In Vitro Efficacy of Myxococcus fulvus ANSM068 to Biotransform Aflatoxin B1
Abstract
:1. Introduction
2. Results and Discussion
2.1. AFB1 Biotransformation by M. fulvus ANSM068
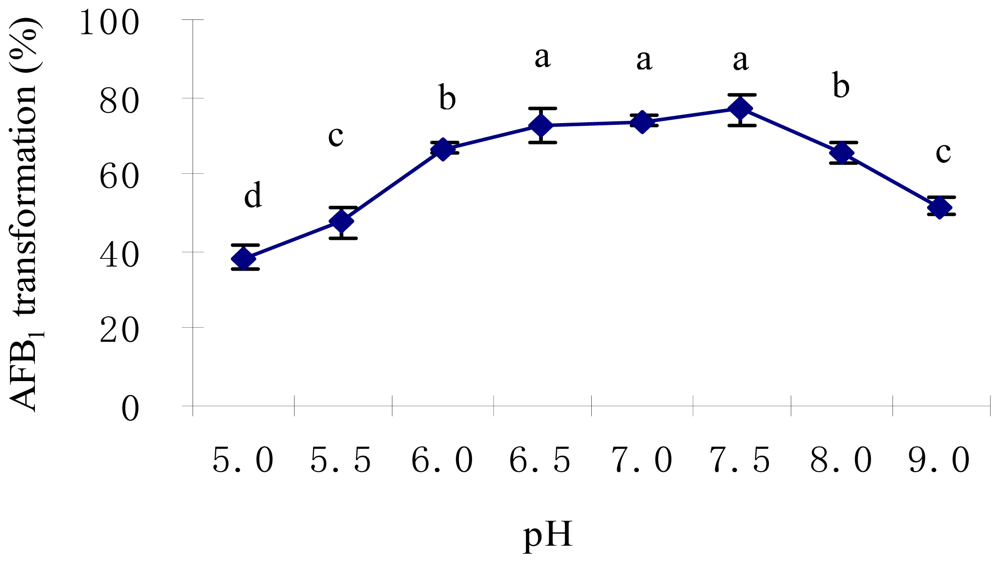
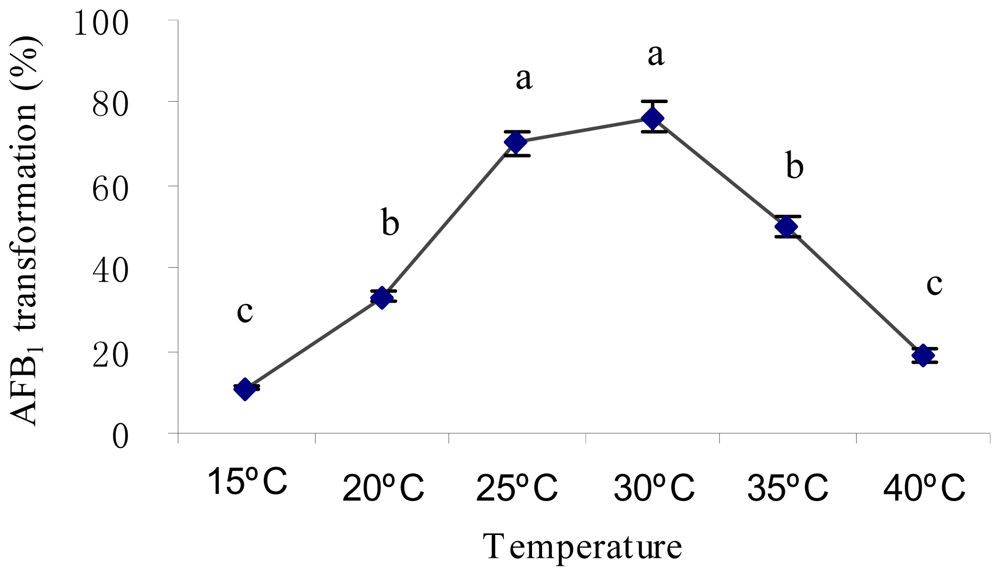
2.2. Optimal Culture Conditions for AFB1 Biotransformation
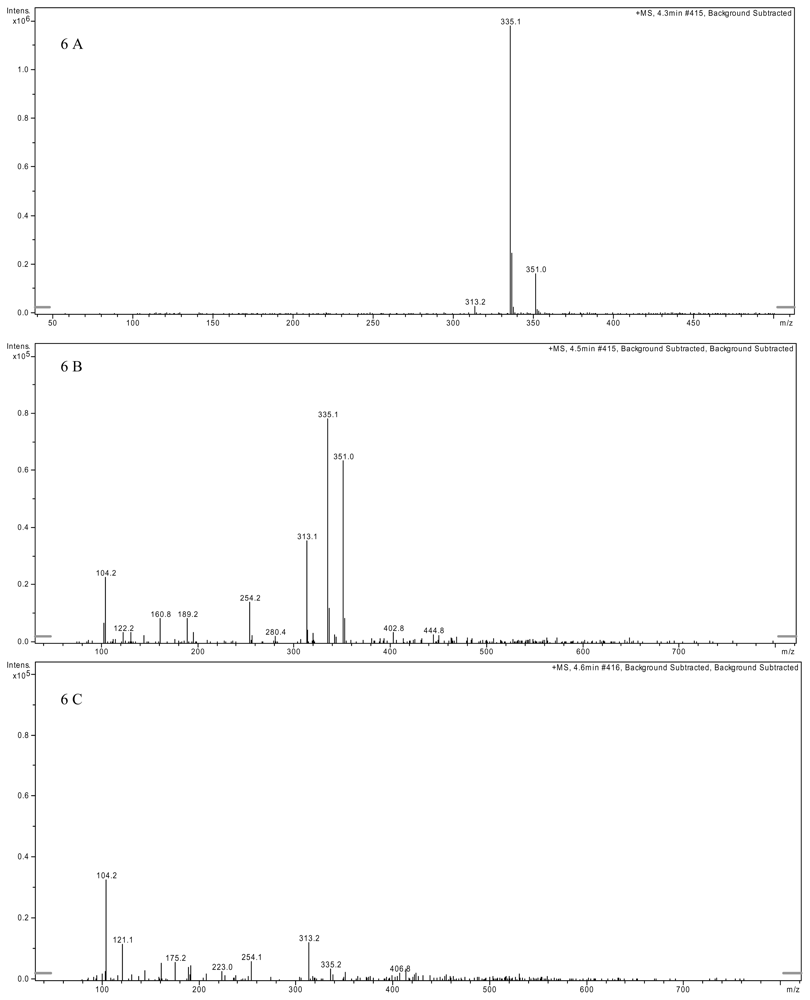
2.3. AFB1 Biotransformation Product Analysis
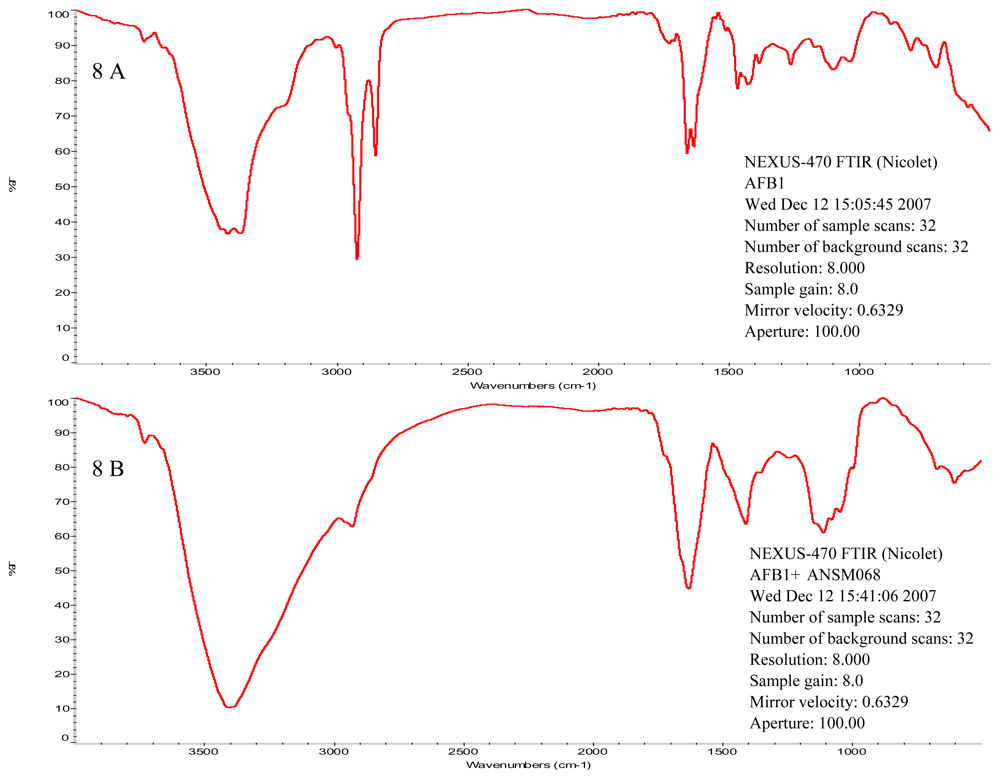
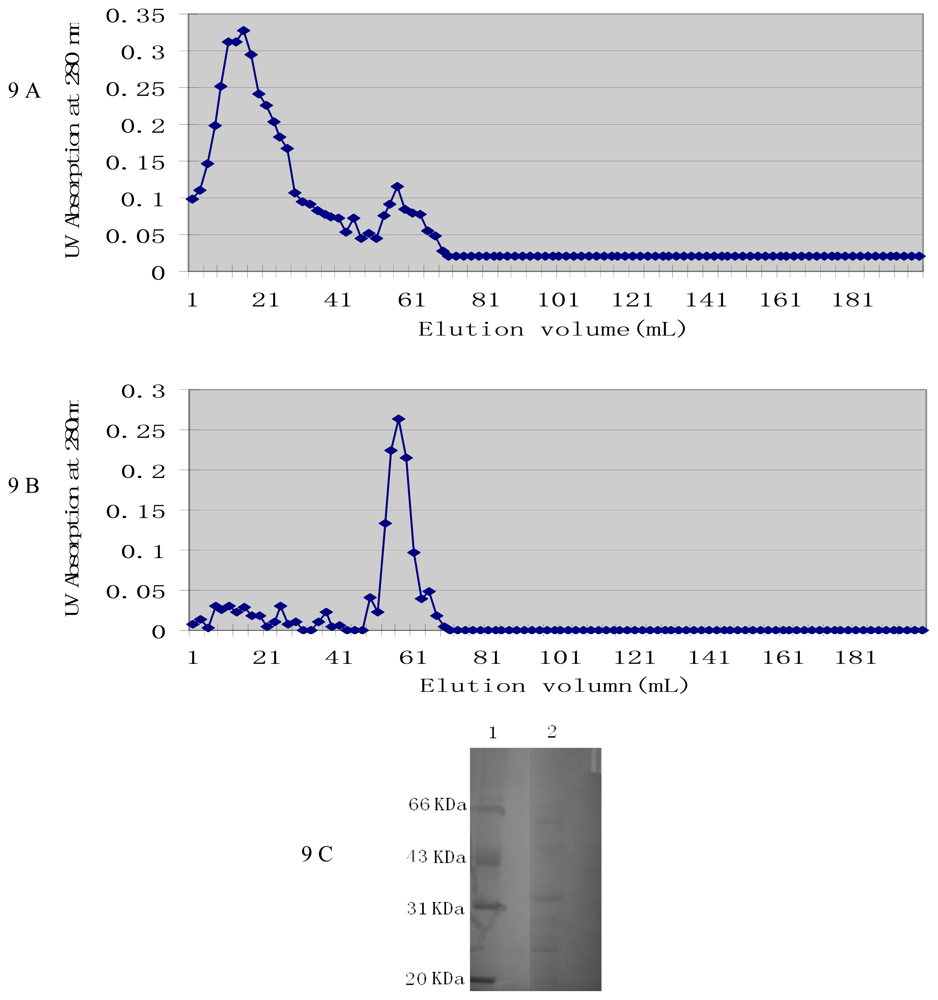
2.4. Characterization of Active Components from Culture Supernatant
3. Experimental Section
3.1. Bacterium, Mycotoxin and Medium
3.2. AFB1 Quantification
3.3. Efficacy of AFB1 Biotransformation by M. fulvus ANSM068
3.4. Culture Conditions Affecting AFB1 Biotransformation
3.5. AFB1 Biotransformation Product Analysis
3.6. Determination of Active Components from Culture Supernatant
3.6.1. Precipitation of Active Components
3.6.2. Purification of the Active Components by Chromatography
3.6.3. Sodium Dodecyl Sulphate-Polyacrylamide Gel Electrophoresis (SDS-PAGE)
3.7. Statistical Analyses
4. Conclusions
Acknowledgements
References
- Eaton, DL; Groopman, JD. The Toxicology of Aflatoxins. Human Health, Veterinary and Agricultural Significance; Academic Press: San Diego, CA, USA, 1994. [Google Scholar]
- Anonymous. Mycotoxins: Risks in Plant, Animal, and Human Systems; Council for Agricultural Science and Technology: Ames, IA, USA, 2003. [Google Scholar]
- Bhatnagar, D; Ehrlich, KC; Cleveland, TE. Molecular genetic analysis and regulation of aflatoxin biosynthesis. Appl. Microbiol. Biotechnol 2003, 61, 83–93. [Google Scholar]
- Marisa, HN; Das, C. A review on biological control and metabolism of aflatoxin. Crit. Rev. Food Sci 2003, 43, 245–264. [Google Scholar]
- Diaz, DE. The Mycotoxin Blue Book; Nottingham University Press: Nottingham, UK, 2005. [Google Scholar]
- Motomura, M; Toyomasu, T; Mizuno, K; Shinozawa, T. Purification and characterization of an aflatoxin degradation enzyme from Pleurotus ostreatus. Microbiol. Res 2003, 158, 237–242. [Google Scholar]
- Zjalic, S; Reverberi, M; Ricelli, A; Granito, VM; Fanelli, C; Fabbri, AA. Trametes versicolor: A possible tool for aflatoxin control. Int. J. Food Microbiol 2006, 107, 243–249. [Google Scholar]
- Varga, J; Peteri, Z; Tabori, K; Teren, J; Vagvolgyi, C. Degradation of ochratoxin A and other mycotoxins by Rhizopus isolates. Int. J. Food Microbiol 2005, 99, 321–328. [Google Scholar]
- Molnar, O; Schatzmayr, G; Elisabeth, F; Prillinger, H. Trichosporon mycotoxinivorans sp. nov., A new yeast species useful in biological detoxification of various mycotoxins. Syst. Appl. Microbiol 2004, 27, 661–671. [Google Scholar]
- Shetty, PH; Jespersen, L. Saccharomyces cerevisiae and lactic acid bacteria as potential mycotoxin decontaminating agents. Trends Food Sci. Tech 2006, 17, 48–55. [Google Scholar]
- Shantha, T. Fungal degradation of aflatoxin B1. Nat. Toxin 1999, 7, 175–178. [Google Scholar]
- Liu, DL; Yao, DS; Liang, R; Ma, L; Cheng, WQ; Gu, LQ. Detoxification of aflatoxin B1 by enzymes isolated from Armillariella tabescens. Food Chem. Toxicol 1998, 36, 563–574. [Google Scholar]
- El-Nezami, H; Kankaanpaa, P; Salminen, S; Ahogas, J. Ability of dairy strains of lactic acid bacteria to bind a common food carcinogen, aflatoxin B1. Food Chem. Toxicol 1998, 36, 321–326. [Google Scholar]
- Gratz, S; Mykkanen, H; El-Nezami, H. Aflatoxin B1 binding by a mixture of Lactobacillus and Propionibacterium: In vitro versus ex vivo. J. Food Prot 2005, 68, 2470–2474. [Google Scholar]
- Peltonen, K; El-Nezami, H; Haskard, C; Ahogas, J; Salminen, S. Aflatoxin B1 binding by dairy strains of lactic acid bacteria and bifidobacteria. J. Dairy Sci 2001, 84, 2152–2156. [Google Scholar]
- Peltonen, K; El-Nezami, H; Salminen, S; Ahogas, J. Binding of aflatoxin B1 by probiotic bacteria. J. Sci. Food Agric 2000, 80, 1942–1945. [Google Scholar]
- El-Nezami, H; Mykkanen, H; Kankaanpaa, P; Salminen, S; Ahokas, J. Ability of Lactobacillus and Propionibacterium strains to remove aflatoxin B1 from the chicken duodenum. J. Food Prot 2000, 63, 549–552. [Google Scholar]
- Pierides, M; El-nezami, H; Peltonen, K; Salminen, S; Ahokas, J. Ability of dairy strains of lactic acid bacteria to bind aflatoxin M1 in a food model. J. Food Prot 2000, 63, 645–650. [Google Scholar]
- Alberts, JF; Engelbrecht, Y; Steyn, PS; Holzapfel, WH; Vanzyl, WH. Biological degradation of aflatoxin B1 by Rhodococcus erythropolis cultures. Int. J. Food Microbiol 2006, 109, 121–126. [Google Scholar]
- Teniola, OD; Addo, PA; Brost, IM; Farber, P; Jany, KD; Alberts, JF; Vanzyl, WH; Steyn, PS; Holzapfel, WH. Degradation of aflatoxin B1 by cell-free extracts of Rhodococcus erythropolis and Mycobacterium fluoranthenivorans sp. nov. DSM 44556T. Int. J. Food Microbiol 2005, 105, 111–117. [Google Scholar]
- Hormisch, D; Brost, I; Kohring, GW; Giffhorn, F; Kroppensted, RM; Stackebrandt, E; Färber, P; Holzapfel, WH. Mycobacterium fluoranthenivorans sp. nov., a fluoranthene and aflatoxin B1 degrading bacterium from contaminated soil of a former coal gas plant. Syst. Appl. Microbiol 2004, 27, 653–660. [Google Scholar]
- Shu, G; Cheng, J; Ting, Z; Junxia, L; Qiugang, M; Tiangui, N. Aflatoxin B1 degradation by Stenotrophomonas Maltophilia and other microbes selected using coumarin medium. Int. J. Mol. Sci 2008, 9, 1489–1503. [Google Scholar]
- D'Souza, DH; Brackett, RE. The role of trace metal ions in aflatoxin B1 degradation by Flavobacterium aurantiacum. J. Food Prot 1998, 61, 1666–1669. [Google Scholar]
- D'Souza, DH; Brackett, RE. The influence of divalent cations and chelators on aflatoxin B1 degradation by Flavobacterium aurantiacum. J. Food Prot 2000, 63, 102–105. [Google Scholar]
- D'Souza, DH; Brackett, RE. Aflatoxin B1 degradation by Flavobacterium aurantiacum in the presence of reducing conditions and seryl and sulfhydryl group inhibitors. J. Food Prot 2001, 64, 268–271. [Google Scholar]
- Smiley, RD; Draughon, FA. Preliminary evidence that degradation of aflatoxin B1 by Flavobacterium aurantiacum is enzymatic. J. Food Prot 2000, 63, 415–418. [Google Scholar]
- Liu, DL; Yao, DS; Liang, YQ; Zhou, TH; Song, YP; Ma, L. Production, purification, and characterization of an intracellular aflatoxin-detoxifizyme from Armillariella tabescens (E-20). Food Chem. Toxicol 2001, 39, 461–466. [Google Scholar]
- Reichenbach, H; Dworkin, M. The myxobacteria. In The Prokaryote; Spriger-Verlag: New York, NY, USA, 1992; pp. 3418–3487. [Google Scholar]
- Reichenbach, H; Hoefle, G. Production of bioactive secondary metabolites. In Myxobacteria; American Society for Microbiology: Washington DC, USA, 1993; pp. 347–397. [Google Scholar]
- Reichenbach, H. Myxobacteria, producers of noval bioactive substances. J. Ind. Microbiol. Biot 2001, 27, 149–156. [Google Scholar]
- Rosenberg, E; Dworkin, M. Autocides and a paracide, antibiotic TA, produced by Myxoxcoccus xanthus. J. Ind. Microbiol. Biot 1996, 17, 424–431. [Google Scholar]
- Bode, HB; Irschik, H; Wenzel, SC; Reichenbach, H; Muller, R; Höfle, G. The leupyrrins: A structurally unique family of secondary metabolites from the myxobacterium Sorangium cellulosum. J. Nat. Prod 2003, 66, 1203–1206. [Google Scholar]
- Jansen, R; Kunze, B; Reichenbach, H; Hoefle, G. Antibiotics from gliding bacteria. Part 93. Chondrochloren A and B, new amino styrenes from Chondromyces crocatus (Myxobacteria). Eur. J. Org. Chem 2003, 14, 2684–2689. [Google Scholar]
- Sasse, F; Steinmetz, H; Hoefle, G. Archazolids, new cytotoxic macrolactones from Archangium gephyra (myxobacteria). Production, isolation, physico-chemical and biological properties. J. Antibiot 2003, 56, 520–525. [Google Scholar]
- Leibold, T; Sasse, F; Reichenbach, H; Höfle, G. Cyrmenins, novel antifungal peptides containing a nitrogen-Linked β-methoxyacrylate pharmacophore: Isolation and structural elucidation. Eur. J. Org. Chem 2004, 2, 431–435. [Google Scholar]
- Kunze, B; Reichenbach, H; Mueller, R; Höfle, G. Aurafuron A and B, new bioactive polyketides from Stigmatella aurantiaca and Archangium gephyra (myxobacteria). Fermentation, isolation, physico-chemical properties, structure and biological activity. J. Antibiot 2005, 58, 244–251. [Google Scholar]
- Reichenbach, H; Hoefle, G. Grabley, S, Ed.; Myxobacteria as producers of secondary metabolites. In Drug Discovery from Nature; Springer Verlag: Berlin, Germany, 1999; pp. 149–179. [Google Scholar]
- Gerth, K; Pradella, S; Perlova, O; Beyer, S; Müller, R. Myxobacteria: proficient producers of novel natural products with various biological activities-past and future biotechnological aspects with the focus on the genus Sorangium. J. Biotechnol 2003, 106, 233–253. [Google Scholar]
- Jansen, R; Kunze, B; Reichenbach, H; Hofle, G. Apicularen A and B, cytotoxic 10-membered lactones with a novel mechanism of action from chondromyces species (myxobacteria)-isolation, structure elucidation, and biosynthesis. Eur. J. Org. Chem 2000, 7, 913–919. [Google Scholar]
- Gupte, M; Kulkarni, P; Ganguli, BN. Antifungal antibiotics. Appl. Microbiol. Biotechnol 2002, 58, 46–57. [Google Scholar]
- Steinmetz, H; Glaser, N; Herdtweck, E; Sasse, F; Reichenbach, H; Hofle, G. Isolation, crystal and solution structure determination, and biosynthesis of tubulysins, powerful inhibitors of tubulin polymerization from myxobacteria. Angew. Chem. Int. Ed 2004, 43, 4888–4892. [Google Scholar]
- Szczepkowska, E. Exobasidium japonicum and myxococcus fulvus on pot grown azalea japonicad. Wszechswiat 1969, 9, 237–238. [Google Scholar]
- Chun, X. Screening of anti-fungal components from marine bacteria. Master thesis, Hua Nan Tropical Agricultural University, Guangzhou, China, 2003. [Google Scholar]
- David, W. Biology and global distribution of myxobacteria in soils. FEMS Microbiol. Rev 2000, 24, 403–427. [Google Scholar]
- Reichenbach, H. The ecology of the myxobacteria. Environ. Microbiol 1999, 1, 15–21. [Google Scholar]
- Ahn, JW; Jang, KH; Yang, HC; Oh, KB; Lee, HS; Shin, JH. Bithiazole metabolites from the myxobacterium Myxococcus fulvus. Chem. Pharm. Bull 2007, 55, 477–479. [Google Scholar]
- Lee, LS; Dunn, JJ; De Lucca, AJ; Ciegler, A. Role of lactone ring of aflatoxin B1 in toxicity and mutagenicity. Experientia 1981, 37, 16–17. [Google Scholar]
- Bol, J; Smith, JE. Biotransformation of aflatoxin. Food Biotechnol 1989, 3, 127–144. [Google Scholar]
- Natural Toxins, 49.4.01 Aflatoxin B1 in feed. In Official Methods of Analysis; AOAC: Washington, DC, USA, 2005; pp. 37–40.




© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Guan, S.; Zhao, L.; Ma, Q.; Zhou, T.; Wang, N.; Hu, X.; Ji, C. In Vitro Efficacy of Myxococcus fulvus ANSM068 to Biotransform Aflatoxin B1. Int. J. Mol. Sci. 2010, 11, 4063-4079. https://doi.org/10.3390/ijms11104063
Guan S, Zhao L, Ma Q, Zhou T, Wang N, Hu X, Ji C. In Vitro Efficacy of Myxococcus fulvus ANSM068 to Biotransform Aflatoxin B1. International Journal of Molecular Sciences. 2010; 11(10):4063-4079. https://doi.org/10.3390/ijms11104063
Chicago/Turabian StyleGuan, Shu, Lihong Zhao, Qiugang Ma, Ting Zhou, Ning Wang, Xinxu Hu, and Cheng Ji. 2010. "In Vitro Efficacy of Myxococcus fulvus ANSM068 to Biotransform Aflatoxin B1" International Journal of Molecular Sciences 11, no. 10: 4063-4079. https://doi.org/10.3390/ijms11104063
APA StyleGuan, S., Zhao, L., Ma, Q., Zhou, T., Wang, N., Hu, X., & Ji, C. (2010). In Vitro Efficacy of Myxococcus fulvus ANSM068 to Biotransform Aflatoxin B1. International Journal of Molecular Sciences, 11(10), 4063-4079. https://doi.org/10.3390/ijms11104063





