Activity of Laccase Immobilized on TiO2-Montmorillonite Complexes
Abstract
:1. Introduction
2. Results and Discussion
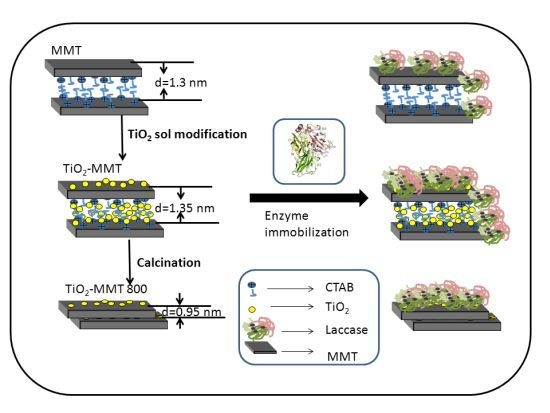
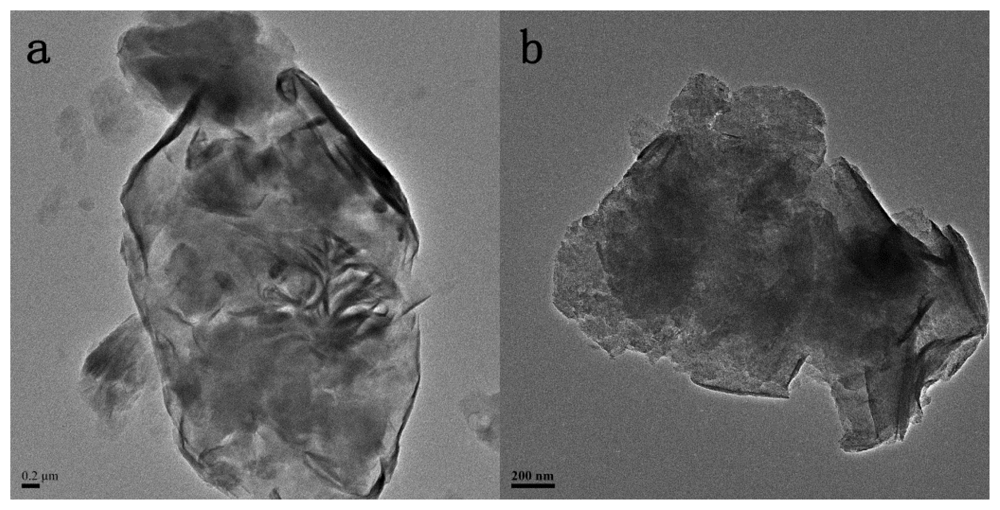
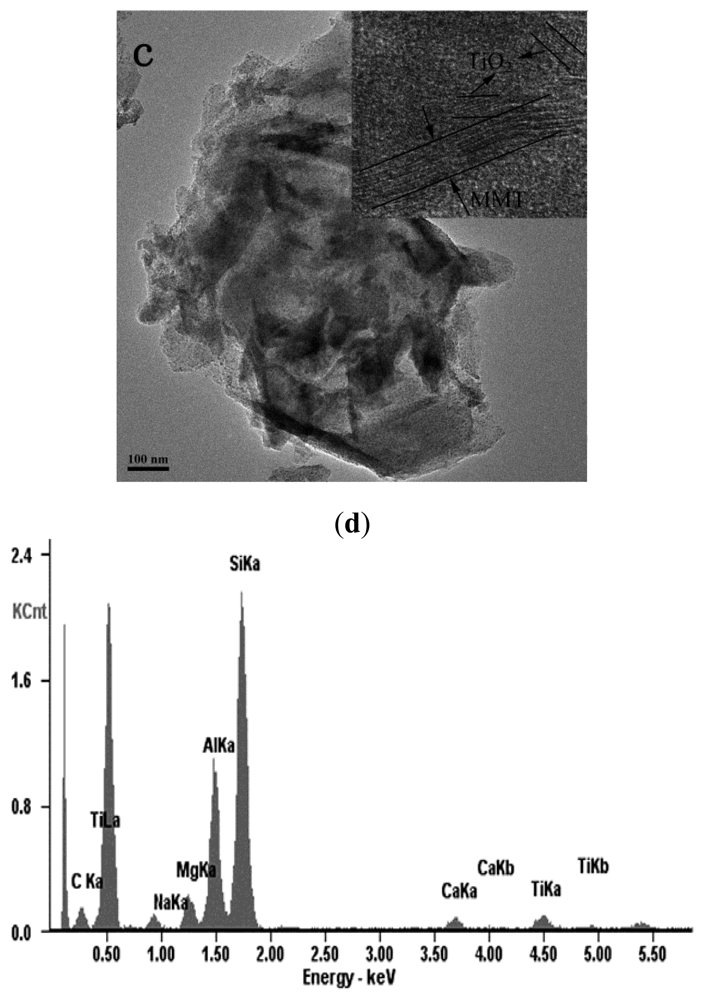
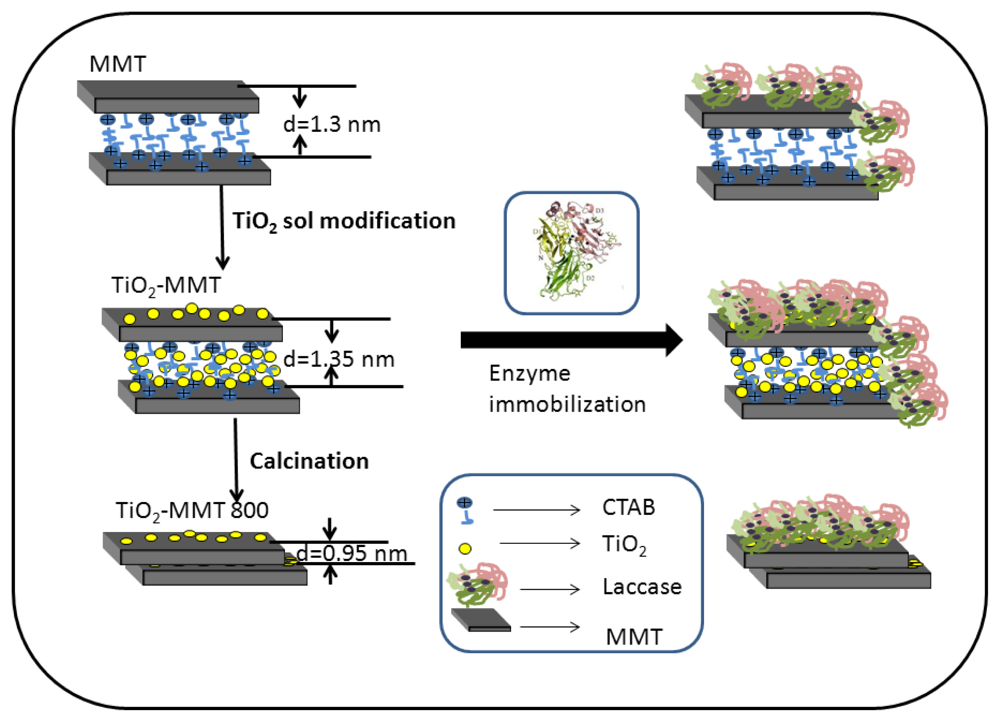
2.1. The Structures of MMT and TiO2-MMT Complexes
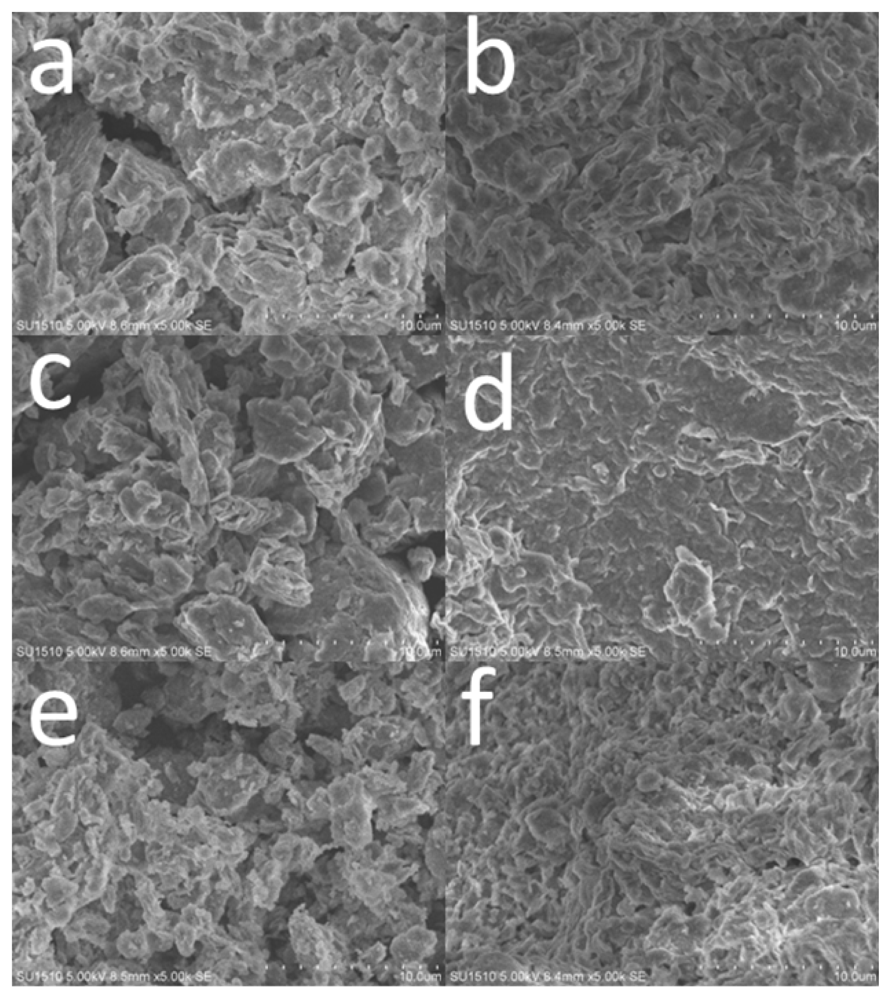
2.2. Surface Morphology of the Nanohybrids before and after Enzyme Immobilization
2.3. Kinetic Properties of Immobilized Enzyme
2.4. The Distribution of Immobilized Laccase on MMT and TiO2-MMT Complexes
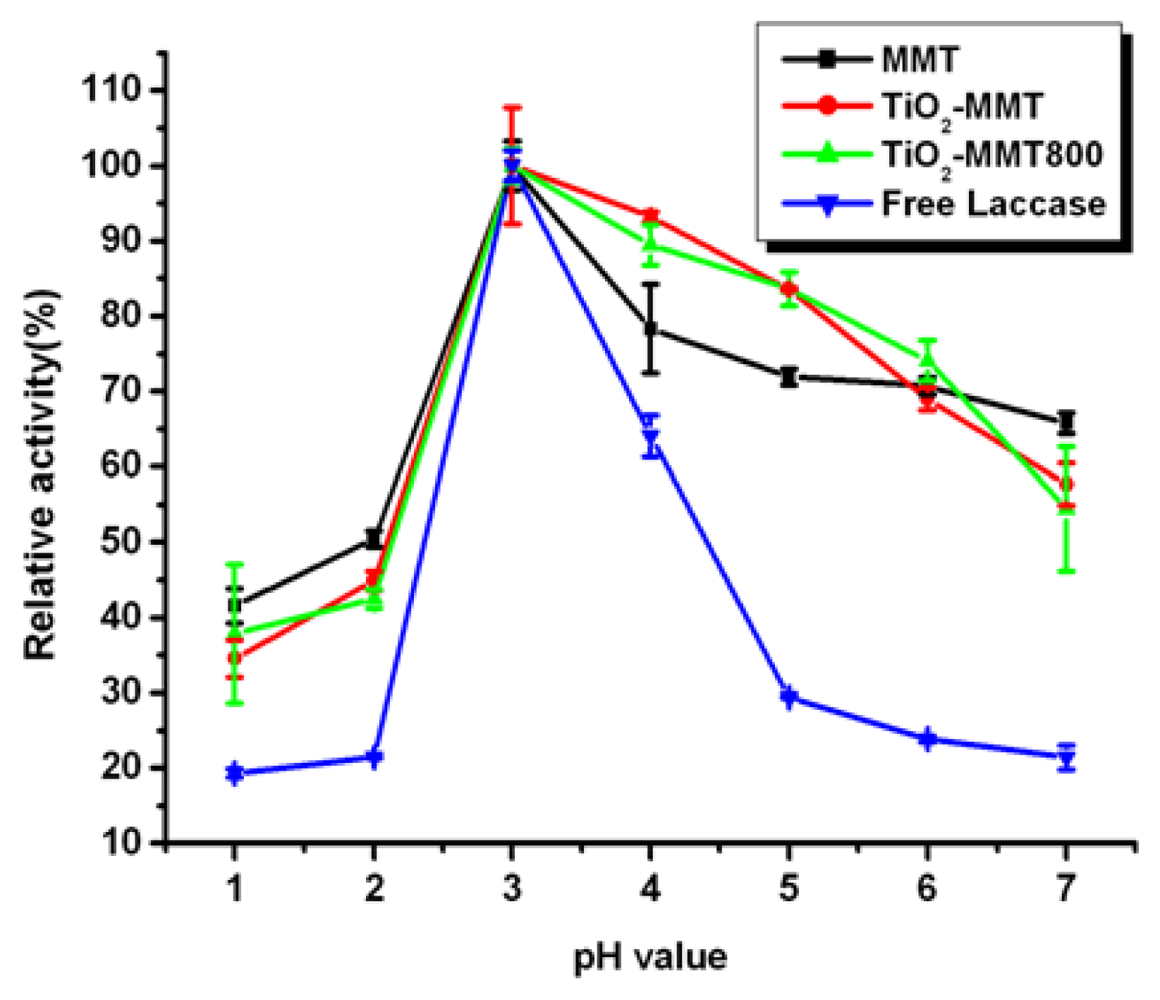
2.5. Effect of pH on Free and Immobilized Laccase
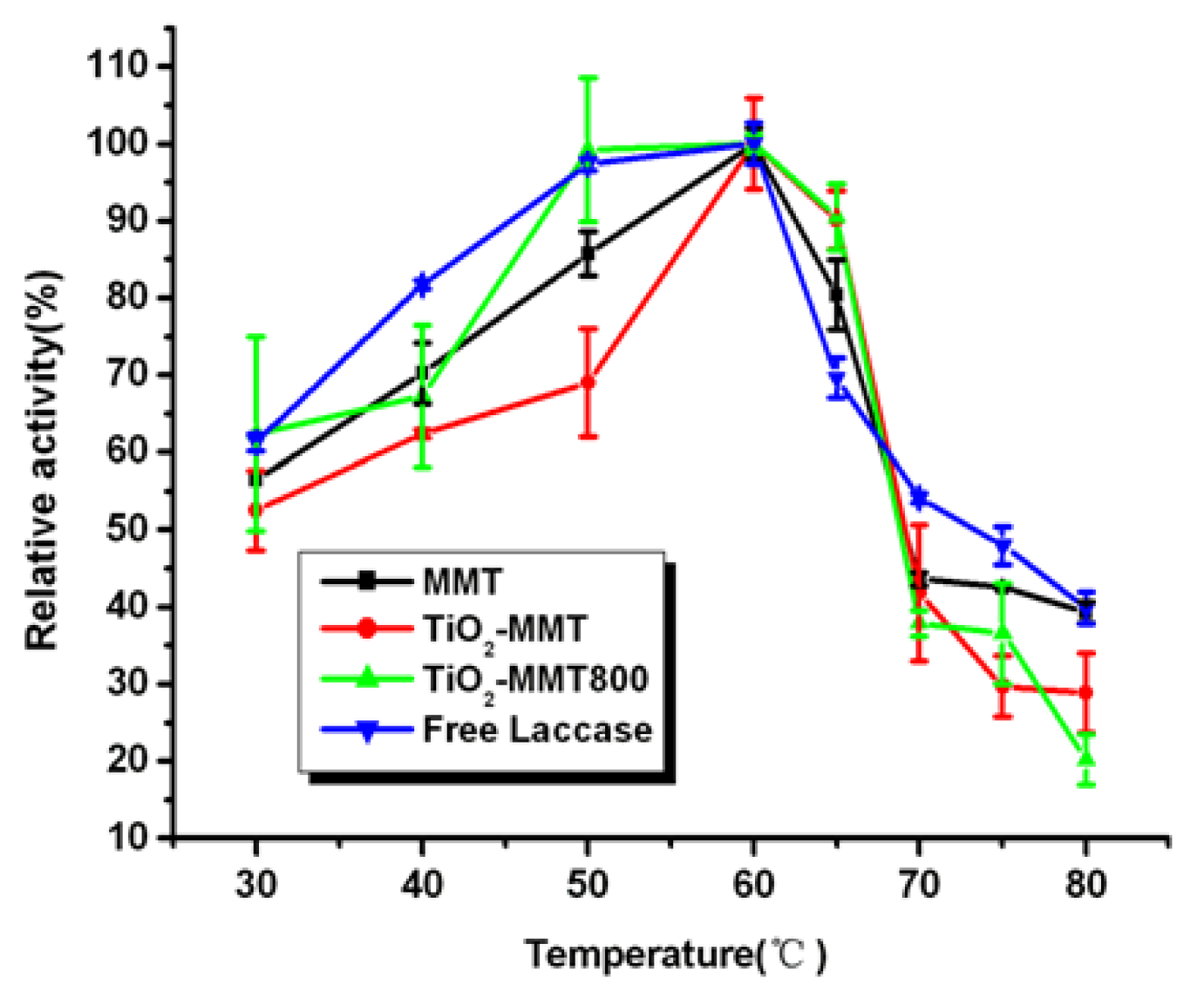
2.6. Effect of Temperature on the Activity of Free and Immobilized Laccase
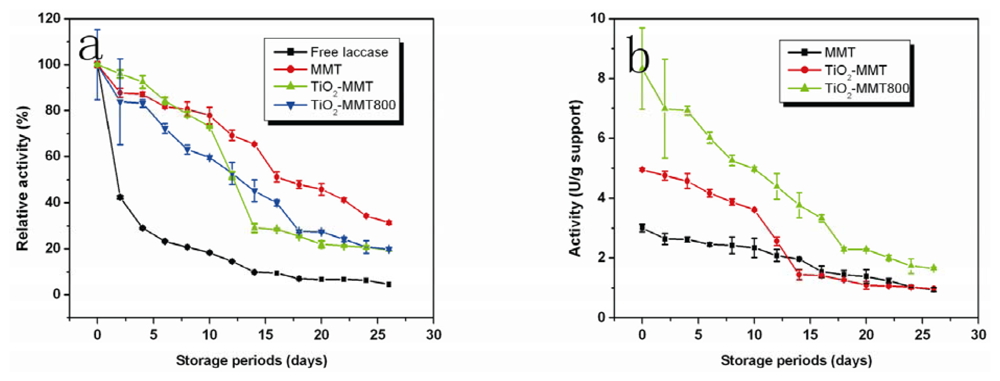
2.7. Storage Stability
3. Experimental Section
3.1. Materials
3.2. Preparation of TiO2–MMT Complexes
3.3. Laccase Immobilization by Physical Adsorption
3.4. Structural Characterization
3.5. Protein Concentration and Laccase Activity
3.5.1. Protein Concentration
3.5.2. Activity Assay of Free and Immobilized Laccase
3.6. Kinetic Parameters
3.7. Effect of Temperature and pH on the Activity of Immobilized and Free Enzyme
3.8. Storage Stability
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Mayer, A.M.; Staples, R.C. Laccase: New functions for an old enzyme. Phytochemistry 2002, 60, 551–565. [Google Scholar]
- Jia, J.B.; Zhang, S.P.; Wang, P.; Wang, H.J. Degradation of high concentration 2,4-dichlorophenol by simultaneous photocatalytic-enzymatic process using TiO2/UV and laccase. J. Hazard. Mater 2012, 205, 150–155. [Google Scholar]
- Brunel, L.; Denele, J.; Servat, K.; Kokoh, K.B.; Jolivalt, C.; Innocent, C.; Cretin, M.; Rolland, M.; Tingry, S. Oxygen transport through laccase biocathodes for a membrane-less glucose/O2 biofuel cell. Electrochem. Commun 2007, 9, 331–336. [Google Scholar]
- Wu, X.M.; Hu, Y.J.; Jin, J.; Zhou, N.L.; Wu, P.; Zhang, H.; Cai, C.X. Electrochemical approach for detection of extracellular oxygen released from erythrocytes based on graphene film integrated with laccase and 2,2-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid). Anal. Chem 2010, 82, 3588–3596. [Google Scholar]
- Feng, Q.A.; Xia, X.; Wei, A.F.; Wang, X.Q.; Wei, Q.F.; Huo, D.Y.; Wei, A.J. Preparation of Cu(II)-chelated poly(vinyl alcohol) nanofibrous membranes for catalase immobilization. J. Appl. Polym. Sci 2011, 120, 3291–3296. [Google Scholar]
- Dai, Y.R.; Niu, J.F.; Liu, J.; Yin, L.F.; Xu, J.J. In situ encapsulation of laccase in microfibers by emulsion electrospinning: Preparation, characterization, and application. Bioresour. Technol 2010, 101, 8942–8947. [Google Scholar]
- Kanwar, S.S.; Pathak, S.; Verma, H.K.; Kumar, S.; Gupta, R.; Chimni, S.S.; Chauhan, G.S. Characteristics of poly(aac(5)-co-hpma(3)-cl-egdma(15)) hydrogel-immobilized lipase of pseudomonas aeruginosa mtcc-4713. J. Appl. Polym. Sci 2006, 100, 4636–4644. [Google Scholar]
- Kim, B.C.; Zhao, X.Y.; Ahn, H.K.; Kim, J.H.; Lee, H.J.; Kim, K.W.; Nair, S.; Hsiao, E.; Jia, H.F.; Oh, M.K.; et al. Highly stable enzyme precipitate coatings and their electrochemical applications. Biosens. Bioelectron 2011, 26, 1980–1986. [Google Scholar]
- Jia, H.F.; Zhu, G.Y.; Wang, P. Catalytic behaviors of enzymes attached to nanoparticles: The effect of particle mobility. Biotechnol. Bioeng 2003, 84, 406–414. [Google Scholar]
- Wang, F.; Manku, S.; Hall, D.G. Solid phase syntheses of polyamine toxins HO-416b and PHTX-433. Use of an efficient polyamide reduction strategy that facilitates access to branched analogues. Org. Lett 2000, 2, 1581–1583. [Google Scholar]
- Verma, M.L.; Barrow, C.J.; Puri, M. Nanobiotechnology as a novel paradigm for enzyme immobilisation and stabilisation with potential applications in biodiesel production. Appl. Microbiol. Biotechnol 2013, 97, 23–39. [Google Scholar]
- Garcia-Galan, C.; Berenguer-Murcia, A.; Fernandez-Lafuente, R.; Rodrigues, R.C. Potential of different enzyme immobilization strategies to improve enzyme performance. Adv. Synth. Catal 2011, 353, 2885–2904. [Google Scholar]
- Gopinath, S.; Sugunan, S. Enzymes immobilized on montmorillonite K 10: Effect of adsorption and grafting on the surface properties and the enzyme activity. Appl. Clay Sci 2007, 35, 67–75. [Google Scholar]
- Chen, G.J.; Yen, M.C.; Wang, J.M.; Lin, J.J.; Chiu, H.C. Layered inorganic/enzyme nanohybrids with selectivity and structural stability upon interacting with biomolecules. Bioconjug. Chem 2008, 19, 138–144. [Google Scholar]
- Naidja, A.; Huang, P.M.; Bollag, J.M. Activity of tyrosinase immobilized on hydroxyaluminum-montmorillonite complexes. J. Mol. Catal 1997, 115, 305–316. [Google Scholar]
- Farre, M.J.; Maldonado, M.I.; Gernjak, W.; Oller, I.; Malato, S.; Domenech, X.; Peral, J. Coupled solar photo-fenton and biological treatment for the degradation of diuron and linuron herbicides at pilot scale. Chemosphere 2008, 72, 622–629. [Google Scholar]
- Oller, I.; Malato, S.; Sanchez-Perez, J.A.; Gernjak, W.; Maldonado, M.L.; Perez-Estrada, L.A.; Pulgarin, C. A combined solar photocatalytic-biological field system for the mineralization of an industrial pollutant at pilot scale. Catal. Today 2007, 122, 150–159. [Google Scholar]
- Guieysse, B.; Viklund, G. Sequential UV-biological degradation of polycyclic aromatic hydrocarbons in two-phases partitioning bioreactors. Chemosphere 2005, 59, 369–376. [Google Scholar]
- Hwang, E.T.; Gu, M.B. Enzyme stabilization by nano/microsized hybrid materials. Eng. Life Sci 2013, 13, 49–61. [Google Scholar]
- Zhang, G.K.; Ding, X.M.; He, F.S.; Yu, X.Y.; Zhou, J.; Hu, Y.J.; Xie, J.W. Low-temperature synthesis and photocatalytic activity of TiO2 pillared montmorillonite. Langmuir 2008, 24, 1026–1030. [Google Scholar]
- Pluta, M.; Galeski, A.; Alexandre, M.; Paul, M.A.; Dubois, P. Polylactide/montmorillonite nanocomposites and microcomposites prepared by melt blending: Structure and some physical properties. J. Appl. Polym. Sci 2002, 86, 1497–1506. [Google Scholar]
- Makas, Y.G.; Kalkan, N.A.; Aksoy, S.; Altinok, H.; Hasirci, N. Immobilization of laccase in kappa-carrageenan based semi-interpenetrating polymer networks. J. Biotechnol 2010, 148, 216–220. [Google Scholar]
- Lu, L.; Zhao, M.; Wang, Y. Immobilization of laccase by alginate-chitosan microcapsules and its use in dye decolorization. World J. Microbiol. Biotechnol 2007, 23, 159–166. [Google Scholar]
- Lee, H.C.; Lin, J.L.; Chin, Y.L.; Sun, T.P. Comparison of carbon and platinum performance as TiO2 membrane substrate material and applied to glucose biosensor using amperometric readout circuit. Sens. Lett 2008, 6, 1001–1004. [Google Scholar]
- Wang, Q.Q.; Gao, D.W.; Gao, C.T.; Wei, Q.F.; Cai, Y.B.; Xu, J.; Liu, X.Y.; Xu, Y. Removal of a cationic dye by adsorption/photodegradation using electrospun PAN/O-MMT composite nanofibrous membranes coated with TiO2. Int. J. Photoenergy 2012, 2012. [Google Scholar] [CrossRef]
| Sample | d (nm) | Langmuir surface area (m2/g) | Adsorbed protein (mg.g−1) | Specific activity (U.mg−1) | Activity retention (%) | Km (mM) | Vmax (μmol/mg protein min) |
|---|---|---|---|---|---|---|---|
| Free laccase | 5.657 | 100 | 0.257 | 62.112 | |||
| Pristine MMT | 1.299 | 77.049 | 27.178 | 2.951 | 52.16 | 5.706 | 4.931 |
| TiO2-MMT | 1.355 | 99.003 | 35.896 | 4.227 | 74.72 | 3.493 | 5.548 |
| TiO2-MMT800 | 0.952 | 32.128 | 33.332 | 5.004 | 88.46 | 1.303 | 7.010 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, Q.; Peng, L.; Li, G.; Zhang, P.; Li, D.; Huang, F.; Wei, Q. Activity of Laccase Immobilized on TiO2-Montmorillonite Complexes. Int. J. Mol. Sci. 2013, 14, 12520-12532. https://doi.org/10.3390/ijms140612520
Wang Q, Peng L, Li G, Zhang P, Li D, Huang F, Wei Q. Activity of Laccase Immobilized on TiO2-Montmorillonite Complexes. International Journal of Molecular Sciences. 2013; 14(6):12520-12532. https://doi.org/10.3390/ijms140612520
Chicago/Turabian StyleWang, Qingqing, Lin Peng, Guohui Li, Ping Zhang, Dawei Li, Fenglin Huang, and Qufu Wei. 2013. "Activity of Laccase Immobilized on TiO2-Montmorillonite Complexes" International Journal of Molecular Sciences 14, no. 6: 12520-12532. https://doi.org/10.3390/ijms140612520
APA StyleWang, Q., Peng, L., Li, G., Zhang, P., Li, D., Huang, F., & Wei, Q. (2013). Activity of Laccase Immobilized on TiO2-Montmorillonite Complexes. International Journal of Molecular Sciences, 14(6), 12520-12532. https://doi.org/10.3390/ijms140612520