Role of Versican, Hyaluronan and CD44 in Ovarian Cancer Metastasis
Abstract
:1. Introduction
2. Ovarian Cancer Metastasis
3. Role of the Extracellular Matrix in Ovarian Cancer Metastasis
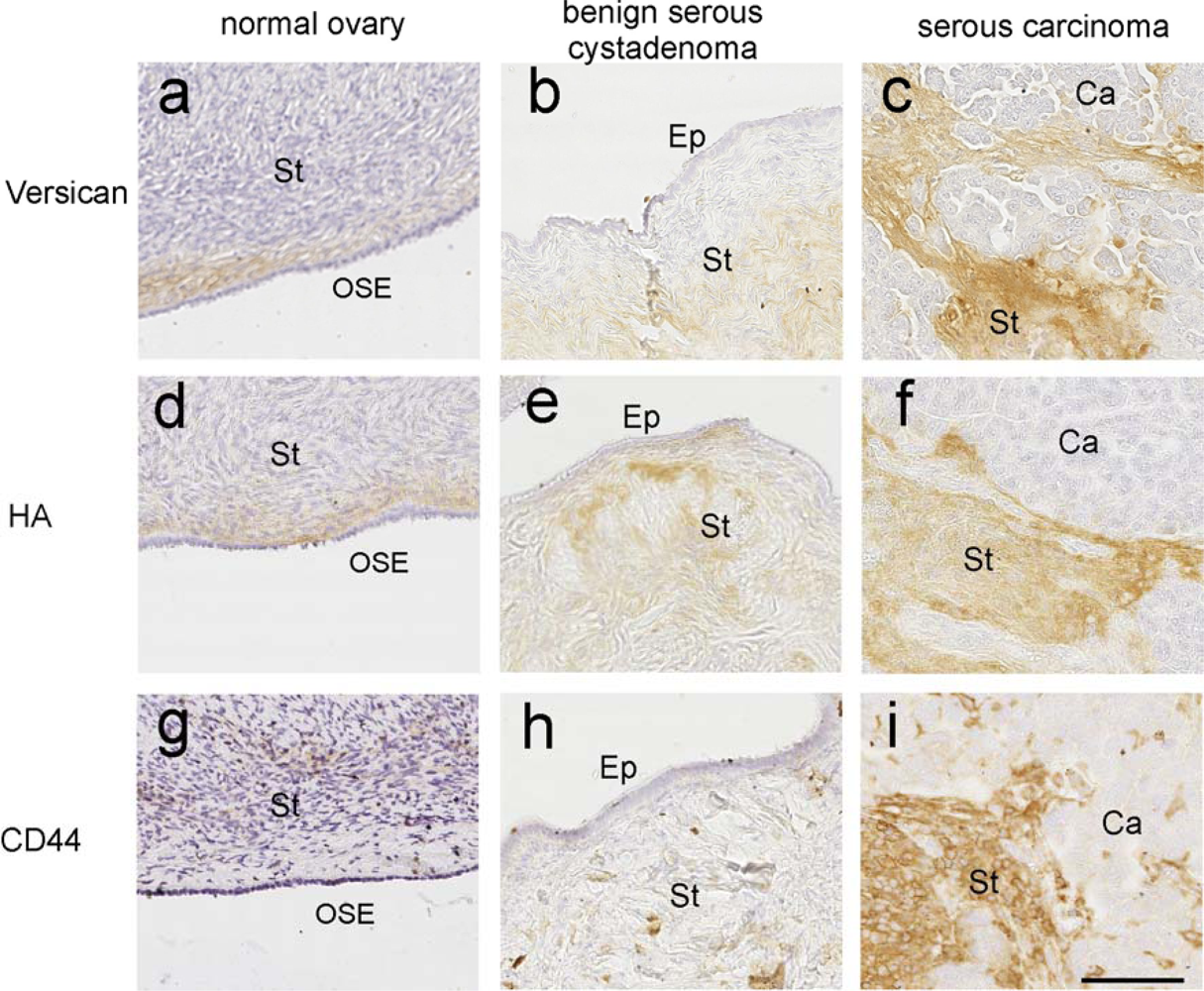
3.1. Role of HA in Ovarian Cancer
3.2. CD44—A Key Receptor for HA
3.3. The Role of CD44 and Its Interactions with HA in Cancer
3.4. Versican—An Interacting Partner of HA
3.5. Versican Is Associated with Poor Outcome
3.6. Role of Versican and HA in Promoting Cancer Cell Motility and Invasion
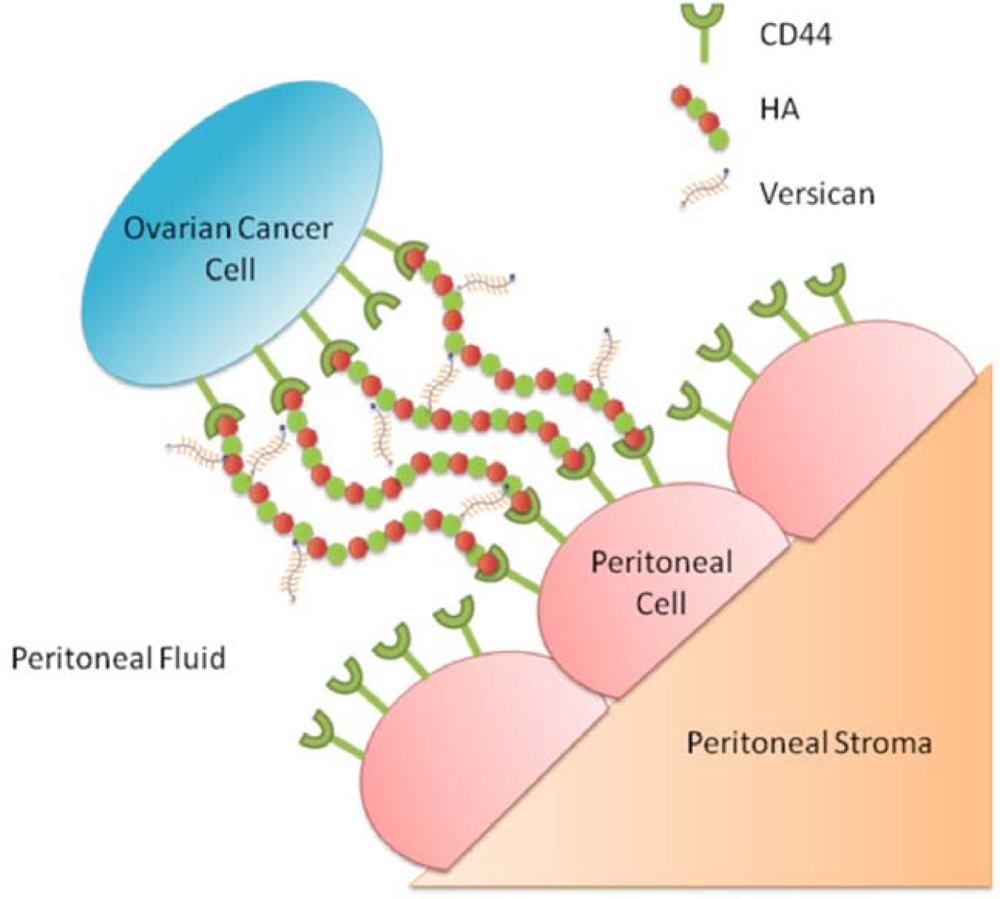
3.7. Involvement of HA and CD44 in the Adhesion of Ovarian Cancer Cells to Peritoneal Cells
4. Cancer Therapies Targeting Extracellular Components
4.1. HA Therapies
4.2. CD44 Therapies
4.3. Versican Therapies
5. Conclusions
Acknowledgments
Abbreviations:
| ECM | extracellular matrix |
| HA | hyaluronan |
| HAS | hyaluronan synthase |
| PEI | ployethyenimine |
| SMCs | smooth muscle cells |
References
- WHO World Cancer Report, Stewart, BW; Kleihues, P (Eds.) IARC Press: Lyon, France, 2003.
- Jemal, A; Siegel, R; Ward, E; Hao, Y; Xu, J; Thun, MJ. Cancer Statistics, 2009. CA Cancer J Clin 2009, 59(4), 225–249. [Google Scholar]
- Amadori, D; Sansoni, E; Amadori, A. Ovarian cancer: natural history and metastatic pattern. Front. Biosci 1997, 2, g8–g10. [Google Scholar]
- Rubin, SC; Randall, TC; Armstrong, KA; Chi, DS; Hoskins, WJ. Ten-year follow-up of ovarian cancer patients after second-look laparotomy with negative findings. Obstet Gynecol 1999, 93(1), 21–24. [Google Scholar]
- Freedman, RS; Deavers, M; Liu, J; Wang, E. Peritoneal inflammation—A microenvironment for Epithelial Ovarian Cancer (EOC). J Transl Med 2004, 2(1), 23. [Google Scholar]
- Gardner, MJ; Catterall, JB; Jones, LM; Turner, GA. Human ovarian tumour cells can bind hyaluronic acid via membrane CD44: a possible step in peritoneal metastasis. Clin Exp Metastasis 1996, 14(4), 325–334. [Google Scholar]
- Strobel, T; Cannistra, SA. Beta1-integrins partly mediate binding of ovarian cancer cells to peritoneal mesothelium in vitro. Gynecol Oncol 1999, 73(3), 362–367. [Google Scholar]
- Said, NA; Najwer, I; Socha, MJ; Fulton, DJ; Mok, SC; Motamed, K. SPARC inhibits LPA-mediated mesothelial-ovarian cancer cell crosstalk. Neoplasia 2007, 9(1), 23–35. [Google Scholar]
- Heyman, L; Kellouche, S; Fernandes, J; Dutoit, S; Poulain, L; Carreiras, F. Vitronectin and its receptors partly mediate adhesion of ovarian cancer cells to peritoneal mesothelium in vitro. Tumour Biol 2008, 29(4), 231–244. [Google Scholar]
- Kenny, HA; Kaur, S; Coussens, LM; Lengyel, E. The initial steps of ovarian cancer cell metastasis are mediated by MMP-2 cleavage of vitronectin and fibronectin. J Clin Invest 2008, 118(4), 1367–1379. [Google Scholar]
- Ween, MP; Lokman, NA; Hoffmann, P; Rodgers, RJ; Ricciardelli, C; Oehler, MK. Transforming growth factor beta-induced protein (TGFBIp) secreted by peritoneal cells increases the metastatic potential of ovarian cancer cells. Int J Cancer 2010. [Google Scholar] [CrossRef]
- Jones, LM; Gardner, MJ; Catterall, JB; Turner, GA. Hyaluronic acid secreted by mesothelial cells: a natural barrier to ovarian cancer cell adhesion. Clin Exp Metastasis 1995, 13(5), 373–380. [Google Scholar]
- Lessan, K; Aguiar, DJ; Oegema, T; Siebenson, L; Skubitz, AP. CD44 and beta1 integrin mediate ovarian carcinoma cell adhesion to peritoneal mesothelial cells. Am J Pathol 1999, 154(5), 1525–1537. [Google Scholar]
- Carpenter, PM; Dao, AV. The role of hyaluronan in mesothelium-induced motility of ovarian carcinoma cells. Anticancer Res 2003, 23(5A), 3985–3990. [Google Scholar]
- Zigrino, P; Loffek, S; Mauch, C. Tumor-stroma interactions: their role in the control of tumor cell invasion. Biochimie 2005, 87(3–4), 321–328. [Google Scholar]
- Liotta, LA; Kohn, EC. The microenvironment of the tumour-host interface. Nature 2001, 411(6835), 375–379. [Google Scholar]
- Ricciardelli, C; Rodgers, RJ. Extracellular matrix of ovarian tumors. Semin Reprod Med 2006, 24(4), 270–282. [Google Scholar]
- Catterall, JB; Gardner, MJ; Jones, LM; Turner, GA. Binding of ovarian cancer cells to immobilized hyaluronic acid. Glycoconj J 1997, 14(7), 867–869. [Google Scholar]
- Yeo, TK; Nagy, JA; Yeo, KT; Dvorak, HF; Toole, BP. Increased hyaluronan at sites of attachment to mesentery by CD44-positive mouse ovarian and breast tumor cells. Am J Pathol 1996, 148(6), 1733–1740. [Google Scholar]
- Anttila, MA; Tammi, RH; Tammi, MI; Syrjanen, KJ; Saarikoski, SV; Kosma, VM. High levels of stromal hyaluronan predict poor disease outcome in epithelial ovarian cancer. Cancer Res 2000, 60(1), 150–155. [Google Scholar]
- Bourguignon, LY; Zhu, H; Zhou, B; Diedrich, F; Singleton, PA; Hung, MC. Hyaluronan promotes CD44v3-Vav2 interaction with Grb2-p185(HER2) and induces Rac1 and Ras signaling during ovarian tumor cell migration and growth. J Biol Chem 2001, 276(52), 48679–48692. [Google Scholar]
- Jojovic, M; Delpech, B; Prehm, P; Schumacher, U. Expression of hyaluronate and hyaluronate synthase in human primary tumours and their metastases in scid mice. Cancer Lett 2002, 188(1–2), 181–189. [Google Scholar]
- Hiltunen, EL; Anttila, M; Kultti, A; Ropponen, K; Penttinen, J; Yliskoski, M; Kuronen, AT; Juhola, M; Tammi, R; Tammi, M; Kosma, VM. Elevated hyaluronan concentration without hyaluronidase activation in malignant epithelial ovarian tumors. Cancer Res 2002, 62(22), 6410–6413. [Google Scholar]
- Yabushita, H; Noguchi, M; Kishida, T; Fusano, K; Noguchi, Y; Itano, N; Kimata, K. Hyaluronan synthase expression in ovarian cancer. Oncol Rep 2004, 12(4), 739–743. [Google Scholar]
- Tzuman, YC; Sapoznik, S; Granot, D; Nevo, N; Neeman, M. Peritoneal adhesion and angiogenesis in ovarian carcinoma are inversely regulated by hyaluronan: the role of gonadotropins. Neoplasia 2010, 12(1), 51–60. [Google Scholar]
- Ween, MP; Hummitzsch, K; Rodgers, RJ; Oehler, MK; Ricciardelli, C. Versican induces a pro-metastatic ovarian cancer cell behavior which can be inhibited by small hyaluronan oligosaccharides. Clin Exp Metastasis 2010. [Google Scholar] [CrossRef]
- Tamakoshi, K; Kikkawa, F; Maeda, O; Suganuma, N; Yamagata, S; Yamagata, T; Tomoda, Y. Hyaluronidase activity in gynaecological cancer tissues with different metastatic forms. Br J Cancer 1997, 75(12), 1807–1811. [Google Scholar]
- Nykopp, TK; Rilla, K; Sironen, R; Tammi, MI; Tammi, RH; Hamalainen, K; Heikkinen, AM; Komulainen, M; Kosma, VM; Anttila, M. Expression of hyaluronan synthases (HAS1-3) and hyaluronidases (HYAL1-2) in serous ovarian carcinomas: inverse correlation between HYAL1 and hyaluronan content. BMC Cancer 2009, 9, 143. [Google Scholar]
- Cannistra, SA; Kansas, GS; Niloff, J; DeFranzo, B; Kim, Y; Ottensmeier, C. Binding of ovarian cancer cells to peritoneal mesothelium in vitro is partly mediated by CD44H. Cancer Res 1993, 53(16), 3830–3838. [Google Scholar]
- Gardner, MJ; Jones, LM; Catterall, JB; Turner, GA. Expression of cell adhesion molecules on ovarian tumour cell lines and mesothelial cells, in relation to ovarian cancer metastasis. Cancer Lett 1995, 91(2), 229–234. [Google Scholar]
- Uhl-Steidl, M; Muller-Holzner, E; Zeimet, AG; Adolf, GR; Daxenbichler, G; Marth, C; Dapunt, O. Prognostic value of CD44 splice variant expression in ovarian cancer. Oncology 1995, 52(5), 400–406. [Google Scholar]
- Strobel, T; Swanson, L; Cannistra, SA. In vivo inhibition of CD44 limits intra-abdominal spread of a human ovarian cancer xenograft in nude mice: a novel role for CD44 in the process of peritoneal implantation. Cancer Res 1997, 57(7), 1228–1232. [Google Scholar]
- Bourguignon, LY; Zhu, H; Chu, A; Iida, N; Zhang, L; Hung, MC. Interaction between the adhesion receptor, CD44, and the oncogene product, p185HER2, promotes human ovarian tumor cell activation. J Biol Chem 1997, 272(44), 27913–27918. [Google Scholar]
- Kayastha, S; Freedman, AN; Piver, MS; Mukkamalla, J; Romero-Guittierez, M; Werness, BA. Expression of the hyaluronan receptor, CD44S, in epithelial ovarian cancer is an independent predictor of survival. Clin Cancer Res 1999, 5(5), 1073–1076. [Google Scholar]
- Saegusa, M; Machida, D; Hashimura, M; Okayasu, I. CD44 expression in benign, premalignant, and malignant ovarian neoplasms: relation to tumour development and progression. J Pathol 1999, 189(3), 326–337. [Google Scholar]
- Casey, RC; Skubitz, AP. CD44 and beta1 integrins mediate ovarian carcinoma cell migration toward extracellular matrix proteins. Clin Exp Metastasis 2000, 18(1), 67–75. [Google Scholar]
- Bourguignon, LY; Zhu, H; Shao, L; Chen, YW. CD44 interaction with c-Src kinase promotes cortactin-mediated cytoskeleton function and hyaluronic acid-dependent ovarian tumor cell migration. J Biol Chem 2001, 276(10), 7327–7336. [Google Scholar]
- Afify, AM; Ferguson, AW; Davila, RM; Werness, BA. Expression of CD44S and CD44v5 is more common in stage III than in stage I serous ovarian carcinomas. Appl Immunohistochem Mol Morphol 2001, 9(4), 309–314. [Google Scholar]
- Ross, JS; Sheehan, CE; Williams, SS; Malfetano, JH; Szyfelbein, WM; Kallakury, BV. Decreased CD44 standard form expression correlates with prognostic variables in ovarian carcinomas. Am J Clin Pathol 2001, 116(1), 122–128. [Google Scholar]
- Rodriguez-Rodriguez, L; Sancho-Torres, I; Mesonero, C; Gibbon, DG; Shih, WJ; Zotalis, G. The CD44 receptor is a molecular predictor of survival in ovarian cancer. Med Oncol 2003, 20(3), 255–263. [Google Scholar]
- Sillanpaa, S; Anttila, MA; Voutilainen, K; Tammi, RH; Tammi, MI; Saarikoski, SV; Kosma, VM. CD44 expression indicates favorable prognosis in epithelial ovarian cancer. Clin Cancer Res 2003, 9(14), 5318–5324. [Google Scholar]
- Bourguignon, LY; Gilad, E; Rothman, K; Peyrollier, K. Hyaluronan-CD44 interaction with IQGAP1 promotes Cdc42 and ERK signaling, leading to actin binding, Elk-1/estrogen receptor transcriptional activation, and ovarian cancer progression. J Biol Chem 2005, 280(12), 11961–11972. [Google Scholar]
- Bourguignon, LY; Peyrollier, K; Gilad, E; Brightman, A. Hyaluronan-CD44 interaction with neural Wiskott-Aldrich syndrome protein (N-WASP) promotes actin polymerization and ErbB2 activation leading to beta-catenin nuclear translocation, transcriptional up-regulation, and cell migration in ovarian tumor cells. J Biol Chem 2007, 282(2), 1265–1280. [Google Scholar]
- Bourguignon, LY; Gilad, E; Peyrollier, K. Heregulin-mediated ErbB2-ERK signaling activates hyaluronan synthases leading to CD44-dependent ovarian tumor cell growth and migration. J Biol Chem 2007, 282(27), 19426–19441. [Google Scholar]
- Chen, H; Hao, J; Wang, L; Li, Y. Coexpression of invasive markers (uPA, CD44) and multiple drug-resistance proteins (MDR1, MRP2) is correlated with epithelial ovarian cancer progression. Br J Cancer 2009, 101(3), 432–440. [Google Scholar]
- Voutilainen, K; Anttila, M; Sillanpaa, S; Tammi, R; Tammi, M; Saarikoski, S; Kosma, VM. Versican in epithelial ovarian cancer: Relation to hyaluronan, clinicopathologic factors and prognosis. Int J Cancer 2003, 107(3), 359–364. [Google Scholar]
- Casey, RC; Oegema, TR, Jr. ; Skubitz, K.M.; Pambuccian, S.E.; Grindle, S.M.; Skubitz, A.P. Cell membrane glycosylation mediates the adhesion, migration, and invasion of ovarian carcinoma cells. Clin Exp Metastasis 2003, 20(2), 143–152. [Google Scholar]
- Lancaster, JM; Dressman, HK; Clarke, JP; Sayer, RA; Martino, MA; Cragun, JM; Henriott, AH; Gray, J; Sutphen, R; Elahi, A; Whitaker, RS; West, M; Marks, JR; Nevins, JR; Berchuck, A. Identification of genes associated with ovarian cancer metastasis using microarray expression analysis. Int J Gynecol Cancer 2006, 16(5), 1733–1745. [Google Scholar]
- Kusumoto, T; Kodama, J; Seki, N; Nakamura, K; Hongo, A; Hiramatsu, Y. Clinical significance of syndecan-1 and versican expression in human epithelial ovarian cancer. Oncol Rep 2010, 23(4), 917–925. [Google Scholar]
- Ghosh, S; Albitar, L; Lebaron, R; Welch, WR; Samimi, G; Birrer, MJ; Berkowitz, RS; Mok, SC. Up-regulation of stromal versican expression in advanced stage serous ovarian cancer. Gynecol. Oncol 2010, 119, 114–120. [Google Scholar]
- Knudson, CB; Knudson, W. Hyaluronan-binding proteins in development, tissue homeostasis, and disease. FASEB J 1993, 7(13), 1233–1241. [Google Scholar]
- Weigel, PH; Hascall, VC; Tammi, M. Hyaluronan synthases. J Biol Chem 1997, 272(22), 13997–14000. [Google Scholar]
- Tammi, MI; Day, AJ; Turley, EA. Hyaluronan and homeostasis: a balancing act. J Biol Chem 2002, 277(7), 4581–4584. [Google Scholar]
- Toole, BP. Hyaluronan: from extracellular glue to pericellular cue. Nat Rev Cancer 2004, 4(7), 528–539. [Google Scholar]
- Boregowda, RK; Appaiah, HN; Siddaiah, M; Kumarswamy, SB; Sunila, S; Thimmaiah, KN; Mortha, K; Toole, B; Banerjee, S. Expression of hyaluronan in human tumor progression. J. Carcinog 2006, 5, 2. [Google Scholar]
- Auvinen, P; Tammi, R; Parkkinen, J; Tammi, M; Agren, U; Johansson, R; Hirvikoski, P; Eskelinen, M; Kosma, VM. Hyaluronan in peritumoral stroma and malignant cells associates with breast cancer spreading and predicts survival. Am J Pathol 2000, 156(2), 529–536. [Google Scholar]
- Ropponen, K; Tammi, M; Parkkinen, J; Eskelinen, M; Tammi, R; Lipponen, P; Agren, U; Alhava, E; Kosma, VM. Tumor cell-associated hyaluronan as an unfavorable prognostic factor in colorectal cancer. Cancer Res 1998, 58(2), 342–347. [Google Scholar]
- Lipponen, P; Aaltomaa, S; Tammi, R; Tammi, M; Agren, U; Kosma, VM. High stromal hyaluronan level is associated with poor differentiation and metastasis in prostate cancer. Eur J Cancer 2001, 37(7), 849–856. [Google Scholar]
- Pirinen, R; Tammi, R; Tammi, M; Hirvikoski, P; Parkkinen, JJ; Johansson, R; Bohm, J; Hollmen, S; Kosma, VM. Prognostic value of hyaluronan expression in non-small-cell lung cancer: Increased stromal expression indicates unfavorable outcome in patients with adenocarcinoma. Int J Cancer 2001, 95(1), 12–17. [Google Scholar]
- Posey, JT; Soloway, MS; Ekici, S; Sofer, M; Civantos, F; Duncan, RC; Lokeshwar, VB. Evaluation of the prognostic potential of hyaluronic acid and hyaluronidase (HYAL1) for prostate cancer. Cancer Res 2003, 63(10), 2638–2644. [Google Scholar]
- Aruffo, A; Stamenkovic, I; Melnick, M; Underhill, CB; Seed, B. CD44 is the principal cell surface receptor for hyaluronate. Cell 1990, 61(7), 1303–1313. [Google Scholar]
- Bourguignon, LY; Zhu, D; Zhu, H. CD44 isoform-cytoskeleton interaction in oncogenic signaling and tumor progression. Front. Biosci 1998, 3, d637–d649. [Google Scholar]
- Screaton, GR; Bell, MV; Jackson, DG; Cornelis, FB; Gerth, U; Bell, JI. Genomic structure of DNA encoding the lymphocyte homing receptor CD44 reveals at least 12 alternatively spliced exons. Proc Natl Acad Sci USA 1992, 89(24), 12160–12164. [Google Scholar]
- Naor, D; Nedvetzki, S; Golan, I; Melnik, L; Faitelson, Y. CD44 in cancer. Crit Rev Clin Lab Sci 2002, 39(6), 527–579. [Google Scholar]
- Stickeler, E; Runnebaum, IB; Mobus, VJ; Kieback, DG; Kreienberg, R. Expression of CD44 standard and variant isoforms v5, v6 and v7 in human ovarian cancer cell lines. Anticancer Res 1997, 17(3C), 1871–1876. [Google Scholar]
- Zhu, D; Bourguignon, LY. Interaction between CD44 and the repeat domain of ankyrin promotes hyaluronic acid-mediated ovarian tumor cell migration. J Cell Physiol 2000, 183(2), 182–195. [Google Scholar]
- Bourguignon, LY; Zhu, H; Shao, L; Chen, YW. Ankyrin-Tiam1 interaction promotes Rac1 signaling and metastatic breast tumor cell invasion and migration. J Cell Biol 2000, 150(1), 177–191. [Google Scholar]
- Kim, Y; Lee, YS; Choe, J; Lee, H; Kim, YM; Jeoung, D. CD44-epidermal growth factor receptor interaction mediates hyaluronic acid-promoted cell motility by activating protein kinase C signaling involving Akt, Rac1, Phox, reactive oxygen species, focal adhesion kinase, and MMP-2. J Biol Chem 2008, 283(33), 22513–22528. [Google Scholar]
- Buess, M; Rajski, M; Vogel-Durrer, BM; Herrmann, R; Rochlitz, C. Tumor-endothelial interaction links the CD44(+)/CD24(−) phenotype with poor prognosis in early-stage breast cancer. Neoplasia 2009, 11(10), 987–1002. [Google Scholar]
- Okayama, H; Kumamoto, K; Saitou, K; Hayase, S; Kofunato, Y; Sato, Y; Miyamoto, K; Nakamura, I; Ohki, S; Sekikawa, K; Takenoshita, S. CD44v6, MMP-7 and nuclear Cdx2 are significant biomarkers for prediction of lymph node metastasis in primary gastric cancer. Oncol Rep 2009, 22(4), 745–755. [Google Scholar]
- Kawano, T; Nakamura, Y; Yanoma, S; Kubota, A; Furukawa, M; Miyagi, Y; Tsukuda, M. Expression of E-cadherin, and CD44s and CD44v6 and its association with prognosis in head and neck cancer. Auris Nasus Larynx 2004, 31(1), 35–41. [Google Scholar]
- Lee, SM; Lee, KE; Chang, HJ; Choi, MY; Cho, MS; Min, SK; Lee, HK; Mun, YC; Nam, EM; Seong, CM; Lee, SN. Prognostic significance of CD44s expression in biliary tract cancers. Ann Surg Oncol 2008, 15(4), 1155–1160. [Google Scholar]
- Gu, H; Shang, P; Zhou, C. Expression of CD44v6 and E-cadherin in prostate carcinoma and metastasis of prostate carcinoma. Zhonghua Nan Ke Xue 2004, 10(1). [Google Scholar]
- Cannistra, SA; Abu-Jawdeh, G; Niloff, J; Strobel, T; Swanson, L; Andersen, J; Ottensmeier, C. CD44 variant expression is a common feature of epithelial ovarian cancer: lack of association with standard prognostic factors. J Clin Oncol 1995, 13(8), 1912–1921. [Google Scholar]
- Zagorianakou, N; Stefanou, D; Makrydimas, G; Zagorianakou, P; Briasoulis, E; Karavasilis, B; Agnantis, NJ. CD44s expression, in benign, borderline and malignant tumors of ovarian surface epithelium. Correlation with p53, steroid receptor status, proliferative indices (PCNA, MIB1) and survival. Anticancer Res 2004, 24(3a), 1665–1670. [Google Scholar]
- Du Cros, DL; Lebaron, RG; Couchman, JR. Association of versican with dermal matrices and its potential role in hair follicle development and cycling. J Invest Dermatol 1995, 105(3), 426–431. [Google Scholar]
- Suwiwat, S; Ricciardelli, C; Tammi, R; Tammi, M; Auvinen, P; Kosma, VM; LeBaron, RG; Raymond, WA; Tilley, WD; Horsfall, DJ. Expression of extracellular matrix components versican, chondroitin sulfate, tenascin, and hyaluronan, and their association with disease outcome in node-negative breast cancer. Clin Cancer Res 2004, 10(7), 2491–2498. [Google Scholar]
- Speiser, P; Wanner, C; Breitenecker, G; Kohlberger, P; Kainz, C. CD-44 is not involved in the metastatic spread of ovarian cancer in vivo. Anticancer Res 1995, 15(6B), 2767–2769. [Google Scholar]
- Sanchez Lockhart, M; Hajos, SE; Basilio, FM; Mongini, C; Alvarez, E. Splice variant expression of CD44 in patients with breast and ovarian cancer. Oncol Rep 2001, 8(1), 145–151. [Google Scholar]
- Ricciardelli, C; Sakko, AJ; Ween, MP; Russell, DL; Horsfall, DJ. The biological role and regulation of versican levels in cancer. Cancer Metastasis Rev 2009, 28(1–2), 233–245. [Google Scholar]
- LeBaron, RG. Versican. Perspect Dev Neurobiol 1996, 3(4), 261–271. [Google Scholar]
- Zimmermann, DR; Ruoslahti, E. Multiple domains of the large fibroblast proteoglycan, versican. EMBO J 1989, 8(10), 2975–2981. [Google Scholar]
- Wu, YJ; La Pierre, DP; Wu, J; Yee, AJ; Yang, BB. The interaction of versican with its binding partners. Cell Res 2005, 15(7), 483–494. [Google Scholar]
- Naso, MF; Zimmermann, DR; Iozzo, RV. Characterization of the complete genomic structure of the human versican gene and functional analysis of its promoter. J BiolChem 1994, 269(52), 32999–33008. [Google Scholar]
- Kischel, P; Waltregny, D; Dumont, B; Turtoi, A; Greffe, Y; Kirsch, S; de Pauw, E; Castronovo, V. Versican overexpression in human breast cancer lesions: known and new isoforms for stromal tumor targeting. Int J Cancer 2010, 126(3), 640–650. [Google Scholar]
- Sakko, AJ; Ricciardelli, C; Mayne, K; Tilley, WD; LeBaron, RG; Horsfall, DJ. Versican accumulation in human prostatic fibroblast cultures is enhanced by prostate cancer cell-derived transforming growth factor beta1. Cancer Res 2001, 61(3), 926–930. [Google Scholar]
- Ricciardelli, C; Brooks, JH; Suwiwat, S; Sakko, AJ; Mayne, K; Raymond, WA; Seshadri, R; LeBaron, RG; Horsfall, DJ. Regulation of stromal versican expression by breast cancer cells and importance to relapse-free survival in patients with node-negative primary breast cancer. Clin Cancer Res 2002, 8(4), 1054–1060. [Google Scholar]
- Ghosh, S; Albitar, L; LeBaron, R; Welch, WR; Samimi, G; Birrer, MJ; Berkowitz, RS; Mok, SC. Up-regulation of stromal versican expression in advanced stage serous ovarian cancer. Gynecol Oncol 2010, 119(1), 114–120. [Google Scholar]
- Knudson, W; Bartnik, E; Knudson, CB. Assembly of pericellular matrices by COS-7 cells transfected with CD44 lymphocyte-homing receptor genes. Proc Natl Acad Sci USA 1993, 90(9), 4003–4007. [Google Scholar]
- Evanko, SP; Angello, JC; Wight, TN. Formation of hyaluronan- and versican-rich pericellular matrix is required for proliferation and migration of vascular smooth muscle cells. Arterioscler Thromb Vasc Biol 1999, 19(4), 1004–1013. [Google Scholar]
- Ricciardelli, C; Russell, DL; Ween, MP; Mayne, K; Suwiwat, S; Byers, S; Marshall, VR; Tilley, WD; Horsfall, DJ. Formation of hyaluronan- and versican-rich pericellular matrix by prostate cancer cells promotes cell motility. J Biol Chem 2007, 282(14), 10814–10825. [Google Scholar]
- Casey, MJ; Bewtra, C; Hoehne, LL; Tatpati, AD; Lynch, HT; Watson, P. Histology of prophylactically removed ovaries from BRCA1 and BRCA2 mutation carriers compared with noncarriers in hereditary breast ovarian cancer syndrome kindreds. Gynecol Oncol 2000, 78 3 Pt 1, 278–287. [Google Scholar]
- Casey, RC; Koch, KA; Oegema, TR, Jr; Skubitz, KM; Pambuccian, SE; Grindle, SM; Skubitz, AP. Establishment of an in vitro assay to measure the invasion of ovarian carcinoma cells through mesothelial cell monolayers. Clin Exp Metastasis 2003, 20(4), 343–356. [Google Scholar]
- Lee, H; Lee, K; Park, TG. Hyaluronic acid-paclitaxel conjugate micelles: synthesis, characterization, and antitumor activity. Bioconjug Chem 2008, 19(6), 1319–1325. [Google Scholar]
- Cohen, MS; Cai, S; Xie, Y; Forrest, ML. A novel intralymphatic nanocarrier delivery system for cisplatin therapy in breast cancer with improved tumor efficacy and lower systemic toxicity in vivo. Am J Surg 2009, 198(6), 781–786. [Google Scholar]
- Banzato, A; Bobisse, S; Rondina, M; Renier, D; Bettella, F; Esposito, G; Quintieri, L; Melendez-Alafort, L; Mazzi, U; Zanovello, P; Rosato, A. A paclitaxel-hyaluronan bioconjugate targeting ovarian cancer affords a potent in vivo therapeutic activity. Clin Cancer Res 2008, 14(11), 3598–3606. [Google Scholar]
- Auzenne, E; Ghosh, SC; Khodadadian, M; Rivera, B; Farquhar, D; Price, RE; Ravoori, M; Kundra, V; Freedman, RS; Klostergaard, J. Hyaluronic acid-paclitaxel: antitumor efficacy against CD44(+) human ovarian carcinoma xenografts. Neoplasia 2007, 9(6), 479–486. [Google Scholar]
- Sun, X; Ma, P; Cao, X; Ning, L; Tian, Y; Ren, C. Positive hyaluronan/PEI/DNA complexes as a target-specific intracellular delivery to malignant breast cancer. Drug Deliv 2009, 16(7), 357–362. [Google Scholar]
- Jiang, G; Park, K; Kim, J; Kim, KS; Hahn, SK. Target specific intracellular delivery of siRNA/PEI-HA complex by receptor mediated endocytosis. Mol Pharm 2009, 6(3), 727–737. [Google Scholar]
- Chono, S; Li, SD; Conwell, CC; Huang, L. An efficient and low immunostimulatory nanoparticle formulation for systemic siRNA delivery to the tumor. J Control Release 2008, 131(1), 64–69. [Google Scholar]
- Baumgartner, G; Gomar-Hoss, C; Sakr, L; Ulsperger, E; Wogritsch, C. The impact of extracellular matrix on the chemoresistance of solid tumors—experimental and clinical results of hyaluronidase as additive to cytostatic chemotherapy. Cancer Lett 1998, 131(1), 85–99. [Google Scholar]
- Maier, U; Baumgartner, G. Metaphylactic effect of mitomycin C with and without hyaluronidase after transurethral resection of bladder cancer: randomized trial. J Urol 1989, 141(3), 529–530. [Google Scholar]
- Thompson, CB; Shepard, HM; O’Connor, PM; Kadhim, S; Jiang, P; Osgood, RJ; Bookbinder, LH; Li, X; Sugarman, BJ; Connor, RJ; Nadjsombati, S; Frost, GI. Enzymatic depletion of tumor hyaluronan induces antitumor responses in preclinical animal models. Mol Cancer Ther 2010, 9(11), 3052–3064. [Google Scholar]
- Guedan, S; Rojas, JJ; Gros, A; Mercade, E; Cascallo, M; Alemany, R. Hyaluronidase expression by an oncolytic adenovirus enhances its intratumoral spread and suppresses tumor growth. Mol Ther 2010, 18(7), 1275–1283. [Google Scholar]
- Homepage of Halozyme. Available online: http://www.halozyme.com/products_oncology.php (accessed on 25 January 2011).
- Lee, SO; Jeong, YJ; Im, HG; Kim, CH; Chang, YC; Lee, IS. Silibinin suppresses PMAinduced MMP-9 expression by blocking the AP-1 activation via MAPK signaling pathways in MCF-7 human breast carcinoma cells. Biochem Biophys Res Commun 2007, 354(1), 165–171. [Google Scholar]
- Mateen, S; Tyagi, A; Agarwal, C; Singh, RP; Agarwal, R. Silibinin inhibits human nonsmall cell lung cancer cell growth through cell-cycle arrest by modulating expression and function of key cell-cycle regulators. Mol Carcinog 2010, 49(3), 247–258. [Google Scholar]
- Kaur, M; Velmurugan, B; Tyagi, A; Deep, G; Katiyar, S; Agarwal, C; Agarwal, R. Silibinin suppresses growth and induces apoptotic death of human colorectal carcinoma LoVo cells in culture and tumor xenograft. Mol Cancer Ther 2009, 8(8), 2366–2374. [Google Scholar]
- Handorean, AM; Yang, K; Robbins, EW; Flaig, TW; Iczkowski, KA. Silibinin suppresses CD44 expression in prostate cancer cells. Am J Transl Res 2009, 1(1), 80–86. [Google Scholar]
- Wallach-Dayan, SB; Rubinstein, AM; Hand, C; Breuer, R; Naor, D. DNA vaccination with CD44 variant isoform reduces mammary tumor local growth and lung metastasis. Mol Cancer Ther 2008, 7(6), 1615–1623. [Google Scholar]
- Hong, SP; Wen, J; Bang, S; Park, S; Song, SY. CD44-positive cells are responsible for gemcitabine resistance in pancreatic cancer cells. Int J Cancer 2009, 125(10), 2323–2331. [Google Scholar]
- Jin, L; Hope, KJ; Zhai, Q; Smadja-Joffe, F; Dick, JE. Targeting of CD44 eradicates human acute myeloid leukemic stem cells. Nat Med 2006, 12(10), 1167–1174. [Google Scholar]
- Subramaniam, V; Vincent, IR; Gilakjan, M; Jothy, S. Suppression of human colon cancer tumors in nude mice by siRNA CD44 gene therapy. Exp Mol Pathol 2007, 83(3), 332–340. [Google Scholar]
- Xie, Z; Choong, PF; Poon, LF; Zhou, J; Khng, J; Jasinghe, VJ; Palaniyandi, S; Chen, CS. Inhibition of CD44 expression in hepatocellular carcinoma cells enhances apoptosis, chemosensitivity, and reduces tumorigenesis and invasion. Cancer Chemother Pharmacol 2008, 62(6), 949–957. [Google Scholar]
- Li, CZ; Liu, B; Wen, ZQ; Li, HY. Inhibition of CD44 expression by small interfering RNA to suppress the growth and metastasis of ovarian cancer cells in vitro and in vivo. Folia Biol. (Praha) 2008, 54(6), 180–186. [Google Scholar]
- Wiranowska, M; Ladd, S; Moscinski, LC; Hill, B; Haller, E; Mikecz, K; Plaas, A. Modulation of Hyaluronan production by CD44 positive Glioma cells. Int J Cancer 2010, 127(3), 532–542. [Google Scholar]
- Yoshida, M; Yasuda, T; Hiramitsu, T; Ito, H; Nakamura, T. Induction of apoptosis by anti-CD44 antibody in human chondrosarcoma cell line SW1353. Biomed Res 2008, 29(1), 47–52. [Google Scholar]
- Marangoni, E; Lecomte, N; Durand, L; de Pinieux, G; Decaudin, D; Chomienne, C; Smadja-Joffe, F; Poupon, MF. CD44 targeting reduces tumour growth and prevents post-chemotherapy relapse of human breast cancers xenografts. Br J Cancer 2009, 100(6), 918–922. [Google Scholar]
- Afify, A; Purnell, P; Nguyen, L. Role of CD44s and CD44v6 on human breast cancer cell adhesion, migration, and invasion. Exp Mol Pathol 2009, 86(2), 95–100. [Google Scholar]
- Guo, Y; Ma, J; Wang, J; Che, X; Narula, J; Bigby, M; Wu, M; Sy, MS. Inhibition of human melanoma growth and metastasis in vivo by anti-CD44 monoclonal antibody. Cancer Res 1994, 54(6), 1561–1565. [Google Scholar]
- Herrera-Gayol, A; Jothy, S. CD44 modulates Hs578T human breast cancer cell adhesion, migration, and invasiveness. Exp Mol Pathol 1999, 66(1), 99–108. [Google Scholar]
- Draffin, JE; McFarlane, S; Hill, A; Johnston, PG; Waugh, DJ. CD44 potentiates the adherence of metastatic prostate and breast cancer cells to bone marrow endothelial cells. Cancer Res 2004, 64(16), 5702–5711. [Google Scholar]
- Orian-Rousseau, V. CD44, a therapeutic target for metastasising tumours. Eur J Cancer 2010, 46(7), 1271–1277. [Google Scholar]
- Schonherr, E; Kinsella, MG; Wight, TN. Genistein selectively inhibits platelet-derived growth factor-stimulated versican biosynthesis in monkey arterial smooth muscle cells. Arch Biochem Biophys 1997, 339(2), 353–361. [Google Scholar]
- Syrokou, A; Tzanakakis, GN; Hjerpe, A; Karamanos, NK. Proteoglycans in human malignant mesothelioma. Stimulation of their synthesis induced by epidermal, insulin and platelet-derived growth factors involves receptors with tyrosine kinase activity. Biochimie 1999, 81(7), 733–744. [Google Scholar]
- Potter-Perigo, S; Baker, C; Tsoi, C; Braun, KR; Isenhath, S; Altman, GM; Altman, LC; Wight, TN. Regulation of proteoglycan synthesis by leukotriene d4 and epidermal growth factor in bronchial smooth muscle cells. Am J Respir Cell Mol Biol 2004, 30(1), 101–108. [Google Scholar]
- Burgess, JK; Oliver, BG; Poniris, MH; Ge, Q; Boustany, S; Cox, N; Moir, LM; Johnson, PR; Black, JL. A phosphodiesterase 4 inhibitor inhibits matrix protein deposition in airways in vitro. J Allergy Clin Immunol 2006, 118(3), 649–657. [Google Scholar]
- Todorova, L; Gurcan, E; Miller-Larsson, A; Westergren-Thorsson, G. Lung fibroblast proteoglycan production induced by serum is inhibited by budesonide and formoterol. Am J Respir Cell Mol Biol 2006, 34(1), 92–100. [Google Scholar]
- Arslan, F; Bosserhoff, AK; Nickl-Jockschat, T; Doerfelt, A; Bogdahn, U; Hau, P. The role of versican isoforms V0/V1 in glioma migration mediated by transforming growth factor-beta2. Br J Cancer 2007, 96(10), 1560–1568. [Google Scholar]
- Nakamura, JL; Haas-Kogan, DA; Pieper, RO. Glioma invasiveness responds variably to irradiation in a co-culture model. Int J Radiat Oncol Biol Phys 2007, 69(3), 880–886. [Google Scholar]
- Almholt, K; Juncker-Jensen, A; Laerum, OD; Dano, K; Johnsen, M; Lund, LR; Romer, J. Metastasis is strongly reduced by the matrix metalloproteinase inhibitor Galardin in the MMTV-PymT transgenic breast cancer model. Mol Cancer Ther 2008, 7(9), 2758–2767. [Google Scholar]
- Vankemmelbeke, MN; Jones, GC; Fowles, C; Ilic, MZ; Handley, CJ; Day, AJ; Knight, CG; Mort, JS; Buttle, DJ. Selective inhibition of ADAMTS-1, -4 and -5 by catechin gallate esters. Eur J Biochem 2003, 270(11), 2394–2403. [Google Scholar]
- Knudson, W; Knudson, CB. Assembly of a chondrocyte-like pericellular matrix on non-chondrogenic cells. Role of the cell surface hyaluronan receptors in the assembly of a pericellular matrix. J Cell Sci 1991, 99 Pt 2, 227–235. [Google Scholar]
- Zeng, C; Toole, BP; Kinney, SD; Kuo, JW; Stamenkovic, I. Inhibition of tumor growth in vivo by hyaluronan oligomers. Int J Cancer 1998, 77(3), 396–401. [Google Scholar]
- Heng, BC; Gribbon, PM; Day, AJ; Hardingham, TE. Hyaluronan binding to link module of TSG-6 and to G1 domain of aggrecan is differently regulated by pH. J Biol Chem 2008, 283(47), 32294–32301. [Google Scholar]
- Hosono, K; Nishida, Y; Knudson, W; Knudson, CB; Naruse, T; Suzuki, Y; Ishiguro, N. Hyaluronan oligosaccharides inhibit tumorigenicity of osteosarcoma cell lines MG-63 and LM-8 in vitro and in vivo via perturbation of hyaluronan-rich pericellular matrix of the cells. Am J Pathol 2007, 171(1), 274–286. [Google Scholar]
- Alaniz, L; Rizzo, M; Malvicini, M; Jaunarena, J; Avella, D; Atorrasagasti, C; Aquino, JB; Garcia, M; Matar, P; Silva, M; Mazzolini, G. Low molecular weight hyaluronan inhibits colorectal carcinoma growth by decreasing tumor cell proliferation and stimulating immune response. Cancer Lett 2009, 278(1), 9–16. [Google Scholar]
- Cui, X; Zhou, S; Xu, H; Zhao, T; Liu, A; Guo, X; Wang, F. Reversal effects of hyaluronan oligosaccharides on adriamycin resistance of K562/A02 cells. Anticancer Drugs 2009, 20(9), 800–806. [Google Scholar]
- Slomiany, MG; Dai, L; Bomar, PA; Knackstedt, TJ; Kranc, DA; Tolliver, L; Maria, BL; Toole, BP. Abrogating drug resistance in malignant peripheral nerve sheath tumors by disrupting hyaluronan-CD44 interactions with small hyaluronan oligosaccharides. Cancer Res 2009, 69(12), 4992–498. [Google Scholar]
- Slomiany, MG; Dai, L; Tolliver, LB; Grass, GD; Zeng, Y; Toole, BP. Inhibition of Functional Hyaluronan-CD44 Interactions in CD133-positive Primary Human Ovarian Carcinoma Cells by Small Hyaluronan Oligosaccharides. Clin Cancer Res 2009, 15(24), 7593–7601. [Google Scholar]

| Molecule | Effect | References |
|---|---|---|
| HA | Ovarian cancer cell bind to HA | [6,18] |
| HA is increased at metastatic sites | [19] | |
| High HA level predicts poor disease outcome | [20] | |
| HA promotes interaction with CD44v3 and HER2 to induce Rac1 and Ras signalling and increased ovarian cancer migration and proliferation | [21] | |
| HA and HAS are increased at metastatic sites | [22] | |
| HA concentration correlates with high tumor grade | [23] | |
| HA promotes motility of ovarian cancer cells | [14] | |
| HAS1 levels are associated with reduced survival | [24] | |
| HA mediates adhesion of ovarian cancer cells to the peritoneum | [25,26] | |
| HA production by mesothelial cells is increased following co-culture with ovarian cancer cells | [14] | |
| Hyaluronidase | Hyaluronidase activity is increased in metastatic gynecological cancers | [27] |
| Hyaluronidase HYAL1 is reduced in serous ovarian cancer and results in HA accumulation | [28] | |
| Hyaluronidase activation is not associated with ovarian cancer aggressiveness | [23] | |
| HA and hyaluronidase synthesis by ovarian cancer cells is increased by gonadotropins | [25] | |
| CD44 | CD44 mediates binding peritoneal mesothelium | [13,29,30] |
| Patients with CD44v positive tumors have reduced disease-free survival | [31] | |
| CD44 inhibition limits intra-abdominal spreading | [32] | |
| CD44-HER2 interactions promote ovarian cancer proliferation | [33] | |
| High levels of CD44s is an independent predictor of survival | [34] | |
| Loss of CD44v3 expression is an independent factor for poor survival. | [35] | |
| CD44 mediates ovarian cancer migration towards ECM | [36] | |
| CD44 interaction with c-src kinase promotes HA dependent ovarian cancer cell migration | [37] | |
| CD44s expression relates with tumor stage | [38] | |
| Decreased CD44s expression correlates with reduced relapse-free survival | [39] | |
| Increased expression CD44s predicts improved survival | [40] | |
| High CD44 expression is associated with a favorable prognosis | [41] | |
| CD44-HA interaction promotes Cdc42 and ERK signaling and ovarian cancer progression | [42] | |
| CD44-HA interactions promotes cell migration via HER2 activation and beta-catenin nuclear translocation | [43] | |
| Heregulin mediated ErbB2-ERK signaling activates HAS and CD44 dependent ovarian tumor growth and migration | [44] | |
| Co-expression of CD44 and multiple drug resistance proteins MDR1 and MDP2 correlates with ovarian cancer progression | [45] | |
| Versican | Elevated stromal versican is associated with reduced overall survival | [46] |
| Versican is increased in primary and metastatic cancers | [47] | |
| Versican correlates with metastatic signature | [48] | |
| Induction of stromal versican expression is associated with tumor progression | [49] | |
| High stromal versican associates with poorer outcome | [50] | |
| Versican promotes ovarian cancer migration and invasion | [26,50] | |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ween, M.P.; Oehler, M.K.; Ricciardelli, C. Role of Versican, Hyaluronan and CD44 in Ovarian Cancer Metastasis. Int. J. Mol. Sci. 2011, 12, 1009-1029. https://doi.org/10.3390/ijms12021009
Ween MP, Oehler MK, Ricciardelli C. Role of Versican, Hyaluronan and CD44 in Ovarian Cancer Metastasis. International Journal of Molecular Sciences. 2011; 12(2):1009-1029. https://doi.org/10.3390/ijms12021009
Chicago/Turabian StyleWeen, Miranda P., Martin K. Oehler, and Carmela Ricciardelli. 2011. "Role of Versican, Hyaluronan and CD44 in Ovarian Cancer Metastasis" International Journal of Molecular Sciences 12, no. 2: 1009-1029. https://doi.org/10.3390/ijms12021009
APA StyleWeen, M. P., Oehler, M. K., & Ricciardelli, C. (2011). Role of Versican, Hyaluronan and CD44 in Ovarian Cancer Metastasis. International Journal of Molecular Sciences, 12(2), 1009-1029. https://doi.org/10.3390/ijms12021009




