Analysis of Epithelial Growth Factor-Receptor (EGFR) Phosphorylation in Uterine Smooth Muscle Tumors: Correlation to Mucin-1 and Galectin-3 Expression
Abstract
:1. Introduction
2. Results and Discussion
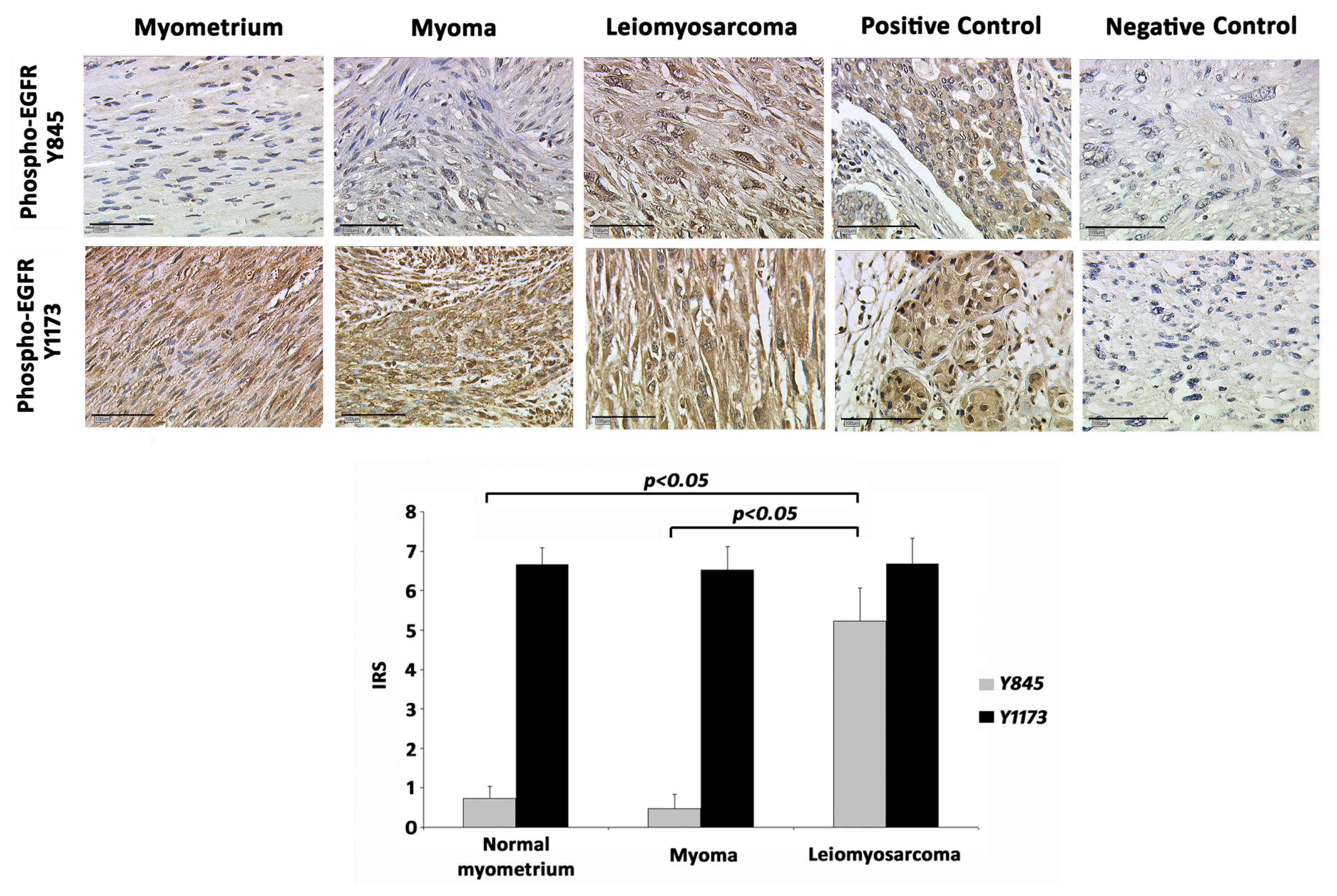
2.1. EGFR-Y845
2.2. EGFR-Y1173
2.3. Correlation Analysis
2.4. Discussion
3. Experimental Section
3.1. Study Population
3.2. Immunohistochemistry
3.3. Statistics
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Hodge, J.C.; Morton, C.C. Genetic heterogeneity among uterine leiomyomata: Insights into malignant progression. Hum. Mol. Genet 2007, 16, R7–R13. [Google Scholar]
- Kanamori, T.; Takakura, K.; Mandai, M.; Kariya, M.; Fukuhara, K.; Kusakari, T.; Momma, C.; Shime, H.; Yagi, H.; Konishi, M.; et al. PEP-19 overexpression in human uterine leiomyoma. Mol. Hum. Reprod 2003, 9, 709–717. [Google Scholar]
- Marshall, L.M.; Spiegelman, D.; Barbieri, R.L.; Goldman, M.B.; Manson, J.E.; Colditz, G.A.; Willett, W.C.; Hunter, D.J. Variation in the incidence of uterine leiomyoma among premenopausal women by age and race. Obstet. Gynecol 1997, 90, 967–973. [Google Scholar]
- Peddada, S.D.; Laughlin, S.K.; Miner, K.; Guyon, J.P.; Haneke, K.; Vahdat, H.L.; Semelka, R.C.; Kowalik, A.; Armao, D.; Davis, B.; et al. Growth of uterine leiomyomata among premenopausal black and white women. Proc. Natl. Acad. Sci. USA 2008, 105, 19887–19892. [Google Scholar]
- Weiss, G.; Noorhasan, D.; Schott, L.L.; Powell, L.; Randolph, J.F., Jr; Johnston, J.M. Racial differences in women who have a hysterectomy for benign conditions. Womens Health Issues 2009, 19, 202–210. [Google Scholar]
- Schwartz, P.E.; Kelly, M.G. Malignant transformation of myomas: Myth or reality? Obstet. Gynecol. Clin. North Am 2006, 33, 183–198. [Google Scholar]
- Indraccolo, U.; Luchetti, G.; Indraccolo, S.R. Malignant transformation of uterine leiomyomata. Eur. J. Gynaecol. Oncol 2008, 29, 543–544. [Google Scholar]
- Jatoi, N. Leiomyosarcoma: A rare malignant change in a leiomyoma. J. Coll. Phys. Surg. Pak 2003, 13, 106–107. [Google Scholar]
- Mittal, K.R.; Chen, F.; Wei, J.J.; Rijhvani, K.; Kurvathi, R.; Streck, D.; Dermody, J.; Toruner, G.A. Molecular and immunohistochemical evidence for the origin of uterine leiomyosarcomas from associated leiomyoma and symplastic leiomyoma-like areas. Mod. Pathol 2009, 22, 1303–1311. [Google Scholar]
- Watanabe, K.; Suzuki, T. Uterine leiomyoma versus leiomyosarcoma: A new attempt at differential diagnosis based on their cellular characteristics. Histopathology 2006, 48, 563–568. [Google Scholar]
- Boerner, J.L.; Demory, M.L.; Silva, C.; Parsons, S.J. Phosphorylation of Y845 on the epidermal growth factor receptor mediates binding to the mitochondrial protein cytochrome c oxidase subunit II. Mol. Cell. Biol 2004, 24, 7059–7071. [Google Scholar]
- Bitler, B.G.; Goverdhan, A.; Schroeder, J.A. MUC1 regulates nuclear localization and function of the epidermal growth factor receptor. J. Cell. Sci 2010, 123, 1716–1723. [Google Scholar]
- Abe, M.; Kuroda, Y.; Hirose, M.; Watanabe, Y.; Nakano, M.; Handa, T. Inhibition of autophosphorylation of epidermal growth factor receptor by small peptides in vitro. Br. J. Pharmacol 2006, 147, 402–411. [Google Scholar]
- Weissenbacher, T.; Kuhn, C.; Mayr, D.; Pavlik, R.; Friese, K.; Scholz, C.; Jeschke, U.; Ditsch, N.; Dian, D. Expression of mucin-1, galectin-1 and galectin-3 in human leiomyosarcoma in comparison to leiomyoma and myometrium. Anticancer Res 2011, 31, 451–457. [Google Scholar]
- Merlin, J.; Stechly, L.; de Beauce, S.; Monte, D.; Leteurtre, E.; van Seuningen, I.; Huet, G.; Pigny, P. Galectin-3 regulates MUC1 and EGFR cellular distribution and EGFR downstream pathways in pancreatic cancer cells. Oncogene 2011, 30, 2514–2525. [Google Scholar]
- Ramasamy, S.; Duraisamy, S.; Barbashov, S.; Kawano, T.; Kharbanda, S.; Kufe, D. The MUC1 and galectin-3 oncoproteins function in a microRNA-dependent regulatory loop. Mol. Cell 2007, 27, 992–1004. [Google Scholar]
- Wu, W.; Graves, L.M.; Gill, G.N.; Parsons, S.J.; Samet, J.M. Src-dependent phosphorylation of the epidermal growth factor receptor on tyrosine 845 is required for zinc-induced Ras activation. J. Biol. Chem 2002, 277, 24252–24257. [Google Scholar]
- Xu, K.P.; Yin, J.; Yu, F.S. SRC-family tyrosine kinases in wound- and ligand-induced epidermal growth factor receptor activation in human corneal epithelial cells. Invest. Ophthalmol. Vis. Sci 2006, 47, 2832–2839. [Google Scholar]
- Sanci, M.; Dikis, C.; Inan, S.; Turkoz, E.; Dicle, N.; Ispahi, C. Immunolocalization of VEGF, VEGF receptors, EGF-R and Ki-67 in leiomyoma, cellular leiomyoma and leiomyosarcoma. Acta Histochem 2011, 113, 317–325. [Google Scholar]
- Moore, A.B.; He, H.; Yoshida, A.; Rico, P.J.; Haseman, J.K.; Dixon, D. Transforming growth factor-alpha, epidermal growth factor receptor, and PCNA immunoexpression in uterine leiomyosarcomas and leiomyomas in B6C3F1 mice. Exp. Toxicol. Pathol 2000, 52, 195–200. [Google Scholar]
- Sato, O.; Wada, T.; Kawai, A.; Yamaguchi, U.; Makimoto, A.; Kokai, Y.; Yamashita, T.; Chuman, H.; Beppu, Y.; Tani, Y.; Hasegawa, T. Expression of epidermal growth factor receptor, ERBB2 and KIT in adult soft tissue sarcomas: A clinicopathologic study of 281 cases. Cancer 2005, 103, 1881–1890. [Google Scholar]
- Sette, G.; Salvati, V.; Memeo, L.; Fecchi, K.; Colarossi, C.; Di Matteo, P.; Signore, M.; Biffoni, M.; D’Andrea, V.; de Antoni, E.; et al. EGFR inhibition abrogates leiomyosarcoma cell chemoresistance through inactivation of survival pathways and impairment of CSC potential. PLoS One 2012, 7, e46891. [Google Scholar]
- Mueller, K.L.; Powell, K.; Madden, J.M.; Eblen, S.T.; Boerner, J.L. EGFR Tyrosine 845 phosphorylation-dependent proliferation and transformation of breast cancer cells require activation of p38 MAPK. Transl. Oncol 2012, 5, 327–334. [Google Scholar]
- Dimri, M.; Naramura, M.; Duan, L.; Chen, J.; Ortega-Cava, C.; Chen, G.; Goswami, R.; Fernandes, N.; Gao, Q.; Dimri, G.P.; et al. Modeling breast cancer-associated c-Src and EGFR overexpression in human MECs: C-Src and EGFR cooperatively promote aberrant three-dimensional acinar structure and invasive behavior. Cancer Res 2007, 67, 4164–4172. [Google Scholar]
- Li, X.; Wang, L.; Nunes, D.P.; Troxler, R.F.; Offner, G.D. Suppression of MUC1 synthesis downregulates expression of the epidermal growth factor receptor. Cancer Biol. Ther 2005, 4, 968–973. [Google Scholar]
- Kong, L.; Deng, Z.; Shen, H.; Zhang, Y. Src family kinase inhibitor PP2 efficiently inhibits cervical cancer cell proliferation through down-regulating phospho-Src-Y416 and phospho-EGFR-Y1173. Mol. Cell. Biochem 2011, 348, 11–19. [Google Scholar]
- De Oliveira, J.T.; de Matos, A.J.; Santos, A.L.; Pinto, R.; Gomes, J.; Hespanhol, V.; Chammas, R.; Aki, M.; Bernardes, E.S.; Albuquerque Reis, C.; et al. Sialylation regulates galectin-3/ligand interplay during mammary tumour progression—A case of targeted uncloaking. Int. J. Dev. Biol 2011, 55, 823–834. [Google Scholar]
- Brustmann, H. Epidermal growth factor receptor expression in serous ovarian carcinoma: An immunohistochemical study with galectin-3 and cyclin D1 and outcome. Int. J. Gynecol. Pathol 2008, 27, 380–389. [Google Scholar]
- Jeschke, U.; Kuhn, C.; Mylonas, I.; Schulze, S.; Friese, K.; Mayr, D.; Speer, R.; Briese, V.; Richter, D.U.; Haase, M.; et al. Development and characterization of monoclonal antibodies for the immunohistochemical detection of glycodelin A in decidual, endometrial and gynaecological tumour tissues. Histopathology 2006, 48, 394–406. [Google Scholar]
- Jeschke, U.; Mylonas, I.; Shabani, N.; Kunert-Keil, C.; Schindlbeck, C.; Gerber, B.; Friese, K. Expression of sialyl lewis X, sialyl Lewis A, E-cadherin and cathepsin-D in human breast cancer: Immunohistochemical analysis in mammary carcinoma in situ, invasive carcinomas and their lymph node metastasis. Anticancer Res 2005, 25, 1615–1622. [Google Scholar]
- Mylonas, I.; Makovitzky, J.; Jeschke, U.; Briese, V.; Friese, K.; Gerber, B. Expression of Her2/neu, steroid receptors (ER and PR), Ki67 and p53 in invasive mammary ductal carcinoma associated with ductal carcinoma in situ (DCIS) versus invasive breast cancer alone. Anticancer Res 2005, 25, 1719–1723. [Google Scholar]
- Ren, W.; Korchin, B.; Lahat, G.; Wei, C.; Bolshakov, S.; Nguyen, T.; Merritt, W.; Dicker, A.; Lazar, A.; Sood, A.; et al. Combined vascular endothelial growth factor receptor/epidermal growth factor receptor blockade with chemotherapy for treatment of local, uterine, and metastatic soft tissue sarcoma. Clin. Cancer Res 2008, 14, 5466–5475. [Google Scholar]
| Antibody | Clone | Isotype | Concentration | Source |
|---|---|---|---|---|
| pEGFR-Y845 | polyclonal | Rabbit IgG | 1.0 mg/mL | R & D Systems, USA |
| pEGFR-Y1173 | polyclonal | Rabbit IgG | 1.0 mg/mL | R & D Systems, USA |
| Galectin-3 | 9C4 | Mouse IgG | 2.0 μg/mL | Novocastra, UK |
| Mucin-1 | VU-4-H5 | Mouse IgG1 | 6.8 μg/mL | Invitrogen, USA |
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Weissenbacher, T.; Vrekoussis, T.; Roeder, D.; Makrigiannakis, A.; Mayr, D.; Ditsch, N.; Friese, K.; Jeschke, U.; Dian, D. Analysis of Epithelial Growth Factor-Receptor (EGFR) Phosphorylation in Uterine Smooth Muscle Tumors: Correlation to Mucin-1 and Galectin-3 Expression. Int. J. Mol. Sci. 2013, 14, 4783-4792. https://doi.org/10.3390/ijms14034783
Weissenbacher T, Vrekoussis T, Roeder D, Makrigiannakis A, Mayr D, Ditsch N, Friese K, Jeschke U, Dian D. Analysis of Epithelial Growth Factor-Receptor (EGFR) Phosphorylation in Uterine Smooth Muscle Tumors: Correlation to Mucin-1 and Galectin-3 Expression. International Journal of Molecular Sciences. 2013; 14(3):4783-4792. https://doi.org/10.3390/ijms14034783
Chicago/Turabian StyleWeissenbacher, Tobias, Thomas Vrekoussis, David Roeder, Antonis Makrigiannakis, Doris Mayr, Nina Ditsch, Klaus Friese, Udo Jeschke, and Darius Dian. 2013. "Analysis of Epithelial Growth Factor-Receptor (EGFR) Phosphorylation in Uterine Smooth Muscle Tumors: Correlation to Mucin-1 and Galectin-3 Expression" International Journal of Molecular Sciences 14, no. 3: 4783-4792. https://doi.org/10.3390/ijms14034783
APA StyleWeissenbacher, T., Vrekoussis, T., Roeder, D., Makrigiannakis, A., Mayr, D., Ditsch, N., Friese, K., Jeschke, U., & Dian, D. (2013). Analysis of Epithelial Growth Factor-Receptor (EGFR) Phosphorylation in Uterine Smooth Muscle Tumors: Correlation to Mucin-1 and Galectin-3 Expression. International Journal of Molecular Sciences, 14(3), 4783-4792. https://doi.org/10.3390/ijms14034783






