Knockdown of FABP3 Impairs Cardiac Development in Zebrafish through the Retinoic Acid Signaling Pathway
Abstract
:1. Introduction
2. Results
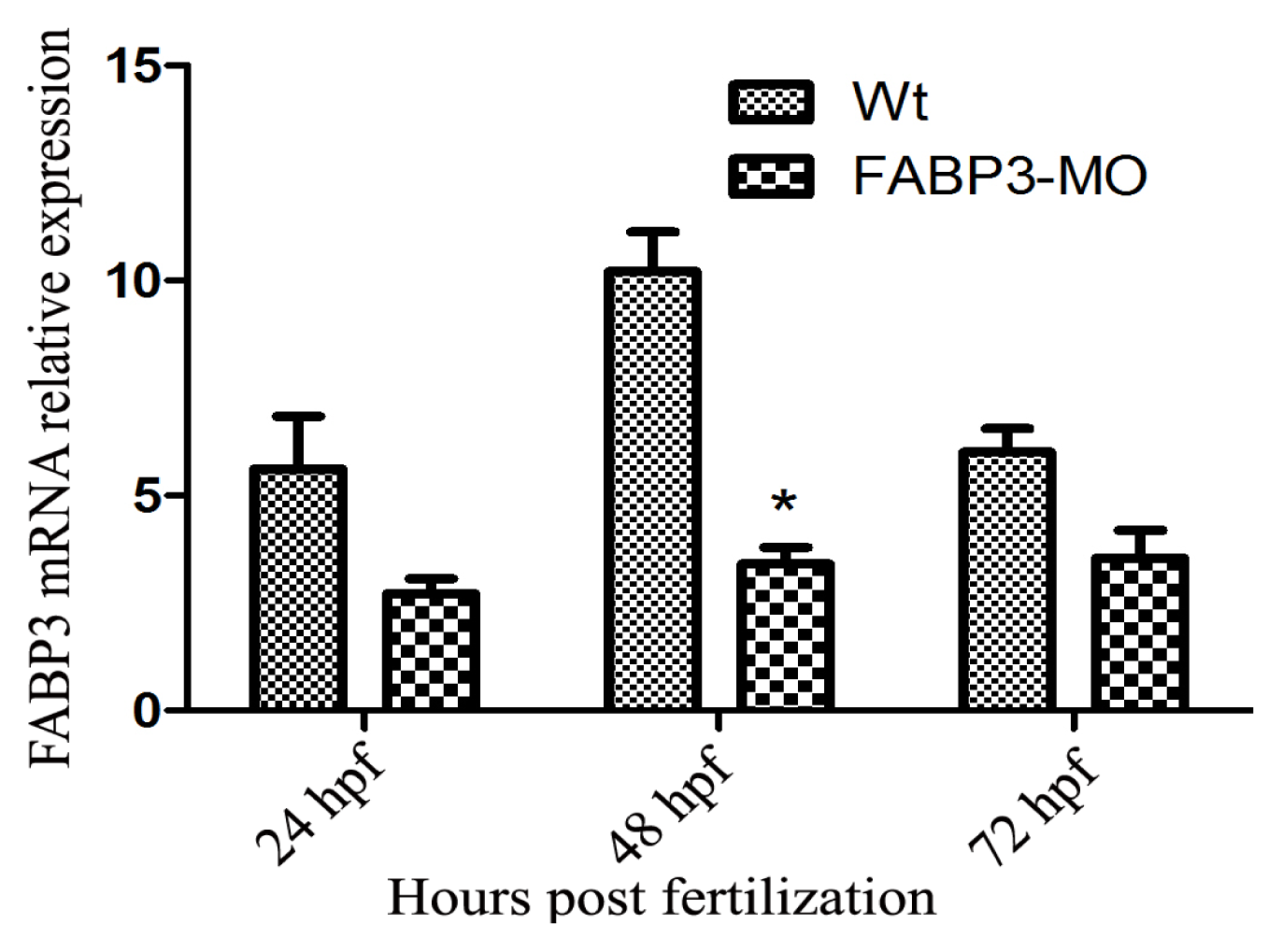
2.1. Verification of FABP3 Knockdown by FABP3-MO Injection in Zebrafish
2.2. FABP3-MO Injection Was Associated with Zebrafish Embryo Lethality
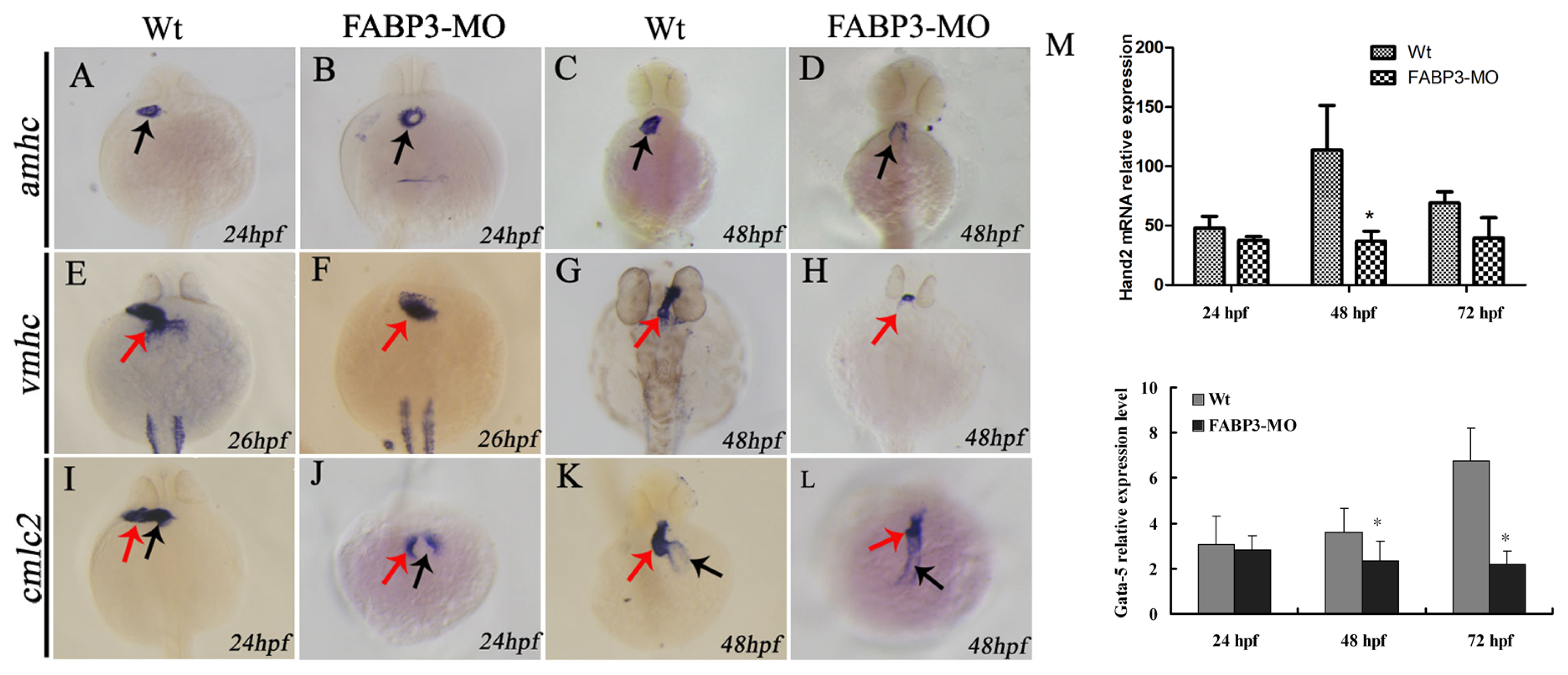
2.3. FABP3-MO Causes Zebrafish Embryonic Malformation, Especially in the Heart
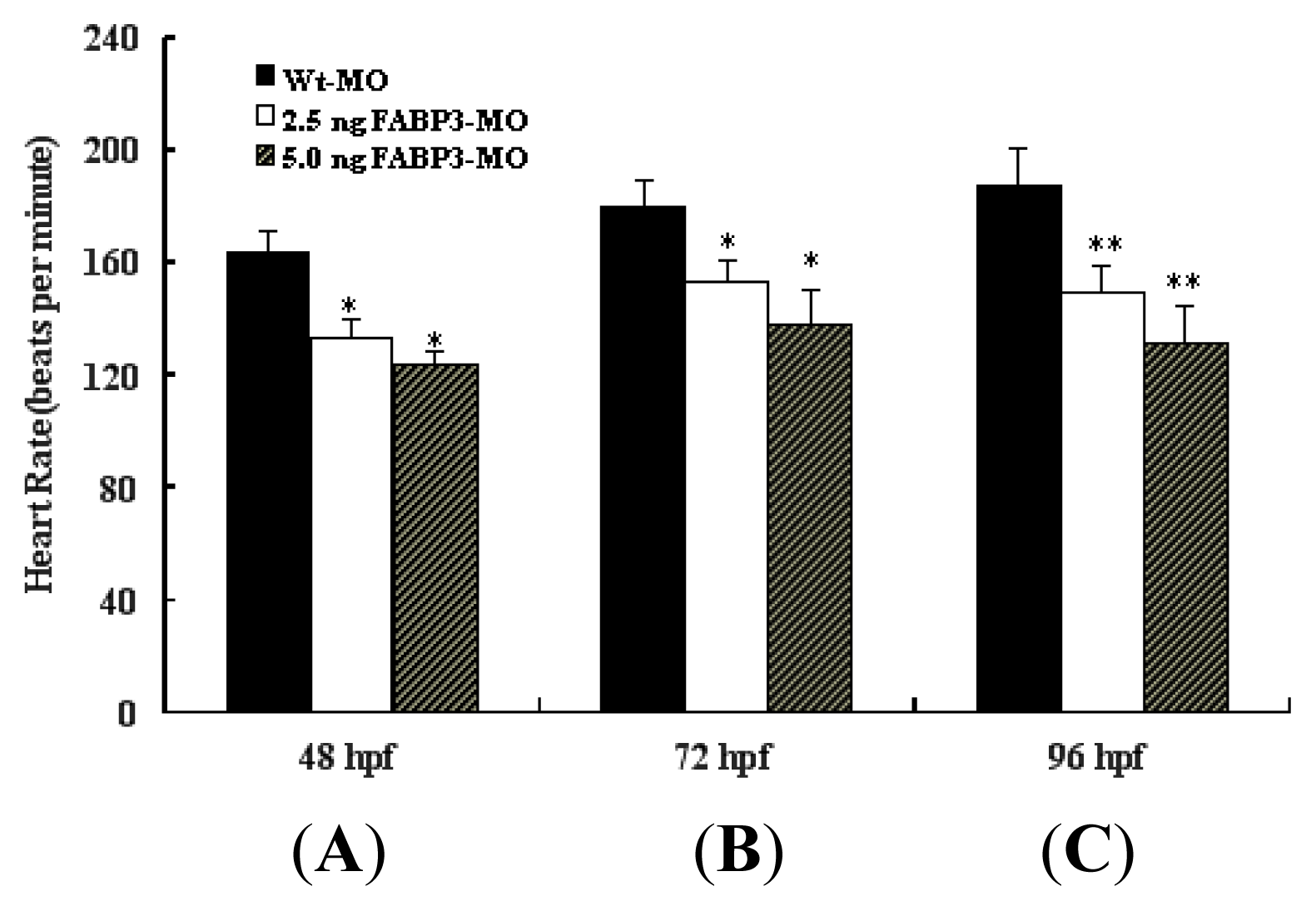
2.4. Decreased Heart Rate in FABP3-MO Injected Embryos
2.5. Effect of FABP3-MO on the Expression of Cardiac-Specific Genes in Zebrafish
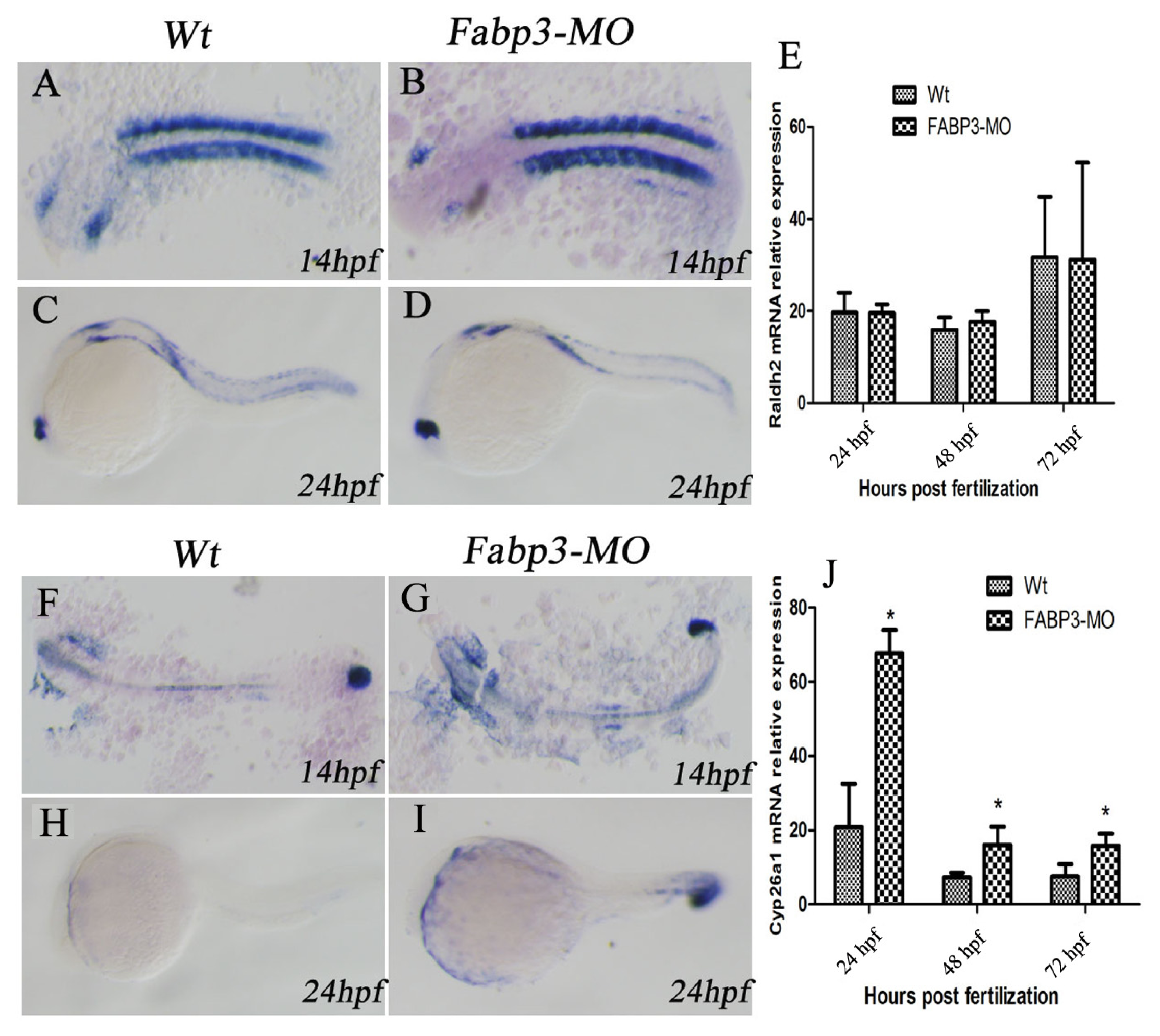
2.6. FABP3-MO Alters the Retinoic Acid Signaling Pathway in Zebrafish Embryos
3. Discussion
4. Experimental Section
4.1. Embryo Maintenance and Transplantation
4.2. Morpholinos and Microinjection
4.3. Determining the Rate of Lethality and Observation of Zebrafish Heart Development
4.4. Heart Rate Measurement
4.5. Preparation of RNA Probes
4.6. Whole-Mount in Situ Hybridization
4.7. Real-Time Polymerase Chain Reaction (PCR) for the Assessment of Cardiac-Specific Gene Expression
4.8. Statistical Analysis
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Hoffman, J.I. Congenital heart disease: Incidence and inheritance. Pediatr. Clin. N. Am 1990, 37, 25–43. [Google Scholar]
- Moller, J.H.; Allen, H.D.; Clark, E.B.; Dajani, A.S.; Golden, A.; Hayman, L.L.; Lauer, R.M.; Marmer, E.L.; McAnulty, J.H.; Oparil, S.; et al. Report of the task force on children and youth. American Heart Association. Circulation 1993, 88, 2479–2486. [Google Scholar]
- Bruneau, B.G. The developmental genetics of congenital heart disease. Nature 2008, 451, 943–948. [Google Scholar]
- Nemer, M. Genetic insights into normal and abnormal heart development. Cardiovasc. Pathol 2008, 17, 48–54. [Google Scholar]
- Srivastava, D.; Olson, E.N. A genetic blueprint for cardiac development. Nature 2000, 407, 221–226. [Google Scholar]
- McCann, C.J.; Glover, B.M.; Menown, I.B.; Moore, M.J.; McEneny, J.; Owens, C.G.; Smith, B.; Sharpe, P.C.; Young, I.S.; Adgey, J.A. Novel biomarkers in early diagnosis of acute myocardial infarction compared with cardiac troponin T. Eur. Heart J 2008, 29, 2843–2850. [Google Scholar]
- Zhang, H.; Zhou, L.; Yang, R.; Sheng, Y.; Sun, W.; Kong, X.; Cao, K. Identification of differentially expressed genes in human heart with ventricular septal defect using suppression subtractive hybridization. Biochem. Biophys. Res. Commun 2006, 342, 135–144. [Google Scholar]
- Shen, Y.H.; Song, G.X.; Liu, Y.Q.; Sun, W.; Zhou, L.J.; Liu, H.L.; Yang, R.; Sheng, Y.H.; Qian, L.M.; Kong, X.Q. Silencing of FABP3 promotes apoptosis and induces mitochondrion impairment in embryonic carcinoma cells. J. Bioenerg. Biomembr 2012, 44, 317–323. [Google Scholar]
- Tang, M.K.; Kindler, P.M.; Cai, D.Q.; Chow, P.H.; Li, M.; Lee, K.K. Heart-type fatty acid binding proteins are upregulated during terminal differentiation of mouse cardiomyocytes, as revealed by proteomic analysis. Cell. Tissue Res 2004, 316, 339–347. [Google Scholar]
- Keegan, B.R.; Feldman, J.L.; Begemann, G.; Ingham, P.W.; Yelon, D. Retinoic acid signaling restricts the cardiac progenitor pool. Science 2005, 307, 247–249. [Google Scholar]
- Sirbu, I.O.; Zhao, X.; Duester, G. Retinoic acid controls heart anteroposterior patterning by down-regulating Isl1 through the Fgf8 pathway. Dev. Dyn 2008, 237, 1627–1635. [Google Scholar]
- Hochgreb, T.; Linhares, V.L.; Menezes, D.C.; Sampaio, A.C.; Yan, C.Y.; Cardoso, W.V.; Rosenthal, N.; Xavier-Neto, J. A caudorostral wave of RALDH2 conveys anteroposterior information to the cardiac field. Development 2003, 130, 5363–5374. [Google Scholar]
- Hu, N.; Sedmera, D.; Yost, H.J.; Clark, E.B. Structure and function of the developing zebrafish heart. Anat. Rec 2000, 260, 148–157. [Google Scholar]
- Fishman, M.C.; Chien, K.R. Fashioning the vertebrate heart: Earliest embryonic decisions. Development 1997, 124, 2099–2117. [Google Scholar]
- Bakkers, J. Zebrafish as a model to study cardiac development and human cardiac disease. Cardiovasc. Res 2011, 91, 279–288. [Google Scholar]
- Liu, R.Z.; Denovan-Wright, E.M.; Wright, J.M. Structure, linkage mapping and expression of the heart-type fatty acid-binding protein gene (fabp3) from zebrafish (Danio rerio). Eur. J. Biochem 2003, 270, 3223–3234. [Google Scholar]
- Liu, R.Z.; Saxena, V.; Sharma, M.K.; Thisse, C.; Thisse, B.; Denovan-Wright, E.M.; Wright, J.M. The fabp4 gene of zebrafish (Danio rerio)—Genomic homology with the mammalian FABP4 and divergence from the zebrafish fabp3 in developmental expression. FEBS J 2007, 274, 1621–1633. [Google Scholar]
- Nasevicius, A.; Ekker, S.C. Effective targeted gene ‘knockdown’ in zebrafish. Nat. Genet 2000, 26, 216–220. [Google Scholar]
- Chen, J.N.; van Eeden, F.J.; Warren, K.S.; Chin, A.; Nusslein-Volhard, C.; Haffter, P.; Fishman, M.C. Left-right pattern of cardiac BMP4 may drive asymmetry of the heart in zebrafish. Development 1997, 124, 4373–4382. [Google Scholar]
- De Pater, E.; Clijsters, L.; Marques, S.R.; Lin, Y.F.; Garavito-Aguilar, Z.V.; Yelon, D.; Bakkers, J. Distinct phases of cardiomyocyte differentiation regulate growth of the zebrafish heart. Development 2009, 136, 1633–1641. [Google Scholar]
- Thaller, C.; Eichele, G. Identification and spatial distribution of retinoids in the developing chick limb bud. Nature 1987, 327, 625–628. [Google Scholar]
- Berdougo, E.; Coleman, H.; Lee, D.H.; Stainier, D.Y.; Yelon, D. Mutation of weak atrium/atrial myosin heavy chain disrupts atrial function and influences ventricular morphogenesis in zebrafish. Development 2003, 130, 6121–6129. [Google Scholar]
- Yelon, D.; Ticho, B.; Halpern, M.E.; Ruvinsky, I.; Ho, R.K.; Silver, L.M.; Stainier, D.Y. The bHLH transcription factor hand2 plays parallel roles in zebrafish heart and pectoral fin development. Development 2000, 127, 2573–2582. [Google Scholar]
- Garavito-Aguilar, Z.V.; Riley, H.E.; Yelon, D. Hand2 ensures an appropriate environment for cardiac fusion by limiting Fibronectin function. Development 2010, 137, 3215–3220. [Google Scholar]
- Rochais, F.; Mesbah, K.; Kelly, R.G. Signaling pathways controlling second heart field development. Circ. Res 2009, 104, 933–942. [Google Scholar]
- Ryckebusch, L.; Wang, Z.; Bertrand, N.; Lin, S.C.; Chi, X.; Schwartz, R.; Zaffran, S.; Niederreither, K. Retinoic acid deficiency alters second heart field formation. Proc. Natl. Acad. Sci. USA 2008, 105, 2913–2918. [Google Scholar]
- Roberts, C.; Ivins, S.; Cook, A.C.; Baldini, A.; Scambler, P.J. Cyp26 genes a1, b1 and c1 are down-regulated in Tbx1 null mice and inhibition of Cyp26 enzyme function produces a phenocopy of DiGeorge Syndrome in the chick. Hum. Mol. Genet 2006, 15, 3394–3410. [Google Scholar]
- Westerfield, M. The Zebrafish Book: A Guide for the Laboratory Use of Zebrafish Danio (Brachydanio) rerio; M. Westerfield: Eugene, OR, USA, 1993. [Google Scholar]
- Kimmel, C.B.; Ballard, W.W.; Kimmel, S.R.; Ullmann, B.; Schilling, T.F. Stages of embryonic development of the zebrafish. Dev. Dyn 1995, 203, 253–310. [Google Scholar]
- Thisse, C.; Thisse, B. High-resolution in situ hybridization to whole-mount zebrafish embryos. Nat. Protoc 2008, 3, 59–69. [Google Scholar]
- Yelon, D.; Horne, S.A.; Stainier, D.Y. Restricted expression of cardiac myosin genes reveals regulated aspects of heart tube assembly in zebrafish. Dev. Biol 1999, 214, 23–37. [Google Scholar]

| Concentration (ng) | Treatment duration | |||||
|---|---|---|---|---|---|---|
| 24 hpf | 48 hpf | 72 hpf | 96 hpf | 120 hpf | ||
| Non-injection | 12(4) | 13(4.3) | 13(4.3) | 15(5) | 15(5) | |
| Wt-MO | 16(5.1) | 19(6.3) | 20(6.7) | 20(6.7) | 20(6.7) | |
| FABP3-MO | 1.0 | 19(6.7) * | 26(8.7) * | 30(10) * | 34(11.3) * | 35(11.7) * |
| 2.5 | 31(10.3) * | 50(16.7) * | 59(19.7) * | 66(22) * | 66(22) * | |
| 5.0 | 55(18.3) * | 78(26) * | 89(29.7) * | 101(33.7) * | 113(37.7) * | |
| 7.5 | 92(30.7) * | 123(41.1) * | 157(52.1) * | 181(60.1) * | 215(71.7) * | |
| 10.0 | 193(64.3) * | 248(82.7) * | 275(91.7) * | 289(96.3) * | 289(96.3) * | |
| χ2 | 533.145 | 720.105 | 945.243 | 917.345 | 981.454 | |
| p | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | |
| χ trend | 398.827 | 568.573 | 724.707 | 781.940 | 860.933 | |
| P | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | |
| Gene name | Sense (5′→3′) | Antisense (5′→3′) |
|---|---|---|
| FABP3 | CGATGAGTACATGAAAGGAA | GTGGATTTGAAAGTGCTGAC |
| Hand2 | TACCATGGCACCTTCGTACA | CCTTTCTTCTTTGGCGTCTG |
| Gata5 | CCACCGAATTCTGATCCGAGACC | GGAGGCTCGAGAAACGATATAATTCC |
| Cyp26a1 | ACCATCGTGCTACCCGTTTT | GGCGGTAGAGGACTTCTGCA |
| Raldh2 | CATTTTTGCAGATGCTGATTTTG | CAAAGATACGGGAACCAGCAGT |
| β-atcin | CAACAGAGAGAAGATGACACAGATCA | GTCACACCATCACCAGAGTCCATCAC |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, X.; Zhou, L.; Jin, J.; Yang, Y.; Song, G.; Shen, Y.; Liu, H.; Liu, M.; Shi, C.; Qian, L. Knockdown of FABP3 Impairs Cardiac Development in Zebrafish through the Retinoic Acid Signaling Pathway. Int. J. Mol. Sci. 2013, 14, 13826-13841. https://doi.org/10.3390/ijms140713826
Wang X, Zhou L, Jin J, Yang Y, Song G, Shen Y, Liu H, Liu M, Shi C, Qian L. Knockdown of FABP3 Impairs Cardiac Development in Zebrafish through the Retinoic Acid Signaling Pathway. International Journal of Molecular Sciences. 2013; 14(7):13826-13841. https://doi.org/10.3390/ijms140713826
Chicago/Turabian StyleWang, Xuejie, Lijuan Zhou, Jin Jin, Yang Yang, Guixian Song, Yahui Shen, Hailang Liu, Ming Liu, Chunmei Shi, and Lingmei Qian. 2013. "Knockdown of FABP3 Impairs Cardiac Development in Zebrafish through the Retinoic Acid Signaling Pathway" International Journal of Molecular Sciences 14, no. 7: 13826-13841. https://doi.org/10.3390/ijms140713826
APA StyleWang, X., Zhou, L., Jin, J., Yang, Y., Song, G., Shen, Y., Liu, H., Liu, M., Shi, C., & Qian, L. (2013). Knockdown of FABP3 Impairs Cardiac Development in Zebrafish through the Retinoic Acid Signaling Pathway. International Journal of Molecular Sciences, 14(7), 13826-13841. https://doi.org/10.3390/ijms140713826




