Crossing the Border: Molecular Control of Motor Axon Exit
Abstract
:1. Introduction
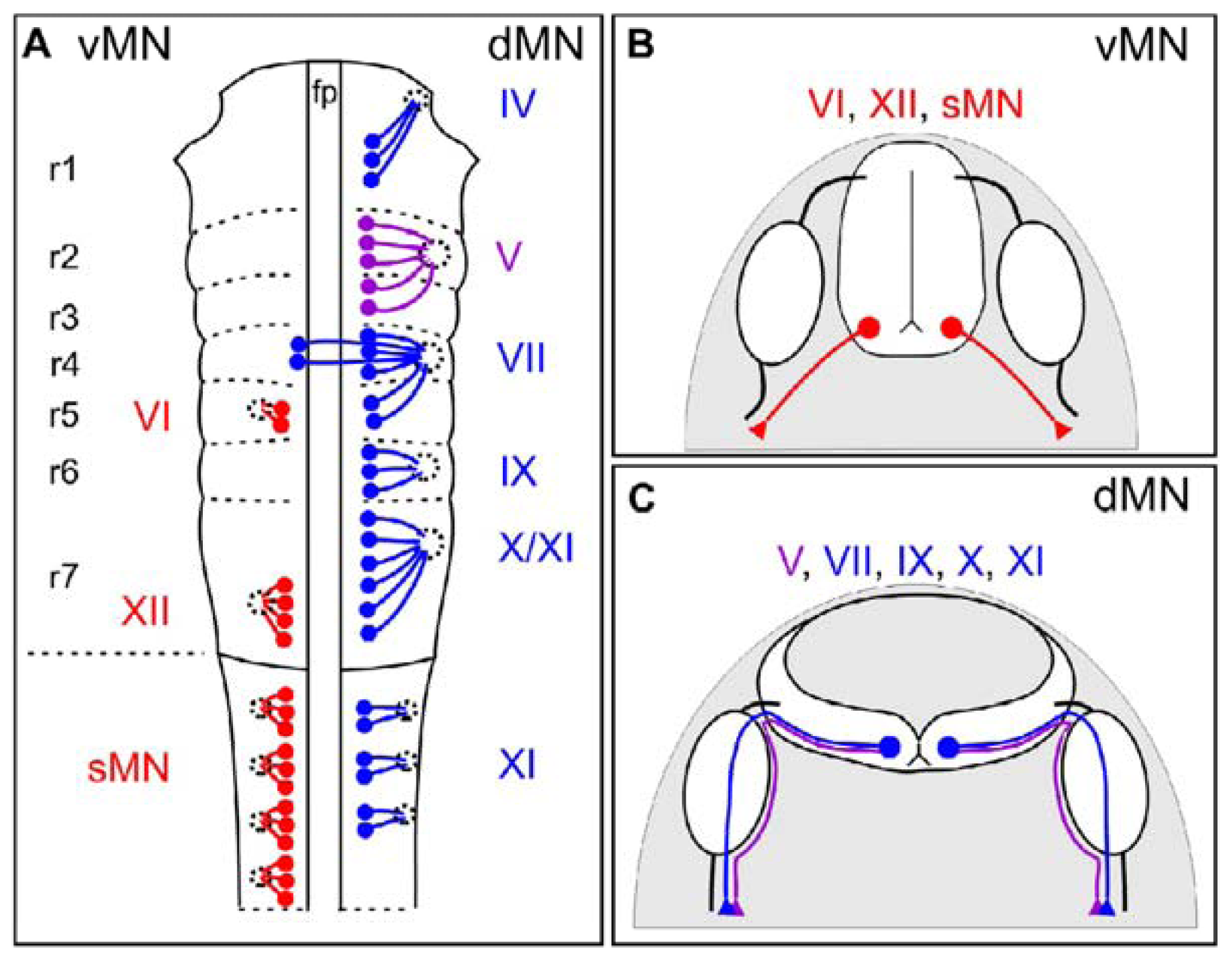
2. Motor Neuron Subtypes: Dorsally- and Ventrally-Exiting Motor Neurons
3. Motor Axon Exit Points
3.1. Motor Exit Point Chemoattractants
3.2. Positioning of Motor Exit Points
3.3. Signaling Molecules That Control the Projection of Motor Axons to Their Appropriate Exit Points
4. Motor Axon Exit
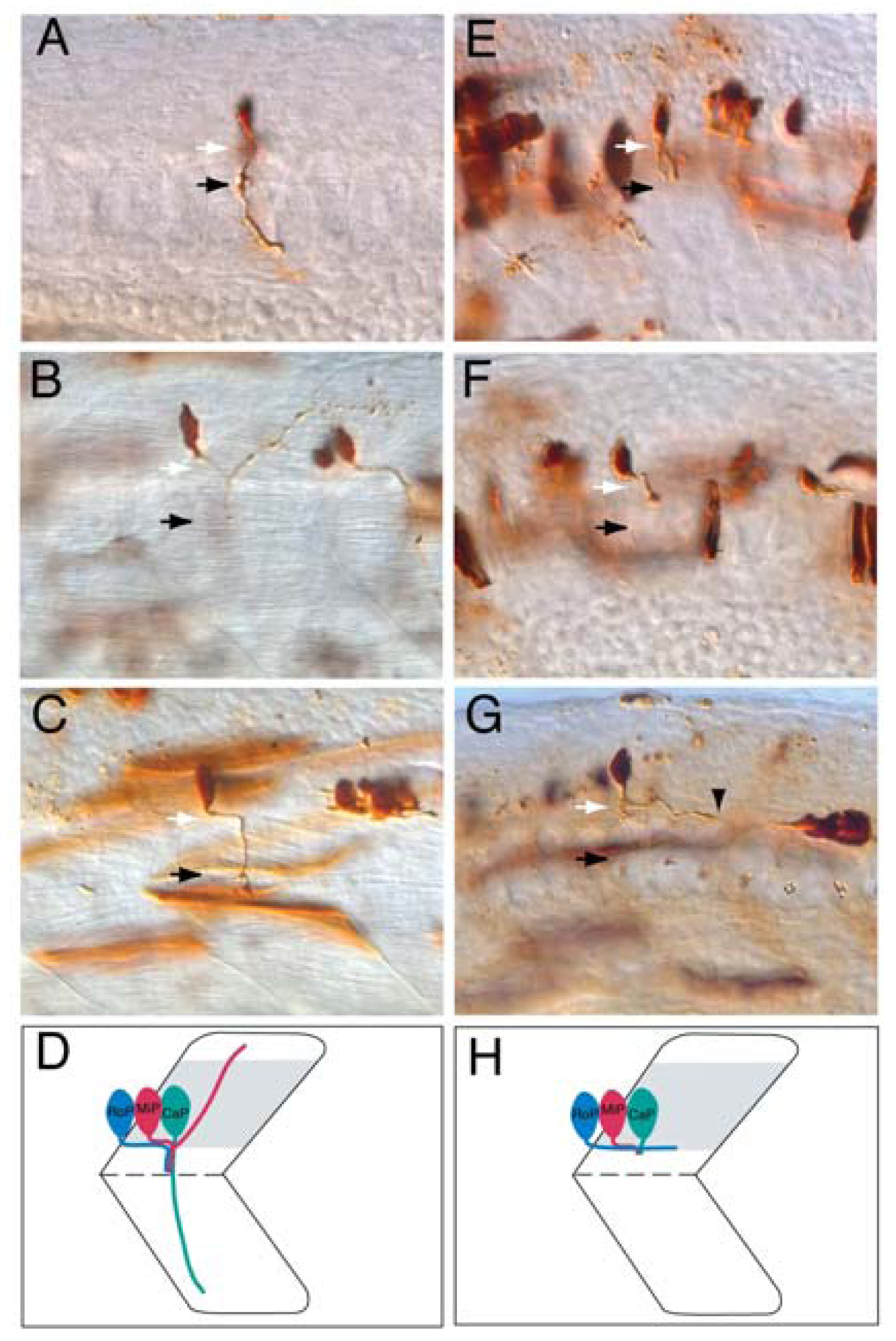
4.1. Projecting into the Periphery
4.2. Transcriptional Control of Motor Axon Exit
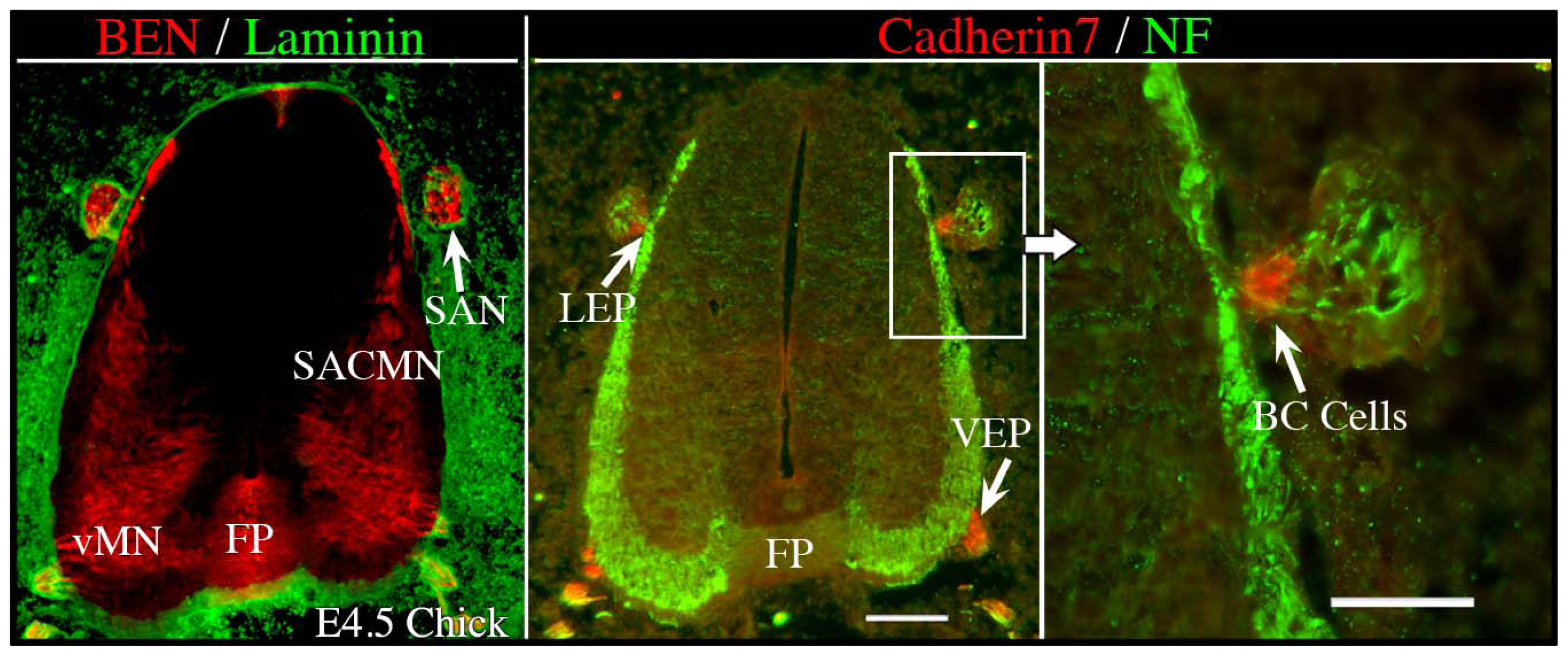
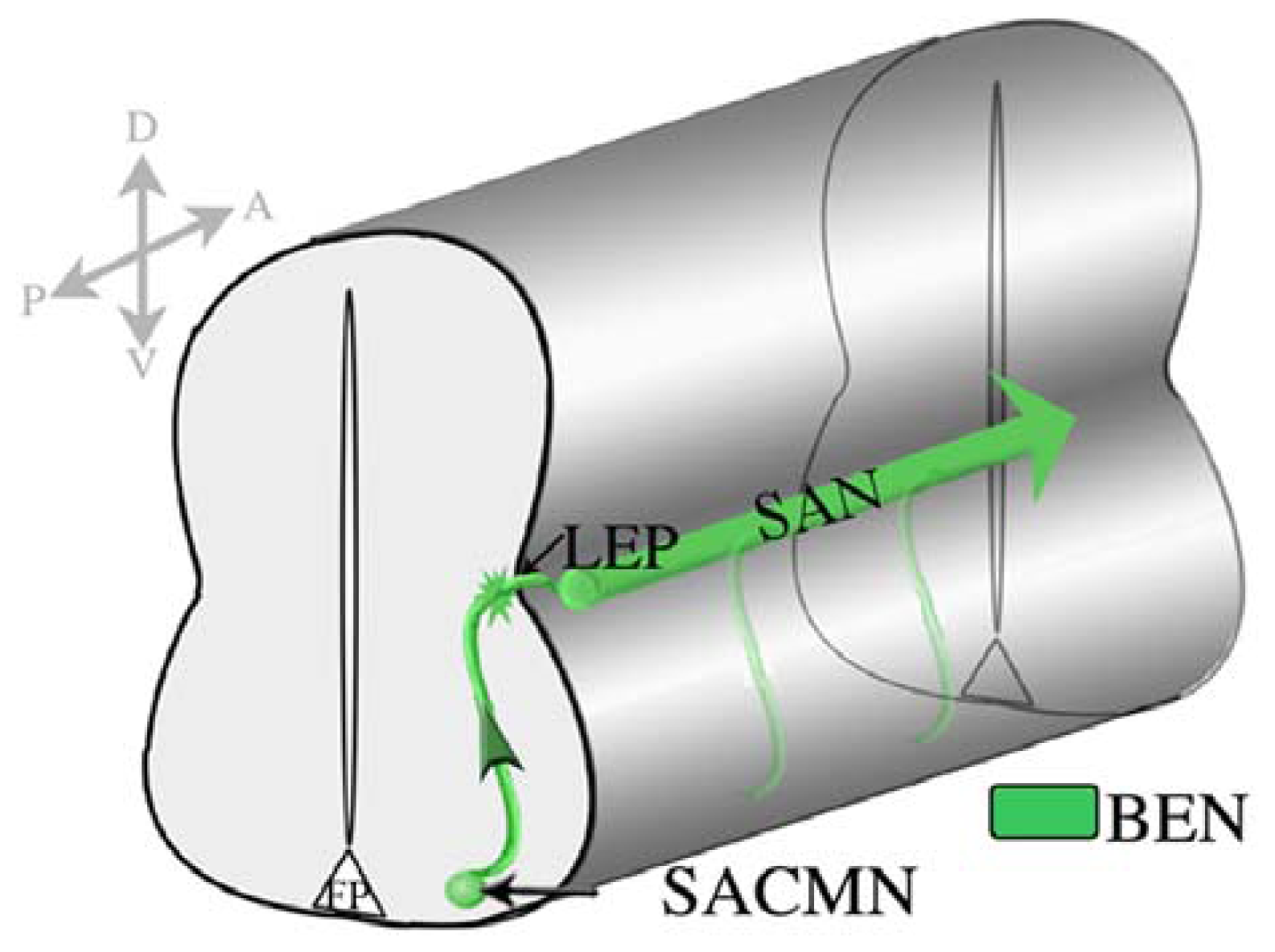
4.3. Spinal Accessory Motor Neurons (SACMN) as a Model System for Elucidating Mechanisms that Regulate Motor Exit Point-Related Guidance Events
4.4. The Homeodomain Transcription Factor, Nkx2.9, Controls Motor Axon Exit from the Mouse Spinal Cord via Robo-Slit Signaling
4.5. Homeobox Genes and Motor Axon Exit
5. Perspectives
5.1. A Novel Role for Robo-Slit Signaling in Motor Axon Exit
5.2. Is There a “Molecular Network” That Controls Motor Axon Exit?
5.3. Crossing the Basal Lamina Border
5.4. Do Motor Neuron Growth Cones Use Invadopodia to Cross the CNS:PNS Barrier?
6. Experimental Section
6.1. Animals
6.2. Immunohistochemistry
6.3. Photodocumentation
Acknowledgments
References
- Cordes, S.P. Molecular genetics of cranial nerve development in mouse. Nat. Rev. Neurosci 2001, 2, 611–623. [Google Scholar]
- Schubert, W.; Kaprielian, Z. Identification and characterization of a cell surface marker for embryonic rat spinal accessory motor neurons. J. Comp. Neurol 2001, 439, 368–383. [Google Scholar]
- Snider, W.D.; Palavali, V. Early axon and dendritic outgrowth of spinal accessory motor neurons studied with DiI in fixed tissues. J. Comp. Neurol 1990, 297, 227–238. [Google Scholar]
- Sharma, K.; Sheng, H.Z.; Lettieri, K.; Li, H.; Karavanov, A.; Potter, S.; Westphal, H.; Pfaff, S.L. LIM homeodomain factors Lhx3 and Lhx4 assign subtype identities for motor neurons. Cell 1998, 95, 817–828. [Google Scholar]
- Dillon, A.K.; Fujita, S.C.; Matise, M.P.; Jarjour, A.A.; Kennedy, T.E.; Kollmus, H.; Arnold, H.-H.; Weiner, J.A.; Sanes, J.R.; Kaprielian, Z. Molecular control of spinal accessory motor neuron/axon development in the mouse spinal cord. J. Neurosci 2005, 25, 10119–10130. [Google Scholar]
- Lieberam, I.; Agalliu, D.; Nagasawa, T.; Ericson, J.; Jessell, T.M. A Cxcl12-Cxcr4 chemokine signaling pathway defines the initial trajectory of mammalian motor axons. Neuron 2005, 47, 667–679. [Google Scholar]
- Chandrasekhar, A. Turning heads: Development of vertebrate branchiomotor neurons. Dev. Dyn 2004, 229, 143–161. [Google Scholar]
- Guthrie, S. Patterning and axon guidance of cranial motor neurons. Nat. Rev. Neurosci 2007, 8, 859–871. [Google Scholar]
- Fritzsch, B.; Northcutt, R.G. Cranial and spinal nerve organization in amphioxus and lampreys: Evidence for an ancestral craniate pattern. Acta Anat (Basel) 1993, 148, 96–109. [Google Scholar]
- Labrador, J.P.; O’Keefe, D.; Yoshikawa, S.; McKinnon, R.D.; Thomas, J.B.; Bashaw, G.J. The homeobox transcription factor even-skipped regulates netrin-receptor expression to control dorsal motor-axon projections in Drosophila. Curr. Biol 2005, 15, 1413–1419. [Google Scholar]
- Schneider, V.A.; Granato, M. The myotomal diwanka (lh3) glycosyltransferase and type XVIII collagen are critical for motor growth cone migration. Neuron 2006, 50, 683–695. [Google Scholar]
- Zeller, J.; Granato, M. The zebrafish diwanka gene controls an early step of motor growth cone migration. Development 1999, 126, 3461–3472. [Google Scholar]
- Murray, A.; Naeem, A.; Barnes, S.H.; Drescher, U.; Guthrie, S. Slit and Netrin-1 guide cranial motor axon pathfinding via Rho-kinase, myosin light chain kinase and myosin II. Neural. Dev 2010, 5, 16–30. [Google Scholar]
- Broihier, H.T.; Kuzin, A.; Zhu, Y.; Odenwald, W.; Skeath, J.B. Drosophila homeodomain protein Nkx6 coordinates motoneuron subtype identity and axonogenesis. Development 2004, 131, 5233–5242. [Google Scholar]
- Sato-Maeda, M.; Obinata, M.; Shoji, W. Position fine-tuning of caudal primary motoneurons in the zebrafish spinal cord. Development 2008, 135, 323–332. [Google Scholar]
- Bron, R.; Vermeren, M.; Kokot, N.; Little, G.E.; Mitchell, K.J.; Andrews, W.; Cohen, J. Boundary cap cells constrain spinal motor neuron somal migration at motor exit points by a semaphorin-plexin mechanism. Neural. Dev 2007, 2, 21–39. [Google Scholar]
- Hirsch, M.-R.; Glover, J.C.; Dufour, H.D.; Brunet, J.-F.; Goridis, C. Forced expression of Phox2 homeodomain transcription factors induces a branchio-visceromotor axonal phenotype. Dev. Biol 2007, 303, 687–702. [Google Scholar]
- Mauti, O.; Domanitskaya, E.; Andermatt, I.; Sadhu, R.; Stoeckli, E.T. Semaphorin6A acts as a gate keeper between the central and peripheral nervous system. Neural. Dev 2007, 2, 28–44. [Google Scholar]
- Palaisa, K.A.; Granato, M. Analysis of zebrafish sidetracked mutants reveals a novel role for Plexin A3 in intraspinal motor axon guidance. Development 2007, 134, 3251–3257. [Google Scholar]
- Layden, M.J.; Odden, J.P.; Schmid, A.; Garces, A.; Thor, S.; Doe, C.Q. Zfh1, a somatic motor neuron transcription factor, regulates axon exit from the CNS. Dev. Biol 2006, 291, 253–263. [Google Scholar]
- Dillon, A.K.; Jevince, A.R.; Hinck, L.; Ackerman, S.L.; Lu, X.; Tessier-Lavigne, M.; Kaprielian, Z. UNC5C is required for spinal accessory motor neuron development. Mol. Cell. Neurosci 2007, 35, 482–489. [Google Scholar]
- Pattyn, A.; Hirsch, M.-R.; Goridis, C.; Brunet, J.-F. Control of hindbrain motor neuron differentiation by the homeobox gene Phox2b. Development 2000, 127, 1349–1358. [Google Scholar]
- Song, M.-R.; Shirasaki, R.; Cai, C.-L.; Ruiz, E.C.; Evans, S.M.; Lee, S.-K.; Pfaff, S.L. T-box transcription factor Tbx20 regulates a genetic program for cranial motor neuron cell body migration. Development 2006, 133, 4945–4955. [Google Scholar]
- Arber, S.; Han, B.; Mendelsohn, M.; Smith, M.; Jessell, T.; Sockanathan, S. Requirement for the homeobox gene Hb9 in the consolidation of motor neuron identity. Neuron 1999, 23, 659–674. [Google Scholar]
- Shirasaki, R.; Pfaff, S.L. Transcriptional codes and the control of neuronal identity. Ann. Rev. Neurosci 2002, 25, 251–281. [Google Scholar]
- Jacob, J.; Hacker, A.; Guthrie, S. Mechanisms and molecules in motor neuron specification and axon pathfinding. BioEssays 2001, 23, 582–595. [Google Scholar]
- Schneider, V.A.; Granato, M. Motor axon migration: A long way to go. Dev. Biol 2003, 263, 1–11. [Google Scholar]
- Niederlander, C.; Lumsden, A. Late emigrating neural crest cells migrate specifically to the exit points of cranial branchiomotor nerves. Development 1996, 122, 2367–2374. [Google Scholar]
- Vermeren, M.; Maro, G.S.; Bron, R.; McGonnell, I.M.; Charnay, P.; Topilko, P.; Cohen, J. Integrity of developing spinal motor columns is regulated by neural crest derivatives at motor exit points. Neuron 2003, 37, 403–415. [Google Scholar]
- Golding, J.P.; Cohen, J. Border controls at the mammalian spinal cord: Late-surviving neural crest boundary cap cells at dorsal root entry sites may regulate sensory afferent ingrowth and entry zone morphogenesis. Mol. Cell. Neurosci 1997, 5, 381–396. [Google Scholar]
- Wilkinson, D.G.; Bhatt, S.; Chavrier, P.; Bravo, R.; Charnay, P. Segment-specific expression of a zinc-finger gene in the developing nervous system of the mouse. Nature 1989, 337, 461–464. [Google Scholar]
- Topilko, P.; Schneider-Maunoury, S.; Levi, G.; Evercooren, A.B.-V.; Chennoufi, A.B.Y.; Seitanidou, T.; Babinet, C.; Charnay, P. Krox-20 controls myelination in the peripheral nervous system. Nature 1994, 371, 796–799. [Google Scholar]
- Redies, C. Cadherins in the central nervous system. Prog. Neurobiol 2000, 61, 611–648. [Google Scholar]
- Nakagawa, S.; Takeichi, M. Neural crest emigration from the neural tube depends on regulated cadherin expression. Development 1998, 125, 2963–2971. [Google Scholar]
- Okafuji, T.; Tanaka, H. Expression pattern of LINGO-1 in the developing nervous system of the chick embryo. Gene Expr. Patterns 2005, 6, 57–62. [Google Scholar]
- Knabe, W.; Siren, A.L.; Ehrenreich, H.; Kuhn, H.J. Expression patterns of erythropoietin and its receptor in the developing spinal cord and dorsal root ganglia. Anat. Embryol (Berl) 2005, 210, 209–219. [Google Scholar]
- Vitalis, T.; Alvarez, C.; Chen, K.; Shih, J.C.; Gaspar, P.; Cases, O. Developmental expression pattern of monoamine oxidases in sensory organs and neural crest derivatives. J. Comp. Neurol 2003, 464, 392–403. [Google Scholar]
- Auerbach, R. Analysis of developmental effects of a lethal mutation in the house mouse. J. Exp. Zool 1954, 127, 305–329. [Google Scholar]
- Henderson, D.J.; Ybot-Gonzalez, P.; Copp, A.J. Overexpression of the chondroitin sulphate proteoglycan versican is associated with defective neural crest migration in the Pax3 mutant mouse (splotch). Mech. Dev 1997, 69, 39–51. [Google Scholar]
- Serbedzija, G.N.; McMahon, A.P. Analysis of neural crest cell migration in Splotch mice using a neural crest-specific LacZ reporter. Dev. Biol 1997, 185, 139–147. [Google Scholar]
- Chauvet, S.; Rougon, G. Semaphorins deployed to repel cell migrants at spinal cord borders. J. Biol 2008, 7, 4:1–4:5. [Google Scholar]
- Fraher, J.P.; Dockery, P.; O’Donoghue, O.; Riedewald, B.; O’Leary, D. Initial motor axon outgrowth from the developing central nervous system. J. Anat 2007, 211, 600–611. [Google Scholar]
- Fraher, J.P.; Rossiter, J.P. Cell clusters on fetal rat ventral roots: Prenatal development. J. Anat 1983, 136, 111–128. [Google Scholar]
- Colamarino, S.A.; Tessier-Lavigne, M. The axonal chemoattractant Netrin-1 is also a chemorepellent for trochlear motor axons. Cell 1995, 81, 621–629. [Google Scholar]
- Chang, S.; Fan, J.; Nayak, J. Pathfinding by cranial nerve VII (facial) motor neurons in the chick hindbrain. Development 1992, 114, 815–823. [Google Scholar]
- Caton, A.; Hacker, A.; Naeem, A.; Livet, J.; Maina, F.; Bladt, F.; Klein, R.; Birchmeier, C.; Guthrie, S. The branchial arches and HGF are growth-promoting and chemoattractant for cranial motor axons. Development 2000, 127, 1751–1760. [Google Scholar]
- Guthrie, S.; Lumsden, A. Motor neuron pathfinding following rhombomere reversals in the chick embryo hindbrain. Development 1992, 114, 663–673. [Google Scholar]
- Shirasaki, R.; Lewcock, J.W.; Lettieri, K.; Pfaff, S.L. FGF as a target-derived chemoattractant for developing motor axons genetically programmed by the LIM code. Neuron 2006, 50, 841–853. [Google Scholar]
- Ebens, A.; Brose, K.; Leonardo, E.D.; Hanson, M.G.; Bladt, F.; Birchmeier, C.; Barres, B.A.; Tessier-Lavigne, M. Hepatocyte growth factor/scatter factor is an axonal chemoattractant and a neurotrophic factor for spinal motor neurons. Neuron 1996, 17, 1157–1172. [Google Scholar]
- Naeem, A.; Abbas, L.; Guthrie, S. Comparison of the effects of HGF, BDNF, CT-1, CNTF, and the brachial arches on the growth of embryonic cranial motor neurons. J. Neurobiol 2002, 51, 101–114. [Google Scholar]
- McCaig, C.D. Myoblasts and myoblast-conditioned medium attract the earliest spinal neurites from frog embryos. J. Physiol 1986, 375, 39–54. [Google Scholar]
- Pollack, E.D.; Muhlach, W.L.; Liebig, V. Neurotrophic influence of mesenchymal limb target tissue on spinal cord neurite growth in vitro. J. Comp. Neurol 1981, 200, 393–405. [Google Scholar]
- Tucker, K.L.; Meyer, M.; Barde, Y.A. Neurotrophins are required for nerve growth during development. Nat. Neurosci 2001, 4, 29–37. [Google Scholar]
- Guthrie, S.; Lumsden, A. Motor neuron pathfinding following rhombomere reversals in the chick embryo hindbrain. Development 1992, 114, 663–673. [Google Scholar]
- Irving, C.; Malhas, A.; Guthrie, S.; Mason, I. Establishing the trochlear motor axon trajectory: Role of the isthmic organiser and Fgf8. Development 2002, 129, 5389–5398. [Google Scholar]
- Birely, J.; Schneider, V.A.; Santana, E.; Dosch, R.; Wagner, D.S.; Mullins, M.C.; Granato, M. Genetic screens for genes controlling motor nerve-muscle development and interactions. Dev. Biol 2005, 280, 162–176. [Google Scholar]
- Kucenas, S.; Takada, N.; Park, H.C.; Woodruff, E.; Broadie, K.; Appel, B. CNS-derived glia ensheath peripheral nerves and mediate motor root development. Nat. Neurosci 2008, 11, 143–151. [Google Scholar]
- Kucenas, S.; Wang, W.D.; Knapik, E.W.; Appel, B. A selective glial barrier at motor axon exit points prevents oligodendrocyte migration from the spinal cord. J. Neurosci 2009, 29, 15187–15194. [Google Scholar]
- Hammond, R.; Vivancos, V.; Naeem, A.; Chilton, J.; Mambitisaeva, E.; Andrews, W.; Sundaresan, V.; Guthrie, S. Slit-mediated repulsion is a key regulator of motor axon pathfinding in the hindbrain. Development 2005, 132, 4483–4495. [Google Scholar]
- Colamarino, S.A.; Tessier-Lavigne, M. The role of the floor plate in axon guidance. Ann. Rev. Neurosci 1995, 18, 497–529. [Google Scholar]
- Burgess, R.W.; Jucius, T.J.; Ackerman, S.L. Motor axon guidance of the mammalian trochlear and phrenic neves: Dependence on the netrin receptor Unc5c and modifier loci. J. Neurosci 2006, 26, 5756–5766. [Google Scholar]
- Brown, J.; Bridgman, P.C. Role of myosin II in axon outgrowth. J. Histochem. Cytochem 2003, 51, 421–428. [Google Scholar]
- Zeller, J.; Schneider, V.; Malayaman, S.; Higashijima, S.; Okamoto, H.; Gui, J.; Lin, S.; Granato, M. Migration of zebrafish spinal motor nerves into the periphery requires multiple myotome-derived cues. Dev. Biol 2002, 252, 241–256. [Google Scholar]
- Butler, S.J.; Tear, G. Getting axons onto the right path: The role of transcription factors in axon guidance. Development 2007, 134, 439–448. [Google Scholar]
- Pabst, O.; Rummelies, J.; winter, B.; Arnold, H.-H. Targeted disruption of the homeobox gene Nkx2.9 reveals a role in development of the spinal accessory nerve. Development 2003, 130, 1193–1202. [Google Scholar]
- Holz, A.; Kollmus, H.; Ryge, J.; Niederkofler, V.; Dias, J.; Ericson, J.; Stoeckli, E.T.; Kiehn, O.; Arnold, H.H. The transcription factors Nkx2.2 and Nkx2.9 play a novel role in floor plate development and commissural axon guidance. Development 2010, 137, 4249–4260. [Google Scholar] [Green Version]
- Geisen, M.J.; Di Meglio, T.; Pasqualetti, M.; Ducret, S.; Brunet, J.F.; Chedotal, A.; Rijli, F.M. Hox paralog group 2 genes control the migration of mouse pontine neurons through slit-robo signaling. PLoS Biol 2008, 6, e142:1178–e142:1194. [Google Scholar]
- Medina-Martinez, O.; Bradley, A.; Ramirez-Solis, R. A large targeted deletion of Hoxb1-Hoxb9 produces a series of single-segment anterior homeotic transformations. Dev. Biol 2000, 222, 71–83. [Google Scholar]
- Ypsilanti, A.R.; Zagar, Y.; Chedotal, A. Moving away from the midline: New developments for Slit and Robo. Development 2010, 137, 1939–1952. [Google Scholar]
- Spitzweck, B.; Brankatschk, M.; Dickson, B.J. Distinct protein domains and expression patterns confer divergent axon guidance functions for Drosophila Robo receptors. Cell 2010, 140, 409–420. [Google Scholar]
- Pappu, K.S.; Zipursky, S.L. Axon guidance: Repulsion and attraction in roundabout ways. Curr. Biol 2010, 20, R400–R402. [Google Scholar]
- Evans, T.A.; Bashaw, G.J. Functional diversity of Robo receptor immunoglobulin domains promotes distinct axon guidance decisions. Curr. Biol 2010, 20, 567–572. [Google Scholar]
- Dickson, B.J.; Gilestro, G.F. Regulation of commissural axon pathfinding by Slit and its Robo receptors. Annu. Rev. Cell. Dev. Biol 2006, 22, 651–675. [Google Scholar]
- Sabatier, C.; Plump, A.S.; Ma, L.; Brose, K.; Tamada, A.; Murakami, F.; Lee, E.Y.-H.P.; Tessier-Lavigne, M. The divergent Robo family protein Rig-1/Robo3 is a negative regulator of Slit responsiveness required for midline crossing by commissural axons. Cell 2004, 117, 157–169. [Google Scholar]
- Pabst, O.; Herbrand, H.; Arnold, H.H. Nkx2-9 is a novel homeobox transcription factor which demarcates ventral domains in the developing mouse CNS. Mech. Dev 1998, 73, 85–93. [Google Scholar]
- Sordino, P.; Duboule, D.; Kondo, T. Zebrafish Hoxa and Evx-2 genes: Cloning, developmental expression and implications for the functional evolution of posterior Hox genes. Mech. Dev 1996, 59, 165–175. [Google Scholar]
- Dolle, P.; Fraulob, V.; Duboule, D. Developmental expression of the mouse Evx-2 gene: Relationship with the evolution of the HOM/Hox complex. Dev. Suppl 1994, 143–153. [Google Scholar]
- Bastian, H.; Gruss, P.; Duboule, D.; Izpisua-Belmonte, J.C. The murine even-skipped-like gene Evx-2 is closely linked to the Hox-4 complex, but is transcribed in the opposite direction. Mamm. Genome 1992, 3, 241–243. [Google Scholar]
- McFarlane, S. Metalloproteases: Carving out a role in axon guidance. Neuron 2003, 37, 559–562. [Google Scholar]
- Weaver, A.M. Invadopodia: Specialized cell structures for cancer invasion. Clin. Exp. Metastasis 2006, 23, 97–105. [Google Scholar]
- Chen, W.T. Proteolytic activity of specialized surface protrusions formed at rosette contact sites of transformed cells. J. Exp. Zool 1989, 251, 167–185. [Google Scholar]
- Oser, M.; Condeelis, J. The cofilin activity cycle in lamellipodia and invadopodia. J. Cell. Biochem 2009, 108, 1252–1262. [Google Scholar]
- Oser, M.; Yamaguchi, H.; Mader, C.C.; Bravo-Cordero, J.J.; Arias, M.; Chen, X.; Desmarais, V.; van Rheenen, J.; Koleske, A.J.; Condeelis, J. Cortactin regulates cofilin and N-WASp activities to control the stages of invadopodium assembly and maturation. J. Cell. Biol 2009, 186, 571–587. [Google Scholar]
- Oser, M.; Mader, C.C.; Gil-Henn, H.; Magalhaes, M.; Bravo-Cordero, J.J.; Koleske, A.J.; Condeelis, J. Specific tyrosine phosphorylation sites on cortactin regulate Nck1-dependent actin polymerization in invadopodia. J. Cell. Sci 2010, 123, 3662–3673. [Google Scholar]
- Weiner, J.A.; Koo, S.J.; Nicolas, S.; Fraboulet, S.; Pfaff, S.L.; Pourquie, O.; Sanes, J.R. Axon fasciculation defects and retinal dysplasias in mice lacking the immunoglobulin superfamily adhesion molecule BEN/ALCAM/SC1. Mol. Cell. Neurosci 2004, 27, 59–69. [Google Scholar]
- Lee, S.-K.; Jurata, L.W.; Funahashi, J.; Ruiz, E.C.; Pfaff, S.L. Analysis of embryonic motoneuron gene regulation: Derepression of general activators function in concert with enhancer factors. Development 2004, 131, 3295–3306. [Google Scholar]
- Wichterle, H.; Lieberam, I.; Porter, J.A.; Jessell, T.M. Directed differentiation of embryonic stem cells into motor neurons. Cell 2002, 110, 385–397. [Google Scholar]
- Lazarenko, R.M.; Milner, T.A.; Depuy, S.D.; Stornetta, R.L.; West, G.H.; Kievits, J.A.; Bayliss, D.A.; Guyenet, P.G. Acid sensitivity and ultrastructure of the retrotrapezoid nucleus in Phox2b-EGFP transgenic mice. J. Comp. Neurol 2009, 517, 69–86. [Google Scholar]



| Name | Organism | Localization | Role in motor neuron development |
|---|---|---|---|
| Cxcl12 | Mouse | mesenchyme | Required for a subset of vMN axons to exit through ventral exit points[6] |
| Cxcr4 | Mouse | vMN | Required for a subset of vMN axons to exit through ventral exit points[6] |
| Eve (TF) | Drosophila | dMNs | Required for dMN axons to exit the CNS[10] |
| LH3 (diwanka) | Zebrafish | myotome | Promotes the exit of a subset of MN axons from the CNS[11,12] |
| Lhx3/Lhx4 (TF) | Mouse | vMNs | Required for vMN specification; sufficient to re-direct dMN axons to and through ventral exit points[4] |
| Myosin II | Chick | growth cone | Required for a subset of dMN axons to project towards their appropriate motor exit point[13] |
| Nkx6 (TF) | Drosophila | vMNs | Required for vMN axons to exit the CNS[14] |
| Nkx2.9 (TF) | Mouse | SACMN derived from Nkx2.9 progenitors | Required for SACMN axon exit * |
| Npn1a | Zebrafish | CaP MN | Required for the proper positioning of motor exit points[15] |
| Npn2 | Chick/Mouse | vMN cell bodies | Required for the confinement of vMN somata to the CNS [16] |
| Phox2a/Phox2b (TF) | Chick | dMNs | Sufficient to re-specify interneurons into MNs[17] |
| PlexinA1 | Chick | vMN axons | Prevents vMN somata from inappropriately migrating out of the CNS[18] |
| PlexinA2 | Chick | vMN cell bodies | Prevents vMN somata from inappropriately migrating out of the CNS[16] |
| PlexinA3 (sidetracked) | Zebrafish | MiP and RoP MNs | Required for the proper positioning of motor exit points[19] |
| RhoA | Chick | growth cone | Required for a subset of dMN axons to project towards their appropriate motor exit point[13] |
| Robo2 | Mouse | SACMN axons | Required for SACMN axon exit * |
| Sema3ab | Zebrafish | Somite | Required for the proper positioning of motor exit points[15] |
| Sema6A | Chick/Mouse | BC Cells | Prevents vMN somata from inappropriately migrating out of the CNS[16,18] |
| Slit1/Slit2 | Mouse | Lateral exit point-associated cells | Required for SACMN axon exit* |
| Zfh1 (TF) | Drosophila | vMNs | Required for a subset of vMN axons to exit the CNS[20] |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bravo-Ambrosio, A.; Kaprielian, Z. Crossing the Border: Molecular Control of Motor Axon Exit. Int. J. Mol. Sci. 2011, 12, 8539-8561. https://doi.org/10.3390/ijms12128539
Bravo-Ambrosio A, Kaprielian Z. Crossing the Border: Molecular Control of Motor Axon Exit. International Journal of Molecular Sciences. 2011; 12(12):8539-8561. https://doi.org/10.3390/ijms12128539
Chicago/Turabian StyleBravo-Ambrosio, Arlene, and Zaven Kaprielian. 2011. "Crossing the Border: Molecular Control of Motor Axon Exit" International Journal of Molecular Sciences 12, no. 12: 8539-8561. https://doi.org/10.3390/ijms12128539
APA StyleBravo-Ambrosio, A., & Kaprielian, Z. (2011). Crossing the Border: Molecular Control of Motor Axon Exit. International Journal of Molecular Sciences, 12(12), 8539-8561. https://doi.org/10.3390/ijms12128539





