In vitro Antioxidant of a Water-Soluble Polysaccharide from Dendrobium fimhriatum Hook.var.oculatum Hook
Abstract
:1. Introduction
2. Results and Discussion
2.1. Extraction of Water-Soluble Crude Polysaccharide
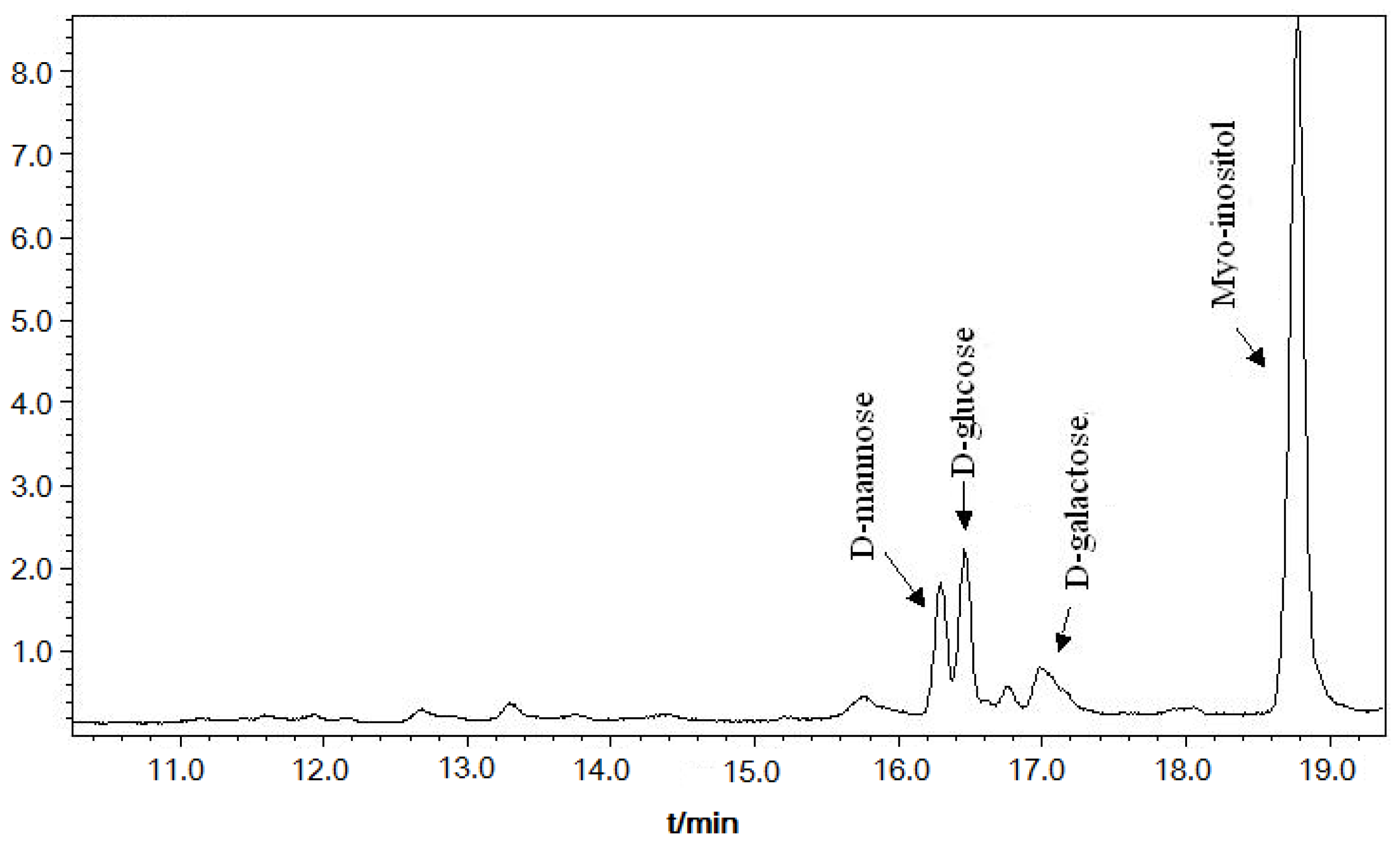
2.2. Monosaccharide Composition of DFHP
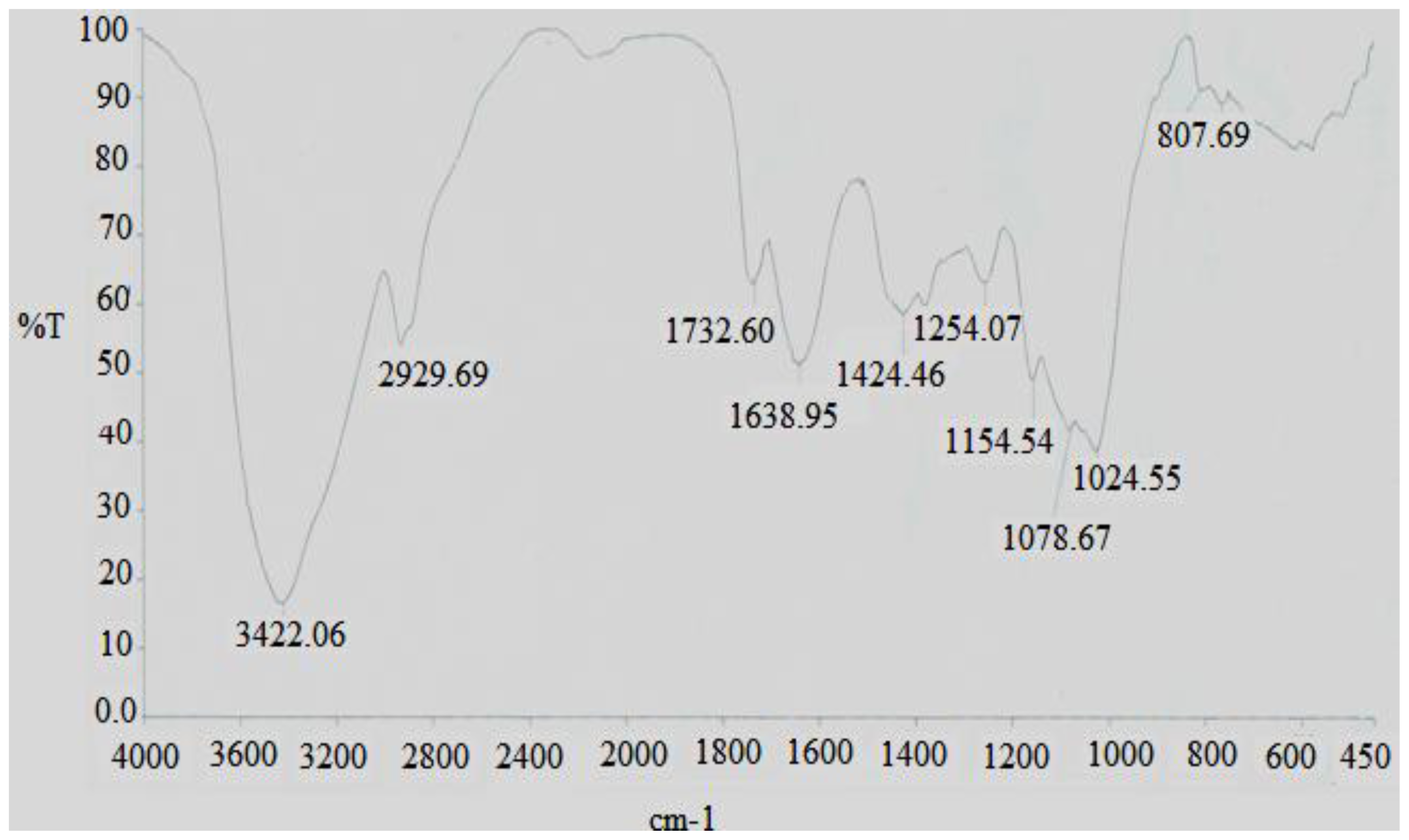
2.3. Infrared spectra of DFHP
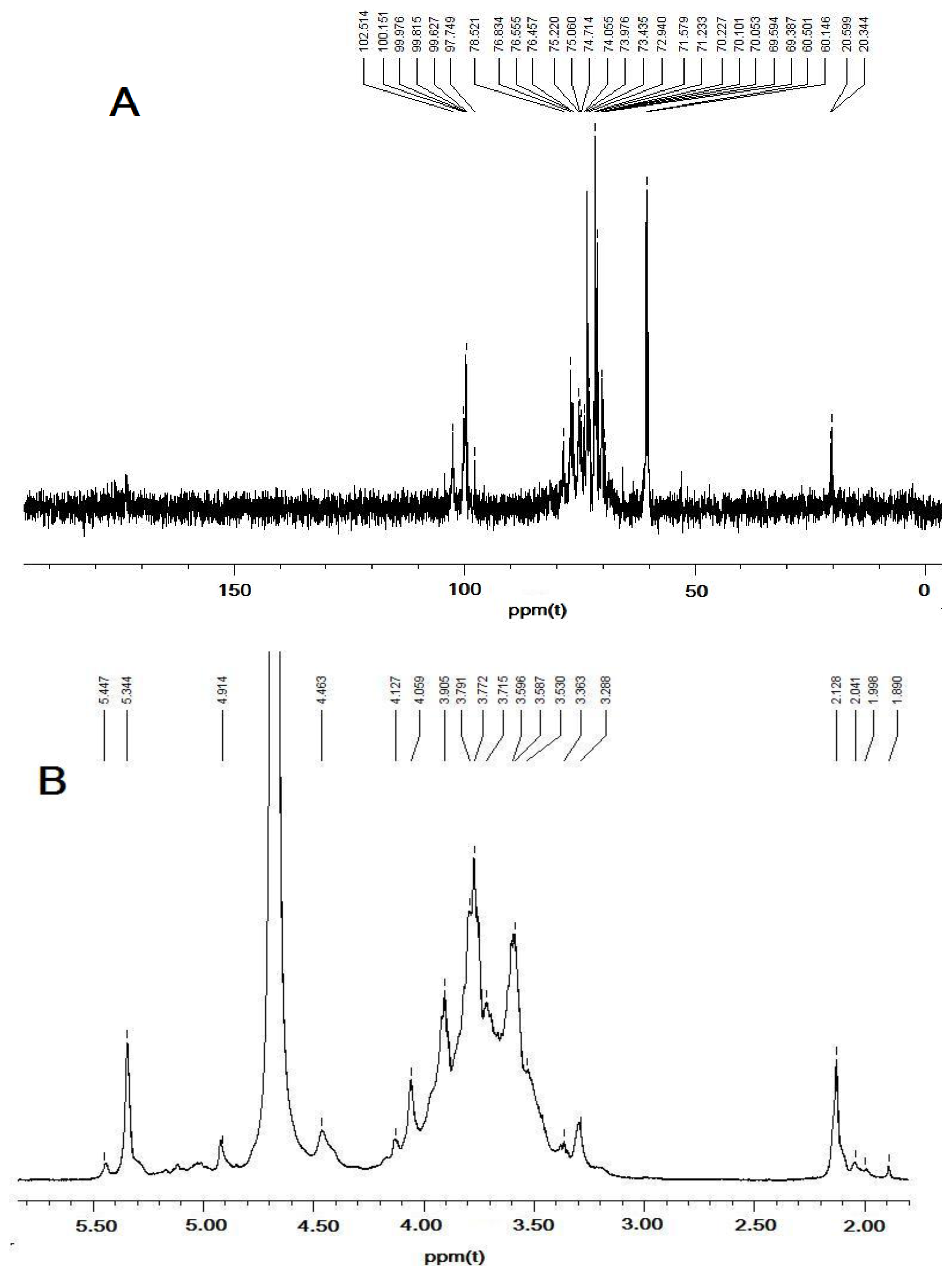
2.4. NMR Identification of DFHP
2.5. Antioxidant Activities Analysis
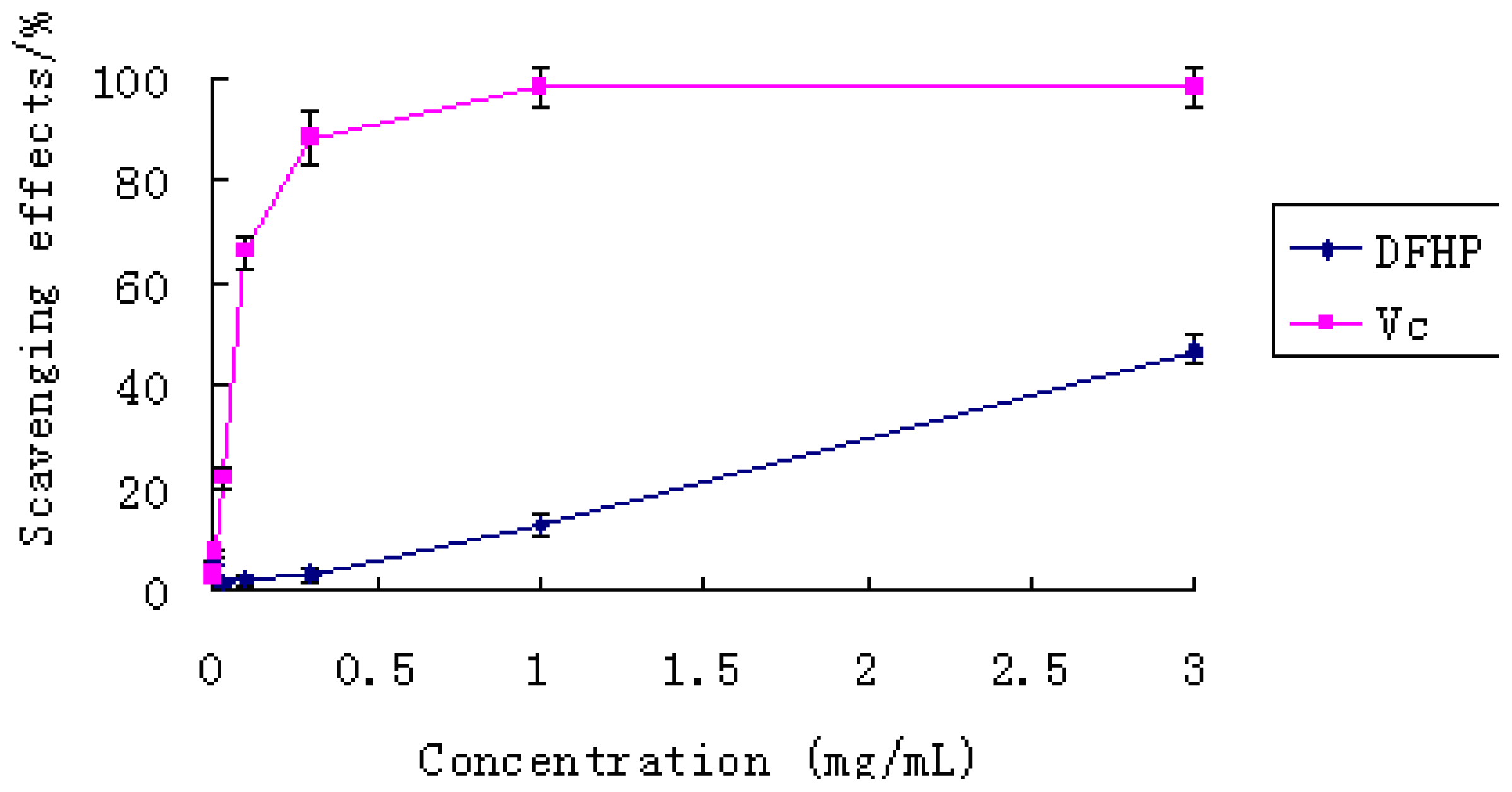
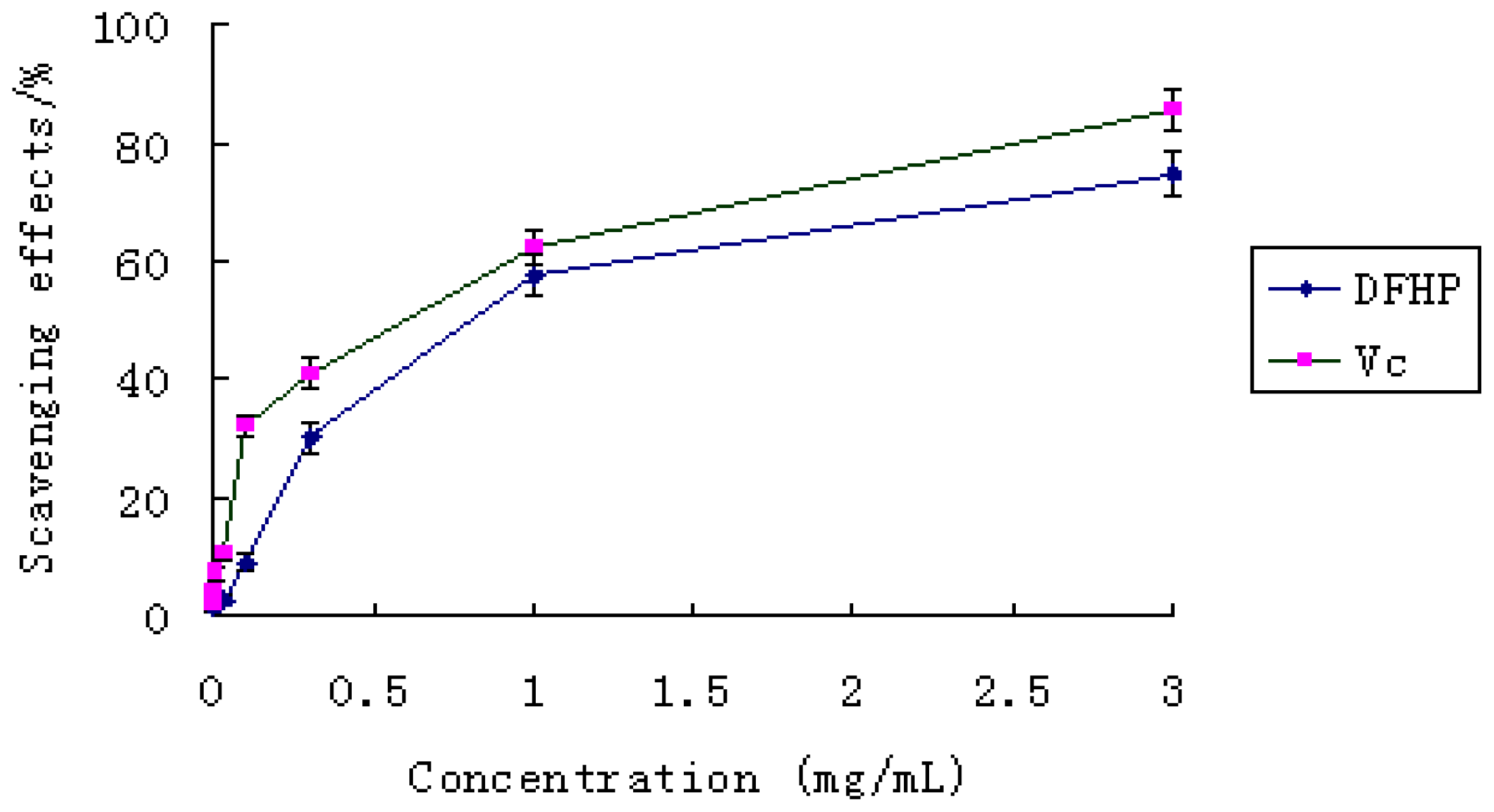
2.5.1. Effect of Scavenging DPPH Radicals
2.5.2. Scavenging Effects of Polysaccharide on Hydroxyl Radicals
2.5.3. Scavenging Effects of Polysaccharide on ABTS
3. Experimental Section
3.1. Materials and Chemicals
3.2. Preparation of the Polysaccharide from Dendrobium fimhriatum Hook.var.Oculatum Hook
3.3. Molecular Weight Determination
3.4. Analysis of Monosaccharide Compositions
3.5. Infrared Spectra of DFHP
3.6. NMR Identification
3.7. Assays for Antioxidant Activities
3.7.1. DPPH Radicals Scavenging Assay
3.7.2. Hydroxyl Radical Scavenging Assay
3.7.3. ABTS Radicals Scavenging Assay
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Majumder, PL; Guha, S; Sen, S. Bibenzyl derivatives from the orchid Dendrobium amoenum. Phytochemistry 1999, 52, 1365–1369. [Google Scholar]
- Yang, L; Qin, LH; Annie Bligh, SW; Bashall, A; Zhang, CF; Zhang, M; Wang, ZT; Xu, LS. A new phenanthrene with a spirolactone from Dendrobium chrysanthum and its anti-inflammatory activities. Bioorg. Med. Chem 2006, 14, 3496–3501. [Google Scholar]
- Wang, Q; Gong, Q; Wu, Q; Shi, J. Neuroprotective effects of Dendrobium alkaloids on rat cortical neurons injured by oxygen-glucose deprivation and reperfusion. Phytomedicin 2010, 17, 108–115. [Google Scholar]
- Fan, YJ; Luo, AX; Luo, AS; Chun, Z. Research on the ultrasonic-assisted extraction process for polysaccharide from D.denneanum. Chin J Hosp Pharm 2009, 22, 1909–1911. [Google Scholar]
- Fan, YJ; Luo, AX. Evaluation of anti-tumor activity of water-soluble polysaccharides from Dendrobium denneanum. Afr. J. Pharm. Pharmaco 2011, 5, 415–420. [Google Scholar]
- Zha, XQ; Luo, JP; Luo, SZ; Jiang, ST. Structure identification of a new immunostimulating polysaccharide from the stems of Dendrobium huoshanense. Carbohyd. Polym 2007, 69, 86–93. [Google Scholar]
- Wang, JH; Luo, JP; Zha, XQ; Feng, BJ. Comparison of antitumor activities of different polysaccharide fractions from the stems of Dendrobium nobile Lindl. Carbohyd. Polym 2010, 79, 114–118. [Google Scholar]
- Luo, AX; Ge, ZF; Fan, YJ; Luo, AS; Chun, Z; He, XJ. In vitro and in vivo antioxidant activity of a water-soluble polysaccharide from dendrobium denneanum. Molecules 2011, 16, 1579–1592. [Google Scholar]
- Zhao, YP; Son, YO; Kim, SS; Jang, YS; Lee, JC. Antioxidant and anti-hyperglycemic activity of polysaccharide isolated from dendrobium chrysotoxum lindl. J. Biochem. Mol. Biol 2007, 40, 670–677. [Google Scholar]
- Fan, YJ; He, XJ; Zhou, SD; Luo, AX; He, T; Chun, Z. Composition analysis and antioxidant activity of polysaccharide from Dendrobium denneanum. Int. J. Biol. Macromol 2009, 45, 169–173. [Google Scholar]
- Santhiya, D; Subramanian, S; Natarajan, KA. Surface chemical studies on sphalerite and galena using extracellular polysaccharides isolated from bacillus polymyxa. J. Colloid Interf. Sci 2002, 256, 237–248. [Google Scholar]
- Park, FS. Application of IR Spectroscopy in Biochemistry, Biology, and Medicine; Plenum Press: New York, NY, USA, 1971; pp. 100–140. [Google Scholar]
- Zhao, MM; Yang, N; Yang, B. Structural characterization of water-soluble olysaccharides from Opuntia monacanthap cladodes in relation to their anti-glycated activities. Food Chem 2007, 105, 1480–1486. [Google Scholar]
- Barker, SA; Bourne, EJ; Stacey, M; Whiffen, DH. Infrared spectra of carbohydrates. Part I. Some derivatives of D-glucopyranose. J. Chem. Soc 1954, 99, 171–176. [Google Scholar]
- Maiti, D; Chandra, K; Mondal, S; Ojha, AK; Das, D; Roy, SK; Ghosh, K; Chakraborty, I; Islam, SS. Isolation and characterization of a heteroglycan from the fruits of Astraeus hygrometricus. Carbohyd. Res 2008, 343, 817–824. [Google Scholar]
- Roy, SK; Maiti, D; Mondal, S; Das, D; Islam, SS. Structural analysis of a polysaccharide isolated from the aqueous extract of an edible mushroom, Pleurotus sajor-caju, cultivar Black Japan. Carbohyd. Res 2008, 343, 1108–1113. [Google Scholar]
- Cao, W; Li, XQ; Liu, L; Yang, TH; Li, C; Fan, HT; Jia, M; Lv, ZG; Mei, QB. Structure of an anti-tumor polysaccharide from Angelica sinensis (Oliv.) Diels. Carbohyd. Polym 2006, 66, 149–159. [Google Scholar]
- He, YM; Liu, CH; Chen, YX; Ji, AC; Shen, ZL; Xi, T; Yao, QS. Isolation and structural characterization of a novel polysaccharide prepared from Arca subcrenata Lischke. J. Biosci. Bioeng 2007, 104, 111–116. [Google Scholar]
- Yoon, S; Kim, MK; Lee, IY; Yun, M; Nam Shin, JE. Production and structural features of a water-soluble polysaccharide from a mutant strain of Agrobacterium sp. J. Ind. Eng. Chem 2008, 14, 759–764. [Google Scholar]
- Kath, F; Kulicke, WM. Mild enzymatic isolation of mannan and glucan from yeast Saccharomyces cerevisiae. Angew. Makromol. Chem 1999, 268, 59–68. [Google Scholar]
- Han, J; Weng, XC; Bi, KS. Antioxidants from a Chinese medicinal herb-Litho-spermum erythrorhizon. Food Chem 2008, 106, 2–10. [Google Scholar]
- Luo, AX; He, XJ; Zhou, SD; Fan, YJ; He, T; Chun, Z. In vitro antioxidant activities of a water-soluble polysaccharide derived from Dendrobium nobile Lindl. Extracts. Int. J. Biol. Macromol 2009, 45, 359–363. [Google Scholar]
- Navarini, L; Gilli, R; Gombac, V; Abatangelo, A; Bosco, M; Toffanin, R. Polysaccharides from hot water extracts of roasted Coffea arabica beans: Isolation characterization. Carbohyd. Polym 1999, 40, 71–81. [Google Scholar]
- Yamamoto, Y; Nunome, T; Yamauchi, R; Kato, K; Sone, Y. Structure of an exocellular polysaccharide of Lactobacillus helveticus TN-4, a spontaneous mutant strain of Lactobacillus helveticus TY1-2. Carbohyd. Res 1995, 275, 319–332. [Google Scholar]
- Pang, XB; Yao, WB; Yang, XB; Xie, C; Liu, D; Zhang, J; Gao, XD. Purification, characterization and biological activity on hepatocytes of a polysaccharide from Flammulina velutipes mycelium. Carbohyd. Res 2007, 70, 291–297. [Google Scholar]
- Luo, AX; He, XJ; Zhou, SD; Fan, YJ; Luo, AS; Chun, Z. Purification, composition analysis and antioxidant activity of the polysaccharides from Dendrobium nobile Lindl. Carbohyd. Polym 2010, 79, 1014–1019. [Google Scholar]
- Shimada, K; Fujikawa, K; Yahara, K; Nakamura, T. Antioxidative properties of xanthan on the autoxidation of soybean oil in cyclodextrin emulsion. J. Agr. Food Chem 1992, 40, 945–948. [Google Scholar]
- Wei, SD; Zhou, HC; Lin, YM. Antioxidant activities of extract and fractions from the hypocotyls of the Mangrove Plant Kandelia candel. Int. J. Mol. Sci 2010, 11, 4080–4093. [Google Scholar]
- Wang, J; Zhang, QB; Zhang, ZS; Li, Z. Antioxidant activity of sulfated polysaccharide fractions extracted from Laminaria japonica. Int. J. Biol. Macromol 2008, 42, 127–132. [Google Scholar]
- Re, R; Pellegrini, N; Proteggente, A; Pannala, A; Yang, M; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Rad. Bio. Med 1999, 26, 1231–1237. [Google Scholar]
- Luo, AX; Fan, YJ; Luo, AS. In vitro free radicals scavenging activities of polysaccharide from polygonum multiflorum thunb. J. Med. Plants Res 2011, 5, 966–972. [Google Scholar]

© 2011 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Luo, A.; Fan, Y. In vitro Antioxidant of a Water-Soluble Polysaccharide from Dendrobium fimhriatum Hook.var.oculatum Hook. Int. J. Mol. Sci. 2011, 12, 4068-4079. https://doi.org/10.3390/ijms12064068
Luo A, Fan Y. In vitro Antioxidant of a Water-Soluble Polysaccharide from Dendrobium fimhriatum Hook.var.oculatum Hook. International Journal of Molecular Sciences. 2011; 12(6):4068-4079. https://doi.org/10.3390/ijms12064068
Chicago/Turabian StyleLuo, Aoxue, and Yijun Fan. 2011. "In vitro Antioxidant of a Water-Soluble Polysaccharide from Dendrobium fimhriatum Hook.var.oculatum Hook" International Journal of Molecular Sciences 12, no. 6: 4068-4079. https://doi.org/10.3390/ijms12064068
APA StyleLuo, A., & Fan, Y. (2011). In vitro Antioxidant of a Water-Soluble Polysaccharide from Dendrobium fimhriatum Hook.var.oculatum Hook. International Journal of Molecular Sciences, 12(6), 4068-4079. https://doi.org/10.3390/ijms12064068



