Plant Defense against Insect Herbivores
Abstract
:1. Introduction
2. Plant/Insect Interactions Induce Early Signaling
2.1. Recognition of Insect Herbivore Attack
2.1.1. Feeding Guilds
2.1.2. Insect Oral Secretions
2.1.3. Oviposition Fluids
2.2. Early Events in the Plant-Insect Interaction
2.2.1. Electrical Signaling
2.2.2. Ca2+ Homeostasis
2.2.3. Signal Transduction Involving Kinases
3. Intracellular Wound Signals
3.1. Systemic Signaling
3.1.1. Systemin and Systemin-Like Peptides
3.1.2. Other Signaling Mechanisms
3.2. Regulation of Defense Responses by Jasmonates
3.2.1. Jasmonate Biosynthesis
3.2.2. Jasmonate Signaling
3.2.3. Jasmonate Regulation of Defense Response
3.3. Oligogalacturonic Acid
3.4. Hydrogen Peroxide
4. Defense Responses
4.1. Direct Defense Response
4.1.1. Bioactive Specialized Compounds
4.1.1.1. Alkaloids
4.1.1.2. Benzoxazinoides
4.1.1.3. Cyanogenic Glucosides
4.1.1.4. Glucosinolates
4.1.1.5. Nonprotein Amino Acids
4.1.1.6. Phenolics
4.1.1.7. Terpenoids
4.1.2. Digestibility Reduction
4.1.2.1. α-Amylase Inhibitors
4.1.2.2. Chitinases
4.1.2.3. Lectins
4.1.2.4. Polyphenol Oxidases
4.1.2.5. Proteinase Inhibitors
4.1.3. Reallocation of Resources
4.1.4. Morphological Features
4.1.4.1. Waxes and Crystals
4.1.4.2. Trichomes
4.1.4.3. Leaf and Root Toughness and Quantity
4.1.4.4. Laticifers and Oleoresins
4.2. Indirect Defense Response
4.2.1. Volatiles
4.2.2. Extrafloral Nectar
4.2.3. Food Bodies
4.2.4. Nesting and Refuge Sites
5. Conclusions and Future Perspectives
Acknowledgments
Conflict of Interest
References
- Ehrlich, P.R.; Raven, P.H. Butterflies and plants: A study in coevolution. Evolution 1964, 18, 586–608. [Google Scholar]
- Labandeira, C.C. Insect mouthparts: Ascertaining the paleobiology of insect feeding strategies. Annu. Rev. Ecol. Syst 1997, 28, 153–193. [Google Scholar]
- Niklas, K.J.; Tiffney, B.H.; Knoll, A.H. Patterns in Vascular Land Plant Diversification: An Analysis at the Species Level. In Phanerozoic Diversity Patterns: Profiles in Macroevolution; Valentine, J.W., Ed.; Princeton University Press: Princeton, NJ, USA, 1985; pp. 97–128. [Google Scholar]
- Schönenberger, J.; Friis, E.M.; Matthews, M.L.; Endress, P.K. Cunoniaceae in the cretaceous of europe: Evidence from fossil flowers. Ann. Bot 2001, 88, 423–437. [Google Scholar]
- Schaller, A.; Howe, G.A. Direct Defenses in Plants and Their Induction by Wounding and Insect Herbivores. In Induced Plant Resistance to Herbivory; Schaller, A., Ed.; Springer Science+Business Media: Berlin, Germany, 2008; pp. 7–29. [Google Scholar]
- Da Costa, C.P.; Jones, C.M. Cucumber beetle resistance controlled by the bitter gene in Cucumis sativa L. Science 1971, 172, 1145–1146. [Google Scholar]
- Feeny, P. Plant Apparency and Chemical Defense. In Biochemical Interaction between Plants and Insects; Wallace, I., Mansell, R.L., Eds.; Plenum Press: New York, NY, USA, 1976; Volume 1–40. [Google Scholar]
- Chew, F.S.; Renwick, J.A.A. Host Plant Choice in Pieris Butterflies. In Chemical Ecology of Insects II; Cardé, R.T., Bell, W.J., Eds.; Chapman and Hall: New York, NY, USA, 1994; pp. 214–238. [Google Scholar]
- Ali, J.G.; Agrawal, A.A. Specialist versus generalist insect herbivores plant defense. Trends Plant Sci 2012, 17, 293–302. [Google Scholar]
- Baldwin, I.T. An ecologically motivated analysis of plant-herbivore interactions in native tobacco. Plant Physiol 2001, 127, 1449–1458. [Google Scholar]
- Rasmann, S.; Agrawal, A.A. In defense of roots: A research agenda for studying plant resistance to belowground herbivory. Plant Physiol 2008, 146, 875–880. [Google Scholar]
- Whittaker, J.B. Root-animal Interactions. In Root Ecology; de Kroon, H., Visser, E.J.W., Eds.; Springer-Verlag: Berlin/Heidelberg, Geramny, 2003; pp. 367–386. [Google Scholar]
- Hajek, A.E.; St. Leger, R.J. Interactions between fungal pathogens and insect hosts. Annu. Rev. Entomol 1994, 39, 293–322. [Google Scholar]
- Guest, D.; Brown, J. Plant Defences against Pathogens. In Plant Pathogens and Plant Diseases; Brown, J.F., Ogle, H.J., Eds.; Rockvale Publications for the Division of Botany, Rockvale Publications for the Division of Botany, School of Rural Science and Natural Resources, University of New England: Armidale New South Wales, New England, UK, 1997. [Google Scholar]
- Dangl, J.L.; Jones, J.D.G. Plant pathogens and integrated defence responses to infection. Nature 2001, 441, 826–833. [Google Scholar]
- Odjakova, M.; Hadjiivanova, C. The complexity of pathogen defense in plants. Bulg. J. Plant Physiol 2001, 27, 101–109. [Google Scholar]
- Heil, M.; Bostock, R.M. Induced systemic resistance against pathogens in the context of induced plant defences. Ann. Bot 2002, 89, 503–512. [Google Scholar]
- Cao, F.Y.; Yoshioka, K.; Desveaux, D. The roles of ABA in plant-pathogen interactions. J. Plant Res 2011, 124, 489–499. [Google Scholar]
- Lucas, J.A. Advances in plant disease and pest management. J. Agric. Sci 2011, 149, 91–114. [Google Scholar]
- Schoonhoven, L.M.; Jermey, T.; van Loon, J.J.A. Insect Plant-biology: From Physiology to Evolution; Chapman and Hall: London, UK, 1998. [Google Scholar]
- Houlihan, D.F. The structure and behaviour of Notiphila riparia and Erioptera squalida, two root-piercing insects. J. Zool 1969, 159, 249–267. [Google Scholar]
- Mithöfer, A.; Wanner, G.; Boland, W. Effects of feeding Spodoptera littoralis on lima bean leaves. II. Continuous mechanical wounding resembling insect feeding is sufficient to elicit herbivory-related volatile emission. Plant Physiol 2005, 137, 1160–1168. [Google Scholar]
- Arimura, G.; Ozawa, R.; Kugimiya, S.; Takabayashi, J.; Bohlmann, J. Herbivore-induced defense response in a model legume. Two-spotted spider mites induce emission of (E)-β-ocimene transcript accumulation of (E)-β-ocimene synthase in Lotus japonicus. Plant Physiol 2004, 135, 1976–1983. [Google Scholar]
- De Moraes, C.M.; Mescher, M.C.; Tumlinson, J.H. Caterpillar-induced nocturnal plant volatiles repel conspecific females. Nature 2001, 410, 577–580. [Google Scholar]
- Turlings, T.C.J.; Wackers, F.L.; Vet, L.E.M.; Lewis, W.J.; Tumlinson, J.H. Learning of Host Location Cues by Insect Parasitoids. In Insect Learning: Ecological and Evolutionary Perspectives; Papaj, D.R., Lewis, A., Eds.; Chapman and Hall: New York, NY, USA, 1993; pp. 51–78. [Google Scholar]
- Alborn, H.T.; Turlings, T.C.J.; Jones, T.H.; Stenhagen, G.; Loughrin, J.H. An elicitor of plant volatiles from beet armyworm oral secretions. Science 1997, 276, 945–949. [Google Scholar]
- Halitschke, R.; Schittko, U.; Pohnert, G.; Boland, W.; Baldwin, I.T. Molecular interactions between the specialist herbivore Manduca sexta (Lepidoptera, Sphingidae) and its natural host Nicotiana attenuata. III. Fatty acid-amino acid conjugates in herbivore oral secretions are necessary and sufficient for herbivore-specific plant responses. Plant Physiol 2001, 125, 711–717. [Google Scholar]
- Mattiacci, L.; Dicke, E.; Posthumus, M.A. β-Glucosidase—An elicitor of herbivore-induced plant odor that attracts host-searching parasitic wasps. Proc. Natl. Acad. Sci. USA 1995, 92, 2036–2040. [Google Scholar]
- Truitt, C.L.; Wei, H.X.; Pare, P.W. A plasma membrane protein from Zea mays binds with the herbivore elicitor volicitin. Plant Cell 2005, 16, 523–532. [Google Scholar]
- Frey, M.; Stettner, C.; Paré, P.W.; Schmelz, E.A.; Tumlinson, J.H.; Gierl, A. An herbivore elicitor activates the gene for indole emission in maize. Proc. Natl. Acad. Sci. USA 2000, 97, 14801–14806. [Google Scholar]
- Schmelz, E.A.; Engelberth, J.; Alborn, H.T.; Tumlinson, J.H.; Teal, P.E.A. Phytohormone-based activity mapping of insect herbivore-produced elicitors. Proc. Natl. Acad. Sci. USA 2009, 106, 653–657. [Google Scholar]
- Schmelz, E.A.; Carroll, M.J.; LeClere, S.; Phipps, S.M.; Meredith, J.; Chourey, P.S.; Alborn, H.T.; Teal, P.E.A. Fragments of ATP synthase mediate plant perception of insect attack. Proc. Natl. Acad. Sci. USA 2007, 103, 8894–8899. [Google Scholar]
- Alborn, H.T.; Hansen, T.V.; Jones, T.H.; Bennett, D.C.; Tumlinson, J.H.; Schmelz, E.A.; Teal, P.E.A. Novel disulfoxy fatty acids from the american bird grasshopper Shistocerca americana, elicitors of plant volatiles. Proc. Natl. Acad. Sci. USA 2007, 104, 12976–12981. [Google Scholar]
- O’Doherty, I.; Yim, J.J.; Schmelz, E.A.; Schroeder, F.C. Synthesis of caeliferins, elicitors of plant immune responses: Accessing lipophilic natural products via cross metathesis. Org. Lett 2011, 13, 5900–5903. [Google Scholar]
- Doss, R.P.; Oliver, J.E.; Proebsting, W.M.; Potter, S.W.; Kuy, S.R.; Clement, S.L.; Williamson, R.T.; Carney, J.R.; DeVilbiss, E.D. Bruchins: Insect-derived plant regulators that stimulate neoplasm formation. Proc. Natl. Acad. Sci. USA 2000, 97, 6218–6223. [Google Scholar]
- Bos, J.I.B.; Prince, D.; Pitino, M.; Maffei, M.E.; Win, J.; Hogenhout, S.A. A functional genomics approach identifies candidate effectors from the aphid species Myzus persicae. PLoS Genet 2010, 6, e1001216. [Google Scholar]
- Eichenseer, H.; Mathews, M.C.; Bi, J.L.; Murphy, J.B.; Felton, G.W. Salivary glucose oxidase: Multifunctional roles for Helicoverpa zea? Arch. Insect Biochem. Physiol 1999, 42, 1520–1527. [Google Scholar]
- Erb, M. Modification of Plant Resistance and Metabolism by Above- and Belowground Herbivores. Ph.D. Dissertation, Université de Neuchâtel, Neuchâtel, Swizerland, 2009. [Google Scholar]
- Erb, M.; Glauser, G.; Robert, C.A.M. Induced immunity against belowground insect herbivores—Activation of defenses in the absence of a jasmonate burst. J. Chem. Ecol 2012, 38, 629–640. [Google Scholar]
- Hilker, M.; Meiners, T. Early herbivore alert: Insect eggs induce plant defense. J. Chem. Ecol 2006, 32, 1379–1397. [Google Scholar]
- Schroder, R.; Cristescu, S.M.; Harren, F.J.; Hilker, M. Reduction of ethylene emission from scots pine elicited by insect egg secretion. J. Exp. Bot 2007, 58, 1835–1842. [Google Scholar]
- Little, D.; Gouhier-Darimont, C.; Bruessow, F.; Reymoind, P. Oviposition by pierid butterflies triggers defense responses in Arabidopsis. Plant Physiol 2007, 143, 784–800. [Google Scholar]
- Fatouros, N.E.; Bukovinszkine’Kiss, G.; Kalkers, L.A.; Gamborena, R.S.; Dicke, M.; Hilker, M. Oviposition-induced plant cues: Do they arrest Trichogramma wasps during host location? Entomol. Exp. Appl 2005, 115, 207–215. [Google Scholar]
- Mithöfer, A.; Boland, W. Recognition of herbivory-associated molecular patterns. Plant Physiol 2008, 146, 825–831. [Google Scholar]
- Ebel, J.; Mithöfer, A. Early events in the elicitation of plant defence. Planta 1998, 206, 335–348. [Google Scholar]
- Shabala, S. Non-invasive Microelectrode ion Flux Measurements in Plant Stress Physiology. In Plant Electrophysiology—Theory and Methods; Volkov, A., Ed.; Springer-Verlag: Berlin, Germany, 2006; pp. 35–37. [Google Scholar]
- Maffei, E.M.; Bossi, S. Electrophysiology and Plant Responses to Biotic Stress. In Plant Electrophysiology—Theory and Methods; Volkov, A., Ed.; Springer-Verlag: Berlin, Germany, 2006; pp. 461–481. [Google Scholar]
- Volkov, A.; Brown, C.L. Electrochemistry of Plant Life. In Plant Electrophysiology—Theory and Methods; Volkov, A., Ed.; Springer-Verlag: Berlin, Germany, 2006; pp. 437–459. [Google Scholar]
- Maffei, E.M.; Mithöfer, A.; Boland, W. Before gene expression: Early events in plant-insect interaction. Trends Plant Sci 2007, 7, 310–316. [Google Scholar]
- Peiffer, M.; Felton, G.W. The host plant as a factor in the synthesis and secretion of salvary glucose oxidase in the larval Helicoverpa zea. Arch. Insect Biochem 2005, 58, 128–137. [Google Scholar]
- Maffei, E.M.; Bossi, S.; Spiteller, D.; Mithöfer, A.; Boland, W. Effects of feeding Spodoptera littoralis on lima bean leaves. I. Membrane potentials, intracellular calcium variations, oral secretions, and regurgitate components. Plant Physiol 2004, 134, 1752–1762. [Google Scholar]
- Maischak, H.; Grigoriev, P.; Vogel, H.; Boland, W.; Mithöfer, A. Oral secretions from herbivorous lepidopteran larvae exhibit ion channel-forming activities. FEBS Lett 2007, 581, 898–904. [Google Scholar]
- Verret, F.; Wheeler, G.; Taylor, A.R.; Farnham, G.; Brownlee, C. Calcium channels in photosynthetic eukaryotes: Implications for evolution of calcium-based signalling. New Phytol 2010, 187, 23–43. [Google Scholar]
- Lecourieux, D.; Ranjeva, R.; Pugin, A. Calcium in the plant defence-signalling pathways. New Phytol 2006, 171, 249–269. [Google Scholar]
- Evans, N.H.; McAinsh, M.R.; Hetherington, A.M. Calcium oscillations in higher plants. Curr. Opin. Plant Biol 2001, 4, 415–420. [Google Scholar]
- Sathyanarayanan, P.V.; Poovaiah, B.W. Decoding Ca2+ signals in plant. Crit. Rev. Plant Sci 2004, 23, 1–11. [Google Scholar]
- Mohanta, T.K.; Occhipinti, A.; Atsbaha Zebelo, S.; Foti, M.; Fliegmann, J.; Boss, S.; Maffei, E.M.; Bertea, C.M. Ginkgo biloba responds to herbivory by activating early signaling and direct defences. PLoS One 2012, 7, e32822. [Google Scholar]
- Arimura, G.; Ozawa, R.; Shimoda, T.; Nishioka, T.; Boland, W.; Takabayashi, J. Herbivory-induced volatiles elicit defence genes in lima bean leaves. Nature 2000, 406, 512–515. [Google Scholar]
- Howe, G.A.; Jander, G. Plant immunity to insect herbivores. Annu. Rev. Plant Biol 2008, 59, 41–66. [Google Scholar]
- Sanders, D.; Pellouxa, J.; Brownleeb, C.; Harperc, J.F. Calcium at the crossroads of signalling. Plant Cell 2002, 14, S401–S417. [Google Scholar]
- Ludwig, A.A.; Saitoh, H.; Felix, G.; Freymark, G.; Miersch, O.; Wasternack, C.; Boller, T.; Jones, J.D.G.; Romeis, T. Ethylene-mediated cross-talk between calcium-dependent protein kinase and MAPK signaling controls stress responses in plants. Proc. Natl. Acad. Sci. USA 2005, 102, 10736–10741. [Google Scholar]
- Wu, J.; Hettenhausen, C.; Meldau, S.; Baldwin, I.T. Herbivory rapidly activates MAPK signaling in attacked and unattacked leaf regions but not between leaves of Nicotiana attenuata. Plant Cell 2007, 19, 1096–1122. [Google Scholar]
- Arimura, G.; Garms, S.; Maffei, M.; Bossi, S.; Schulze, B.; Leitner, M.; Mithöfer, A.; Boland, W. Herbivore-induced terpenoid emission in Medicago truncatula: Concerted action of jasmonate, ethylene and calcium signalling. Planta Med 2008, 227, 453–464. [Google Scholar]
- Seo, S.; Okamoto, M.; Seto, H.; Ishizuka, K.; Sano, H.; Ohashi, Y. Tobacco MAP kinase—A possible mediator in wound signal-transduction pathways. Science 1995, 270, 1988–1992. [Google Scholar]
- Ryan, C.A. The systemin signalling pathway: Differential activation of plant defensive genes. Biochim. Biophys. Acta 2000, 1477, 112–121. [Google Scholar]
- Colcombet, J.; Hirt, H. Arabidopsis mapks: A complex signalling network involved in multiple biological processes. Biochem. J 2008, 413, 217–226. [Google Scholar]
- Seo, S.; Asno, H.; Ohashi, Y. Jasmonate-based wound signal transduction requires activation of wipk, a tobacco mitogen-activated protein kinase. Plant Cell 1999, 11, 289–298. [Google Scholar]
- Arimura, G.I.; Ozawa, R.; Maffei, M.E. Recent advances in plant early signaling in response to herbivory. Int. J. Mol. Sci 2012, 12, 3723–3739. [Google Scholar]
- Arimura, G.I.; Matsui, K.; Takabayashi, J. Chemical and molecular ecology of herbivore-induced plant volatiles: Proximate factors and their ultimate functions. Plant Cell Physiol 2009, 50, 911–923. [Google Scholar]
- Doares, S.H.; Syrovets, T.; Weiler, E.W.; Ryan, C.A. Oligogalacturonides and chitosan activate plant defensive genes through the octadecanoid pathway. Proc. Natl. Acad. Sci. USA 1995, 92, 4095–4098. [Google Scholar]
- Bostock, R.M.; Karban, R.; Thaler, J.S.; Weyman, P.D.; Gilchrist, D. Signal interactions in induced resistance to pathogens and insect herbivores. Eur. J. Plant Pathol 2001, 107, 103–111. [Google Scholar]
- Von Dahl, C.; Baldwin, I.T. Deciphering the role of ethylene in plant-herbivore interactions. J. Plant Growth Regul 2007, 26, 201–209. [Google Scholar]
- Kahl, J.; Siemens, D.H.; Aerts, R.J.; Gäbler, R.; Kühnemann, F.; Preston, C.A.; Baldwin, I.T. Herbivore-induced ethylene suppresses a direct defense but not a putative indirect defense against an adapted herbivore. Planta 2000, 210, 336–342. [Google Scholar]
- Green, T.R.; Ryan, C.A. Wound-induced proteinase inhibitor in plant leaves: A possible defense mechanism against insects. Science 1972, 175, 776–777. [Google Scholar]
- Pearce, G.; Strydom, D.; Johnson, S.; Ryan, C.A. A polypeptide from tomato leaves induces woung-inducible proteinase inhibitor proteins. Science 1991, 253, 895–898. [Google Scholar]
- Ryan, C.A.; Pearce, G. Polypeptide hormones. Plant Physiol 2001, 125, 65–68. [Google Scholar]
- McGurl, B.; Pearce, G.; Orozco-Cárdenas, M.; Ryan, C.A. Structure, expression and antisense inhibition of the systemin precursor gene. Science 1992, 255, 1570–1573. [Google Scholar]
- Ryan, C.A.; Pearce, G. Systemins: A functionally defined family of peptide signal that regulate defensive genes in solanaceae species. Proc. Natl. Acad. Sci. USA 2003, 100, 14577–14580. [Google Scholar]
- Bergey, D.R.; Orozco-Cardenas, M.; de Moura, D.S.; Ryan, C.A. A wound- and systemin-inducible polygalacturonase in tomato leaves. Proc. Natl. Acad. Sci. USA 1999, 96, 1756–1760. [Google Scholar]
- Farmer, E.E.; Ryan, C.A. Interplant communication: Airborne methyl jasmonate induces synthesis of proteinase inhibitors in plant leaves. Proc. Natl. Acad. Sci. USA 1990, 87, 7713–7716. [Google Scholar]
- Farmer, E.E.; Johnson, R.R.; Ryan, C.A. Regulation of expression of proteinase inhibitor genes by methyl jasmonate and jasmonic acid. Plant Physiol 1991, 98, 995–1002. [Google Scholar]
- Farmer, E.E.; Ryan, C.A. Octadecanoid precursors of jasmonic acid activate the synthesis of wound-inducible proteinase inhibitors. Plant Cell 1992, 4, 129–134. [Google Scholar]
- Gatehouse, J.A. Plant resistance towards insect herbivores: A dynamic interaction. New Phytol 2002, 156, 145–169. [Google Scholar]
- Falco, M.C.; Marbach, P.A.S.; Pompermayes, P.; Lopes, F.C.C.; Silva-Filho, M.C. Mechanisms of sugarcane response to herbivory. Genet. Mol. Biol 2001, 24, 113–122. [Google Scholar]
- Ryan, C.A.; Pearce, G. Systemin: A polypeptide signalling in the wound response. Annu. Rev. Cell Dev. Biol 1998, 14, 1–17. [Google Scholar]
- Jacinto, T.; McGurl, B.; Franceschi, V.; DelanoFreier, J.; Ryan, C.A. Tomato prosystemin promoter confers wound-inducible, vascular bundle-specific expression of the β-glucuronidase gene in transgenic tomato plants. Planta 1997, 203, 406–412. [Google Scholar]
- Narváez-Vásquez, J.; Ryan, C.A. The cellular localization of prosystemin: A functional role for phloem parenchyma in systemic wound signalling. Planta 2004, 218, 360–369. [Google Scholar]
- Schilmiller, A.L.; Howe, G.A. Systemic signalling in the wound response. Curr. Opin. Plant Biol 2005, 8, 369–377. [Google Scholar]
- Scheer, J.M.; Ryan, C.A. A 160-kd systemin receptor on the surface of Lycopersicon peruvianum suspension-cultured cells. Plant Cell 1999, 11, 1525–1536. [Google Scholar]
- Narváez-Vásquez, J.; Orozco-cárdenas, M.L.; Pearce, G. Plant Peptide Signals. In Amino Acids, Peptides and Proteins in Organic Chemistry; Wiley-VCH: Weinheim, Germany, 2009; pp. 597–629. [Google Scholar]
- Pearce, G. Systemin, hydroxyproline-rich systemin and the induction of protease inhibitors. Curr. Protein Pept. Sci 2011, 12, 399–408. [Google Scholar]
- Narváez-Vásquez, J.; Orozco-Cárdenas, M.L. Systemins and Atpeps: Defense-related Peptide Signals. In Induced Plant Resistance to Herbivory; Schaller, A., Ed.; Springer: Berlin, Germany, 2008; pp. 313–328. [Google Scholar]
- Pearce, G.; Ryan, C.A. Systemic signaling in tomato plants for defense against herbivores: Isolation and characterization of three novel defense-signaling glycopeptide hormones coded in a single precursor gene. J. Biol. Chem 2003, 278, 30044–30050. [Google Scholar]
- Pearce, G.; Moura, D.S.; Stratmann, J.; Ryan, C.A. Production of multiple plant hormones from a single polyprotein precursor. Nature 2001, 411, 817–820. [Google Scholar]
- Umemoto, N.; Kakitani, M.; Iwamatsu, A.; Yoshikawa, M.; Yamaoka, N.; Ishida, L. The structure and function of a soya bean β-glucan-elicitor-binding protein. Proc. Natl. Acad. Sci. USA 1997, 94, 1029–1034. [Google Scholar]
- Schmidt, W.E.; Ebel, J. Specific binding of a fungal glucan phytoalexin elicitor to membrane fractions from soya bean Glycine max. Proc. Natl. Acad. Sci. USA 1987, 84, 4117–4121. [Google Scholar]
- Cheong, J.J.; Birberg, W.; Fügedi, P.; Pilotti, Å.; Garegg, P.J.; Hong, N.; Ogawa, T.; Hahn, M.G. A specific, high-affinity binding site for the hepta-β-glucoside elicitor exists in soya bean membranes. Plant Cell 1991, 3, 127–136. [Google Scholar]
- Yoshikawa, M.; Yamauchi, K.; Masago, H. Release of a soluble phytoalexin elicitor from mycelial walls of Phytophthora megasperma var. Sojae by soya bean tissues. Plant Cell Physiol 1993, 34, 1229–1237. [Google Scholar]
- Fletcher, J.C.; Brandu, U.; Running, M.P.; Simon, R.; Meyerowitz, E.M. Signaling of cell fate decisions by CLAVATA3 in Arabidopsis shoot meristems. Science 2000, 283, 1911–1914. [Google Scholar]
- Li, J.; Chory, J. A putative leucine-rich repeat receptor kinase involved in brassinosteroid signal transduction. Cell 1997, 90, 929–938. [Google Scholar]
- Mu, J.H.; Lee, H.S.; Kao, T.H. Characterization of a pollen-expressed receptor-like kinase gene of Petunia inflata and the activity of its encoded kinase. Plant Cell 1994, 6, 709–721. [Google Scholar]
- Schmidt, E.D.L.; Guzzo, F.; Toonen, M.A.J.; de Bries, S.C. A leucine-rich repeat containing receptor-like kinase marks somatic plant cells competent to form embryos. Development 1997, 124, 2049–2062. [Google Scholar]
- Koiwa, H.; Bressan, R.A.; Hasegawa, P.M. Regulation of protease inhibitors and plant defense. Trends Plant Sci 1997, 2, 379–384. [Google Scholar]
- Anderson, J.P.; Badruzsaufari, E.; Schenk, P.M.; Manners, J.M.; Desmond, O.J.; Ehlert, C.; Maclean, D.J.; Ebert, P.R.; Kazan, K. Antagonistic interaction between abscisic acid jasmonate-ethylene signalling pathways modulates defense gene expression disease resistance in Arabidopsis. Plant Cell 2004, 16, 3460–3479. [Google Scholar]
- Sommerville, C.; Browse, J.; Jaworski, J.G.; Ohlrogge, J.B. Lipids. In Biochemistry and Molecular Biology of Plants; Buchanan, B.B., Gruissem, W., Jones, R.L., Eds.; American Society of Plant Physiologists: Rockville, MD, USA, 2000; Volume 1, pp. 456–527. [Google Scholar]
- Pearce, G.; Yamaguchi, Y.; Barona, G.; Ryan, C.A. A subtilisin-like protein from soybean contains an embedded, cryptic signal that activates defense-related genes. Proc. Natl. Acad. Sci. USA 2010, 107, 14921–14925. [Google Scholar]
- Koo, A.J.K.; Howe, G.A. The wound hormone jasmonate. Phytochemistry 2009, 70, 1571–1580. [Google Scholar]
- Glauser, G.; Grata, E.; Dubugnon, L.; Rudaz, S.; Farmer, E.E.; Wolfender, J.L. Spatial and temporal dynamics of jasmonate synthesis and accumulation in Arabidopsis in response to wounding. J. Biol. Chem 2008, 283, 16400–16407. [Google Scholar]
- Tretner, C.; Huth, U.; Hause, B. Mechanostimulation of Medicago truncatula leads to enhanced levels of jasmonic acid. J. Exp. Bot 2008, 59, 2847–2856. [Google Scholar]
- Bonaventure, G.; VanDoorn, A.; Baldwin, I.T. Herbivore-associated elicitors: Fac signaling and metabolism. Trends Plant Sci 2011, 16, 294–299. [Google Scholar]
- Erb, M.; Flors, V.; Karlen, D.; de Lange, E.; Planchamp, C.; D’Alessandro, M.; Turlings, T.C.J.; Ton, J. Signal signature of aboveground-induced resistance upon belowground herbivory in maize. Plant J 2009, 59, 292–302. [Google Scholar]
- Schulze, B.; Ryan, L.; Mesmin Mekem, S.; Annik, S.; Wilhelm, B. Profiling of structurally labile oxylipins in plants by in situ derivatization with pentafluorobenzyl hydroxylamine. Anal. Biochem 2006, 348, 269–283. [Google Scholar]
- Schulze, B.; Dabrowska, P.; Boland, W. Rapid enzymatic isomerization of 12-oxophyto-dienoic acid in the gut of feeding lepidopteran larvae. ChemBioChem 2007, 8, 208–216. [Google Scholar]
- Hause, B.; Stenzel, I.; Miersch, O.; Maucher, H.; Kramell, R.; Ziegler, J.; Wasternack, C. Tissue-specific oxylipin signature of tomato flowers: Allene oxide cyclase is highly expressed in distinct flower organs and vascular bundles. Plant J 2000, 24, 113–126. [Google Scholar]
- Bachmann, A.; Hause, B.; Maucher, H.; Garbe, E.; Vörös, K.; Weichert, H.; Wasternack, C.; Feussner, I. Jasmonate-induced lipid peroxidation in barley leaves initiated by distinct 13-lox forms of chloroplasts. Biol. Chem 2002, 383, 1645–1657. [Google Scholar]
- Brash, A.R.; Ingram, C.D.; Harris, T.M. Analysis of a specific oxygenation reaction of soybean lipoxygenase-1 with fatty acids esterified in phospholipids. Biochemistry 1987, 26, 5467–5471. [Google Scholar]
- Dave, A.; Graham, I.A. Oxylipin signaling: A distinct role for the jasmonic acid precursor cis-12-oxo-phytodienoic acid. Front. Plant Physiol. 2012, 3, 42. [Google Scholar] [CrossRef]
- Kodama, H.; Nishiuchi, T.; Seio, S.; Ohashi, Y.; Iba, K. Possible involvement of protein phosphorylation in the wound-responsive expression of Arabidopsis plastid omega-3 fatty acid desaturase gene. Plant Sci 2000, 155, 153–160. [Google Scholar]
- Engelberth, J. Secondary Metabolites and Plant Defense. In Plant Physiology; Taiz, L., Zeiger, E., Eds.; Sinauer Associates: Sunderland, UK, 2006; Volume 4, pp. 315–344. [Google Scholar]
- Blee, E. Impact of phyto-oxylipins in plant defense. Trends Plant Sci 2002, 7, 315–321. [Google Scholar]
- Feussner, I.; Wasternack, C. The lipoxygenase pathway. Annu. Rev. Plant Biol 2002, 53, 275–297. [Google Scholar]
- Narváez-Vásquez, J.; Florin-Christensen, J.; Ryan, C.A. Positional specificity of a phospholipase a activity induced by wounding, systemin, and oligosaccharide elicitors in tomato leaves. Plant Cell 1999, 11, 2249–2260. [Google Scholar]
- Weber, H.; Vick, B.A.; Farmer, E.E. Dinor-oxo-phytodienoic acid: A new hexadecanoid signal in the jasmonate family. Proc. Natl. Acad. Sci. USA 1997, 94, 10473–10478. [Google Scholar]
- Zolman, B.K.; Silva, I.D.; Bartel, B. The Arabidopsis pxa1 mutant is defective in an ATP-binding cassette transporter-like protein required for peroxisomal fatty acid β-oxidation. Plant Physiol 2001, 127, 1266–1278. [Google Scholar]
- Hayashi, M.; Nito, K.; Takei-Hoshi, R.; Yagi, M.; Kondo, M.; Suenaga, A.; Yamaya, T.; Nishimura, M. Ped3p is a peroxisomal ATP-binding cassette transporter that might supply substrates for fatty acid β-oxidation. Plant Cell Physiolol 2002, 43, 1–11. [Google Scholar]
- Theodoulou, F.L.; Job, K.; Slocombe, S.P.; Footitt, S.; Holdsworth, M.; Baker, A.; Larson, T.R.; Graham, I.A. Jasmonoic acid levels are reduced in comatose ATP-binding cassette transporter mutants. Implications for transport of jasmonate precursors into peroxisomes. Plant Physiol 2005, 137, 835–840. [Google Scholar]
- Schaller, F.; Biesgen, C.; Müssig, C.; Altmann, T.; Weiler, E.W. 12-oxophytodienoate reductase 3 (OPR3) is the isoenzyme involved in jasmonate biosynthesis. Planta 2000, 210, 979–984. [Google Scholar]
- Koo, A.J.K.; Chung, H.S.; Kobayashi, Y.; Howe, G.A. Identification of a peroxisomal acylactivating enzyme involved in the biosynthesis of jasmonic acid in Arabidopsis. J. Biol. Chem 2006, 281, 33511–33520. [Google Scholar]
- Schaller, A.; Stintzi, A. Jasmonate Biosynthesis and Signalling for Induced Plant Defence against Herbivory. In Induced Plant Resistance to Herbivory; Schaller, A., Ed.; Springer Science+Business Media: Berlin, Germany, 2008; pp. 349–366. [Google Scholar]
- Browse, J.; Howe, G.A. New weapons and a rapid response against insect attack. Plant Physiol 2008, 146, 832–838. [Google Scholar]
- Acosta, I.; Farmer, E.E. Jasmonates. In The Arabidopsis Book; American Society of Plant Biologists: Rockville, MD, USA, 2010; Volume 8. [Google Scholar]
- Thines, B.; Katsir, L.; Melotto, M.; Niu, Y.; Mandaokar, A.; Liu, G.H.; Nomura, K.; He, S.Y.; Howe, G.A.; Browse, J. JAZ repressor proteins are targets of the scfcoi1 complex during jasmonate signalling. Nature 2007, 448, 661–662. [Google Scholar]
- Schaller, A.; Stintzi, A. Enzymes in jasmonate biosynthesis—Structure, function, regulation. Phytochemistry 2009, 70, 1532–1538. [Google Scholar]
- Hause, B.; Hause, G.; Kutter, C.; Miersch, O.; Wasternack, C. Enzymes of jasmonate biosynthesis occur in tomato sieve elements. Plant Cell Physiol 2003, 44, 643–648. [Google Scholar]
- Davis, J.M.; Gordon, M.P.; Smit, B.A. Assimilate movement dictates remote sites of wound-induced gene expression in poplar leaves. Proc. Natl. Acad. Sci. USA 1991, 88, 2393–2396. [Google Scholar]
- Farmer, E.E. Plant biology: Jasmonate perception machines. Nature 2007, 448, 659–660. [Google Scholar]
- Wasternack, C. Jasmonates: An update on biosynthesis, signal transduction and action in plant stress response, growth and development. Ann. Bot 2007, 100, 681–697. [Google Scholar]
- Yoshihara, T.; Omer, E.A.; Koshino, H.; Sakamura, S.; Kikuta, Y.; Koda, Y. Structure of a tuber-inducing stimulus from potato leaves (Solanum tuberosum l.). Agric. Biol. Chem 1989, 53, 2835–2837. [Google Scholar]
- Staswick, P.E.; Tiryaki, I. The oxylipin signal jasmonic acid is activated by an enzyme that conjugates it to isolecuine in Arabidopsis. Plant Cell 2004, 16, 2117–2127. [Google Scholar]
- Kang, J.H.; Wang, L.; Giri, A.; Baldwin, I.T. Silencing threonine deaminase and JAR4 in nicotiana attenuata impairs jasmonic acid-isoleucine-mediated defenses against Manduca sexta. Plant Cell 2006, 18, 3303–3320. [Google Scholar]
- Fonseca, S.; Chini, A.; Hamberg, M.; Adie, B.; Porzel, A.; Kramell, R.; Miersch, O.; Wasternack, C.; Solano, R. (+)-7-iso-jasmonoyl-l-isoleucine is the endogenous bioactive jasmonate. Nat. Chem. Biol 2009, 5, 344–350. [Google Scholar]
- Stuhlfelder, C.; Lottspeich, F.; Mueller, M.J. Purification and partial amino acid sequences of an esterase from tomato. Phytochemistry 2002, 60, 233–240. [Google Scholar]
- Cheong, J.J.; Choi, Y.D. Methyl jasmonate as a vital substance in plants. Trends Genet 2003, 19, 409–413. [Google Scholar]
- Boter, M.; Ruiz-Rivero, O.; Abdeen, A.; Prat, S. Conserved MYC transcription factors play a key role in jasmonate signaling both in tomato and Arabidopsis. Genes Dev. Biol 2004, 18, 1577–1591. [Google Scholar]
- Lorenzo, O.; Chico, J.M.; Sanchez-Serrano, J.J.; Solano, R. Jasmonate-insensitive1 encodes a myc transcripton factor essential to discriminate between different jasmonate-regulated defense responses in Arabidopsis. Plant Cell 2004, 16, 1938–1950. [Google Scholar]
- Staswick, P.E.; Tiryaki, I.; Rowe, M.L. Jasmonate response locus JAR1 and several related Arabidopsis genes encode enzymes of the firefly luciferase superfamily that show activity on jasmonic, salicylic, and indole-3-acetic acids in an assay for adenylation. Plant Cell 2002, 14, 1405–1415. [Google Scholar]
- Xie, D.X.; Feys, B.F.; James, S.; Nieto-Rostro, M.; Turner, J.G. Coi1: An Arabidopsis gene required for jasmonate-regulated defense and fertility. Science 1998, 280, 1091–1094. [Google Scholar]
- Chini, A.; Fonseca, S.; Fernandez, G.; Adie, B.; Chico, J.M.; Lorenzo, O.; Garcia-Casado, G.; Lopez-Vidriero, I.; Lozano, F.M.; Ponce, M.R.; et al. The JAZ family of repressors is the missing link in jasmonate signalling. Nature 2007, 448, 666–672. [Google Scholar]
- Devoto, A.; Turner, J.G. Jasmonate-regulated Arabidopsis stress signalling network. Physiol. Plant 2005, 123, 161–172. [Google Scholar]
- Chown, S.L.; Sinclair, B.J.; Leinaas, H.P.; Gaston, K.J. Hemispheric asymmetries in biodiversity—A serious matter for ecology. PLoS Biol 2004, 2, e406. [Google Scholar]
- Hou, X.; Lee, L.Y.C.; Xia, K.; Yan, Y.; Yu, H. Dellas modulate jasmonate signalling via competitive binding to jazs. Dev. Cell 2010, 19, 884–894. [Google Scholar]
- O’Donnell, P.J.; Calvert, C.; Atzorn, R.; Wasternack, C.; Leyser, H.M.O.; Bowles, D.J. Ethylene as a signal mediating the wound response of tomato plants. Science 1996, 274, 1914–1917. [Google Scholar]
- Lorenzo, O.; Piqueras, R.; Sánchez-Serrano, J.J.; Solano, R. Ethylene response factor1 integrates signals from ethylene and jasmonate pathways in plant defence. Plant Cell 2003, 15, 165–178. [Google Scholar]
- Orozco-Cárdenas, M.L.; Narvaez-Vasquez, J.; Ryan, C.A. Hydrogen peroxide acts as a second messenger for the induction of defense genes in tomato plants in response to wounding, systemin, and methyl jasmonate. Plant Cell 2001, 13, 179–191. [Google Scholar]
- Sagi, M.; Davydov, O.; Orazova, S.; Yesbergenova, Z.; Ophir, R.; Stratmann, J.W.; Fluhr, R. Plant respiratory burst oxidase homologs impinge on wound responsiveness and development in Lycopersicon esculentum. Plant Cell 2004, 16, 616–628. [Google Scholar]
- Lorenzo, O.; Solano, R. Molecular players regulating the jasmonate signalling network. Curr. Opin. Plant Biol 2005, 8, 532–540. [Google Scholar]
- Bai, Y.; Meng, Y.; Huang, D.; Qi, Y.; Chen, M. Origin and evolutionary analysis of the plant-specific tify transcription factor family. Genomics 2011, 98, 128–136. [Google Scholar]
- Oh, Y.; Baldwin, I.T.; Gális, I. Najazh regulates a subset of defense responses against herbivores and spontaneous leaf necrosis in Nicotiana attenuata plants. Plant Physiol 2012, 159, 769–788. [Google Scholar]
- Apostol, I.; Heinstein, P.F.; Low, P.S. Rapid stimulation of an oxidative burst during elicitation of cultured plant cells: Role in defense and signal transduction. Plant Physiol 1989, 90, 109–116. [Google Scholar]
- Horn, M.A.; Heinstein, P.F.; Low, P.S. Receptor-mediated endocytosis in plant cells. Plant Cell 1989, 1, 1003–1009. [Google Scholar]
- Legendre, L.; Heinstein, P.F.; Low, P.S. Evidence for participation of GTP-binding proteins in elicitation of the rapid oxidative burst in cultured soya bean cells. J. Biol. Chem 1992, 267, 20140–20147. [Google Scholar]
- Thain, J.F.; Gubb, I.R.; Wildon, D.C. Depolarization of tomato leaf cells by oligogalaturonide elicitors. Plant Cell Environ 1995, 18, 211–214. [Google Scholar]
- Chandra, S.; Low, P.S. Role of phosphorylation in elicitation of the oxidative burst in cultured soya bean cells. Proc. Natl. Acad. Sci. USA 1995, 92, 4120–4123. [Google Scholar]
- Legendre, L.; Yueh, Y.G.; Crain, R.; Haddock, N.; Heinstein, P.F.; Low, P.S. Phospholipase c activation during elicitation of the oxidative burst in cultured plant cells. J. Biol. Chem 1993, 268, 24559–24563. [Google Scholar]
- John, M.; Rohrig, H.; Schmidt, J.; Walden, R.; Schell, J. Cell signalling by oligosaccharides. Trends Plant Sci 1997, 2, 111–115. [Google Scholar]
- Orozco-Cárdenas, M.; Ryan, C.A. Hydrogen peroxide is generated systemically in plant leaves by wounding and systemin via the octadecanoid pathway. Proc. Natl. Acad. Sci. USA 1999, 96, 6553–6557. [Google Scholar]
- Shibuya, N.; Minami, E. Oligosacchraide signalling for defence responses in plant. Physiol. Mol. Plant Pathol 2001, 59, 223–233. [Google Scholar]
- Kauss, H.; Jeblick, W.; Domard, A. The degree of polymerization and N-acetylation of chitosan determine its ability to elicit callose formation in suspension cells and protoplasts of Cathalanthus roseus. Planta 1989, 178, 385–392. [Google Scholar]
- Baydoun, E.A.H.; Fry, S.C. The immobility of pectic substances in injured tomato leaves and its bearing on the identity of the wound hormone. Planta 1985, 165, 269–276. [Google Scholar]
- Lamb, C.; Dixon, R.A. The oxidative burst in plant disease resistance. Annu. Rev. Plant Physiol. Plant Mol. Biol 1997, 48, 251–275. [Google Scholar]
- Bi, J.L.; Felton, G.W. Foliar oxidative stress and insect herbivory—Primary compounds, secondary metabolites, and reactive oxygen species as components of induced resistance. J. Chem. Ecol 1995, 21, 1511–1530. [Google Scholar]
- Waetzig, G.H.; Sobczak, M.; Grundler, F.M.W. Localization of hydrogen peroxide during the defence response of Arabidopsis thaliana against the plant-parasitic nematode Heterodera glycines. Nematology 1999, 1, 681–686. [Google Scholar]
- Stennis, M.J.; Chandra, S.; Ryan, C.A.; Low, P.S. Systemin potentiates the oxidative burst in cultured tomato cells. Plant Physiol 1998, 117, 1031–1036. [Google Scholar]
- Lee, S.; Choi, H.; Suh, S.; Doo, I.S.; Oh, K.Y.; Choi, E.J.; Taylor, A.T.S.; Low, P.S.; Lee, Y. Oligogalacturonic acid and chitosan reduce stomatal aperture by inducing the evolution of reactive oxygen species from guard cells of tomato and Commelina communis. Plant Physiol 1999, 121, 147–152. [Google Scholar]
- Legendre, L.; Rueter, S.; Heinstein, P.F.; Low, P.S. Characterization of the oligogalacturonide-induced oxidative burst in cultured soya bean (Glycine max) cells. Plant Physiol 1993, 102, 233–240. [Google Scholar]
- Doke, N.; Miura, Y.; Sanchez, L.M.; Park, H.J.; Noritake, T.; Yoshioka, H.; Kawakita, K. The oxidative burst protects plants against pathogen attack: Mechanism and role as an emergency signal for plant bio-defence—A review. Gene 1996, 179, 45–51. [Google Scholar]
- Wu, G.S.; Shortt, B.J.; Lawrence, E.B.; Leon, J.; Fitzsimmons, K.C.; Levine, E.B.; Raskin, I.; Shah, D.M. Activation of host defense mechanisms by elevated production of H2O2 in transgenic plants. Plant Physiol 1997, 115, 427–435. [Google Scholar]
- Wu, G.S.; Shortt, B.J.; Lawrence, E.B.; Levine, E.B.; Fitzsimmons, K.C.; Shah, D.M. Disease resistance conferred by expression of a gene encoding H2O2-generating glucose oxidase in transgenic potato plants. Plant Cell 1995, 7, 1357–1368. [Google Scholar]
- Desikan, R.; Clarke, A.; Hancock, J.T.; Neill, S.J. H2o2 activates a map kinase-like enzyme in Arabidopsis thaliana suspension cultures. J. Exp. Bot 1999, 50, 1863–1866. [Google Scholar]
- Chico, J.M.; Raices, M.; Tellez-Inon, M.T.; Ulloa, R.M. A calcium dependent protein kinase is systemically induced upon wounding in tomato plants. Plant Physiol 2002, 128, 256–270. [Google Scholar]
- Delledonne, M. No news is good news for plants. Curr. Opin. Plant Biol 2005, 8, 390–396. [Google Scholar]
- Rosenthal, G.A.; Berenbaum, M.R. Herbivores: Their Interactions with Secondary Plant Metabolites. Ecological and Evolutionary Processes; Academic Press: San Diego, CA, USA, 1992. [Google Scholar]
- Fraenkel, G.S. The raison d’etre of secondary plant substances: These odd chemicals arose as a means of protecting plants from insects and now guide insects to food. Science 1959, 129, 1466–1470. [Google Scholar]
- Kessler, A.; Baldwin, I.T. Plants responses to insect herbivory: The emerging molecular analysis. Annu. Rev. Plant Biol 2002, 53, 299–328. [Google Scholar]
- Ziegler, J.; Facchini, P.J. Alkaloid biosynthesis: Metabolism and trafficking. Annu. Rev. Plant Biol 2008, 59, 736–769. [Google Scholar]
- Fattorusso, E.; Taglialatela-Scafati, O. Modern Alkaloids: Structure, Isolation, Synthesis and Biology; Wiley: Hoboken, NJ, USA, 2007. [Google Scholar]
- Petterson, D.S.; Harris, D.J.; Allen, D.G. Alkaloids. In Toxic Substances in Crop Plants; D’Mello, J.P.F., Duffus, C.M., Duffus, J.H., Eds.; The Royal Society of Chemistry: Cambridge, UK, 1991; pp. 148–179. [Google Scholar]
- Harborne, J.B. Introduction to Ecological Biochemistry, 3rd ed; Academic Press: London, UK, 1988; p. 356. [Google Scholar]
- Caspi, R.; Foerester, H.; Fulcher, C.A.; Kaipa, P.; Krummenacker, M.; Latendresse, M.; Paley, S.; Rhee, S.Y.; Schearer, A.G.; Tissier, C.; et al. The MeTaCyC database of metabolic pathways and enzymes and the biocyc collection of pathway/genome databases. Nucleic Acids Res. 2008, 36, D623–D631. [Google Scholar]
- Hartmann, T. Chemical ecology of pyrrolizidine alkaloids. Planta 1999, 207, 483–495. [Google Scholar]
- Johnson, M.T.J. Evolutionary ecology of plant defences against herbivores. Funct. Ecol 2011, 25, 305–311. [Google Scholar]
- Eisner, T.; Eisner, M.; Rossini, C.; Iyengar, V.K.; Roach, B.L.; Benedikt, E.; Meinwald, J. Chemical defense against predation in an insect egg. Proc. Natl. Acad. Sci. USA 2000, 97, 1634–1639. [Google Scholar]
- Dutartre, L.; JHilliou, F.; Feyereisen, R. Phylogenomics of the benzoxazinoid biosynthetic pathway of poaceae: Gene duplications and origin of the Bx cluster. BMC Eolutionary Biol 2012, 12, 64. [Google Scholar]
- Seigler, D.S. Cyanide and Cyanogenic Glucosides. In Herbivores: Their Interactions with Secondary Plant Metabolites; Rosenthal, G.A., Berenbaum, M.R., Eds.; Academic Press: San Diego, CA, USA, 1991; pp. 35–77. [Google Scholar]
- Niemeyer, H.M. Hydroxamic acids (4-hydroxy-1,4-benzoxazin-3-ones), defence chemicals in the gramineae. Phytochemistry 1988, 27, 3349–3358. [Google Scholar]
- Robert, C.A.M.; Veyrat, N.; Glauser, G.; Marti, G.; Doyen, G.R.; Villard, N.; Gaillard, M.D.P.; Köllner, T.G.; Giron, D.; Body, M.; et al. A specialist root herbivore exploits defensive metabolites to locate nutritious tissues. Ecol. Lett. 2012, 15, 55–64. [Google Scholar]
- Bjostad, L.B.; Hibbard, B.E. 6-methoxy-2-benzoxazolinone: A semiochemical for host location by western corn rootworm larvae. J. Chem. Ecol 1992, 18, 931–944. [Google Scholar]
- Oikawa, A.; Ishihara, A.; Iwamura, H. Induction of hdmboa-glc accumulation and dimboa-glc 4-O-methyltransferase by jasmonic acid in poaceous plants. Phytochemistry 2002, 61, 331–337. [Google Scholar]
- Glauser, G.; Marti, G.; Villard, N.; Doyen, G.A.; Wolfender, J.L.; Turlings, T.C.J.; Erb, M. Induction and detoxification of maize 1,4-benzoxazin-3-ones by insect herbivores. Plant J 2011, 68, 901–911. [Google Scholar]
- Orcutt, D.M.; Nilsen, E.T. The Physiology of Plants under Stress: Soil and Biotic Factors; John Whiley & Sons: Hoboken, NJ, USA, 2000; Volume 2. [Google Scholar]
- Lindberg Møller, B. Functional diversifications of cyanogenic glucosides. Curr. Opin. Plant Biol 2010, 13, 337–346. [Google Scholar]
- Dustan, W.R.; Henry, T.A. Cyanogenesis in plants, II. The great millet, Sorghwtin vulgare. Chem. News 1902, 85, 399–410. [Google Scholar]
- Forslund, K.; Morant, M.; Jørgensen, B.; Olsen, C.E.; Asamizu, E.; Sato, S.; Tabata, S.; Bak, S. Biosynthesis of the nitrile glucosides rhodiocyanoside A and D and the cyanogenic glucosides lotaustralin and linamarin in Lotus japonicus. Plant Physiol 2004, 135, 71–84. [Google Scholar]
- McMahon, J.M.; White, W.L.B.; Sayre, R.T. Cyanogenesis in cassava (Manihot esculenta crantz). J. Exp. Bot 1995, 46, 731–741. [Google Scholar]
- Sánchez-Pérez, R.; Jørgensen, K.; Olsen, C.E.; Dicenta, F.; Lindberg Møller, B. Bitterness in almonds. Plant Physiol 2008, 146, 1040–1052. [Google Scholar]
- Vetter, J. Plant cyanogenic glycosides. Toxicon 2000, 38, 11–36. [Google Scholar]
- Brattsten, L.B.; Samuelian, J.H.; Long, K.Y.; Kincaid, S.A.; Evans, C.K. Cyanide as a feeding stimulant for the southern armyworm, Spodoptera eridania. Ecol. Entomol 1983, 8, 125–132. [Google Scholar]
- Davis, R.H.; Nahrstedt, A. Cyanogenesis in Insects. In Comprehensive Insect Physiology, Biochemistry Pharmacology; Kerkut, G.A., Gilbert, L.I., Eds.; Pergamon Press: Oxford, UK, 1985; pp. 635–654. [Google Scholar]
- Zagrobelny, M.; Dreon, A.L.; Gomiero, T.; Marcazzan, G.L.; Glaring, M.A.; Linberg Møller, B.; Paoletti, M.G. Toxic moths: Source of a truly safe delicacy. J. Ethnobiol 2009, 29, 64–76. [Google Scholar]
- Forslund, K.; Jonsson, L. Cyanogenic glucosides their metabolic enzymes in barley, in relation to nitrogen levels. Physiol. Plant 1997, 101, 367–372. [Google Scholar]
- Kamp Busk, P.; Lindberg Møller, B. Dhurrin synthesis in sorghum is regulated at the transcriptional level and induced by nitrogen fertilization in older plants. Plant Physiol 2002, 129, 1222–1231. [Google Scholar]
- Bellotti, A.C.; Arias, B. The possible Role of HCN on the Biology and Feeding Behaviours of the Cassava Burrowing Bug (Cyrtomenus Bergi Froeschner). Proceedings on the 1st International Scientific Meeting of the Cassava Biotechnology Network, Cali, Columbia, 25–28 August 1992; Roca, W.M., Thro, A.M., Eds.; Cali: Columbia; pp. 406–409.
- Malagón, J.; Garrido, A. Relación entre el contenido de glicósidos cianogénicos y la resistencia a Capnodis tenebrionis l. En frutales de hueso. Bol. Sanid. Veg. Plagas 1990, 16, 499–503. [Google Scholar]
- Ellsbury, M.M.; Pedersen, G.A.; Fairbrother, T.E. Resistance to foliar-feeding hyperine weevils (coleoptera: Curculionidae) in cyanogenic white clover. J. Econ. Entomol 1992, 85, 2467–2472. [Google Scholar]
- Zagrobelny, M.; Bak, S.; Ekstrøm, C.T.; Olsen, C.E.; Lindberg Møller, B. The cyanogenic glucoside composition of Zygaena filipendulae (lepidoptera: Zygaenidae) as effected by feeding on wild-type and transgenic lotus populations with variable cyanogenic glucoside profiles. Insect Biochem. Mol. Biol 2007, 37, 10–18. [Google Scholar]
- Jensen, N.B.; Zagrobelny, M.; Hjernø, K.; Olsen, C.E.; Houghton-Larsen, J.; Borch, J.; Lindberg Møller, B.; Bak, S. Convergent evolution in biosynthesis of cyanogenic defence compounds in plants and insects. Nat. Commun 2011, 2, 273. [Google Scholar]
- Kakes, P. An analysis of the costs and benefits of the cyanogenic system in Trifolium repens. Theor. Appl. Genet 1989, 77, 111–118. [Google Scholar]
- Lieberi, R. Cyanogenesis of Hevea brasiliensis during infection with Microcyclus ulei. J. Phytopathol 1986, 115, 134–146. [Google Scholar]
- Hopkins, R.J.; van Dam, N.M.; van Loon, J.J.A. Role of glucosinolates in insect-plant relationships and multitrophic interactions. Annu. Rev. Entomol 2009, 54, 57–83. [Google Scholar]
- Dam, N.M.; Tytgat, T.O.G.; Kirkegaard, J.A. Root and shoot glucosinolates: A comparison of their diversity, function and interactions in natural and managed ecosystems. Phytochem. Rev 2009, 8, 171–186. [Google Scholar]
- Karban, R.; Agrawal, A.A.; Thaler, J.S.; Adler, L.S. Induced plant responses and information content about risk of herbivory. Trends Ecol. Evol 1999, 14, 443–447. [Google Scholar]
- Grob, K.; Matile, P.H. Vacuolar location of glucosinolates in horseradish root cells. Plant Sci. Lett 1979, 14, 327–335. [Google Scholar]
- Bennett, R.N.; Wallsgrove, R.M. Tansley review no. 72. Secondary metabolites in plant defence mechanisms. New Phytol 1994, 127, 617–633. [Google Scholar]
- Glen, D.M.; Jones, H.; Fieldsend, J.K. Damage to oilseed rape seedling by the field slug Deroceras reticulatum in relation to glucosinolate concentration. Ann. Appl. Biol 1990, 117, 197–207. [Google Scholar]
- Newman, R.M.; Hanscom, Z.; Kerfoot, W.C. The watercress glucosinolate-myrosinase system: A feeding deterrent to caddisflies, snails and amphipods. Oecologia 1992, 92, 1–7. [Google Scholar]
- Bodnaryk, R.P. Developmental profile of sinalbin in mustard seedlings, Sinapis alba L., and its relationship to insect resistance. J. Chem. Ecol 1991, 17, 1543–1556. [Google Scholar]
- Bartlet, E.; Williams, I.H. Factors restricting the feeding of the cabbage stem flea beetle (Psylliodes chrysocephala). Entomol. Exp. Appl 1991, 60, 233–238. [Google Scholar]
- Blight, M.M.; Pickett, J.A.; Wadhams, L.J.; Woodcock, C.M. Antennal responses of Ceutorhynchus assimilis and Psylliodes chrysocephala to volatiles from oilseed rape. Aspects Appl. Biol 1989, 23, 329–334. [Google Scholar]
- Moyes, C.L.; Raybould, A.F. The role of spatial scale and intraspecific variation in secondary chemistry in host plant location by Ceutorhynchus assimilis (coleoptera: Curculionidae). Proc. R. Soc. Lond 2001, 268, 1567–1573. [Google Scholar]
- Wittstock, U.; Agerbrick, N.; Stauber, E.J.; Olsen, C.E.; Hippler, M. Successful herbivore attack due to metabolic diversion of plant chemical defence. Proc. Natl. Acad. Sci. USA 2004, 101, 4859–4864. [Google Scholar]
- Ratzka, A.; Vogel, H.; Kliebenstein, D.J.; Mitchell-Olds, T.; Kroymann, J. Disarming the mustard oil bomb. Proc. Natl. Acad. Sci. USA 2002, 99, 11223–11228. [Google Scholar]
- Li, Q.; Eigenbrode, S.D.; Stringham, G.R.; Thingarajah, M.R. Feeding and growth of Plutella xylostella and Spodopetra eridania on Brassica juncea with warying glucosinolate concentrations and myrosinase activities. J. Chem. Ecol 2000, 26, 2401–2419. [Google Scholar]
- Barth, C.; Jander, G. Arabidopsis myrosinases TGG1 and TGG2 have redundant function in glycosinolate breakdown and insect defence. Plant J 2006, 46, 549–562. [Google Scholar]
- Kim, J.H.; Jander, G. Myzus persicae (green peach aphid) feeding on Arabidopsis induces the formation of a repellent indole glucosinolate. Plant J 2007, 49, 1008–1019. [Google Scholar]
- Halkier, B.A.; Gershenzon, J. Biology and biochemistry of glucosinolates. Annu. Rev. Plant Biol 2006, 57, 303–333. [Google Scholar]
- D’Mello, J.P.F. Toxicity of Non-protein Amino Acids from Plants. In Amino Acids and Their Derivatives in Higher Plants; Wallsgrove, R.M., Ed.; Cambridge University Press: Cambridge, UK, 1994. [Google Scholar]
- Rosenthal, G.A. Nonprotein Amino Acids as Protective Allelochemicals. In Herbivores: Their Interactions with Secondary Plant Metabolites; Rosenthal, G.A., Berenbaum, M.R., Eds.; Academic Press: San Diego, CA, USA, 1991; Volume 1, pp. 1–34. [Google Scholar]
- Quirk, M.F.; Bushell, J.J.; Jones, R.J.; Megarrity, R.G.; Buttler, K.L. Live-weight gains on leucaena and native grass pastures after dosing cattle with rumen bacteria capable of degrading dhp, a ruminal metabolite from leucaena. J. Agric. Sci 1988, 111, 165–170. [Google Scholar]
- Rincón, M.T.; Allison, M.J.; Michelangeli, F.; de Sanctis, Y.; Domínguez-Bello, M.G. Anaerobic degradation of mimosine-derived hydroxypyridines by cell free extracts of the rumen bacterium Synergistes jonesii. FEMS Microbiol. Ecol 1998, 27, 127–132. [Google Scholar]
- Huang, T.; Jander, G.; de Vos, M. Non-protein amino acids in plant defense against insect herbivores: Representative cases and opportunities for further functional analysis. Phytochemistry 2011, 72, 1531–1537. [Google Scholar]
- Nuessly, G.S.; Scully, B.T.; Hentz, M.G.; Beiriger, R.; Snook, M.E.; Widstrom, N.W. Resistance to Spodoptera frugiperda (lepidoptera: Noctuidae) and Euxesta stigmatias (diptera: Ulidiidae) in sweet corn derived from exogenous and endogenous genetic systems. J. Econ. Entomol 2007, 100, 1887–1895. [Google Scholar]
- Cheeke, P.R. Toxicants of Plant Origin, Volume IV, Phenolics; Boca Raton; CRC Press: Boca Raton, FL, USA, 1989. [Google Scholar]
- Leszczynski, B. The influence of phenolic compounds on the preference of winter wheat cultivars by cereal aphids. Insect Sci. Its Appl 1995, 6, 157–158. [Google Scholar]
- Larsson, S.; Wiren, A.; Lundgren, L.; Ericsson, T. Effects of light and nutrient stress on leaf phenolic chemistry in Salix dasyclados and susceptibility to Galerucella lincola. Oikos 1986, 47, 205–210. [Google Scholar]
- Ruuhola, T.; Tikkanen, O.; Tahvanainen, O. Differences in host use efficiency of larvae of a generalist moth, Operophtera brumata on three chemically divergent salix species. J. Chem. Ecol 2001, 27, 1595–1615. [Google Scholar]
- Luczynski, A.; Isman, M.B.; Rawirth, D.A. Strawberry foliar phenolics and their relationship to development of the two-spotted spider mite. J. Econ. Entomol 1990, 83, 557–563. [Google Scholar]
- Abou-Donia, M.B. Gossypol. In Toxicants of Plant Origin—Volume 5: Phenolics; Cheeke, P.R., Ed.; Boca Raton; CRC Press: Boca Raton, FL, USA, 1989; pp. 2–22. [Google Scholar]
- Maxwell, F.G.; Lafever, H.N.; Jenkins, J.N. Blister beetles on glandless cotton. J. Econ. Entomol 1965, 58, 792–798. [Google Scholar]
- Aharoni, A.; Jongsma, M.A.; Bouwmeester, H.J. Volatile science? Metabolic engineering of terpenoids in plants. Trends Plant Sci 2005, 10, 594–602. [Google Scholar]
- Cherrett, J.M. Some factors involved in the selection of vegetable substrate by Atta cephalotes (L.) (hymenoptera: Formicidae) in tropical rain forest. J. Anim. Ecol 1972, 41, 647–660. [Google Scholar]
- Trapp, S.; Croteau, R. Defensive resin biosynthesis in conifers. Annu. Rev. Plant Physiol. Plant Mol. Biol 2001, 52, 689–724. [Google Scholar]
- Canals, D.; Irurre-Santilari, J.; Casas, J. The first cytochrome P450 in ferns. FEBS J 2005, 272, 4817–4825. [Google Scholar]
- Cruickshank, P.A. Insect juvenile hormone analogues: Effects of some terpenoid amide derivatives. Bull. World Health Org 1971, 44, 395–396. [Google Scholar]
- Hummelbrunner, L.A.; Isman, M.B. Acute, sublethal, antifeedant, and synergistic effects of monoterpenoid essential oil compounds on the tobacco cutworm, Spodoptera litura (Lep., Noctuidae). J. Agric. Food Chem 2001, 49, 715–720. [Google Scholar]
- Duffey, S.S.; Stout, M.J. Antinutritive and toxic components of plant defence against insects. Arch. Insect Biochem. Physiol 1996, 32, 3–37. [Google Scholar]
- Bowles, D.J. Defense-related proteins in higher plants. Annu. Rev. Biochem 1990, 59, 873–907. [Google Scholar]
- Morton, R.L.; Schroeder, H.E.; Bateman, K.S.; Chrispeels, M.J.; Armstrong, E.; Higgings, T.J.V. Bean α-amylase inhibitor 1 in transgenic peas (Pisum sativum) provides complete protection from pea weevil (Bruchus pisorum) under field conditions. Proc. Natl. Acad. Sci. USA 2000, 97, 3820–3825. [Google Scholar]
- Kluh, I.; Horn, M.; Hýblová, J.; Hubert, J.; Dolecková-Maresová, L.; Voburka, Z.; Kudlíková, I.; Kocourek, F.; Mares, M. Inhibitory specificity and insecticidal selectivity of α-amylase inhibitor from Phaseolus vulgaris. Phytochemistry 2005, 66, 31–39. [Google Scholar]
- Shade, R.E.; Schroeder, H.E.; Pueyo, J.J.; Tabe, L.M.; Murdock, L.L.; Higgins, T.J.V.; Chrispeels, M.J. Transgenic pea seeds expressing the α-amylase inhibitor of the common bean are resistant to bruchid beetles. Biotechnology 1994, 12, 739–769. [Google Scholar]
- Schroeder, H.E.; Gollasch, S.; Moore, A.; Tabe, L.M.; Craig, S.; Hardie, D.C.; Chrispeels, M.J.; Spencer, D.; Higgins, T.J.V. Bean α-amylase inhibitor confers resistance to the pea weevil (Bruchus pisorum) in transgenic peas (Pisum sativum l.). Plant Physiol 1995, 107, 1233–1239. [Google Scholar]
- Kramer, K.J.; Muthukrishnan, S. Insect chitinases: Molecular biology and potential use as biopesticides. Insect Biochem. Mol. Biol 1997, 27, 887–900. [Google Scholar]
- Jouanin, L.; Bonade-Bottino, M.; Girard, C.; Morrot, G.; Giband, M. Transgenic plants for insect resistance. Plant Sci. Lett 1998, 131, 1–11. [Google Scholar]
- Lawrence, S.D.; Novak, N.G. Expression of poplar chitinase in tomato leads to inhibition of development in colorado potato beetle. Biotechnol. Lett 2006, 28, 593–599. [Google Scholar]
- Ding, X.; Gopalakrishnan, B.; Johnson, L.B.; White, F.F.; Wang, X.; Morgan, T.D.; Kramer, K.J.; Muthukrishnan, S. Insect resistance of transgenic tobacco expressing an insect chitinase gene. Transgenic Res 1998, 7, 77–84. [Google Scholar]
- Gatehouse, A.M.R.; Davison, G.M.; Newell, C.A.; Merryweather, A.; Hamilton, W.D.O.; Burgess, E.P.J.; Gilbert, R.J.C.; Gatehouse, J.A. Transgenic potato plants with enhanced resistance to the tomato moth, Lacanobia oleracea: Growth room trials. Mol. Breed 1997, 3, 49–63. [Google Scholar]
- Arason, M.J. Lectins as defence molecules in vertebrates invertebrates. Fish Shellfish Immunol 1996, 6, 277–289. [Google Scholar]
- Edelman, G.M.; Cunningham, B.A.; Reeke, G.N.; Becker, J.W.; Waxdal, M.J.; Wang, J.L. The covalent and three-dimensional structure of concanavalin A. Proc. Natl. Acad. Sci. USA 1972, 69, 2580–2584. [Google Scholar]
- Gatehouse, A.M.R.; Davison, G.M.; Stewart, J.N.; Gatehouse, L.N.; Kumar, A.; Geoghegan, L.E.; Birch, A.N.E.; Gatehouse, J.A. Concanavalin A inhibits development of tomato moth and peach-potato aphid when expressed in transgenic potato plants. Mol. Breed 1997, 5, 153–165. [Google Scholar]
- Gatehouse, A.M.R.; Gatehouse, J.A. Identifying proteins with insecticidal activity: Use of encoding genes to produce insect-resistant transgenic crops. Pestic. Sci 1998, 52, 165–175. [Google Scholar]
- Osborn, T.C.; Alexander, D.C.; Sun, S.S.M.; Cardona, C.; Bliss, F.A. Insecticidal activity and lectin homology of arcelin seed protein. Science 1988, 240, 207–210. [Google Scholar]
- Czapla, T.H.; Lang, B.A. Effect of plant lectins on the larval development of european corn borer (lepidopteran: Pyralidae) and southern corn borer (coleoptera: Chrysomelidae). J. Econ. Entomol 1990, 83, 2480–2485. [Google Scholar]
- Eismann, C.H.; Donaldson, R.A.; Pearson, R.D.; Cadagon, L.C.; Vuocolo, T.; Tellam, R.L. Larvicidal activity of lectinson Lucilia cuprina: Mechanism of action. Entomol. Exp. Appl 1994, 72, 1–11. [Google Scholar]
- Down, R.E.; Gatehouse, A.M.R.; Hamilton, W.D.O.; Gatehouse, J.A. Snowdrop lectin inhibits development and decreases fecundity of the glasshouse potato aphid when administered in vitro and via transgenic plants both in laboratory and glasshouse trials. J. Insect Physiol 1996, 42, 11–12. [Google Scholar]
- Chrispeels, M.J.; Raikhel, N.V. Lectins, lectin genes, and their role in plant defense. Plant Cell 1991, 3, 1–9. [Google Scholar]
- Mahanil, S.; Attajarusit, J.; Stout, M.J.; Thipyapong, P. Overexpression of tomato polyphenol oxidase increases resistance to common cutworm. Plant Sci 2008, 174, 456–466. [Google Scholar]
- Castañera, P.; Steffens, J.C.; Tingey, W.M. Biological performance of colorado potato beetle larvae on potato genotypes with differing levels of polyphenol oxidase. J. Chem. Ecol 1996, 22, 91–101. [Google Scholar]
- Alba-Meraz, A.; Choe, T.H. Systemic effects on oxidative enzymes in Phaseolus vulgaris leaves that have been wounded by the grasshopper Melanoplus differentialis or have had a foliar application of jasmonic acid. Int. J. Plant Sci 2002, 163, 317–328. [Google Scholar]
- Felton, G.W.; Donato, K.K.; Broadway, R.M.; Duffey, S.S. Impact of oxidized plant phenolics on the nutritional quality of dietary protein to a noctuid herbivore, Spodoptera exigua. J. Insect Physiol 1992, 38, 277–285. [Google Scholar]
- Thipyapong, P.; Melkonian, J.; Wolfe, D.W.; Steffens, J.C. Suppression of polyphenol oxidase increases stress tolerance in tomato. Plant Sci 2004, 167, 693–703. [Google Scholar]
- Li, L.; Steffens, J.C. Overexpression of polyphenol oxidase in transgenic tomato plants results in enhanced disease susceptibility. Planta 2002, 220, 105–117. [Google Scholar]
- Constabel, C.P.; Yip, L.; Patton, J.J.; Christopher, M.E. Polyphenol oxidase from hybrid poplar. Cloning and expression in response to wounding and herbivory. Plant Physiol 2000, 124, 285–295. [Google Scholar]
- Baldwin, I.T.; Preston, C.A. The eco-physiological complexity of plant responses to insect herbivores. Planta Med 1999, 208, 137–145. [Google Scholar]
- Coppedge, B.R.; Jones, J.M.; Felton, G.W.; Stephen, F.M. Examination of midgut proteinases of the adult southern pine-beetle (coleoptera, scolytidae). J. Entomol. Sci 1994, 29, 457–465. [Google Scholar]
- Terra, W.R.; Ferreira, C. Insect digestive enzymes—Properties, compartmentalization and function. Comp. Biochem. Physiol 1994, B 109, 411–425. [Google Scholar]
- Rodrigues Macedo, M.L.; das Graças Machado Freire, M. Insect digestive enzymes as a target for pest control. Invertebr. Surv. J 2011, 8, 190–198. [Google Scholar]
- Wolfson, J.L.; Murdock, L.L. Potential use of protease inhibitors for host-plant resistance: A test case. Environ. Entomol 1995, 24, 52–57. [Google Scholar]
- Broadway, R.M.; Duffey, S.S. Plant proteinase inhibitors: Mechanism of action and effect on the growth and digestive physiology of larval Heliothis zea and Spodoptera exigua. J. Insect Physiol 1986, 32, 827–833. [Google Scholar]
- Brown, W.E.; Ryan, C.A. Isolation and characterization of a wound-induced trypsin inhibitor from alfalfa leaves. Biochemistry 1984, 23, 8–22. [Google Scholar]
- Lipke, H.; Fraenkel, G.S.; Liener, I.E. Effects of soybean inhibitors on growth of Tribolium confusum. J. Sci. Food Agric 1954, 2, 410–415. [Google Scholar]
- Hilder, V.A.; Gatehouse, A.M.R.; Sheerman, S.E.; Barker, R.F.; Boulter, D. A novel mechanism of insect resistance engineered into tobacco. Nature 1987, 300, 160–163. [Google Scholar]
- Ryan, C.A. Protease inhibitors in plants: Genes for improving defenses against insects and pathogens. Annu. Rev. Phytopathol 1990, 28, 425–449. [Google Scholar]
- Newingham, B.A.; Callaway, R.M.; BassiriRad, H. Allocating nitrogen away from a herbivore: A novel compensatory response to root herbivory. Oecologia 2007, 153, 913–920. [Google Scholar]
- Poveda, K.; Jiménez, M.I.G.; Kessler, A. The enemy as ally: Herbivore-induced increase in crop yield. Ecol. Appl 2010, 20, 1787–1793. [Google Scholar]
- Schwachtje, J.; Minchin, P.E.H.; Jahnke, S.; van Dongen, J.T.; Schittko, U.; Baldwin, I.T. Snf1-related kinases allow plants to tolerate herbivory by allocating carbon to roots. Proc. Natl. Acad. Sci. USA 2006, 103, 12935–12940. [Google Scholar]
- Babst, B.A.; Ferrieri, R.A.; Gray, D.W.; Lerdau, M.; Schlyer, D.J.; Schueller, M.; Thorpe, M.R.; Orians, C.M. Jasmonic acid induces rapid changes in carbon transport and partitioning in populus. New Phytol 2005, 167, 63–72. [Google Scholar]
- Henkes, G.J.; Thorpe, M.R.; Minchin, P.E.H.; Schurr, U.; Röse, U.S.R. Jasmonic acid treatment to part of the root system is consistent with simulated leaf herbivory, diverting recently assimilated carbon towards untreated roots within an hour. Plant Cell Environ 2008, 31, 1229–1236. [Google Scholar]
- Kaplan, I.; Halitschke, R.; Kessler, A.; Rehill, B.J.; Sardanelli, S.; Denno, R.F. Physiological integration of roots and shoots in plant defense strategies links above- and belowground herbivory. Ecol. Lett 2008, 11, 841–851. [Google Scholar]
- Lalonde, S.; Wipf, D.; Frommer, W.B. Transport mechanisms for organic forms of carbon and nitrogen between source and sink. Annu. Rev. Plant Biol 2004, 55, 341–372. [Google Scholar]
- Erb, M.; Lenk, C.; Degenhardt, J.; Turlings, T.C.J. The underestimated role of roots in defense against leaf attackers. Trends Plant Sci 2009, 14, 653–659. [Google Scholar]
- Müller, C. Plant-insect Interactions on Cuticular Surfaces. In Biology of the Plant Cuticle; Rieder, M., Müller, C., Eds.; Blackwell Publishing Ltd: Oxford, UK, 2007. [Google Scholar]
- Blenn, B.; Bandoly, M.; Küffner, A.; Otte, T.; Geiselhardt, S.; Fatouros, N.E.; Hilker, M. Insect egg deposition induces indirect defense and epicuticular wax changes in Arabidopsis thaliana. J. Chem. Ecol 2012, 38, 882–892. [Google Scholar]
- Myers, J.H.; Bazely, D. Thorns, Spines, Prickles, and Hairs: Are They Stimulated by Herbivory and Do They Deter Herbivores? In Phytochemical Induction by Herbivores; Tallamy, D.W., Raupp, M.J., Eds.; John Wiley & Sons: New York, NY, USA, 1991; pp. 325–344. [Google Scholar]
- Fordyce, J.A.; Agrawal, A.A. The role of plant trichomes and caterpillar group size on growth and defence of the pipevine swallowtail Battus philenor. J. Anim. Ecol 2001, 70, 997–1005. [Google Scholar]
- Weinhold, A.; Baldwin, I.T. Trichome-derived O-acyl sugars are a first meal for caterpillars that tags them for predation. Proc. Natl. Acad. Sci. USA 2011, 108, 7855–7859. [Google Scholar]
- Raupp, M.J. Effects of leaf toughness on mandibular wear of the leaf beetle, Plagiodera versicolora. Ecol. Entomol 2008, 10, 73–79. [Google Scholar]
- Nichols-Orians, C.M.; Schultz, J.C. Interactions among leaf toughness, chemistry, and harvesting by attine ants. Ecol. Entomol 1990, 15, 311–320. [Google Scholar]
- Read, J.; Stokes, A. Plant biomechanics in an ecological context. Am. J. Bot 2006, 93, 1546–1565. [Google Scholar]
- McNaughton, S.J.; Tarrants, J.L. Grass leaf silicification: Natural selection for an inducible defense against herbivores. Proc. Natl. Acad. Sci. USA 1983, 80, 790–791. [Google Scholar]
- Schoonhoven, L.M.; van Loon, J.J.A.; Dicke, M. Insect-plant Biology; Oxford University Press: Oxford, UK, 2005. [Google Scholar]
- Care, D.A.; Crush, J.R.; Hardwick, S.; Nichols, S.N.; Ouyang, L. Interaction between Clover Root Weevil and Clover Root Type. In New Zealand Plant Protection; Zydenbos, S.M., Ed.; New Zealand Plant Protection Society: Christchurch, New Zealand, 2000; Volume 53, pp. 420–424. [Google Scholar]
- Quinn, M.A.; Hall, M.H. Compensatory response of a legume root-nodule system to nodule herbivory by Sitona hispidulus. Entomol. Exp. Appl 1992, 64, 167–176. [Google Scholar]
- Johnson, S.N.; Hallett, P.D.; Gillespie, T.L.; Halpin, C. Below-ground herbivory and root toughness: A potential model system using lignin-modified tobacco. Physiol. Entomol 2010, 35, 186–191. [Google Scholar]
- Agrawal, A.A.; Konno, K. Latex: A model for understanding mechanisms, ecology, and evolution of plant defense against herbivory. Annu. Rev. Ecol. Syst 2009, 40, 311–331. [Google Scholar]
- Dussourd, D.E.; Hoyle, A.M. Poisoned plusiines: Toxicity of milkweed latex and cardenolides to some generalist caterpillars. Chemoecology 2000, 10, 11–16. [Google Scholar]
- Buttery, B.R.; Boatman, S.G. Water Deficits and Flow of Latex. In Water Deficits and Plant Growth; Kozlowski, T.T., Ed.; Academic Press: New York, NY, USA, 1976; Volume 4, pp. 233–289. [Google Scholar]
- Carroll, C.R.; Hoffman, C.A. Chemical feeding repellent mobilized in response to insect herbivory and counteradaptation by Epilachna tredecimnotata. Science 1980, 209, 414–416. [Google Scholar]
- Dussourd, D.E.; Denno, R.F. Deactivation of plant defense: Correspondence between insect behavior and secretory canal architecture. Ecology 1991, 72, 1383–1396. [Google Scholar]
- Williams, C.E. Host plant latex and the feeding behavior of Chrysochus auratus (coleoptera: Chrysomelidae). Coleopt. Bull 1991, 45, 195–196. [Google Scholar]
- Mithöfer, A.; Boland, W. Plant defense against herbivores: Chemical aspects. Annu. Rev. Plant Biol 2012, 63, 431–450. [Google Scholar]
- Phillips, M.A.; Croteau, R.B. Resin-based defenses in conifers. Trends Plant Sci 1999, 4, 184–190. [Google Scholar]
- Raffa, K.F.; Induced, Defensive. Reactions in Conifer-bark Beetle Systems. In Phytochemical Induction by Herbivores; Tallamy, D.W., Raup, M.J., Eds.; John Wiley & Sons: New York, NY, USA, 1991. [Google Scholar]
- Raffa, K.F.; Berryman, A.A. The role of host plant resistance in the colonization behavior and ecology of bark beetles. Ecol. Monogr 1983, 53, 27–49. [Google Scholar]
- Dicke, M.; Sabelis, M.W. How plants obtain predatory mites as bodyguards. Netherlands J. Zool 1988, 38, 149–165. [Google Scholar]
- Dudareva, N.; Negre, F.; Nagegowda, D.A.; Orlova, I. Plant volatiles: Recent advances and future perspectives. Crit. Rev. Plant Sci 2006, 25, 417–440. [Google Scholar]
- Pichersky, E.; Noel, K.P.; Dudareva, N. Biosynthesis of plant volatiles: Nature’s diversity and ingenuity. Science 2006, 311, 808–811. [Google Scholar]
- Dicke, M.; van Poecke, R.M.P.; de Boer, J.G. Inducible indirect defence of plants: From mechanism to ecological function. Basic Appl. Ecol 2003, 4, 27–42. [Google Scholar]
- Kessler, A.; Baldwin, I.T. Defensive function of herbivore-induced plant volatile emissions in nature. Science 2001, 291, 2141–2144. [Google Scholar]
- Karban, R.; Baldwin, I.T.; Baxter, K.J.; Laue, G.; Felton, G.W. Communication between plants: Induced resistance in wild tobacco plats following clipping of neighboring sagebrush. Oecologia 2000, 125, 66–71. [Google Scholar]
- Engelberth, J.; Alborn, H.T.; Schmelz, E.A.; Tumlinson, J.H. Airborne signals prime plants against insect herbivore attack. Proc. Natl. Acad. Sci. USA 2004, 101, 1781–1785. [Google Scholar]
- Baldwin, I.T.; Schultz, J.C. Rapid changes in tree leaf chemistry induce by damage: Evidence for communication between plants. Science 1983, 221, 277–279. [Google Scholar]
- Turlings, T.C.J.; Loughrin, J.H.; McCall, P.J.; Röse, U.S.; Lewis, W.J.; Tumlinson, J.H. How caterpillar-damaged plants protect themselves by attracting parsitic wasps. Proc. Natl. Acad. Sci. USA 1995, 92, 4169–4174. [Google Scholar]
- Tumlinson, J.H.; Paré, P.W.; Lewis, W.J. Plant Production of Volatile Semiochemicals in Response to Insect-derived Elicitors. In Insect-plant Interactions and Induced Plant Defence; Chadwick, D.J., Goode, J.A., Eds.; John Wiley & Sons: Chichester, UK, 1999; Volume 8, pp. 95–109. [Google Scholar]
- Paré, P.W.; Tumlinson, J.H. Induced synthesis of plant volatiles. Nature 1997, 385, 30–31. [Google Scholar]
- Turlings, T.C.J.; Lengwiler, U.B.; Bernasconi, M.L.; Wechsler, D. Timing of induced volatile emissions in maize seedlings. Planta 1998, 207, 146–152. [Google Scholar]
- Walling, L.L. The myriad plant responses to herbivores. J. Plant Growth Regul 2000, 19, 195–216. [Google Scholar]
- Rasmann, S.; Köllner, T.G.; Degenhardt, J.; Hiltpold, I.; Toepfer, S.; Kuhlmann, U.; Gershenzon, J.; Turlings, T.C.J. Recruitment of entomopathogenic nematodes by insect-damaged maize roots. Nature 2005, 434, 732–737. [Google Scholar]
- Erb, M.; Foresti, N.; Turlings, T.C.J. A tritrophic signal that attracts parasitoids to host-damaged plants withstands disruption by non-host herbivores. BMC Plant Biol. 2010. [Google Scholar] [CrossRef]
- Ali, J.G.; Alborn, H.T.; Stelinski, L.L. Constitutive and induced subterranean plant volatiles attract both entomopathogenic and plant parasitic nematodes. J. Ecol 2011, 99, 26–35. [Google Scholar]
- Hilker, M.; Kobs, C.; Varma, M.; Schrank, K. Insect egg deposition induces Pinus sylvestris to attract egg parasitoids. J. Exp. Biol 2002, 205, 455–461. [Google Scholar]
- Mumm, R.; Schrank, K.; Wegener, R.; Schulz, S.; Hilker, M. Chemical analysis of volatiles emitted by Pinus sylvestris after induction by insect oviposition. J. Chem. Ecol 2003, 29, 1235–1252. [Google Scholar]
- Schnee, C.; Köllner, T.G.; Held, M.; Turlings, T.C.J.; Gershenzon, J.; Degerhardt, J. The products of a single maize sesquiterpene synthase form a volatile defense signal that attracts natural enemies of maize herbivores. Proc. Natl. Acad. Sci. USA 2006, 103, 1129–1134. [Google Scholar]
- Schuman, M.C.; Barthel, K.; Baldwin, I.T. Herbivory-induced volatiles function as defenses increasing fitness of the native plant Nicotiana attenuata in nature. eLife 2012, 1, e00007. [Google Scholar]
- Hulcr, J.; Pollet, M.; Ubik, K.; Vrkoc, J. Exploitation of kairomones and synomones by Medetera spp. (diptera: Dolichopodidae, predators of spruce bark beetles. Eur J. Entomol 2005, 102, 655–662. [Google Scholar]
- Moayeri, H.R.S.; Ashouri, A.; Brodsgaard, H.F.; Enkegaard, A. Males of the predatory mirid bug Macrolophus caliginosus exploit plant volatiles induced by conspecifics as a sexual synomone. Entomol. Exp. Appl 2007, 123, 49–55. [Google Scholar]
- Shimoda, T.; Takabayashi, J.; Ashihara, W.; Takafuji, A. Response of predatory insect Scolothrips takahashii towards herbivore-induced plant volatiles under laboratory and field conditions. J. Chem. Ecol 1997, 23, 2033–2048. [Google Scholar]
- Ali, J.G.; Alborn, H.T.; Stelinski, L.L. Subterranean herbivore-induced volatiles released by citrus roots upon feeding by Diaprepes abbreviatus recruit entomopathogenic nematodes. J. Chem. Ecol 2010, 36, 361–368. [Google Scholar]
- Seybold, S.J.; Bohlmann, J.; Raffa, K.F. Biosynthesis of coniferophagous bark beetle pheromones and conifer isoprenoids: Evolutionary perspective and synthesis. Can. Entomol 2000, 132, 697–753. [Google Scholar]
- Dicke, M. Local and systemic production of volatile herbivore-induced terpenoids: Their role in plant-carnivore mutualism. J. Plant Physiol 1994, 143, 465–472. [Google Scholar]
- Kahn, R.A.; Bak, S.; Svendsen, I.; Halkier, B.A.; Lindberg Møller, B. Isolation and reconstitution of cytochrome P450ox and in vitro reconstitution of the entire biosynthetic pathway of the cyanogenic glucoside dhurrin from sorghum. Plant Physiol 1997, 115, 1661–1670. [Google Scholar]
- Zimmerli, L.; Jakab, G.; Métraux, J.P.; Mauch-Mani, B. Potentiation of pathogen-specific defense mechanisms in Arabidopsis by β-aminobutyric acid. Proc. Natl. Acad. Sci. USA 2000, 97, 12920–12925. [Google Scholar]
- Kessler, A.; Halitschke, R.; Diesel, C.; Baldwin, I.T. Priming of plant defence responses in nature by airborne signalling between Artemisia tridenta and Nicotiana attenuata. Oecologia 2006, 148, 280–292. [Google Scholar]
- Frost, C.; Appel, H.; Carlson, J.; de Moraes, C.; Mescher, M.; Schultz, J. Within-plant signalling by volatiles overcomes vascular constraints on systemic signalling and primes response against herbivores. Ecol. Lett 2007, 10, 490–498. [Google Scholar]
- Orians, C. Herbivores, vascular pathways, and systemic induction: Facts and artifacts. J. Chem. Ecol 2005, 31, 2231–2242. [Google Scholar]
- Karban, R.; Shiojiri, K.; Huntzinger, M.; McCall, A.C. Damage-induced resistance in sagebrush: Volatiles are key to intra- and interplant communictation. Ecology 2006, 87, 922–930. [Google Scholar]
- Khan, Z.R.; AmpongNyarko, K.; Chiliswa, P.; Hassanali, A.; Kimani, S.; Lwande, W.; Overholt, W.A.; Pickett, J.A.; Smart, L.E.; Wadhams, L.J. Intercropping increases parasitism of pests. Nature 1997, 388, 631–632. [Google Scholar]
- Bernasconi Ockroy, M.L.; Turlings, T.C.J.; Edwards, P.J.; Fritzsche-Hoballah, M.E.; Ambrosetti, L.; Bassetti, P.; Dorn, S. Response of natural populations of predators and parasitoids to artificially induced volatile emissions in maize plants (Zea mays l.). Agric. For. Entomol 2001, 3, 201–209. [Google Scholar]
- Tinzaara, W.; Gold, C.S.; Dicke, M.; vas Huis, A. Olfactory responses of banana weevil predators to volatiles pseudostem tissue and synthetic pheromone. J. Chem. Ecol 2005, 31, 1537–1553. [Google Scholar]
- Heil, M. Induction of two indirect defences benefits lima bean in nature. J. Ecol 2004, 92, 527–536. [Google Scholar]
- Heil, M. Indirect defence—Recent development and open questions. Progress Bot 2007, 69, 360–395. [Google Scholar]
- González-Teuber, M.; Heil, M. Nectar chemistry is tailored for both attraction of mutualists and protection from exploiters. Plant Signal. Behav 2009, 4, 809–813. [Google Scholar]
- Bentley, B.L. Extrafloral nectaries and protection by pugnacious bodyguards. Annu. Rev. Ecol. Syst 1977, 8, 407–427. [Google Scholar]
- Fægri, K.; van der Pijl, L. The Principles of Pollination Ecology; Pergamon Press: Oxford, UK, 1979; Volume 3. [Google Scholar]
- Wagner, D.; Kay, A. Do extrafloral nectaries distract ants from visiting flowers? An experimental test of an overlooked hypothesis. Evol. Ecol. Res 2002, 4, 293–305. [Google Scholar]
- Heil, M. Indirect defence via tritrophic interactions. New Phytol 2007, 178, 41–61. [Google Scholar]
- Baker, H.G.; Baker, I. Floral Nectar Sugar Constituents in Relation to Pollinator Type. In Handbook of Experimental Pollination Biology; Jones, C.E., Little, R.J., Eds.; Van Nostrand Reinhold: New York, NY, USA, 1983; pp. 117–141. [Google Scholar]
- Baker, H.G.; Baker, I.M.H. Chemical Constituents of Nectar in Relation to Pollination Mechanisms and Phylogeny. In Biochemical Aspects of Evolutionary Biology; Nitecki, M.H., Ed.; University of Chicago Press: Chicago, IL, USA, 1982; pp. 131–171. [Google Scholar]
- Davis, A.R.; Pylatuik, J.D.; Paradis, J.C.; Low, N.H. Nectar-carbohydrate production and composition vary in relation to nectary anatomy and locations within individual flowers of several species of brassicaceae. Planta 1998, 205, 305–318. [Google Scholar]
- Guerrant, E.O., Jr; Fiedler, P.L. Flower defenses against nectar-pilferage by ants. Biotropica 1981, 13, 25–33. [Google Scholar]
- Stephenson, A.G. Iridoid glycosides in the nectar of Catalpa speciosa are unpalatable to nectar thieves. J. Chem. Ecol 1982, 8, 1025–1034. [Google Scholar]
- Heil, M.; Fiala, B.; Baumann, B.; Linsemair, K.E. Temporal, spatial and biotic variations in extrafloral nectar secretion by Macaranga tanarius. Funct. Ecol 2000, 14, 749–757. [Google Scholar]
- Wäckers, F.L.; Zuber, D.; Wunderlin, R.; Keller, F. The effect of herbivory on temporal and spatial dynamics of foliar nectar production in cotton and castor. Ann. Bot 2001, 87, 365–370. [Google Scholar]
- Choh, Y.; Kugimyya, S.; Takabayashi, J. Induced production of extrafloral nectar in intact lima bean plants in response to volatiles from spider mite-infested conspecific plants as a possible indirect defence against spider mites. Oecologia 2006, 147, 445–460. [Google Scholar]
- Heil, M.; Koch, T.; Hilpert, A.; Fiala, B.; Boland, W.; Linsemair, K.E. Extrafloral nectar production of the ant-associated plant, Macaranga tanarius, is an induced, indirect, defensive response elicited by jasmonic acid. Proc. Natl. Acad. Sci. USA 2001, 97, 1083–1088. [Google Scholar]
- O’Dowd, D.J. Pearl bodies as ant food: An ecological role for some leaf emergencies of tropical plants. Biotropica 1982, 14, 40–49. [Google Scholar]
- O’Dowd, D.J. Pearl bodies of a neotropical tree, Ochroma pyramidale: Ecological implications. Am. J. Bot 1980, 67, 543–549. [Google Scholar]
- Heil, M.; Fiala, B.; Linsenmair, K.E.; Zotz, G.; Menke, P.; Maschwitz, U. Food body production in Macaranga triloba: A plant investment in anti-herbivore defence via mutualistic ant partners. J. Ecol 1997, 85, 847–861. [Google Scholar]
- Risch, J.R.; Rickson, F.R. Mutualism in which ants must be present before plants produce food bodies. Nature 1981, 291, 149–150. [Google Scholar]
- Fischer, R.C.; Richter, A.; Wanek, W.; Mayer, V. Plants feed ants: Food bodies of myrmecophytic piper and their significance for the interaction with Pheidole bicornis ants. Oecologia 2002, 133, 186–192. [Google Scholar]
- Romero, G.Q.; Benson, W.W. Biotic interactions of mites, plants and leaf domatia. Curr. Opin. Plant Biol 2005, 8, 436–440. [Google Scholar]
- Grostal, P.; O’Dowd, D.J. Plants, mites and mutualism: Leaf domatia and the abundance and reproduction of mites Viburnum tinus. Oecologia 1994, 97, 308–315. [Google Scholar]
- Agrawal, A.A.; Karban, R.; Colfer, R.G. How leaf domatia and induced plant resistance affect herbivores, natural enemies and plant performance. Oikos 2003, 89, 70–80. [Google Scholar]
- Gaume, L.; Zacharias, M.; Grosbois, V.; Borges, R.M. The fitness consequences of bearing domatia and having the right ant partner: Experiments with protective and non-protective ants in a semi-myrmecophyte. Oecologia 2005, 145, 76–86. [Google Scholar]
- Bolton, M.D. Primary metabolism and plant defense—Fuel for the fire. Mol. Plant Microbe Interact 2009, 22, 487–497. [Google Scholar]
- Fujita, M.; Fujita, Y.; Noutoshi, Y.; Takahashi, F.; Narusaka, Y.; Yamaguchi-Shinozaki, K.; Shinozaki, K. Crosstalk between abiotic and biotic stress responses: A current view from the points of convergence in the stress signaling networks. Curr. Opin. Plant Biol 2006, 9, 436–442. [Google Scholar]
- Fraire-Velázquez, S.; Rodríguez-Guerra, R.; Sánchez-Calderón, L. Abiotic and Biotic Stress Response Crosstalk in Plants. In Abiotic Stress Response in Plants—Physiological, Biochemical and Genetic Perspectives; Shanker, A., Ed.; InTech: Rijeka, Croatia, 2011; p. 346. [Google Scholar]
- Hufford, M.B.; Xu, X.; van Heerwaarden, J.; Pyhajarvi, T.; Chia, J.M.; Cartwright, R.A.; Elshire, R.J.; Glaubitz, J.C.; Guill, K.E.; Kaeppler, S.M.; et al. Comparative population genomics of maize domestication and improvement. Nat. Genet 2012, 44, 808–811. [Google Scholar]
- Lyons, R.; Manners, J.M.; Kazan, K. Jasmonate biosynthesis and signaling in monocots: A comparative overview. Plant Cell Rep. 2013, 1–13. [Google Scholar]
- Yoshii, M.; Yamazaki, M.; Rakwal, R.; Kishi-Kaboshi, M.; Miyao, A.; Hirochika, H. The NAC transcription factor RIM1 of rice is a new regulator of jasmonate signaling. Plant J 2010, 61, 804–815. [Google Scholar]
- Coley, P.D.; Aide, T.M. Comparison of Herbivory and Plant Defenses in Temperate and Tropical Broad-leaved Forests. In Plant-Animal Interactions: Evolutionary Ecology in Tropical and Temperate Regions; Price, P.W., Lewinsohn, T.M., Fernandes, G.W., Benson, W.W., Eds.; John Wiley & Sons: New York, NY, USA, 1991; pp. 25–49. [Google Scholar]
- Andrew, N.R.; Roberts, I.R.; Hill, S.J. Insect herbivory along environmental gradients. Open J. Ecol 2012, 2, 202–213. [Google Scholar]
- Marchin, R.; Zeng, H.; Hoffmann, W. Drought-deciduous behavior reduces nutrient losses from temperate deciduous trees under severe drought. Oecologia 2010, 163, 845–854. [Google Scholar]
- Andrew, N.R.; Hughes, L. Herbivore damage along a latitudinal gradient: Relative impacts of different feeding guilds. Oikos 2005, 108, 176–182. [Google Scholar]
- Andrew, N.R.; Hughes, L. Potential host colonization by insect herbivores in a warmer climate: A transplant experiment. Glob. Change Biol 2007, 13, 1539–1549. [Google Scholar]
- Grant, M.R.; Kazan, K.; Manners, J.M. Exploiting pathogens’ tricks of the trade for engineering of plant disease resistance: Challenges and opportunities. Microb. Biotechnol. 2013, 212–222. [Google Scholar]
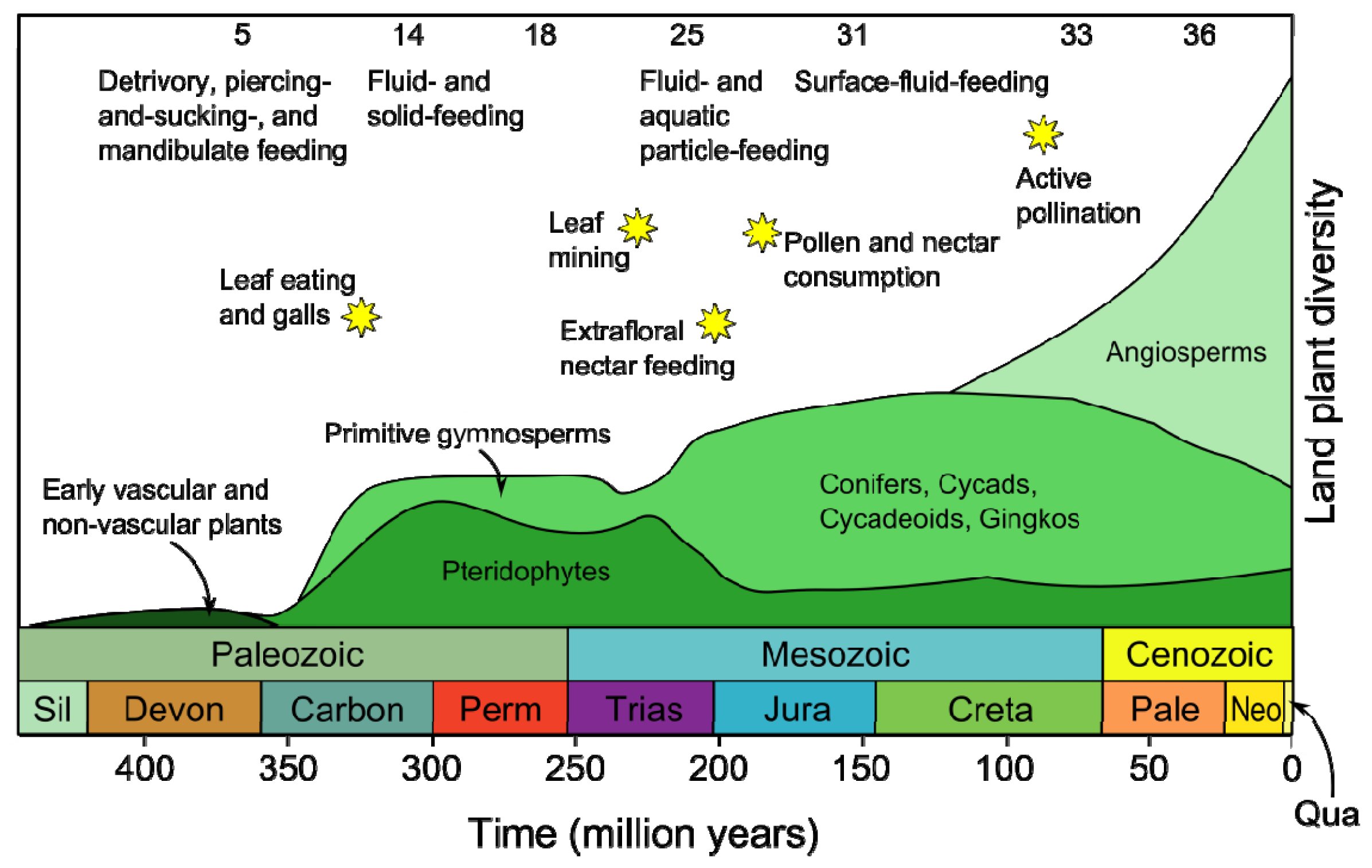
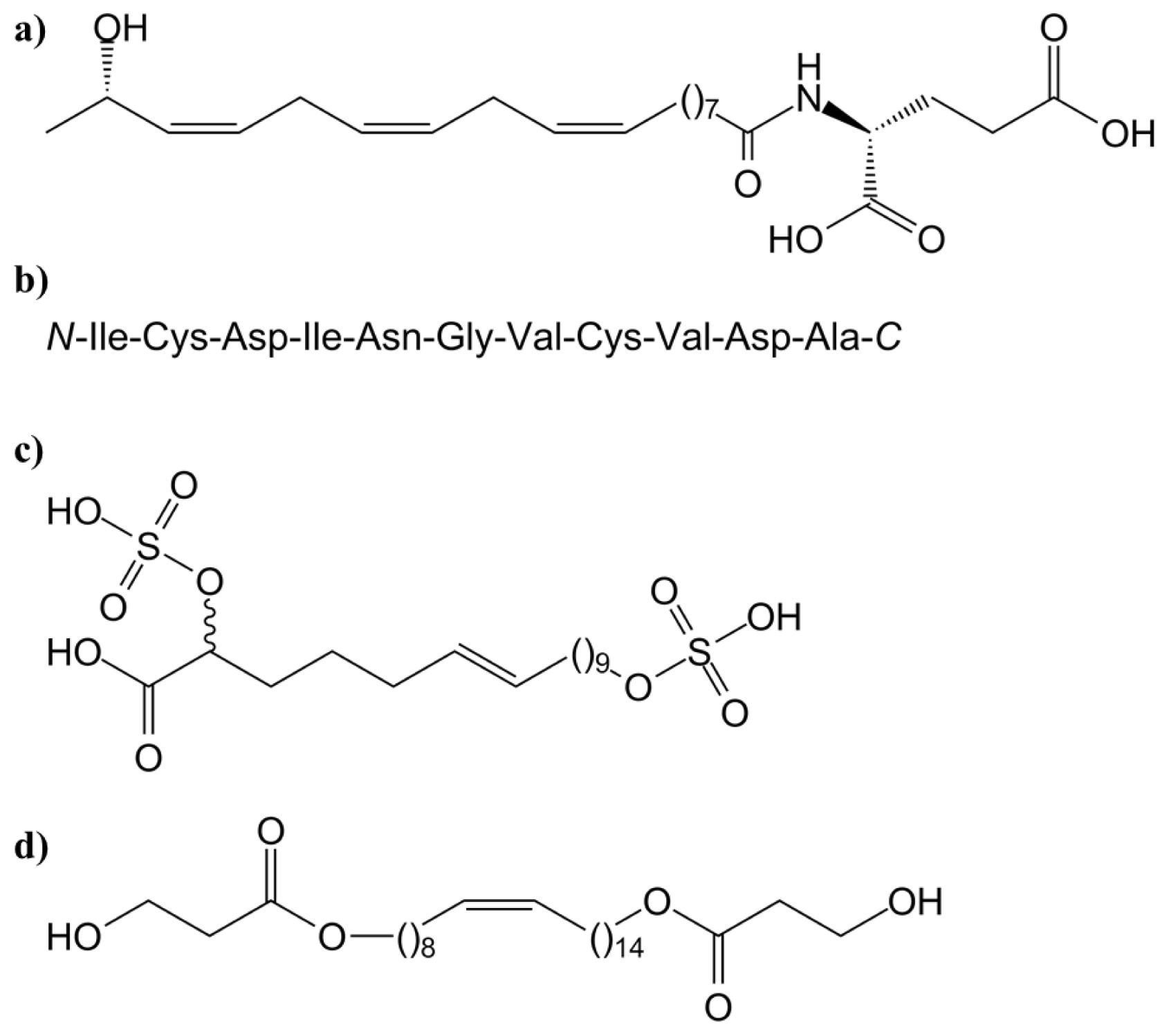
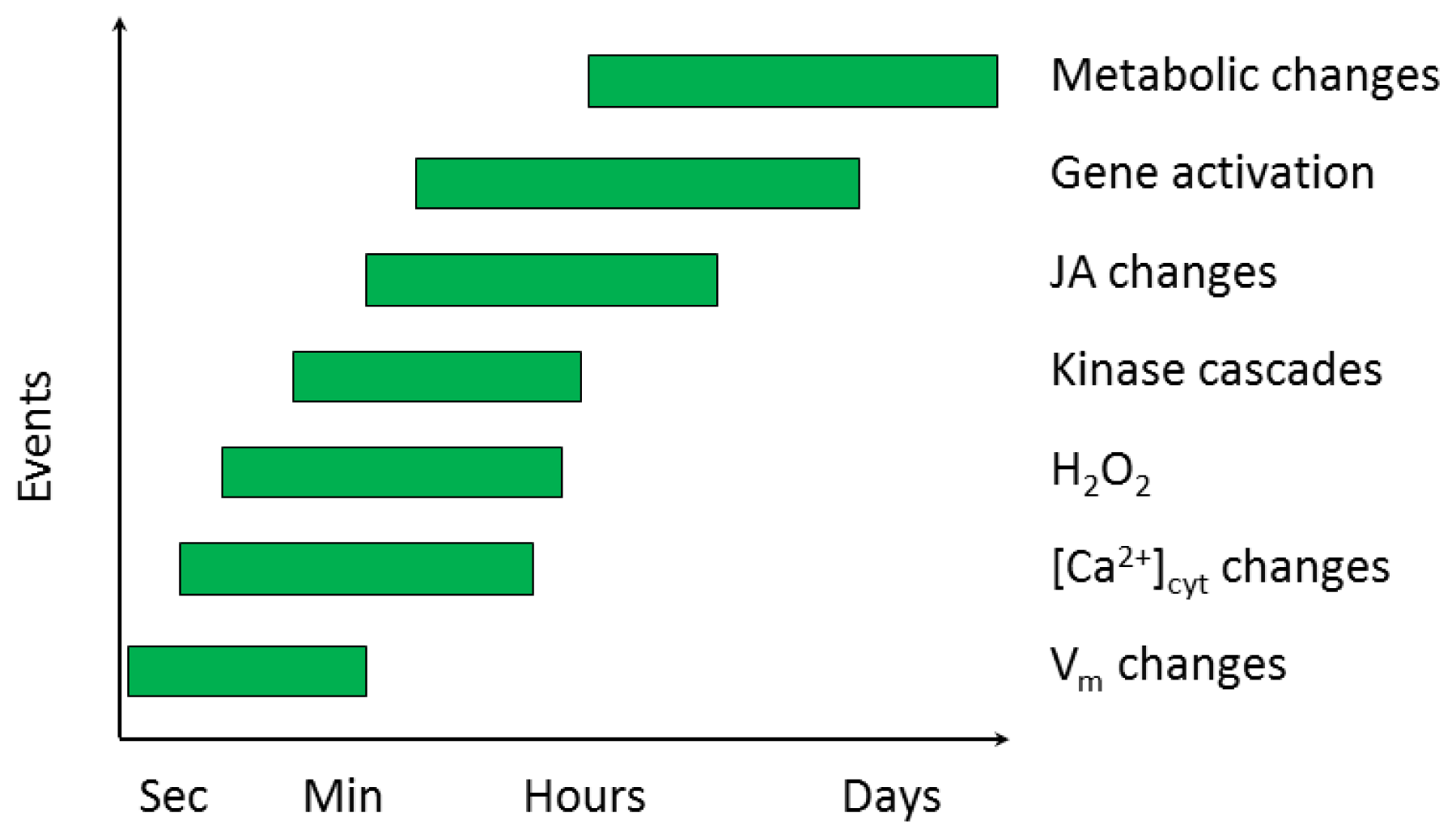
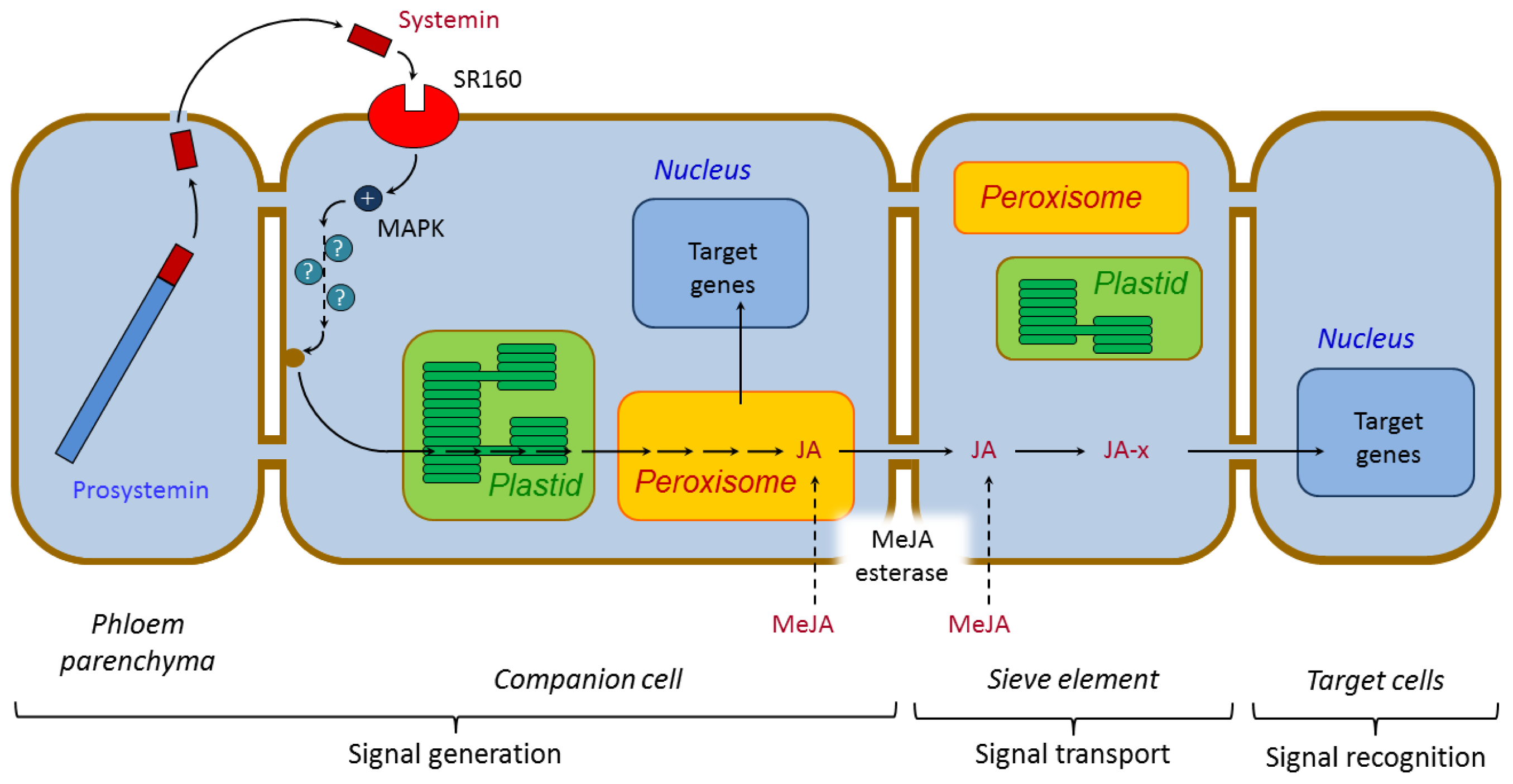
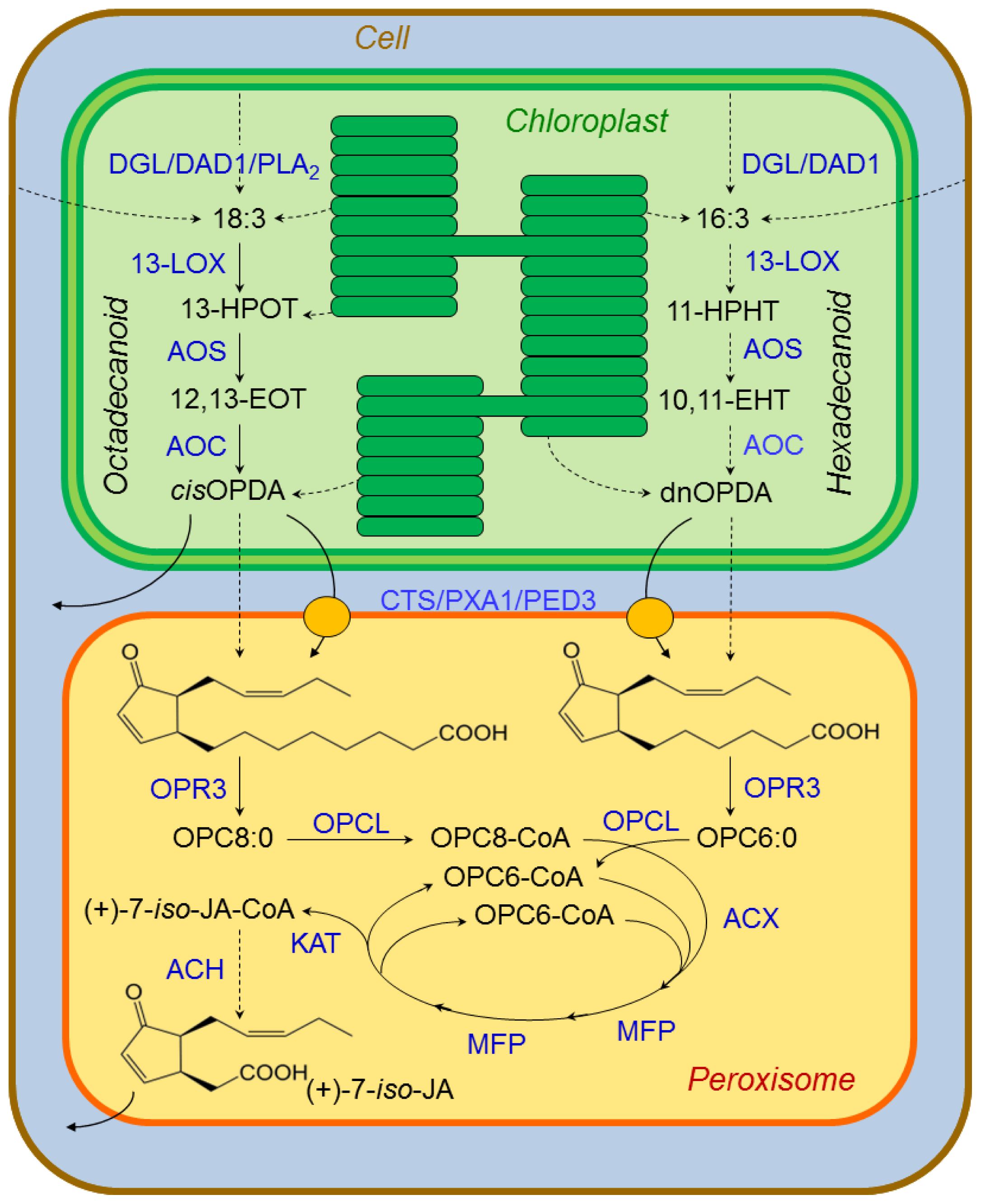
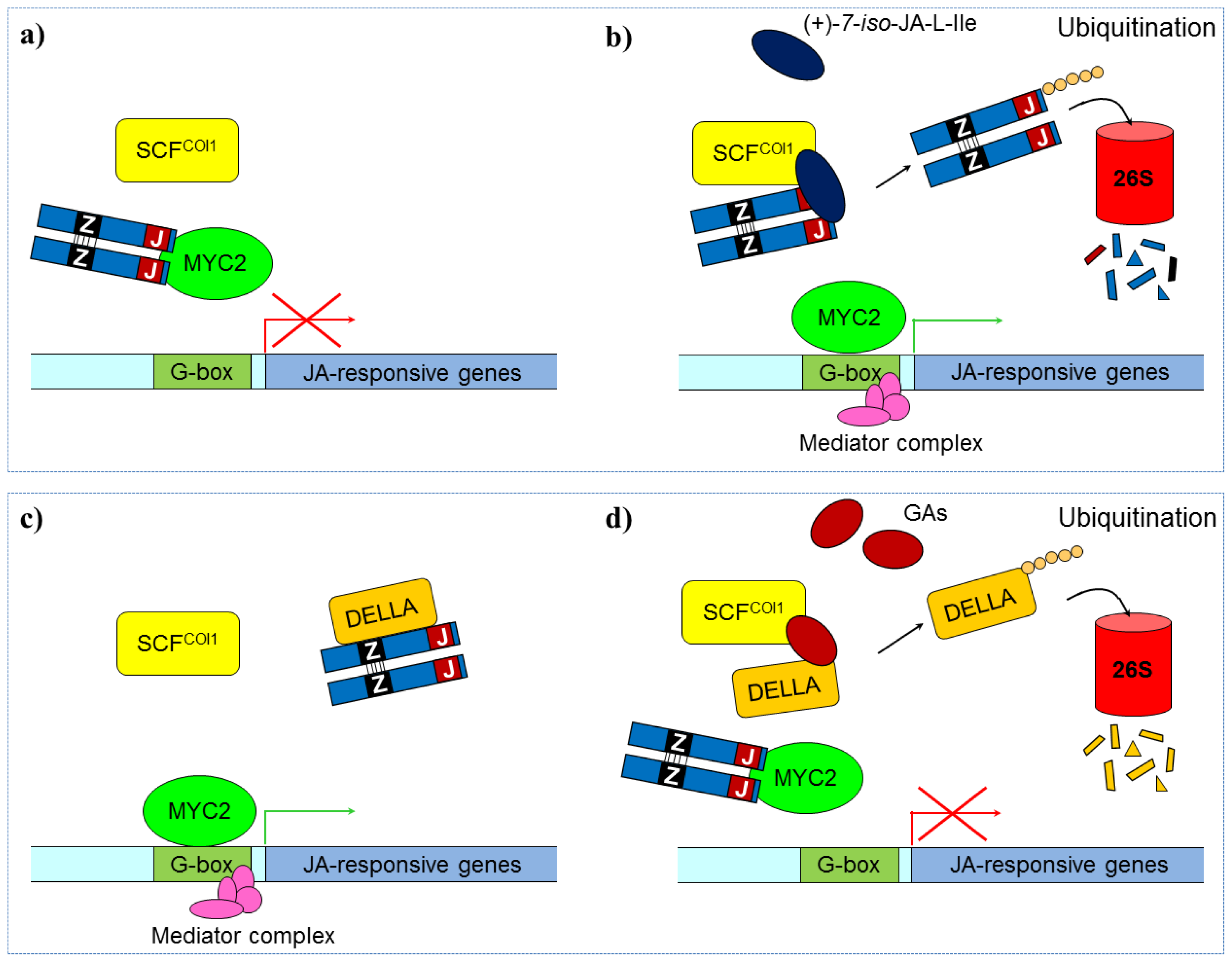
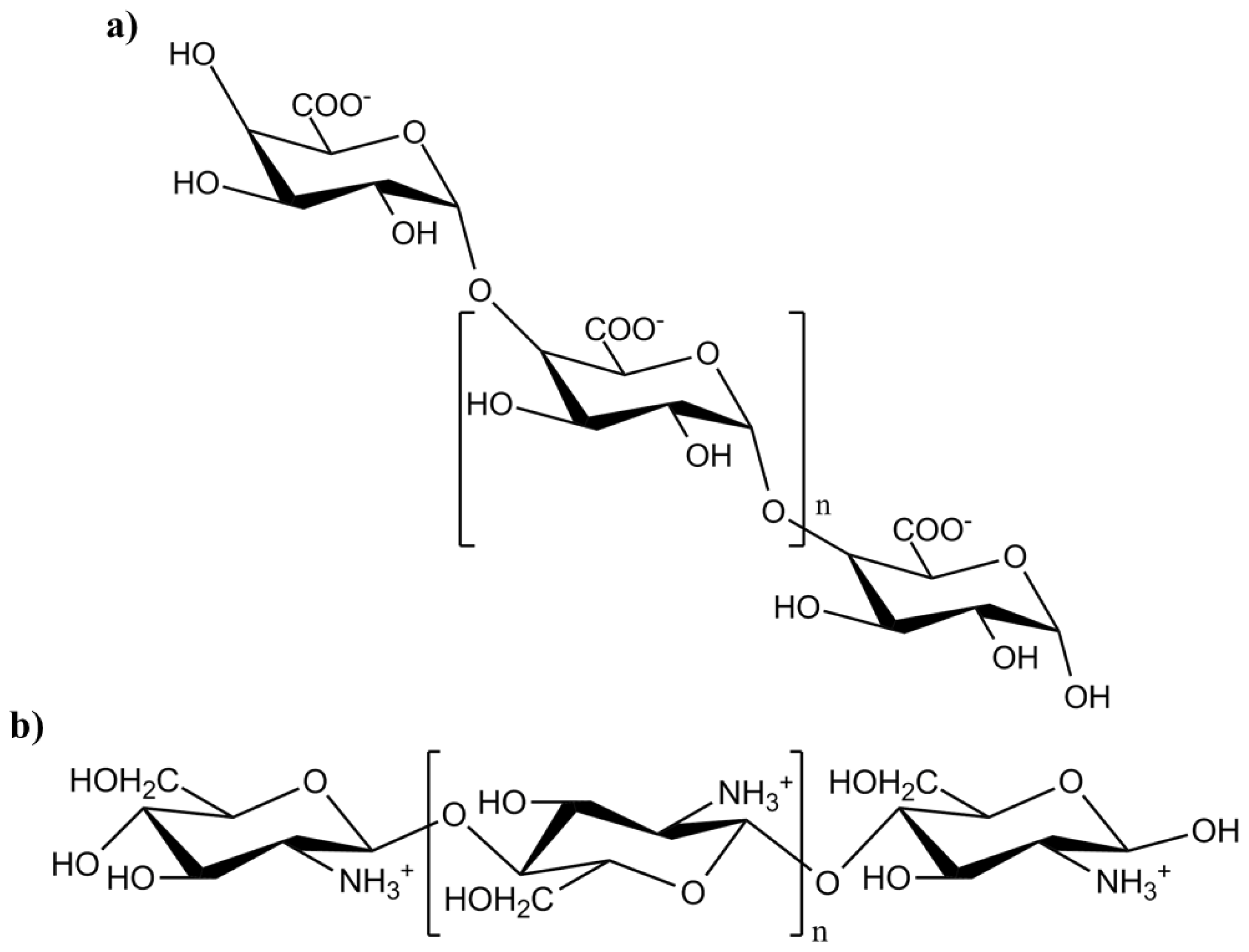
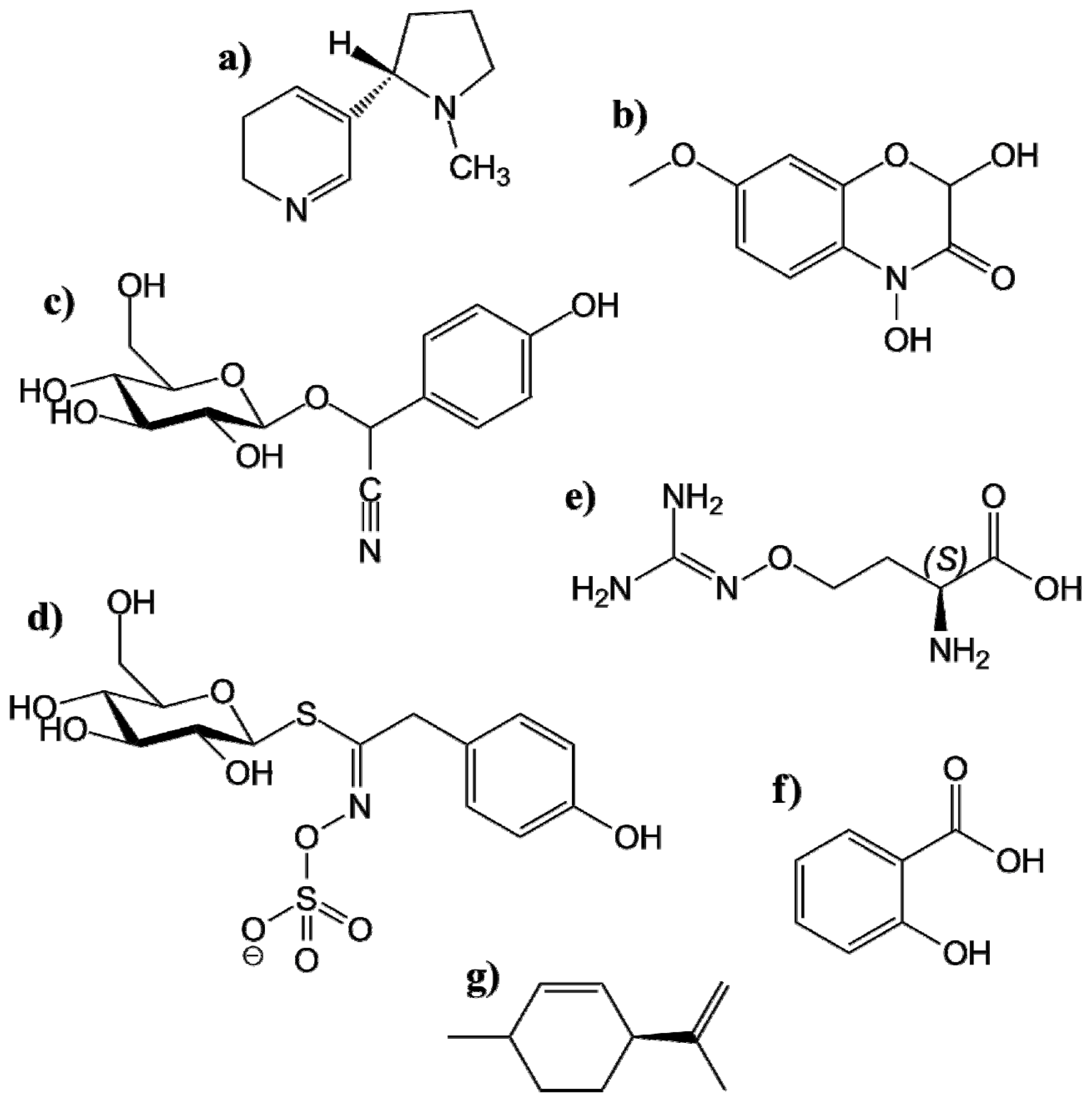
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fürstenberg-Hägg, J.; Zagrobelny, M.; Bak, S. Plant Defense against Insect Herbivores. Int. J. Mol. Sci. 2013, 14, 10242-10297. https://doi.org/10.3390/ijms140510242
Fürstenberg-Hägg J, Zagrobelny M, Bak S. Plant Defense against Insect Herbivores. International Journal of Molecular Sciences. 2013; 14(5):10242-10297. https://doi.org/10.3390/ijms140510242
Chicago/Turabian StyleFürstenberg-Hägg, Joel, Mika Zagrobelny, and Søren Bak. 2013. "Plant Defense against Insect Herbivores" International Journal of Molecular Sciences 14, no. 5: 10242-10297. https://doi.org/10.3390/ijms140510242
APA StyleFürstenberg-Hägg, J., Zagrobelny, M., & Bak, S. (2013). Plant Defense against Insect Herbivores. International Journal of Molecular Sciences, 14(5), 10242-10297. https://doi.org/10.3390/ijms140510242



