Optimization to Low Temperature Activity in Psychrophilic Enzymes
Abstract
:1. Introduction
2. Kinetic Properties of Cold Enzymes
2.1. General Properties
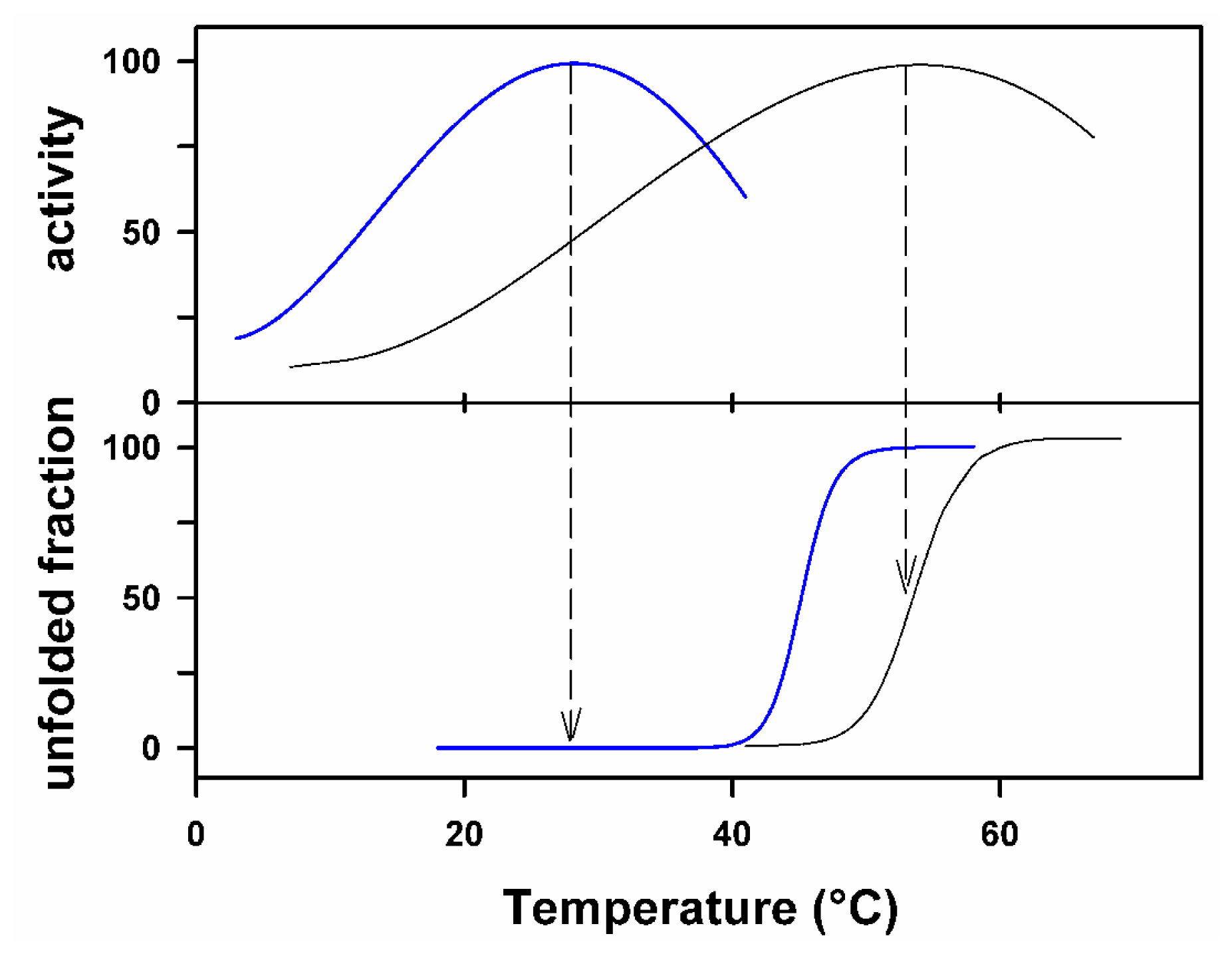
2.2. Heat-Labile and Unstable Cold Enzymes
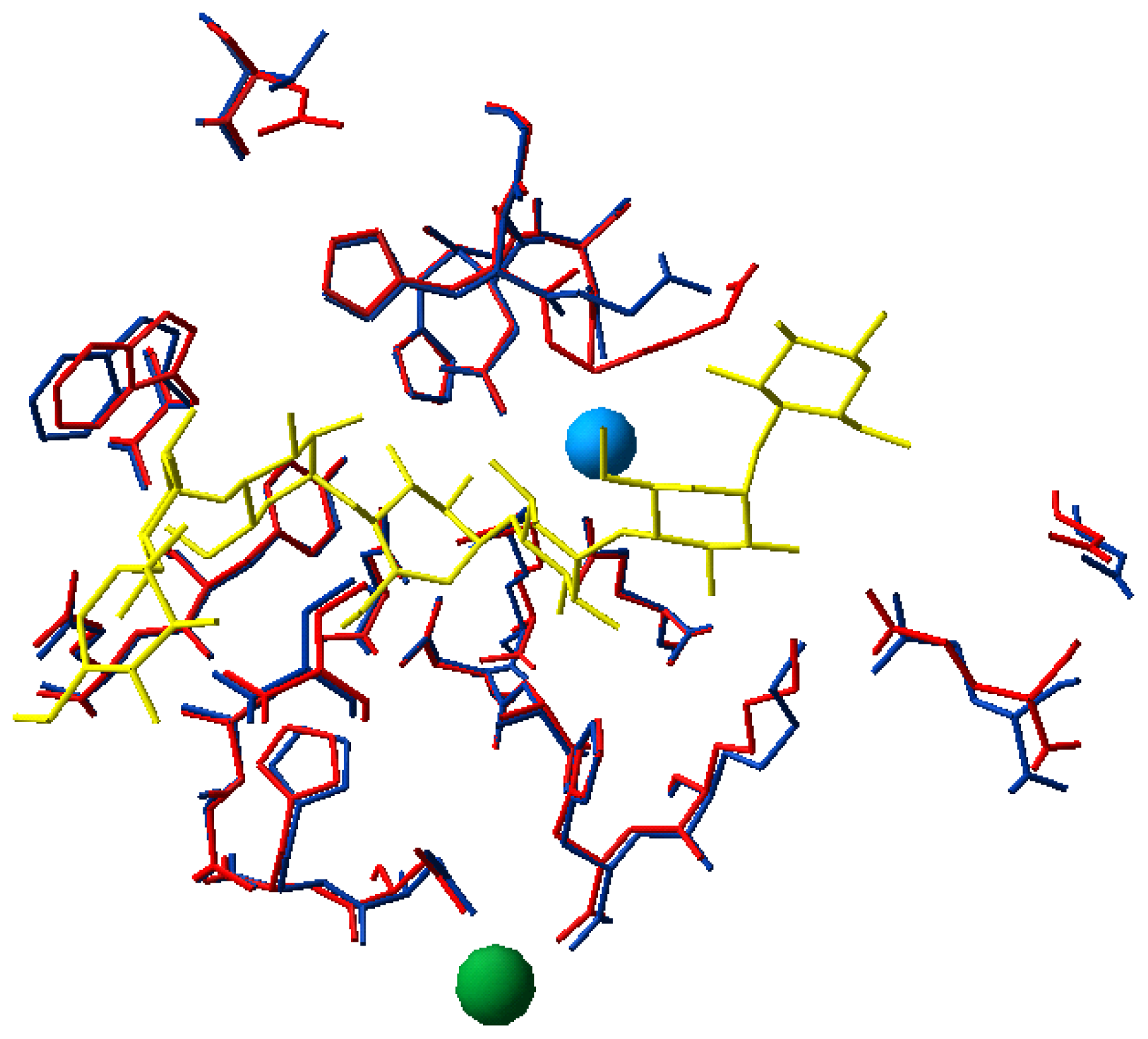
2.3. Active Site Structure
2.4. Active Site Dynamics
2.5. Adaptive Drift of Substrate Affinity
2.6. Adaptive Optimization of Substrate Affinity
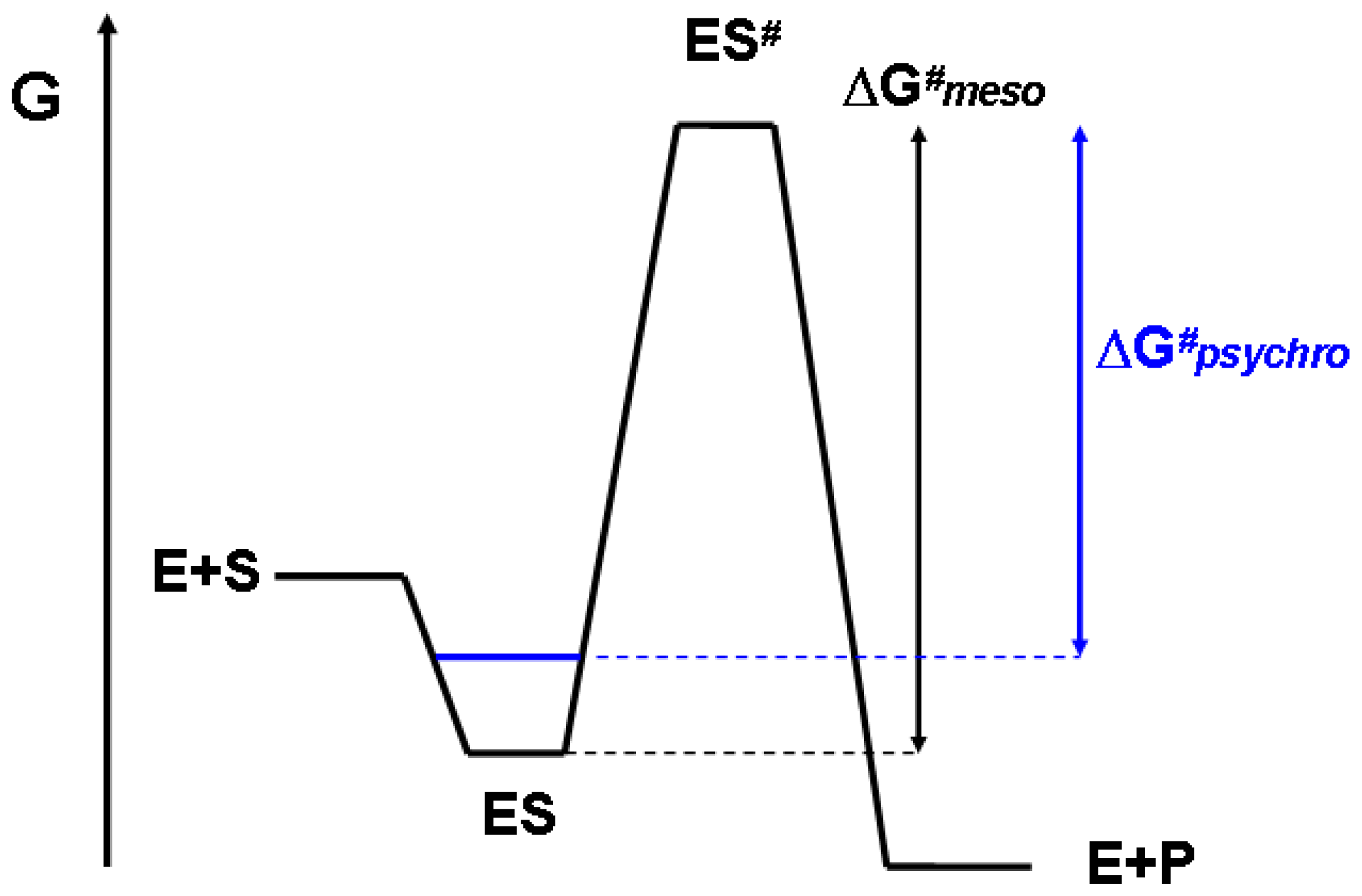
3. Thermodynamic Origin of Optimization
4. Structural Origin of Cold Activity
5. Biotechnological Usefulness of Psychrophilic Enzymes
5.1. Molecular Biology
5.2. Industrial Enzymes
5.3. Food Technology
5.4. Engineering Cold Activity
6. Conclusions
Acknowledgments
References
- Gilichinsky, D.; Rivkina, E.; Bakermans, C.; Shcherbakova, V.; Petrovskaya, L.; Ozerskaya, S.; Ivanushkina, N.; Kochkina, G.; Laurinavichuis, K.; Pecheritsina, S.; et al. Biodiversity of cryopegs in permafrost. FEMS Microbiol. Ecol 2005, 53, 117–128. [Google Scholar]
- Deming, J.W. Psychrophiles and polar regions. Curr. Opin. Microbiol 2002, 5, 301–309. [Google Scholar]
- Friedmann, E.I. Endolithic microorganisms in the Antarctic cold desert. Science 1982, 215, 1045–1053. [Google Scholar]
- Cary, S.C.; McDonald, I.R.; Barrett, J.E.; Cowan, D.A. On the rocks: The microbiology of Antarctic Dry Valley soils. Nat. Rev. Microbiol 2010, 8, 129–138. [Google Scholar]
- Rodrigues, D.F.; Tiedje, J.M. Coping with our cold planet. Appl. Environ. Microbiol 2008, 74, 1677–1686. [Google Scholar]
- Cowan, D.A.; Casanueva, A.; Stafford, W. Ecology and Biodiversity of Cold-adapted Microorganisms. In Physiology and Biochemistry of Extremophiles; Gerday, C., Glansdorff, N., Eds.; ASM Press: Washington, DC, USA, 2007; pp. 119–132. [Google Scholar]
- Margesin, R.; Schinner, F.; Marx, J.C.; Gerday, C. Psychrophiles, from Biodiversity to Biotechnology; Springer-Verlag: Berlin/Heidelberg, Germany, 2008. [Google Scholar]
- Feller, G.; Gerday, C. Psychrophilic enzymes: Hot topics in cold adaptation. Nat. Rev. Microbiol 2003, 1, 200–208. [Google Scholar]
- Siddiqui, K.S.; Cavicchioli, R. Cold-adapted enzymes. Annu. Rev. Biochem 2006, 75, 403–433. [Google Scholar]
- Feller, G. Protein stability and enzyme activity at extreme biological temperatures. J. Phys. Condens. Mat 2010, 22. [Google Scholar] [CrossRef]
- Feller, G.; Lonhienne, T.; Deroanne, C.; Libioulle, C.; Van Beeumen, J.; Gerday, C. Purification, characterization, and nucleotide sequence of the thermolabile α-amylase from the antarctic psychrotroph Alteromonas haloplanctis A23. J. Biol. Chem 1992, 267, 5217–5221. [Google Scholar]
- D’Amico, S.; Collins, T.; Marx, J.C.; Feller, G.; Gerday, C. Psychrophilic microorganisms: Challenges for life. EMBO Rep 2006, 7, 385–389. [Google Scholar]
- Collins, T.; Meuwis, M.A.; Gerday, C.; Feller, G. Activity, stability and flexibility in glycosidases adapted to extreme thermal environments. J. Mol. Biol 2003, 328, 419–428. [Google Scholar]
- D’Amico, S.; Marx, J.C.; Gerday, C.; Feller, G. Activity-stability relationships in extremophilic enzymes. J. Biol. Chem 2003, 278, 7891–7896. [Google Scholar]
- Georlette, D.; Damien, B.; Blaise, V.; Depiereux, E.; Uversky, V.N.; Gerday, C.; Feller, G. Structural and functional adaptations to extreme temperatures in psychrophilic, mesophilic, and thermophilic DNA ligases. J. Biol. Chem 2003, 278, 37015–37023. [Google Scholar]
- Siddiqui, K.S.; Feller, G.; D’Amico, S.; Gerday, C.; Giaquinto, L.; Cavicchioli, R. The active site is the least stable structure in the unfolding pathway of a multidomain cold-adapted alpha-amylase. J. Bacteriol 2005, 187, 6197–6205. [Google Scholar]
- Fields, P.A.; Somero, G.N. Hot spots in cold adaptation: Localized increases in conformational flexibility in lactate dehydrogenase A(4) orthologs of Antarctic notothenioid fishes. Proc. Natl. Acad. Sci. USA 1998, 95, 11476–11481. [Google Scholar]
- Chiuri, R.; Maiorano, G.; Rizzello, A.; del Mercato, L.L.; Cingolani, R.; Rinaldi, R.; Maffia, M.; Pompa, P.P. Exploring local flexibility/rigidity in psychrophilic and mesophilic carbonic anhydrases. Biophys. J 2009, 96, 1586–1596. [Google Scholar]
- Fedoy, A.E.; Yang, N.; Martinez, A.; Leiros, H.K.; Steen, I.H. Structural and functional properties of isocitrate dehydrogenase from the psychrophilic bacterium Desulfotalea psychrophila reveal a cold-active enzyme with an unusual high thermal stability. J. Mol. Biol 2007, 372, 130–149. [Google Scholar]
- Tosco, A.; Birolo, L.; Madonna, S.; Lolli, G.; Sannia, G.; Marino, G. GroEL from the psychrophilic bacterium Pseudoalteromonas haloplanktis TAC 125: Molecular characterization and gene cloning. Extremophiles 2003, 7, 17–28. [Google Scholar]
- Piette, F.; D’Amico, S.; Struvay, C.; Mazzucchelli, G.; Renaut, J.; Tutino, M.L.; Danchin, A.; Leprince, P.; Feller, G. Proteomics of life at low temperatures: Trigger factor is the primary chaperone in the Antarctic bacterium Pseudoalteromonas haloplanktis TAC125. Mol. Microbiol 2010, 76, 120–132. [Google Scholar]
- Falasca, P.; Evangelista, G.; Cotugno, R.; Marco, S.; Masullo, M.; De Vendittis, E.; Raimo, G. Properties of the endogenous components of the thioredoxin system in the psychrophilic eubacterium Pseudoalteromonas haloplanktis TAC 125. Extremophiles 2012, 16, 539–552. [Google Scholar]
- Aghajari, N.; Feller, G.; Gerday, C.; Haser, R. Crystal structures of the psychrophilic α-amylase from Alteromonas haloplanctis in its native form and complexed with an inhibitor. Protein Sci 1998, 7, 564–572. [Google Scholar]
- Aghajari, N.; Feller, G.; Gerday, C.; Haser, R. Structures of the psychrophilic Alteromonas haloplanctis α-amylase give insights into cold adaptation at a molecular level. Structure 1998, 6, 1503–1516. [Google Scholar]
- Aghajari, N.; Roth, M.; Haser, R. Crystallographic evidence of a transglycosylation reaction: Ternary complexes of a psychrophilic alpha-amylase. Biochemistry 2002, 41, 4273–4280. [Google Scholar]
- Qian, M.; Haser, R.; Buisson, G.; Duee, E.; Payan, F. The active center of a mammalian alpha-amylase. Structure of the complex of a pancreatic alpha-amylase with a carbohydrate inhibitor refined to 2.2 Å resolution. Biochemistry 1994, 33, 6284–6294. [Google Scholar]
- Feller, G. Enzyme Function at Low Temperatures in Psychrophiles. In Protein Adaptation in Extremophiles; Siddiqui, K.S., Thomas, T., Eds.; Nova Science Publishers: New York, NY, USA, 2008; pp. 35–69. [Google Scholar]
- Russell, R.J.; Gerike, U.; Danson, M.J.; Hough, D.W.; Taylor, G.L. Structural adaptations of the cold-active citrate synthase from an Antarctic bacterium. Structure 1998, 6, 351–361. [Google Scholar]
- Aghajari, N.; van Petegem, F.; Villeret, V.; Chessa, J.P.; Gerday, C.; Haser, R.; Van Beeumen, J. Crystal structures of a psychrophilic metalloprotease reveal new insights into catalysis by cold-adapted proteases. Proteins 2003, 50, 636–647. [Google Scholar]
- Kim, S.Y.; Hwang, K.Y.; Kim, S.H.; Sung, H.C.; Han, Y.S.; Cho, Y.J. Structural basis for cold adaptation. Sequence, biochemical properties, and crystal structure of malate dehydrogenase from a psychrophile Aquaspirillium arcticum. J. Biol. Chem 1999, 274, 11761–11767. [Google Scholar]
- Leiros, I.; Moe, E.; Lanes, O.; Smalas, A.O.; Willassen, N.P. The structure of uracil-DNA glycosylase from Atlantic cod (Gadus morhua) reveals cold-adaptation features. Acta Crystallogr. D Biol. Crystallogr 2003, 59, 1357–1365. [Google Scholar]
- Smalas, A.O.; Leiros, H.K.; Os, V.; Willassen, N.P. Cold adapted enzymes. Biotechnol. Annu. Rev 2000, 6, 1–57. [Google Scholar]
- Gorfe, A.A.; Brandsdal, B.O.; Leiros, H.K.; Helland, R.; Smalas, A.O. Electrostatics of mesophilic and psychrophilic trypsin isoenzymes: Qualitative evaluation of electrostatic differences at the substrate binding site. Proteins 2000, 40, 207–217. [Google Scholar]
- Brandsdal, B.O.; Smalas, A.O.; Aqvist, J. Electrostatic effects play a central role in cold adaptation of trypsin. FEBS Lett 2001, 499, 171–175. [Google Scholar]
- Tsigos, I.; Velonia, K.; Smonou, I.; Bouriotis, V. Purification and characterization of an alcohol dehydrogenase from the Antarctic psychrophile Moraxella sp. TAE123. Eur. J. Biochem 1998, 254, 356–362. [Google Scholar]
- Merlino, A.; Russo Krauss, I.; Castellano, I.; de Vendittis, E.; Rossi, B.; Conte, M.; Vergara, A.; Sica, F. Structure and flexibility in cold-adapted iron superoxide dismutases: The case of the enzyme isolated from Pseudoalteromonas haloplanktis. J. Struct. Biol 2010, 172, 343–352. [Google Scholar]
- Aurilia, V.; Rioux-Dube, J.F.; Marabotti, A.; Pezolet, M.; D’Auria, S. Structure and dynamics of cold-adapted enzymes as investigated by FT-IR spectroscopy and MD. The case of an esterase from Pseudoalteromonas haloplanktis. J. Phys. Chem. B 2009, 113, 7753–7761. [Google Scholar]
- Mereghetti, P.; Riccardi, L.; Brandsdal, B.O.; Fantucci, P.; de Gioia, L.; Papaleo, E. Near native-state conformational landscape of psychrophilic and mesophilic enzymes: Probing the folding funnel model. J. Phys. Chem. B 2010, 114, 7609–7619. [Google Scholar]
- Spiwok, V.; Lipovova, P.; Skalova, T.; Duskova, J.; Dohnalek, J.; Hasek, J.; Russell, N.J.; Kralova, B. Cold-active enzymes studied by comparative molecular dynamics simulation. J. Mol. Model 2007, 13, 485–497. [Google Scholar]
- Tiberti, M.; Papaleo, E. Dynamic properties of extremophilic subtilisin-like serine-proteases. J. Struct. Biol 2011, 174, 69–83. [Google Scholar]
- D’Amico, S.; Sohier, J.S.; Feller, G. Kinetics and energetics of ligand binding determined by microcalorimetry: Insights into active site mobility in a psychrophilic alpha-amylase. J. Mol. Biol 2006, 358, 1296–1304. [Google Scholar]
- Sun, K.; Camardella, L.; Di Prisco, G.; Herve, G. Properties of aspartate transcarbamylase from TAD1, a psychrophilic bacterial strain isolated from Antarctica. FEMS Microbiol. Lett 1998, 164, 375–382. [Google Scholar]
- Xu, Y.; Zhang, Y.; Liang, Z.; van de Casteele, M.; Legrain, C.; Glansdorff, N. Aspartate carbamoyltransferase from a psychrophilic deep-sea bacterium, Vibrio strain 2693: Properties of the enzyme, genetic organization and synthesis in Escherichia coli. Microbiology 1998, 144, 1435–1441. [Google Scholar]
- Alvarez, M.; Zeelen, J.P.; Mainfroid, V.; Rentier-Delrue, F.; Martial, J.A.; Wyns, L.; Wierenga, R.K.; Maes, D. Triose-phosphate isomerase (TIM) of the psychrophilic bacterium Vibrio marinus. Kinetic and structural properties. J.Biol. Chem 1998, 273, 2199–2206. [Google Scholar]
- Narinx, E.; Baise, E.; Gerday, C. Subtilisin from psychrophilic Antarctic bacteria: Characterization and site-directed mutagenesis of residues possibly involved in the adaptation to cold. Protein Eng 1997, 10, 1271–1279. [Google Scholar]
- Coquelle, N.; Fioravanti, E.; Weik, M.; Vellieux, F.; Madern, D. Activity, stability and structural studies of lactate dehydrogenases adapted to extreme thermal environments. J. Mol. Biol 2007, 374, 547–562. [Google Scholar]
- Georlette, D.; Jonsson, Z.O.; van Petegem, F.; Chessa, J.; van Beeumen, J.; Hubscher, U.; Gerday, C. A DNA ligase from the psychrophile Pseudoalteromonas haloplanktis gives insights into the adaptation of proteins to low temperatures. Eur. J. Biochem 2000, 267, 3502–3512. [Google Scholar]
- Masullo, M.; Arcari, P.; de Paola, B.; Parmeggiani, A.; Bocchini, V. Psychrophilic elongation factor Tu from the antarctic Moraxella sp. Tac II 25: Biochemical characterization and cloning of the encoding gene. Biochemistry 2000, 39, 15531–15539. [Google Scholar]
- Ruggiero, I.; Raimo, G.; Palma, M.; Arcari, P.; Masullo, M. Molecular and functional properties of the psychrophilic elongation factor G from the Antarctic Eubacterium Pseudoalteromonas haloplanktis TAC 125. Extremophiles 2007, 11, 699–709. [Google Scholar]
- Ciardiello, M.A.; Camardella, L.; Carratore, V.; di Prisco, G. l-Glutamate dehydrogenase from the antarctic fish Chaenocephalus aceratus. Primary structure, function and thermodynamic characterisation: Relationship with cold adaptation. Biochim. Biophys. Acta 2000, 1543, 11–23. [Google Scholar]
- Di Fraia, R.; Wilquet, V.; Ciardiello, M.A.; Carratore, V.; Antignani, A.; Camardella, L.; Glansdorff, N.; Di Prisco, G. NADP+-dependent glutamate dehydrogenase in the Antarctic psychrotolerant bacterium Psychrobacter sp. TAD1. Characterization, protein and DNA sequence, and relationship to other glutamate dehydrogenases. Eur. J. Biochem 2000, 267, 121–131. [Google Scholar]
- D’Amico, S.; Gerday, C.; Feller, G. Structural determinants of cold adaptation and stability in a large protein. J. Biol. Chem 2001, 276, 25791–25796. [Google Scholar]
- Xu, Y.; Feller, G.; Gerday, C.; Glansdorff, N. Moritella cold-active dihydrofolate reductase: Are there natural limits to optimization of catalytic efficiency at low temperature? J. Bacteriol 2003, 185, 5519–5526. [Google Scholar]
- Garsoux, G.; Lamotte, J.; Gerday, C.; Feller, G. Kinetic and structural optimization to catalysis at low temperatures in a psychrophilic cellulase from the Antarctic bacterium Pseudoalteromonas haloplanktis. Biochem. J 2004, 384, 247–253. [Google Scholar]
- Altermark, B.; Niiranen, L.; Willassen, N.P.; Smalas, A.O.; Moe, E. Comparative studies of endonuclease I from cold-adapted Vibrio salmonicida and mesophilic Vibrio cholerae. FEBS J 2007, 274, 252–263. [Google Scholar]
- Birolo, L.; Tutino, M.L.; Fontanella, B.; Gerday, C.; Mainolfi, K.; Pascarella, S.; Sannia, G.; Vinci, F.; Marino, G. Aspartate aminotransferase from the Antarctic bacterium Pseudoalteromonas haloplanktis TAC 125. Cloning, expression, properties, and molecular modelling. Eur. J. Biochem 2000, 267, 2790–2802. [Google Scholar]
- Watanabe, S.; Yasutake, Y.; Tanaka, I.; Takada, Y. Elucidation of stability determinants of cold-adapted monomeric isocitrate dehydrogenase from a psychrophilic bacterium, Colwellia maris, by construction of chimeric enzymes. Microbiology 2005, 151, 1083–1094. [Google Scholar]
- Collins, T.; Meuwis, M.A.; Stals, I.; Claeyssens, M.; Feller, G.; Gerday, C. A novel family 8 xylanase, functional and physicochemical characterization. J. Biol. Chem 2002, 277, 35133–35139. [Google Scholar]
- Xu, Y.; Feller, G.; Gerday, C.; Glansdorff, N. Metabolic enzymes from psychrophilic bacteria: Challenge of adaptation to low temperatures in ornithine carbamoyltransferase from Moritella abyssi. J. Bacteriol 2003, 185, 2161–2168. [Google Scholar]
- Gerike, U.; Danson, M.J.; Russell, N.J.; Hough, D.W. Sequencing and expression of the gene encoding a cold-active citrate synthase from an Antarctic bacterium, strain DS2-3R. Eur. J. Biochem 1997, 248, 49–57. [Google Scholar]
- Li, X.; Jiang, X.; Li, H.; Ren, D. Purine nucleoside phosphorylase from Pseudoalteromonas sp. Bsi590: Molecular cloning, gene expression and characterization of the recombinant protein. Extremophiles 2008, 12, 325–333. [Google Scholar]
- Cartier, G.; Lorieux, F.; Allemand, F.; Dreyfus, M.; Bizebard, T. Cold adaptation in DEAD-box proteins. Biochemistry 2010, 49, 2636–2646. [Google Scholar]
- Tang, M.A.; Motoshima, H.; Watanabe, K. Fluorescence studies on the stability, flexibility and substrate-induced conformational changes of acetate kinases from psychrophilic and mesophilic bacteria. Protein J 2012, 31, 337–344. [Google Scholar]
- D’Amico, S.; Gerday, C.; Feller, G. Temperature adaptation of proteins: Engineering mesophilic-like activity and stability in a cold-adapted alpha-amylase. J. Mol. Biol 2003, 332, 981–988. [Google Scholar]
- Cipolla, A.; D’Amico, S.; Barumandzadeh, R.; Matagne, A.; Feller, G. Stepwise adaptations to low temperature as revealed by multiple mutants of psychrophilic alpha-amylase from Antarctic Bacterium. J. Biol. Chem 2011, 286, 38348–38355. [Google Scholar]
- Asgeirsson, B.; Cekan, P. Microscopic rate-constants for substrate binding and acylation in cold-adaptation of trypsin I from Atlantic cod. FEBS Lett 2006, 580, 4639–4644. [Google Scholar]
- Lonhienne, T.; Gerday, C.; Feller, G. Psychrophilic enzymes: Revisiting the thermodynamic parameters of activation may explain local flexibility. Biochim. Biophys. Acta 2000, 1543, 1–10. [Google Scholar]
- Bjelic, S.; Brandsdal, B.O.; Aqvist, J. Cold adaptation of enzyme reaction rates. Biochemistry 2008, 47, 10049–10057. [Google Scholar]
- Tehei, M.; Franzetti, B.; Madern, D.; Ginzburg, M.; Ginzburg, B.Z.; Giudici-Orticoni, M.T.; Bruschi, M.; Zaccai, G. Adaptation to extreme environments: Macromolecular dynamics in bacteria compared in vivo by neutron scattering. EMBO Rep 2004, 5, 66–70. [Google Scholar]
- Russell, N.J. Toward a molecular understanding of cold activity of enzymes from psychrophiles. Extremophiles 2000, 4, 83–90. [Google Scholar]
- Gianese, G.; Bossa, F.; Pascarella, S. Comparative structural analysis of psychrophilic and meso- and thermophilic enzymes. Proteins 2002, 47, 236–249. [Google Scholar]
- Goldstein, R.A. Amino-acid interactions in psychrophiles, mesophiles, thermophiles, and hyperthermophiles: Insights from the quasi-chemical approximation. Protein Sci 2007, 16, 1887–1895. [Google Scholar]
- Kulakova, L.; Galkin, A.; Nakayama, T.; Nishino, T.; Esaki, N. Cold-active esterase from Psychrobacter sp. Ant300: Gene cloning, characterization, and the effects of Gly→Pro substitution near the active site on its catalytic activity and stability. Biochim. Biophys. Acta 2004, 1696, 59–65. [Google Scholar]
- Mavromatis, K.; Tsigos, I.; Tzanodaskalaki, M.; Kokkinidis, M.; Bouriotis, V. Exploring the role of a glycine cluster in cold adaptation of an alkaline phosphatase. Eur. J. Biochem 2002, 269, 2330–2335. [Google Scholar]
- Sakaguchi, M.; Matsuzaki, M.; Niimiya, K.; Seino, J.; Sugahara, Y.; Kawakita, M. Role of proline residues in conferring thermostability on aqualysin I. J. Biochem 2007, 141, 213–220. [Google Scholar]
- Paredes, D.I.; Watters, K.; Pitman, D.J.; Bystroff, C.; Dordick, J.S. Comparative void-volume analysis of psychrophilic and mesophilic enzymes: Structural bioinformatics of psychrophilic enzymes reveals sources of core flexibility. BMC Struct. Biol 2011, 11, 42. [Google Scholar]
- Feller, G.; D’Amico, D.; Gerday, C. Thermodynamic stability of a cold-active α-amylase from the Antarctic bacterium Alteromonas haloplanctis. Biochemistry 1999, 38, 4613–4619. [Google Scholar]
- Yip, K.S.; Stillman, T.J.; Britton, K.L.; Artymiuk, P.J.; Baker, P.J.; Sedelnikova, S.E.; Engel, P.C.; Pasquo, A.; Chiaraluce, R.; Consalvi, V. The structure of Pyrococcus furiosus glutamate dehydrogenase reveals a key role for ion-pair networks in maintaining enzyme stability at extreme temperatures. Structure 1995, 3, 1147–1158. [Google Scholar]
- Vetriani, C.; Maeder, D.L.; Tolliday, N.; Yip, K.S.; Stillman, T.J.; Britton, K.L.; Rice, D.W.; Klump, H.H.; Robb, F.T. Protein thermostability above 100 degrees C: A key role for ionic interactions. Proc. Natl. Acad. Sci. USA 1998, 95, 12300–12305. [Google Scholar]
- Papaleo, E.; Tiberti, M.; Invernizzi, G.; Pasi, M.; Ranzani, V. Molecular determinants of enzyme cold adaptation: Comparative structural and computational studies of cold- and warm-adapted enzymes. Curr. Protein Pept. Sci 2011, 12, 657–683. [Google Scholar]
- Bell, G.S.; Russell, R.J.; Connaris, H.; Hough, D.W.; Danson, M.J.; Taylor, G.L. Stepwise adaptations of citrate synthase to survival at life’s extremes. From psychrophile to hyperthermophile. Eur. J. Biochem 2002, 269, 6250–6260. [Google Scholar]
- Bae, E.; Phillips, G.N., Jr. Structures and analysis of highly homologous psychrophilic, mesophilic, and thermophilic adenylate kinases. J. Biol. Chem. 2004, 279, 28202–28208. [Google Scholar]
- Mandrich, L.; Pezzullo, M.; Del Vecchio, P.; Barone, G.; Rossi, M.; Manco, G. Analysis of thermal adaptation in the HSL enzyme family. J. Mol. Biol 2004, 335, 357–369. [Google Scholar]
- Tronelli, D.; Maugini, E.; Bossa, F.; Pascarella, S. Structural adaptation to low temperatures-analysis of the subunit interface of oligomeric psychrophilic enzymes. FEBS J 2007, 274, 4595–4608. [Google Scholar]
- Zheng, B.; Yang, W.; Zhao, X.; Wang, Y.; Lou, Z.; Rao, Z.; Feng, Y. Crystal structure of hyperthermophilic endo-beta-1,4-glucanase: Implications for catalytic mechanism and thermostability. J. Biol. Chem 2012, 287, 8336–8346. [Google Scholar]
- De Vendittis, E.; Castellano, I.; Cotugno, R.; Ruocco, M.R.; Raimo, G.; Masullo, M. Adaptation of model proteins from cold to hot environments involves continuous and small adjustments of average parameters related to amino acid composition. J. Theor. Biol 2008, 250, 156–171. [Google Scholar]
- Saelensminde, G.; Halskau, O., Jr; Helland, R.; Willassen, N.P.; Jonassen, I. Structure-dependent relationships between growth temperature of prokaryotes and the amino acid frequency in their proteins. Extremophiles 2007, 11, 585–596. [Google Scholar]
- Saunders, N.F.; Thomas, T.; Curmi, P.M.; Mattick, J.S.; Kuczek, E.; Slade, R.; Davis, J.; Franzmann, P.D.; Boone, D.; Rusterholtz, K.; et al. Mechanisms of thermal adaptation revealed from the genomes of the Antarctic Archaea Methanogenium frigidum and Methanococcoides burtonii. Genome Res 2003, 13, 1580–1588. [Google Scholar]
- Jahandideh, M.; Barkooie, S.M.; Jahandideh, S.; Abdolmaleki, P.; Movahedi, M.M.; Hoseini, S.; Asadabadi, E.B.; Jouni, F.J.; Karami, Z.; Firoozabadi, N.H. Elucidating the protein cold-adaptation: Investigation of the parameters enhancing protein psychrophilicity. J. Theor. Biol 2008, 255, 113–118. [Google Scholar]
- Metpally, R.P.; Reddy, B.V. Comparative proteome analysis of psychrophilic versus mesophilic bacterial species: Insights into the molecular basis of cold adaptation of proteins. BMC Genomics 2009, 10. [Google Scholar] [CrossRef]
- Russell, N.J. Molecular adaptations in psychrophilic bacteria: Potential for biotechnological applications. Adv. Biochem. Eng. Biotechnol 1998, 61, 1–21. [Google Scholar]
- Margesin, R.; Schinner, F. Biotechnological Applications of Cold-adapted Organisms; Springer-Verlag: Berlin/Heidelberg, Germany, 1999. [Google Scholar]
- Gerday, C.; Aittaleb, M.; Bentahier, M.; Chessa, J.P.; Claverie, P.; Collins, T.; D’Amico, S.; Dumont, J.; Garsoux, G.; Georlette, D.; et al. Cold-adapted enzymes: From fundamentals to biotechnology. Trends Biotechnol 2000, 18, 103–107. [Google Scholar]
- Allen, D.; Huston, A.L.; Weels, L.E.; Deming, J.W. Biotechnological Use of Psychrophiles. In Encyclopedia of Environmental Microbiology; Bitton, G., Ed.; John Wiley and Soons: New York, NY, USA, 2002; pp. 1–17. [Google Scholar]
- Cavicchioli, R.; Siddiqui, K.S.; Andrews, D.; Sowers, K.R. Low-temperature extremophiles and their applications. Curr. Opin. Biotechnol 2002, 13, 253–261. [Google Scholar]
- Marx, J.C.; Collins, T.; D’Amico, S.; Feller, G.; Gerday, C. Cold-adapted enzymes from marine Antarctic microorganisms. Mar. Biotechnol 2007, 9, 293–304. [Google Scholar]
- Cavicchioli, R.; Charlton, T.; Ertan, H.; Mohd Omar, S.; Siddiqui, K.S.; Williams, T.J. Biotechnological uses of enzymes from psychrophiles. Microb. Biotechnol 2011, 4, 449–460. [Google Scholar]
- Bioprospecting Information Resource, United Nations University: Tokyo, Japan. Available online: http://www.bioprospector.org/bioprospector/ accessed on 14 September 2012.
- Kobori, H.; Sullivan, C.W.; Shizuya, H. Heat-labile alkaline phosphatase from Antarctic bacteria: Rapid 5′ end labelling of nucleic acids. Proc. Natl. Acad. Sci. USA 1984, 81, 6691–6695. [Google Scholar]
- Rina, M.; Pozidis, C.; Mavromatis, K.; Tzanodaskalaki, M.; Kokkinidis, M.; Bouriotis, V. Alkaline phosphatase from the Antarctic strain TAB5. Properties and psychrophilic adaptations. Eur. J. Biochem 2000, 267, 1230–1238. [Google Scholar]
- Wang, E.; Koutsioulis, D.; Leiros, H.K.; Andersen, O.A.; Bouriotis, V.; Hough, E.; Heikinheimo, P. Crystal structure of alkaline phosphatase from the Antarctic bacterium TAB5. J. Mol. Biol 2007, 366, 1318–1331. [Google Scholar]
- Koutsioulis, D.; Wang, E.; Tzanodaskalaki, M.; Nikiforaki, D.; Deli, A.; Feller, G.; Heikinheimo, P.; Bouriotis, V. Directed evolution on the cold adapted properties of TAB5 alkaline phosphatase. Protein Eng. Des. Sel 2008, 21, 319–327. [Google Scholar]
- Babu, J.; Ramteke, P.W.; Thomas, G. Cold active microbial lipases: Some hot issues and recent developments. Biotechnol. Adv 2008, 26, 457–470. [Google Scholar]
- Lohan, D.; Johnston, S. UNU-IAS Report: Bioprospecting in Antarctica, 2005. Available online: http://www.ias.unu.edu/binaries2/antarctic_bioprospecting.pdf accessed on 14 September 2014.
- Davail, S.; Feller, G.; Narinx, E.; Gerday, C. Cold adaptation of proteins. Purification, characterization, and sequence of the heat-labile subtilisin from the Antarctic psychrophile Bacillus TA41. J. Biol. Chem 1994, 269, 17448–17453. [Google Scholar]
- Van Petegem, F.; Collins, T.; Meuwis, M.A.; Gerday, C.; Feller, G.; Van Beeumen, J. The structure of a cold-adapted family 8 xylanase at 1.3 Å resolution. Structural adaptations to cold and investigation of the active site. J. Biol. Chem 2003, 278, 7531–7539. [Google Scholar]
- Collins, T.; de Vos, D.; Hoyoux, A.; Savvides, S.N.; Gerday, C.; Van Beeumen, J.; Feller, G. Study of the active site residues of a glycoside hydrolase family 8 xylanase. J. Mol. Biol 2005, 354, 425–435. [Google Scholar]
- De Vos, D.; Collins, T.; Nerinckx, W.; Savvides, S.N.; Claeyssens, M.; Gerday, C.; Feller, G.; Van Beeumen, J. Oligosaccharide binding in family 8 glycosidases: Crystal structures of active-site mutants of the beta-1,4-xylanase pXyl from Pseudoaltermonas haloplanktis TAH3a in complex with substrate and product. Biochemistry 2006, 45, 4797–4807. [Google Scholar]
- Collins, T.; Hoyoux, A.; Dutron, A.; Georis, J.; Genot, B.; Dauvrin, T.; Arnaut, F.; Gerday, C.; Feller, G. Use of glycoside hydrolase family 8 xylanases in baking. J. Cereal Sci 2006, 43, 79–84. [Google Scholar]
- Hoyoux, A.; Jennes, I.; Dubois, P.; Genicot, S.; Dubail, F.; Francois, J.M.; Baise, E.; Feller, G.; Gerday, C. Cold-adapted beta-galactosidase from the Antarctic psychrophile Pseudoalteromonas haloplanktis. Appl. Environ. Microbiol 2001, 67, 1529–1535. [Google Scholar]
- Wintrode, P.L.; Arnold, F.H. Temperature adaptation of enzymes: Lessons from laboratory evolution. Adv. Protein Chem 2000, 55, 161–225. [Google Scholar]
- Siddiqui, K.S.; Cavicchioli, R. Improved thermal stability and activity in the cold-adapted lipase B from Candida antarctica following chemical modification with oxidized polysaccharides. Extremophiles 2005, 9, 471–476. [Google Scholar]
- Siddiqui, K.S.; Parkin, D.M.; Curmi, P.M.; de Francisci, D.; Poljak, A.; Barrow, K.; Noble, M.H.; Trewhella, J.; Cavicchioli, R. A novel approach for enhancing the catalytic efficiency of a protease at low temperature: Reduction in substrate inhibition by chemical modification. Biotechnol. Bioeng 2009, 103, 676–686. [Google Scholar]
- Siddiqui, K.S.; Poljak, A.; Cavicchioli, R. Improved activity and stability of alkaline phosphatases from psychrophilic and mesophilic organisms by chemically modifying aliphatic or amino groups using tetracarboxy-benzophenone derivatives. Cell. Mol. Biol 2004, 50, 657–667. [Google Scholar]

| Substrate | Relative activity (%) | |
|---|---|---|
| AHA | PPA | |
| Macromolecular substrates | ||
| Starch | 100 | 100 |
| Amylopectin | 96 | 68 |
| Amylose | 324 | 214 |
| Dextrin | 108 | 95 |
| Glycogen | 74 | 59 |
| Short oligosaccharides | ||
| Maltotetraose G4 | 17 | 22 |
| Maltopentose G5 | 69 | 145 |
| Maltohexaose G6 | 94 | 147 |
| Maltoheptaose G7 | 119 | 155 |
| Maltooligosaccharides (G4 to G10 mix) | 64 | 101 |
| AHA | PPA | |||||
|---|---|---|---|---|---|---|
| Substrate | kcat s−1 | Km mg L−1 | kcat/Km s−1mg−1 L | kcat s−1 | Km mg L−1 | kcat/Km s−1mg−1 L |
| Starch | 663 | 155 | 4.3 | 327 | 41 | 8.0 |
| Amylopectin | 636 | 258 | 2.5 | 222 | 53 | 4.2 |
| Amylose | 2148 | 178 | 12.1 | 700 | 36 | 19.4 |
| Dextrin | 716 | 586 | 1.2 | 311 | 61 | 5.1 |
| Glycogen | 491 | 1344 | 0.3 | 193 | 46 | 4.2 |
| kcat s−1 | ΔG# kcal mol−1 | ΔH# kcal mol−1 | TΔS# kcal mol−1 | |
|---|---|---|---|---|
| Psychrophile | 294 | 13.3 | 8.3 | −5.0 |
| Mesophile | 97 | 14.0 | 11.1 | −2.9 |
| Thermophile | 14 | 15.0 | 16.8 | 1.8 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Struvay, C.; Feller, G. Optimization to Low Temperature Activity in Psychrophilic Enzymes. Int. J. Mol. Sci. 2012, 13, 11643-11665. https://doi.org/10.3390/ijms130911643
Struvay C, Feller G. Optimization to Low Temperature Activity in Psychrophilic Enzymes. International Journal of Molecular Sciences. 2012; 13(9):11643-11665. https://doi.org/10.3390/ijms130911643
Chicago/Turabian StyleStruvay, Caroline, and Georges Feller. 2012. "Optimization to Low Temperature Activity in Psychrophilic Enzymes" International Journal of Molecular Sciences 13, no. 9: 11643-11665. https://doi.org/10.3390/ijms130911643
APA StyleStruvay, C., & Feller, G. (2012). Optimization to Low Temperature Activity in Psychrophilic Enzymes. International Journal of Molecular Sciences, 13(9), 11643-11665. https://doi.org/10.3390/ijms130911643





