Phyto-SERM Constitutes from Flemingia macrophylla
Abstract
:1. Introduction
2. Results and Discussion
2.1. Structure Elucidation
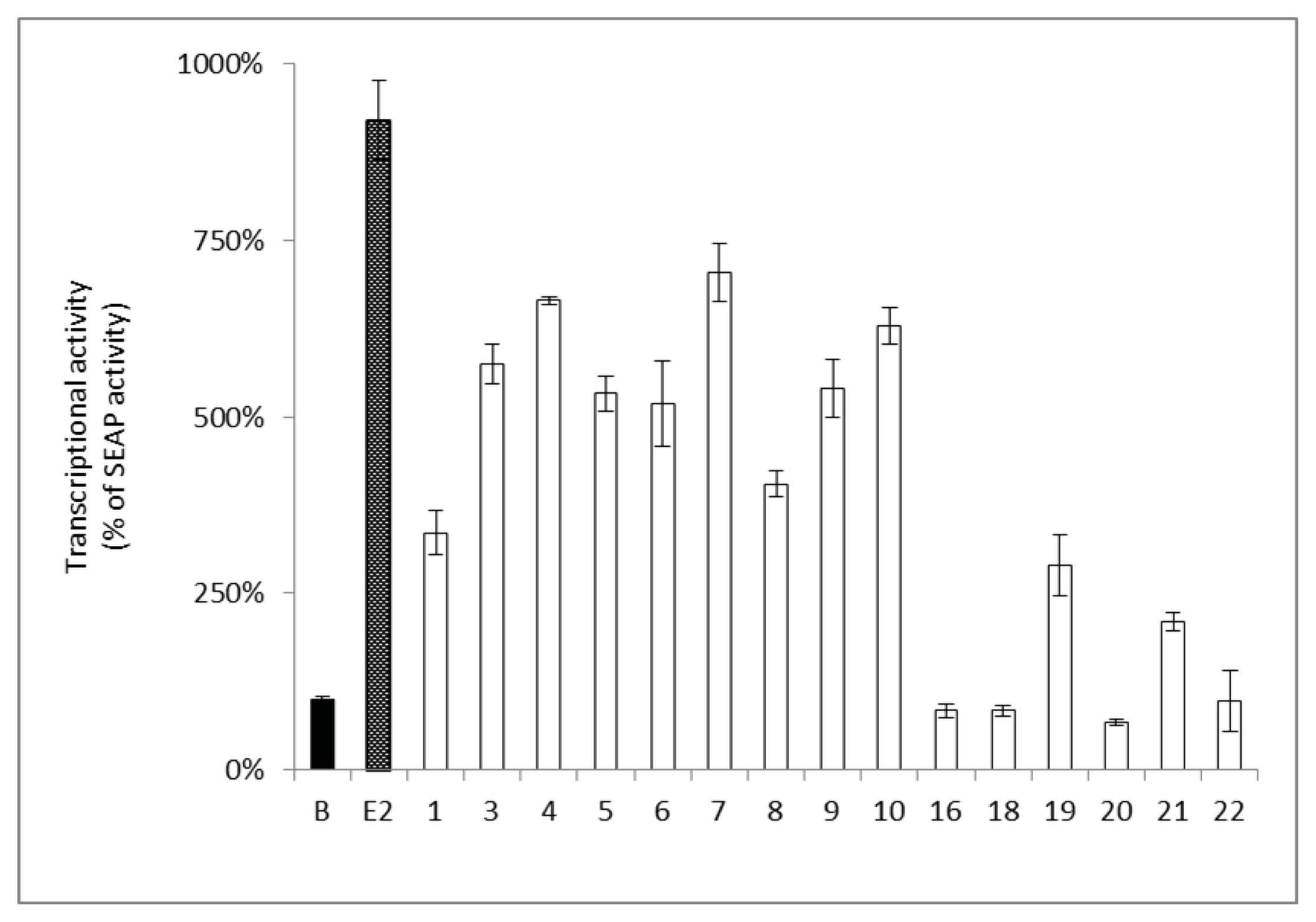
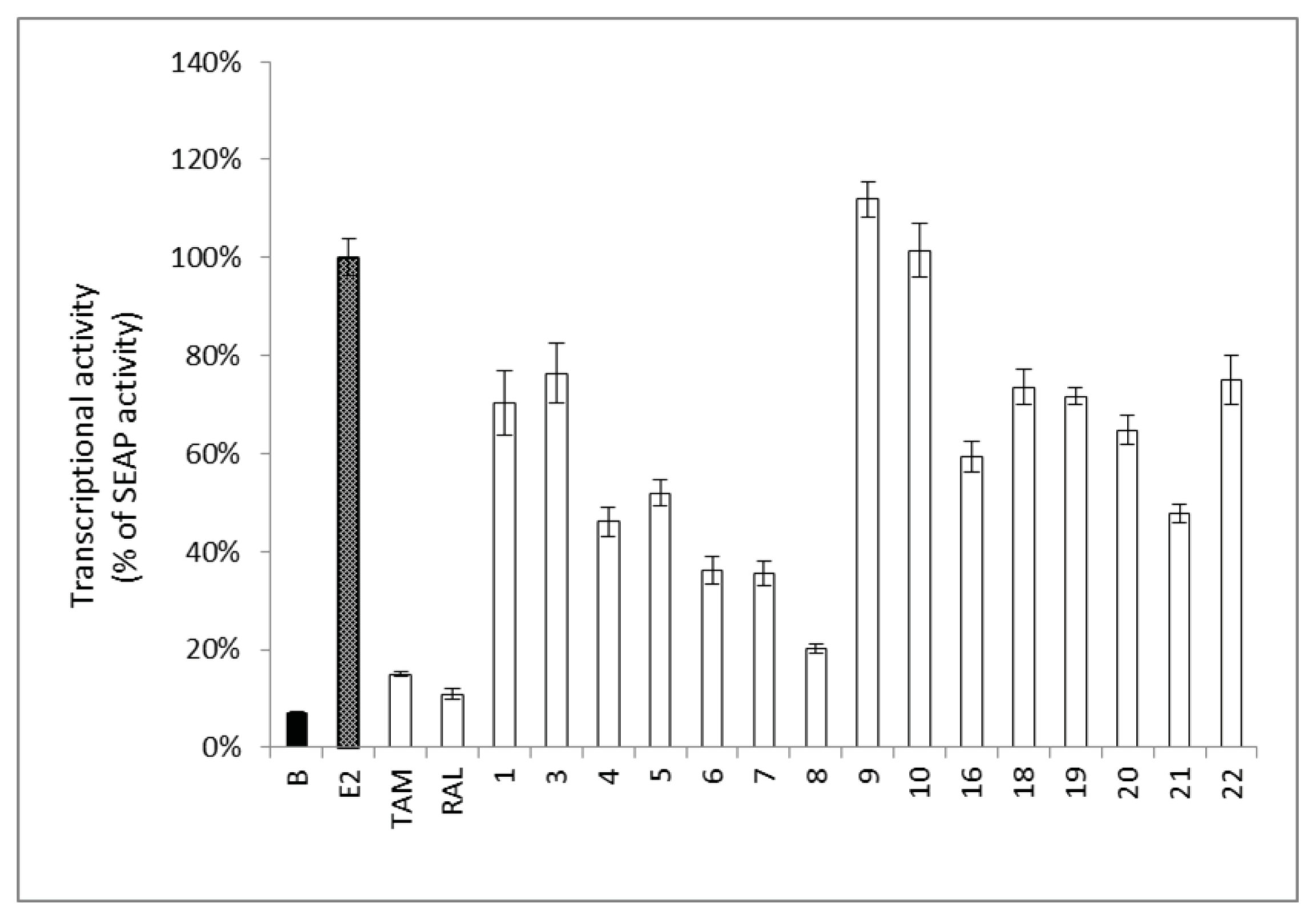
2.2. Estrogenic and Anti-Estrogenic Activities
2.3. Anti–Inflammatory and Cytotoxic Activities
3. Experimental Section
3.1. General Experimental Procedures
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Transgenic Plant Material and Estrogen–Like Reporter Assay
3.5. Histochemical Assay
3.6. Reporter Gene Assay
3.7. Human Neutrophil Superoxide Anion Generation and Elastase Release
3.8. Cytotoxicity
4. Conclusions
Supplementary Information
ijms-14-15578-s001.pdfAcknowledgments
Conflict of Interest
References
- Nelson, H.D.; Humphrey, L.L.; Nygren, P.; Teutsch, S.M.; Allan, J.D. Postmenopausal hormone replacement therapy. J. Am. Med. Assoc 2002, 288, 872–881. [Google Scholar]
- Brann, D.W.; Dhandapani, K.; Wakade, C.; Mahesh, V.B.; Khan, M.M. Neurotrophic and neuroprotective actions of estrogen: Basic mechanisms and clinical implications. Steroids 2007, 72, 381–405. [Google Scholar]
- Welty, F.K.; Lee, K.S.; Lew, N.S.; Zhou, J.R. Effect of soy nuts on blood pressure and lipid levels in hypertensive, prehypertensive, and normotensive postmenopausal women. Arch. Intern. Med 2007, 167, 1060–1067. [Google Scholar]
- Siow, R.C.; Li, F.Y.; Rowlands, D.J.; de Winter, P.; Mann, G.E. Cardiovascular targets for estrogens and phytoestrogens: Transcriptional regulation of nitric oxide synthase and antioxidant defense genes. Free Radic. Biol. Med 2007, 42, 909–925. [Google Scholar]
- Widyarini, S.; Allanson, M.; Gallagher, N.L.; Pedley, J.; Boyle, G.M.; Parsons, P.G.; Whiteman, D.C.; Walker, C.; Reeve, V.E. Isoflavonoid photoprotection in mouse and human skin is dependent on metallothionein. J. Invest. Dermatol 2006, 126, 198–204. [Google Scholar]
- Usui, T. Pharmaceutical prospects of phytoestrogens. Endocr. J 2006, 53, 7–20. [Google Scholar]
- Brand, L.; Hörler, M.; Nüesch, E.; Vassalli, S.; Barrell, P.; Yang, W.; Jefferson, R.A.; Grossniklaus, U.; Curtis, M.D. A versatile and reliable two–component system for tissue-specific gene induction in Arabidopsis. Plant Physiol 2006, 141, 1194–1204. [Google Scholar]
- Lai, W.C.; Wang, H.C.; Chen, G.Y.; Yang, J.C.; Korinek, M.; Hsieh, C.J.; Nozaki, H.; Hayashi, K.I.; Wu, C.C.; Wu, Y.C.; et al. Using the pER8:GUS reporter system to screen for phytoestrogens from Caesalpinia sappan. J. Nat. Prod 2011, 74, 1698–1706. [Google Scholar]
- Yang, Y.P.; Lu, S.Y. Flora of Taiwan, 2nd ed; Editorial Committee of the Flora of Taiwan: Taipei, Taiwan, 1995; Volume 3, pp. 284–285. [Google Scholar]
- Shiao, Y.J.; Wang, C.N.; Wang, W.Y.; Lin, Y.L. Neuroprotective flavonoids from Flemingia macrophylla. Planta Med 2005, 71, 835–840. [Google Scholar]
- Ko, Y.J.; Lu, T.C.; Kitanaka, S.; Liu, C.Y.; Wu, J.B.; Kuo, C.L.; Cheng, H.Y.; Lin, Y.C.; Peng, W.H. Analgesic and anti-inflammatory activities of the aqueous extracts from three Flemingia species. Am. J. Chin. Med 2010, 38, 625–638. [Google Scholar]
- Ho, H.Y.; Wu, J.B.; Lin, W.C. Flemingia macrophylla extract ameliorates experimental osteoporosis in ovariectomized rats. Evid. Based Complement. Alternat. Med. 2011. [Google Scholar] [CrossRef]
- Babu, S.S.; Rao, J.M.; Rao, K.V.J. Flemichin E, a new chromenoflavanone from the roots of Flemingia wallichii W and A. Indian J. Chem 1979, 18B, 388–389. [Google Scholar]
- Li, H.; Yang, M.; Miao, J.; Ma, X. Prenylated isoflavones from Flemingia philippinensis. Magn. Reson. Chem 2008, 46, 1203–1207. [Google Scholar]
- Lewis, P.; Kaltia, S.; Wähälä, K. The phase transfer catalysed synthesis of isoflavone-O-glucosides. J. Chem. Soc., Perkin Trans. 1 1998, 16, 2481–2484. [Google Scholar]
- Wang, H.; Nair, M.G.; Strasburg, G.M.; Booren, A.M.; Gray, J.I. Antioxidant polyphenols from tart cherries (Prunus cerasus). J. Agric. Food Chem 1999, 47, 840–844. [Google Scholar]
- Pistelli, L.; Giachi, I.; Potenza, D.; Morelli, I. A new isoflavone from Genista corsica. J. Nat. Prod 2000, 63, 504–506. [Google Scholar]
- Dahiya, J.S.; Strange, R.N.; Bilyard, K.G.; Cooksey, C.J.; Garratt, P.J. Two isoprenylated isoflavone phytoalexins from Cajanus cajan. Phytochemistry 1984, 23, 871–873. [Google Scholar]
- Emami, S.A.; Amin-Ar-Ramimeh, E.; Ahi, A.; Kashy, M.R.; Schneider, B.; Iranshahi, M. Prenylated flavonoids and flavonostilbenes from Sophora pachycarpa roots. Pharm. Biol 2007, 45, 453–457. [Google Scholar]
- Ahn, E.M.; Nakamura, N.; Akao, T.; Komatsu, K.; Qui, M.H.; Hattori, M. Prenylated flavonoids from Moghania philippinensis. Phytochemistry 2003, 64, 1389–1394. [Google Scholar]
- Vitor, R.F.; Mota-Filipe, H.; Teixeira, G.; Borges, C.; Rodrigues, A.I.; Teixeira, A.; Paulo, A. Flavonoids of an extract of Pterospartum tridentatum showing endothelial protection against oxidative injury. J. Ethnopharmacol 2004, 93, 363–370. [Google Scholar]
- Dai, J.Q.; Hou, Z.F.; Zhu, Q.X.; Yang, L.; Li, Y. Sesquiterpenes and flavonoids from Serratula strangulata. J. Chin. Chem. Soc 2001, 48, 249–252. [Google Scholar]
- Kinoshita, T.; Ichinose, K.; Takahashi, C.; Ho, F.C.; Wu, J.B.; Sankawa, U. Chemical studies on Sophora tomentosa: The isolation of a new class of isoflavonoid. Chem. Pharm. Bull 1990, 38, 2756–2759. [Google Scholar]
- Lane, G.A.; Newman, R.H. Isoflavones from Lupinus angustifolius root. Phytochemistry 1986, 26, 295–300. [Google Scholar]
- Brink, A.J.; Rall, G.J.H.; Engelbrecht, J.P. Phenolic neorautanenia isoflavanoids: The isolation and structures of neorauflavene, (−)-neorauflavane and neoraufurane, three novel isoflavanoids from Neorautanenia edulis. Tetrahedron 1974, 30, 311–314. [Google Scholar]
- Rao, K.N.; Srimannarayana, G. Fleminone, a flavanone from the stems of Flemingia macrophylla. Phytochemistry 1983, 22, 2287–2290. [Google Scholar]
- Li, J.; Wang, M. Two flavanones from the root bark of Lespedeza davidii. Phytochemistry 1989, 28, 3564–3566. [Google Scholar]
- Slade, D.; Ferreira, D.; Marais, J.P. Circular dichroism, a powerful tool for the assessment of absolute configuration of flavonoids. Phytochemistry 2005, 66, 2177–2215. [Google Scholar]
- Ryu, S.Y.; Lee, H.S.; Kim, Y.K.; Kim, S.H. Determination of isoprenyl and lavandulyl positions of flavonoids from Sophora flavescens by NMR experiment. Arch. Pharm. Res 1997, 20, 491–495. [Google Scholar]
- Garcia, J.; Massoma, T.; Morin, C.; Mpondo, T.N.; Nyassé, B. 4′-O-methylgallocatechin from Panda oleosa. Phytochemistry 1993, 32, 1626–1628. [Google Scholar]
- De–Eknamkul, W.; Potduang, B. Biosynthesis of [beta]-sitosterol and stigmasterol in Croton sublyratus proceeds via a mixed origin of isoprene units. Phytochemistry 2003, 62, 389–398. [Google Scholar]
- Rahman, M.M.; Sarker, S.D.; Byres, M.; Gray, A.I. New salicylic acid and isoflavone derivatives from Flemingia paniculata. J. Nat. Prod 2004, 67, 402–406. [Google Scholar]
- Wuttke, W.; Jarry, H.; Westphalen, S.; Christoffel, V.; Seidlová-Wuttke, D. Phytoestrogens for hormone replacement therapy? J. Steroid Biochem. Mol. Biol 2002, 83, 133–147. [Google Scholar]
- Cornwell, T.; Cohick, W.; Raskin, I. Dietary phytoestrogens and health. Phytochemistry 2004, 65, 995–1016. [Google Scholar]
- Vaya, J.; Tamir, S. The relation between the chemical structure of flavonoids and their estrogen-like activities. Curr. Med. Chem 2004, 11, 1333–1343. [Google Scholar]
- De–Eknamkul, W.; Umehara, K.; Monthakantirat, O.; Toth, R.; Frecer, V.; Knapic, L.; Braiuca, P.; Noguchi, H.; Miertus, S. QSAR study of natural estrogen-like isoflavonoids and diphenolics from Thai medicinal plants. J. Mol. Graph. Model 2011, 29, 784–794. [Google Scholar]
- Fang, H.; Tong, W.; Shi, L.M.; Blair, R.; Perkins, R.; Branham, W.; Hass, B.S.; Xie, Q.; Dial, S.L.; Moland, C.L.; et al. Structure-activity relationships for a large diverse set of natural, synthetic, and environmental estrogens. Chem. Res. Toxicol 2001, 14, 280–294. [Google Scholar]
- Choi, S.Y.; Ha, T.Y.; Ahn, J.Y.; Kim, S.R.; Kang, K.S.; Hwang, I.K.; Kim, S. Estrogenic activities of isoflavones and flavones and their structure-activity relationships. Planta Med 2008, 74, 25–32. [Google Scholar]
- Collins-Burow, B.M.; Burow, M.E.; Duong, B.N.; McLachlan, J.A. Estrogenic and antiestrogenic activities of flavonoid phytochemicals through estrogen receptor binding-dependent and -independent mechanisms. Nutr. Cancer 2000, 38, 229–244. [Google Scholar]
- Pratesi, F.; Dioni, I.; Tommasi, C.; Alcaro, M.C.; Paolini, I.; Barbetti, F.; Boscaro, F.; Panza, F.; Puxeddu, I.; Rovero, P.; et al. Antibodies from patients with rheumatoid arthritis target citrullinated histone 4 contained in neutrophils extracellular traps. Ann. Rheum. Dis. 2013. [Google Scholar] [CrossRef]
- Sadik, C.D.; Kim, N.D.; Iwakura, Y.; Luster, A.D. Neutrophils orchestrate their own recruitment in murine arthritis through C5aR and FcγR signaling. Proc. Natl. Acad. Sci. USA 2012, 109, 3177–3185. [Google Scholar]
- Li, J.; Gang, D.; Yu, X.; Hu; Yue, Y.; Cheng, W.; Pan, X.; Zhang, P. Genistein: The potential for efficacy in rheumatoid arthritis. Clin. Rheumatol. 2013, 32, 535–540. [Google Scholar]
- Chang, F.R.; Hayashi, K.; Chua, N.H.; Kamio, S.; Huang, Z.Y.; Nozaki, H.; Wu, Y.C. The transgenic Arabidopsis plant system, pER8-GFP, as a powerful tool in searching for natural product estrogen–agonists/antagonists. J. Nat. Prod 2005, 68, 971–973. [Google Scholar]
- Jefferson, R.A.; Kavanagh, T.A.; Bevan, M.W. GUS fusions: Beta-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 1987, 6, 3901–3907. [Google Scholar]
- Alley, M.C.; Scudiero, D.A.; Monks, A.; Hursey, M.L.; Czerwinski, M.J.; Fine, D.L.; Abbott, B.J.; Mayo, J.G.; Shoemaker, R.H.; Boyd, M.R. Feasibility of drug screening with panels of human tumor cell lines using a microculture tetrazolium assay. Cancer Res 1988, 48, 589–601. [Google Scholar]
- Scudiero, D.A.; Shoemaker, R.H.; Paull, K.D.; Monks, A.; Tierney, S.; Nofziger, T.H.; Currens, M.J.; Seniff, D.; Boyd, M.R. Evaluation of a soluble tetrazolium/formazan assay for cell growth and drug sensitivity in culture using human and other tumor cell lines. Cancer Res 1988, 48, 4827–4833. [Google Scholar]
- Hwang, T.L.; Yeh, S.H.; Leu, Y.L.; Chern, C.Y.; Hsu, H.C. Inhibition of superoxide anion and elastase release in human neutrophils by 3′-isopropoxychalcone via a cAMP-dependent pathway. Br. J. Pharmacol 2006, 148, 78–87. [Google Scholar]
- Chang, H.L.; Chang, F.R.; Chen, J.S.; Wang, H.P.; Wu, Y.H.; Wang, C.C.; Wu, Y.C.; Hwang, T.L. Inhibitory effects of 16-hydroxycleroda-3,13(14)E-dien-15-oic acid on superoxide anion and elastase release in human neutrophils through multiple mechanisms. Eur. J. Pharmacol 2008, 586, 332–339. [Google Scholar]
- Sladowski, D.; Steer, S.J.; Clothier, R.H.; Balls, M. An improved MTT assay. J. Immunol. Methods 1993, 157, 203–207. [Google Scholar]
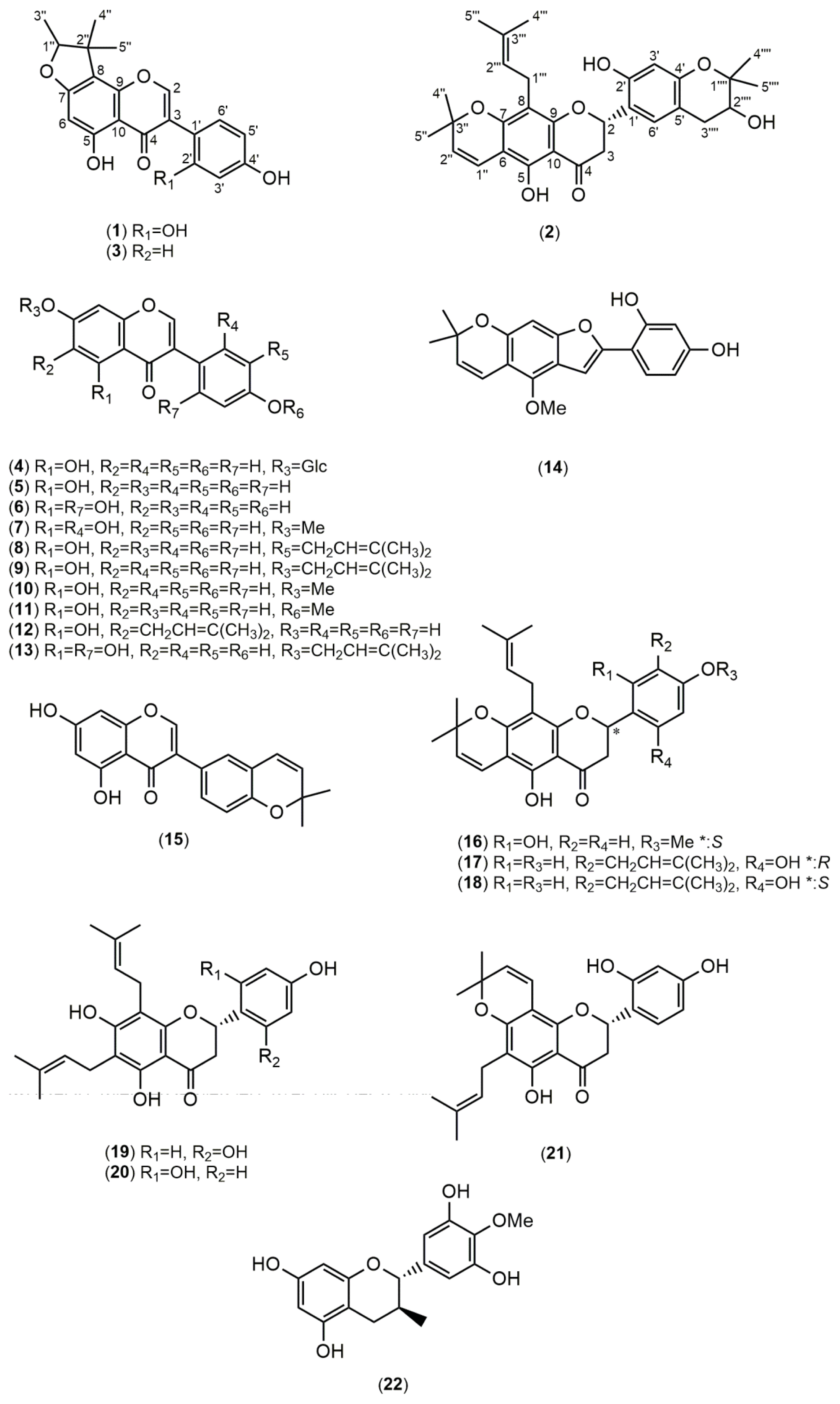
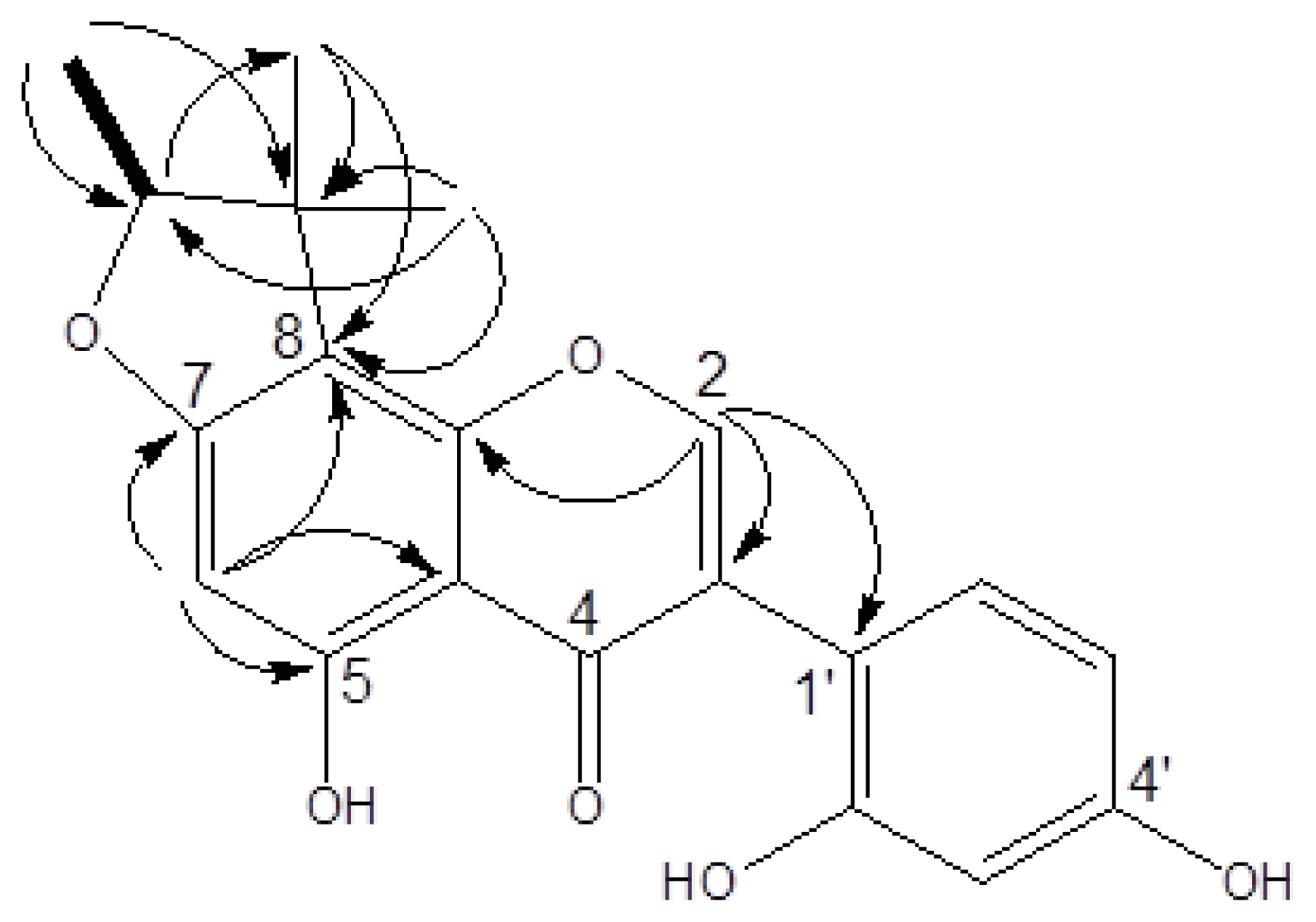
| Position | δC (mult.) a | δH (mult., J in Hz) b | HMBC (1H→13C) |
|---|---|---|---|
| 2 | 155.1 | 8.33 (s) | 3, 4, 9, 1′ |
| 3 | 121.7 | ||
| 4 | 181.9 | ||
| 5 | 165.2 | ||
| 6 | 94.7 | 6.53 (s) | 5, 7, 8, 10 |
| 7 | 164.0 | ||
| 8 | 112.8 | ||
| 9 | 153.5 | ||
| 10 | 106.8 | ||
| 1′ | 110.0 | ||
| 2′ | 158.3 | ||
| 3′ | 104.4 | 7.08 (d, 2.4) | 2′, 4′, 1′, 5′ |
| 4′ | 160.7 | ||
| 5′ | 107.8 | 6.94 (dd, 8.4, 2.4) | 1′, 3′ |
| 6′ | 133.4 | 7.62 (d, 8.4) | 3, 2′, 4′ |
| 1″ | 90.9 | 4.43 (q, 6.4) | 4″, 5″ |
| 2″ | 43.8 | ||
| 3″ | 14.2 | 1.28 (d, 6.4) | 1″, 2″, 4″, 5″ |
| 4″ | 25.5 | 1.37 (s) | 8, 1″, 2″, 5″ |
| 5″ | 21.3 | 1.14 (s) | 8, 1″, 2″, 3″, 4″ |
| Position | δC (mult.) a | δH (mult., J in Hz) b | HMBC (1H→13C) |
|---|---|---|---|
| 2 | 77.2 | 5.52 (dd, 13.2, 3.0) | |
| 3 | 42.0 | 3.13 (dd, 17.1, 3.0) | 2, 4 |
| 2.82 (dd, 17.1, 13.2) | 4 | ||
| 4 | 196.5 | ||
| 5 | 156.7 | ||
| 5-OH | 12.9 (s) | 5, 10 | |
| 6 | 103.2 | ||
| 7 | 158.7 | ||
| 8 | 108.7 | ||
| 9 | 159.7 | ||
| 10 | 102.6 | ||
| 1′ | 117.5 | ||
| 2′ | 153.6 | ||
| 3′ | 105.3 | 6.39 (s) | 1′, 4′, 5′ |
| 4′ | 153.9 | ||
| 5′ | 111.0 | ||
| 6′ | 128.3 | 6.94 (s) | 2, 4′, 3‴′ |
| 1″ | 115.6 | 6.64 (d, 10.2) | 7, 3″ |
| 2″ | 126.2 | 5.51 (d, 10.2) | 6, 3″ |
| 3″ | 78.2 | ||
| 4″-CH3 | 28.3 | 1.43 (s) | 2″, 3″, 5″ |
| 5″-CH3 | 28.4 | 1.45 (s) | 2″, 3″, 4″ |
| 1‴ | 21.4 | 3.20 (2H, d, 7.8) | 7, 8, 9, 2‴, 3‴ |
| 2‴ | 122.3 | 5.10 (td, 7.8, 1.8) | 4‴, 5‴ |
| 3‴ | 131.7 | ||
| 4‴-CH3 | 25.8 | 1.67 (d, 1.8) | 2‴, 3‴, 5‴ |
| 5‴-CH3 | 17.8 | 1.68 (s) | 2‴, 3‴, 4‴ |
| 1‴′ | 77.1 | ||
| 2‴′ | 69.7 | 3.80 (dd, 5.4, 4.8) | |
| 3‴′ | 30.7 | 3.01 (dd,16.8, 4.8) | 4′, 5′, 6′, 2‴′ |
| 2.71 (dd,16.8, 5.4) | 4′, 5′, 6′, 1‴′, 2‴′ | ||
| 4‴′-CH3 | 22.2 | 1.37 (s) | 1‴′, 2‴′, 5‴′ |
| 5‴′-CH3 | 24.8 | 1.33 (s) | 1‴′, 2‴′, 4‴′ |
| Compounds | MAC (μM) |
|---|---|
| 17β-estradiol | 5.93 × 10−4 |
| 1 | 35.31 |
| 3 | 36.98 |
| 4 | 0.0139 |
| 5 | 0.0037 |
| 6 | 0.042 |
| 7 | 2.6 |
| 8 | 36.98 |
| 12 | 59.17 |
| 13 | 28.25 |
| 16 | >500 |
| 19 | 58.96 |
| 21 | 118.48 |
| 22 | 312.50 |
| Compounds | IC50 (g/mL) or Inh% | |
|---|---|---|
| Superoxide anion | Elastase release | |
| 3 | 3.74 ± 0.30 a,*** | 23.31 ± 3.82 c,*** |
| 4 | 6.82 ± 1.41 ** | 11.9 ± 5.02 |
| 5 | 0.66 ± 0.06 a | 4.25 ± 1.23 a |
| 6 | 1.89 ± 0.05 a | 4.32 ± 0.14 a |
| 7 | 8.86 ± 1.34 a | 4.22 ± 0.72 a |
| 8 | 2.18 ± 0.24 a | 10.31 ± 3.82 b,*** |
| 16 | 10.31 ± 3.82 | 40.92 ± 11.08 b |
| 19 | 73.08 ± 4.89 ** | 55.26 ± 5.94 c,** |
| 21 | ND | 92.87 ± 2.27 b,** |
| 22 | 29.37 ± 4.42 ** | −4.34 ± 5.95 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lai, W.-C.; Tsui, Y.-T.; Singab, A.N.B.; El-Shazly, M.; Du, Y.-C.; Hwang, T.-L.; Wu, C.-C.; Yen, M.-H.; Lee, C.-K.; Hou, M.-F.; et al. Phyto-SERM Constitutes from Flemingia macrophylla. Int. J. Mol. Sci. 2013, 14, 15578-15594. https://doi.org/10.3390/ijms140815578
Lai W-C, Tsui Y-T, Singab ANB, El-Shazly M, Du Y-C, Hwang T-L, Wu C-C, Yen M-H, Lee C-K, Hou M-F, et al. Phyto-SERM Constitutes from Flemingia macrophylla. International Journal of Molecular Sciences. 2013; 14(8):15578-15594. https://doi.org/10.3390/ijms140815578
Chicago/Turabian StyleLai, Wan-Chun, Ya-Ting Tsui, Abdel Nasser B. Singab, Mohamed El-Shazly, Ying-Chi Du, Tsong-Long Hwang, Chin-Chung Wu, Ming-Hong Yen, Ching-Kuo Lee, Ming-Feng Hou, and et al. 2013. "Phyto-SERM Constitutes from Flemingia macrophylla" International Journal of Molecular Sciences 14, no. 8: 15578-15594. https://doi.org/10.3390/ijms140815578
APA StyleLai, W.-C., Tsui, Y.-T., Singab, A. N. B., El-Shazly, M., Du, Y.-C., Hwang, T.-L., Wu, C.-C., Yen, M.-H., Lee, C.-K., Hou, M.-F., Wu, Y.-C., & Chang, F.-R. (2013). Phyto-SERM Constitutes from Flemingia macrophylla. International Journal of Molecular Sciences, 14(8), 15578-15594. https://doi.org/10.3390/ijms140815578








