4-androstene-3, 17-dione) | Granulosa cells collected from infertilite women |
| [74] |
| In vitro | MWCNTs (diameter ~10 nm) | In order to observe if the MWNT sheets have any toxic effect on cells, CHO cells were allowed to grow on the substrates until about 90% confluence | CHO |
| [75] |
| In vitro | Anatase TiO2-NPs (<25 nm) | Cells were maintained under exponential growth conditions and were continuously exposed for 1, 2, or 60 days to either 0, 10, 20, 40, 100 and 200 μg/mL of NPs | CHO-K1 |
| [76] |
| In vitro | Anatase TiO2-NPs (<25 nm) | Treatment with different concentrations of NPs (0, 25, 50, 100 and 200 μg/mL). Cells were incubated for 24 h and 48 h. | CHO-K1 |
| [77] |
| In vitro | Naked mesoporous silica NPs (10 nm and 50 nm); carboxyl or amine modified mesoporous silica NPs; carboxyl-modified polystyrene-NPs (30 nm); polystyrene-NPs functionalized with amine groups (50 nm) | Exposure of NIH-OVCAR3 for 48 h to 30 and 75 μg/mL of naked mesoporous silica NPs (10 nm); exposure of NIH-OVCAR3 and SKOV3 cells for 1 and 24 h to 20 μg/mL of naked mesoporous silica NPs (50 nm) and of carboxyl or amine modified mesoporous silica NPs; exposure of NIH-OVCAR3 and SKOV3 cells for 24 and 48 h to 75 μg/mL of carboxyl-modified polystyrene-NPs and polystyrene-NPs functionalized with amine groups | Ovarian NIHOVCAR3 epithelial cancer cells; SKOV3 cancer cells |
| [78] |
| In vitro | MWCNTs (average diameter: 67 nm; surface area: 26 m2/g; carbon purity: 99.79 wt%) | Exposure for 24 h to 1, 10 and 100 μg/mL of NPs | MARCO-transfected CHO-K1 cells |
| [79] |
| Effects on oogenesis and follicle maturation | |||||
| In vitro | TiO2-NPs (25 nm) | Exposure to increasing concentrations (12.5–50 μg/mL) of TiO2-NPs | Rat preantral follicles |
| [80] |
| In vitro | CdSe-core-QDs and ZnS-coated CdSe QDs (3.5 nm) | Exposure for 24 h to 0, 125, 250 and 500 nM of CdSe-core-QDs and to 500 nM of ZnS-coated CdSe QDs | Cumulus-oocyte complexes collected from female ICR mice |
| [81] |
| In vitro | Lysine coated CdSe/CdS/ZnS QDs (~20 nm) | Exposure for 4, 8, 16 and 24 h to 5.78 nmol/L and 29.8 nmol/L of Lysine coated CdSe/CdS/ZnS QDs | Immature oocytes of 28 days Kunming mice |
| [82] |
| In vitro | CdSe/CdS/ZnS QDs | Exposure for 4, 8 and 20 h to 28.9 nmol/L of QDs | Immature oocytes were collected from Kunming mice |
| [83] |
| In vitro | CdTe/ZnTe QD-Transferrin bioconjugates | Preantral follicles were treated with escalating concentrations of (0.0289, 0.289, 2.89 and 28.9 nmol/L) CdTe/ZnTe QD-Transferrin bioconjugates for 8 days | Pre antral follicles were collected from female Kunming mice |
| [84] |
| In vivo | TiO2-NPs (anatase; NPs were found to aggregate in culture medium and consequently the mean sizes were 240–280 nm (0.1 mg/L) and 259–360 nm (1.0 mg/L) | Reproductively active fish were exposed for 13 weeks to 0.1 and 1.0 mg/L | Zebrafish Danio rerio |
| [85] |
| In vivo | TiO2-NPs (anatase, average particle size: 6 nm, surface area: 174.8 m2/g) | TiO2-NPs suspensions at different concentrations (2.5, 5, and 10 mg/kg of body weight) were administered to mice by intragastric administration for 90 consecutive days. | CD-1 (ICR) female mice |
| [86] |
| In vivo | Ag-NPs (3 and 35 nm) | Exposure to 10 μg/L of Ag-NPs for 35 days | Sheepshead minnows (Cyprinodon Variegatus) |
| [87] |
| In vivo | TiO2-NPs (anatase-75% rutile-25%, average primary particle size: 21 nm, specific surface area: 50 ± 15 m2/g) | Reproductively active fish were exposed for 14 days to 0.1 and 1.0 mg/L | Zebrafish Danio rerio |
| [88] |
| Disruption of normal levels of sex hormones | |||||
| In vitro | Au-NPs (10 nm) | Exposure for 1,3,5 and 24 h to 2.85 × 1010 NPs/mL | Rat granulosa cells |
| [89] |
| In vitro | Calcium phosphate-NPs (20–30 nm, less than 10% particles were greater than 100 nm) | Cells were divided into different groups of exposure: control (treated with 4-androstene-3,17-dione), group II (treated with 10 μM of Calcium phosphate-NPs and 4-androstene-3, 17-dione) and group III (treated with 100 μM of Calcium phosphate-NPs and 4-androstene-3, 17-dione) for 48 h | Granulosa cells collected from infertile women |
| [74] |
| In vitro | CdTe/ZnTe QD-Transferrin bioconjugates | Preantral follicles were treated with escalating concentrations of (0.0289, 0.289, 2.89 and 28.9 nmol/L) CdTe/ZnTe QD-Transferrin bioconjugates for 8 days | Pre antral follicles were collected from female Kunming mice |
| [84] |
| In vivo | TiO2-NPs (anatase, average particle size: 6 nm, surface area: 174.8 m2/g) | TiO2-NP suspensions at different concentrations (2.5, 5, and 10 mg/kg of body weight) were administered to mice by intragastric administration for 90 consecutive days. | CD-1 (ICR) female mice |
| [86] |
| In vivo | NRDE-NPs (22–27 nm; particle composition showed a higher percentage of organic carbon than elemental carbon) | Inhalation exposure, for 5 h daily from day 1 to 19 of gestation, to 148.86 μg/m3 | Pregnant Fischer 344 rats |
| [90] |
| In vivo | ZnO-NPs (20–30 nm) | Oral administration of 333.33 mg/kg of ZnO-NPs | Wistar rats |
| [91] |
| Type of study | Type and physico-chemical properties of NPs | Experimental protocol | Cell line/laboratory animals | Results | References |
|---|---|---|---|---|---|
| In vitro | CdTe-QDs (~3 nm) | Treatment with 0.5 and 10 μg/mL for time periods ranging from 5 min to 96 h | Human breast cancer MCF-7 cells |
| [99] |
| In vivo | CdS-QDs (4.2 ± 1 nm) | Exposure for 21 days to 5, 50 and 500 μg/L of CdS-QDs | Male sticklebacks (Gasterosteus aculeatus) |
| [95] |
| In vivo | NRDE-NPs—Averages of mode diameter and number concentrations for low, middle and high doses were 22.48, 26.13 and 27.06 nm; 2.27 × 105, 5.11 × 105 and 1.36 × 106, respectively | Inhalation exposure to low (15.37), middle (36.35) and high (168.84) μg/m3 doses for 4, 8, or 12 weeks (5 hours/day, 5 days/week) | Fischer F344 rats |
| [33] |
| In vivo | NRDE-NPs—Averages of mode diameter and number concentrations were 26.81 nm and 1.83 × 106, respectively | Inhalation exposure to 148.86 μg/m3 for 5 h daily for 19 gestational days | Pregnant Fischer F344 rats |
| [34] |
| In vivo | (CdS)/CdTe capped-QDs | Exposure for 48 h to increasing concentrations of (CdS)/CdTe capped-QDs (1, 2 and 6 μg/L) | Juvenile rainbow trout (Oncorhynchus mykiss) |
| [94] |
| In vivo | C60 fullerene | Male zebrafish (Danio rerio) were fed for 5 days with brine shrimp preparations that had accumulated a mixture of C60 fullerene (10% v/v of the 600 mg C60/900 mL water) and 1 μg/L of 17α-ethinylestradiol or C60 fullerene or 17α-ethinylestradiol alone. | Male zebrafish (Danio rerio) |
| [97] |
| In vivo | CB-NPs (50–60 nm) | Intratracheal instillation at a total dose of 11, 54 and 268 μg/animal during gestation (7, 10, 15 and 18 gestation days) | C57BL/6 mice |
| [100] |
| In vivo | C60 fullerene | Exposure of male zebrafish (Danio rerio) to increasing concentrations of C60 fullerene. See Park et al. (2010) | Male zebrafish (Danio rerio) |
| [98] |
| In vivo | Ag-NPs (20 nm) | Exposure to increasing concentrations of Ag-NPs (0.06, 0.6 and 6 μg/L) for 96 h | Juvenile rainbow trout (Oncorhynchus mykiss) |
| [93] |
| In vivo | NRDE-NPs Low dose group (average of mode diameter: 22.78 ± 0.39 nm; mass concentration: 41.73 ± 0.58 μg/m3; number concentration: 8.21 × 105 ± 3.1 × 105), High dose group (average of mode diameter: 26.31 ± 0.38 nm; mass concentration: 152.01 ± 1.18 μg/m3; number concentration: 1.8 × 106 ± 5.18 × 105) | Exposure for 8 weeks (5 h/day, 5 days/week) to 41.73 μg/m3 and 152.01 μg/m3 of NRDE-NPs | Male C57BL/Jcl mice |
| [96] |
| In vivo | Ag-NPs (23.5 ± 4.4 nm) | Exposure to 1 and 25 μg/L of Ag-NPs for 28 days | Male Medaka (Oryzias latipes) fish |
| [92] |
| Type of study | Type and physico-chemical properties of NPs | Experimental protocol | Cell line/laboratory animals | Results | References |
|---|---|---|---|---|---|
| Effects on thyroid function | |||||
| In vitro | Ag-NPs (2–6 and 10 nm), ZnO-NPs (2–10 and 9 nm) and QDs composed of cadmium telluride (2–10 and 10–15 nm) | Exposure of cultured tail fin biopsy to 0.06 μg/L–5.5 mg/L of Ag-NPs, 0.19–10 mg/L of ZnO-NPs and 0.25 μg/L–22 mg/L of QDs for 48 h | Cultured tail fin biopsy assay derived from Rana catesbeiana tadpoles |
| [105] |
| In vivo | Cr-NPs (40–70 nm) | Oral administration of 150, 300 and 450 μg/Kg of Cr-NPs for 8 weeks | Male Sprague Dawley rats |
| [106] |
| Effects on insulin action and metabolism | |||||
| In vitro | TiO2-NPs (21 nm) | Exposure for 2 h to 50 and 200 μg/mL of TiO2-NPs | Fao rat hepatoma cells |
| [110] |
| In vitro | CeO2-NPs (100 nm) | Exposure to 100 nmol/L of CeO2-NPs, alone or in combination with 30 nmol/L of sodium selenite for 1–6 days | Pancreatic islets |
| [111] |
| In vivo | CrCl3-NPs (40–70 nm) | Dietary supplementation for 35 days with 200 μg/kg of CrCl3-NPs | Crossbred pigs (Duroc X Landrace X Yorkshire) |
| [112] |
| In vivo | Cr-NPs (40–50 nm) | Oral administration for 6 weeks of 75, 150, 300, 450, 600 and 1200 ppb of Cr-NPs | Male Sprague Dawley rats |
| [113] |
| In vivo | Cr-NPs (40–70 nm) | Oral administration of 150, 300 and 450 μg/Kg of Cr-NPs for 8 weeks | Male Sprague Dawley rats |
| [106] |
| In vivo | Double walled-CNTs (0.5–2.5 nm for inner tubes and 1.2–3.2 nm for outer tubes) | Intranasal instillation of 1.5 mg/kg | Male Swiss mice |
| [114] |
| In vivo | CeO2-NPs | Intraperitoneal injection of 60 mg/Kg of CeO2-NPs for 2 weeks, alone or in combination with sodium selenite (5 μmol/kg/day) | Male Wistar rats |
| [115] |
| Effects on neuroendocrine system | |||||
| In vitro | C60 fullerene | Exposure for 4 h to 100 μM of C60 fullerene | Adrenal chromaffin cells obtained from Wistar rats |
| [116] |
| In vitro | MnO-NPs (40 nm) and Ag-NPs (15 nm) | Exposure for 24 h to increasing concentrations (1–100 μg/mL ) of NPs | PC-12 cells derived from Rattus norvegicus pheochromocytoma (CRL-1721) |
| [117] |
| In vitro | Cu-NPs (90.9 ± 19.3 nm), Mn-NPs (52.1 ± 23.8 nm) and Ag-NPs (18.3 ± 7.3 nm) | Treatment for 24 h with 10 μg/mL of NPs and with increasing concentrations of Cu-NPs (2.5, 5, 7.5, 10 and 25 μg/mL) | PC-12 cells |
| [118] |
| In vitro | Au-NPs (28 nm) and Ag-NPs (61 nm) | Treatment for 24 and 48 h with 0.01–1 nM of NPs | Primary culture murine adrenal medullary chormaffin cells harvested from wild-type brown male mice (C57BL/6J) |
| [119] |
| In vitro | Carboxyl QDs with CdSe core and ZnS shell (7–8 nm) | Exposure for 24 h to increasing concentrations of QDs (5, 8, 16 and 36 nM) | Mouse chromaffin cells obtained from young C57BL/6J male mice |
| [120] |
| In vitro | Ag-NPs coated with citrate (6 nm) or polyvinylpyrrolidone (21 nm) | Exposure for 24 h to different concentrations (1–30 μM) of Ag-NPs | PC-12 cells |
| [121] |
| In vitro | Silica NPs (15 nm) | Exposure for 24 h to 25–200 μg/mL of silica NPs | PC-12 cells |
| [122] |
| In vitro | Citrated-capped Ag-NPs (from 15 to 60 nm); PEG (25.6 ± 7.8 nm) and heparin (20.6 ± 15.3 nm) surface modified Au NPs. | Exposure for 24 h to citrated-capped Ag-NPs at 1 nM concentration and to PEG and heparin Au NPs at 10 μg/mL | Primary culture murine adrenal medullary chormaffin cells harvested from wild-type brown male mice (C57BL/6J) |
| [123] |
| Effects on pituitary gland | |||||
| In vivo | CrCl3-NPs (40–70 nm) | Dietary supplementation for 35 days with 200 μg/kg of CrCl3-NPs | Crossbred pigs (Duroc X Landrace X Yorkshire) |
| [112] |
| In vivo | CrCl3-NPs (40–70 nm) | Dietary supplementation for 35 days with 200 μg/kg of CrCl3-NPs | Crossbred pigs (Duroc X Landrace X Yorkshire) |
| [124] |
| In vivo | Cr-NPs (40–70 nm) | Oral administration of 150, 300 and 450 μg/Kg of Cr-NPs for 8 weeks | Male Sprague Dawley rats |
| [106] |
| Effects on adrenal gland | |||||
| In vivo | NRDE-NPs—Averages of mode diameter and number concentrations for low, middle and high doses were 22.48, 26.13 and 27.06 nm; 2.27 × 105, 5.11 × 105 and 1.36 × 106, respectively | Inhalation exposure to low (15.37), middle (36.35) and high (168.84) μg/m3 doses for 4, 8, or 12 weeks (5 h/day, 5 days/week) | Fischer F344 rats |
| [33] |
| In vivo | NRDE-NPs—Averages of mode diameter and number concentrations were 26.81 nm and 1.83 × 106, respectively | Inhalation exposure to 148.86 μg/m3for 5 h daily for 19 gestational days | Pregnant Fischer F344 rats |
| [34] |
| In vivo | Cr-NPs (40–70 nm) | Oral administration of 150, 300 and 450 μg/Kg of Cr-NPs for 8 weeks | Male Sprague Dawley rats |
| [106] |
| In vivo | NRDE-NPs Low dose group (average of mode diameter: 22.78 ± 0.39 nm; mass concentration: 41.73 ± 0.58 μg/m3; number concentration: 8.21 × 105 ± 3.1 × 105), High dose group (average of mode diameter: 26.31 ± 0.38 nm; mass concentration: 152.01 ± 1.18 μg/m3; number concentration: 1.8 × 106 ± 5.18 × 105) | Exposure for 8 weeks (5 h/day, 5 days/week) to 41.73 μg/m3 and 152.01 μg/m3 of NRDE-NPs | Male C57BL/Jcl mice |
| [96] |
| In vivo | NRDE-NPs (22–27 nm; particle composition showed a higher percentage of organic carbon than elemental carbon) | Inhalation exposure to 148.86 μg/m3 for 5 h daily from day 1 to 19 of gestation | Pregnant Fischer 344 rats |
| [90] |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Iavicoli, I.; Fontana, L.; Leso, V.; Bergamaschi, A. The Effects of Nanomaterials as Endocrine Disruptors. Int. J. Mol. Sci. 2013, 14, 16732-16801. https://doi.org/10.3390/ijms140816732
Iavicoli I, Fontana L, Leso V, Bergamaschi A. The Effects of Nanomaterials as Endocrine Disruptors. International Journal of Molecular Sciences. 2013; 14(8):16732-16801. https://doi.org/10.3390/ijms140816732
Chicago/Turabian StyleIavicoli, Ivo, Luca Fontana, Veruscka Leso, and Antonio Bergamaschi. 2013. "The Effects of Nanomaterials as Endocrine Disruptors" International Journal of Molecular Sciences 14, no. 8: 16732-16801. https://doi.org/10.3390/ijms140816732
APA StyleIavicoli, I., Fontana, L., Leso, V., & Bergamaschi, A. (2013). The Effects of Nanomaterials as Endocrine Disruptors. International Journal of Molecular Sciences, 14(8), 16732-16801. https://doi.org/10.3390/ijms140816732




