Impact of Dietary Polyphenols on Carbohydrate Metabolism
Abstract
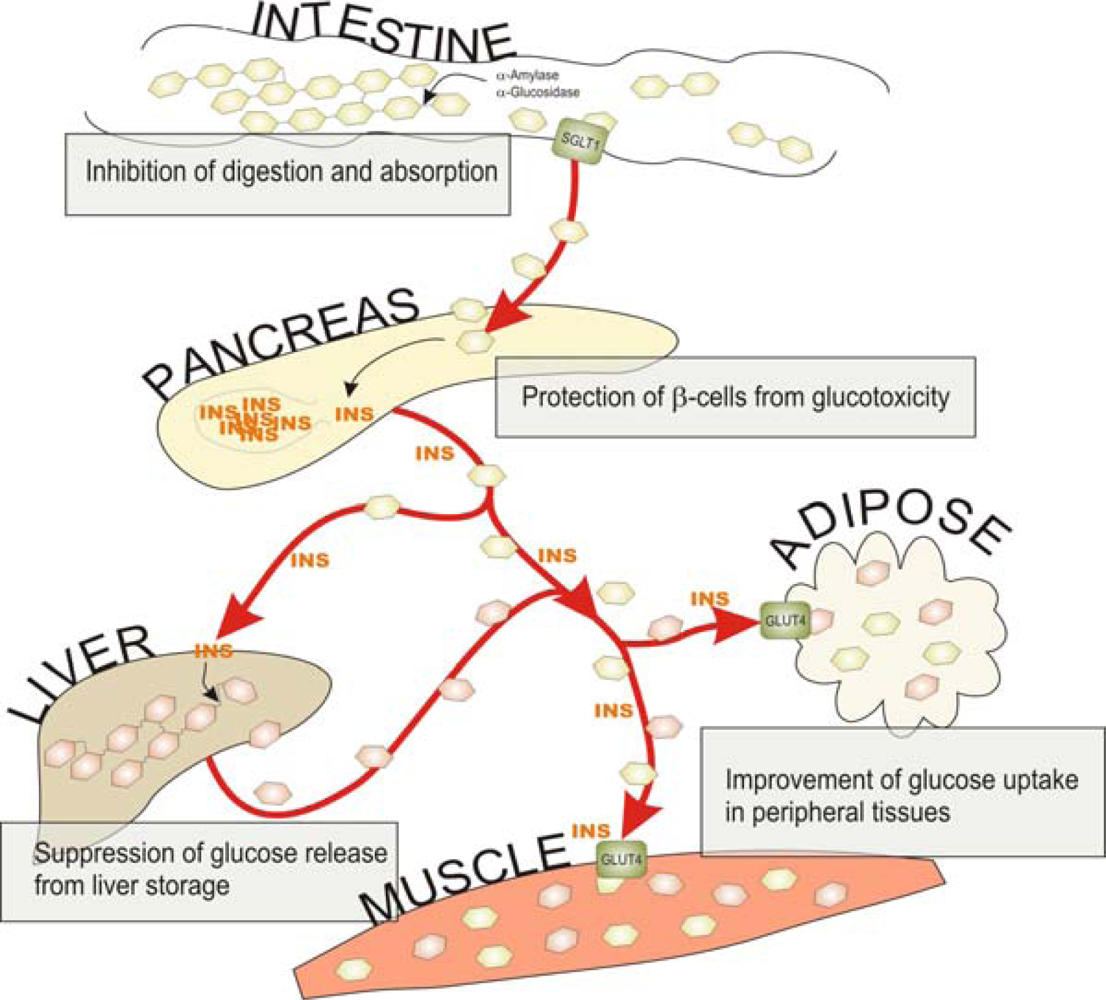
:1. Introduction
2. Influence of Polyphenols on Carbohydrate Digestion and Glucose Absorption in the Intestine
2.1. Carbohydrate Digestion
2.2. Glucose Absorption
2.3. Postprandial Glycemia
3. Influence of Polyphenols on Pancreatic β-cell Function
3.1. In Vivo Studies with Animal Models
3.2. Effects Observed in Cell Culture Analyses
3.3. Effects Observed in Isolated/Perfused Pancreas
4. Influence of Polyphenols on Tissue Uptake of Glucose
4.1. Effects of Pure Compounds on Glucose Uptake
4.2. Effects of Polyphenol Containing Foods and Plant Extracts on Glucose Uptake
5. Influence of Polyphenols on Liver Function to Maintain Glucose Homeostasis
5.1. Effects of Green Tea and Epigallocatechin Gallate (EGCG)
5.2. Effect of Soy Isoflavones, Genistein and Daidzein
5.3. Effect of Citrus Flavonoids, Grape Polyphenols and Phenolic Acids
6. Impact of Polyphenols on Maintenance of Glucose Homeostasis
6.1. Evidence from Epidemiological Studies
6.2. Evidence from Clinical Trials
6.3. Evidence from Animal Experiments
7. Conclusions and Future Prospects
- There are indications for positive effects on glucose homeostasis with polyphenols and polyphenol-rich plant extracts from in vitro & animal studies.
- Epidemiological evidence supports beneficial effects of polyphenol- rich diets.
- Clinical studies so far have not undoubtedly succeeded in pointing out any specific polyphenols or food products in reducing the risk of insulin resistance.
- It is evident that in clinical studies whole diets instead of single compounds or food components should be addressed.
- Combination of specific clinical measurements determining glucose tolerance and insulin sensitivity together with systems biology profiling technologies is needed to get a holistic view on the health effects of diets and foods rich in polyphenols.
Acknowledgments
References and Notes
- Ovaskainen, ML; Torronen, R; Koponen, JM; Sinkko, H; Hellstrom, J; Reinivuo, H; Mattila, P. Dietary intake and major food sources of polyphenols in Finnish adults. J. Nutr 2008, 138, 562–566. [Google Scholar]
- Scalbert, A; Manach, C; Morand, C; Remesy, C; Jimenez, L. Dietary polyphenols and the prevention of diseases. Crit. Rev. Food Sci. Nutr 2005, 45, 287–306. [Google Scholar]
- Crozier, A; Jaganath, IB; Clifford, MN. Dietary phenolics: Chemistry, bioavailability and effects on health. Nat. Prod. Rep 2009, 26, 1001–1043. [Google Scholar]
- Clifford, MN. Diet-derived phenols in plasma and tissues and their implications for health. Planta Med 2004, 70, 1103–1114. [Google Scholar]
- Knekt, P; Kumpulainen, J; Jarvinen, R; Rissanen, H; Heliovaara, M; Reunanen, A; Hakulinen, T; Aromaa, A. Flavonoid intake and risk of chronic diseases. Am. J. Clin. Nutr 2002, 76, 560–568. [Google Scholar]
- Selma, MV; Espin, JC; Tomas-Barberan, FA. Interaction between phenolics and gut microbiota: role in human health. J. Agric. Food Chem 2009, 57, 6485–6501. [Google Scholar]
- Lila, MA. From beans to berries and beyond: Teamwork between plant chemicals for protection of optimal human health. Ann. NY Acad. Sci 2007, 1114, 372–380. [Google Scholar]
- Liu, RH. Health benefits of fruit and vegetables are from additive and synergistic combinations of phytochemicals. Am. J. Clin. Nutr 2003, 78, 517S–520S. [Google Scholar]
- Eckel, RH; Grundy, SM; Zimmet, PZ. The metabolic syndrome. Lancet 2005, 365, 1415–1428. [Google Scholar]
- Uusitupa, M. Gene-diet interaction in relation to the prevention of obesity and type 2 diabetes: Evidence from the Finnish Diabetes Prevention Study. Nutr. Metab. Cardiovasc. Dis 2005, 15, 225–233. [Google Scholar]
- McCarthy, MI. Progress in defining the molecular basis of type 2 diabetes mellitus through susceptibility-gene identification. Hum Mol Genet 2004, 13(Spec No 1), R33–R41. [Google Scholar]
- Laaksonen, DE; Niskanen, L; Lakka, HM; Lakka, TA; Uusitupa, M. Epidemiology and treatment of the metabolic syndrome. Ann. Med 2004, 36, 332–346. [Google Scholar]
- Lann, D; Gallagher, E; Leroith, D. Insulin resistance and the metabolic syndrome. Minerva Med 2008, 99, 253–262. [Google Scholar]
- Wild, S; Roglic, G; Green, A; Sicree, R; King, H. Global prevalence of diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care 2004, 27, 1047–1053. [Google Scholar]
- Tuomilehto, J; Lindstrom, J; Eriksson, JG; Valle, TT; Hamalainen, H; Ilanne-Parikka, P; Keinanen-Kiukaanniemi, S; Laakso, M; Louheranta, A; Rastas, M; Salminen, V; Uusitupa, M. Finnish Diabetes Prevention Study Group Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N. Engl. J. Med 2001, 344, 1343–1350. [Google Scholar]
- Lindström, J; Ilanne-Parikka, P; Peltonen, M; Aunola, S; Eriksson, JG; Hemiö, K; Hämäläinen, H; Härkönen, P; Keinänen-Kiukaanniemi, S; Laakso, M; Louheranta, A; Mannelin, M; Paturi, M; Sundvall, J; Valle, TT; Uusitupa, M; Tuomilehto, J. Sustained reduction in the incidence of type 2 diabetes by lifestyle intervention: Follow-up of the Finnish Diabetes Prevention Study. Lancet 2006, 368, 1673–1679. [Google Scholar]
- Orgaard, A; Jensen, L. The effects of soy isoflavones on obesity. Exp. Biol. Med. (Maywood) 2008, 233, 1066–1080. [Google Scholar]
- Wolfram, S. Effects of green tea and EGCG on cardiovascular and metabolic health. J. Am. Coll. Nutr 2007, 26, 373S–388S. [Google Scholar]
- van Dam, RM; Hu, FB. Coffee consumption and risk of type 2 diabetes: A systematic review. JAMA 2005, 294, 97–104. [Google Scholar]
- Zunino, S. Type 2 diabetes and glycemic response to grapes or grape products. J. Nutr 2009, 139, 1794S–800S. [Google Scholar]
- Boyer, J; Liu, RH. Apple phytochemicals and their health benefits. Nutr. J 2004, 3, 5. [Google Scholar]
- Hui, H; Tang, G; Go, VL. Hypoglycemic herbs and their action mechanisms. Chin. Med 2009, 4, 11. [Google Scholar]
- Ghosh, D; Konishi, T. Anthocyanins and anthocyanin-rich extracts: Role in diabetes and eye function. Asia Pac. J. Clin. Nutr 2007, 16, 200–208. [Google Scholar]
- Bondia-Pons, I; Aura, A; Vuorela, S; Kolehmainen, M; Mykkanen, H; Poutanen, K. Rye phenolics in nutrition and health. J. Cereal Sci 2009, 49, 323–336. [Google Scholar]
- Maritim, AC; Sanders, RA; Watkins, JB, III. Diabetes, oxidative stress, and antioxidants: A review. J. Biochem. Mol. Toxicol 2003, 17, 24–38. [Google Scholar]
- Vincent, HK; Innes, KE; Vincent, KR. Oxidative stress and potential interventions to reduce oxidative stress in overweight and obesity. Diabetes Obes. Metab 2007, 9, 813–839. [Google Scholar]
- Ludwig, DS. The glycemic index: Physiological mechanisms relating to obesity, diabetes, and cardiovascular disease. JAMA 2002, 287, 2414–2423. [Google Scholar]
- Quezada-Calvillo, R; Robayo-Torres, CC; Ao, Z; Hamaker, BR; Quaroni, A; Brayer, GD; Sterchi, EE; Baker, SS; Nichols, BL. Luminal substrate “brake” on mucosal maltase-glucoamylase activity regulates total rate of starch digestion to glucose. J. Pediatr. Gastroenterol. Nutr 2007, 45, 32–43. [Google Scholar]
- Quezada-Calvillo, R; Robayo-Torres, CC; Opekun, AR; Sen, P; Ao, Z; Hamaker, BR; Quaroni, A; Brayer, GD; Wattler, S; Nehls, MC; Sterchi, EE; Nichols, BL. Contribution of mucosal maltase-glucoamylase activities to mouse small intestinal starch alpha-glucogenesis. J. Nutr 2007, 137, 1725–1733. [Google Scholar]
- Quezada-Calvillo, R; Sim, L; Ao, Z; Hamaker, BR; Quaroni, A; Brayer, GD; Sterchi, EE; Robayo-Torres, CC; Rose, DR; Nichols, BL. Luminal starch substrate “brake” on maltase-glucoamylase activity is located within the glucoamylase subunit. J. Nutr 2008, 138, 685–692. [Google Scholar]
- Levin, RJ. Digestion and absorption of carbohydrates--from molecules and membranes to humans. Am. J. Clin. Nutr 1994, 59, 690S–698S. [Google Scholar]
- Drozdowski, LA; Thomson, ABR. Intestinal sugar transport. World J. Gastroenterol 2006, 12, 1657–1670. [Google Scholar]
- Welsch, CA; Lachance, PA; Wasserman, BP. Dietary phenolic compounds: Inhibition of Na+- dependent D-glucose uptake in rat intestinal brush border membrane vesicles. J. Nutr 1989, 119, 1698–1704. [Google Scholar]
- Cermak, R; Landgraf, S; Wolffram, S. Quercetin glucosides inhibit glucose uptake into brush-border-membrane vesicles of porcine jejunum. Br. J. Nutr 2004, 91, 849–855. [Google Scholar]
- Kobayashi, Y; Suzuki, M; Satsu, H; Arai, S; Hara, Y; Suzuki, K; Miyamoto, Y; Shimizu, M. Green tea polyphenols inhibit the sodium-dependent glucose transporter of intestinal epithelial cells by a competitive mechanism. J. Agric. Food Chem 2000, 48, 5618–5623. [Google Scholar]
- Shimizu, M; Kobayashi, Y; Suzuki, M; Satsu, H; Miyamoto, Y. Regulation of intestinal glucose transport by tea catechins. Biofactors 2000, 13, 61–65. [Google Scholar]
- Johnston, K; Sharp, P; Clifford, M; Morgan, L. Dietary polyphenols decrease glucose uptake by human intestinal Caco-2 cells. FEBS Lett 2005, 579, 1653–1657. [Google Scholar]
- Li, JM; Che, CT; Lau, CB; Leung, PS; Cheng, CH. Inhibition of intestinal and renal Na+- glucose cotransporter by naringenin. Int. J. Biochem. Cell Biol 2006, 38, 985–995. [Google Scholar]
- Song, J; Kwon, O; Chen, S; Daruwala, R; Eck, P; Park, JB; Levine, M. Flavonoid inhibition of sodium-dependent vitamin C transporter 1 (SVCT1) and glucose transporter isoform 2 (GLUT2), intestinal transporters for vitamin C and Glucose. J. Biol. Chem 2002, 277, 15252–15260. [Google Scholar]
- Matsui, T; Ebuchi, S; Kobayashi, M; Fukui, K; Sugita, K; Terahara, N; Matsumoto, K. Antihyperglycemic effect of diacylated anthocyanin derived from Ipomoea batatas cultivar Ayamurasaki can be achieved through the alpha-glucosidase inhibitory action. J. Agric. Food Chem 2002, 50, 7244–7248. [Google Scholar]
- Matsui, T; Tanaka, T; Tamura, S; Toshima, A; Tamaya, K; Miyata, Y; Tanaka, K; Matsumoto, K. alpha-Glucosidase inhibitory profile of catechins and theaflavins. J. Agric. Food Chem 2007, 55, 99–105. [Google Scholar]
- Hanamura, T; Mayama, C; Aoki, H; Hirayama, Y; Shimizu, M. Antihyperglycemic effect of polyphenols from Acerola (Malpighia emarginata DC.) fruit. Biosci. Biotechnol. Biochem 2006, 70, 1813–1820. [Google Scholar]
- Ishikawa, A; Yamashita, H; Hiemori, M; Inagaki, E; Kimoto, M; Okamoto, M; Tsuji, H; Memon, AN; Mohammadio, A; Natori, Y. Characterization of inhibitors of postprandial hyperglycemia from the leaves of Nerium indicum. J. Nutr. Sci. Vitaminol. (Tokyo) 2007, 53, 166–173. [Google Scholar]
- Tanaka, S; Han, LK; Zheng, YN; Okuda, H. Effects of the flavonoid fraction from Ginkgo biloba extract on the postprandial blood glucose elevation in rats. Yakugaku Zasshi 2004, 124, 605–611. [Google Scholar]
- Johnston, KL; Clifford, MN; Morgan, LM. Possible role for apple juice phenolic, compounds in the acute modification of glucose tolerance and gastrointestinal hormone secretion in humans. J. Sci. Food Agric 2002, 82, 1800–1805. [Google Scholar]
- Torronen, R; Sarkkinen, E; Tapola, N; Hautaniemi, E; Kilpi, K; Niskanen, L. Berries modify the postprandial plasma glucose response to sucrose in healthy subjects. Br J Nutr 2009. [Google Scholar]
- Wilson, T; Singh, AP; Vorsa, N; Goettl, CD; Kittleson, KM; Roe, CM; Kastello, GM; Ragsdale, FR. Human glycemic response and phenolic content of unsweetened cranberry juice. J. Med. Food 2008, 11, 46–54. [Google Scholar]
- Gin, H; Rigalleau, V; Caubet, O; Masquelier, J; Aubertin, J. Effects of red wine, tannic acid, or ethanol on glucose tolerance in non-insulin-dependent diabetic patients and on starch digestibility in vitro. Metabolism 1999, 48, 1179–1183. [Google Scholar]
- Holt, S; Jong, VD; Faramus, E; Lang, T; Brand Miller, J. A bioflavonoid in sugar cane can reduce the postprandial glycaemic response to a high-GI starchy food. Asia Pac. J. Clin. Nutr 2003, 12, S66. [Google Scholar]
- Hlebowicz, J; Darwiche, G; Bjorgell, O; Almer, LO. Effect of cinnamon on postprandial blood glucose, gastric emptying, and satiety in healthy subjects. Am. J. Clin. Nutr 2007, 85, 1552–1556. [Google Scholar]
- Hlebowicz, J; Hlebowicz, A; Lindstedt, S; Bjorgell, O; Hoglund, P; Holst, JJ; Darwiche, G; Almer, LO. Effects of 1 and 3 g cinnamon on gastric emptying, satiety, and postprandial blood glucose, insulin, glucose-dependent insulinotropic polypeptide, glucagon-like peptide 1, and ghrelin concentrations in healthy subjects. Am. J. Clin. Nutr 2009, 89, 815–821. [Google Scholar]
- Johnston, KL; Clifford, MN; Morgan, LM. Coffee acutely modifies gastrointestinal hormone secretion and glucose tolerance in humans: Glycemic effects of chlorogenic acid and caffeine. Am. J. Clin. Nutr 2003, 78, 728–733. [Google Scholar]
- van Dijk, AE; Olthof, MR; Meeuse, JC; Seebus, E; Heine, RJ; van Dam, RM. Acute effects of decaffeinated coffee and the major coffee components chlorogenic acid and trigonelline on glucose tolerance. Diabetes Care 2009, 32, 1023–1025. [Google Scholar]
- Thom, E. The effect of chlorogenic acid enriched coffee on glucose absorption in healthy volunteers and its effect on body mass when used long-term in overweight and obese people. J. Int. Med. Res 2007, 35, 900–908. [Google Scholar]
- Aldughpassi, A; Wolever, TM. Effect of coffee and tea on the glycaemic index of foods: No effect on mean but reduced variability. Br. J. Nutr 2009, 101, 1282–1285. [Google Scholar]
- Battram, DS; Arthur, R; Weekes, A; Graham, TE. The glucose intolerance induced by caffeinated coffee ingestion is less pronounced than that due to alkaloid caffeine in men. J. Nutr 2006, 136, 1276–1280. [Google Scholar]
- Moisey, LL; Kacker, S; Bickerton, AC; Robinson, LE; Graham, TE. Caffeinated coffee consumption impairs blood glucose homeostasis in response to high and low glycemic index meals in healthy men. Am. J. Clin. Nutr 2008, 87, 1254–1261. [Google Scholar]
- Bryans, JA; Judd, PA; Ellis, PR. The effect of consuming instant black tea on postprandial plasma glucose and insulin concentrations in healthy humans. J. Am. Coll. Nutr 2007, 26, 471–477. [Google Scholar]
- Rutter, GA. Nutrient-secretion coupling in the pancreatic islet beta-cell: Recent advances. Mol. Aspects Med 2001, 22, 247–284. [Google Scholar]
- Rutter, GA. Visualising insulin secretion. The Minkowski Lecture 2004. Diabetologia 2004, 47, 1861–1872. [Google Scholar]
- Chang-Chen, KJ; Mullur, R; Bernal-Mizrachi, E. Beta-cell failure as a complication of diabetes. Rev. Endocr Metab. Disord 2008, 9, 329–343. [Google Scholar]
- Choi, MS; Jung, UJ; Yeo, J; Kim, MJ; Lee, MK. Genistein and daidzein prevent diabetes onset by elevating insulin level and altering hepatic gluconeogenic and lipogenic enzyme activities in non-obese diabetic (NOD) mice. Diabetes Metab. Res. Rev 2008, 24, 74–81. [Google Scholar]
- Kim, DJ; Jeong, YJ; Kwon, JH; Moon, KD; Kim, HJ; Jeon, SM; Lee, MK; Park, YB; Choi, MS. Beneficial effect of chungkukjang on regulating blood glucose and pancreatic beta-cell functions in C75BL/KsJ-db/db mice. J. Med. Food 2008, 11, 215–223. [Google Scholar]
- Lu, MP; Wang, R; Song, X; Chibbar, R; Wang, X; Wu, L; Meng, QH. Dietary soy isoflavones increase insulin secretion and prevent the development of diabetic cataracts in streptozotocin-induced diabetic rats. Nutr. Res 2008, 28, 464–471. [Google Scholar]
- Cai, EP; Lin, JK. Epigallocatechin Gallate (EGCG) and rutin suppress the glucotoxicity through activating IRS2 and AMPK signaling in rat pancreatic beta cells. J Agric Food Chem 2009. [Google Scholar]
- Qa’dan, F; Verspohl, EJ; Nahrstedt, A; Petereit, F; Matalka, KZ. Cinchonain Ib isolated from Eriobotrya japonica induces insulin secretion in vitro and in vivo. J. Ethnopharmacol 2009, 124, 224–227. [Google Scholar]
- Adisakwattana, S; Moonsan, P; Yibchok-Anun, S. Insulin-releasing properties of a series of cinnamic acid derivatives in vitro and in vivo. J. Agric. Food Chem 2008, 56, 7838–7844. [Google Scholar]
- Liu, IM; Chen, WC; Cheng, JT. Mediation of beta-endorphin by isoferulic acid to lower plasma glucose in streptozotocin-induced diabetic rats. J. Pharmacol. Exp. Ther 2003, 307, 1196–1204. [Google Scholar]
- Fu, Z; Liu, D. Long-term exposure to genistein improves insulin secretory function of pancreatic beta-cells. Eur. J. Pharmacol 2009, 616, 321–327. [Google Scholar]
- Jayaprakasam, B; Vareed, SK; Olson, LK; Nair, MG. Insulin secretion by bioactive anthocyanins and anthocyanidins present in fruits. J. Agric. Food Chem 2005, 53, 28–31. [Google Scholar]
- Kawano, A; Nakamura, H; Hata, S; Minakawa, M; Miura, Y; Yagasaki, K. Hypoglycemic effect of aspalathin, a rooibos tea component from Aspalathus linearis, in type 2 diabetic model db/db mice. Phytomedicine 2009, 16, 437–443. [Google Scholar]
- Martineau, LC; Couture, A; Spoor, D; Benhaddou-Andaloussi, A; Harris, C; Meddah, B; Leduc, C; Burt, A; Vuong, T; Mai Le, P; Prentki, M; Bennett, SA; Arnason, JT; Haddad, PS. Anti-diabetic properties of the Canadian lowbush blueberry Vaccinium angustifolium Ait. Phytomedicine 2006, 13, 612–623. [Google Scholar]
- Kim, EK; Kwon, KB; Song, MY; Han, MJ; Lee, JH; Lee, YR; Lee, JH; Ryu, DG; Park, BH; Park, JW. Flavonoids protect against cytokine-induced pancreatic beta-cell damage through suppression of nuclear factor kappaB activation. Pancreas 2007, 35, e1–e9. [Google Scholar]
- Stanley Mainzen Prince, P; Kamalakkannan, N. Rutin improves glucose homeostasis in streptozotocin diabetic tissues by altering glycolytic and gluconeogenic enzymes. J. Biochem. Mol. Toxicol 2006, 20, 96–102. [Google Scholar]
- Kobori, M; Masumoto, S; Akimoto, Y; Takahashi, Y. Dietary quercetin alleviates diabetic symptoms and reduces streptozotocin-induced disturbance of hepatic gene expression in mice. Mol. Nutr. Food Res 2009, 53, 859–868. [Google Scholar]
- Coskun, O; Kanter, M; Korkmaz, A; Oter, S. Quercetin, a flavonoid antioxidant, prevents and protects streptozotocin-induced oxidative stress and beta-cell damage in rat pancreas. Pharmacol. Res 2005, 51, 117–123. [Google Scholar]
- Vessal, M; Hemmati, M; Vasei, M. Antidiabetic effects of quercetin in streptozocin-induced diabetic rats. Comp. Biochem. Physiol. C. Toxicol. Pharmacol 2003, 135C, 357–364. [Google Scholar]
- Chakravarthy, BK; Gupta, S; Gode, KD. Functional beta cell regeneration in the islets of pancreas in alloxan induced diabetic rats by (−)-epicatechin. Life Sci 1982, 31, 2693–2697. [Google Scholar]
- Hii, CS; Howell, SL. Effects of flavonoids on insulin secretion and 45Ca2+ handling in rat islets of Langerhans. J. Endocrinol 1985, 107, 1–8. [Google Scholar]
- Zunino, SJ; Storms, DH; Stephensen, CB. Diets rich in polyphenols and vitamin A inhibit the development of type I autoimmune diabetes in nonobese diabetic mice. J. Nutr 2007, 137, 1216–1221. [Google Scholar]
- Hamden, K; Allouche, N; Damak, M; Elfeki, A. Hypoglycemic and antioxidant effects of phenolic extracts and purified hydroxytyrosol from olive mill waste in vitro and in rats. Chem. Biol. Interact 2009, 180, 421–432. [Google Scholar]
- Sharma, B; Balomajumder, C; Roy, P. Hypoglycemic and hypolipidemic effects of flavonoid rich extract from Eugenia jambolana seeds on streptozotocin induced diabetic rats. Food Chem. Toxicol 2008, 46, 2376–2383. [Google Scholar]
- Kang, YJ; Jung, UJ; Lee, MK; Kim, HJ; Jeon, SM; Park, YB; Chung, HG; Baek, NI; Lee, KT; Jeong, TS; Choi, MS. Eupatilin, isolated from Artemisia princeps Pampanini, enhances hepatic glucose metabolism and pancreatic beta-cell function in type 2 diabetic mice. Diabetes Res. Clin. Pract 2008, 82, 25–32. [Google Scholar]
- Krisanapun, C; Peungvicha, P; Temsiririrkkul, R; Wongkrajang, Y. Aqueous extract of Abutilon indicum Sweet inhibits glucose absorption and stimulates insulin secretion in rodents. Nutr. Res 2009, 29, 579–587. [Google Scholar]
- Esmaeili, MA; Zohari, F; Sadeghi, H. Antioxidant and protective effects of major flavonoids from Teucrium polium on beta-cell destruction in a model of streptozotocin-induced diabetes. Planta Med 2009, 75, 1418–1420. [Google Scholar]
- Zaid, H; Antonescu, CN; Randhawa, VK; Klip, A. Insulin action on glucose transporters through molecular switches, tracks and tethers. Biochem. J 2008, 413, 201–215. [Google Scholar]
- Bjornholm, M; Zierath, JR. Insulin signal transduction in human skeletal muscle: Identifying the defects in Type II diabetes. Biochem. Soc. Trans 2005, 33, 354–357. [Google Scholar]
- Uldry, M; Thorens, B. The SLC2 family of facilitated hexose and polyol transporters. Pflugers Arch 2004, 447, 480–489. [Google Scholar]
- Konrad, D; Somwar, R; Sweeney, G; Yaworsky, K; Hayashi, M; Ramlal, T; Klip, A. The antihyperglycemic drug alpha-lipoic acid stimulates glucose uptake via both GLUT4 translocation and GLUT4 activation: Potential role of p38 mitogen-activated protein kinase in GLUT4 activation. Diabetes 2001, 50, 1464–1471. [Google Scholar]
- Liu, W; Hsin, C; Tang, F. A molecular mathematical model of glucose mobilization and uptake. Math. Biosci 2009, 221, 121–129. [Google Scholar]
- Taniguchi, CM; Kondo, T; Sajan, M; Luo, J; Bronson, R; Asano, T; Farese, R; Cantley, LC; Kahn, CR. Divergent regulation of hepatic glucose and lipid metabolism by phosphoinositide 3-kinase via Akt and PKClambda/zeta. Cell. Metab 2006, 3, 343–353. [Google Scholar]
- Dugani, CB; Randhawa, VK; Cheng, AW; Patel, N; Klip, A. Selective regulation of the perinuclear distribution of glucose transporter 4 (GLUT4) by insulin signals in muscle cells. Eur. J. Cell Biol 2008, 87, 337–351. [Google Scholar]
- Saltiel, AR; Kahn, CR. Insulin signalling and the regulation of glucose and lipid metabolism. Nature 2001, 414, 799–806. [Google Scholar]
- Prabhakar, PK; Doble, M. Synergistic effect of phytochemicals in combination with hypoglycemic drugs on glucose uptake in myotubes. Phytomedicine 2009, 16, 1119–1126. [Google Scholar]
- Zhang, ZF; Li, Q; Liang, J; Dai, XQ; Ding, Y; Wang, JB; Li, Y. Epigallocatechin-3-O-gallate (EGCG) protects the insulin sensitivity in rat L6 muscle cells exposed to dexamethasone condition. Phytomedicine 2010, 17, 14–18. [Google Scholar]
- Park, CE; Kim, MJ; Lee, JH; Min, BI; Bae, H; Choe, W; Kim, SS; Ha, J. Resveratrol stimulates glucose transport in C2C12 myotubes by activating AMP-activated protein kinase. Exp. Mol. Med 2007, 39, 222–229. [Google Scholar]
- Deng, JY; Hsieh, PS; Huang, JP; Lu, LS; Hung, LM. Activation of estrogen receptor is crucial for resveratrol-stimulating muscular glucose uptake via both insulin-dependent and - independent pathways. Diabetes 2008, 57, 1814–1823. [Google Scholar]
- Breen, DM; Sanli, T; Giacca, A; Tsiani, E. Stimulation of muscle cell glucose uptake by resveratrol through sirtuins and AMPK. Biochem. Biophys. Res. Commun 2008, 374, 117–122. [Google Scholar]
- Lagouge, M; Argmann, C; Gerhart-Hines, Z; Meziane, H; Lerin, C; Daussin, F; Messadeq, N; Milne, J; Lambert, P; Elliott, P; Geny, B; Laakso, M; Puigserver, P; Auwerx, J. Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1alpha. Cell 2006, 127, 1109–1122. [Google Scholar]
- Milne, JC; et al. Small molecule activators of SIRT1 as therapeutics for the treatment of type 2 diabetes. Nature 2007, 450, 712–716. [Google Scholar]
- Fang, XK; Gao, J; Zhu, DN. Kaempferol and quercetin isolated from Euonymus alatus improve glucose uptake of 3T3-L1 cells without adipogenesis activity. Life Sci 2008, 82, 615–622. [Google Scholar]
- Zanatta, L; Rosso, A; Folador, P; Figueiredo, MS; Pizzolatti, MG; Leite, LD; Silva, FR. Insulinomimetic effect of kaempferol 3-neohesperidoside on the rat soleus muscle. J. Nat. Prod 2008, 71, 532–535. [Google Scholar]
- Vishnu Prasad, CN; Suma Mohan, S; Banerji, A; Gopalakrishnapillai, A. Kaempferitrin inhibits GLUT4 translocation and glucose uptake in 3T3-L1 adipocytes. Biochem. Biophys. Res. Commun 2009, 380, 39–43. [Google Scholar]
- Tzeng, YM; Chen, K; Rao, YK; Lee, MJ. Kaempferitrin activates the insulin signaling pathway and stimulates secretion of adiponectin in 3T3-L1 adipocytes. Eur. J. Pharmacol 2009, 607, 27–34. [Google Scholar]
- Jorge, AP; Horst, H; de Sousa, E; Pizzolatti, MG; Silva, FR. Insulinomimetic effects of kaempferitrin on glycaemia and on 14C-glucose uptake in rat soleus muscle. Chem. Biol. Interact 2004, 149, 89–96. [Google Scholar]
- Bazuine, M; van den Broek, PJ; Maassen, JA. Genistein directly inhibits GLUT4-mediated glucose uptake in 3T3-L1 adipocytes. Biochem. Biophys. Res. Commun 2005, 326, 511–514. [Google Scholar]
- Cao, H; Hininger-Favier, I; Kelly, MA; Benaraba, R; Dawson, HD; Coves, S; Roussel, AM; Anderson, RA. Green tea polyphenol extract regulates the expression of genes involved in glucose uptake and insulin signaling in rats fed a high fructose diet. J. Agric. Food Chem 2007, 55, 6372–6378. [Google Scholar]
- Pinent, M; Blay, M; Blade, MC; Salvado, MJ; Arola, L; Ardevol, A. Grape seed-derived procyanidins have an antihyperglycemic effect in streptozotocin-induced diabetic rats and insulinomimetic activity in insulin-sensitive cell lines. Endocrinology 2004, 145, 4985–4990. [Google Scholar]
- Montagut, G; Onnockx, S; Vaque, M; Blade, C; Blay, M; Fernandez-Larrea, J; Pujadas, G; Salvado, MJ; Arola, L; Pirson, I; Ardevol, A; Pinent, M. Oligomers of grape-seed procyanidin extract activate the insulin receptor and key targets of the insulin signaling pathway differently from insulin. J Nutr Biochem 2009. [Google Scholar]
- Cummings, E; Hundal, HS; Wackerhage, H; Hope, M; Belle, M; Adeghate, E; Singh, J. Momordica charantia fruit juice stimulates glucose and amino acid uptakes in L6 myotubes. Mol. Cell. Biochem 2004, 261, 99–104. [Google Scholar]
- Roffey, BW; Atwal, AS; Johns, T; Kubow, S. Water extracts from Momordica charantia increase glucose uptake and adiponectin secretion in 3T3-L1 adipose cells. J. Ethnopharmacol 2007, 112, 77–84. [Google Scholar]
- Kumar, R; Balaji, S; Uma, TS; Sehgal, PK. Fruit extracts of Momordica charantia potentiate glucose uptake and up-regulate Glut-4, PPAR gamma and PI3K. J. Ethnopharmacol 2009, 126, 533–537. [Google Scholar]
- Martin, LJ; Matar, C. Increase of antioxidant capacity of the lowbush blueberry (Vaccinium angustifolium) during fermentation by a novel bacterium from the fruit microflora. J. Sci. Food Agric 2005, 85, 1477–1484. [Google Scholar]
- Vuong, T; Martineau, LC; Ramassamy, C; Matar, C; Haddad, PS. Fermented Canadian lowbush blueberry juice stimulates glucose uptake and AMP-activated protein kinase in insulin-sensitive cultured muscle cells and adipocytes. Can. J. Physiol. Pharmacol 2007, 85, 956–965. [Google Scholar]
- Anderson, RA; Broadhurst, CL; Polansky, MM; Schmidt, WF; Khan, A; Flanagan, VP; Schoene, NW; Graves, DJ. Isolation and characterization of polyphenol type-A polymers from cinnamon with insulin-like biological activity. J. Agric. Food Chem 2004, 52, 65–70. [Google Scholar]
- Imparl-Radosevich, J; Deas, S; Polansky, MM; Baedke, DA; Ingebritsen, TS; Anderson, RA; Graves, DJ. Regulation of PTP-1 and insulin receptor kinase by fractions from cinnamon: Implications for cinnamon regulation of insulin signalling. Horm. Res 1998, 50, 177–182. [Google Scholar]
- Cao, H; Polansky, MM; Anderson, RA. Cinnamon extract and polyphenols affect the expression of tristetraprolin, insulin receptor, and glucose transporter 4 in mouse 3T3-L1 adipocytes. Arch. Biochem. Biophys 2007, 459, 214–222. [Google Scholar]
- Qin, B; Nagasaki, M; Ren, M; Bajotto, G; Oshida, Y; Sato, Y. Cinnamon extract (traditional herb) potentiates in vivo insulin-regulated glucose utilization via enhancing insulin signaling in rats. Diabetes Res. Clin. Pract 2003, 62, 139–148. [Google Scholar]
- Qin, B; Nagasaki, M; Ren, M; Bajotto, G; Oshida, Y; Sato, Y. Cinnamon extract prevents the insulin resistance induced by a high-fructose diet. Horm. Metab. Res 2004, 36, 119–125. [Google Scholar]
- Lee, MS; Kim, CH; Hoang, DM; Kim, BY; Sohn, CB; Kim, MR; Ahn, JS. Genistein-derivatives from Tetracera scandens stimulate glucose-uptake in L6 myotubes. Biol. Pharm. Bull 2009, 32, 504–508. [Google Scholar]
- Li, BG; Hasselgren, PO; Fang, CH. Insulin-like growth factor-I inhibits dexamethasone-induced proteolysis in cultured L6 myotubes through PI3K/Akt/GSK-3beta and PI3K/Akt/mTOR-dependent mechanisms. Int. J. Biochem. Cell Biol 2005, 37, 2207–2216. [Google Scholar]
- Purintrapiban, J; Suttajit, M; Forsberg, NE. Differential activation of glucose transport in cultured muscle cells by polyphenolic compounds from Canna indica L. Root. Biol. Pharm. Bull 2006, 29, 1995–1998. [Google Scholar]
- Chang, L; Chiang, SH; Saltiel, AR. Insulin signaling and the regulation of glucose transport. Mol. Med 2004, 10, 65–71. [Google Scholar]
- Cazarolli, LH; Zanatta, L; Alberton, EH; Figueiredo, MS; Folador, P; Damazio, RG; Pizzolatti, MG; Silva, FR. Flavonoids: Cellular and molecular mechanism of action in glucose homeostasis. Mini Rev. Med. Chem 2008, 8, 1032–1038. [Google Scholar]
- Cherrington, AD. Banting Lecture 1997. Control of glucose uptake and release by the liver in vivo. Diabetes 1999, 48, 1198–1214. [Google Scholar]
- Pilkis, SJ; Claus, TH. Hepatic gluconeogenesis/glycolysis: Regulation and structure/function relationships of substrate cycle enzymes. Annu. Rev. Nutr 1991, 11, 465–515. [Google Scholar]
- Dentin, R; Liu, Y; Koo, SH; Hedrick, S; Vargas, T; Heredia, J; Yates, J, III; Montminy, M. Insulin modulates gluconeogenesis by inhibition of the coactivator TORC2. Nature 2007, 449, 366–369. [Google Scholar]
- Lam, TK; Pocai, A; Gutierrez-Juarez, R; Obici, S; Bryan, J; Aguilar-Bryan, L; Schwartz, GJ; Rossetti, L. Hypothalamic sensing of circulating fatty acids is required for glucose homeostasis. Nat. Med 2005, 11, 320–327. [Google Scholar]
- Pocai, A; Lam, TK; Gutierrez-Juarez, R; Obici, S; Schwartz, GJ; Bryan, J; Aguilar-Bryan, L; Rossetti, L. Hypothalamic K(ATP) channels control hepatic glucose production. Nature 2005, 434, 1026–1031. [Google Scholar]
- Postic, C; Girard, J. Contribution of de novo fatty acid synthesis to hepatic steatosis and insulin resistance: Lessons from genetically engineered mice. J. Clin. Invest 2008, 118, 829–838. [Google Scholar]
- Stewart, LK; Wang, Z; Ribnicky, D; Soileau, JL; Cefalu, WT; Gettys, TW. Failure of dietary quercetin to alter the temporal progression of insulin resistance among tissues of C57BL/6J mice during the development of diet-induced obesity. Diabetologia 2009, 52, 514–523. [Google Scholar]
- Roghani, M; Baluchnejadmojarad, T. Hypoglycemic and hypolipidemic effect and antioxidant activity of chronic epigallocatechin-gallate in streptozotocin-diabetic rats. Pathophysiology 2010, 17, 55–59. [Google Scholar]
- Shrestha, S; Ehlers, SJ; Lee, JY; Fernandez, ML; Koo, SI. Dietary green tea extract lowers plasma and hepatic triglycerides and decreases the expression of sterol regulatory element-binding protein-1c mRNA and its responsive genes in fructose-fed, ovariectomized rats. J. Nutr 2009, 139, 640–645. [Google Scholar]
- Bose, M; Lambert, JD; Ju, J; Reuhl, KR; Shapses, SA; Yang, CS. The major green tea polyphenol, (−)-epigallocatechin-3-gallate, inhibits obesity, metabolic syndrome, and fatty liver disease in high-fat-fed mice. J. Nutr 2008, 138, 1677–1683. [Google Scholar]
- Wolfram, S; Raederstorff, D; Preller, M; Wang, Y; Teixeira, SR; Riegger, C; Weber, P. Epigallocatechin gallate supplementation alleviates diabetes in rodents. J. Nutr 2006, 136, 2512–2518. [Google Scholar]
- Collins, QF; Liu, HY; Pi, J; Liu, Z; Quon, MJ; Cao, W. Epigallocatechin-3-gallate (EGCG), a green tea polyphenol, suppresses hepatic gluconeogenesis through 5′-AMP-activated protein kinase. J. Biol. Chem 2007, 282, 30143–30149. [Google Scholar]
- Waltner-Law, ME; Wang, XL; Law, BK; Hall, RK; Nawano, M; Granner, DK. Epigallocatechin gallate, a constituent of green tea, represses hepatic glucose production. J. Biol. Chem 2002, 277, 34933–34940. [Google Scholar]
- Lin, CL; Lin, JK. Epigallocatechin gallate (EGCG) attenuates high glucose-induced insulin signaling blockade in human hepG2 hepatoma cells. Mol. Nutr. Food Res 2008, 52, 930–939. [Google Scholar]
- Ae Park, S; Choi, MS; Cho, SY; Seo, JS; Jung, UJ; Kim, MJ; Sung, MK; Park, YB; Lee, MK. Genistein and daidzein modulate hepatic glucose and lipid regulating enzyme activities in C57BL/KsJ-db/db mice. Life Sci 2006, 79, 1207–1213. [Google Scholar]
- Cederroth, CR; Vinciguerra, M; Gjinovci, A; Kühne, F; Klein, M; Cederroth, M; Caille, D; Suter, M; Neumann, D; James, RW; Doerge, DR; Wallimann, T; Meda, P; Foti, M; Rohner-Jeanrenaud, F; Vassalli, JD; Nef, S. Dietary phytoestrogens activate AMP-activated protein kinase with improvement in lipid and glucose metabolism. Diabetes 2008, 57, 1176–1185. [Google Scholar]
- Liang, H; Ward, WF. PGC-1alpha: A key regulator of energy metabolism. Adv. Physiol. Educ 2006, 30, 145–151. [Google Scholar]
- Kim, JA. Mechanisms underlying beneficial health effects of tea catechins to improve insulin resistance and endothelial dysfunction. Endocr Metab. Immune Disord. Drug Targets 2008, 8, 82–88. [Google Scholar]
- Jung, UJ; Lee, MK; Park, YB; Kang, MA; Choi, MS. Effect of citrus flavonoids on lipid metabolism and glucose-regulating enzyme mRNA levels in type-2 diabetic mice. Int. J. Biochem. Cell Biol 2006, 38, 1134–1145. [Google Scholar]
- Jung, UJ; Lee, MK; Jeong, KS; Choi, MS. The hypoglycemic effects of hesperidin and naringin are partly mediated by hepatic glucose-regulating enzymes in C57BL/KsJ-db/db mice. J. Nutr 2004, 134, 2499–2503. [Google Scholar]
- Purushotham, A; Tian, M; Belury, MA. The citrus fruit flavonoid naringenin suppresses hepatic glucose production from Fao hepatoma cells. Mol. Nutr. Food Res 2009, 53, 300–307. [Google Scholar]
- Ganjam, GK; Dimova, EY; Unterman, TG; Kietzmann, T. FoxO1 and HNF-4 are involved in regulation of hepatic glucokinase gene expression by resveratrol. J Biol Chem 2009. [Google Scholar]
- Frescas, D; Valenti, L; Accili, D. Nuclear trapping of the forkhead transcription factor FoxO1 via Sirt-dependent deacetylation promotes expression of glucogenetic genes. J. Biol. Chem 2005, 280, 20589–20595. [Google Scholar]
- Motta, MC; Divecha, N; Lemieux, M; Kamel, C; Chen, D; Gu, W; Bultsma, Y; McBurney, M; Guarente, L. Mammalian SIRT1 represses forkhead transcription factors. Cell 2004, 116, 551–563. [Google Scholar]
- Rodgers, JT; Puigserver, P. Fasting-dependent glucose and lipid metabolic response through hepatic sirtuin 1. Proc. Natl. Acad. Sci. USA 2007, 104, 12861–12866. [Google Scholar]
- Jung, EH; Kim, SR; Hwang, IK; Ha, TY. Hypoglycemic effects of a phenolic acid fraction of rice bran and ferulic acid in C57BL/KsJ-db/db mice. J. Agric. Food Chem 2007, 55, 9800–9804. [Google Scholar]
- Lin, CL; Huang, HC; Lin, JK. Theaflavins attenuate hepatic lipid accumulation through activating AMPK in human HepG2 cells. J. Lipid Res 2007, 48, 2334–2343. [Google Scholar]
- Hwang, JT; Kwon, DY; Yoon, SH. AMP-activated protein kinase: A potential target for the diseases prevention by natural occurring polyphenols. N. Biotechnol 2009, 26, 17–22. [Google Scholar]
- Zang, M; Xu, S; Maitland-Toolan, KA; Zuccollo, A; Hou, X; Jiang, B; Wierzbicki, M; Verbeuren, TJ; Cohen, RA. Polyphenols stimulate AMP-activated protein kinase, lower lipids, and inhibit accelerated atherosclerosis in diabetic LDL receptor-deficient mice. Diabetes 2006, 55, 2180–2191. [Google Scholar]
- Ajmo, JM; Liang, X; Rogers, CQ; Pennock, B; You, M. Resveratrol alleviates alcoholic fatty liver in mice. Am. J. Physiol. Gastrointest. Liver Physiol 2008, 295, G833–G842. [Google Scholar]
- Cheng, Z; Guo, S; Copps, K; Dong, X; Kollipara, R; Rodgers, JT; Depinho, RA; Puigserver, P; White, MF. Foxo1 integrates insulin signaling with mitochondrial function in the liver. Nat. Med 2009, 15, 1307–1311. [Google Scholar]
- Song, Y; Manson, JE; Buring, JE; Sesso, HD; Liu, S. Associations of dietary flavonoids with risk of type 2 diabetes, and markers of insulin resistance and systemic inflammation in women: A prospective study and cross-sectional analysis. J. Am. Coll. Nutr 2005, 24, 376–384. [Google Scholar]
- Murtaugh, MA; Jacobs, DR, Jr; Jacob, B; Steffen, LM; Marquart, L. Epidemiological support for the protection of whole grains against diabetes. Proc. Nutr. Soc 2003, 62, 143–149. [Google Scholar]
- de Munter, JS; Hu, FB; Spiegelman, D; Franz, M; van Dam, RM. Whole grain, bran, and germ intake and risk of type 2 diabetes: A prospective cohort study and systematic review. PLoS Med 2007, 4, e261. [Google Scholar]
- Pereira, MA; Parker, ED; Folsom, AR. Coffee consumption and risk of type 2 diabetes mellitus: An 11-year prospective study of 28 812 postmenopausal women. Arch. Int. Med 2006, 166, 1311–1316. [Google Scholar]
- Jing, Y; Han, G; Hu, Y; Bi, Y; Li, L; Zhu, D. Tea consumption and risk of type 2 diabetes: A meta-analysis of cohort studies. J. Gen. Intern. Med 2009, 24, 557–562. [Google Scholar]
- Polychronopoulos, E; Zeimbekis, A; Kastorini, CM; Papairakleous, N; Vlachou, I; Bountziouka, V; Panagiotakos, DB. Effects of black and green tea consumption on blood glucose levels in non-obese elderly men and women from Mediterranean Islands (MEDIS epidemiological study). Eur. J. Nutr 2008, 47, 10–16. [Google Scholar]
- van Dieren, S; Uiterwaal, CS; van der Schouw, YT; van der A, DL; Boer, JM; Spijkerman, A; Grobbee, DE; Beulens, JW. Coffee and tea consumption and risk of type 2 diabetes. Diabetologia 2009, 52, 2561–2569. [Google Scholar]
- Brown, AL; Lane, J; Coverly, J; Stocks, J; Jackson, S; Stephen, A; Bluck, L; Coward, A; Hendrickx, H. Effects of dietary supplementation with the green tea polyphenol epigallocatechin-3-gallate on insulin resistance and associated metabolic risk factors: Randomized controlled trial. Br. J. Nutr 2009, 101, 886–894. [Google Scholar]
- Fukino, Y; Shimbo, M; Aoki, N; Okubo, T; Iso, H. Randomized controlled trial for an effect of green tea consumption on insulin resistance and inflammation markers. J. Nutr. Sci. Vitaminol. (Tokyo) 2005, 51, 335–342. [Google Scholar]
- Nagao, T; Meguro, S; Hase, T; Otsuka, K; Komikado, M; Tokimitsu, I; Yamamoto, T; Yamamoto, K. A catechin-rich beverage improves obesity and blood glucose control in patients with type 2 diabetes. Obesity (Silver Spring) 2009, 17, 310–317. [Google Scholar]
- Grassi, D; Lippi, C; Necozione, S; Desideri, G; Ferri, C. Short-term administration of dark chocolate is followed by a significant increase in insulin sensitivity and a decrease in blood pressure in healthy persons. Am. J. Clin. Nutr 2005, 81, 611–614. [Google Scholar]
- Grassi, D; Desideri, G; Necozione, S; Lippi, C; Casale, R; Properzi, G; Blumberg, JB; Ferri, C. Blood pressure is reduced and insulin sensitivity increased in glucose-intolerant, hypertensive subjects after 15 days of consuming high-polyphenol dark chocolate. J. Nutr 2008, 138, 1671–1676. [Google Scholar]
- Muniyappa, R; Hall, G; Kolodziej, TL; Karne, RJ; Crandon, SK; Quon, MJ. Cocoa consumption for 2 wk enhances insulin-mediated vasodilatation without improving blood pressure or insulin resistance in essential hypertension. Am. J. Clin. Nutr 2008, 88, 1685–1696. [Google Scholar]
- Kar, P; Laight, D; Rooprai, HK; Shaw, KM; Cummings, M. Effects of grape seed extract in Type 2 diabetic subjects at high cardiovascular risk: A double blind randomized placebo controlled trial examining metabolic markers, vascular tone, inflammation, oxidative stress and insulin sensitivity. Diabet. Med 2009, 26, 526–531. [Google Scholar]
- Andersen, G; Koehler, P; Somoza, V. Postprandial glucose and free fatty acid response is improved by wheat bread fortified with germinated wheat seedlings. Curr. Topics Nutraceut. Res 2008, 6, 15–21. [Google Scholar]
- Nahas, R; Moher, M. Complementary and alternative medicine for the treatment of type 2 diabetes. Can. Fam. Physician 2009, 55, 591–596. [Google Scholar]
- Unno, K; Yamamoto, H; Maeda, K; Takabayashi, F; Yoshida, H; Kikunaga, N; Takamori, N; Asahina, S; Iguchi, K; Sayama, K; Hoshino, M. Protection of brain and pancreas from high-fat diet: Effects of catechin and caffeine. Physiol. Behav 2009, 96, 262–269. [Google Scholar]
- Zhang, HJ; Ji, BP; Chen, G; Zhou, F; Luo, YC; Yu, HQ; Gao, FY; Zhang, ZP; Li, HY. A combination of grape seed-derived procyanidins and gypenosides alleviates insulin resistance in mice and HepG2 cells. J. Food Sci 2009, 74, H1–H7. [Google Scholar]
- DeFuria, J; Bennett, G; Strissel, KJ; Perfield, JW, II; Milbury, PE; Greenberg, AS; Obin, MS. Dietary blueberry attenuates whole-body insulin resistance in high fat-fed mice by reducing adipocyte death and its inflammatory sequelae. J. Nutr 2009, 139, 1510–1516. [Google Scholar]
- Noriega-Lopez, L; Tovar, AR; Gonzalez-Granillo, M; Hernandez-Pando, R; Escalante, B; Santillan-Doherty, P; Torres, N. Pancreatic insulin secretion in rats fed a soy protein high fat diet depends on the interaction between the amino acid pattern and isoflavones. J. Biol. Chem 2007, 282, 20657–20666. [Google Scholar]
- Rivera, L; Moron, R; Sanchez, M; Zarzuelo, A; Galisteo, M. Quercetin ameliorates metabolic syndrome and improves the inflammatory status in obese Zucker rats. Obesity (Silver Spring) 2008, 16, 2081–2087. [Google Scholar]
- Seymour, M; Tanone, I; Lewis, S; Urcuyo-Llanes, D; Bolling, SF; Bennink, MR. Blueberry-enriched diets reduce metabolic syndrome and insulin resistance in rats. FASEB J. 2009, p. 23.
- Liu, IM; Tzeng, TF; Liou, SS; Lan, TW. Myricetin, a naturally occurring flavonol, ameliorates insulin resistance induced by a high-fructose diet in rats. Life Sci 2007, 81, 1479–1488. [Google Scholar]
- Kannappan, S; Anuradha, CV. Insulin sensitizing actions of fenugreek seed polyphenols, quercetin & metformin in a rat model. Indian J. Med. Res 2009, 129, 401–408. [Google Scholar]
- Tsai, HY; Wu, LY; Hwang, LS. Effect of a proanthocyanidin-rich extract from longan flower on markers of metabolic syndrome in fructose-fed rats. J. Agric. Food Chem 2008, 56, 11018–11024. [Google Scholar]
- Wu, LY; Juan, CC; Ho, LT; Hsu, YP; Hwang, LS. Effect of green tea supplementation on insulin sensitivity in Sprague-Dawley rats. J. Agric. Food Chem 2004, 52, 643–648. [Google Scholar]
- Bain, JR; Stevens, RD; Wenner, BR; Ilkayeva, O; Muoio, DM; Newgard, CB. Metabolomics applied to diabetes research: Moving from information to knowledge. Diabetes 2009, 58, 2429–2443. [Google Scholar]
- Iwai, K; Kim, MY; Onodera, A; Matsue, H. Alpha-glucosidase inhibitory and antihyperglycemic effects of polyphenols in the fruit of Viburnum dilatatum Thunb. J. Agric. Food Chem 2006, 54, 4588–4592. [Google Scholar]
- Tadera, K; Minami, Y; Takamatsu, K; Matsuoka, T. Inhibition of alpha-glucosidase and alpha-amylase by flavonoids. J. Nutr. Sci. Vitaminol. (Tokyo) 2006, 52, 149–153. [Google Scholar]
- Lo Piparo, E; Scheib, H; Frei, N; Williamson, G; Grigorov, M; Chou, CJ. Flavonoids for controlling starch digestion: Structural requirements for inhibiting human alpha-amylase. J. Med. Chem 2008, 51, 3555–3561. [Google Scholar]
- Kim, JS; Kwon, CS; Son, KH. Inhibition of alpha-glucosidase and amylase by luteolin, a flavonoid. Biosci. Biotechnol. Biochem 2000, 64, 2458–2461. [Google Scholar]
- Funke, I; Melzig, MF. Effect of different phenolic compounds on alpha-amylase activity: Screening by microplate-reader based kinetic assay. Pharmazie 2005, 60, 796–797. [Google Scholar]
- Narita, Y; Inouye, K. Kinetic analysis and mechanism on the inhibition of chlorogenic acid and its components against porcine pancreas alpha-amylase isozymes I and II. J. Agric. Food Chem 2009, 57, 9218–9225. [Google Scholar]
- McDougall, GJ; Shpiro, F; Dobson, P; Smith, P; Blake, A; Stewart, D. Different polyphenolic components of soft fruits inhibit alpha-amylase and alpha-glucosidase. J. Agric. Food Chem 2005, 53, 2760–2766. [Google Scholar]
- Lee, YA; Cho, EJ; Tanaka, T; Yokozawa, T. Inhibitory activities of proanthocyanidins from persimmon against oxidative stress and digestive enzymes related to diabetes. J. Nutr. Sci. Vitaminol. (Tokyo) 2007, 53, 287–292. [Google Scholar]
- Adisakwattana, S; Charoenlertkul, P; Yibchok-Anun, S. alpha-Glucosidase inhibitory activity of cyanidin-3-galactoside and synergistic effect with acarbose. J. Enzyme Inhib. Med. Chem 2009, 24, 65–69. [Google Scholar]
- Adisakwattana, S; Ngamrojanavanich, N; Kalampakorn, K; Tiravanit, W; Roengsumran, S; Yibchok-Anun, S. Inhibitory activity of cyanidin-3-rutinoside on alpha-glucosidase. J. Enzyme Inhib. Med. Chem 2004, 19, 313–316. [Google Scholar]
- Matsui, T; Ueda, T; Oki, T; Sugita, K; Terahara, N; Matsumoto, K. alpha-Glucosidase inhibitory action of natural acylated anthocyanins. 2. alpha-Glucosidase inhibition by isolated acylated anthocyanins. J. Agric. Food Chem 2001, 49, 1952–1956. [Google Scholar]
- Matsui, T; Ueda, T; Oki, T; Sugita, K; Terahara, N; Matsumoto, K. alpha-Glucosidase inhibitory action of natural acylated anthocyanins. 1. Survey of natural pigments with potent inhibitory activity. J. Agric. Food Chem 2001, 49, 1948–1951. [Google Scholar]
- Welsch, CA; Lachance, PA; Wasserman, BP. Effects of native and oxidized phenolic compounds on sucrase activity in rat brush border membrane vesicles. J. Nutr 1989, 119, 1737–1740. [Google Scholar]
- Hanamura, T; Hagiwara, T; Kawagishi, H. Structural and functional characterization of polyphenols isolated from acerola (Malpighia emarginata DC.) fruit. Biosci. Biotechnol. Biochem 2005, 69, 280–286. [Google Scholar]
- Lee, DS; Lee, SH. Genistein, a soy isoflavone, is a potent alpha-glucosidase inhibitor. FEBS Lett 2001, 501, 84–86. [Google Scholar]
- Adisakwattana, S; Chantarasinlapin, P; Thammarat, H; Yibchok-Anun, S. A series of cinnamic acid derivatives and their inhibitory activity on intestinal alpha-glucosidase. J. Enzyme Inhib. Med. Chem 2009, 24, 1194–1200. [Google Scholar]
- Chauhan, A; Gupta, S; Mahmood, A. Effect of tannic acid on brush border disaccharidases in mammalian intestine. Indian J. Exp. Biol 2007, 45, 353–358. [Google Scholar]
- Schafer, A; Hogger, P. Oligomeric procyanidins of French maritime pine bark extract (Pycnogenol) effectively inhibit alpha-glucosidase. Diabetes Res. Clin. Pract 2007, 77, 41–46. [Google Scholar]
- Ohno, T; Kato, N; Ishii, C; Shimizu, M; Ito, Y; Tomono, S; Kawazu, S. Genistein augments cyclic adenosine 3′5′-monophosphate(cAMP) accumulation and insulin release in MIN6 cells. Endocr. Res 1993, 19, 273–285. [Google Scholar]
- Liu, D; Zhen, W; Yang, Z; Carter, JD; Si, H; Reynolds, KA. Genistein acutely stimulates insulin secretion in pancreatic beta-cells through a cAMP-dependent protein kinase pathway. Diabetes 2006, 55, 1043–1050. [Google Scholar]
- Jonas, JC; Plant, TD; Gilon, P; Detimary, P; Nenquin, M; Henquin, JC. Multiple effects and stimulation of insulin secretion by the tyrosine kinase inhibitor genistein in normal mouse islets. Br. J. Pharmacol 1995, 114, 872–880. [Google Scholar]
- Sorenson, RL; Brelje, TC; Roth, C. Effect of tyrosine kinase inhibitors on islets of Langerhans: Evidence for tyrosine kinases in the regulation of insulin secretion. Endocrinology 1994, 134, 1975–1978. [Google Scholar]
- Hsu, FL; Liu, IM; Kuo, DH; Chen, WC; Su, HC; Cheng, JT. Antihyperglycemic effect of puerarin in streptozotocin-induced diabetic rats. J. Nat. Prod 2003, 66, 788–792. [Google Scholar]
- Li, Y; Kim, J; Li, J; Liu, F; Liu, X; Himmeldirk, K; Ren, Y; Wagner, TE; Chen, X. Natural anti-diabetic compound 1,2,3,4,6-penta-O-galloyl-D-glucopyranose binds to insulin receptor and activates insulin-mediated glucose transport signaling pathway. Biochem. Biophys. Res. Commun 2005, 336, 430–437. [Google Scholar]
- Pinto Mda, S; Kwon, YI; Apostolidis, E; Lajolo, FM; Genovese, MI; Shetty, K. Potential of Ginkgo biloba L. leaves in the management of hyperglycemia and hypertension using in vitro models. Bioresour. Technol 2009, 100, 6599–6609. [Google Scholar]
- Kashket, S; Paolino, VJ. Inhibition of salivary amylase by water-soluble extracts of tea. Arch. Oral Biol 1988, 33, 845–846. [Google Scholar]
- Koh, LW; Wong, LL; Loo, YY; Kasapis, S; Huang, D. Evaluation of different teas against starch digestibility by mammalian glycosidases. J. Agric. Food Chem 2009, 58, 148–154. [Google Scholar]
- Kusano, R; Andou, H; Fujieda, M; Tanaka, T; Matsuo, Y; Kouno, I. Polymer-like polyphenols of black tea and their lipase and amylase inhibitory activities. Chem. Pharm. Bull. (Tokyo) 2008, 56, 266–272. [Google Scholar]
- Kwon, YI; Apostolidis, E; Kim, YC; Shetty, K. Health benefits of traditional corn, beans, and pumpkin: In vitro studies for hyperglycemia and hypertension management. J. Med. Food 2007, 10, 266–275. [Google Scholar]
- Kwon, YI; Apostolidis, E; Shetty, K. In vitro studies of eggplant (Solanum melongena) phenolics as inhibitors of key enzymes relevant for type 2 diabetes and hypertension. Bioresour. Technol 2008, 99, 2981–2988. [Google Scholar]
- da Silva Pinto, M; Kwon, YI; Apostolidis, E; Lajolo, FM; Genovese, MI; Shetty, K. Functionality of bioactive compounds in Brazilian strawberry (Fragaria x ananassa Duch.) cultivars: Evaluation of hyperglycemia and hypertension potential using in vitro models. J. Agric. Food Chem 2008, 56, 4386–4392. [Google Scholar]
- Yao, Y; Sang, W; Zhou, M; Ren, G. Antioxidant and α-Glucosidase inhibitory activity of colored grains in china. J. Agric. Food Chem 2009, 58, 770–774. [Google Scholar]
- Kwon, Y; Apostolidis, E; Shetty, K. Inhibitory potential of wine and tea against alpha-amylase and alpha-glucosidase for management of hyperglycemia linked to type 2 diabetes. J. Food Biochem 2008, 32, 15–31. [Google Scholar]
- Nordentoft, I; Jeppesen, PB; Hong, J; Abudula, R; Hermansen, K. Increased insulin sensitivity and changes in the expression profile of key insulin regulatory genes and beta cell transcription factors in diabetic KKAy-mice after feeding with a soy bean protein rich diet high in isoflavone content. J. Agric. Food Chem 2008, 56, 4377–4385. [Google Scholar]
- Roffey, B; Atwal, A; Kubow, S. Cinnamon water extracts increase glucose uptake but inhibit adiponectin secretion in 3T3-L1 adipose cells. Mol. Nutr. Food Res 2006, 50, 739–745. [Google Scholar]
- Khan, SA; Priyamvada, S; Arivarasu, NA; Khan, S; Yusufi, AN. Influence of green tea on enzymes of carbohydrate metabolism, antioxidant defense, and plasma membrane in rat tissues. Nutrition 2007, 23, 687–695. [Google Scholar]
- Govorko, D; Logendra, S; Wang, Y; Esposito, D; Komarnytsky, S; Ribnicky, D; Poulev, A; Wang, Z; Cefalu, WT; Raskin, I. Polyphenolic compounds from Artemisia dracunculus L. inhibit PEPCK gene expression and gluconeogenesis in an H4IIE hepatoma cell line. Am. J. Physiol. Endocrinol. Metab 2007, 293, E1503–E1510. [Google Scholar]
| Inhibiotion of α–Amylase activity | Inhibiotion of α–Glucosidase activity | Inhibition of glucose absorption from intestine | Protection of β-cells in cell culture | Protection of β-cells in islets or pancreas | Increased insulin secretion/content from cultured cells | Increased insulin secretion/content in isolated islets/pancreas | Improved GU in muscle cells | Improved GU in adypocytes | Induction of hepatic glucokinase activity | Supression of gluconeogenetic enzyme expression | Activation of hepatic AMPK | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Anthocyanins | ||||||||||||
| Cyanidin 3-galactoside | [191] | |||||||||||
| Cyanidin 3-rutinoside | [192] | |||||||||||
| Cyanidin 3-sambubioside | [183] | [183] | ||||||||||
| Cyanidin 3-glucoside | [70] | |||||||||||
| Acylated anthocyanins | [193,194] | |||||||||||
| Delphinidin 3-glucoside | [70] | |||||||||||
| Catechins | ||||||||||||
| Catechin | [43] | [33,37] | ||||||||||
| Epicatechin | [78] | [78,79] | ||||||||||
| Catechin gallate | [43] | |||||||||||
| Epi(gallo)catechin | [33,43,195] | [37] | ||||||||||
| Epi(gallo)catechin gallate | [41,184] | [35–37] | [65] | [65] | [95] | [135] | [135–137] | [136,138,151] | ||||
| Theaflavin | [151] | |||||||||||
| Theaflavin gallate | [41] | [151] | ||||||||||
| Theaflavin digallate | [151] | |||||||||||
| Flavonols, flavones, flavanones | [184] | |||||||||||
| Naringenin | [38] | |||||||||||
| Naringin | [143,144] | [143,144] | ||||||||||
| Quercetin | [184,185] | [43,184] | [37,39] | [73] | [76,77] | [73,76,79] | [101] | |||||
| Quercetin 3-glucoside | [34] | |||||||||||
| Quercetin 4’-glucoside | [34] | |||||||||||
| Quercetin 3-rhamnoside | [196] | |||||||||||
| Rutin | [65, 74] | [65] | [85] | |||||||||
| Myricetin | [184,185] | [184] | [37] | |||||||||
| Hesperidin | [143,144] | [144] | ||||||||||
| Apigenin | [37] | [73] | [73,85] | |||||||||
| Luteolin | [184–187] | [184,186] | [73] | [73] | ||||||||
| Luteolin 7-glucoside | [186] | [186] | ||||||||||
| Puerarin | [205] | |||||||||||
| Kaempferol | [101] | |||||||||||
| Kaempferol 3-neohesperidoside | [102] | |||||||||||
| Isoflavones | ||||||||||||
| Genistein | [184,197] | [69,201,202] | [62,69,202–204] | [139] | [62,139] | |||||||
| Daidzein | [184,186] | [62,139] | [62,139] | |||||||||
| 3,5-Diprenylgenistein | [120] | |||||||||||
| 6,8-Diprenylgenistein | [120] | |||||||||||
| Alpinumisoflavone | [120] | |||||||||||
| Derrone | [120] | |||||||||||
| Phenolic acids | ||||||||||||
| Caffeic acid | [188] | [43,195,198] | [33] | |||||||||
| Chlorogenic acid | [187,188] | [43,183] | [33,43] | [94] | ||||||||
| p-Coumaric acid | [195] | |||||||||||
| Ferulic acid | [195, 198] | [33] | [67] | [67] | [94] | [150] | ||||||
| Gallic acid | [195] | |||||||||||
| Tannic acid | [187] | [195,199] | [33] | |||||||||
| Ellagitannins | [189] | |||||||||||
| Proanthocyanidins | [190] | [190, 200] | ||||||||||
| Other phenolics | ||||||||||||
| Aspalathin | [71] | [71] | ||||||||||
| Penta-galloyl-glucose | [206] | |||||||||||
| Resveratrol | [96,98] | [154] |
| Inhibiotion of a–Amylase activity | Inhibiotion of a–Glucosidase activity | Inhibition of glucose absorption from intestine | Protection of beta-cells in cell culture | Protection of beta-cells in islets or pancreas | Increased insulin secretion/content from cultured cells | Increased insulin secretion/content in isolated islets/pancreas | Improved GU in muscle cells | Improved GU in adypocytes | Induction of hepatic glucokinase activity | Supression of gluconeogenetic enzyme expression | Activation of hepatic AMPK | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acerola | [196] | [42] | ||||||||||
| Berries; strawberry, raspberry, blueberry, blackcurrant | [189] | [189,213] | ||||||||||
| Blueberry | [72] | [72] | [114] | [114] | ||||||||
| Black rice | [214] | |||||||||||
| Rice bran | [150] | |||||||||||
| Gingko biloba | [44,207] | [44,207] | ||||||||||
| Red wine | [189] | [215] | ||||||||||
| Tea green, black | [189,208, 209,210] | [209,215] | [181] | [218] | [218] | |||||||
| Vegetables; pumpkin, beans, maize, eggplant | [211,212] | [211,212] | ||||||||||
| Soy | [64] | [63,64,216] | [63] | [63] | [140] | |||||||
| Grape | [80] | |||||||||||
| Grape seed | [108, 109] | [108] | [173] | |||||||||
| Bitter melon | [110] | [111] | ||||||||||
| Canna indica root | [122] | |||||||||||
| Cinnamon | [217] | |||||||||||
| Artemisia dracunculus | [219] |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hanhineva, K.; Törrönen, R.; Bondia-Pons, I.; Pekkinen, J.; Kolehmainen, M.; Mykkänen, H.; Poutanen, K. Impact of Dietary Polyphenols on Carbohydrate Metabolism. Int. J. Mol. Sci. 2010, 11, 1365-1402. https://doi.org/10.3390/ijms11041365
Hanhineva K, Törrönen R, Bondia-Pons I, Pekkinen J, Kolehmainen M, Mykkänen H, Poutanen K. Impact of Dietary Polyphenols on Carbohydrate Metabolism. International Journal of Molecular Sciences. 2010; 11(4):1365-1402. https://doi.org/10.3390/ijms11041365
Chicago/Turabian StyleHanhineva, Kati, Riitta Törrönen, Isabel Bondia-Pons, Jenna Pekkinen, Marjukka Kolehmainen, Hannu Mykkänen, and Kaisa Poutanen. 2010. "Impact of Dietary Polyphenols on Carbohydrate Metabolism" International Journal of Molecular Sciences 11, no. 4: 1365-1402. https://doi.org/10.3390/ijms11041365
APA StyleHanhineva, K., Törrönen, R., Bondia-Pons, I., Pekkinen, J., Kolehmainen, M., Mykkänen, H., & Poutanen, K. (2010). Impact of Dietary Polyphenols on Carbohydrate Metabolism. International Journal of Molecular Sciences, 11(4), 1365-1402. https://doi.org/10.3390/ijms11041365






