Characterization and Emulsification Properties of Rhamnolipid and Sophorolipid Biosurfactants and Their Applications
Abstract
:1. Introduction
2. Surfactant Characterization
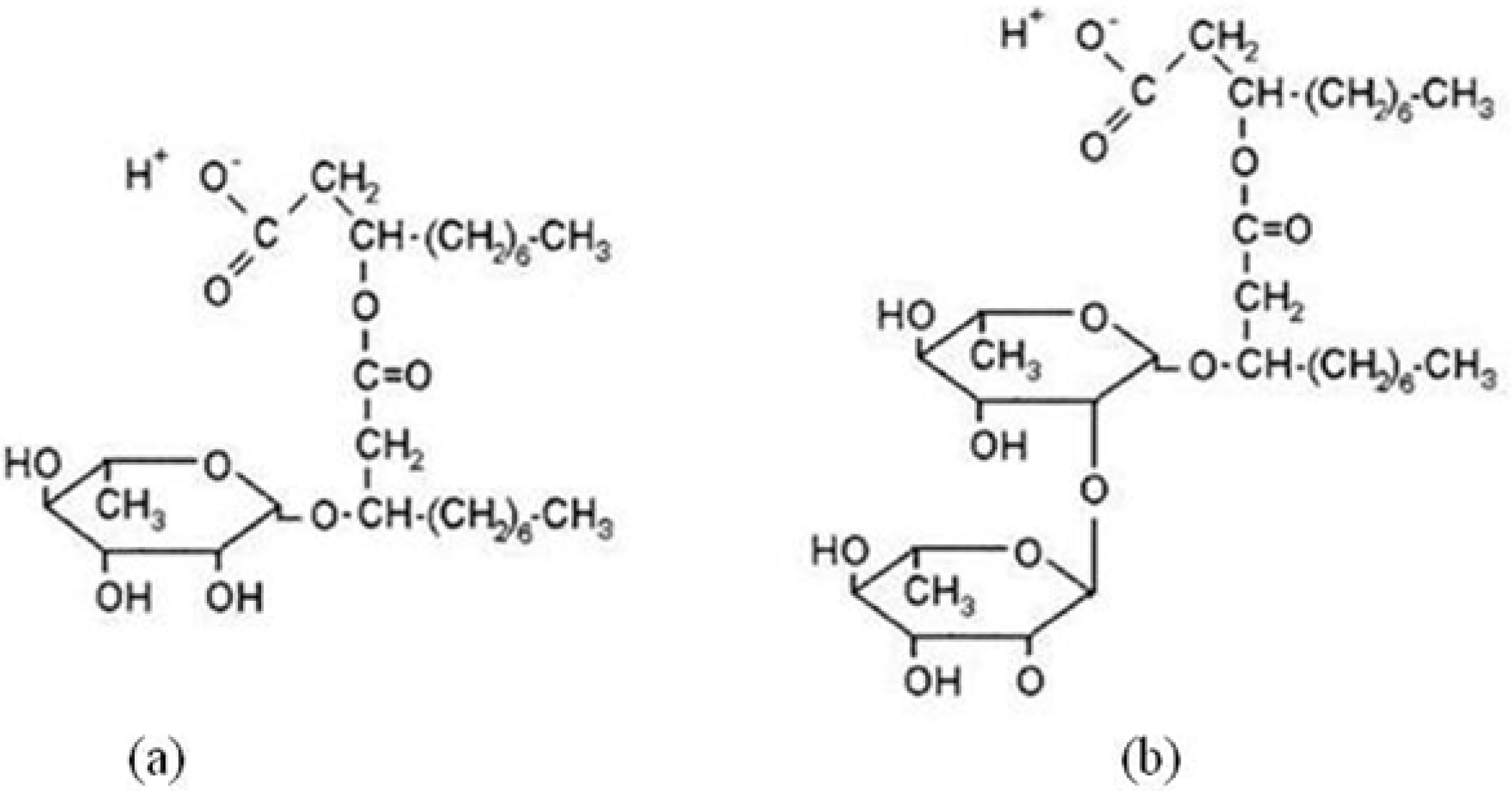
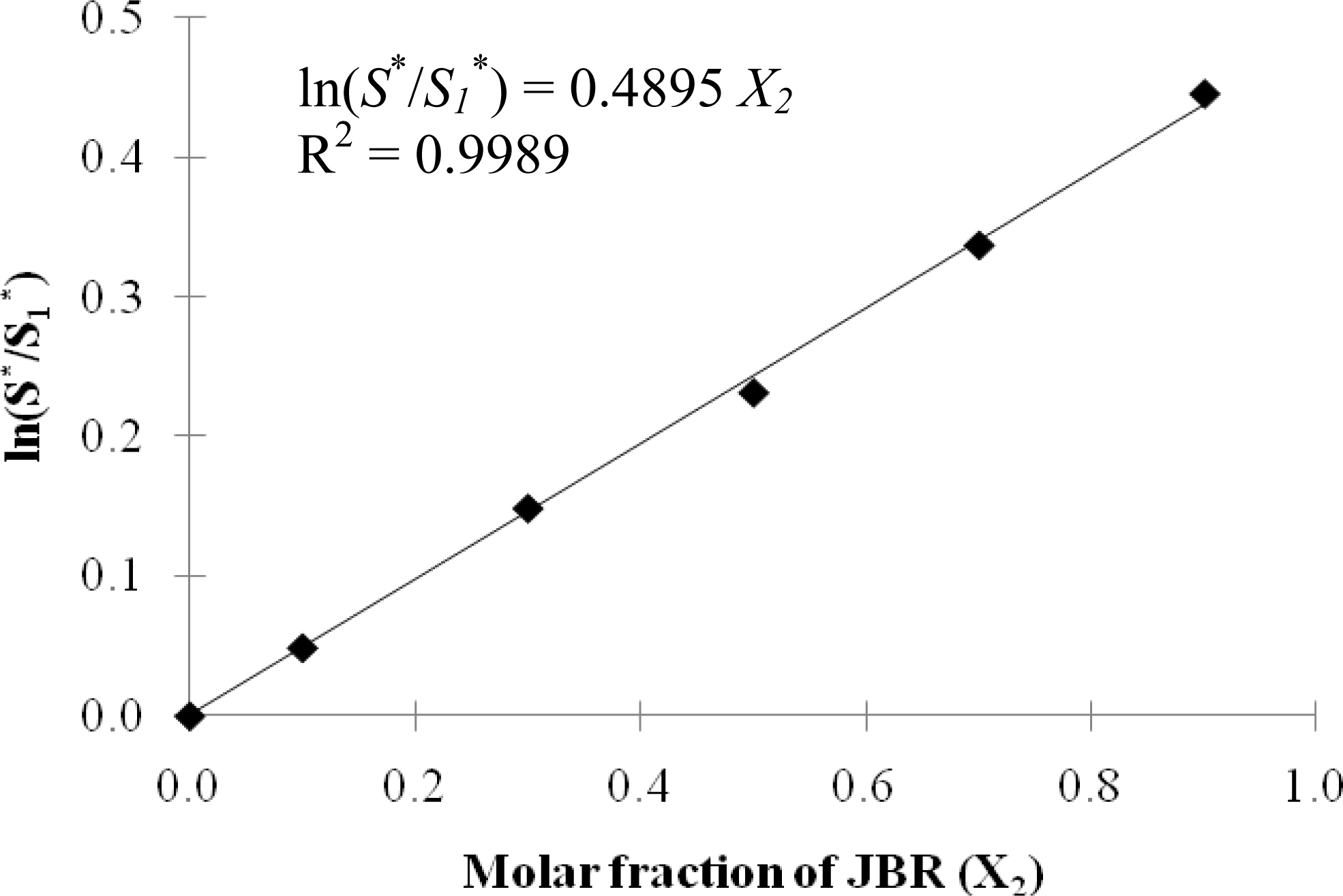
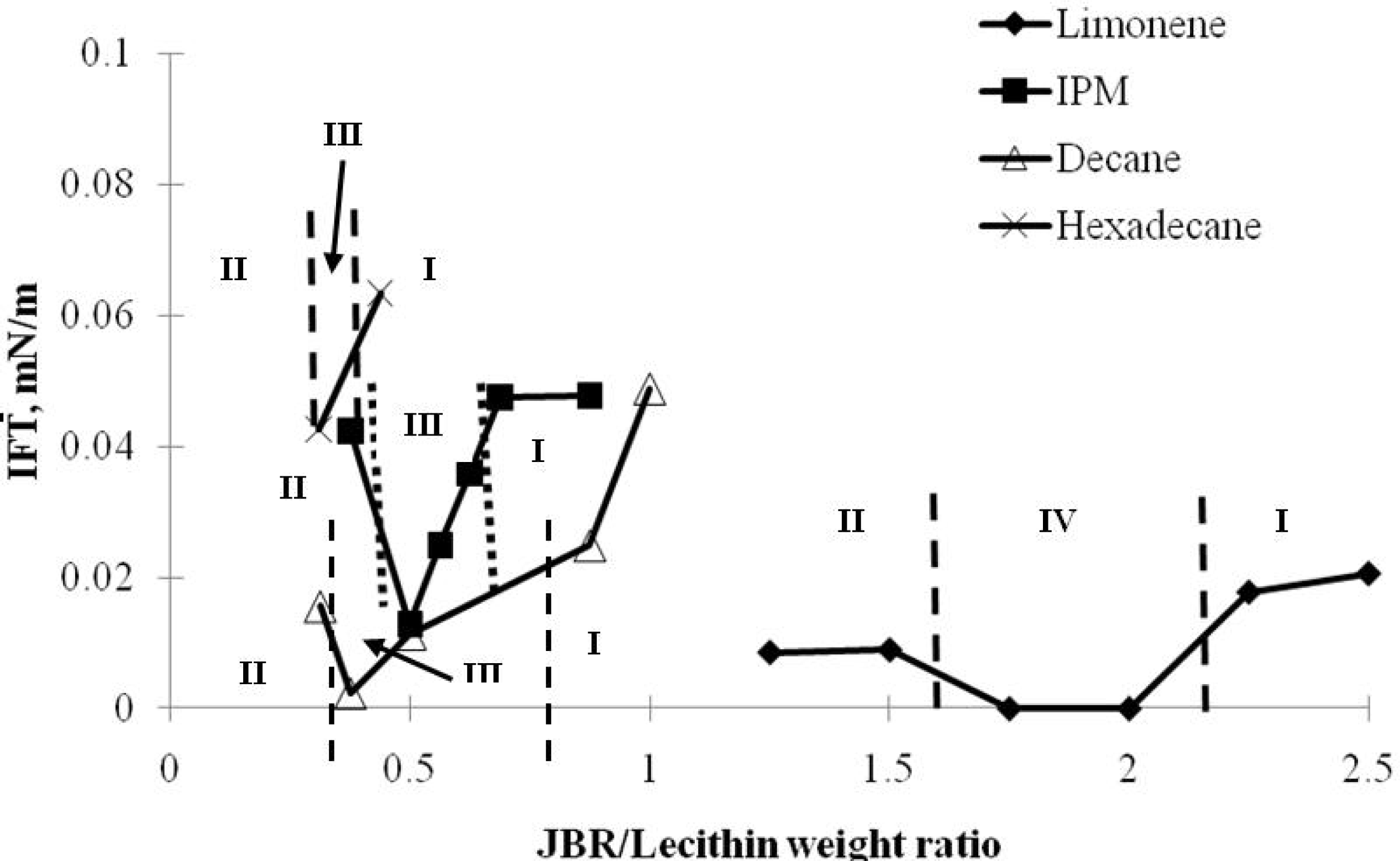
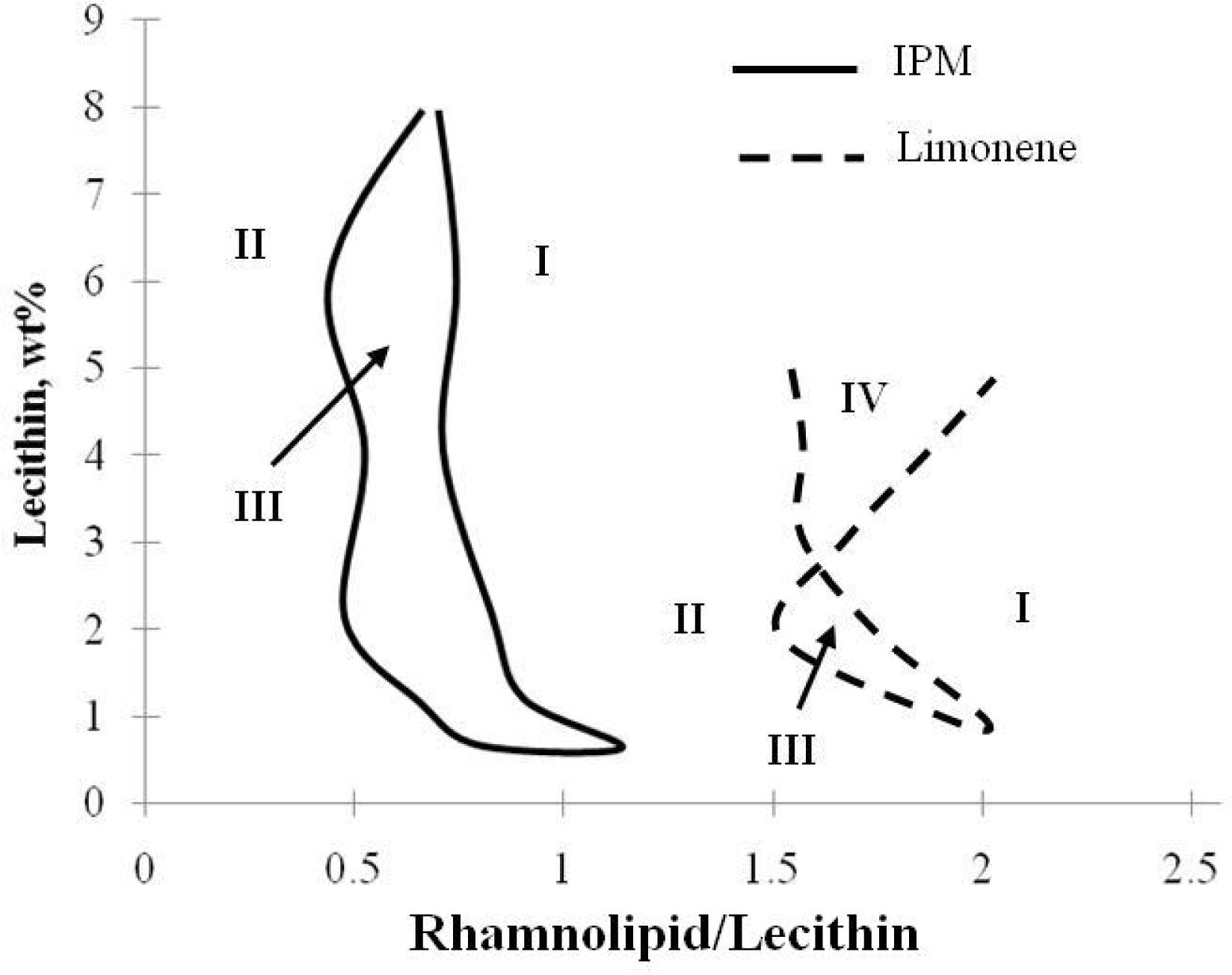
2.1. Characteristic Curvature and Rhamnolipid Characterization
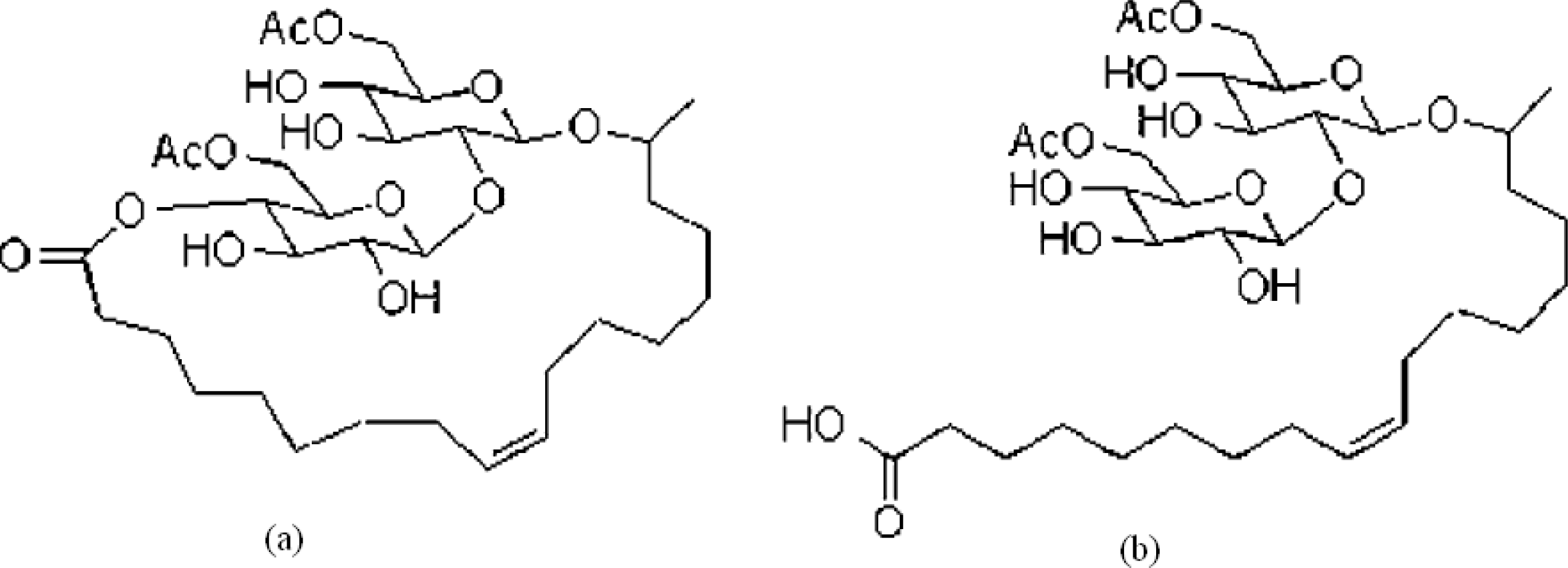
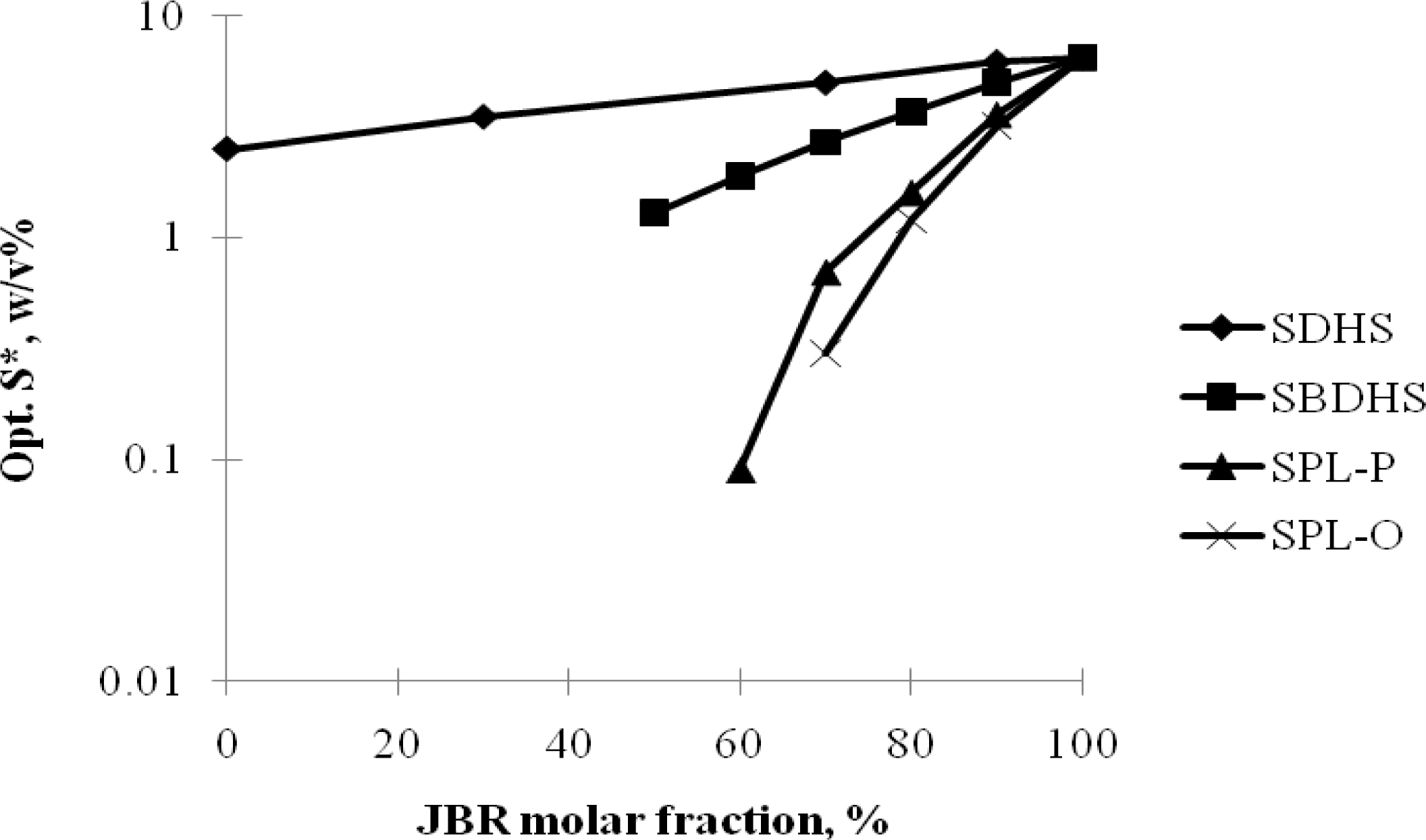
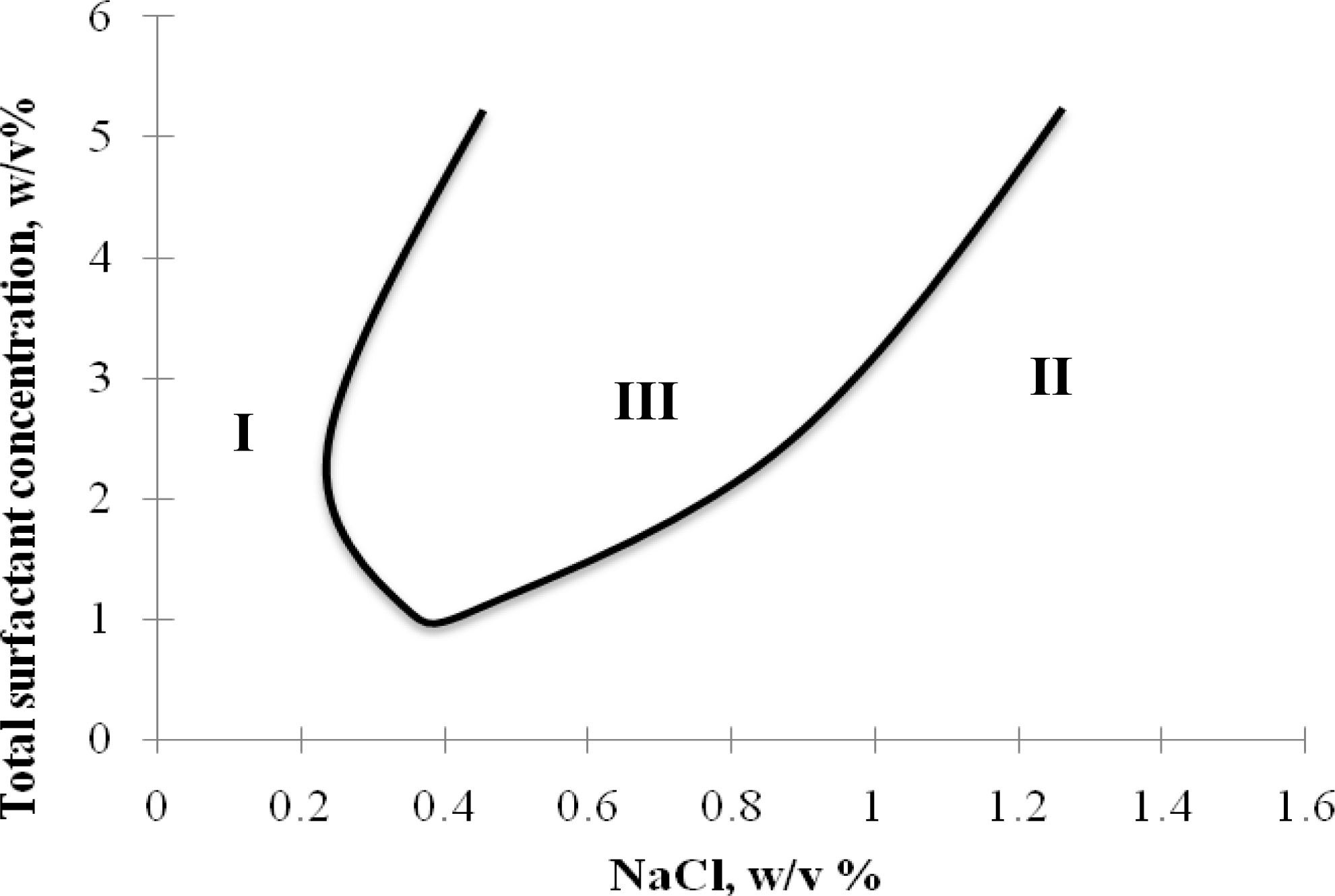
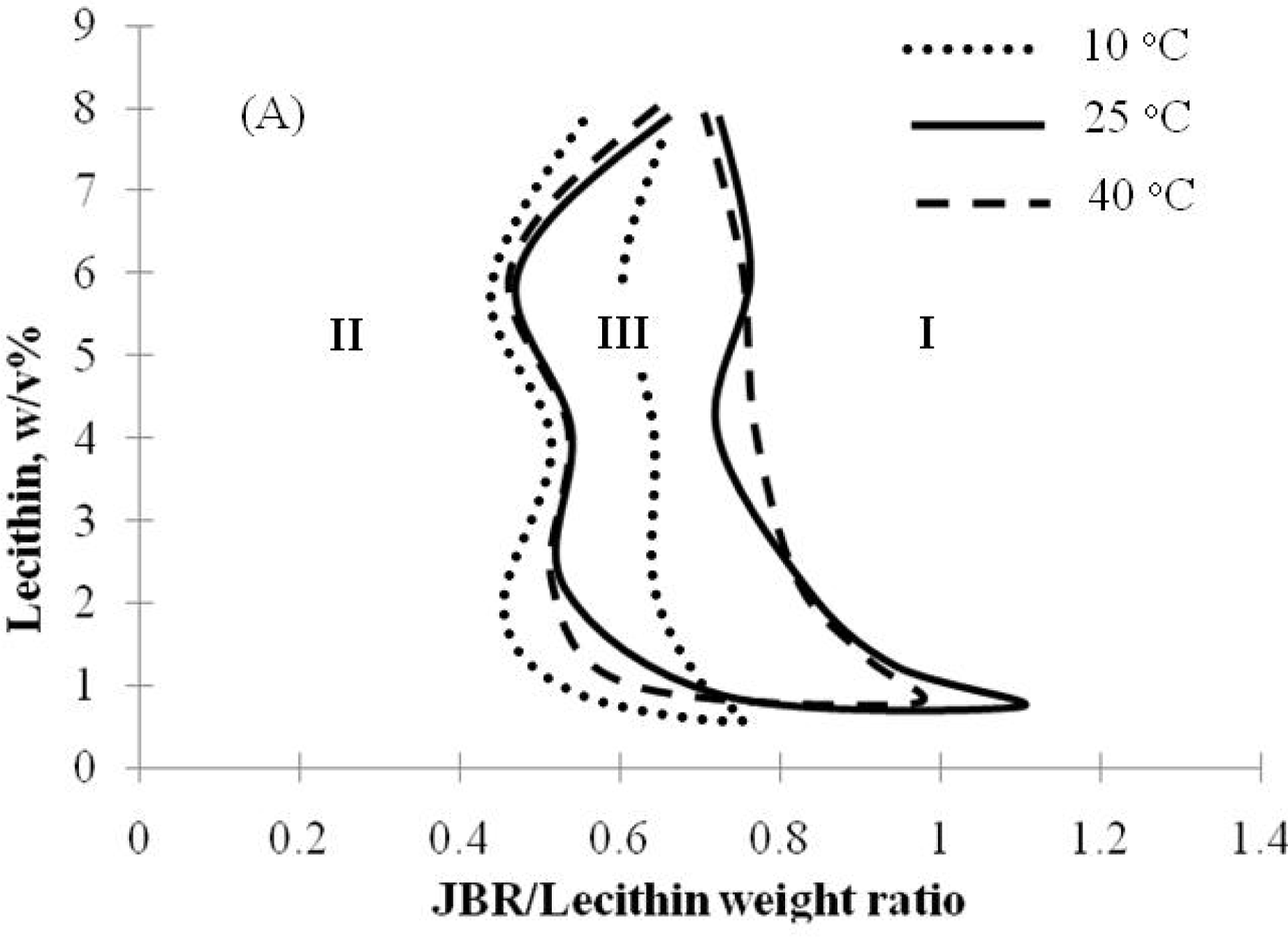
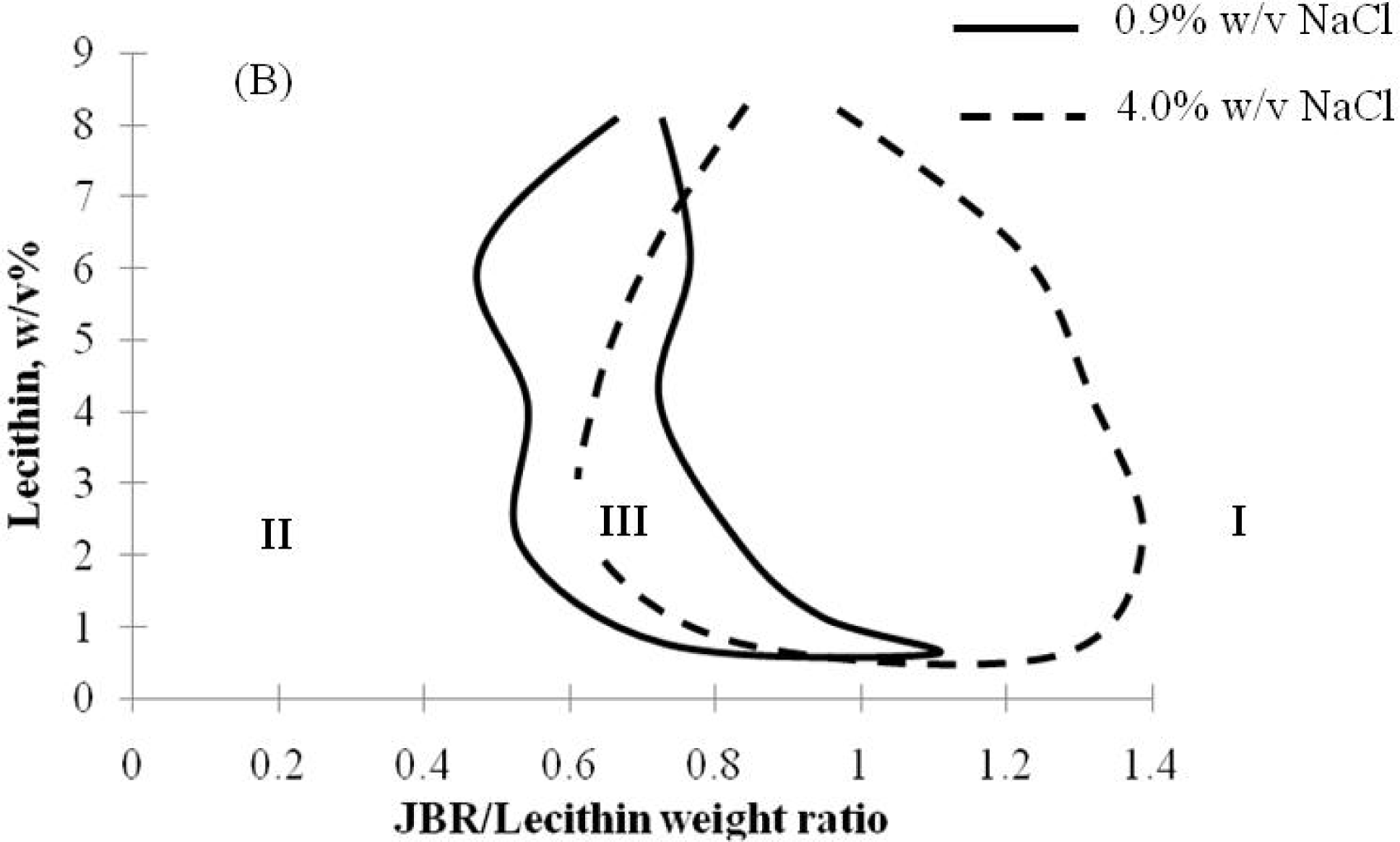
2.2. Winsor R-ratio, Optimum Salinity and Sophorolipid Characterization
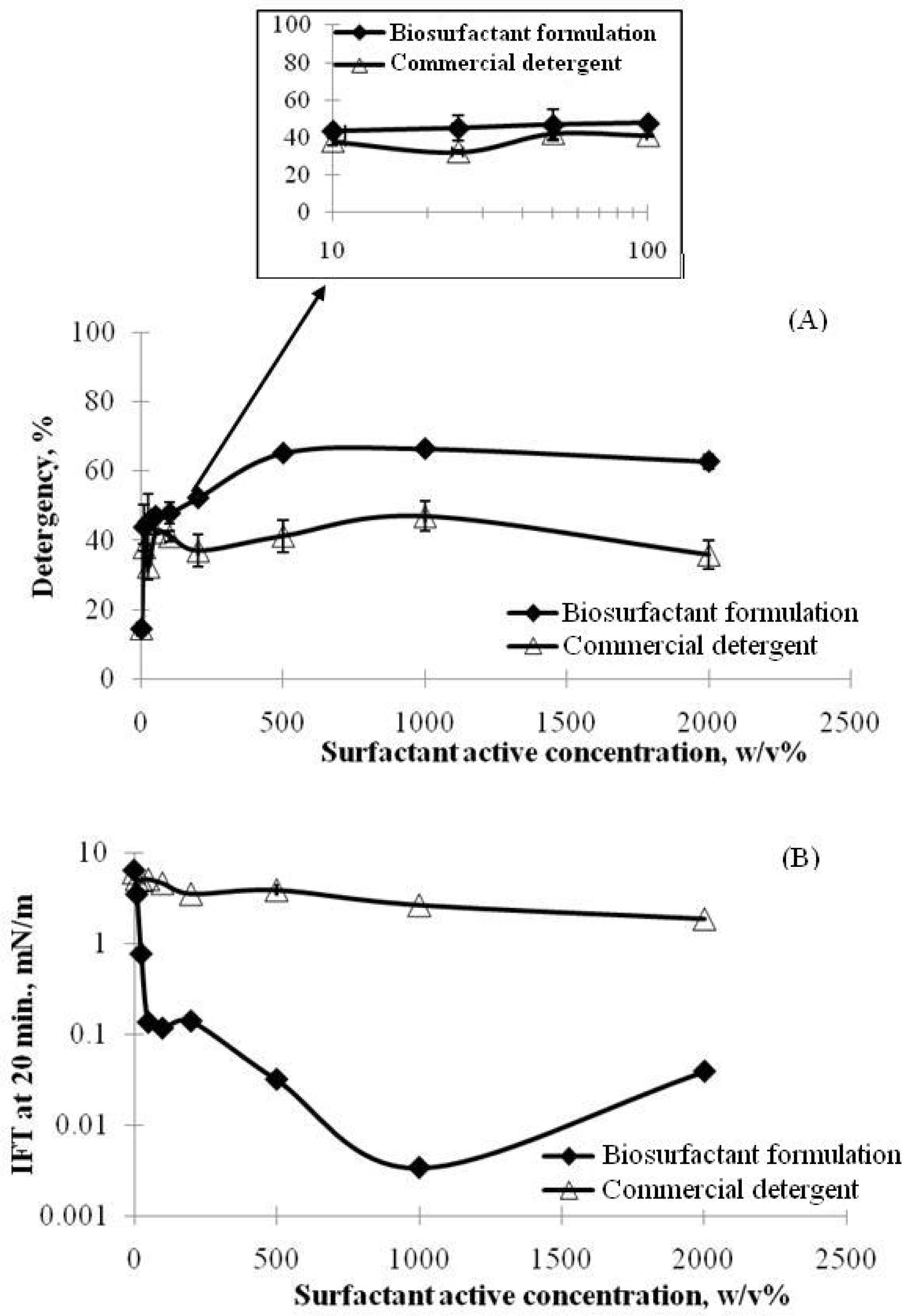
3. Applications
4. Conclusions
References
- Helvaci, ŞŞ; Pecker, S; Özdemir, G. Effect of electrolytes on the surface behavior of rhamnolipids R1 and R2. Colloid. Surface. B 2004, 35, 225–233. [Google Scholar]
- Ashby, RD; Solaiman, DKY; Foglia, TA. Property control of sophorolipids: Influence of fatty acid substrate and blending. Biotechnol. Lett 2008, 30, 1093–1100. [Google Scholar]
- Xie, Y; Li, Y; Ye, R. Effect of alcohols on the phase behavior of microemulsions formed by a biosurfactant—Rhamnolipid. J. Dispersion Sci. Technol 2005, 26, 455–461. [Google Scholar]
- Özdemir, G; Peker, S; Helvaci, ŞŞ. Effect of pH on the surface and interfacial behavior of rhamnolipids R1 and R2. Colloids Surf. A: Physicochem Eng. Aspects 2004, 234, 135–143. [Google Scholar]
- Rosen, MJ. Surfactants and Interfacial Phenomena, 2nd ed; John Wiley & Sons, Inc: New York, NY, USA, 1989. [Google Scholar]
- Bourrel, M; Schechter, RS. Microemulsions and Related Systems: Formulation, Solvency, and Physical Properties; Surfactant Sci Ser 30; Marcel Dekker: New York, NY, USA, 1988; pp. 229–302. [Google Scholar]
- Banat, IM; Makkar, RS; Cameotra, SS. Potential commercial applications of microbial surfactants. Appl. Microbiol. Biotechnol 2000, 53, 495–508. [Google Scholar]
- Brown, MJ. Biosurfactants for cosmetic applications. Int. J. Cosmet. Sci 1991, 13, 61–64. [Google Scholar]
- Desai, JD; Banat, IM. Microbiol production of surfactants and their commercial potential. Microbiol. Molecular Biol. Rev 1997, 61, 47–64. [Google Scholar]
- Maier, RM; Soberón-Chávez, G. Pseudomonas Aeruginosa rhamnolipids: Biosynthesis and potential applications. Appl. Microbiol. Biotechnol 2000, 54, 625–633. [Google Scholar]
- Makkar, RS; Cameotra, SS. An update on the use of unconventional substrates for biosurfactant production and their new applications. Appl. Microbiol. Biotechnol 2002, 58, 428–434. [Google Scholar]
- Rodrigues, L; Banat, IM; Teixeira, J; Oliveira, R. Biosurfactants: Potential applications in medicine. J. Antimicrobial. Chemother 2006, 57, 609–618. [Google Scholar]
- Acosta, EJ; Yuan, JS; Bhakta, AS. The characteristic curvature of ionic surfactants. J. Surfactants Deterg 2008, 11, 145–158. [Google Scholar]
- Salager, JL; Morgan, JC; Schechter, RS; Wade, WH; Vasquez, E. Optimum formulation of surfactant/water/oil systems for minimum interfacial tension or phase behavior. Soc. Petrol. Eng. J 1979, 19, 107–115. [Google Scholar]
- Salager, JL. Ionic microemulsions. In Handbook of Microemulsion Science and Technology; Kumar, P, Mittal, KL, Eds.; Marcel Dekker: New York, NY, USA, 1999; pp. 247–280. [Google Scholar]
- Nguyen, TT; Sabatini, DA. Formulating alcohol-free microemulsions using rhamnolipid biosurfactant and rhamnolipid mixtures. J. Surfactants Deterg 2009, 12, 109–115. [Google Scholar]
- Nguyen, TT; Youssef, NH; McInerney, MJ; Sabatini, DA. Rhamnolipid biosurfactant mixtures for environmental remediation. Water Res 2008, 42, 1735–1743. [Google Scholar]
- Winsor, P. Solvent Properties of Amphiphilic Compounds; Butterworth: London, UK, 1954. [Google Scholar]
- Salager, JL; Antón, RE; Sabatini, DA; Harwell, JH; Acosta, EJ; Tolosa, LI. Enhancing solubilization in microemulsions—State of the art and current trends. J. Surfactants Deterg 2005, 8, 3–21. [Google Scholar]
- Nguyen, TT; Edelen, A; Neighbors, B; Sabatini, DA. Biocompatible lecithin-based microemulsions with rhamnolipid and sophorolipid biosurfactants: Formulation and potential applications. J. Colloid Interface Sci 2010, 348, 498–504. [Google Scholar]
- Nguyen, T; Do, L; Sabatini, DA. Biodiesel production via peanut oil extraction using diesel-based reverse-micellar microemulsions. Fuel 2010, 89, 2285–2291. [Google Scholar]
- Thompson, L. The role of oil detachment mechanism in determining optimum detergency conditions. J. Colloid Interface Sci 1994, 163, 61–73. [Google Scholar]
- Tongcumpou, C; Acosta, EJ; Quencer, LB; Joseph, AF; Scamehorn, JF; Sabatini, DA; Chavadej, S; Yanumet, N. Microemulsion formation and detergency with oily soils: II. Detergency formulation and performance. J. Surfactants Deterg 2003, 6, 205–214. [Google Scholar]


© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nguyen, T.T.; Sabatini, D.A. Characterization and Emulsification Properties of Rhamnolipid and Sophorolipid Biosurfactants and Their Applications. Int. J. Mol. Sci. 2011, 12, 1232-1244. https://doi.org/10.3390/ijms12021232
Nguyen TT, Sabatini DA. Characterization and Emulsification Properties of Rhamnolipid and Sophorolipid Biosurfactants and Their Applications. International Journal of Molecular Sciences. 2011; 12(2):1232-1244. https://doi.org/10.3390/ijms12021232
Chicago/Turabian StyleNguyen, Thu T., and David A. Sabatini. 2011. "Characterization and Emulsification Properties of Rhamnolipid and Sophorolipid Biosurfactants and Their Applications" International Journal of Molecular Sciences 12, no. 2: 1232-1244. https://doi.org/10.3390/ijms12021232




