In Vitro Control of Post-Harvest Fruit Rot Fungi by Some Plant Essential Oil Components
Abstract
:1. Introduction
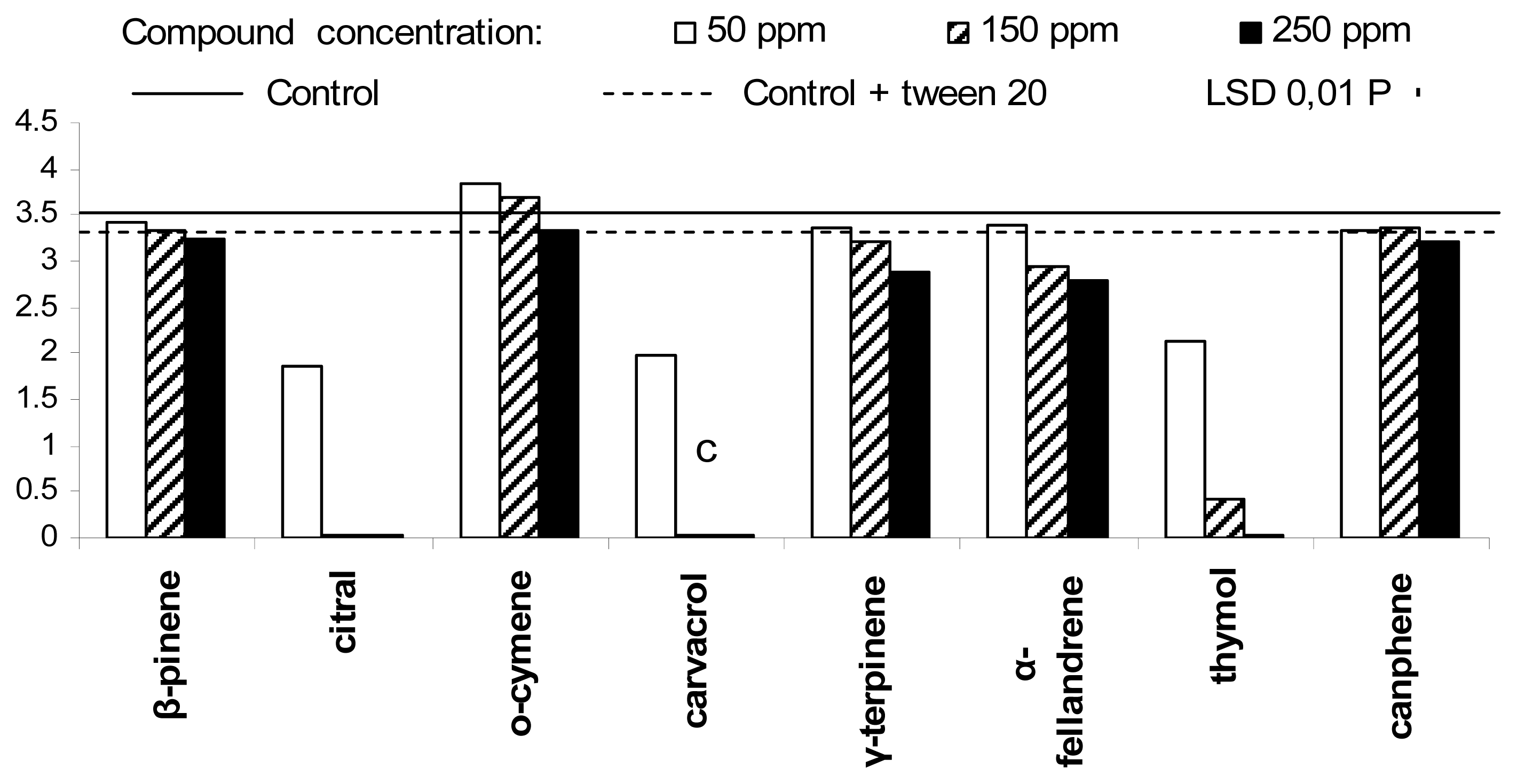
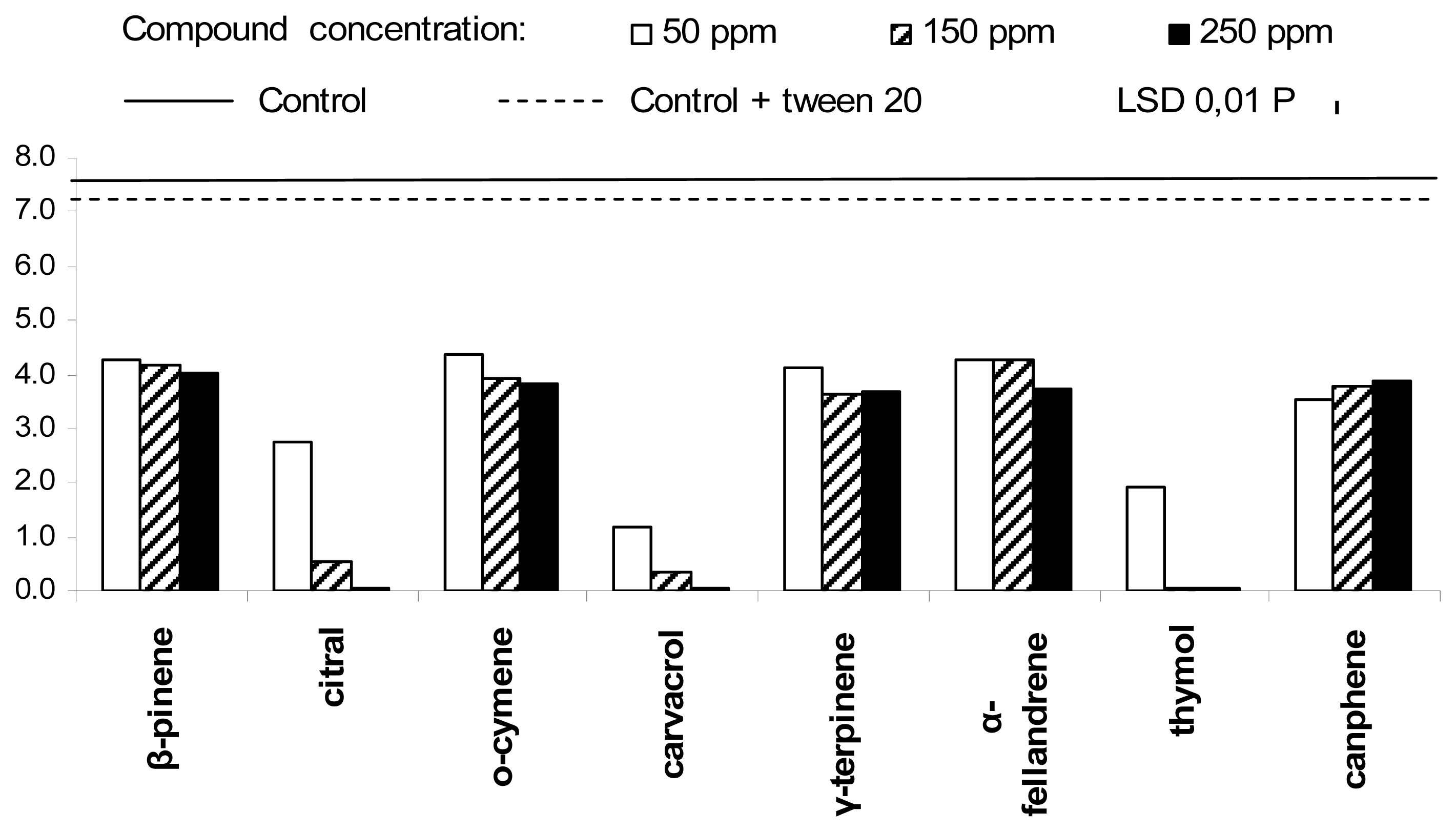
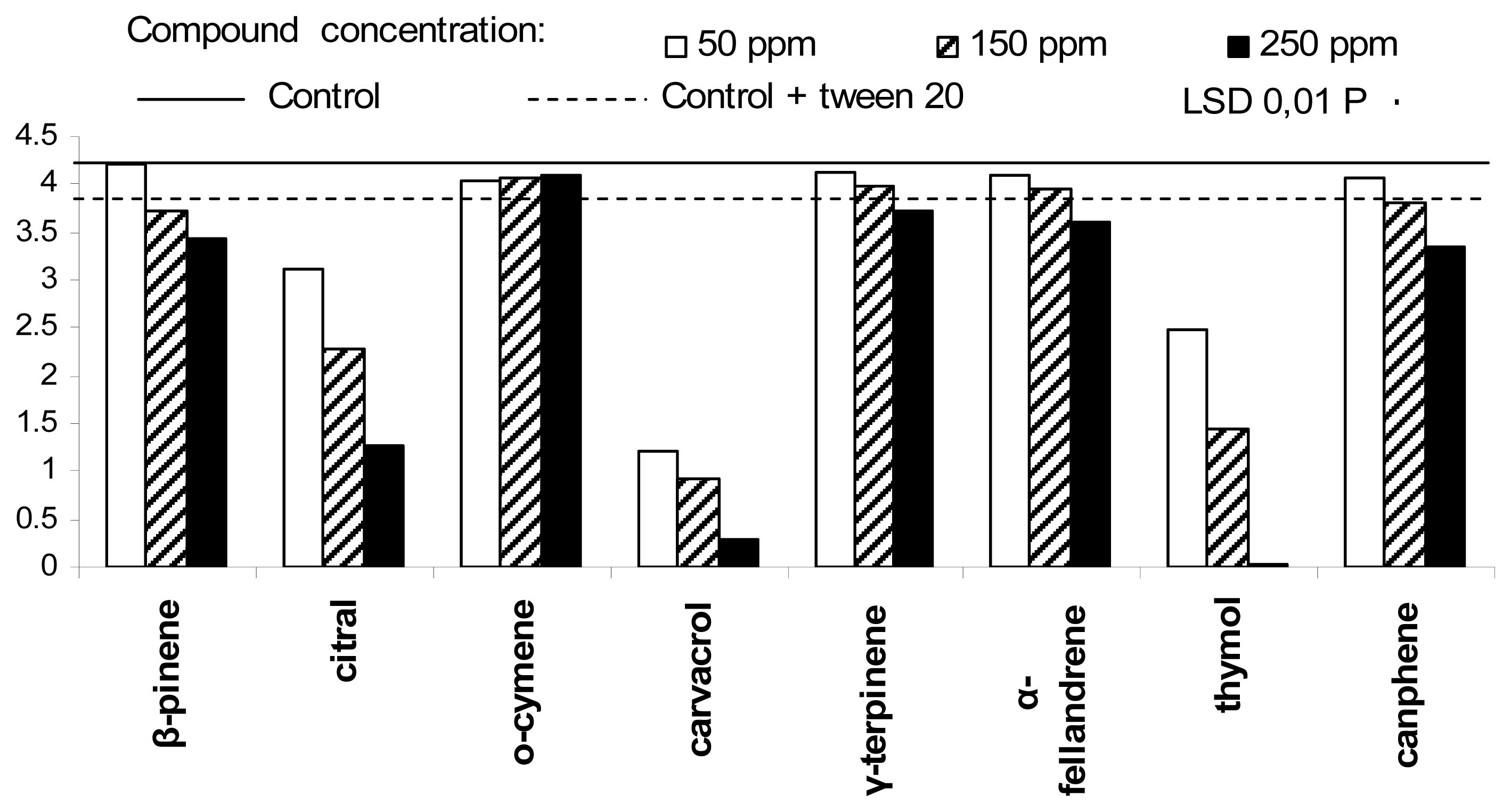
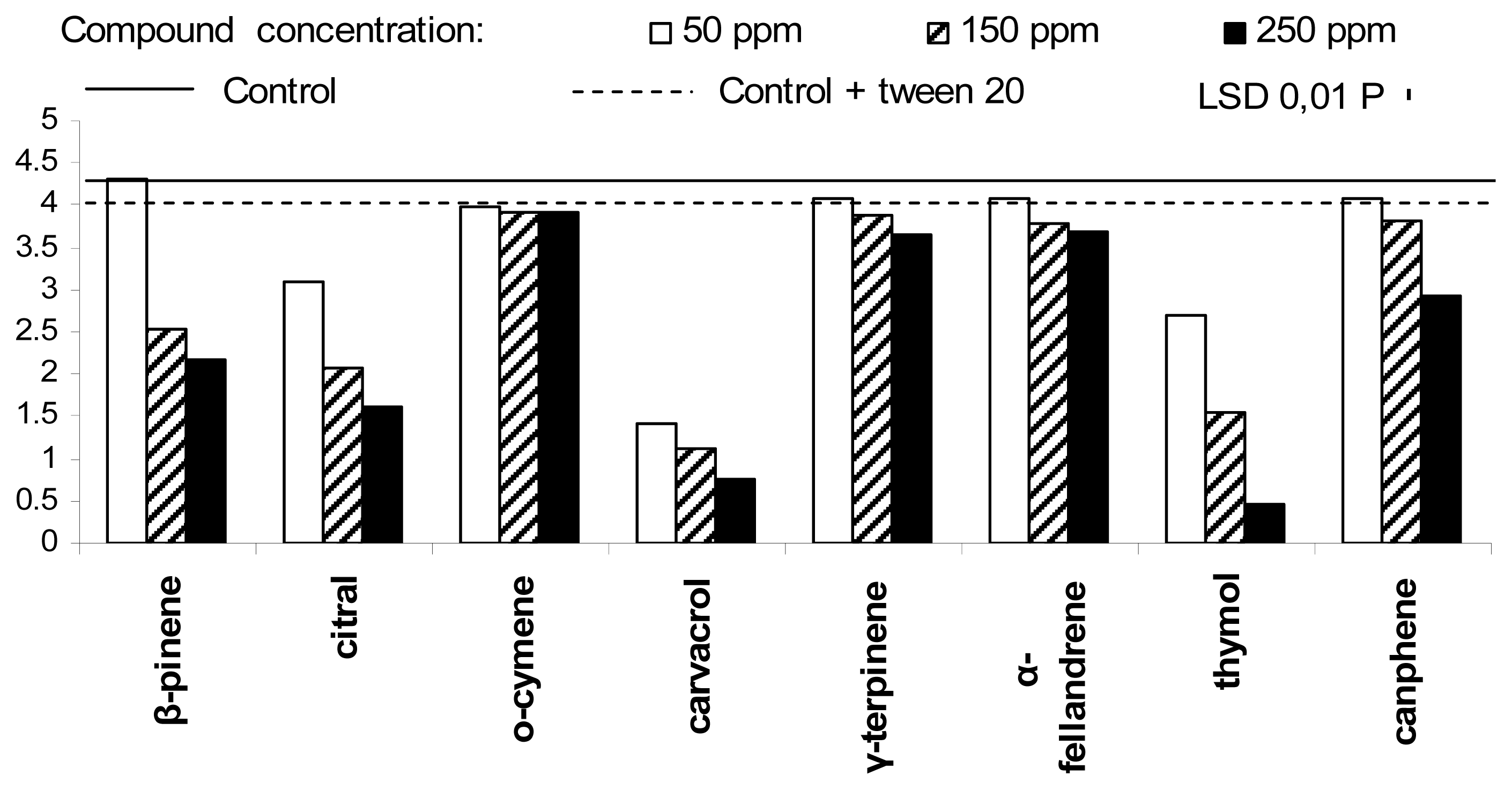
2. Results and Discussion
3. Experimental Section
3.1. Chemicals
3.2. Fungal and Stramenopilus Isolates
3.3. In Vitro Tests
3.4. Evaluation of Fungicidal Effect of Some PEO Components
3.5. Statistical Analysis
4. Conclusions
Abbreviations
| PEOs | plant essential oils |
| PDA | potato dextrose agar |
| CMA | corn meal agar |
References and Notes
- Chang, H.-T.; Cheng, Y.-H.; Wua, C.-L.; Chang, S.-T.; Chang, T.-T.; Su, Y.-C. Antifungal activity of essential oil and its constituents from Calocedrus macrolepis var. formosana Florin leaf against plant pathogenic fungi. Bioresource Technol 2008, 99, 6266–6270. [Google Scholar]
- Lee, Y.-S.; Kim, J.; Lee, S.-G.; Oh, E.; Shin, S.-C.; Park, I.-K. Effects of plant essential oils and components from Oriental sweetgum (Liquidambar orientalis) on growth and morphogenesis of three phytopathogenic fungi. Pestic. Biochem. Phys 2009, 93, 138–143. [Google Scholar]
- Lee, Y.-S.; Kim, J.; Shin, S.-C.; Lee, S.-G.; Park, I.-K. Antifungal activity of Myrtaceae essential oils and their components against three phytopathogenic fungi. Flavour Fragr. J 2008, 23, 23–28. [Google Scholar]
- Kim, M.-K.; Choi, G.-J.; Lee, H.-S. Fungicidal Property of Curcuma longa L. Rhizome-Derived Curcumin against Phytopathogenic Fungi in a Greenhouse. J. Agric. Food Chem 2003, 51, 1578–1581. [Google Scholar]
- Lee, H.-S.; Lee, S.-W.; Cho, K.-Y.; Kim, M.-K.; Ahn, Y.-J. Fungicidal activities of 51 fruit extracts against six phytopathogenic fungi. Agric. Chem. Biotechnol 2001, 44, 147–153. [Google Scholar]
- Isman, M.B. Plant essential oils for pest and disease management. Crop Prot 2000, 19, 603–608. [Google Scholar]
- Plotto, S.; Roberts, D.D.; Roberts, R.G. Evolution of plant essential oil as natural postharvest disease control of tomato (Lycopersicon esculentum). Acta Hortic 2003, 628, 737–745. [Google Scholar]
- Lee, S.-O.; Park, I.-K.; Choi, G.-J.; Lim, H.-K.; Jang, K.-S.; Cho, K.-Y.; Shin, S.-C.; Kim, J.-C. Fumigant activity of essential oils and components of Illicium verum and Schizonepeta tenuifolia against Botrytis cinerea and Colletotrichum gloeosporioides. J. Microbiol. Biotechnol 2007, 17, 1568–1572. [Google Scholar]
- Alilou, H.; Akssira, M.; Hassani, L.I.; Chebli, B.; El Hakmoui, A.; Mellouki, F.; Rouhi, R.; Boira, H.; Blázquez, M.A. Chemical composition and antifungal activity of Bubonium imbricatum volatile oil. Phytopathol. Med 2008, 47, 3–10. [Google Scholar]
- Kim, J.; Lee, Y.-S.; Lee, S.-G.; Shin, S.-C.; Park, I.-K. Fumigant antifungal activity of plant essential oils and components from West Indian bay (Pimenta racemosa) and thyme (Thymus vulgaris) oils against two phytopathogenic fungi. Flavour Fragr. J 2008, 23, 272–277. [Google Scholar]
- Bajpai, V.K.; Lee, T.J.; Kang, S.C. Chemical composition and in vitro control of agricultural plant pathogens by the essential oil and various extracts of Nandina domestica Thunb. J. Sci. Food Agric 2009, 89, 109–116. [Google Scholar]
- Bhaskara-Reddy, M.V.; Angers, P.; Gosselin, A.; Arul, J. Characterization and use of essential oil from Thymus vulgaris against Botrytis cinerea and Rhyzopus stolonifer in strawberry fruits. Phytochemistry 1998, 42, 1515–1520. [Google Scholar]
- Chebli, B.; Hmamouchi, M.; Achouri, M.; Hassani, L.M.I. Composition and in vitro fungitoxic activity of 19 essential oils against two postharvest pathogens. J. Essent. Oil Res 2004, 16, 507–511. [Google Scholar]
- Tzortzakis, N.G. Maintaining postharvest quality of fresh produce with volatile compounds. Innovat. Food Sci. Emerg. Technol 2007, 8, 111–116. [Google Scholar]
- Lopez-Reyes, J.G.; Spadaro, D.; Gullino, M.L.; Garibaldi, A. Efficacy of plant essential oils on postharvest control of rot caused by fungi on four cultivars of apples in vivo. Flavour Fragr. J 2010, 25, 171–177. [Google Scholar]
- Conte, A.; Speranza, B.S.; Sinigaglia, M.; Deinobile, M.A. Effect of lemon extract on food borne microorganisms. J. Food Protect 2007, 70, 1896–1900. [Google Scholar]
- Zambonelli, A.D.; Aulerio, A.Z.; Bianchi, A.; Albasini, A. Effect of essential oils on phytopathogenic fungi in vitro. J. Phytopathol 2008, 144, 491–494. [Google Scholar]
- Camele, I.; De Feo, V.; Altieri, L.; Mancini, E.; De Martino, L.; Rana, G.L. An Attempt of Postharvest Orange Fruit Rot Control Using Essential Oils from Mediterranean Plants. J. Med. Food 2010, 13, 1–9. [Google Scholar]
- Woolf, A. Essential oils poisoning. Clin. Toxicol 1999, 37, 721–727. [Google Scholar]
- Landelle, C.; Francony, G.; Sam-Lai, N.F.; Gaillard, Y.; Vincent, F.; Wrobeleski, I.; Danel, V. Poisoning by lavandin extract in a 18-month-old boy. Clin. Toxicol 2008, 46, 279–281. [Google Scholar]
- Espinosa-Garcia, F.J.; Langenheim, J.H. Effects of sabinene and γ-terpinene from coastal redwood leaves acting singly or in mixtures on the growth of some of their fungus endophytes. Biochem. Syst. Ecol 1991, 19, 643–650. [Google Scholar]
- Pitarokili, D.; Tzakou, O.; Loukis, A. Composition of the essential oil of spontaneous Rosmarinus officinalis from Greece and antifungal activity against phytopathogenic fungi. J. Essent. Oil Res 2008, 20, 457–459. [Google Scholar]
- Kordali, S.; Cakir, A.; Ozer, H.; Cakmakci, R.; Kesdek, M.; Mete, E. Antifungal, phytotoxic and insecticidal properties of essential oil isolated from Turkish Origanum acutidens and its three components, carvacrol, thymol and p-cymene. Bioresource Technol 2008, 99, 8788–8795. [Google Scholar]
- Bouchra, C.; Achouri, M.; Idrissi Hassani, L.M.; Hmamouchi, M. Chemical composition and antifungal activity of essential oils of seven Moroccan Labiatae against Botrytis cinerea Pers: Fr. J. Ethnopharmacol 2003, 89, 165–169. [Google Scholar]
- Tsao, R.; Zhou, T. Antifungal activity of monoterpenoids against postharvest pathogens Botrytis cinerea and Monilinia fructicola. J. Essent. Oil Res 2000, 12, 113–121. [Google Scholar]
- Mueller-Riebau, F.; Berger, B.; Yegen, O. Chemical Composition and Fungitoxic Properties to Phytopathogenic Fungi of Essential Oils of Selected Aromatic Plants Growing Wild in Turkey. J. Agric. Food Chem 1995, 43, 2262–2266. [Google Scholar]
- Martin, J.A.; Solla, A.; Witzell, J.; Gil, L.; Garcia-Vallejo, M.C. Antifungal effect and reduction of Ulmus minor symptoms to Ophiostoma novo-ulmi by carvacrol and salicylic acid. Eur. J. Plant Pathol 2010, 127, 21–32. [Google Scholar]
- Soković, M.D.; Vukojević, J.; Marin, P.D.; Brkić, D.D.; Vajs, V.; van Griensven, L.J.L.D. Chemical Composition of Essential Oils of Thymus and Mentha Species and Their Antifungal Activities. Molecules 2009, 14, 238–249. [Google Scholar]
- Arras, G.; Usai, M. Fungitoxic activity of 12 essential oils against four postharvest citrus pathogens: chemical analysis of Thymus capitatus oil and its effect in subatmospheric pressure conditions. J. Food Protect 2001, 64, 1025–1029. [Google Scholar]
- Caccioni, D.R.L.; Guizzardi, M. Inhibition of germination and growth of fruit and vegetable postharvest pathogenic fungi by essential oil components. J. Essent. Oil Res 1994, 6, 173–179. [Google Scholar]
- Tabanca, N.; Demirci, B.; Crockett, S.L.; Baser, K.H.C.; Wedge, D.E. Chemical Composition and Antifungal Activity of Arnica longifolia, Aster hesperius, and Chrysothamnus nauseosus Essential Oils. J. Agric. Food Chem 2007, 55, 8430–8435. [Google Scholar]
- Sokovic, M.; Griensven, L.J.L.D. Antimicrobial activity of essential oils and their components against the three major pathogens of the cultivated button mushroom, Agaricus bisporus. Eur. J. Plant Pathol 2006, 116, 211–224. [Google Scholar]
- Regnier, T.; Combrinck, S. In vitro and in vivo screening of essential oils for the control of wet bubble disease of Agaricus bisporus. S. Afr. J. Bot 2010, 76, 681–685. [Google Scholar]
- Alzate, O.; Diego, A.; Mier, M.; Gonzalo, I.L.; Afanador, K.; Durango, R.; Diego, L.; Garcia, P.; Carlos, M. Evaluation of phytotoxicity and antifungal activity against Colletotrichum acutatum of essential oils of thyme (Thymus vulgaris), lemongrass (Cymbopogon citratus), and their main constituents. Vitae 2009, 16, 116–125. [Google Scholar]
- Venturini, M.E.; Blanco, D.; Oria, R. In vitro antifungal activity of several antimicrobial compounds against Penicillium expansum. J. Food Protect 2002, 65, 834–839. [Google Scholar]
- Saddiq, A.A.; Khayyat, S.A. Chemical and antimicrobial studies of monoterpene: Citral. Pestic. Biochem. Phys 2010, 98, 89–93. [Google Scholar]
- Moleyar, V.; Narasimham, P. Fungitoxicity of binary mixtures of citral, cinnamic aldehyde, menthol and lemon grass oil against Aspergillus niger and Rhizopus stolonifer. Lebensm. Wiss. Technol 1988, 21, 100–102. [Google Scholar]
- Kishore, G.K.; Pande, S.; Harish, S. Evaluation of essential oils and their components for broad-spectrum antifungal activity and control of late leaf spot and crown rot diseases in peanut. Plant Dis 2007, 91, 375–379. [Google Scholar]
- Lee, H.-C.; Cheng, S.-S.; Chang, S.-T. Antifungal property of the essential oils and their constituents from Cinnamomum osmophloeum leaf against tree pathogenic fungi. J. Sci. Food Agric 2005, 85, 2047–2053. [Google Scholar]
- Wuryatmo, E.; Klieber, A.; Scott, E.S. Inhibition of Citrus Postharvest Pathogens by Vapor of Citral and Related Compounds in Culture. J. Agric. Food Chem 2003, 51, 2637–2640. [Google Scholar]
- Luo, M.; Jiang, L.-K.; Zou, G.-L. The mechanism of loss of germination ability of A. flavus spore with citral. Zhongguo Shengwu Huaxue Yu Fenzi Shengwu Xuebao 2002, 18, 227–233. Chem. Abs 2002, 137, 60144. [Google Scholar]
- Kurita, N.; Miyaji, M.; Kurane, R.; Takahara, Y.; Ichimura, K. Antifungal activity and molecular orbital energies of aldehyde compounds from oils of higher plants. Agric. Biol. Chem 1979, 43, 2365–2371. [Google Scholar]
- Krupa, S.; Nylund, J.E. Ectomycorrhizae of pine. III. Growth inhibition of two root pathogenic fungi by volatile organic constituents of ectomycorrhizal root systems of Pinus sylvestris. Eur. J. Forest. Pathol 1972, 2, 88–94. [Google Scholar]

© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Camele, I.; Altieri, L.; De Martino, L.; De Feo, V.; Mancini, E.; Rana, G.L. In Vitro Control of Post-Harvest Fruit Rot Fungi by Some Plant Essential Oil Components. Int. J. Mol. Sci. 2012, 13, 2290-2300. https://doi.org/10.3390/ijms13022290
Camele I, Altieri L, De Martino L, De Feo V, Mancini E, Rana GL. In Vitro Control of Post-Harvest Fruit Rot Fungi by Some Plant Essential Oil Components. International Journal of Molecular Sciences. 2012; 13(2):2290-2300. https://doi.org/10.3390/ijms13022290
Chicago/Turabian StyleCamele, Ippolito, Luciana Altieri, Laura De Martino, Vincenzo De Feo, Emilia Mancini, and Gian Luigi Rana. 2012. "In Vitro Control of Post-Harvest Fruit Rot Fungi by Some Plant Essential Oil Components" International Journal of Molecular Sciences 13, no. 2: 2290-2300. https://doi.org/10.3390/ijms13022290
APA StyleCamele, I., Altieri, L., De Martino, L., De Feo, V., Mancini, E., & Rana, G. L. (2012). In Vitro Control of Post-Harvest Fruit Rot Fungi by Some Plant Essential Oil Components. International Journal of Molecular Sciences, 13(2), 2290-2300. https://doi.org/10.3390/ijms13022290







