Risks of Using Antifouling Biocides in Aquaculture
Abstract
: Biocides are chemical substances that can deter or kill the microorganisms responsible for biofouling. The rapid expansion of the aquaculture industry is having a significant impact on the marine ecosystems. As the industry expands, it requires the use of more drugs, disinfectants and antifoulant compounds (biocides) to eliminate the microorganisms in the aquaculture facilities. The use of biocides in the aquatic environment, however, has proved to be harmful as it has toxic effects on the marine environment. Organic booster biocides were recently introduced as alternatives to the organotin compounds found in antifouling products after restrictions were imposed on the use of tributyltin (TBT). The replacement products are generally based on copper metal oxides and organic biocides. The biocides that are most commonly used in antifouling paints include chlorothalonil, dichlofluanid, DCOIT (4,5-dichloro-2-n-octyl-4-isothiazolin-3-one, Sea-nine 211®), Diuron, Irgarol 1051, TCMS pyridine (2,3,3,6-tetrachloro-4-methylsulfonyl pyridine), zinc pyrithione and Zineb. There are two types of risks associated with the use of biocides in aquaculture: (i) predators and humans may ingest the fish and shellfish that have accumulated in these contaminants and (ii) the development of antibiotic resistance in bacteria. This paper provides an overview of the effects of antifouling (AF) biocides on aquatic organisms. It also provides some insights into the effects and risks of these compounds on non-target organisms.1. Introduction
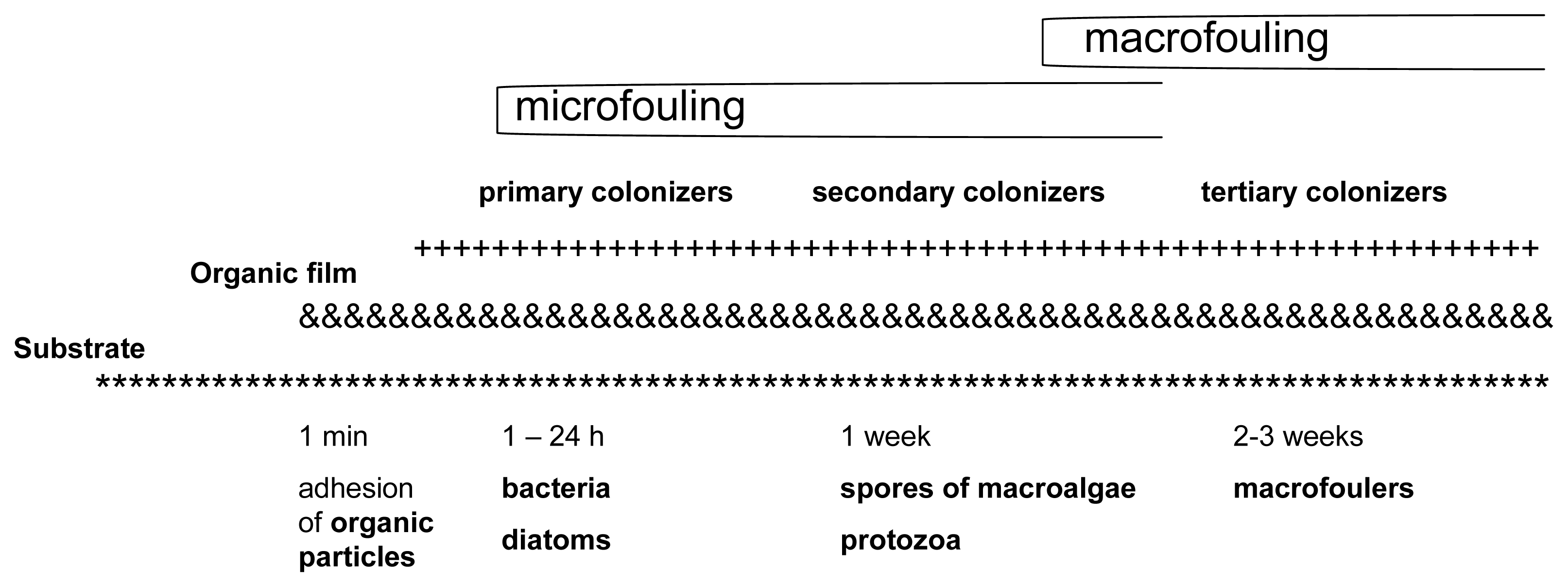
According to the Biocides Directive (98/8/EC) [1], biocides are active substances or preparations that are intended to destroy, deter, render harmless and exercise control or prevent the action of any other harmful organism through chemical or biological means. Biocides are classified into 23 different product types, each of which is comprised of multiple subgroups. Biocides are used because of their potential to destroy a wide range of organisms and for their relatively easy applicability to vessels [2] and aquaculture systems. The settlement of microorganisms, plants and animals is a natural phenomenon that occurs continuously and vigorously on immersed surfaces. This process is called biofouling [3] (Figure 1). Biofouling is a problem for any structure placed in the aquatic environment. It can be controlled through the use of both chemical biocides and non-biocidal technologies [4].
Aquaculture in general, and the fish farming industry in particular, suffer significantly from the effects of biofouling [6]. The aquaculture industry makes periodic discharges of wastes from farm activities. These waste products include detergents, effluent from net washing, antifoulants, heavy metals and even chemicals, such as drugs [7]. The chemicals are essential for aquaculture as they help increase and control the production of seeds in hatcheries, increase feeding efficiency, improve survival rates, control pathogens and diseases and reduce transport stress [8,9].
Nevertheless, despite the beneficial effects of the chemicals to aquaculture, they may also cause potential harm to aquatic organisms [10–12] and even to humans. The chemicals may be ingested by farmed fish and shellfish, which are, in turn, consumed by humans. Ingestion of the contaminated fish and shellfish can pose a great risk to human health [9,13]. The conditions and locations of the aquaculture farms play a significant role on the spread of these chemicals and heavy metals into the environment [14].
Marine pollution caused by the chemicals utilised in aquaculture activities, however, is not yet well documented. In addition, available information indicates low concentrations (low ng L−1 level) [15] of these compounds in the environment. This is due to factors, such as the complexity of the matrix, the high dilution factor, and degradation phenomena [9,16]. Nevertheless, the health risk in animals and humans may increase when bacterial resistance to antibiotics and heavy metals caused by the use of biocides occurs [17–19]. The aim of this study is to review the main effects and risks of using antifouling biocides in aquaculture on aquatic systems, shellfish, fish and humans.
2. Present Situation of Biocide Use in Aquaculture
Aquaculture is the farming of aquatic organisms, including fish, molluscs, crustaceans and aquatic plants, using techniques designed to increase the production and productivity of these organisms beyond the natural capacity of the environment [9]. Since wild fish stocks are reaching the limits of exploitation, we have to rely to a far greater extent on products produced from aquaculture [20]. However, the practice of aquaculture has become so widespread that it has begun to have significant impact on the environment and on natural resources. A number of concerns have been expressed by both environmental activists and scientists regarding this practice [21–25].
With the rapid expansion of the aquaculture industry and with the tightening of the legislation on the use of antifouling (AF) biocides, the problems of aquaculture biofouling have increased [5,6,26,27]. The herbicides or fungicides currently used in aquaculture were originally developed for use in agriculture or as additives for boat anti-fouling paints. As such, the published data regarding their occurrence in marine waters are mainly related to such activities [15,28,29]. Accordingly, many studies have investigated and demonstrated the presence of pesticides and biocides in surface waters [30–34].
With the gradual elimination of triorganotin-based formulations (e.g., tributyltin (TBT)), copper has become the principal biocidal component of most AF paints. It usually comes in the form of copper oxide (Cu2O) [5]. Inorganic zinc is often used in combination with copper to increase the overall toxicity of the formulation or to facilitate the leaching process [35]. Organic booster biocides, such as Irgarol 1051®, Sea Nine 211®, dichlofluanid, chlorothalonil, zinc pyrithione, and Zineb are also added to the paint to enhance its effectiveness [36]. The main AFs used in aquaculture and their effect are shown in Table 1. Nevertheless, these alternatives to TBT are also toxic and their contamination of the aquatic environment has been a topic of increasing importance in recent years [29]. Several studies have evaluated the toxicity of booster biocides on non-target species and have found most of them to be growth inhibitors for freshwater and marine autotrophs [37], influencing key species, such as sea grasses [38] and corals [39]. Therefore, there is increasing interest in the impact of these compounds on the aquatic ecosystems [40].
In the aquatic environment, fishes have been found appropriate to be used as a model for the immunotoxicity testing because they are representatives of aquatic organisms and, therefore, bioindicators of aquatic animal health. As vertebrates that have immune systems strikingly similar to those of mammals, they can also be used to identify potential threats to terrestrial wildlife and humans [41,42]. The risk to predators and humans through the consumption of fish is very low, especially for humans, since the latter are less exposed to the dangers of contamination due to the fact that fish constitutes only a small part of their diet [9]. However, the risk may be increased by mechanisms of resistance.
3. The Main Type of Antifouling Used in Aquaculture and Its Effect on Aquatic Organisms
3.1. Chorothalonil
Chlorothalonil (2,4,5,6-tetrachloroisophthalonitrile) is a pesticide used widely in agriculture, silviculture and urban settings. This pesticide can enter surface waters through rainfall runoff, spray drift or atmospheric deposition, subsequently impacting aquatic biota [77]. It is used as a booster biocide in marine paints as one of the chemicals replacing the widely banned organotin fungicides, such as tributyltin, resulting in greater potential for chlorothalonil contamination of marine waters and sediments [78,79]. Chlorothalonil is a broad-spectrum fungicide with a Kow of 2.64–4.28 and a water solubility of 0.9 mg L−1 [80].
Chlorothalonil can be acutely toxic (50% lethal concentration, LC50) to fish following 96 h exposures ranging from 8.2 to 76 μg L−1, depending on the species and the exposure conditions [48,51]. Chlorothalonil can accumulate in the tissue of fish. Bioaccumulation factors have been reported to be 18 for willow shiner (Gnathopogon caerulescens) and 25 for carp (Cyprinus carpio) following sublethal exposures (1.1–1.4 μg L−1) [81]. It has been suggested that leukocytes may be a potential target of toxicity because significant decreases in leukocyte values were found in the Australian freshwater fish Pseudaphritis urvulii, which was exposed for 10 d to 4.4 μg L−1 chlorothalonil [51]. In vitro studies have demonstrated that the exposure of fish (Morone saxatilus) macrophages and oyster hemocytes to chlorothalonil (10 ± 500 μg L−1) suppressed immunostimulated ROS (reactive oxygen species) and baseline NADPH (nicotinamide adenine dinucleotide phosphate) concentration but did not inhibit phagocytosis [82,83]. There are numerous toxicity studies for chlorothalonil on marine animals, such as crustaceans [43–46], molluscs [44–48], tunicates [47] and teleosts [44–46,49–51].
3.2. Copper Oxide
Copper is an essential metal. However, although it is an effective biocide, it may also affect non-target organisms and cause environmental concerns [84]. The toxicity of copper in water is greatly affected by the chemical form or speciation of the copper and to what degree it is bound to various ligands that may be in the water, making the copper unavailable to organisms [85]. The speciation is essential for understanding the copper’s bioavailability and subsequent toxicity to aquatic organisms [4]. Copper oxide leaches from the boat surfaces and enters the water as a free copper ion (Cu+ ), which is immediately oxidised to Cu2+ and forms complexes with inorganic and organic ligands [4].
Copper is a trace element needed at miniscule levels for the proper functioning of all organisms [84]. However, it can be toxic at higher concentrations [85]. Copper is generally toxic to aquatic organisms, with a lethal concentration 50 (LC50) value varying from 5 to 100,000 μg L−1 [86,87]. However, organisms have different mechanisms by which they cope with and process copper [84]. Generally, copper is actively regulated in fish, decapod crustaceans and algae. It is stored in bivalves, barnacles and aquatic insects [84,88].
The bioavailability, biodistribution to various parts of the organism and bioaccumulation of copper are dramatically influenced by water chemistry. Therefore, water pH, hardness, organic content and salinity play important roles in copper-induced toxicity [84,85]. Thus, increased pH accentuates copper toxicity because of the reduced competition between copper and hydrogen ions at the cell surface [84,89]. In a similar manner, cations that are involved in water hardness also compete with Cu2+ for biological binding sites [84,90].
Copper bound to organic matter is widely thought to be non-bioavailable and, therefore, non-toxic [4,91,92]. Dissolved organic carbon (DOC) content is among the most important factors in reducing copper toxicity in both fresh- and salt-water species [84]. DOC forms organic complexes with copper, thereby reducing copper’s bioavailability [84]. The effects of DOC on reducing the toxicity of copper have been reported in fish [93,94], bivalves [92], echinoderms [95], macroalgae [96], unicellular algae [97], estuarine copepod [98] and planktonic crustaceans [99]. Some authors confirm that water salinity influences the biodistribution and bioaccumulation of copper, affecting its toxicity [54,98,100–102]. Therefore, in oysters, copper accumulation was inversely related to salinity [100].
Copper causes toxicity by impairing the osmoregulation and ion regulation in the gill of numerous aquatic animals [54,55]. In brine shrimp, copper inhibited the Na/K ATPase and Mg2+ ATPase enzyme activity [52]. In mussel, Mytilus galloprovincialis, copper interfered with Ca2+ homeostasis in the gill, causing alterations in the Na/K ATPase and Ca2+ ATPase [53]. Copper depresses the transcription of key genes within the olfactory signal transduction pathway [103]. Additionally, copper toxicity can be induced by generating reactive oxygen species (ROS) [53,104].
It seems remarkable that phytoplankton species have different sensitivities to copper toxicity: resistant (diatoms), intermediate sensitivity (coccolithophores and dinoflagellates) and most sensitive (cyanobacteria) [105,106].
3.3. Dichlofluanid
Dichlofluanid (N-dichlorofluoromethylthio-N0-dimethyl-N-phenylsulphamide) has been commonly used as a herbicide on crops (Lee et al., 2010). Dichlofluanid has a lower toxicity compared with other AF agents, although some studies have identified its toxic effects [107–109], such as embryotoxicity in sea urchin, Glyptocidaris crenularis [56].
3.4. DCOIT (Sea Nine 211®)
One of the new alternative biocides is 4,5-dichloro-2-n-octyl-4-isothiazolin-3-one (DCOIT), the active ingredient of the Sea Nine 211® AF Agent manufactured by Rohm and Haas Company [110]. Aquatic microcosm and marine sediment studies demonstrate that the predominant route of DCOIT dissipation in the marine environment is its rapid biodegradation [110]. DCOIT predominantly undergoes biotic degradation under both aerobic and anaerobic conditions with biological degradation over 200 times faster than hydrolysis or photolysis [4,58,111]. Biodegradation is a very effective mechanism for the detoxification of the compound since the resulting metabolites are five orders of magnitude less toxic than the parent compound [112,113]. However, Sea-Nine antifoulant is acutely toxic to a wide range of aquatic organisms although no chronic toxicological effects have been observed in the extensive toxicology tests conducted on it [114]. DCOIT has a log KOW of 2.8 and an aqueous solubility of 14 mg L−1 [4].
There are numerous studies that have investigated the toxicity and effects of DCOIT on marine animals. These studies demonstrated the following: larval mortality in crustaceans [57,58]: embryo-larva immobility and embryotoxicity in molluscs [46,47], embryotoxicity in echinoderms [59], embryotoxicity and inhibition of larval settlement in tunicates [47] and mortality in teleosts [46,115].
3.5. Diuron
Diuron (1-(3,4-dichlorophenyl)-3,3-dimethylurea) also persists in seawater, but it is less persistent in marine sediments with a half-life of 14 days [116,117]. Diuron is relatively soluble in water (35 mg L−1) and has a reported log KOW of 2.8 [4]. Diuron is present at high concentrations in marine surface waters but it has only been detected at low concentrations in sediments [118,119]. Diuron is persistent in the marine environment and partitions poorly between water and sediments. It can remain suspended and available for uptake by marine organisms [120].
While the toxic effect of the antifoulant herbicide diuron to the photosynthetic aquatic biota has been widely studied, its sublethal effects on the different life stages of fish have been under-reported [121]. Diuron has been proven to be very toxic for the reproduction of the green freshwater alga Scenedesmus vacuolatus [60]. It has also been proven to affect planktonic and periphytic microalgae by reducing the chlorophyll a levels [61–63]. Moreover, it has been proven to be toxic to certain bacterial species [122–124].
3.6. Irgarol-1051®
Irgarol-1051 (2-methylthio-4-terbutylamino-6-cyclopropylamino-s-triazine) is a slightly soluble and moderately lipophilic triazine herbicide used in concert with copper to control fouling on boat hulls [125]. Irgarol inhibits electron transport in the photosystem II (PSII) [126] by binding to the D1 protein [127]. Irgarol may affect non-target photosynthetic organisms, such as phytoplankton, periphyton and aquatic macrophytes [128] when leaching into the marine environment [129].
Only a few studies have addressed the possible effect of Irgarol on marine non-target algae [130]. The effect of Irgarol on green alga Dunaliella tertiolecta [65], Synechococcus sp and Emiliania huxleyi [66], natural phytoplankton communities [131], periphyton colonization [129] and phytoplankton species [130,132,133] has been investigated and the results showed a decrease in growth, inhibition in cell number and a decrease in the photosynthetic activity of these organisms. These effects have been seen in many different marine plants and algae, such as the eelgrass Zostera marina [38,67], the brown macroalga Fucus serratus [69], the green macroalga Enteromorpha intestinalis [70] and the green macroalga Ulva intestinalis [71].
3.7. TCMS Pyridine
TCMS (2,3,5,6-tetrachloro-4-methylsulphonyl pyridine), which was used in both the textile and leather industries, is one of the more recent AF compounds introduced to the market [134]. The toxicity of TCMS towards living organisms has already been evidenced [29,135,136] and substantiated in in vitro studies [137,138]. TCMS has been found to cause immunotoxic effects at concentrations higher than 10 μM in haemocyte cultures of the colonial ascidian Botryllus schlosseri, causing oxidative stress in the process [71,72].
Both diuron and TCMS pyridine exerted immunosuppressant effects on the Botryllus hemocytes when used at concentrations higher than 250 μM and 10 μM, respectively, causing (i) deep changes in the cytoskeleton that irreversibly affect cell morphology and phagocytosis; (ii) induction of DNA damage; and (iii) leakage of oxidative and hydrolytic enzymes due to membrane alteration. Unlike organotin compounds, diuron and TCMS pyridine do not inhibit cytochrome-c-oxidase and only TCMS pyridine triggers oxidative stress.
3.8. Zinc Pyrithione
Zinc pyrithione (ZnPT) (bis(1hydroxy-2(1H)-pyridethionato-o,s)-(T-4)zinc), one of the most popular surrogate AF biocides, has long been widely used as algaecide, bactericide and fungicide [5,139]. ZnPT was found to be highly toxic to aquatic plants and animals [140], but it was assumed to be environmentally neutral because it could easily photo-degrade to less toxic compounds [140,141]. ZnPT is toxic to Japanese medaka fish (Oryzias latipes) and also causes teratogenic effects, such as spinal cord deformities in embryos and on the larvae of zebra fish (Danio rerio) [74] at very low sublethal concentrations [73]. However, there is a lack of data on the toxicity of ZnPT [139].
3.9. Zineb
Zineb (zinc ethylenebis-(dithiocarbamate)) is a widely used foliar fungicide with prime agricultural and industrial applications [142]. Zineb has been registered for use on fruits, vegetables, field crops, ornamental plants and for the treatment of many seeds [142]. It has also been registered as a fungicide in paints and for mould control on fabrics, leather, linen, painted and wood surfaces, and so on [143]. The occurrence of the dithiocarbamates in coastal environments was not reported until 2009 [144] although it is known that these compounds exhibit teratogenicity in fish embryos at relatively low concentrations [75].
3.10. Capsaicin, Econea and Medetomidine
Capsaicin, Econea and medetomidine can be collectively termed as “emerging” biocides [4]. Capsaicin (8-methyl-n-vanillyl-6-nonenamide) is a compound that may emerge as an AF biocide in the future. It has even been evaluated as a marine AF [4,145]. Econea (2-(p-chlorophenyl)-3-cyano-4- bromo-5-trifluoromethyl pyrrole) is being marketed as a metal-free biocidal additive replacement for copper [4]. Medetomidine (4-[1-(2,3-dimethylphenyl)ethyl]-3Himidazole), on the other hand, is a neuroactive catemine that has been shown to be effective in preventing barnacle cyprid settlement by interfering with the regulation of cement production [4,146].
4. Bioaccumulation
The bioconcentration of pesticides and other chemicals into aquatic organisms mainly proceeds by passive diffusion through gills, epithelial tissues, or the gastrointestinal tract [147]. Bioconcentration factors (BCFs) are available for certain biocides in specific tissues. They represent the concentration of a biocide in the tissue per concentration of the biocide in water (L kg−1) [4]. DCOIT has been shown to bioaccumulate in fish at very low levels following exposure to radiolabelled DCOIT [58]. There are no reports of the bioaccumulation of diuron with BCFs of 75 and 22 L kg−1, suggesting that its accumulation in aquatic organisms is unlikely [4,148]. Irgarol 1051 accumulates in freshwater macrophytes [149] and marine macrophytes [67] with BCFs of up to 30,000 L kg−1. It also accumulates in the green alga Tetraselmis suecica under laboratory conditions with BCFs of up to 150,000 mL g−1 [150]. In addition, the accumulation of Zineb in trout (Salmo gairdneri) is reported to be low with a BCF of <100 L kg−1 [4].
5. Resistance
Scientific evidence from bacteriological, biochemical and genetic data indicate that the use of active molecules in the biocidal products may contribute to the increased occurrence of antibiotic resistant bacteria. The selective stress exerted by biocides may favour the existence of bacteria expressing resistance mechanisms and their dissemination. Some biocides have the capacity to maintain the presence of mobile genetic elements that carry genes involved in cross-resistance between biocides and antibiotics. The dissemination of these mobile elements, their genetic organisation and the formation of biofilms, provide conditions that could create a potential risk of development of cross-resistance between antibiotics and biocides [151].
6. Conclusions
Biocides are used as components in paints to coat the structures of vessels, as a means of disinfecting aquaculture facilities and cages, as well as in controlling the biofouling phenomenon (antifouling). The use of biocides is not as well-regulated as drug use in aquaculture because the information available on the effects of these agents to the marine ecosystems is still limited. Hence, it is important to know the risks associated with the existence of those biocides in the marine environment. It is also important to evaluate the effects of these compounds through the continuous monitoring of biocide concentration profiles in water, sediment and biota to provide information that could lead to concerted action to ban or regulate their use.
Acknowledgements
This work was partly supported by projects of the Ministry of Education and Science (AGL2008-05119-C02-01) and the Fundación Séneca (04538/GERM/06). A. Cuesta thanks the Ministry of Science and Innovation of Spain the Ramón y Cajal contract.
References
- European Parliament and of the Council. Directive 98/8/EC of the European Parliament and of the Council of 16 February Concerning the Placing of Biocidal Products on the Market. Official Journal of the European Communities L 123/1 of 24.04.98 1998.
- La Carbona, S.; Viitasalo-Frösen, S.; Masson, D.; Sassi, J.; Pineau, S.; Lehtiniemi, M.; Corroler, D. Efficacy and environmental acceptability of two ballast water treatment chemicals and an alkylamine based-biocide. Sci. Total Environ 2010, 409, 247–255. [Google Scholar]
- Löschau, M.; Krätke, R. Efficacy and toxicity of self-polishing biocide-free antifouling paints. Environ. Pollut 2005, 138, 206–267. [Google Scholar]
- Thomas, K.V.; Brooks, S. The environmental fate and effects of antifouling paint biocides. Biofouling 2010, 26, 73–88. [Google Scholar]
- Yebra, D.M.; Kiil, S.; Dam-Johansen, K. Antifouling technology-past, present and future step towards efficient and environmentally friendly antifouling coatings. Prog. Org. Coat 2004, 50, 75–104. [Google Scholar]
- Bazes, A.; Silkina, A.; Defer, D.; Bernède-Bauduin, C.; Quéméner, E.; Braud, J.P.; Bourgougnon, N. Active substances from Ceramium botryocarpum used as antifouling products in aquaculture. Aquaculture 2006, 258, 664–674. [Google Scholar]
- Read, P.; Fernandes, T. Management of environmental impacts of marine aquaculture in Europe. Aquaculture 2003, 226, 139–163. [Google Scholar]
- Huntington, T.C.; Roberts, H.; Cousins, N.; Pitta, V.; Marchesi, N.; Sanmamed, A.; Hunter-Rowe, T.; Fernandes, T.F.; Tett, P.; McCue, J.; et al. Some Aspects of the Environmental Impact of Aquaculture in Sensitive Areas; Report to the DG Fish and Maritime Affairs of the European Commission: Brussels, Belgium, 2006. [Google Scholar]
- Muñoz, I.; Martínez Bueno, M.J.; Agüera, A.; Fernández-Alba, A.R. Environmental and human health risk assessment of organic micro-pollutants occurring in a Spanish marine fish farm. Environ. Pollut 2010, 158, 1809–1816. [Google Scholar]
- Liess, M.; Schulz, R. Linking insecticide contamination and population response in an agricultural stream. Environ. Toxicol. Chem 1999, 18, 1948–1955. [Google Scholar]
- Chèvre, N.; Loepfe, C.; Singer, H.; Stamm, C.; Fenner, K.; Escher, B.I. Including mixtures in the determination of water quality criteria for herbicides in surface water. Environ. Sci. Technol 2006, 40, 426–435. [Google Scholar]
- Wittmer, I.K.; Scheidegger, R.; Bader, H.P.; Singer, H.; Stamm, C. Loss rates of urban biocides can exceed those of agricultural pesticides. Sci. Total Environ 2011, 409, 920–932. [Google Scholar]
- Hites, R.A.; Foran, J.A.; Carpenter, D.O.; Hamilton, M.C.; Knuth, B.A.; Schwager, S.J. Global assessment of organic contaminants in farmed salmon. Science 2004, 303, 226–229. [Google Scholar]
- Gräslund, S.; Bengtsson, B. Chemicals and biological products used in southeast Asian shrimp farming, and their potential impact on the environment—A review. Sci. Total Environ 2001, 280, 93–131. [Google Scholar]
- Hernando, M.D.; De Vettori, S.; Bueno, M.J.M.; Fernández-Alba, A.R. Toxicity evaluation with Vibrio fischeri test of organic chemicals used in aquaculture. Chemosphere 2007, 68, 724–730. [Google Scholar]
- Pouliquen, H.; Delépée, R.; Larhantec-Verdier, M.; Morvan, M.; Le Bris, H. Comparative hydrolysis and photolysis of four antibacterial agents (oxytetracycline oxolinic acid, flumequine and florfenicol) in deionised water, freshwater and seawater under abiotic conditions. Aquaculture 2007, 262, 23–28. [Google Scholar]
- Brözel, V.S.; Cloete, T.E. Resistance of Pseudomonas aeruginosa to isothiazolone. J. Appl. Bacteriol 1994, 76, 576–582. [Google Scholar]
- Bruins, M.R.; Kapil, S.; Oehme, F.W. Microbial resistance to metals in the environment. Ecotoxicol. Environ. Saf. Mar 2000, 45, 198–207. [Google Scholar]
- Chelossi, E.; Faimali, M. Comparative assessment of antimicrobial efficacy of new potential biocides for treatment of cooling and ballast waters. Sci. Total Environ 2006, 356, 1–10. [Google Scholar]
- Penders, J.; Stobberingh, E.E. Antibiotic resistance of motile aeromonads in indoor catfish and eel farms in the southern part of The Netherlands. Int. J. Antimicrob. Agents 2008, 31, 261–265. [Google Scholar]
- Dierberg, F.E.; Kiattisimukul, W. Issues, impacts, and implications of shrimp aquaculture in Thailand. Environ. Manag 1996, 20, 649–666. [Google Scholar]
- Goldburg, R.; Triplett, T. Murky Waters: Environmental Effects of Aquaculture in the United States; Environmental Defense Fund: Washington, DC, USA, 1997; pp. 1–198. [Google Scholar]
- Naylor, R.L.; Goldburg, R.J.; Mooney, H.; Beveridge, M.; Clay, J.; Folke, C.; Kautsky, N.; Lubchenco, J.; Primavera, J.; Williams, M. Nature’s subsidies to shrimp and salmon farming. Science 1998, 282, 883–884. [Google Scholar]
- Naylor, R.L.; Goldburg, R.J.; Primavera, J.H.; Kautsky, N.; Beveridge, M.C.M.; Clay, J.; Folke, C.; Lubchenco, J.; Mooney, H.; Troell, M. Effect of aquaculture on world fish supplies. Nature 2000, 405, 1017–1024. [Google Scholar]
- Boyd, C.E. Guidelines for aquaculture effluent management at the farm-level. Aquaculture 2003, 226, 101–112. [Google Scholar]
- Fusetani, N. Biofouling and antifouling. Nat. Prod. Rep 2004, 21, 94–104. [Google Scholar]
- Braithwaite, R.A.; McEvoy, L.A. Marine biofouling on fish farms and its remediation. Adv. Mar. Biol 2005, 47, 215–252. [Google Scholar]
- Martinez, K.; Ferrer, I.; Hernando, M.D.; Fernandez-Alba, A.R.; Marce, R.M.; Borrull, F.; Barcelo, D. Occurrence of antifouling biocides in the Spanish Mediterranean marine environment. Environ. Technol 2001, 22, 543–552. [Google Scholar]
- Konstantinou, I.K.; Albanis, T.A. Worldwide occurrence and effects of antifouling paint booster biocides in the aquatic environment. Environ. Int 2004, 30, 235–248. [Google Scholar]
- Hoffmann, R.S.; Capel, D.C.; Larson, S.J. Comparison of pesticides in eight U.S. urban streams. Environ. Toxicol. Chem 2000, 19, 2249–2258. [Google Scholar]
- Blanchoud, H.; Moreau-Guigon, E.; Farrugia, F.; Chevreuil, M.; Mouchel, J.M. Contribution by urban and agricultural pesticide uses to water contamination at the scale of the Marne watershed. Sci. Total Environ 2007, 375, 168–179. [Google Scholar]
- Singer, H.; Jaus, S.; Hanke, I.; Lück, A.; Hollender, J.; Alder, A.C. Determination of biocides and pesticides by on-line solid phase extraction coupled with mass spectrometry and their behaviour in wastewater and surface water. Environ. Pollut 2010, 158, 3054–3064. [Google Scholar]
- Wittmer, I.K.; Bader, H.P.; Scheidegger, R.; Singer, H.; Lück, A.; Hanke, I.; Carlsson, C.; Stamm, C. Significance of urban and agricultural land use for biocide and pesticide dynamics in surface waters. Water Res 2010, 44, 2850–2862. [Google Scholar]
- Wittmer, I.K.; Scheidegger, R.; Bader, H.P.; Singer, H.; Stamm, C. Loss rates of urban biocides can exceed those of agricultural pesticides. Sci. Total Environ 2011, 409, 920–932. [Google Scholar]
- Watermann, B.T.; Daehne, B.; Sievers, S.; Dannenberg, R.; Overbeke, J.C.; Klijnstra, J.W.; Heemken, O. Bioassays and selected chemical analysis of biocidefree antifouling coatings. Chemosphere 2005, 60, 1530–1541. [Google Scholar]
- Parks, R.; Donnier-Marechal, M.; Frickers, P.E.; Turner, A.; Readman, J.W. Antifouling biocides in discarded marine paint particles. Mar. Pollut. Bull 2010, 60, 1226–1230. [Google Scholar]
- Okamura, H.; Nishida, T.; Ono, Y.; Shim, W.J. Phytotoxic effects of antifouling compounds on nontarget plant species. Bull. Environ. Contam. Toxicol 2003, 71, 881–886. [Google Scholar]
- Chesworth, J.C.; Donkin, M.E.; Brown, M.T. The interactive effects of the antifouling herbicides Irgarol 1051 and Diuron on the seagrass Zostera marina (L.). Aquat. Toxicol 2004, 66, 293–305. [Google Scholar]
- Owen, R.; Knap, A.; Toaspern, M.; Carbery, K. Inhibition of coral photosynthesis by the antifoling herbicide Irgarol 1051. Mar. Pollut. Bull 2002, 44, 623–632. [Google Scholar]
- Sánchez-Rodríguez, A.; Sosa-Ferrera, Z.; Santana-del Pino, A.; Santana-Rodríguez, J.J. Probabilistic risk assessment of common booster biocides in surface waters of the harbours of Gran Canaria (Spain). Mar. Pollut. Bull 2011, 62, 985–991. [Google Scholar]
- Anderson, D.P.; Zeeman, M.G. Immunotoxicology in Fish. In Fundamentals of Aquatic Toxicology: Effects, Environmental Fate, and Risk Assessment, 2nd ed; Rand, G.M., Ed.; Taylor & Francis: Washington, DC, USA, 1995; pp. 371–404. [Google Scholar]
- Luebke, R.W.; Hodson, P.V.; Faisal, M.; Ross, P.S.; Grasman, K.A.; Zelikoff, J. Aquatic pollution-induced immunotoxicity in wildlife species. Fundam. Appl. Toxicol 1997, 37, 1–15. [Google Scholar]
- Armstrong, D.A.; Buchanan, D.V.; Caldwell, R.S. A mycosis caused by Lagneidium sp. in laboratory-reared larvae of the dungeness crab, Cancer magister, and possible chemical treatments. J. Invertebr. Pathol 1976, 28, 329–336. [Google Scholar]
- Mayer, F.L. Acute Toxicity Handbook of Chemicals to Estuarine Organisms, Gulf Breeze, Florida; US Environmental Protection Agency: Washington, DC, USA, 1987. [Google Scholar]
- Montforts, M.H.M.M. Chlorothalonil; CSR Advisory Report 06105A00: Bilthoven, The Netherlands, 1999. [Google Scholar]
- U.S. EPA Office of Pesticide Programs. Pesticide Ecotoxicity Database (Formerly: Environmental Effects Database (EEDB)); Environmental Fate and Effects Division, U.S. EPA: Washington, DC, USA, 2000.
- Bellas, J. Comparative toxicity of alternative antifouling biocides on embryos and larvae of marine invertebrates. Sci. Total Environ 2006, 367, 573–585. [Google Scholar]
- Ernst, W.; Doe, K.; Jonah, P.; Young, J.; Julien, G.; Hennigar, P. The toxicity of chlorothalonil to aquatic fauna and the impact of its operational use on a pond ecosystem. Arch. Environ. Contam. Toxicol 1991, 21, 1–9. [Google Scholar]
- Yokoyama, T.; Saka, H.; Fujita, S.; Nishiuchi, Y. Sensitivity of Japanese eel, Anguilla japonica, to 68 kinds of agricultural chemicals. Bull. Agric. Chem. Insp. Stn 1988, 28, 26–33. [Google Scholar]
- Davies, P.E.; White, R.W.G. The toxicology and metabolism of chlorothalonil in fish. I. Lethal levels for Salmo gairdneri, Galaxias maculatus, G. truttaceus and G. auratus and the fate of 14C-TCIN in S. gairdneri. Aquat. Toxicol 1985, 7, 93–105. [Google Scholar]
- Davies, P.E.; Cook, L.S.J.; Goenarso, D. Sublethal responses to pesticides of several species of Australian freshwater fish and crustaceans and rainbow trout. Environ. Toxicol. Chem 1994, 13, 1341–1354. [Google Scholar]
- Katranitsas, A.; Castritsi-Catharios, J.; Persoone, G. The effects of a copper-based antifouling paint on mortality and enzymatic activity of a non-target marine organism. Mar. Pollut. Bull 2003, 46, 1491–1494. [Google Scholar]
- Viarengo, A.; Pertica, M.; Mancinelli, G.; Burlando, B.; Canesi, L.; Orunesu, M. In vivo effects of copper on calcium homeostasis mechanisms of mussel gill cell plasma membranes. Comp. Biochem. Physiol. Part C 1996, 113, 421–425. [Google Scholar]
- Blanchard, J.; Grosell, M. Effects of salinity on copper accumulation in the common killifish (Fundulus heteroclitus). Environ. Toxicol. Chem 2005, 24, 1403–1413. [Google Scholar]
- McIntyre, J.K.; Baldwin, D.H.; Meador, J.P.; Scholz, N.L. Chemosensory deprivation in juvenile Coho salmon exposed to dissolved copper under varying water chemistry conditions. Environ. Sci. Technol 2008, 42, 1352–1358. [Google Scholar]
- Xu, X.; Wang, X.; Li, Y.; Wang, Y.; Wang, Y. Acute toxicity and synergism of binary mixtures of antifouling biocides with heavy metals to embryos of sea urchin Glyptocidaris crenularis. Hum. Exp. Toxicol 2011, 30, 1009–1021. [Google Scholar]
- Willemsen, P.R.; Overbeke, K.; Suurmond, A. Repetitive testing of TBTO, Sea-Nine 211 and farnesol using Balanus amphitrite (Darwin) cypris larvae: variability in larval sensitivity. Biofouling 1998, 12, 133–147. [Google Scholar]
- Jacobson, A.H.; Willingham, G.L. Sea-Nine antifoulant: An environmentally acceptable alternative to organotin antifoulants. Sci. Total Environ 2000, 258, 103–110. [Google Scholar]
- Kobayashi, N.; Okamura, H. Effects of new antifouling compounds on the development of sea urchin. Mar. Pollut. Bull 2002, 44, 748–751. [Google Scholar]
- Backhaus, T.; Faust, M.; Scholze, M.; Gramatica, P.; Vighi, M.; Grimme, L.H. Joint algal toxicity of phenylurea herbicides is equally predictable by concentration addition and independent action. Environ. Toxicol. Chem 2004, 23, 258–264. [Google Scholar]
- Molander, S.; Blanck, H. Detection of pollution-induced community tolerance (PICT) in marine periphyton communities established under diuron exposure. Aquat. Toxicol 1992, 22, 129–144. [Google Scholar]
- Arrhenius, A.; Grönvall, F.; Scholze, M.; Backhaus, T.; Blanck, H. Predictability of the mixture toxicity of 12 similarly acting congeneric inhibitors of photosystem II in marine periphyton and epipsammon communities. Aquat. Toxicol 2004, 68, 351–367. [Google Scholar]
- Perschbacher, P.W.; Ludwig, G.M. Effects of diuron and other aerially applied cotton herbicides and defoliants on the plankton communities of aquaculture ponds. Aquaculture 2004, 233, 197–203. [Google Scholar]
- Bretaud, S.; Toutant, J.P.; Saglio, P. Effects of Carbofuran, Diuron, and Nicosulfuron on Acetylcholinesterase Activity in Goldfish (Carassius auratus). Ecotoxicol. Environ. Saf 2000, 47, 117–124. [Google Scholar]
- Gatidou, G.; Kotrikla, A.; Rontogianni, V.; Thomaidis, N.S.; Lekkas, T.D. The Toxic Effects of the Antifouling Biocide Irgarol 1051 and Its Principal Metabolite on the Green Alga Dunaliella Tertiotecta. Proceedings of 8th International Conference on Environmental Science and Technology, Lemnos island, Greece, 8–10 September 2003.
- Devilla, R.A.; Brown, M.T.; Donkin, M.; Tarran, G.A.; Aiken, J.; Readman, J.W. Impact of antifouling booster biocides on single microalgal species and on a natural marine phytoplankton community. Mar. Ecol. Prog. Ser 2005, 256, 1–12. [Google Scholar]
- Scarlett, A.; Donkin, P.; Fileman, T.W.; Morris, R.J. Occurrence of the Antifouling Herbicide, Irgarol 1051, within Coastal-water Seagrasses from Queensland, Australia. Mar. Pollut. Bull 1999, 38, 687–691. [Google Scholar]
- Braithwaite, R.A.; Fletcher, R.L. The toxicity of Irgarol 1051 and Sea-Nine 211 to the non-target macroalga Fucus serratus Linnaeus, with the aid of an image capture and analysis system. J. Exp. Mar. Bio. Ecol 2005, 322, 111–121. [Google Scholar]
- Scarlett, A.; Donkin, M.E.; Fileman, T.W.; Donkin, P. Occurrence of the marine antifouling agent irgarol 1051 within the Plymouth Sound locality: Implications for the green macroalga Enteromorpha intestinalis. Mar. Pollut. Bull 1997, 34, 645–651. [Google Scholar]
- Tolhurst, L.E.; Barry, J.; Dyer, R.A.; Thomas, K.V. The effect of resuspending sediment contaminated with antifouling paint particles containing Irgarol 1051 on the marine macrophyte Ulva intestinalis. Chemosphere 2007, 68, 1519–1524. [Google Scholar]
- Menin, A.; Ballarin, L.; Marin, M.G.; Cima, F. Immunotoxicity in ascidians of antifouling compounds alternative to organotins, the case of Diuron and TCMS pyridine. J. Environ. Sci. Health B 2008, 43, 644–654. [Google Scholar]
- Menin, A.; Ballarin, L. Immunomodulatory molecules in the compound ascidian Botryllus schlosseri: Evidence from conditioned media. J. Invertebr. Pathol 2008, 99, 275–280. [Google Scholar]
- Sánchez-Bayo, F.; Goka, K. Unexpected effects of zinc pyrithione and imidacloprid on Japanese medaka fish (Oryzias latipes). Aquat. Toxicol 2005, 74, 285–293. [Google Scholar]
- Goka, K. Embryotoxicity of zinc pyrithione, an antidandruff chemical, in Fish. Environ. Res 1999, 81, 81–83. [Google Scholar]
- van Leeuwen, C.J.; Espeldoorn, A.; Mol, F. Aquatic toxicological aspects of dithiocarbamates and related compounds. III. Embryolarval studies with rainbow trout (Salmo gairdneri). Aquat. Toxicol 1986, 9, 129–145. [Google Scholar]
- Dafforn, K.A.; Lewis, J.A.; Johnston, E.L. Antifouling strategies: History and regulation, ecological impacts and mitigation. Mar. Pollut. Bull 2011, 62, 453–465. [Google Scholar]
- Reregistration Eligibility Decision: Chlorothalonil, U.S. Environmental Protection Agency (USEPA) ed.; Office of Prevention, Pesticides and Toxic Substances, EPA: Washington, DC, USA, 1999.
- Voulvoulis, N.; Scrimshaw, M.D.; Lester, J.N. Occurrence of four biocides utilized in antifouling paints, as alternatives to organotin compounds, in waters and sediments of a commercial estuary in the UK. Mar. Pollut. Bull 2000, 40, 938–946. [Google Scholar]
- Voulvoulis, N.; Scrimshaw, M.D.; Lester, J.N. Partitioning of selected antifouling in the aquatic environment. Mar. Environ. Res 2000, 53, 1–16. [Google Scholar]
- Caux, P.Y.; Kent, R.A.; Fan, G.T.; Stephenson, G.L. Environmental fate and effects of chlorothalonil: a Canadian perspective. Crit. Rev. Environ. Sci. Technol 1996, 26, 45–93. [Google Scholar]
- Tsuda, T.; Aoki, S.; Kojima, M.; Fujita, T. Accumulation and excretion of pesticides used in golf courses by carp (Cyprinus carpio) and willow shiner (Gnathopogon caerulescens). Comp. Biochem. Physiol. C 1992, 101, 63–66. [Google Scholar]
- Baier-Anderson, C.; Anderson, R.S. Suppression of superoxide production by chlorothalonil in striped bass (Morone saxatilus) macrophages: the role of cellular sulfhydryls and oxidative stress. Aquat. Toxicol 2000, 50, 85–96. [Google Scholar]
- Baier-Anderson, C.; Anderson, R.S. The effects of chlorothalonilon oyster hemocyte activation: Phagocytosis, reduced pyridine nucleotides, and reactive oxygen species production. Environ. Res 2000, 83, 72–78. [Google Scholar]
- Kiaune, L.; Singhasemanon, N. Pesticidal copper (I) oxide: Environmental fate and aquatic toxicity. Rev. Environ. Contam. Toxicol 2011, 213, 1–26. [Google Scholar]
- Burridge, L.; Weis, J.; Cabello, F.; Pizarro, J. Chemical use in salmon aquaculture: A review of current practices and possible environmental effects. Aquaculture 2010, 306, 7–23. [Google Scholar]
- Ambient Water Quality Criteria for Copper, U.S. Environmental Protection Agency (EPA) ed.; Office of Water and Office of Science and Technology, U.S. Government Printing Office: Washington, DC, USA, 1985.
- Ambient Water Quality Criteria - Saltwater Copper Addendum, U.S. Environmental Protection Agency (EPA) ed.; Office of Water and Office of Science and Technology, U.S. Government Printing Office: Washington, DC, USA, 1995.
- Brix, K.V.; DeForest, D.K. Critical Review of the Use of Bioconcentration Factors for Hazard Classification of Metals and Metal Compounds; Parametrix Inc.: Washington, DC, USA, 2000. [Google Scholar]
- Wilde, K.L.; Stauber, J.L.; Markich, S.J.; Franklin, N.M.; Brown, P.L. The effects of pH on the uptake and toxicity of copper and zinc in a tropical freshwater alga (Chlorella sp). Arch. Environ. Contam. Toxicol 2006, 51, 174–185. [Google Scholar]
- Boulanger, B.; Nikolaidis, N.P. Mobility and aquatic toxicity of copper in an urban watershed. J. Am. Water Resour. Assoc 2003, 39, 325–336. [Google Scholar]
- Arnold, W.R.; Santore, R.C.; Cotsifas, J.S. Predicting copper toxicity in estuarine and marine waters using the biotic ligand model. Mar. Pollut. Bull 2005, 50, 1634–1640. [Google Scholar]
- Brooks, S.J.; Bolam, T.; Tolhurst, L.; Bassett, J.; La Roche, J.; Waldock, M.; Barry, J.; Thomas, K.V. The effects of dissolved organic carbon on the toxicity of copper to the developing embryos of the Pacific oyster, Crassostrea gigas. Environ. Toxicol. Chem 2007, 26, 1756–1763. [Google Scholar]
- Playle, R.C.; Dixon, D.G.; Burnison, K. Copper and cadmium binding to fish gills: Modification by dissolved organic carbon and synthetic ligands. Can. J. Fish. Aquat. Sci 1993, 50, 2667–2677. [Google Scholar]
- Playle, R.C.; Dixon, D.G.; Burnison, K. Copper and cadmium binding to fish gills: Estimates of metal-gill stability constants and modelling of metal accumulation. Can. J. Fish. Aquat. Sci 1993, 50, 2678–2687. [Google Scholar]
- Lorenzo, J.I.; Nieto, O.; Beiras, R. Anodic stripping voltammetry measures copper bioavailability for sea urchin larvae in the presence of fulvic acids. Environ. Toxicol. Chem 2006, 25, 73–81. [Google Scholar]
- Brooks, S.J.; Bolam, T.; Tolhurst, L.; Bassett, J.; La Roche, J.; Waldock, M.; Barry, J.; Thomas, K.V. Dissolved organic carbon reduces the toxicity of copper to germlings of the macroalgae, Fucus vesiculosus. Ecotoxicol. Environ. Saf 2008, 70, 88–98. [Google Scholar]
- Florence, T.M.; Stauber, J.L. Toxicity of copper complexes to the marine diatom Nitzschia closterium. Aquat. Toxicol 1986, 8, 11–26. [Google Scholar]
- Hall, L.W., Jr; Anderson, R.D.; Lewis, B.L.; Arnold, W.R. The influence of salinity and dissolved organic carbon on the toxicity of copper to the estuarine copepod, Eurytemora affinis. Arch. Environ. Contam. Toxicol. 2008, 54, 44–56. [Google Scholar]
- Kramer, K.J.M.; Jak, R.G.; van Hattum, B.; Hoftman, R.N.; Zwolsman, J.J.G. Copper toxicity in relation to surface water-dissolved organic matter: biological effects to Daphnia magna. Environ. Toxicol. Chem 2004, 23, 2971–2980. [Google Scholar]
- Amiard-Triquet, C.; Berthet, B.; Martoja, R. Influence of salinity on trace metal (Cu, Zn, Ag) accumulation at the molecular, cellular and organism level in the oyster Crassostrea gigas Thunberg. Biol. Met 1991, 4, 144–150. [Google Scholar]
- Grosell, M.; Blanchard, J.; Brix, K.V.; Gerdes, R. Physiology is pivotal for interactions between salinity and acute copper toxicity to fish and invertebrates. Aquat. Toxicol 2007, 84, 162–172. [Google Scholar]
- Polo, A.D.; Scrimshaw, M. Challenges for the development of a biotic ligand model predicting copper toxicity in estuaries and seas. Environ. Toxicol. Chem 2011. in press. [Google Scholar]
- Tilton, F.; Tilton, S.C.; Bammler, T.K.; Beyer, R.; Farin, F.; Stapleton, P.L.; Gallagher, E.P. Transcription biomarkers and mechanisms of copper-induced olfactory injury in zebrafish. Environ. Sci. Technol 2008, 42, 9404–9411. [Google Scholar]
- Bopp, S.K.; Abicht, H.K.; Knauer, K. Copper-induced oxidative stress in rainbow trout gill cells. Aquat. Toxicol 2008, 86, 197–204. [Google Scholar]
- Brand, L.; Sunda, W.G.; Guillard, R.R.L. Reduction of marine phytoplankton reproduction rates by copper and cadmium. J. Exp. Mar. Biol. Ecol 1986, 96, 225–250. [Google Scholar]
- Beck, N.G.; Bruland, K.W.; Rue, E.L. Short-term biogeochemical influence of a diatom bloom on the nutrient and trace metal concentrations in South San Francisco Bay microcosm experiments. Estuaries 2002, 25, 1063–1076. [Google Scholar]
- Wang, H.; Li, Y.; Huang, H.; Xu, X.; Wang, Y. Toxicity evaluation of single and mixed antifouling biocides using the Strongylocentrotus intermedius sea urchin embryo test. Environ. Toxicol. Chem 2011, 30, 692–703. [Google Scholar]
- Lee, S.E.; Won, H.S.; Lee, Y.W. Study on the new antifouling compounds in Korean coasts. Bull. Environ. Contam. Toxicol 2010, 85, 538–543. [Google Scholar]
- Lee, S.; Chung, J.; Won, H.; Lee, D.; Lee, Y.W. Analysis of antifouling agents after regulation of tributyltin compounds in Korea. J. Hazard. Mater 2011, 185, 1318–1325. [Google Scholar]
- Steen, R.J.; Ariese, F.; van Hattum, B.; Jacobsen, J.; Jacobson, A. Monitoring and evaluation of the environmental dissipation of the marine antifoulant 4,5-dichloro-2-n-octyl-4-isothiazolin-3-one (DCOIT) in a Danish Harbor. Chemosphere 2004, 57, 513–521. [Google Scholar]
- Callow, M.E.; Willingham, G.L. Degradation of antifouling biocides. Biofouling 1996, 10, 239–249. [Google Scholar]
- Jacobson, A. Environmentally Preferable Alternatives to TBT and Their Impact on the Environment. Proceedings of IBC UK Conferences, Marine Environmental Regulations: The Cost to the Shipping Industry, LM 1120, London, UK, 23–24 September 1998; IBC UK Limited: London, UK, 1998. [Google Scholar]
- Jacobson, A.; Williams, T.M. Environmental fate of isothiazolone biocides. Chem. Oggi 2000, 18, 105–108. [Google Scholar]
- Shade, W.D.; Hurt, S.S.; Jacobson, A.H.; Reinert, K.H. Ecological Risk Assessment of a Novel Marine Antifoulant. In Environmental Toxicology and Risk Assessment; Gorsuch, J.W., Dwyer, F.W., Ingersoll, C.M., LaPoint, T.W., Eds.; American Society for Testing and Materials: Philadelphia, PA, USA, 1994; Volume 2, pp. 381–408. [Google Scholar]
- Cima, F.; Bragadin, M.; Ballarin, L. Toxic effects of new antifouling compounds on tunicate haemocytes I. Sea-Nine 211TM and chlorothalonil. Aquat. Toxicol 2008, 86, 299–312. [Google Scholar]
- Callow, M.E.; Finlay, J.A. A simple method to evaluate the potential for degradation of antifouling biocides. Biofouling 1995, 9, 153–165. [Google Scholar]
- Thomas, K.V.; McHugh, M.; Waldock, M. Antifouling paint booster biocides in UK coastal waters: inputs, occurrence and environmental fate. Sci. Total Environ 2002, 293, 117–127. [Google Scholar]
- Thomas, K.V.; Blake, S.J.; Waldock, M.J. Antifouling paint booster biocide contamination in UK marine sediments. Mar. Pollut. Bull 2000, 40, 739–745. [Google Scholar]
- Lamoree, M.H.; Swart, C.P.; van der Horst, A.; van Hattum, B. Determination of diuron and the antifouling paint biocide Irgarol 1051 in Dutch marinas and coastal waters. J. Chromatogr. A 2002, 970, 183–190. [Google Scholar]
- Okamura, H.; Watanabe, T.; Aoyama, I.; Hasobe, M. Toxicity evaluation of new antifouling compounds using suspension-cultured fish. Chemosphere 2002, 46, 945–951. [Google Scholar]
- Gagnon, M.M.; Rawson, C.A. Diuron increases spinal deformity in early-life-stage pink snapper Pagrus auratus. Mar. Pollut. Bull 2009, 58, 1078–1095. [Google Scholar]
- Canna-Michaelidou, S.; Nicolaou, A.S. Evaluation of the genotoxicity potential (by MutatoxTM test) of ten pesticides found as water pollutants in Cyprus. Sci. Total Environ 1996, 193, 27–35. [Google Scholar]
- Strachan, G.; Preston, S.; Maciel, H.; Porter, A.J.R.; Paton, G.I. Use of bacterial biosensors to interpret the toxicity and mixture toxicity of herbicides in freshwater. Water Res 2001, 35, 3490–3495. [Google Scholar]
- Tixier, C.; Sancelme, M.; Bonnemoy, F.; Cuer, A.; Veschambre, H. Degradation products of a phenylurea herbicide, diuron: synthesis, ecotoxicity, and biotransformation. Environ. Toxicol. Chem 2001, 20, 1381–1389. [Google Scholar]
- Hall, L.W., Jr; Killen, W.D.; Anderson, R.D.; Balcomb, R.; Gardinali, P. Ecological risk of Irgarol 1051 and its major metabolite in coastal California marinas and reference areas. Mar. Pollut. Bull. 2009, 58, 702–710. [Google Scholar]
- Holt, J.S. Mechanisms and agronomic aspects of herbicide resistance. Ann. Rev. Plant Physiol. Plant Mol. Biol 1993, 44, 203–229. [Google Scholar]
- Ranke, J.; Jastorff, B. Multidimensional risk analysis of antifouling biocides. Environ. Sci. Poll. Res 2000, 7, 109–110. [Google Scholar]
- Hall, L.W., Jr; Gidding, J.M.; Solomon, K.R.; Balcomb, R. An ecological risk assessment for the use of Irgarol 1051 as an algaecide for antifoulingpaints. Crit. Rev. Toxicol. 1999, 29, 367–437. [Google Scholar]
- Dahl, B.; Blanck, H. Toxic effects of the antifouling agent Irgarol 1051 on periphyton communities in coastal water microcosms. Mar. Poll. Bul 1996, 32, 342–350. [Google Scholar]
- Buma, A.G.J.; Sjollema, S.B.; van de Poll, W.H.; Klamer, H.J.C.; Bakker, J.F. Impact of the antifouling agent Irgarol 1051 on marine phytoplankton species. J. Sea Res 2009, 61, 133–139. [Google Scholar]
- Readman, J.W.; Devilla, R.A.; Tarran, G.; Llewellyn, C.A.; Fileman, T.W.; Easton, A.; Burkill, P.H.; Mantoura, R.F.C. Flow cytometry and pigment analysis as tools to investigate the toxicity of herbicides to natural phytoplankton communities. Mar. Environ. Res 2004, 58, 353–358. [Google Scholar]
- Devilla, R.A.; Brown, M.T.; Donkin, M.; Readman, J.W. The effects of a PSII inhibitor on phytoplankton community structure as assessed by HPLC pigment analyses, microscopy and flow cytometry. Aquat. Toxicol 2005, 71, 25–38. [Google Scholar]
- Zamora-Ley, I.M.; Gardinali, P.R.; Jochem, F.J. Assessing the effects of Irgarol 1051 on marine phytoplankton populations in Key Largo Harbor, Florida. Mar. Poll. Bull 2006, 52, 935–941. [Google Scholar]
- Bragadin, M.; Iero, A.; Cima, F.; Ballarin, L.; Manente, S. TCMS inhibits ATP synthesis in mitochondria: A systematic analysis of the inhibitory mechanism. Toxicol. In Vitro 2007, 21, 1127–1133. [Google Scholar]
- Huh, W.K.; Masuji, Y.; Tada, J.; Arata, J.; Kaniwa, M. Allergic contact dermatitis froma pyridine derivative in polyvinyl chloride leather. Am. J. Contact Dermat 2001, 12, 35–37. [Google Scholar]
- Voulvoulis, N.; Scrimshaw, M.D.; Lester, J.N. Partitioning of selected antifouling in the aquatic environment. Mar. Environ. Res 2001, 53, 1–16. [Google Scholar]
- Karlsson, J.; Eklund, B. New biocide antifouling paints are toxic. Mar. Pollut. Bull 2004, 49, 456–464. [Google Scholar]
- Sabev, H.A.; Handley, P.S.; Robson, G.D. In situ quantification of biocide efficacy using GFP transformed Aureobasidium pullutans. J. Appl. Microbiol 2004, 97, 1132–1139. [Google Scholar]
- Bao, W.W.V.; Leung, K.M.Y.; Kwok, K.W.H.; Zhang, A.Q.; Lui, G.C.S. Synergistic toxic effects of zinc pyrithione and copper to three marine species: Implications on setting appropriate water quality criteria. Mar. Pollut. Bull 2008, 57, 616–623. [Google Scholar]
- Turley, P.A.; Fenn, R.J.; Ritter, J.C. Pyrithiones as antifoulants: Environmental chemistry and preliminary risk assessment. Biofouling 2000, 15, 175–182. [Google Scholar]
- Turley, P.A.; Fenn, R.J.; Ritter, J.C.; Callow, M.E. Pyrithiones as antifoulants: environmental fate and loss of toxicity. Biofouling 2005, 21, 31–40. [Google Scholar]
- Andrioli, N.B.; Soloneski, S.; Larramendy, M.L.; Mudry, M.D. Cytogenetic and microtubule array effects of the zineb-containing commercial fungicide formulation Azzurro® on meristematic root cells of Allium cepa L. Mutat. Res 2011. in press. [Google Scholar]
- U.S. Environmental Protection Agency (EPA). Compendium of Registered Pesticides; US Government Printing Office: Washington, DC, USA, 1974.
- Mochida, K.; Fujii, K. Futher Effects of Alternative Biocides on Aquatic Organisms. In Ecotoxicology of Antifouling Biocides; Arai, T., Harino, H., Ohji, M., Langston, W., Eds.; Springer: Tokyo, Japan, 2009; pp. 383–396. [Google Scholar]
- Xu, Q.; Barrios, C.A.; Cutright, T.; Newby, B.Z. Evaluation of toxicity of capsaicin and zosteric acid and their potential application as antifoulants. Environ. Toxicol 2005, 20, 467–474. [Google Scholar]
- Dahlström, M.; Jonsson, P.R.; Lausmaa, J.; Arnebrant, T.; Sjögren, M.; Holmberg, K.; Martensson, L.G.E.; Elwing, H. Impact of polymer surface affinity of novel antifouling agents. Biotechnol. Bioeng 2004, 86, 1–8. [Google Scholar]
- Katagi, T. Bioconcentration, bioaccumulation, and metabolism of pesticides in aquatic organisms. Rev. Environ. Contam. Toxicol 2010, 204, 1–132. [Google Scholar]
- Kenaga, E. Predicted bioconcentration factors and soil sorption coefficients of pesticides and other chemicals. Ecotoxicol. Environ. Saf 1980, 4, 26–38. [Google Scholar]
- Tóth, S.; Becker-van Slooten, K.; Spack, L.; de Alencastro, L.; Tarradellas, J. Irgarol 1051, an antifouling compound in freshwater, sediment, and biota of Lake Geneva. Bull. Environ. Toxicol. Chem 1996, 57, 426–433. [Google Scholar]
- Dyer, R.A.; Tolhurst, L.E.; Hilton, M.J.; Thomas, K.V. Bioaccumulation of the antifouling paint booster biocide Irgarol 1051 by the green alga Tetraselmis suecica. Bull. Environ. Contam. Toxicol 2006, 77, 1519–1524. [Google Scholar]
- Assessment of the Antibiotic Resistance Effects of Biocides; Scientific Committee on Emerging and Newly Identified Health Risks (SCENIHR), January 2009. Available online: http://ec.europa.eu/health/ph_risk/committees/04_scenihr/docs/scenihr_o_021.pdf accessed on 12 November 2011.


|
Table 1. The main antifouling biocides used in aquaculture and their effect on aquatic organisms. |
| Common Names a | Application a | Mode of Action a | Species | Effects |
|---|---|---|---|---|
| Chorothalonil | Fungicide | Inhibition of mitochondrial electron transport | Crustaceans | |
| Behaviour | ||||
| Cancer magister | Larval mortality [43] | |||
| Penaeus duoramun | Mortality [44] | |||
| Molluscs | ||||
| Crassostrea virginica | Growth [45] | |||
| Embryotoxicity [46] | ||||
| Mytilus edulis | Embryotoxicity [47] | |||
| Mortality [48] | ||||
| Tunicates | ||||
| Embryotoxicity | ||||
| Ciona intestinalis | Inhibition of larval settlement [47] | |||
| Teleosts | ||||
| Anguilla japonica | Mortality [49] | |||
| Cyprinodon variegates | Mortality [45] | |||
| Galaxias auratus | Mortality [50] | |||
| Galaxias maculates | Mortality [50] | |||
| Galaxias truttaceus | Mortality [50] | |||
| Leiostomus xanthurus | Mortality [44] | |||
| Pseudaphritis urvillii | Juvenile mortality [51] | |||
| Copper pyrithione (CuPT) | Microbicide | Multi-site inhibitor (metabolic processes) | Crustaceans | |
| Artemia salina | Inhibition of Na/K ATPase and Mg2+ ATPase enzyme activities [52] | |||
| Molluscs | ||||
| Mytilus galloprovincialis | Alterations in Na/K ATPase and Ca2+ ATPase activities [53] | |||
| Teleosts | ||||
| Fundulus heteroclitus | Alterations in gill osmoregulation [54] | |||
| Oncorhynchus kisutch | Alterations in gill osmoregulation [55] | |||
| Dichlofuanid | Fungicide | Inhibitor of PS II electron transport | Echinoderms | |
| Glyptocidaris crenularis | Embryotoxicity [56] | |||
| DCOIT (Sea-Nine 211®) | Herbicide | Inhibitor of PS II electron transport | Crustaceans | |
| Balanus amphitrite | Larva mortality [57,58] | |||
| Molluscs | ||||
| Crassostrea virginica | Embryo-larva immobility [46] | |||
| Mytilus edulis | Embryo-larva immobility and embryotoxicity [46,47] | |||
| Echinoderms | ||||
| Hemicentrotus pulcherrimus | Embryotoxicity [59] | |||
| Anthocidaris crassispina | Embryotoxicity [59] | |||
| Tunicates | ||||
| Ciona intestinalis | Embryotoxicity and inhibition of larval settlement [47] | |||
| Teleosts | ||||
| Cyprinodon variegatus | Mortality [46] | |||
| Diuron | Herbicide | Inhibitor of PS II electron transport | Algae | |
| Scenedesmus vacuolatus | Toxic for the reproduction [60] | |||
| Microalgae | ||||
| planktonic periphytic | Reduction of chlorophyll a levels [61–63] | |||
| Teleosts | ||||
| Carassius auratus | Acetylcholinesterase inhibition [64] | |||
| Irgarol-1051 | Herbicide | Inhibitor of PS II electron transport | Algae | |
| Dunaliella tertiolecta | Decreasing in growth, inhibition of cell number and decrease in the photosynthetic activity [38,65–70] | |||
| Synechococcus sp | ||||
| Emiliania huxleyi | ||||
| Zostera marina | ||||
| Fucus vesiculosus | ||||
| Enteromorpha intestinalis | ||||
| Ulva intestinalis | ||||
| TCMS pyridine (2,3,3,6-tetrachloro-4- methylsulfonylpyridine) | Fungicide | Inhibitor of mitochondrial electron transport | Tunicates | |
| Botryllus schlosseri | Immunotoxic [71,72] | |||
| Zinc pyrithione (ZnPT) | Microbicide | Multi-site inhibitor (metabolic processes) | Teleosts | |
| Oryzias latipes | Embryotoxicity [73] | |||
| Danio rerio | Embryo-larva [74] | |||
| Zineb | Fungicide | Multi-site inhibitor (metabolic processes) | Teleosts | |
| Salmo gairdneri | Embryotoxicity [75] | |||
aFrom [76].
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Guardiola, F.A.; Cuesta, A.; Meseguer, J.; Esteban, M.A. Risks of Using Antifouling Biocides in Aquaculture. Int. J. Mol. Sci. 2012, 13, 1541-1560. https://doi.org/10.3390/ijms13021541
Guardiola FA, Cuesta A, Meseguer J, Esteban MA. Risks of Using Antifouling Biocides in Aquaculture. International Journal of Molecular Sciences. 2012; 13(2):1541-1560. https://doi.org/10.3390/ijms13021541
Chicago/Turabian StyleGuardiola, Francisco Antonio, Alberto Cuesta, José Meseguer, and Maria Angeles Esteban. 2012. "Risks of Using Antifouling Biocides in Aquaculture" International Journal of Molecular Sciences 13, no. 2: 1541-1560. https://doi.org/10.3390/ijms13021541
APA StyleGuardiola, F. A., Cuesta, A., Meseguer, J., & Esteban, M. A. (2012). Risks of Using Antifouling Biocides in Aquaculture. International Journal of Molecular Sciences, 13(2), 1541-1560. https://doi.org/10.3390/ijms13021541





