Inorganic-Organic Hybrid Nanomaterials for Therapeutic and Diagnostic Imaging Applications
Abstract
:1. Introduction
2. Solid Silica Nanoparticles (SNPs)
2.1. Synthesis
2.2. Therapeutic Applications
2.3. Diagnostic Imaging Applications
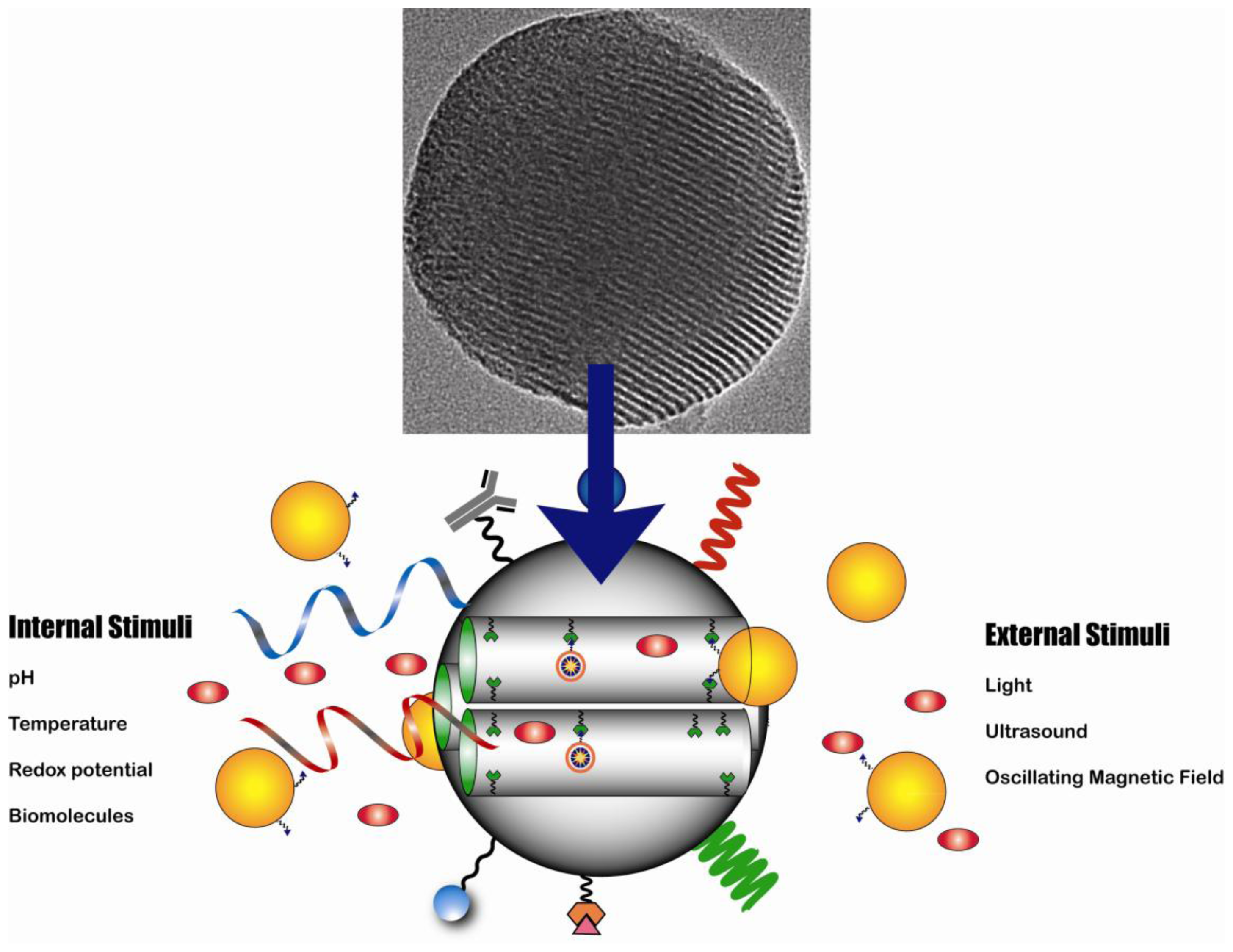
3. Mesoporous Silica Nanoparticles (MSNs)
3.1. Synthesis
3.2. Therapeutic Applications
3.3. Diagnostic Imaging Applications
3.4. Theranostic Applications
4. Gold Nanoparticles (AuNPs)
4.1. Synthesis
4.2. Therapeutic Applications
4.3. Diagnostic Imaging Applications
5. Iron Oxide Nanoparticles (IONPs)
5.1. Synthesis of Magnetite (Fe3O4) Nanoparticles
5.2. Therapeutic Applications
5.3. Diagnostic Imaging Applications
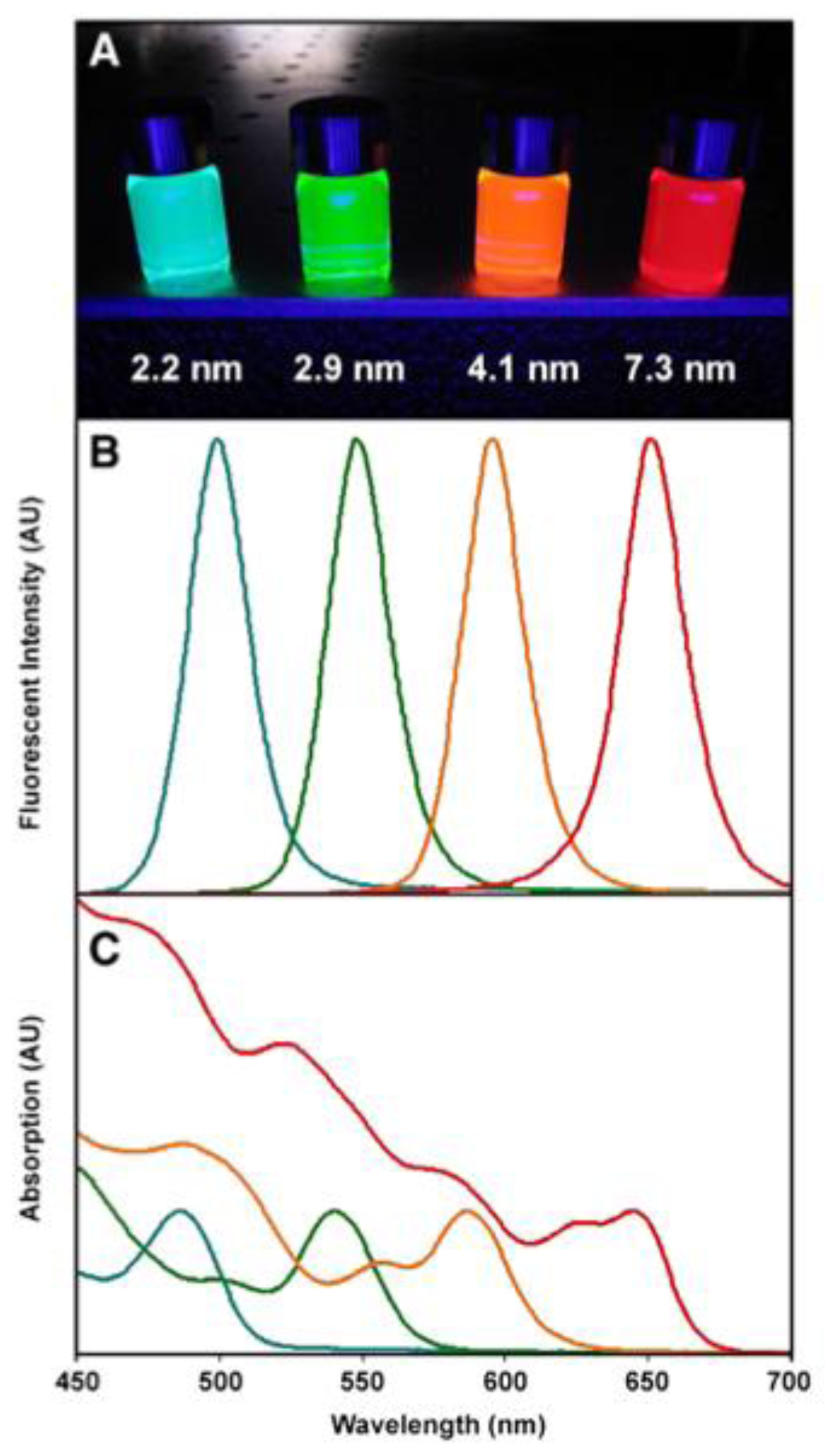
6. Quantum Dots (QDs)
6.1. Synthesis
6.2. Therapeutic Applications
6.3. Diagnostic Imaging Applications
7. Conclusions
Acknowledgments
References
- Smith, AM; Duan, HW; Mohs, AM; Nie, SM. Bioconjugated quantum dots for in vivo molecular and cellular imaging. Adv. Drug Deliv. Rev 2008, 60, 1226–1240. [Google Scholar]
- Ghosh, P; Han, G; De, M; Kim, CK; Rotello, VM. Gold nanoparticles in delivery applications. Adv. Drug Deliv. Rev 2008, 60, 1307–1315. [Google Scholar]
- Cobley, CM; Au, L; Chen, J-Y; Xia, Y-N. Targeting gold nanocages to cancer cells for photothermal destruction and drug delivery. Expet. Opin. Drug Deliv 2010, 7, 577–587. [Google Scholar]
- Jain, PK; Huang, X; El-Sayed, IH; El-Sayed, MA. Noble metals on the nanoscale: Optical and photothermal properties and some applications in imaging, sensing, biology, and medicine. Acc. Chem. Res 2008, 41, 1578–1586. [Google Scholar]
- Sun, C; Lee, JSH; Zhang, M. Magnetic nanoparticles in MR imaging and drug delivery. Adv. Drug Deliv. Rev 2008, 60, 1252–1265. [Google Scholar]
- Lin, W; Hyeon, T; Lanza, GM; Zhang, M; Meade, TJ. Magnetic nanoparticles for early detection of cancer by magnetic resonance imaging. MRS Bull 2009, 34, 441–448. [Google Scholar]
- Fendler, JH. Feldheim, DL, Foss, CA, Eds.; Metal Nanoparticles Synthesis, Characterization, and Applications; CRC Press: Boca Raton, FL, USA, 2002; Volume 14. [Google Scholar]
- Burns, A; Ow, H; Wiesner, U. Fluorescent core-shell silica nanoparticles: Towards “Lab on a Particle” architectures for nanobiotechnology. Chem. Soc. Rev 2006, 35, 1028–1042. [Google Scholar]
- Slowing, II; Vivero-Escoto, JL; Wu, C-W; Lin, VSY. Mesoporous silica nanoparticles as controlled release drug delivery and gene transfection carriers. Adv. Drug Deliv. Rev 2008, 60, 1278–1288. [Google Scholar]
- Trewyn, BG; Slowing, II; Giri, S; Chen, H-T; Lin, VSY. Synthesis and functionalization of a mesoporous silica nanoparticle based on the sol-gel process and applications in controlled release. Acc. Chem. Res 2007, 40, 846–853. [Google Scholar]
- Stoeber, W; Fink, A; Bohn, E. Controlled growth of monodisperse silica spheres in the micron size range. J. Colloid Interface Sci 1968, 26, 62–69. [Google Scholar]
- Wang, X-D; Shen, Z-X; Sang, T; Cheng, X-B; Li, M-F; Chen, L-Y; Wang, Z-S. Preparation of spherical silica particles by Stoeber process with high concentration of tetra-ethylorthosilicate. J. Colloid Interface Sci 2010, 341, 23–29. [Google Scholar]
- Arriagada, FJ; Osseo-Asare, K. Phase and dispersion stability effects in the synthesis of silica nanoparticles in a non-ionic reverse microemulsion. Colloid. Surface 1992, 69, 105–115. [Google Scholar]
- Arriagada, FJ; Osseo-Asare, K. Synthesis of nanometer-sized silica by controlled hydrolysis in reverse micellar systems. Adv. Chem 1994, 234, 113–128. [Google Scholar]
- Arriagada, FJ; Osseo-Asare, K. Synthesis of nanosize silica in a nonionic water-in-oil microemulsion: Effects of the water/surfactant molar ratio and ammonia concentration. J. Colloid Interface Sci 1999, 211, 210–220. [Google Scholar]
- Bagwe, RP; Yang, C; Hilliard, LR; Tan, W. Optimization of dye-doped silica nanoparticles prepared using a reverse microemulsion method. Langmuir 2004, 20, 8336–8342. [Google Scholar]
- Wang, L; Zhao, W; Tan, W. Bioconjugated silica nanoparticles: Development and applications. Nano Res 2008, 1, 99–115. [Google Scholar]
- Jin, Y; Li, A; Hazelton, SG; Liang, S; John, CL; Selid, PD; Pierce, DT; Zhao, JX. Amorphous silica nanohybrids: Synthesis, properties and applications. Coord. Chem. Rev 2009, 253, 2998–3014. [Google Scholar]
- Guerrero-Martinez, A; Perez-Juste, J; Liz-Marzan, LM. Recent progress on silica coating of nanoparticles and related nanomaterials. Adv. Mater 2010, 22, 1182–1195. [Google Scholar]
- Taylor-Pashow, KML; Della Rocca, J; Huxford, RC; Lin, W. Hybrid nanomaterials for biomedical applications. Chem. Commun 2010, 46, 5832–5849. [Google Scholar]
- Kim, S; Ohulchanskyy, TY; Pudavar, HE; Pandey, RK; Prasad, PN. Organically modified silica nanoparticles co-encapsulating photosensitizing drug and aggregation-enhanced two-photon absorbing fluorescent dye aggregates for two-photon photodynamic therapy. J. Am. Chem. Soc 2007, 129, 2669–2675. [Google Scholar]
- He, X; Wu, X; Wang, K; Shi, B; Hai, L. Methylene blue-encapsulated phosphonate-terminated silica nanoparticles for simultaneous in vivo imaging and photodynamic therapy. Biomaterials 2009, 30, 5601–5609. [Google Scholar]
- Mintzer, MA; Simanek, EE. Nonviral vectors for gene delivery. Chem. Rev 2009, 109, 259–302. [Google Scholar]
- Lee, KG; Kim, JC; Wi, R; Min, JS; Ahn, JK; Kim, DH. Synthesis of stable silica-dye hybrid nanomaterial as DNA carrier. J. Nanosci. Nanotech 2011, 11, 686–690. [Google Scholar]
- Ravi Kumar, MNV; Sameti, M; Mohapatra, SS; Kong, X; Lockey, RF; Bakowsky, U; Lindenblatt, G; Schmidt, H; Lehr, CM. Cationic silica nanoparticles as gene carriers: Synthesis, characterization and transfection efficiency in vitro and in vivo. J. Nanosci. Nanotech 2004, 4, 876–881. [Google Scholar]
- Bharali, DJ; Klejbor, I; Stachowiak, EK; Dutta, P; Roy, I; Kaur, N; Bergey, EJ; Prasad, PN; Stachowiak, MK. Organically modified silica nanoparticles: A nonviral vector for in vivo gene delivery and expression in the brain. Proc. Natl. Acad. Sci. USA 2005, 102, 11539–11544. [Google Scholar]
- Zhao, X; Hilliard, LR; Wang, K; Tan, W. Bioconjugated silica nanoparticles for bioanalysis. Encycloped. Nanosci. Nanotech 2004, 1, 255–268. [Google Scholar]
- Smith, JE; Medley, CD; Tang, Z; Shangguan, D; Lofton, C; Tan, W. Aptamer-conjugated nanoparticles for the collection and detection of multiple cancer cells. Anal. Chem 2007, 79, 3075–3082. [Google Scholar]
- Zhao, X; Tapec-Dytioco, R; Tan, W. Ultrasensitive DNA detection using highly fluorescent bioconjugated nanoparticles. J. Am. Chem. Soc 2003, 125, 11474–11475. [Google Scholar]
- Zhou, X; Zhou, J. Improving the signal sensitivity and photostability of DNA hybridizations on microarrays by using dye-doped core-shell silica nanoparticles. Anal. Chem 2004, 76, 5302–5312. [Google Scholar]
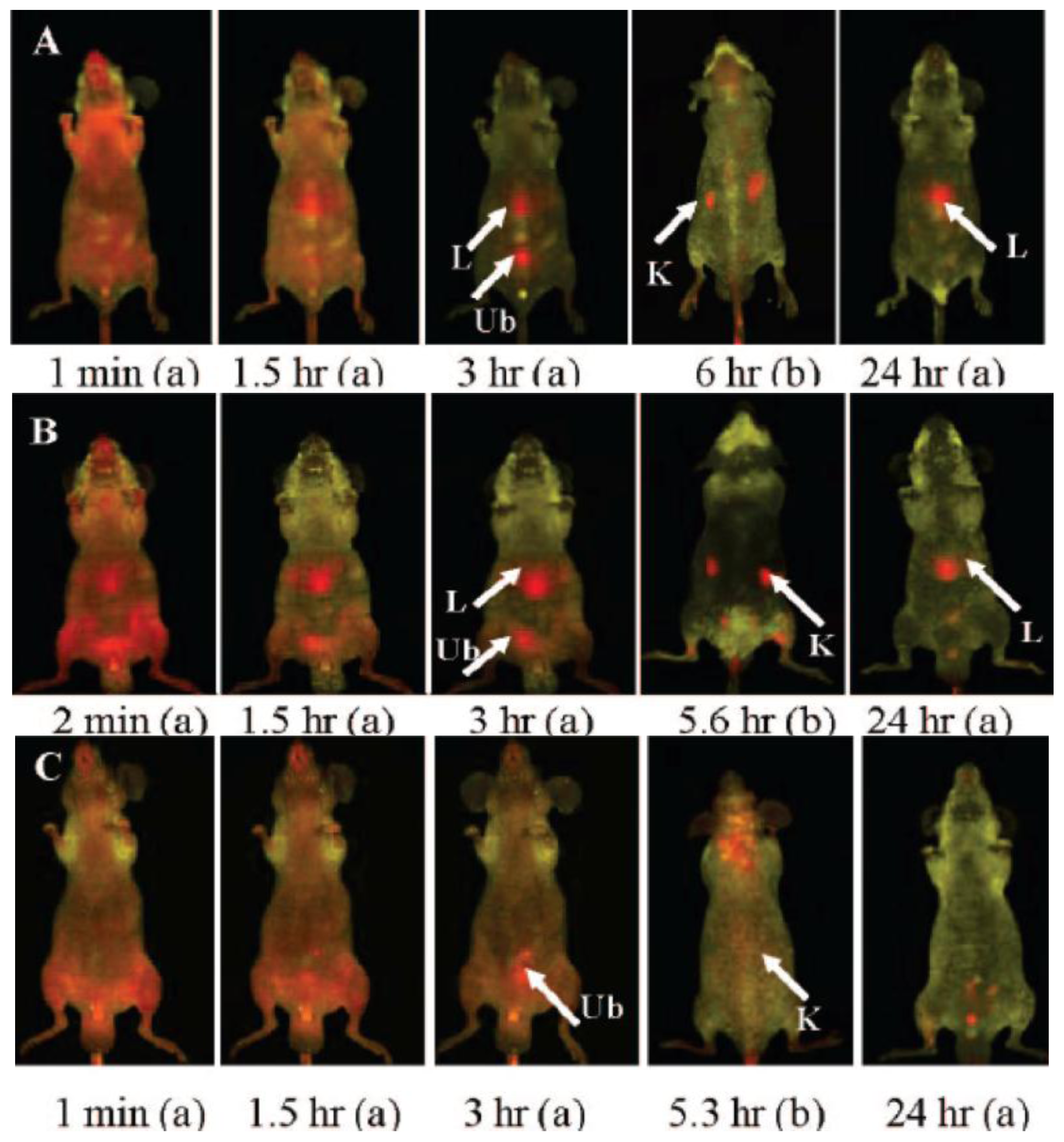
- He, X; Nie, H; Wang, K; Tan, W; Wu, X; Zhang, P. In vivo study of biodistribution and urinary excretion of surface-modified silica nanoparticles. Anal. Chem 2008, 80, 9597–9603. [Google Scholar]
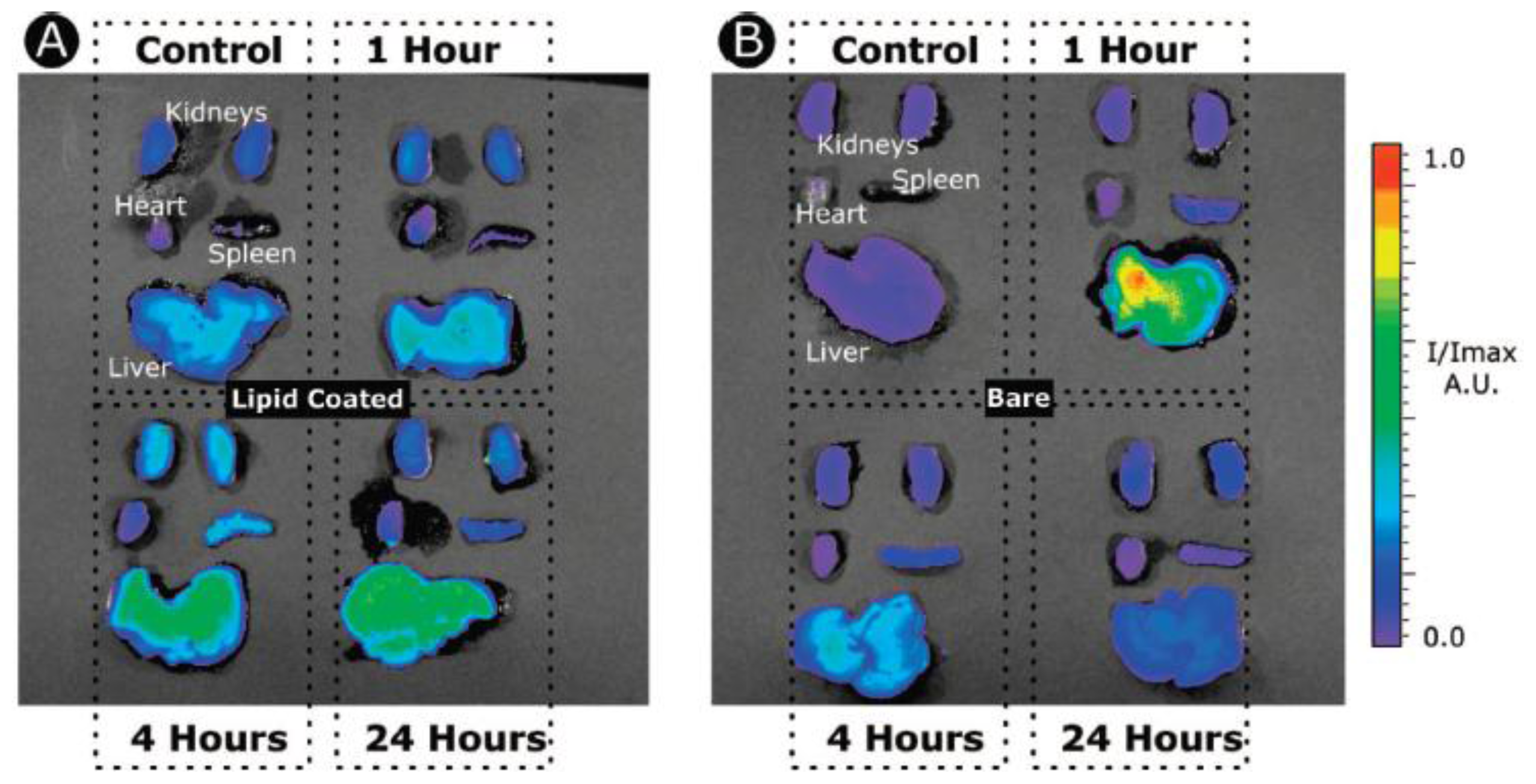
- Van Schooneveld, MM; Vucic, E; Koole, R; Zhou, Y; Stocks, J; Cormode, DP; Tang, CY; Gordon, RE; Nicolay, K; Meijerink, A; et al. Improved biocompatibility and pharmacokinetics of silica nanoparticles by means of a lipid coating: A multimodality investigation. Nano Lett 2008, 8, 2517–2525. [Google Scholar]
- Cho, M; Cho, W-S; Choi, M; Kim, SJ; Han, BS; Kim, SH; Kim, HO; Sheen, YY; Jeong, J. The impact of size on tissue distribution and elimination by single intravenous injection of silica nanoparticles. Toxicol. Lett 2009, 189, 177–183. [Google Scholar]
- Kumar, R; Roy, I; Ohulchanskky, TY; Vathy, LA; Bergey, EJ; Sajjad, M; Prasad, PN. In vivo biodistribution and clearance studies using multimodal organically modified silica nanoparticles. ACS Nano 2010, 4, 699–708. [Google Scholar]
- Vivero-Escoto, JL; Trewyn, BG; Lin, VSY. Mesoporous silica nanoparticles: Synthesis and applications. Annu. Rev. Nano Res 2010, 3, 191–231. [Google Scholar]
- Coti, KK; Belowich, ME; Liong, M; Ambrogio, MW; Lau, YA; Khatib, HA; Zink, JI; Khashab, NM; Stoddart, JF. Mechanised nanoparticles for drug delivery. Nanoscale 2009, 1, 16–39. [Google Scholar]
- Hom, C; Lu, J; Tamanoi, F. Silica nanoparticles as a delivery system for nucleic acid-based reagents. J. Mater. Chem 2009, 19, 6308–6316. [Google Scholar]
- Slowing, II; Vivero-Escoto, JL; Trewyn, BG; Lin, VSY. Mesoporous silica nanoparticles: Structural design and applications. J. Mater. Chem 2010, 20, 7924–7937. [Google Scholar]
- Vivero-Escoto, JL; Slowing, II; Lin, VSY. Tuning the cellular uptake and cytotoxicity properties of oligonucleotide intercalator-functionalized mesoporous silica nanoparticles with human cervical cancer cells HeLa. Biomaterials 2010, 31, 1325–1333. [Google Scholar]
- Vivero-Escoto, JL; Slowing, II; Trewyn, BG; Lin, VSY. Mesoporous silica nanoparticles for intracellular controlled drug delivery. Small 2010, 6, 1952–1967. [Google Scholar]
- Chen, H-T; Huh, S; Lin, VS-Y. Regalbuto, J, Ed.; Fine-tuning the functionalization of mesoporous silica. In Catalyst Preparation; CRC Press: Boca Raton, FL, USA, 2007; pp. 45–73. [Google Scholar]
- Descalzo, AB; Martinez-Manez, R; Sancenon, F; Hoffmann, K; Rurack, K. The supramolecular chemistry of organic-inorganic hybrid materials. Angew. Chem. Int. Ed 2006, 45, 5924–5948. [Google Scholar]
- Huh, S; Wiench, JW; Trewyn, BG; Song, S; Pruski, M; Lin, VSY. Tuning of particle morphology and pore properties in mesoporous silicas with multiple organic functional groups. Chem. Commun 2003, 18, 2364–2365. [Google Scholar]
- Huh, S; Wiench, JW; Yoo, J-C; Pruski, M; Lin, VSY. Organic functionalization and morphology control of mesoporous silicas via a Co-condensation synthesis method. Chem. Mater 2003, 15, 4247–4256. [Google Scholar]
- Huang, Y; Xu, S; Lin, VSY. Bifunctionalized mesoporous materials with site-separated brønsted acids and bases: Catalyst for a two-step reaction sequence. Angew. Chem. Int. Ed 2011, 50, 661–664. [Google Scholar]
- Vallet-Regi, M; Ramila, A; del Real, RP; Perez-Pariente, J. A new property of MCM-41: Drug delivery system. Chem. Mater 2001, 13, 308–311. [Google Scholar]
- Lai, C-Y; Trewyn, BG; Jeftinija, DM; Jeftinija, K; Xu, S; Jeftinija, S; Lin, VSY. A mesoporous silica nanosphere-based carrier system with chemically removable CdS nanoparticle caps for stimuli-responsive controlled release of neurotransmitters and drug molecules. J. Am. Chem. Soc 2003, 125, 4451–4459. [Google Scholar]
- Liu, R; Zhang, Y; Zhao, X; Agarwal, A; Mueller, LJ; Feng, P. pH-responsive nanogated ensemble based on gold-capped mesoporous silica through an acid-labile acetal linker. J. Am. Chem. Soc 2010, 132, 1500–1501. [Google Scholar]
- Torney, F; Trewyn, BG; Lin, VSY; Wang, K. Mesoporous silica nanoparticles deliver DNA and chemicals into plants. Nat. Nanotech 2007, 2, 295–300. [Google Scholar]
- Vivero-Escoto, JL; Slowing, II; Wu, C-W; Lin, VSY. Photoinduced intracellular controlled release drug delivery in human cells by gold-capped mesoporous silica nanosphere. J. Am. Chem. Soc 2009, 131, 3462–3463. [Google Scholar]
- Giri, S; Trewyn, BG; Stellmaker, MP; Lin, VSY. Stimuli-responsive controlled-release delivery system based on mesoporous silica nanorods capped with magnetic nanoparticles. Angew. Chem. Int. Ed 2005, 44, 5038–5044. [Google Scholar]
- Lee, JE; Lee, N; Kim, H; Kim, J; Choi, SH; Kim, JH; Kim, T; Song, IC; Park, SP; Moon, WK; et al. Uniform mesoporous dye-doped silica nanoparticles decorated with multiple magnetite nanocrystals for simultaneous enhanced magnetic resonance imaging, fluorescence imaging, and drug delivery. J. Am. Chem. Soc 2010, 132, 552–557. [Google Scholar]
- Radu, DR; Lai, C-Y; Jeftinija, K; Rowe, EW; Jeftinija, S; Lin, VSY. A polyamidoamine dendrimer-capped mesoporous silica nanosphere-based gene transfection reagent. J. Am. Chem. Soc 2004, 126, 13216–13217. [Google Scholar]
- Zhao, Y; Trewyn, BG; Slowing, II; Lin, VSY. Mesoporous silica nanoparticle-based double drug delivery system for glucose-responsive controlled release of insulin and cyclic AMP. J. Am. Chem. Soc 2009, 131, 8398–8400. [Google Scholar]
- Liu, R; Zhang, Y; Feng, P. Multiresponsive Supramolecular nanogated ensembles. J. Am. Chem. Soc 2009, 131, 15128–15129. [Google Scholar]
- Liu, R; Zhang, Y; Wu, T; Feng, P. Tunable redox-responsive hybrid nanogated ensembles. J. Am. Chem. Soc 2008, 130, 14418–14419. [Google Scholar]
- Radu, DR; Lai, C-Y; Wiench, JW; Pruski, M; Lin, VSY. Gatekeeping layer effect: A poly(lactic acid)-coated mesoporous silica nanosphere-based fluorescence probe for detection of amino-containing neurotransmitters. J. Am. Chem. Soc 2004, 126, 1640–1641. [Google Scholar]
- Ambrogio, MW; Pecorelli, TA; Patel, K; Khashab, NM; Trabolsi, A; Khatib, HA; Botros, YY; Zink, JI; Stoddart, JF. Snap-top nanocarriers. Org. Lett 2010, 12, 3304–3307. [Google Scholar]
- Zhao, Y; Vivero-Escoto, JL; Slowing, II; Trewyn, BG; Lin, VSY. Capped mesoporous silica nanoparticles as stimuli-responsive controlled release systems for intracellular drug/gene delivery. Expet. Opin. Drug Deliv 2010, 7, 1013–1029. [Google Scholar]
- Lu, J; Liong, M; Li, Z; Zink, JI; Tamanoi, F. Biocompatibility, biodistribution, and drug-delivery efficiency of mesoporous silica nanoparticles for cancer therapy in animals. Small 2010, 6, 1794–1805. [Google Scholar]
- Cheon, J; Lee, J-H. Synergistically integrated nanoparticles as multimodal probes for nanobiotechnology. Acc. Chem. Res 2008, 41, 1630–1640. [Google Scholar]
- Weissleder, R. Molecular imaging in cancer. Science 2006, 312, 1168–1171. [Google Scholar]
- Lee, C-H; Cheng, S-H; Wang, Y-J; Chen, Y-C; Chen, N-T; Souris, J; Chen, C-T; Mou, C-Y; Yang, C-S; Lo, L-W. Near-infrared mesoporous silica nanoparticles for optical imaging: Characterization and in vivo biodistribution. Adv. Funct. Mater 2009, 19, 215–222. [Google Scholar]
- Souris, JS; Lee, C-H; Cheng, S-H; Chen, C-T; Yang, C-S; Ho, J-A; Mou, C-Y; Lo, L-W. Surface charge-mediated rapid hepatobiliary excretion of mesoporous silica nanoparticles. Biomaterials 2010, 31, 5564–5574. [Google Scholar]
- Na, HB; Song, IC; Hyeon, T. Inorganic nanoparticles for MRI contrast agents. Adv. Mater 2009, 21, 2133–2148. [Google Scholar]
- Villaraza, AJL; Bumb, A; Brechbiel, MW. Macromolecules, dendrimers, and nanomaterials in magnetic resonance imaging: The interplay between size, function, and pharmacokinetics. Chem. Rev 2010, 110, 2921–2959. [Google Scholar]
- Na, HB; Hyeon, T. Nanostructured T1 MRI contrast agents. J. Mater. Chem 2009, 19, 6267–6273. [Google Scholar]
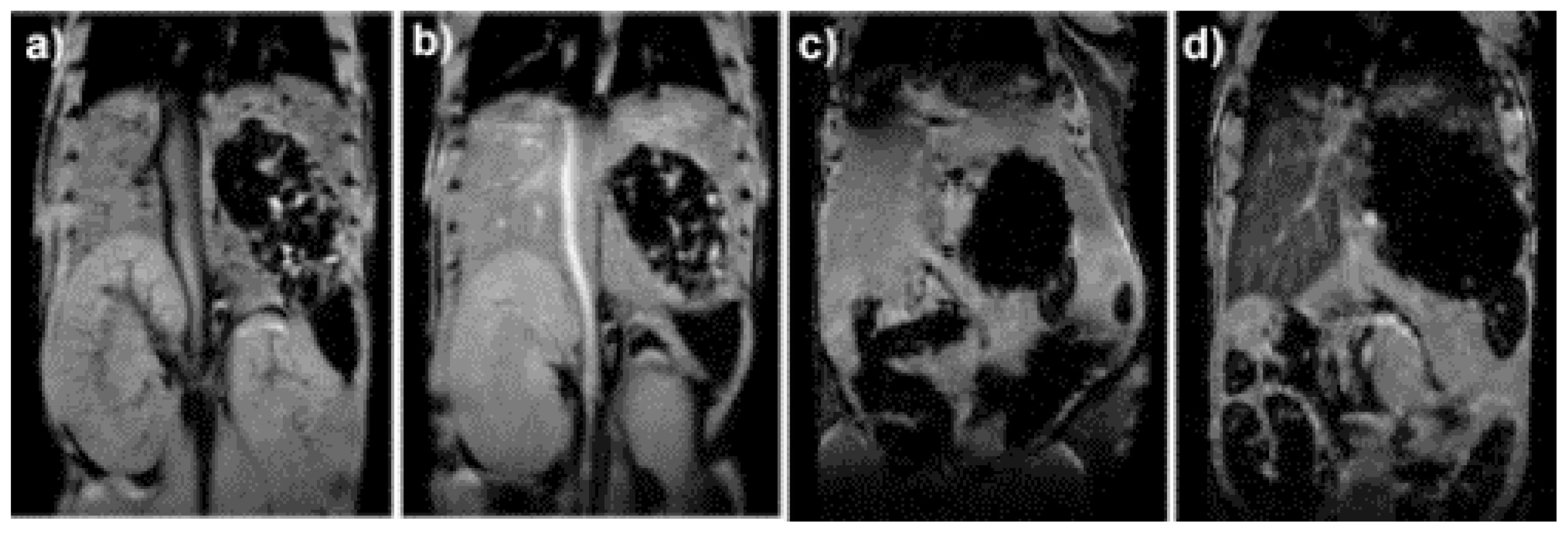
- Taylor, KML; Kim, JS; Rieter, WJ; An, H; Lin, W; Lin, W. Mesoporous silica nanospheres as highly efficient MRI contrast agents. J. Am. Chem. Soc 2008, 130, 2154–2155. [Google Scholar]
- Hsiao, J-K; Tsai, C-P; Chung, T-H; Hung, Y; Yao, M; Liu, H-M; Mou, C-Y; Yang, C-S; Chen, Y-C; Huang, D-M. Mesoporous silica nanoparticles as a delivery system of gadolinium for effective human stem cell tracking. Small 2008, 4, 1445–1452. [Google Scholar]
- Liu, H-M; Wu, S-H; Lu, C-W; Yao, M; Hsiao, J-K; Hung, Y; Lin, Y-S; Mou, C-Y; Yang, C-S; Huang, D-M; et al. Mesoporous silica nanoparticles improve magnetic labeling efficiency in human stem cells. Small 2008, 4, 619–626. [Google Scholar]
- Carniato, F; Tei, L; Cossi, M; Marchese, L; Botta, M. A chemical strategy for the relaxivity enhancement of GdIII chelates anchored on mesoporous silica nanoparticles. Chem. A Eur. J 2010, 16, 10727–10734. [Google Scholar]
- Steinbacher, JL; Lathrop, SA; Cheng, K; Hillegass, JM; Butnor, KJ; Kauppinen, RA; Mossman, BT; Landry, CC. Gd-labeled microparticles in MRI: In vivo imaging of microparticles after intraperitoneal injection. Small 2010, 6, 2678–2682. [Google Scholar]
- Kim, J; Kim, HS; Lee, N; Kim, T; Kim, H; Yu, T; Song, IC; Moon, WK; Hyeon, T. Multifunctional uniform nanoparticles composed of a magnetite nanocrystal core and a mesoporous silica shell for magnetic resonance and fluorescence imaging and for drug delivery. Angew. Chem. Int. Ed 2008, 47, 8438–8441. [Google Scholar]
- Edwards, PP; Thomas, JM. Gold in a metallic divided state—From Faraday to present-day nanoscience. Angew. Chem. Int. Ed 2007, 46, 5480–5486. [Google Scholar]
- Mie, G. Articles on the optical characteristics of turbid tubes, especially colloidal metal solutions. Ann. Phys. Berlin 1908, 25, 377–445. [Google Scholar]
- Kelly, KL; Coronado, E; Zhao, LL; Schatz, GC. The optical properties of metal nanoparticles: The influence of size, shape, and dielectric environment. J. Phys. Chem. B 2003, 107, 668–677. [Google Scholar]
- Turkevich, J; Stevenson, PC; Hillier, J. A study of the nucleation and growth processes in the synthesis of colloidal gold. Discuss. Faraday Soc 1951, 11, 55–75. [Google Scholar]
- Haes, AJ; Stuart, DA; Nie, SM; van Duyne, RP. Using solution-phase nanoparticles, surface-confined nanoparticle arrays and single nanoparticles as biological sensing platforms. J. Fluoresc 2004, 14, 355–367. [Google Scholar]
- Haes, AJ; Hall, WP; Chang, L; Klein, WL; van Duyne, RP. A localized surface plasmon resonance biosensor: First steps toward an assay for Alzheimer‘s disease. Nano Lett 2004, 4, 1029–1034. [Google Scholar]
- Murray, RW. Nanoelectrochemistry: Metal nanoparticles, nanoelectrodes, and nanopores. Chem. Rev 2008, 108, 2688–2720. [Google Scholar]
- Frens, G. Controlled nucleation for regulation of particle-size in monodisperse gold suspensions. Nat. Phys. Sci 1973, 241, 20–22. [Google Scholar]
- Hussain, I; Graham, S; Wang, ZX; Tan, B; Sherrington, DC; Rannard, SP; Cooper, AI; Brust, M. Size-controlled synthesis of near-monodisperse gold nanoparticles in the 1–4 nm range using polymeric stabilizers. J. Am. Chem. Soc 2005, 127, 16398–16399. [Google Scholar]
- Wang, ZX; Tan, BE; Hussain, I; Schaeffer, N; Wyatt, MF; Brust, M; Cooper, AI. Design of polymeric stabilizers for size-controlled synthesis of monodisperse gold nanoparticles in water. Langmuir 2007, 23, 885–895. [Google Scholar]
- Jana, NR; Peng, XG. Single-phase and gram-scale routes toward nearly monodisperse Au and other noble metal nanocrystals. J. Am. Chem. Soc 2003, 125, 14280–14281. [Google Scholar]
- Templeton, AC; Wuelfing, MP; Murray, RW. Monolayer protected cluster molecules. Accounts Chem. Res 2000, 33, 27–36. [Google Scholar]
- Connor, EE; Mwamuka, J; Gole, A; Murphy, CJ; Wyatt, MD. Gold nanoparticles are taken up by human cells but do not cause acute cytotoxicity. Small 2005, 1, 325–327. [Google Scholar]
- Chithrani, BD; Ghazani, AA; Chan, WCW. Determining the size and shape dependence of gold nanoparticle uptake into mammalian cells. Nano Lett 2006, 6, 662–668. [Google Scholar]
- Sandhu, KK; McIntosh, CM; Simard, JM; Smith, SW; Rotello, VM. Gold nanoparticle-mediated transfection of mammalian cells. Bioconjugate Chem 2002, 13, 3–6. [Google Scholar]
- Bowman, MC; Ballard, TE; Ackerson, CJ; Feldheim, DL; Margolis, DM; Melander, C. Inhibition of HIV fusion with multivalent gold nanoparticles. J. Am. Chem. Soc 2008, 130, 6896–6897. [Google Scholar]
- Kim, CK; Ghosh, P; Pagliuca, C; Zhu, ZJ; Menichetti, S; Rotello, VM. Entrapment of hydrophobic drugs in nanoparticle monolayers with efficient release into cancer cells. J. Am. Chem. Soc 2009, 131, 1360–1361. [Google Scholar]
- Tkachenko, AG; Xie, H; Coleman, D; Glomm, W; Ryan, J; Anderson, MF; Franzen, S; Feldheim, DL. Multifunctional gold nanoparticle-peptide complexes for nuclear targeting. J. Am. Chem. Soc 2003, 125, 4700–4701. [Google Scholar]
- Tkachenko, AG; Xie, H; Liu, YL; Coleman, D; Ryan, J; Glomm, WR; Shipton, MK; Franzen, S; Feldheim, DL. Cellular trajectories of peptide-modified gold particle complexes: Comparison of nuclear localization signals and peptide transduction domains. Bioconjugate Chem 2004, 15, 482–490. [Google Scholar]
- Nitin, N; Javier, DJ; Richards-Kortum, R. Oligonucleotide-coated metallic nanoparticles as a flexible platform for molecular imaging agents. Bioconjugate Chem 2007, 18, 2090–2096. [Google Scholar]
- El-Sayed, IH; Huang, XH; El-Sayed, MA. Selective laser photo-thermal therapy of epithelial carcinoma using anti-EGFR antibody conjugated gold nanoparticles. Cancer Lett 2006, 239, 129–135. [Google Scholar]
- Cormode, DP; Skajaa, T; van Schooneveld, MM; Koole, R; Jarzyna, P; Lobatto, ME; Calcagno, C; Barazza, A; Gordon, RE; Zanzonico, P; et al. Nanocrystal core high-density lipoproteins: a multimodality contrast agent platform. Nano Lett 2008, 8, 3715–3723. [Google Scholar]
- Thaxton, CS; Daniel, WL; Giljohann, DA; Thomas, AD; Mirkin, CA. Templated spherical high density lipoprotein nanoparticles. J. Am. Chem. Soc 2009, 131, 1384–1385. [Google Scholar]
- Goodman, CM; McCusker, CD; Yilmaz, T; Rotello, VM. Toxicity of gold nanoparticles functionalized with cationic and anionic side chains. Bioconjugate Chem 2004, 15, 897–900. [Google Scholar]
- Giljohann, DA; Seferos, DS; Daniel, WL; Massich, MD; Patel, PC; Mirkin, CA. Gold nanoparticles for biology and medicine. Angew. Chem. Int. Ed 2010, 49, 3280–3294. [Google Scholar]
- Rosi, NL; Giljohann, DA; Thaxton, CS; Lytton-Jean, AKR; Han, MS; Mirkin, CA. Oligonucleotide-modified gold nanoparticles for intracellular gene regulation. Science 2006, 312, 1027–1030. [Google Scholar]
- Grzelczak, M; Perez-Juste, J; Mulvaney, P; Liz-Marzan, LM. Shape control in gold nanoparticle synthesis. Chem. Soc. Rev 2008, 37, 1783–1791. [Google Scholar]
- Perez-Juste, J; Pastoriza-Santos, I; Liz-Marzan, LM; Mulvaney, P. Gold nanorods: Synthesis, characterization and applications. Coord. Chem. Rev 2005, 249, 1870–1901. [Google Scholar]
- Jain, PK; El-Sayed, IH; El-Sayed, MA. Au nanoparticles target cancer. Nano Today 2007, 2, 18–29. [Google Scholar]
- Murphy, CJ; Gole, AM; Stone, JW; Sisco, PN; Alkilany, AM; Goldsmith, EC; Baxter, SC. Gold nanoparticles in biology: Beyond toxicity to cellular imaging. Accounts Chem. Res 2008, 41, 1721–1730. [Google Scholar]
- Huang, XH; El-Sayed, IH; Qian, W; El-Sayed, MA. Cancer cell imaging and photothermal therapy in the near-infrared region by using gold nanorods. J. Am. Chem. Soc 2006, 128, 2115–2120. [Google Scholar]
- Saito, G; Swanson, JA; Lee, KD. Drug delivery strategy utilizing conjugation via reversible disulfide linkages: Role and site of cellular reducing activities. Adv. Drug Deliv. Rev 2003, 55, 199–215. [Google Scholar]
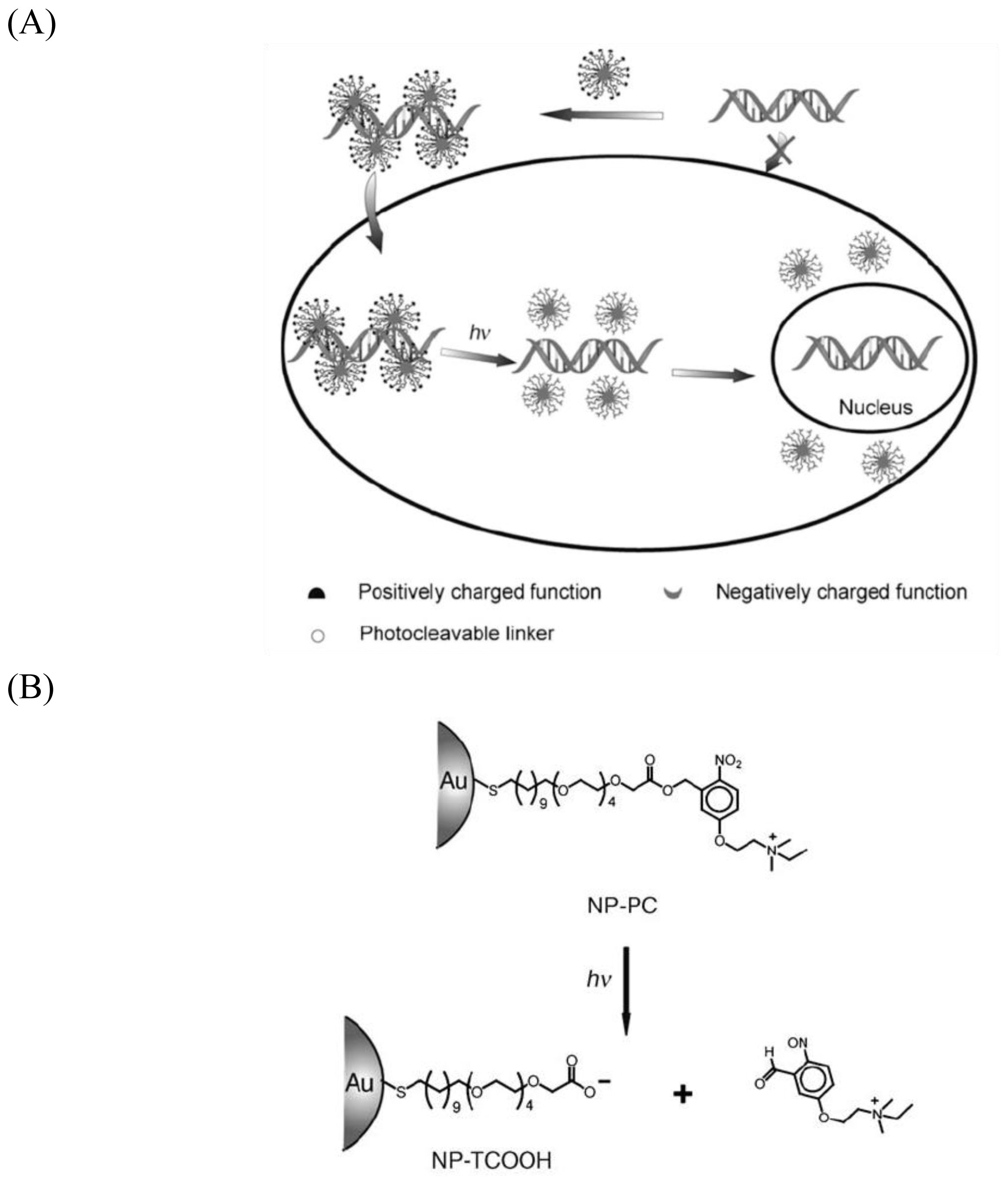
- Han, G; You, CC; Kim, BJ; Turingan, RS; Forbes, NS; Martin, CT; Rotello, VM. Light-regulated release of DNA and its delivery to nuclei by means of photolabile gold nanoparticles. Angew. Chem. Int. Ed 2006, 45, 3165–3169. [Google Scholar]
- Radt, B; Smith, TA; Caruso, F. Optically addressable nanostructured capsules. Adv. Mater 2004, 16, 2184–2189. [Google Scholar]
- Angelatos, AS; Radt, B; Caruso, F. Light-responsive polyelectrolyte/gold nanoparticle microcapsules. J. Phys. Chem. B 2005, 109, 3071–3076. [Google Scholar]
- Skirtach, AG; Dejugnat, C; Braun, D; Susha, AS; Rogach, AL; Parak, WJ; Mohwald, H; Sukhorukov, GB. The role of metal nanoparticles in remote release of encapsulated materials. Nano Lett 2005, 5, 1371–1377. [Google Scholar]
- Skirtach, AG; Javier, AM; Kreft, O; Kohler, K; Alberola, AP; Mohwald, H; Parak, WJ; Sukhorukov, GB. Laser-induced release of encapsulated materials inside living cells. Angew. Chem. Int. Ed 2006, 45, 4612–4617. [Google Scholar]
- Jain, RK; Booth, MF. What brings pericytes to tumor vessels? J. Clin. Invest 2003, 112, 1134–1136. [Google Scholar]
- O‘Neal, DP; Hirsch, LR; Halas, NJ; Payne, JD; West, JL. Photo-thermal tumor ablation in mice using near infrared-absorbing nanoparticles. Cancer Lett 2004, 209, 171–176. [Google Scholar]
- Love, JC; Estroff, LA; Kriebel, JK; Nuzzo, RG; Whitesides, GM. Self-assembled monolayers of thiolates on metals as a form of nanotechnology. Chem. Rev 2005, 105, 1103–1169. [Google Scholar]
- Pissuwan, D; Valenzuela, SM; Cortie, MB. Therapeutic possibilities of plasmonically heated gold nanoparticles. Trends Biotech 2006, 24, 62–67. [Google Scholar]
- Govorov, AO; Zhang, W; Skeini, T; Richardson, H; Lee, J; Kotov, NA. Gold nanoparticle ensembles as heaters and actuators: Melting and collective plasmon resonances. Nanoscale Res. Lett 2006, 1, 84–90. [Google Scholar]
- El-Sayed, MA. Some interesting properties of metals confined in time and nanometer space of different shapes. Accounts Chem. Res 2001, 34, 257–264. [Google Scholar]
- Loo, C; Lin, A; Hirsch, L; Lee, MH; Barton, J; Halas, NJ; West, J; Drezek, R. Nanoshell-enabled photonics-based imaging and therapy of cancer. Technol. Cancer Res. Treat 2004, 3, 33–40. [Google Scholar]
- Sershen, SR; Westcott, SL; Halas, NJ; West, JL. Temperature-sensitive polymer-nanoshell composites for photothermally modulated drug delivery. J. Biomed. Mater. Res 2000, 51, 293–298. [Google Scholar]
- Weissleder, R. A clearer vision for in vivo imaging. Nat. Biotech 2001, 19, 316–317. [Google Scholar]
- Lee, KS; El-Sayed, MA. Dependence of the enhanced optical scattering efficiency relative to that of absorption for gold metal nanorods on aspect ratio, size, end-cap shape, and medium refractive index. J. Phys. Chem. B 2005, 109, 20331–20338. [Google Scholar]
- Brioude, A; Jiang, XC; Pileni, MP. Optical properties of gold nanorods: DDA simulations supported by experiments. J. Phys. Chem. B 2005, 109, 13138–13142. [Google Scholar]
- Oldenburg, SJ; Averitt, RD; Westcott, SL; Halas, NJ. Nanoengineering of optical resonances. Chem. Phys. Lett 1998, 288, 243–247. [Google Scholar]
- Grainger, RG. Intravascular contrast-media: The past, the present and the future. Br. J. Radiol 1982, 55, 1–18. [Google Scholar]
- Bugaj, JE; Achilefu, S; Dorshow, RB; Rajagopalan, R. Novel fluorescent contrast agents for optical imaging of in vivo tumors based on a receptor-targeted dye-peptide conjugate platform. J. Biomed. Opt 2001, 6, 122–133. [Google Scholar]
- Faulk, WP; Taylor, GM. Immunocolloid method for electronmicroscope. Immunochemistry 1971, 8, 1081–1083. [Google Scholar]
- Daneels, G; Moeremans, M; Deraeymaeker, M; Demey, J. Sequential immunostaining (gold-silver) and complete protein staining (aurodye) on western blots. J. Immunol. Meth 1986, 89, 89–91. [Google Scholar]
- Weng, HA; Wu, CC; Chen, CG; Ho, CC; Ding, SJ. Preparation and properties of gold nanoparticle-electrodeposited titanium substrates with Arg-Gly-Asp-Cys peptides. J. Mater. Sci. Mater. Med 2010, 21, 1511–1519. [Google Scholar]
- Felsenfeld, DP; Choquet, D; Sheetz, MP. Ligand binding regulates the directed movement of beta 1 integrins on fibroblasts. Nature 1996, 383, 438–440. [Google Scholar]
- Lasne, D; Blab, GA; Berciaud, S; Heine, M; Groc, L; Choquet, D; Cognet, L; Lounis, B. Single nanoparticle photothermal tracking (SNaPT) of 5-nm gold beads in live cells. Biophys. J 2006, 91, 4598–4604. [Google Scholar]
- Cognet, L; Tardin, C; Boyer, D; Choquet, D; Tamarat, P; Lounis, B. Single metallic nanoparticle imaging for protein detection in cells. Proc. Natl. Acad. Sci. USA 2003, 100, 11350–11355. [Google Scholar]
- Hainfeld, JF; Slatkin, DN; Focella, TM; Smilowitz, HM. Gold nanoparticles: A new x-ray contrast agent. Br. J. Radiol 2006, 79, 248–253. [Google Scholar]
- Kim, D; Park, S; Lee, JH; Jeong, YY; Jon, S. Antibiofouling polymer-coated gold nanoparticles as a contrast agent for in vivo x-ray computed tomography imaging. J. Am. Chem. Soc 2007, 129, 7661–7665. [Google Scholar]
- Albrechtbuehler, G. Phagokinetic tracks of 3T3 cells-parallels between orientation of track segments and of cellular structiures which contain actin or tubulin. Cell 1977, 12, 333–339. [Google Scholar]
- Albrechtbuehler, G. Phagokinetic tracks of 3T3 cells. Cell 1977, 11, 395–404. [Google Scholar]
- Albrechtbuehler, G. Angular-distribution of directional changes of guided 3T3-cells. J. Cell. Biol 1979, 80, 53–60. [Google Scholar]
- Sperling, RA; Gil, PR; Zhang, F; Zanella, M; Parak, WJ. Biological applications of gold nanoparticles. Chem. Soc. Rev 2008, 37, 1896–1908. [Google Scholar]
- Roth, J. The silver anniversary of gold: 25 years of the colloidal gold marker system for immunocytochemistry and histochemistry. Histochem. Cell. Biol 1996, 106, 1–8. [Google Scholar]
- Souza, GR; Christianson, DR; Staquicini, FI; Ozawa, MG; Snyder, EY; Sidman, RL; Miller, JH; Arap, W; Pasqualini, R. Networks of gold nanoparticles and bacteriophage as biological sensors and cell-targeting agents. Proc. Natl. Acad. Sci. USA 2006, 103, 1215–1220. [Google Scholar]
- Boisselier, E; Astruc, D. Gold nanoparticles in nanomedicine: Preparations, imaging, diagnostics, therapies and toxicity. Chem. Soc. Rev 2009, 38, 1759–1782. [Google Scholar]
- Ray, PC; Darbha, GK; Ray, A; Walker, J; Hardy, W. Gold nanoparticle based FRET for DNA detection. Plasmonics 2007, 2, 173–183. [Google Scholar]
- Pons, T; Medintz, IL; Sapsford, KE; Higashiya, S; Grimes, AF; English, DS; Mattoussi, H. On the quenching of semiconductor quantum dot photoluminescence by proximal gold nanoparticles. Nano Lett 2007, 7, 3157–3164. [Google Scholar]
- You, CC; Miranda, OR; Gider, B; Ghosh, PS; Kim, IB; Erdogan, B; Krovi, SA; Bunz, UHF; Rotello, VM. Detection and identification of proteins using nanoparticle-fluorescent polymer ‘chemical nose’ sensors. Nat. Nanotech 2007, 2, 318–323. [Google Scholar]
- Mi, CC; Zhang, JP; Gao, HY; Wu, XL; Wang, M; Wu, YF; Di, YQ; Xu, ZR; Mao, CB; Xu, SK. Multifunctional nanocomposites of superparamagnetic (Fe3O4) and NIR-responsive rare earth-doped up-conversion fluorescent (NaYF4 : Yb, Er) nanoparticles and their applications in biolabeling and fluorescent imaging of cancer cells. Nanoscale 2010, 2, 1141–1148. [Google Scholar]
- Sun, P; Zhang, HY; Liu, C; Fang, J; Wang, M; Chen, J; Zhang, JP; Mao, CB; Xu, SK. Preparation and characterization of Fe3O4/CdTe magnetic/fluorescent nanocomposites and their applications in immuno-labeling and fluorescent imaging of cancer cells. Langmuir 2010, 26, 1278–1284. [Google Scholar]
- Chen, FH; Gao, Q; Ni, JZ. The grafting and release behavior of doxorubincin from Fe3O4@SiO2 core-shell structure nanoparticles via an acid cleaving amide bond: The potential for magnetic targeting drug delivery. Nanotechnology 2008, 19. [Google Scholar]
- Lin, BL; Shen, XD; Cui, S. Application of nanosized Fe3O4 in anticancer drug carriers with target-orientation and sustained-release properties. Biomed. Mater 2007, 2, 132–134. [Google Scholar]
- Wang, J; Zhang, K; Peng, ZM; Chen, QW. Magnetic properties improvement in Fe3O4 nanoparticles grown under magnetic fields. J. Cryst. Growth 2004, 266, 500–504. [Google Scholar]
- Feng, B; Hong, RY; Wang, LS; Guo, L; Li, HZ; Ding, J; Zheng, Y; Wei, DG. Synthesis of Fe3O4/APTES/PEG diacid functionalized magnetic nanoparticles for MR imaging. Colloid. Surf. A-Physicochem. Eng. Asp 2008, 328, 52–59. [Google Scholar]
- Nishimura, K; Hasegawa, M; Ogura, Y; Nishi, T; Kataoka, K; Handa, H; Abe, M. 4 degrees C preparation of ferrite nanoparticles having protein molecules immobilized on their surfaces. J. Appl. Phys 2002, 91, 8555–8556. [Google Scholar]
- Lai, CW; Wang, YH; Lai, CH; Yang, MJ; Chen, CY; Chou, PT; Chan, CS; Chi, Y; Chen, YC; Hsiao, JK. Iridium-complex-functionalized Fe3O4/SiO2 core/shell nanoparticles: A facile three-in-one system in magnetic resonance imaging, luminescence imaging, and photodynamic therapy. Small 2008, 4, 218–224. [Google Scholar]
- Ankamwar, B; Lai, TC; Huang, JH; Liu, RS; Hsiao, M; Chen, CH; Hwu, YK. Biocompatibility of Fe3O4 nanoparticles evaluated by in vitro cytotoxicity assays using normal, glia and breast cancer cells. Nanotechnology 2010, 21, 075102. [Google Scholar]
- Jiang, J; Gu, HW; Shao, HL; Devlin, E; Papaefthymiou, GC; Ying, JY. Manipulation bifunctional Fe3O4-Ag heterodimer nanoparticles for two-photon fluorescence imaging and magnetic manipulation. Adv. Mater 2008, 20, 4403–4407. [Google Scholar]
- Lv, G; He, F; Wang, XM; Gao, F; Zhang, G; Wang, T; Jiang, H; Wu, CH; Guo, DD; Li, XM; et al. Novel nanocomposite of nano Fe3O4 and polylactide nanofibers for application in drug uptake and induction of cell death of leukemia cancer cells. Langmuir 2008, 24, 2151–2156. [Google Scholar]
- Gang, J; Park, SB; Hyung, W; Choi, EH; Wen, J; Kim, HS; Shul, YG; Haam, S; Song, Y. Magnetic poly epsilon-caprolactone nanoparticles containing Fe3O4 and gemcitabine enhance anti-tumor effect in pancreatic cancer xenograft mouse model. J. Drug Target 2007, 15, 445–453. [Google Scholar]
- Chen, FH; Zhang, LM; Chen, QT; Zhang, Y; Zhang, ZJ. Synthesis of a novel magnetic drug delivery system composed of doxorubicin-conjugated Fe3O4 nanoparticle cores and a PEG-functionalized porous silica shell. Chem. Commun 2010, 46, 8633–8635. [Google Scholar]
- Nigam, S; Barick, KC; Bahadur, D. Development of citrate-stabilized Fe3O4 nanoparticles: Conjugation and release of doxorubicin for therapeutic applications. J. Magn. Magn. Mater 2011, 323, 237–243. [Google Scholar]
- Guo, MA; Que, CL; Wang, CH; Liu, XZ; Yan, HS; Liu, KL. Multifunctional superparamagnetic nanocarriers with folate-mediated and pH-responsive targeting properties for anticancer drug delivery. Biomaterials 2011, 32, 185–194. [Google Scholar]
- Wang, XM; Zhang, RY; Wu, CH; Dai, YY; Song, M; Gutmann, S; Gao, F; Lv, G; Li, JY; Li, XM; et al. The application of Fe3O4 nanoparticles in cancer research: A new strategy to inhibit drug resistance. J. Biomed. Mater. Res. A 2007, 80A, 852–860. [Google Scholar]
- Kim, GC; Li, YY; Chu, YF; Cheng, SX; Zhuo, RX; Zhang, XZ. Nanosized temperature-responsive Fe3O4-UA-g-P(UA-co-NIPAAm) magnetomicelles for controlled drug release. Eur. Polym. J 2008, 44, 2761–2767. [Google Scholar]
- Purushotham, S; Ramanujan, RV. Modeling the performance of magnetic nanoparticles in multimodal cancer therapy. J Appl Phys 2010, 107. [Google Scholar]
- Shi, DL; Cho, HS; Chen, Y; Xu, H; Gu, HC; Lian, J; Wang, W; Liu, GK; Huth, C; Wang, LM; et al. Fluorescent polystyrene-Fe3O4 composite nanospheres for in vivo imaging and hyperthermia. Adv. Mater 2009, 21, 2170–2173. [Google Scholar]
- Hou, Y; Liu, Y; Chen, Z; Gu, N; Wang, J. Manufacture of IRDye800CW-coupled Fe3O4 nanoparticles and their applications in cell labeling and in vivo imaging. J Nanobiotech 2010, 8. [Google Scholar]
- Tan, H; Xue, JM; Shuter, B; Li, X; Wang, J. Synthesis of PEOlated Fe3O4@SiO2 nanoparticles via bioinspired silification for magnetic resonance imaging. Adv. Funct. Mater 2010, 20, 722–731. [Google Scholar]
- Hong, S; Chang, Y; Rhee, I. Chitosan-coated Ferrite (Fe3O4) nanoparticles as a T-2 contrast agent for magnetic resonance imaging. J. Korean Phys. Soc 2010, 56, 868–873. [Google Scholar]
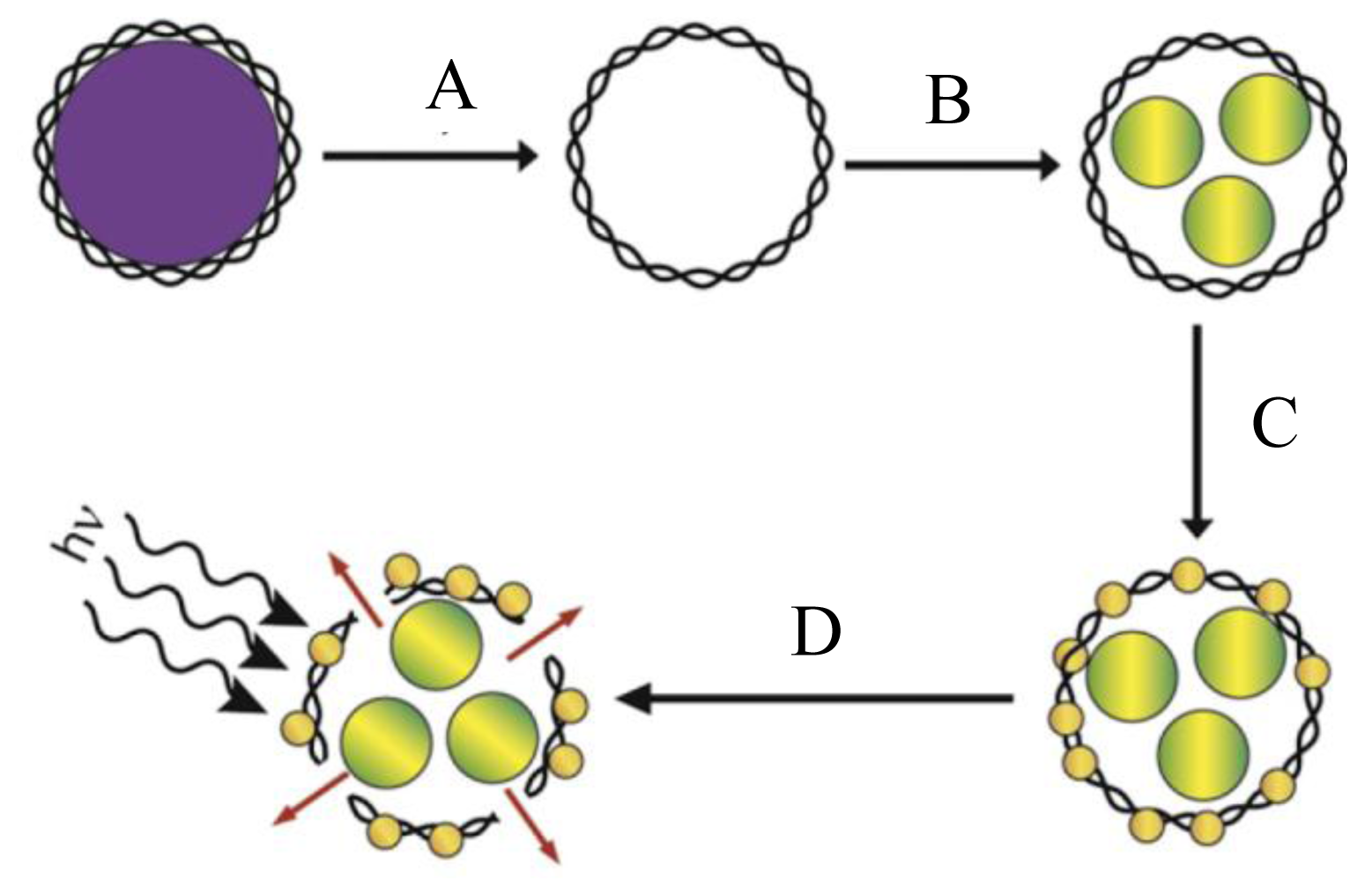
- Wang, CG; Chen, J; Talavage, T; Irudayaraj, J. Gold Nanorod/Fe3O4 nanoparticle “nano-pearl-necklaces” for simultaneous targeting, dual-mode imaging, and photothermal ablation of cancer cells. Angew. Chem. Int. Ed 2009, 48, 2759–2763. [Google Scholar]
- Brus, LE. Electron-electron and electron-hole interactions in small semiconductor crystallites—The size dependence of the lowest excited electronic state. J. Chem. Phys 1984, 80, 4403–4409. [Google Scholar]
- Dabbousi, BO; RodriguezViejo, J; Mikulec, FV; Heine, JR; Mattoussi, H; Ober, R; Jensen, KF; Bawendi, MG. (CdSe)ZnS core-shell quantum dots: Synthesis characterization of a size series of highly luminescent nanocrystallites. J. Phys. Chem B 1997, 101, 9463–9475. [Google Scholar]
- Dahan, M; Laurence, T; Pinaud, F; Chemla, DS; Alivisatos, AP; Sauer, M; Weiss, S. Time-gated biological imaging by use of colloidal quantum dots. Opt. Lett 2001, 26, 825–827. [Google Scholar]
- Smith, AM; Duan, H; Mohs, AM; Nie, S. Bioconjugated quantum dots for in vivo molecular and cellular imaging. Adv. Drug Deliv. Rev 2008, 60, 1226–1240. [Google Scholar]
- Rosenthal, SJ; McBride, J; Pennycook, SJ; Feldman, LC. Synthesis, surface studies, composition and structural characterization of CdSe, core/shell and biologically active nanocrystals. Surf. Sci. Rep 2007, 62, 111–157. [Google Scholar]
- Chen, FQ; Gerion, D. Fluorescent CdSe/ZnS nanocrystal-peptide conjugates for long-term, nontoxic imaging and nuclear targeting in living cells. Nano Lett 2004, 4, 1827–1832. [Google Scholar]
- Hild, WA; Breunig, M; Goepferich, A. Quantum dots—Nano-sized probes for the exploration of cellular and intracellular targeting. Eur. J. Pharm. Biopharm 2008, 68, 153–168. [Google Scholar]
- Zrazhevskiy, P; Sena, M; Gao, XH. Designing multifunctional quantum dots for bioimaging, detection, and drug delivery. Chem. Soc. Rev 2010, 39, 4326–4354. [Google Scholar]
- Duonghong, D; Ramsden, J; Gratzel, M. Dynamics of interfacial electron-transfer processes in colloidal semiconductor systems. J. Am. Chem. Soc 1982, 104, 2977–2985. [Google Scholar]
- Peng, XG; Manna, L; Yang, WD; Wickham, J; Scher, E; Kadavanich, A; Alivisatos, AP. Shape control of CdSe nanocrystals. Nature 2000, 404, 59–61. [Google Scholar]
- Halder, N; Rashmi, R; Chakrabarti, S; Stanley, CR; Herrera, M; Browning, ND. A comprehensive study of the effect of in situ annealing at high growth temperature on the morphological and optical properties of self-assembled InAs/GaAs QDs. Appl. Phys. A Mater. Sci. Process 2009, 95, 713–720. [Google Scholar]
- Murray, CB; Norris, DJ; Bawendi, MG. Synthesis and characterization of nearly monodisperse CDE (E = S, Se, Te) semiconductor nanocrystallites. J. Am. Chem. Soc 1993, 115, 8706–8715. [Google Scholar]
- Hines, MA; Guyot-Sionnest, P. Synthesis and characterization of strongly luminescing ZnS-Capped CdSe nanocrystals. J. Phys. Chem 1996, 100, 468–471. [Google Scholar]
- Peng, XG; Schlamp, MC; Kadavanich, AV; Alivisatos, AP. Epitaxial growth of highly luminescent CdSe/CdS core/shell nanocrystals with photostability and electronic accessibility. J. Am. Chem. Soc 1997, 119, 7019–7029. [Google Scholar]
- Peng, ZA; Peng, XG. Formation of high-quality CdTe, CdSe, and CdS nanocrystals using CdO as precursor. J. Am. Chem. Soc 2001, 123, 183–184. [Google Scholar]
- Qu, LH; Peng, ZA; Peng, XG. Alternative routes toward high quality CdSe nanocrystals. Nano Lett 2001, 1, 333–337. [Google Scholar]
- Mekis, I; Talapin, DV; Kornowski, A; Haase, M; Weller, H. One-pot synthesis of highly luminescent CdSe/CdS core-shell nanocrystals via organometallic and “greener” chemical approaches. J. Phys. Chem. B 2003, 107, 7454–7462. [Google Scholar]
- Yu, WW; Peng, XG. Formation of high-quality CdS and other II-VI semiconductor nanocrystals in noncoordinating solvents: Tunable reactivity of monomers. Angew. Chem. Int. Ed 2002, 41, 2368–2371. [Google Scholar]
- Colvin, VL. The potential environmental impact of engineered nanomaterials. Nat. Biotech 2003, 21, 1166–1170. [Google Scholar]
- Derfus, AM; Chan, WCW; Bhatia, SN. Probing the cytotoxicity of semiconductor quantum dots. Nano Lett 2004, 4, 11–18. [Google Scholar]
- Weng, KC; Noble, CO; Papahadjopoulos-Sternberg, B; Chen, FF; Drummond, DC; Kirpotin, DB; Wang, DH; Hom, YK; Hann, B; Park, JW. Targeted tumor cell internalization and imaging of multifunctional quantum dot-conjugated immunoliposomes in vitro and in vivo. Nano Lett 2008, 8, 2851–2857. [Google Scholar]
- Al-Jamal, WT; Al-Jamal, KT; Bomans, PH; Frederik, PM; Kostarelos, K. Functionalized-quantum-dot-liposome hybrids as multimodal nanoparticles for cancer. Small 2008, 4, 1406–1415. [Google Scholar]
- Bentolila, LA; Doose, S; Ebenstein, Y; Iyer, G; Li, JJ; Michalet, X; Pinaud, F; Tsay, J; Weiss, S. Mattoussi, H, Cheon, J, Eds.; Peptide-functionalized quantum dots for live diagnostic imaging and therapeutic applications. In Inorganic Nanoprobes for Biological Sensing and Imaging; Artech House Publisher: Boston, MA, USA, 2009; pp. 45–69. [Google Scholar]
- Yang, K; Li, ZG; Cao, Y; Yu, XL; Mei, J. Effect of peptide-conjugated near-infrared fluorescent quantum dots (NIRF-QDs) on the invasion and metastasis of human tongue squamous cell carcinoma cell line Tca8113 in vitro. Int. J. Mol. Sci 2009, 10, 4418–4427. [Google Scholar]
- Efros, AL; Rosen, M. Random telegraph signal in the photoluminescence intensity of a single quantum dot. Phys. Rev. Lett 1997, 78, 1110–1113. [Google Scholar]
- Michalet, X; Pinaud, F; Lacoste, TD; Dahan, M; Bruchez, MP; Alivisatos, AP; Weiss, S. Properties of fluorescent semiconductor nanocrystals and their application to biological labeling. Single Mol 2001, 2, 261–276. [Google Scholar]
- Pinaud, F; Michalet, X; Bentolila, LA; Tsay, JM; Doose, S; Li, JJ; Iyer, G; Weiss, S. Advances in fluorescence imaging with quantum dot bio-probes. Biomaterials 2006, 27, 1679–1687. [Google Scholar]
- Hanaki, K; Momo, A; Oku, T; Komoto, A; Maenosono, S; Yamaguchi, Y; Yamamoto, K. Semiconductor quantum dot/albumin complex is a long-life and highly photostable endosome marker. Biochem. Biophys. Res. Commun 2003, 302, 496–501. [Google Scholar]
- Kaul, Z; Yaguchi, T; Kaul, SC; Hirano, T; Wadhwa, R; Taira, K. Mortalin imaging in normal and cancer cells with quantum dot immuno-conjugates. Cell Res 2003, 13, 503–507. [Google Scholar]
- Medintz, IL; Uyeda, HT; Goldman, ER; Mattoussi, H. Quantum dot bioconjugates for imaging, labelling and sensing. Nat. Mater 2005, 4, 435–446. [Google Scholar]
- Dubertret, B; Skourides, P; Norris, DJ; Noireaux, V; Brivanlou, AH; Libchaber, A. In vivo imaging of quantum dots encapsulated in phospholipid micelles. Science 2002, 298, 1759–1762. [Google Scholar]
- Dahan, M; Levi, S; Luccardini, C; Rostaing, P; Riveau, B; Triller, A. Diffusion dynamics of glycine receptors revealed by single-quantum dot tracking. Science 2003, 302, 442–445. [Google Scholar]
- Jaiswal, JK; Mattoussi, H; Mauro, JM; Simon, SM. Long-term multiple color imaging of live cells using quantum dot bioconjugates. Nat. Biotech 2003, 21, 47–51. [Google Scholar]
- Pellegrino, T; Parak, WJ; Boudreau, R; Le Gros, MA; Gerion, D; Alivisatos, AP; Larabell, CA. Quantum dot-based cell motility assay. Differentiation 2003, 71, 542–548. [Google Scholar]
- Jares-Erijman, EA; Jovin, TM. FRET imaging. Nat. Biotech 2003, 21, 1387–1395. [Google Scholar]
- Chen, AA; Derfus, AM; Khetani, SR; Bhatia, SN. Quantum dots to monitor RNAi delivery and improve gene silencing. Nucl. Acids Res 2005, 33, e190. [Google Scholar]
- Howarth, M; Takao, K; Hayashi, Y; Ting, AY. Targeting quantum dots to surface proteins in living cells with biotin ligase. Proc. Natl. Acad. Sci. USA 2005, 102, 7583–7588. [Google Scholar]
- Edgar, R; McKinstry, M; Hwang, J; Oppenheim, AB; Fekete, RA; Giulian, G; Merril, C; Nagashima, K; Adhya, S. High-sensitivity bacterial detection using biotin-tagged phage and quantum-dot nanocomplexes. Proc. Natl. Acad. Sci. USA 2006, 103, 4841–4845. [Google Scholar]
- Olek, M; Busgen, T; Hilgendorff, M; Giersig, M. Quantum dot modified multiwall carbon nanotubes. J. Phys. Chem. B 2006, 110, 12901–12904. [Google Scholar]
- Pinaud, F; King, D; Moore, HP; Weiss, S. Bioactivation and cell targeting of semiconductor CdSe/ZnS nanocrystals with phytochelatin-related peptides. J. Am. Chem. Soc 2004, 126, 6115–6123. [Google Scholar]
- Liu, HY; Vu, TQ. Identification of quantum dot bioconjugates and cellular protein co-localization by hybrid gel blotting. Nano Lett 2007, 7, 1044–1049. [Google Scholar]
- Bakalova, R; Zhelev, Z; Ohba, H; Baba, Y. Quantum dot-based western blot technology for ultrasensitive detection of tracer proteins. J. Am. Chem. Soc 2005, 127, 9328–9329. [Google Scholar]
- Makrides, SC; Gasbarro, C; Bello, JM. Bioconjugation of quantum dot luminescent probes for western blot analysis. Biotechniques 2005, 39, 501–506. [Google Scholar]
- Frangioni, JV. In vivo near-infrared fluorescence imaging. Curr. Opin. Chem. Biol 2003, 7, 626–634. [Google Scholar]
- Voura, EB; Jaiswal, JK; Mattoussi, H; Simon, SM. Tracking metastatic tumor cell extravasation with quantum dot nanocrystals and fluorescence emission-scanning microscopy. Nat. Med 2004, 10, 993–998. [Google Scholar]
- Stroh, M; Zimmer, JP; Duda, DG; Levchenko, TS; Cohen, KS; Brown, EB; Scadden, DT; Torchilin, VP; Bawendi, MG; Fukumura, D; et al. Quantum dots spectrally distinguish multiple species within the tumor milieu in vivo. Nat. Med 2005, 11, 678–682. [Google Scholar]
- Smith, JD; Fisher, GW; Waggoner, AS; Campbell, PG. The use of quantum dots for analysis of chick CAM vasculature. Microvasc. Res 2007, 73, 75–83. [Google Scholar]
- Akerman, ME; Chan, WCW; Laakkonen, P; Bhatia, SN; Ruoslahti, E. Nanocrystal targeting in vivo. Proc. Natl. Acad. Sci. USA 2002, 99, 12617–12621. [Google Scholar]
- Michalet, X; Pinaud, FF; Bentolila, LA; Tsay, JM; Doose, S; Li, JJ; Sundaresan, G; Wu, AM; Gambhir, SS; Weiss, S. Quantum dots for live cells, in vivo imaging, and diagnostics. Science 2005, 307, 538–544. [Google Scholar]
- Chan, WCW; Nie, SM. Quantum dot bioconjugates for ultrasensitive nonisotopic detection. Science 1998, 281, 2016–2018. [Google Scholar]
- Pathak, S; Choi, SK; Arnheim, N; Thompson, ME. Hydroxylated quantum dots as luminescent probes for in situ hybridization. J. Am. Chem. Soc 2001, 123, 4103–4104. [Google Scholar]
- Wu, XY; Liu, HJ; Liu, JQ; Haley, KN; Treadway, JA; Larson, JP; Ge, NF; Peale, F; Bruchez, MP. Immunofluorescent labeling of cancer marker Her2 and other cellular targets with semiconductor quantum dots. Nat. Biotech 2003, 21, 41–46. [Google Scholar]
- Gao, XH; Cui, YY; Levenson, RM; Chung, LWK; Nie, SM. In vivo cancer targeting and imaging with semiconductor quantum dots. Nat. Biotech 2004, 22, 969–976. [Google Scholar]
- Gerion, D; Pinaud, F; Williams, SC; Parak, WJ; Zanchet, D; Weiss, S; Alivisatos, AP. Synthesis and properties of biocompatible water-soluble silica-coated CdSe/ZnS semiconductor quantum dots. J. Phys. Chem. B 2001, 105, 8861–8871. [Google Scholar]
- Gao, XH; Chan, WCW; Nie, SM. Quantum-dot nanocrystals for ultrasensitive biological labeling and multicolor optical encoding. J. Biomed. Opt 2002, 7, 532–537. [Google Scholar]
- Pellegrino, T; Manna, L; Kudera, S; Liedl, T; Koktysh, D; Rogach, AL; Keller, S; Radler, J; Natile, G; Parak, WJ. Hydrophobic nanocrystals coated with an amphiphilic polymer shell: A general route to water soluble nanocrystals. Nano Lett 2004, 4, 703–707. [Google Scholar]
- Osaki, F; Kanamori, T; Sando, S; Sera, T; Aoyama, Y. A quantum dot conjugated sugar ball and its cellular uptake on the size effects of endocytosis in the subviral region. J. Am. Chem. Soc 2004, 126, 6520–6521. [Google Scholar]
- Mattoussi, H; Mauro, JM; Goldman, ER; Anderson, GP; Sundar, VC; Mikulec, FV; Bawendi, MG. Self-assembly of CdSe-ZnS quantum dot bioconjugates using an engineered recombinant protein. J. Am. Chem. Soc 2000, 122, 12142–12150. [Google Scholar]
- Sukhanova, A; Devy, M; Venteo, L; Kaplan, H; Artemyev, M; Oleinikov, V; Klinov, D; Pluot, M; Cohen, JHM; Nabiev, I. Biocompatible fluorescent nanocrystals for immunolabeling of membrane proteins and cells. Anal. Biochem 2004, 324, 60–67. [Google Scholar]
- Mitchell, GP; Mirkin, CA; Letsinger, RL. Programmed assembly of DNA functionalized quantum dots. J. Am. Chem. Soc 1999, 121, 8122–8123. [Google Scholar]
- Bruchez, M; Moronne, M; Gin, P; Weiss, S; Alivisatos, AP. Semiconductor nanocrystals as fluorescent biological labels. Science 1998, 281, 2013–2016. [Google Scholar]
- Wang, S; Jarrett, BR; Kauzlarich, SM; Louie, AY. Core/shell quantum dots with high relaxivity and photoluminescence for multimodality imaging. J. Am. Chem. Soc 2007, 129, 3848–3856. [Google Scholar]
- Selvan, ST; Patra, PK; Ang, CY; Ying, JY. Synthesis of silica-coated semiconductor and magnetic quantum dots and their use in the imaging of live cells. Angew. Chem. Int. Ed 2007, 46, 2448–2452. [Google Scholar]
- Mulder, WJM; Koole, R; Brandwijk, RJ; Storm, G; Chin, PTK; Strijkers, GJ; Donega, CD; Nicolay, K; Griffioen, AW. Quantum dots with a paramagnetic coating as a bimodal molecular imaging probe. Nano Lett 2006, 6, 1–6. [Google Scholar]
- van Tilborg, GAF; Mulder, WJM; Chin, PTK; Storm, G; Reutelingsperger, CP; Nicolay, K; Strijkers, GJ. Annexin A5-conjugated quantum dots with a paramagnetic lipidic coating for the multimodal detection of apoptotic cells. Bioconjugate Chem 2006, 17, 865–868. [Google Scholar]
- Prinzen, L; Miserus, R; Dirksen, A; Hackeng, TM; Deckers, N; Bitsch, NJ; Megens, RTA; Douma, K; Heemskerk, JW; Kooi, ME; et al. Optical and magnetic resonance imaging of cell death and platelet activation using annexin A5-functionalized quantum dots. Nano Lett 2007, 7, 93–100. [Google Scholar]
- Wang, DS; He, JB; Rosenzweig, N; Rosenzweig, Z. Superparamagnetic Fe2O3 Beads-CdSe/ZnS quantum dots core-shell nanocomposite particles for cell separation. Nano Lett 2004, 4, 409–413. [Google Scholar]
- Gu, HW; Zheng, RK; Zhang, XX; Xu, B. Facile one-pot synthesis of bifunctional heterodimers of nanoparticles: A conjugate of quantum dot and magnetic nanoparticles. J. Am. Chem. Soc 2004, 126, 5664–5665. [Google Scholar]

© 2011 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Vivero-Escoto, J.L.; Huang, Y.-T. Inorganic-Organic Hybrid Nanomaterials for Therapeutic and Diagnostic Imaging Applications. Int. J. Mol. Sci. 2011, 12, 3888-3927. https://doi.org/10.3390/ijms12063888
Vivero-Escoto JL, Huang Y-T. Inorganic-Organic Hybrid Nanomaterials for Therapeutic and Diagnostic Imaging Applications. International Journal of Molecular Sciences. 2011; 12(6):3888-3927. https://doi.org/10.3390/ijms12063888
Chicago/Turabian StyleVivero-Escoto, Juan L., and Yu-Tzu Huang. 2011. "Inorganic-Organic Hybrid Nanomaterials for Therapeutic and Diagnostic Imaging Applications" International Journal of Molecular Sciences 12, no. 6: 3888-3927. https://doi.org/10.3390/ijms12063888
APA StyleVivero-Escoto, J. L., & Huang, Y.-T. (2011). Inorganic-Organic Hybrid Nanomaterials for Therapeutic and Diagnostic Imaging Applications. International Journal of Molecular Sciences, 12(6), 3888-3927. https://doi.org/10.3390/ijms12063888



