Exosomes as Intercellular Signaling Organelles Involved in Health and Disease: Basic Science and Clinical Applications
Abstract
:1. Introduction
2. Origin, Molecular Composition and Delivery of Exosomes to Target Cells
3. Exosomes in Cell Physiology
3.1. Exosomes as Immune-Modulators
3.2. Exosomes in Biologic Fluids
3.3. Exosomes and Genetic Materials: RNA Delivery and the Role of miRNA
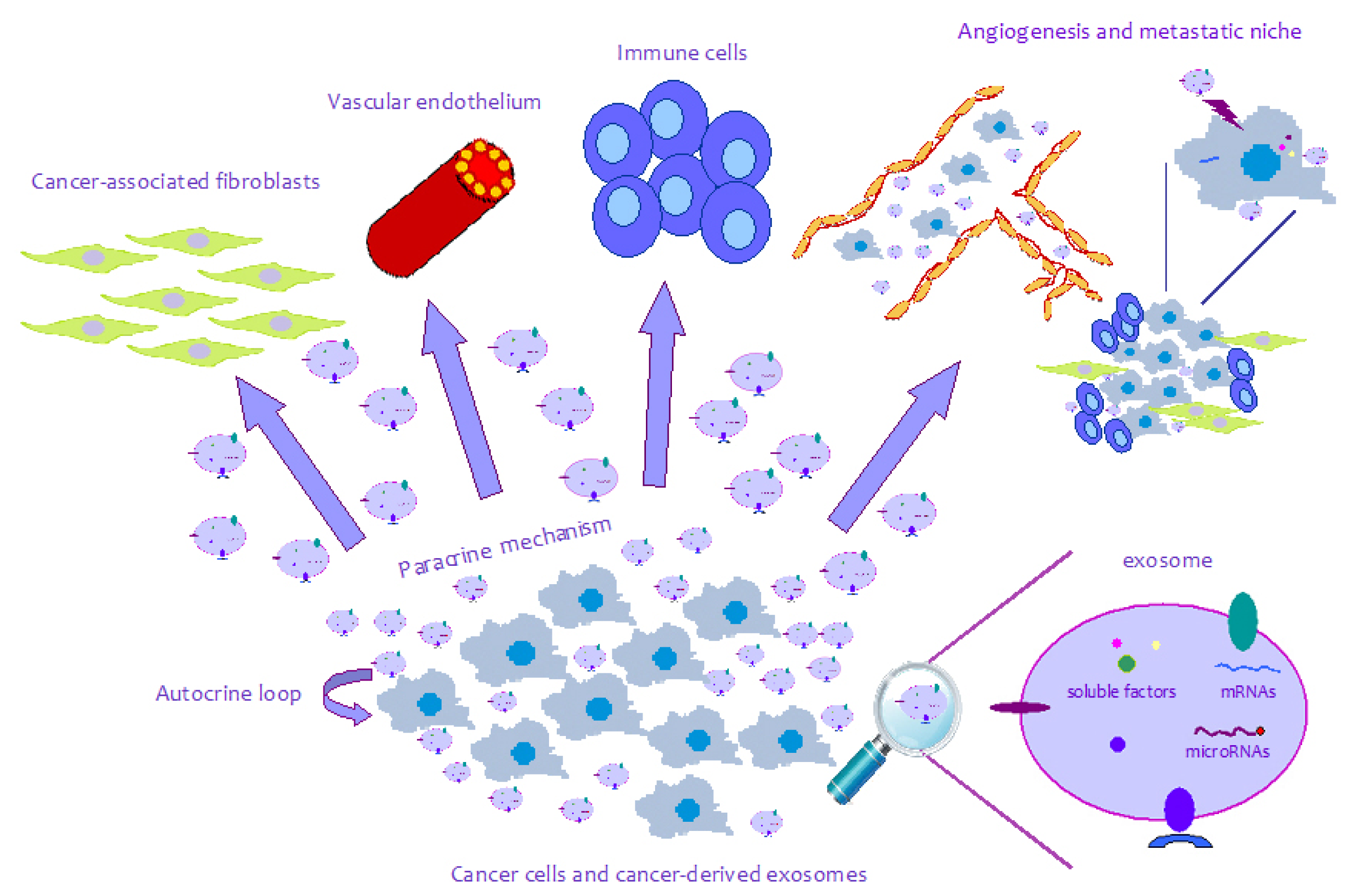
4. Exosomes in Pathology: Exosomes and Cancer
5. Exosomes in Pathology: Exosomes and Neurological, Cardiovascular and Rheumatologic Diseases
6. Exosomes in Clinical Diagnostic and Therapeutic Approaches
6.1. Diagnostics and Companion Diagnostics
6.2. Therapy and Regenerative Medicine
6.3. Immunotherapy and Vaccines
Acknowledgements
Conflict of Interest
References
- Alessandro, R.; Kohn, E.C. Signal transduction targets in invasion. Clin. Exp. Metastasis 2002, 19, 265–273. [Google Scholar]
- Simons, M.; Raposo, G. Exosomes—Vesicular carriers for intercellular communication. Curr. Opin. Cell Biol 2009, 21, 575–581. [Google Scholar]
- Mathivanan, S.; Ji, H.; Simpson, R.J. Exosomes: Extracellular organelles important in intercellular communication. J. Proteomics 2010, 73, 1907–1920. [Google Scholar]
- Harding, C.; Heuser, J.; Stahl, P. Receptor-mediated endocytosis of transferrin and recycling of the transferrin receptor in rat reticulocytes. J. Cell Biol 1983, 97, 329–339. [Google Scholar]
- Pan, B.T.; Teng, K.; Wu, C.; Adam, M.; Johnstone, R.M. Electron microscopic evidence for externalization of the transferrin receptor in vesicular form in sheep reticulocytes. J. Cell Biol 1985, 101, 942–948. [Google Scholar]
- Trams, E.G.; Lauter, C.J.; Salem, N., Jr; Ursula, H. Exfoliation of membrane ecto-enzymes in the form of micro-vesicles. Biochim. Biophys. Acta 1981, 645, 63–70. [Google Scholar]
- Johnstone, R.M.; Adam, M.; Hammond, J.R.; Orr, L.; Turbide, C. Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes). J. Biol. Chem 1987, 262, 9412–9420. [Google Scholar]
- Raposo, G.; Nijman, H.W.; Stoorvogel, W.; Liejendekker, R.; Harding, C.V.; Melief, C.J.; Geuze, H.J. B lymphocytes secrete antigen-presenting vesicles. J. Exp. Med 1996, 183, 1161–1172. [Google Scholar]
- Zech, D.; Rana, S.; Buchler, M.W.; Zoller, M. Tumor-exosomes and leukocyte activation: An ambivalent crosstalk. Cell Commun. Signal 2012, 10, 37. [Google Scholar]
- Thery, C.; Regnault, A.; Garin, J.; Wolfers, J.; Zitvogel, L.; Ricciardi-Castagnoli, P.; Raposo, G.; Amigorena, S. Molecular characterization of dendritic cell-derived exosomes. Selective accumulation of the heat shock protein hsc73. J. Cell Biol 1999, 147, 599–610. [Google Scholar]
- Carroll-Portillo, A.; Surviladze, Z.; Cambi, A.; Lidke, D.S.; Wilson, B.S. Mast cell synapses and exosomes: Membrane contacts for information exchange. Front. Immunol 2012, 3, 46. [Google Scholar]
- Lai, R.C.; Arslan, F.; Lee, M.M.; Sze, N.S.; Choo, A.; Chen, T.S.; Salto-Tellez, M.; Timmers, L.; Lee, C.N.; El Oakley, R.M.; et al. Exosome secreted by msc reduces myocardial ischemia/reperfusion injury. Stem. Cell Res 2010, 4, 214–222. [Google Scholar]
- Mallegol, J.; van Niel, G.; Lebreton, C.; Lepelletier, Y.; Candalh, C.; Dugave, C.; Heath, J.K.; Raposo, G.; Cerf-Bensussan, N.; Heyman, M. T84-intestinal epithelial exosomes bear mhc class II/peptide complexes potentiating antigen presentation by dendritic cells. Gastroenterology 2007, 132, 1866–1876. [Google Scholar]
- Kapsogeorgou, E.K.; Abu-Helu, R.F.; Moutsopoulos, H.M.; Manoussakis, M.N. Salivary gland epithelial cell exosomes: A source of autoantigenic ribonucleoproteins. Arthritis Rheum 2005, 52, 1517–1521. [Google Scholar]
- Wang, G.; Dinkins, M.; He, Q.; Zhu, G.; Poirier, C.; Campbell, A.; Mayer-Proschel, M.; Bieberich, E. Astrocytes secrete exosomes enriched with proapoptotic ceramide and prostate apoptosis response 4 (par-4): Potential mechanism of apoptosis induction in Alzheimer disease (ad). J. Biol. Chem 2012, 287, 21384–21395. [Google Scholar]
- Hergenreider, E.; Heydt, S.; Treguer, K.; Boettger, T.; Horrevoets, A.J.; Zeiher, A.M.; Scheffer, M.P.; Frangakis, A.S.; Yin, X.; Mayr, M.; et al. Atheroprotective communication between endothelial cells and smooth muscle cells through mirnas. Nat. Cell Biol 2012, 14, 249–256. [Google Scholar]
- Ristorcelli, E.; Beraud, E.; Verrando, P.; Villard, C.; Lafitte, D.; Sbarra, V.; Lombardo, D.; Verine, A. Human tumor nanoparticles induce apoptosis of pancreatic cancer cells. FASEB J 2008, 22, 3358–3369. [Google Scholar]
- Clayton, A.; Mitchell, J.P.; Court, J.; Linnane, S.; Mason, M.D.; Tabi, Z. Human tumor-derived exosomes down-modulate nkg2d expression. J. Immunol 2008, 180, 7249–7258. [Google Scholar]
- Peinado, H.; Aleckovic, M.; Lavotshkin, S.; Matei, I.; Costa-Silva, B.; Moreno-Bueno, G.; Hergueta-Redondo, M.; Williams, C.; Garcia-Santos, G.; Ghajar, C.; et al. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through met. Nat. Med 2012, 18, 883–891. [Google Scholar]
- Al-Nedawi, K.; Meehan, B.; Rak, J. Microvesicles: Messengers and mediators of tumor progression. Cell Cycle 2009, 8, 2014–2018. [Google Scholar]
- Taverna, S.; Flugy, A.; Saieva, L.; Kohn, E.C.; Santoro, A.; Meraviglia, S.; de Leo, G.; Alessandro, R. Role of exosomes released by chronic myelogenous leukemia cells in angiogenesis. Int. J. Cancer 2012, 130, 2033–2043. [Google Scholar]
- Dear, J.W.; Street, J.M.; Bailey, M.A. Urinary exosomes: A reservoir for biomarker discovery and potential mediators of intra-renal signaling. Proteomics 2012. [Google Scholar] [CrossRef]
- Gallo, A.; Tandon, M.; Alevizos, I.; Illei, G.G. The majority of micrornas detectable in serum and saliva is concentrated in exosomes. PLoS One 2012, 7, e30679. [Google Scholar]
- Torregrosa Paredes, P.; Esser, J.; Admyre, C.; Nord, M.; Rahman, Q.K.; Lukic, A.; Radmark, O.; Gronneberg, R.; Grunewald, J.; Eklund, A.; et al. Bronchoalveolar lavage fluid exosomes contribute to cytokine and leukotriene production in allergic asthma. Allergy 2012, 67, 911–919. [Google Scholar]
- Wahlgren, J.; de Karlson, L.T.; Brisslert, M.; vaziri Sani, F.; Telemo, E.; Sunnerhagen, P.; Valadi, H. Plasma exosomes can deliver exogenous short interfering rna to monocytes and lymphocytes. Nucleic Acids Res 2012, 40, e130. [Google Scholar]
- Skriner, K.; Adolph, K.; Jungblut, P.; Burmester, G. Association of citrullinated proteins with synovial exosomes. Arthritis Rheum 2006, 54, 3809–3814. [Google Scholar]
- Mathivanan, S.; Simpson, R.J. Exocarta: A compendium of exosomal proteins and RNA. Proteomics 2009, 9, 4997–5000. [Google Scholar]
- Savina, A.; Fader, C.M.; Damiani, M.T.; Colombo, M.I. Rab11 promotes docking and fusion of multivesicular bodies in a calcium-dependent manner. Traffic 2005, 6, 131–143. [Google Scholar]
- Hurley, J.H.; Odorizzi, G. Get on the exosome bus with alix. Nat. Cell Biol 2012, 14, 654–655. [Google Scholar]
- Petersen, S.H.; Odintsova, E.; Haigh, T.A.; Rickinson, A.B.; Taylor, G.S.; Berditchevski, F. The role of tetraspanin cd63 in antigen presentation via mhc class II. Eur. J. Immunol 2011, 41, 2556–2561. [Google Scholar]
- Lugini, L.; Cecchetti, S.; Huber, V.; Luciani, F.; Macchia, G.; Spadaro, F.; Paris, L.; Abalsamo, L.; Colone, M.; Molinari, A.; et al. Immune surveillance properties of human nk cell-derived exosomes. J. Immunol 2012, 189, 2833–2842. [Google Scholar]
- Mathivanan, S.; Lim, J.W.; Tauro, B.J.; Ji, H.; Moritz, R.L.; Simpson, R.J. Proteomics analysis of a33 immunoaffinity-purified exosomes released from the human colon tumor cell line lim1215 reveals a tissue-specific protein signature. Mol. Cell Proteomics 2010, 9, 197–208. [Google Scholar]
- Faure, J.; Lachenal, G.; Court, M.; Hirrlinger, J.; Chatellard-Causse, C.; Blot, B.; Grange, J.; Schoehn, G.; Goldberg, Y.; Boyer, V.; et al. Exosomes are released by cultured cortical neurones. Mol. Cell Neurosci 2006, 31, 642–648. [Google Scholar]
- Yang, J.M.; Gould, S.J. The cis-acting signals that target proteins to exosomes and microvesicles. Biochem. Soc. Trans 2013, 41, 277–82. [Google Scholar]
- Shen, B.; Wu, N.; Yang, J.M.; Gould, S.J. Protein targeting to exosomes/microvesicles by plasma membrane anchors. J. Biol. Chem 2011, 286, 14383–95. [Google Scholar]
- Fehon, R.G.; McClatchey, A.I.; Bretscher, A. Organizing the cell cortex: The role of ERM proteins. Nat. Rev. Mol. Cell Biol 2010, 11, 276–87. [Google Scholar]
- Rana, S.; Yue, S.; Stadel, D.; Zoller, M. Toward tailored exosomes: The exosomal tetraspaninwebcontributes to target cell selection. Int. J. Biochem. Cell Biol 2012, 44, 1574–1584. [Google Scholar]
- Nabhan, J.F.; Hu, R.; Oh, R.S.; Cohen, S.N.; Lu, Q. Formation and release of arrestin domain-containing protein 1-mediated microvesicles (ARMMs) at plasma membrane by recruitment of TSG101 protein. Proc. Natl. Acad. Sci. USA 2012, 109, 4146–4151. [Google Scholar]
- Baietti, M.F.; Zhang, Z.; Mortier, E.; Melchior, A.; Degeest, G.; Geeraerts, A.; Ivarsson, Y.; Depoortere, F.; Coomans, C.; Vermeiren, E.; et al. Syndecan-syntenin-ALIX regulates the biogenesis of exosomes. Nat. Cell Biol 2012, 14, 677–85. [Google Scholar]
- Geminard, C.; de Gassart, A.; Blanc, L.; Vidal, M. Degradation of ap2 duringreticulocyte maturationenhances binding of hsc70 and alix to a common site on tfr for sorting into exosomes. Traffic 2004, 5, 181–193. [Google Scholar]
- Irion, U.; St Johnston, D. Bicoid RNA localization requires specific binding of an endosomal sorting complex. Nature 2007, 445, 554–558. [Google Scholar]
- Turchinovich, A.; Weiz, L.; Langheinz, A.; Burwinkel, B. Characterization of extracellular circulating microrna. Nucleic Acids Res 2011, 398, 7223–7233. [Google Scholar]
- Savina, A.; Furlan, M.; Vidal, M.; Colombo, M.I. Exosome release is regulated by a calcium-dependent mechanism in k562 cells. J. Biol. Chem 2003, 278, 20083–20090. [Google Scholar]
- Yu, X.; Riley, T.; Levine, A.J. The regulation of the endosomal compartment by p53 the tumor suppressor gene. FEBS J 2009, 276, 2201–2212. [Google Scholar]
- Feng, D.; Zhao, W.L.; Ye, Y.Y.; Bai, X.C.; Liu, R.Q.; Chang, L.F.; Zhou, Q.; Sui, S.F. Cellular internalization of exosomes occurs through phagocytosis. Traffic 2010, 11, 675–687. [Google Scholar]
- Parolini, I.; Federici, C.; Raggi, C.; Lugini, L.; Palleschi, S.; de Milito, A.; Coscia, C.; Iessi, E.; Logozzi, M.; Molinari, A.; et al. Microenvironmental ph is a key factor for exosome traffic in tumor cells. J. Biol. Chem 2009, 284, 34211–34222. [Google Scholar]
- Montecalvo, A.; Larregina, A.T.; Shufesky, W.J.; Stolz, D.B.; Sullivan, M.L.; Karlsson, J.M.; Baty, C.J.; Gibson, G.A.; Erdos, G.; Wang, Z.; et al. Mechanism of transfer of functional micrornas between mouse dendritic cells via exosomes. Blood 2012, 119, 756–766. [Google Scholar]
- Segura, E.; Guerin, C.; Hogg, N.; Amigorena, S.; Thery, C. Cd8+ dendritic cells use lfa-1 to capture mhc-peptide complexes from exosomes in vivo. J. Immunol 2007, 179, 1489–1496. [Google Scholar]
- Calzolari, A.; Raggi, C.; Deaglio, S.; Sposi, N.M.; Stafsnes, M.; Fecchi, K.; Parolini, I.; Malavasi, F.; Peschle, C.; Sargiacomo, M.; et al. TfR2 localizes in lipid raft domains and is released in exosomes to activate signal transduction along the MAPK pathway. J. Cell Sci 2006, 119, 4486–98. [Google Scholar]
- Clayton, A.; Turkes, A.; Dewitt, S.; Steadman, R.; Mason, M.D.; Hallett, M.B. Adhesion and signaling by B cell-derived exosomes: The role of integrins. FASEB J 2004, 18, 977–979. [Google Scholar]
- Muntasell, A.; Berger, A.C.; Roche, P.A. T cell-induced secretion of mhc class II-peptide complexes on b cell exosomes. EMBO J 2007, 26, 4263–4272. [Google Scholar]
- Nolte-’t Hoen, E.N.; Buschow, S.I.; Anderton, S.M.; Stoorvogel, W.; Wauben, M.H. Activated T cells recruit exosomes secreted by dendritic cells via lfa-1. Blood 2009, 113, 1977–1981. [Google Scholar]
- Karlsson, M.; Lundin, S.; Dahlgren, U.; Kahu, H.; Pettersson, I.; Telemo, E. “Tolerosomes” are produced by intestinal epithelial cells. Eur. J. Immunol. 2001, 2892–2900. [Google Scholar]
- Almqvist, N.L.A.; Hultkrantz, S.; Rask, C.; Telemo, E. Serum-derived exosomes from antigen-fed mice prevent allergic sensitization in a model of allergic asthma. Immunology 2008, 125, 21–27. [Google Scholar]
- Frängsmyr, L.; Baranov, V.; Nagaeva, O.; Stendahl, U.; Kjellberg, L.; Mincheva-Nilsson, L. Cytoplasmic microvesicular form of fas ligand in human early placenta: Switching the tissue immune privilege hypothesis from cellular to vesicular level. Mol. Human Reprod 2005, 11, 35–41. [Google Scholar]
- Abrahams, V.M.; Straszewski-Chavez, S.L.; Guller, S.; Mor, G. First trimester trophoblast cells secrete fas ligand which induces immune cell apoptosis. Mol. Human Reprod 2004, 10, 55–63. [Google Scholar]
- Hedlund, M.; Stenqvist, A.C.; Nagaeva, O.; Kjellberg, L.; Wulff, M.; Baranov, V.; Mincheva-Nilsson, L. Human placenta expresses and secretes nkg2d ligands via exosomes that down-modulate the cognate receptor expression: Evidence for immunosuppressive function. J. Immul 2009, 183, 340–351. [Google Scholar]
- Reinhardt, T.A.; Lippolis, J.D.; Nonnecke, B.J.; Sacco, R.E. Bovine milk exosome proteome. J. Proteomics 2012, 75, 1486–1492. [Google Scholar]
- Hata, T.; Murakami, K.; Nakatani, H.; Yamamoto, Y.; Matsuda, T.; Aoki, N. Isolation of bovine milk-derived microvesicles carrying mrnas and micrornas. Biochem. Biophys. Res. Commun 2010, 396, 528–533. [Google Scholar]
- Zhou, Q.; Li, M.; Wang, X.; Li, Q.; Wang, T.; Zhu, Q.; Zhou, X.; Gao, X.; Li, X. Immune-related micrornas are abundant in breast milk exosomes. Int. J. Biol. Sci 2012, 8, 118–123. [Google Scholar]
- Valadi, H.; Ekström, K.; Bossios, A.; Sjöstrand, M.; Lee, J.J.; Lötvall, J.O. Exosome-mediated transfer of mrnas and micrornas is a novel mechanism of genetic exchange between cells. Nat. Cell Biol 2007, 9, 654–659. [Google Scholar]
- Luga, V.; Zhang, L.; Viloria-Petit, A.M.; Ogunjimi, A.A.; Inanlou, M.R.; Chiu, E.; Buchanan, M.; Hosein, A.N.; Basik, M.; Wrana, J.L. Exosomes mediate stromal mobilization of autocrine wnt-pcp signaling in breast cancer cell migration. Cell 2012, 51, 1542–1556. [Google Scholar]
- Fabbri, M.; Paone, A.; Calore, F.; Galli, R.; Gaudio, E.; Santhanam, R.; Lovat, F.; Fadda, P.; Mao, C.; Nuovo, G.J.; et al. Micrornas bind to toll-like receptors to induce prometastatic inflammatory response. Proc. Natl. Acad. Sci. USA 2012, 109, E2110–E2116. [Google Scholar]
- Corrado, C.; Flugy, A.; Taverna, S.; Raimondo, S.; Guggino, G.; Karmali, R.; de Leo, G.; Alessandro, R. Carboxyamidotriazole-orotate inhibits the growth of imatinib-resistant chronic myeloid leukaemia cells and modulates exosomes-stimulated angiogenesis. PLoS One 2012, 7, e42310. [Google Scholar]
- Mineo, M.; Garfield, S.; Taverna, S.; Flugy, A.; de Leo, G.; Alessandro, R.; Kohn, E. Exosomes released by k562 chronic myeloid leukemia cells promote angiogenesis in a src-dependent fashion. Angiogenesis 2012, 15, 33–45. [Google Scholar]
- Huan, J.; Hornick, N.; Skinner, A.; Goloviznina, N.; Roberts, C.; Kurre, P. RNA trafficking by acute myeloid leukemia exosomes. Cancer Res. 2012. [Google Scholar] [CrossRef]
- Surgucheva, I.; Sharov, V.; Surguchov, A. γ-synuclein: Seeding of α-synuclein aggregation and transmission between cells. Biochemistry 2012, 51, 4743–4754. [Google Scholar]
- Zhang, H.; Liu, C.; Su, K.; Yu, S.; Zhang, L.; Zhang, S.; Wang, J.; Cao, X.; Grizzle, W.; Kimberly, R. A membrane form of TNF-α presented by exosomes delays T cell activation-induced cell death. J. Immunol 2006, 176, 7385–7393. [Google Scholar]
- Wan, S.; Zhou, Z.; Duan, B.; Morel, L. Direct b cell stimulation by dendritic cells in a mouse model of lupus. Arthritis Rheum 2008, 58, 1741–1750. [Google Scholar]
- Saunderson, S.C.; Schuberth, P.C.; Dunn, A.C.; Miller, L.; Hock, B.D.; MacKay, P.A.; Koch, N.; Jack, R.W.; McLellan, A.D. Induction of exosome release in primary B cells stimulated via cd40 and the il-4 receptor. J. Immunol 2008, 180, 8146–8152. [Google Scholar]
- Zitvogel, L.; Regnault, A.; Lozier, A.; Wolfers, J.; Flament, C.; Tenza, D.; Ricciardi-Castagnoli, P.; Raposo, G.; Amigorena, S. Eradication of established murine tumors using a novel cell-free vaccine: Dendritic cell-derived exosomes. Nat. Med 1998, 4, 594–600. [Google Scholar]
- Yin, W.; Ouyang, S.; Li, Y.; Xiao, B.; Yang, H. Immature dendritic cell-derived exosomes: A promise subcellular vaccine for autoimmunity. Inflammation 2012. [Google Scholar] [CrossRef]
- Kim, S.H.; Bianco, N.; Menon, R.; Lechman, E.R.; Shufesky, W.J.; Morelli, A.E.; Robbins, P.D. Exosomes derived from genetically modified dc expressing fasl are anti-inflammatory and immunosuppressive. Mol. Ther 2006, 13, 289–300. [Google Scholar]
- Kim, S.H.; Bianco, N.R.; Shufesky, W.J.; Morelli, A.E.; Robbins, P.D. Effective treatment of inflammatory disease models with exosomes derived from dendritic cells genetically modified to express il-4. J. Immunol. 2007, 179, 2242–2249.0. [Google Scholar]
- Cai, Z.; Zhang, W.; Yang, F.; Yu, L.; Yu, Z.; Pan, J.; Wang, L.; Cao, X.; Wang, J. Immunosuppressive exosomes from TGF-β1 gene-modified dendritic cells attenuate th17-mediated inflammatory autoimmune disease by inducing regulatory t cells. Cell Res 2012, 22, 607–661. [Google Scholar]
- Pêche, H.; Renaudin, K.; Beriou, G.; Merieau, E.; Amigorena, S.; Cuturi, M.C. Induction of tolerance by exosomes and shortterm immunosuppression in a fully mhc-mismatched rat cardiac allograft model. Am. J. Transplant 2006, 6, 1541–1550. [Google Scholar]
- Yang, X.; Meng, S.; Jiang, H.; Zhu, C.; Wu, W. Exosomes derived from immature bone marrow dendritic cells induce tolerogenicity of intestinal transplantation in rats. J. Surg. Res 2011, 171, 826–832. [Google Scholar]
- Li, X.; Li, J.J.; Yang, J.Y.; Wang, D.S.; Zhao, W.; Song, W.J.; Li, W.M.; Wang, J.F.; Han, W.; Zhang, Z.C.; et al. Tolerance induction by exosomes from immature dendritic cells and rapamycin in a mouse cardiac allograft model. PLoS One 2012, 7, e44045. [Google Scholar]
- Montecalvo, A.; Shufesky, W.J.; Stolz, D.B.; Sullivan, M.G.; Wang, Z.; Divito, S.J.; Papworth, G.D.; Watkins, S.C.; Robbins, P.D.; Larregina, A.T.; et al. Exosomes as a short-range mechanism to spread alloantigen between dendritic cells during T cell allorecognition. J. Immunol 2008, 180, 3081–3090. [Google Scholar]
- Caby, M.P.; Lankar, D.; Vincendeau-Scherrer, C.; Raposo, G.; Bonnerot, C. Exosomal-like vesicles are present in human blood plasma. Int. Immunol 2005, 17, 879–887. [Google Scholar]
- Pisitkun, T.; Shen, R.F.; Knepper, M.A. Identification and proteomic profiling of exosomes in human urine. Proc. Natl. Acad. Sci. USA 2004, 101, 13368–13373. [Google Scholar]
- Gonzales, P.A.; Pisitkun, T.; Hoffert, J.D.; Tchapyjnikov, D.; Star, R.A.; Kleta, R.; Wang, N.S.; Knepper, M.A. Large-scale proteomics and phosphoproteomics of urinary exosomes. 2009, 20, 363–379. [Google Scholar]
- Ogawa, Y.; Kanai-Azuma, M.; Akimoto, Y.; Kawakami, H.; Yanoshita, R. Exosome-like vesicles with dipeptidyl peptidase iv in human saliva. Biol. Pharm. Bull 2008, 31, 1059–1062. [Google Scholar]
- Admyre, C.; Johansson, S.M.; Qazi, K.R.; Filen, J.J.; Lahesmaa, R.; Norman, M.; Neve, E.P.A.; Scheynius, A.; Gabrielsson, S. Exosomes with immune modulatory features are present in human breast milk. J. Immunol 2007, 179, 1969–1978. [Google Scholar]
- Admyre, C.; Grunewald, J.; Thyberg, J.; Gripenbäck, S.; Tornling, G.; Eklund, A.; Scheynius, A.; Gabrielsson, S. Exosomes with major histocompatibility complex class II and co-stimulatory molecules are present in human bal fluid. Eur. Respir. J 2003, 22, 578–583. [Google Scholar]
- Gatti, J.L.; Métayer, S.; Belghazi, M.; Dacheux, F.; Dacheux, J.L. Identification, proteomic profiling, and origin of ram epididymal fluid exosome-like vesicles. Biol. Reprod 2005, 72, 1452–1465. [Google Scholar]
- Gonzalez-Begne, M.; Lu, B.; Han, X.; Hagen, F.K.; Hand, A.R.; Melvin, J.E.; Yates, J.R. Proteomic analysis of human parotid gland exosomes by multidimensional protein identification technology (mudpit). J. Prot. Res 2009, 8, 1304–1314. [Google Scholar]
- Tarazona, R.; Delgado, E.; Guarnizo, M.C.; Roncero, R.G.; Morgado, S.; Sánchez-Correa, B.; Gordillo, J.J.; Dejulián, J.; Casado, J.G. Human prostasomes express cd48 and interfere with nk cell function. Immunobiology 2011, 216, 41–46. [Google Scholar]
- Taylor, D.D.; Akyol, S.; Gercel-Taylor, C. Pregnancy-associated exosomes and their modulation of T cell signaling. J. Immunol 2006, 176, 1534–1542. [Google Scholar]
- Lässer, C.; Alikhani, V.S.; Ekström, K.; Eldh, M.; Paredes, P.T.; Bossios, A.; Sjöstrand, M.; Gabrielsson, S.; Lötvall, J.; Valadi, H. Human saliva, plasma and breast milk exosomes contain rna: Uptake by macrophages. J. Transl. Med 2011, 9, 1–8. [Google Scholar]
- Xiao, H.; Wonga, D.T. Proteomic analysis of microvesicles in human saliva by gel electrophoresis with liquid chromatography-mass spectrometry. Anal. Chim. Acta 2012, 723, 61–67. [Google Scholar]
- Kim, S.H.; Bianco, N.R.; Shufesky, W.J.; Morelli, A.E.; Robbins, P.D. Mhc class II+ exosomes in plasma suppress inflammation in an antigen-specific and fas ligand/fas-dependent manner. J. Immunol 2007, 179, 2235–2241. [Google Scholar]
- Ostman, S.; Taube, M.; Telemo, E. Tolerosome-induced oral tolerancies mhc dependent. Immunology 2005, 116, 464–476. [Google Scholar]
- Prado, N.; Marazuel, E.; Segura, E.; Fernández-García, H.; Villalba, M.; Théry, C.; Rodríguez, R.; Batanero, E. Exosomes from bronchoalveolar fluid of tolerized mice prevent allergic reaction. J. Immunol 2008, 181, 1519–1525. [Google Scholar]
- Street, J.; Birkhoff, W.; Menzies, R.; Webb, D.; Bailey, M.; Dear, J. Exosomal transmission of functional aquaporin 2 in kidney cortical collecting duct cells. J. Physiol 2011, 589, 6119–6127. [Google Scholar]
- Wang, Z.; Hill, S.; Luther, J.; Hachey, D.; Schey, K. Proteomic analysis of urine exosomes by multidimensional protein identification technology (mudpit). Proteomics 2012, 12, 329–338. [Google Scholar]
- Belting, M.; Wittrup, A. Nanotubes, exosomes, and nucleic acid-binding peptides provide novel mechanisms of intercellular communication in eukaryotic cells: Implications in health and disease. J. Cell Biol 2008, 183, 1187–1191. [Google Scholar]
- Wang, K.; Zhang, S.; Weber, J.; Baxter, D.; Galas, D.J. Export of micrornas and microrna-protective protein by mammalian cells. Nucleic Acids Res 2010, 38, 7248–7259. [Google Scholar]
- Chen, X.; Liang, H.; Zhang, J.; Zen, K.; Zhang, C.Y. Horizontal transfer of micrornas: Molecular mechanisms and clinical applications. Protein Cell 2012, 3, 28–37. [Google Scholar]
- Eldh, M.; Ekström, K.; Valadi, H.; Sjöstrand, M.; Olsson, B.; Jernås, M.; Lötvall, J. Exosomes communicate protective messages during oxidative stress; possible role of exosomal shuttle RNA. PLoS One 2010, 5, 1–8. [Google Scholar]
- Mittelbrunn, M.; Gutierrez-Vazquez, C.; Villarroya-Beltri, C.; Gonzalez, S.; Sanchez-Cabo, F.; Gonzalez, M.A.; Bernad, A.; Sanchez-Madrid, F. Unidirectional transfer of microrna-loaded exosomes from t cells to antigen-presenting cells. Nat. Commun 2011, 2, 282–292. [Google Scholar]
- Li, Q.J.; Chau, J.; Ebert, P.J.; Sylvester, G.; Min, H.; Liu, G.; Braich, R.; Manoharan, M.; Soutschek, J.; Skare, P.; et al. Mir-181a is an intrinsic modulator of t cell sensitivity and selection. Cell 2007, 129, 147–161. [Google Scholar]
- Chen, C.Z.; Li, L.; Lodish, H.F.; Bartel, D.P. Micrornas modulate hematopoietic lineage differentiation. Science 2004, 303, 83–86. [Google Scholar]
- Vigorito, E.; Perks, K.L.; Abreu-Goodger, C.; Bunting, S.; Xiang, Z.; Kohlhaas, S.; Das, P.P.; Miska, E.A.; Rodriguez, A.; Bradley, A.; et al. Microrna-155 regulates the generation of immunoglobulin class-switched plasma cells. Immunity 2007, 27, 847–859. [Google Scholar]
- Kosaka, N.I.H.; Sekine, K.; Ochiya, T. Microrna as a new immune-regulatory breast milk. Silence 2010, 1, 1–8. [Google Scholar]
- Hass, R.; Otte, A. Mesenchymal stem cells as all-round supporters in a normal and neoplastic microenvironment. Cell Commun. Signal 2012, 10, 26–39. [Google Scholar]
- Taylor, D.D.; Gercel-Taylor, C. Exosomes/microvesicles: Mediators of cancer-associated immunosuppressive microenvironments. Semin. Immunopathol 2011, 33, 441–454. [Google Scholar]
- Zhu, W.X.W.; Jiang, R.; Qian, H.; Chen, M.; Hu, J.; Cao, W.; Han, C.; Chen, Y. Mesenchymal stem cells derived from bone marrow favor tumor cell growth in vivo. Exp. Mol. Pathol 2006, 80, 267–274. [Google Scholar]
- Karnoub, A.E.; Dash, A.B.; Vo, A.P.; Sullivan, A.; Brooks, M.W.; Bell, G.W.; Richardson, A.L.; Polyak, K.; Tubo, R.; Weinberg, R.A. Mesenchymal stem cells within tumor stroma promote breast cancer metastasis. Nature 2007, 449, 557–563. [Google Scholar]
- Shinagawa, K.; Kitadai, Y.; Tanaka, M.; Sumida, T.; Kodama, M.; Higashi, Y.; Tanaka, S.; Yasui, W.; Chayama, K. Mesenchymal stem cells enhance growth and metastasis of colon cancer. Int. J. Cancer 2010, 127, 2323–2333. [Google Scholar]
- Psaila, B.; Lyden, D. The metastatic niche: Adapting the foreign soil. Nat. Rev. Cancer 2009, 9, 285–293. [Google Scholar]
- Zhu, W.; Huang, L.; Li, Y.; Zhang, X.; Gu, J.; Yan, Y.; Xu, X.; Wang, M.; Qian, H.; Xu, W. Exosomes derived from human bone marrow mesenchymal stem cells promote tumor growth in vivo. Cancer Lett 2012, 315, 28–37. [Google Scholar]
- Demory Beckler, M.; Higginbotham, J.; Franklin, J.; Ham, A.-J.; Halvey, P.; Imasuen, I.; Whitwell, C.; Li, M.; Liebler, D.; Coffey, R.J. Proteomic analysis of exosomes from mutant kras colon cancer cells identifies intercellular transfer of mutant kras. Mol. Cell. Proteomics 2012, 12, 343–355. [Google Scholar]
- Qu, J.; Qu, X.; Zhao, M.; Teng, Y.; Zhang, Y.; Hou, K.; Jiang, Y.; Yang, X.; Liu, Y. Gastric cancer exosomes promote tumor cell proliferation through pi3k/akt and mapk/erk activation. Digest. Liver Dis 2009, 41, 875–880. [Google Scholar]
- Umezu, T.; Ohyashiki, K.; Kuroda, M.; Ohyashiki, J. Leukemia cell to endothelial cell communication via exosomal mirnas. Oncogene 2012. [Google Scholar] [CrossRef]
- Harris, A.L. Hypoxia—A key regulatory factor in tumor growth. Nat. Rev. Cancer 2002, 2, 38–47. [Google Scholar]
- Kai, R.; Gang, S.; Gaoliang, O. Role of hypoxia in the hallmarks of human cancer. J. Cell. Biochem 2009, 107, 1053–1062. [Google Scholar]
- King, H.W.; Michael, M.Z.; Gleadle, J.M. Hypoxic enhancement of exosome release by breast cancer cells. BMC Cancer 2012, 12, 421. [Google Scholar] [Green Version]
- Crosby, M.E.K.R.; Ivan, M.; Glazer, P.M. Microrna regulation of DNA repair gene expression in hypoxic stress. Cancer Res 2009, 69, 1221–1229. [Google Scholar]
- Park, J.E.; Tan, H.S.; Datta, A.; Lai, R.C.; Zhang, H.; Meng, W.; Lim, S.K.; Sze, S.K. Hypoxic tumor cell modulates its microenvironment to enhance angiogenic and metastatic potential by secretion of proteins and exosomes. Mol. Cell. Proteomics 2010, 9, 1085–1099. [Google Scholar]
- Camps, C.; Buffa, F.M.; Colella, S.; Moore, J.; Sotiriou, C.; Sheldon, H.; Harris, A.L.; Gleadle, J.M.; Ragoussis, J. Hsa-mir-210 is induced by hypoxia and is an independent prognostic factor in breast cancer. Clin. Cancer Res 2008, 14, 1340–1348. [Google Scholar]
- Safaei, R.; Larson, B.; Cheng, T.; Gibson, M.; Otani, S.; Naerdemann, W.; Howell, S. Abnormal lysosomal trafficking and enhanced exosomal export of cisplatin in drug-resistant human ovarian carcinoma cells. Mol. Cancer Ther 2005, 4, 1595–1604. [Google Scholar]
- Yan, X.; Yin, J.; Yao, H.; Mao, N.; Yang, Y.; Pan, L. Increased expression of annexin a3 is a mechanism of platinum resistance in ovarian cancer. Cancer Res 2010, 70, 1616–1624. [Google Scholar]
- Yin, J.; Yan, X.; Yao, X.; Zhang, Y.; Shan, Y.; Mao, N.; Yang, Y.; Pan, L. Secretion of annexin a3 from ovarian cancer cells and its association with platinum resistance in ovarian cancer patients. J. Cell Mol. Med 2012, 16, 337–348. [Google Scholar]
- Battke, C.; Ruiss, R.; Welsch, U.; Wimberger, P.; Lang, S.; Jochum, S.; Zeidler, R. Tumor exosomes inhibit binding of tumor-reactive antibodies to tumor cells and reduce adcc. Cancer Immunol. Immunother 2011, 60, 639–648. [Google Scholar]
- Taylor, A.R.; Robinson, M.B.; Gifondorwa, D.J.; Tytell, M.; Milligan, C.E. Regulation of heat shock protein 70 release in astrocytes: Role of signaling kinases. Dev. Neurobiol 2007, 67, 1815–1829. [Google Scholar]
- Potolicchio, I.; Carven, G.J.; Xu, X.; Stipp, C.; Riese, R.J.; Stern, L.J.; Santambrogio, L. Proteomic analysis of microglia-derived exosomes: Metabolic role of the aminopeptidase cd13 in neuropeptide catabolism. J. Immunol 2005, 175, 2237–2243. [Google Scholar]
- Lachenal, G.; Pernet-Gallay, K.; Chivet, M.; Hemming, F.J.; Belly, A.; Bodon, G.; Blot, B.; Haase, G.; Goldberg, Y.; Sadoul, R. Release of exosomes from differentiated neurons and its regulation by synaptic glutamatergic activity. Mol. Cell. Neurosci 2011, 46, 409–418. [Google Scholar] [Green Version]
- Weissmann, C.; Enari, M.; Klöhn, P.C.; Rossi, D.; Flechsig, E. Transmission of prions. Proc. Natl. Acad. Sci. USA 2002, 99, 16378–16383. [Google Scholar]
- Fevrier, B.; Vilette, D.; Archer, F.; Loew, D.; Faigle, W.; Vidal, M.; Laude, H.; Raposo, G. Cells release prions in association with exosomes. Proc. Natl. Acad. Sci. USA 2004, 101, 9683–9688. [Google Scholar]
- Vella, L.J.; Sharples, R.A.; Lawson, V.A.; Masters, C.L.; Cappai, R.; Hill, A.F. Packaging of prions into exosomes is associated with a novel pathway of prp processing. J. Pathol 2007, 211, 582–590. [Google Scholar]
- Aguzzi, A.; Rajendran, L. The transcellular spread of cytosolic amyloids, prions, and prionoids. Neuron 2009, 64, 783–790. [Google Scholar]
- Alvarez-Erviti, L.S.Y.; Schapira, A.H.; Gardiner, C.; Sargent, I.L.; Wood, M.J.; Cooper, J.M. Lysosomal dysfunction increases exosome-mediated alpha-synuclein release and transmission. Neurobiol. Dis 2011, 42, 360–367. [Google Scholar]
- Selkoe, D.J. Alzheimer’s disease results from the cerebral accumulation and cytotoxicity of amyloid beta-protein. J. Alzheimers Dis 2001, 3, 75–80. [Google Scholar]
- Rajendran, L.; Honsho, M.; Zahn, T.R.; Keller, P.; Geiger, K.D.; Verkade, P.; Simons, K. Alzheimer’s disease b-amyloid peptides are released in association with exosomes. Proc. Natl. Acad. Sci. USA 2006, 103, 11172–11177. [Google Scholar]
- Saman, S.; Kim, W.; Raya, M.; Visnick, Y.; Miro, S.; Saman, S.; Jackson, B.; McKee, A.; Alvarez, V.; Lee, N.; et al. Exosome-associated tau is secreted in tauopathy models and is selectively phosphorylated in cerebrospinal fluid in early alzheimer disease. J. Biol. Chem 2012, 287, 3842–3849. [Google Scholar]
- Chen, J.; Mandel, E.; Thomson, J.; Wu, Q.; Callis, T.; Hammond, S.; Conlon, F.; Wang, D. The role of microrna-1 and microrna-133 in skeletal muscle proliferation and differentiation. Nat. Genet 2006, 38, 228–233. [Google Scholar]
- Kuwabara, Y.; Ono, K.; Horie, T.; Nishi, H.; Nagao, K.; Kinoshita, M.; Watanabe, S.; Baba, O.; Kojima, Y.; Shizuta, S.; et al. Increased microrna-1 and microrna-133a levels in serum of patients with cardiovascular disease indicate myocardial damage. Circ. Cardiovasc. Genet 2011, 4, 446–454. [Google Scholar]
- Azevedo, L.; Janiszewski, M.; Pontieri, V.; Pedro, A.; Bassi, E.; Tucci, P.; Laurindo, F. Platelet-derived exosomes from septic shock patients induce myocardial dysfunction. Crit. Care 2007, 11, R120. [Google Scholar]
- Martinez-Lostao, L.; García-Alvarez, F.; Basáñez, G.; Alegre-Aguarón, E.; Desportes, P.; Larrad, L.; Naval, J.; Martínez-Lorenzo, M.J.; Anel, A. Liposome-bound apo2l/trail is an effective treatment in a rabbit model of rheumatoid arthritis. Arthritis Rheum 2010, 62, 2272–2282. [Google Scholar]
- Van Venrooij, W.J.; van Beers, J.J.; Pruijn, G.J. Anti-ccp antibodies: The past, the present and the future. Nat. Rev. Rheumatol 2011, 7, 391–398. [Google Scholar]
- Ireland, J.; Herzog, J.; Unanue, E.R. Cutting edge: Unique T cells that recognize citrullinated peptides are a feature of protein immunization. J. Immunol 2006, 177, 1421–1425. [Google Scholar]
- Pant, S.; Hilton, H.; Burczynski, M.E. The multifaceted exosome: Biogenesis, role in normal and aberrant cellular function, and frontiers for pharmacological and biomarker opportunities. Biochem. Pharmacol 2012, 83, 1484–1494. [Google Scholar]
- Vlassov, A.V.; Magdaleno, S.; Setterquist, R.; Conrad, R. Exosomes: Current knowledge of their composition, biological functions, and diagnostic and therapeutic potentials. Biochim. Biophys. Acta 2012, 1820, 940–948. [Google Scholar]
- Fais, S.; Logozzi, M.; Lugini, L.; Federici, C.; Azzarito, T.; Zarovni, N.; Chiesi, A. Exosomes: The ideal nanovectors for biodelivery. Biol. Chem 2013, 394, 1–15. [Google Scholar]
- Kalra, H.; Simpson, R.; Ji, H.; Aikawa, E.; Altevogt, P.; Askenase, P.; Bond, V.C.; Borràs, F.E.; Breakefield, X.; Budnik, V.; et al. Vesiclepedia: A compendium for extracellular vesicles with continuous community annotation. PLoS Biol 2012, 10, e1001450. [Google Scholar]
- Raimondo, F.; Morosi, L.; Chinello, C.; Magni, F.; Pitto, M. Advances in membranous vesicle and exosome proteomics improving biological understanding and biomarker discovery. Proteomics 2011, 11, 709–720. [Google Scholar]
- Smalheiser, N. Exosomal transfer of proteins and RNAs at synapses in the nervous system. Biol. Direct 2007, 2, 1–15. [Google Scholar]
- Rabinowits, G.; Gerçel-Taylor, C.; Day, J.; Taylor, D.; Kloecker, G. Exosomal microrna: A diagnostic marker for lung cancer. Clin. Lung Cancer 2009, 10, 42–46. [Google Scholar]
- Taylor, D.; Gercel-Taylor, C. Microrna signatures of tumor-derived exosomes as diagnostic biomarkers of ovarian cancer. Gynecol. Oncol 2008, 110, 13–21. [Google Scholar]
- Smalley, D.M.; Sheman, N.E.; Nelson, K.; Theodorescu, D. Isolation and identificationof potential urinary microparticle biomarkers of bladder cancer. J. Prot. Res 2008, 7, 2088–2096. [Google Scholar]
- Liang, B.; Peng, P.; Chen, S.; Li, L.; Zhang, M.; Cao, D.; Yang, J.; Li, H.; Gui, T.; Li, X.; et al. Characterization and proteomic analysis of ovarian cancer-derived exosomes. J. Proteomics 2013, 80, 171–182. [Google Scholar]
- Chen, C.L.; Lai, Y.F.; Tang, P.; Chien, K.Y.; Yu, J.S.; Tsai, C.H.; Chen, H.W.; Wu, C.C.; Chung, T.; Hsu, C.W.; et al. Comparative and targeted proteomic analyses of urinary microparticles from bladder cancer and herniapatients. J. Prot. Res 2012, 11, 5611–5629. [Google Scholar]
- Alvarez-Erviti, L.; Seow, Y.; Yin, H.; Betts, C.; Lakhal, S.; Wood, M.J. Delivery of sirna to the mouse brain by systemic injection of targeted exosomes. Nat. Biotechnol 2011, 29, 341–345. [Google Scholar]
- El-Andaloussi, S.; Lee, Y.; Lakhal-Littleton, S.; Li, J.; Seow, Y.; Gardiner, C.; Alvarez-Erviti, L.; Sargent, I.L.; Wood, M.J. Exosome-mediated delivery of sirna in vitro and in vivo. Nat. Protoc 2012, 7, 2112–2126. [Google Scholar]
- Ohno, S.; Takanashi, M.; Sudo, K.; Ueda, S.; Ishikawa, A.; Matsuyama, N.; Fujita, K.; Mizutani, T.; Ohgi, T.; Ochiya, T.; et al. Systemically injected exosomes targeted to egfr deliver antitumor microrna to breast cancer cells. Mol. Ther 2013, 21, 185–191. [Google Scholar]
- Sun, D.; Zhuang, X.; Xiang, X.; Liu, Y.; Zhang, S.; Liu, C.; Barnes, S.; Grizzle, W.; Miller, D.; Zhang, H.G. A novel nanoparticle drug delivery system: The anti-inflammatory activity of curcumin is enhanced when encapsulated in exosomes. Mol. Ther 2010, 18, 1606–1614. [Google Scholar]
- Lai, C.P.; Breakefield, X.O. Role of exosomes/microvesicles in the nervous system and use in emerging therapies. Front. Physiol 2012, 3, 228. [Google Scholar]
- Biancone, L.; Bruno, S.; Deregibus, M.C.; Tetta, C.; Camussi, G. Therapeutic potential of mesenchymal stem cell-derived microvesicles. Nephrol. Dial. Transplant 2012, 27, 3037–3042. [Google Scholar]
- Ratajczak, M.Z.; Kucia, M.; Jadczyk, T.; Greco, N.J.; Wojakowski, W.; Tendera, M.; Ratajczak, J. Pivotal role of paracrine effects in stem cell therapies in regenerative medicine: Can we translate stem cell-secreted paracrine factors and microvesicles into better therapeutic strategies? Leukemia 2012, 26, 1166–1173. [Google Scholar]
- Chen, T.S.; Arslan, F.; Yin, Y.; Tan, S.S.; Lai, R.C.; Choo, A.B.; Padmanabhan, J.; Lee, C.N.; de Kleijn, D.P.; Lim, S.K. Enabling a robust scalable manufacturing process for therapeutic exosomes through oncogenic immortalization of human esc-derived mscs. J. Transl. Med 2011, 9, 1–10. [Google Scholar]
- Andre, F.; Chaput, N.; Schartz, N.E.; Flament, C.; Aubert, N.; Bernard, J.; Lemonnier, F.; Raposo, G.; Escudier, B.; Hsu, D.H.; et al. Exosomes as potent cell-free peptide-based vaccine. I. Dendritic cell-derived exosomes transfer functional MHC class I/peptide complexes to dendritic cells. J. Immunol 2004, 172, 2126–2136. [Google Scholar]
- Schartz, N.E.; Chaput, N.; André, F.; Zitvogel, L. From the antigen-presenting cell to the antigen-presenting vesicle: The exosomes. Curr. Opin. Mol. Ther 2002, 4, 372–381. [Google Scholar]
- Cho, J.A.; Yeo, D.J.; Son, H.Y.; Kim, H.W.; Jung, D.S.; Ko, J.K.; Koh, J.S.; Kim, Y.N.; Kim, C.W. Exosomes: A new delivery system for tumor antigens in cancer immunotherapy. Int. J. Cancer 2005, 114, 613–622. [Google Scholar]
- Hosseini, H.M.; Fooladi, A.A.; Nourani, M.R.; Ghanezadeh, F. Role of exosome in infectious disease. Inflamm. Allergy Drug Targets 2012, in press. [Google Scholar]
- Martin-Jaular, L.; Nakayasu, E.S.; Ferrer, M.; Almeida, I.C.; del Portillo, H.A. Exosomes from plasmodium yoelii-infected reticulocytes protect mice from lethal infections. PLoS One 2011, 6, e26588. [Google Scholar]
- Nanjundappa, R.H.; Wang, R.; Xie, Y.; Umeshappa, C.S.; Chibbar, R.; Wei, Y.; Liu, Q.; Xiang, J. Gp120-specific exosome-targeted T cell-based vaccine capable of stimulating dc- and cd4(+) T-independent ctl responses. Vaccine 2011, 29, 3538–3547. [Google Scholar]
- Lattanzi, L.; Federico, M. A strategy of antigen incorporation into exosomes: Comparing cross-presentation levels of antigens delivered by engineered exosomes and by lentiviral virus-like particles. Vaccine 2012, 30, 7229–7237. [Google Scholar]
| Tissue/cell derived-exosomes | Target | Function | References |
|---|---|---|---|
| Activated B cells | CD4+ T cells | Modulation of immune response and maintaining antigen specific memory T cells | [51,52] |
| Mature DCs | CD8+ T cells | Vehicle of antigen transfer between different DCs | [47] |
| Plasma | T cells | Suppression of Th1-type hypersensitivity response; suppression of Th2-type allergic response | [53,54] |
| Plasma | Monocytes and lymphocytes | Delivering of specific exogenous siRNAs targeting MAPK pathway | [25] |
| Placenta | Fetus and child | Modulation of T cell activity; immune surveillance and recognition of paternal antigens | [55–57] |
| Breast milk | Infant | Modulation of infant’s immune cell function via miRNAs involved in T cell regulation and B cell differentiation | [58–60] |
| Mouse mast cells | Primary bone marrow-derived mouse mast cells | Regulated exchange of genetic material (mRNAs and miRNAs) | [61] |
| Cancer Associated Fibroblasts (CAFs) | Breast cancer cells | Promotion of cells’ protrusive activity and motility | [62] |
| Metastatic melanoma cells | Bone marrow progenitor cells | Support of tumor vasculogenesis, invasion and metastasis through MET | [19] |
| Lung cancer cells | Toll Like receptor (TLR) family in immune cells | Activation of prometastatic inflammatory response through specific miRNAs | [63] |
| Chronic myelogenous leukaemia cells | Human vascular endothelial cells | Induction of an angiogenic phenotype through the release of IL8 (21, 110) or activation of Src Kinase (111) | [21,64,65] |
| Acute myeloid leukaemia blasts and cells | Ba/F3 progenitor cells | Alteration of proliferative, angiogenic and migratory responses through specif miRNAs | [66] |
| Neuronal cells | Glial cells | Transmission of γ-sinuclein thus promoting the aggregation of intracellular protein | [67] |
| Endothelial cells | smooth muscle cells (SMCs) | Transfer of specific miRNAs thus preventing SMC de-differentiation | [16] |
| Synovial fibroblast from AR patients | CD4+ T cells | Induction of AKT and NF-κB pathways leading to apoptosis resistance | [68] |
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Corrado, C.; Raimondo, S.; Chiesi, A.; Ciccia, F.; De Leo, G.; Alessandro, R. Exosomes as Intercellular Signaling Organelles Involved in Health and Disease: Basic Science and Clinical Applications. Int. J. Mol. Sci. 2013, 14, 5338-5366. https://doi.org/10.3390/ijms14035338
Corrado C, Raimondo S, Chiesi A, Ciccia F, De Leo G, Alessandro R. Exosomes as Intercellular Signaling Organelles Involved in Health and Disease: Basic Science and Clinical Applications. International Journal of Molecular Sciences. 2013; 14(3):5338-5366. https://doi.org/10.3390/ijms14035338
Chicago/Turabian StyleCorrado, Chiara, Stefania Raimondo, Antonio Chiesi, Francesco Ciccia, Giacomo De Leo, and Riccardo Alessandro. 2013. "Exosomes as Intercellular Signaling Organelles Involved in Health and Disease: Basic Science and Clinical Applications" International Journal of Molecular Sciences 14, no. 3: 5338-5366. https://doi.org/10.3390/ijms14035338
APA StyleCorrado, C., Raimondo, S., Chiesi, A., Ciccia, F., De Leo, G., & Alessandro, R. (2013). Exosomes as Intercellular Signaling Organelles Involved in Health and Disease: Basic Science and Clinical Applications. International Journal of Molecular Sciences, 14(3), 5338-5366. https://doi.org/10.3390/ijms14035338






