Amino Acid Synthesis in a Supercritical Carbon Dioxide - Water System
Abstract
:1. Introduction
2. Results and Discussion
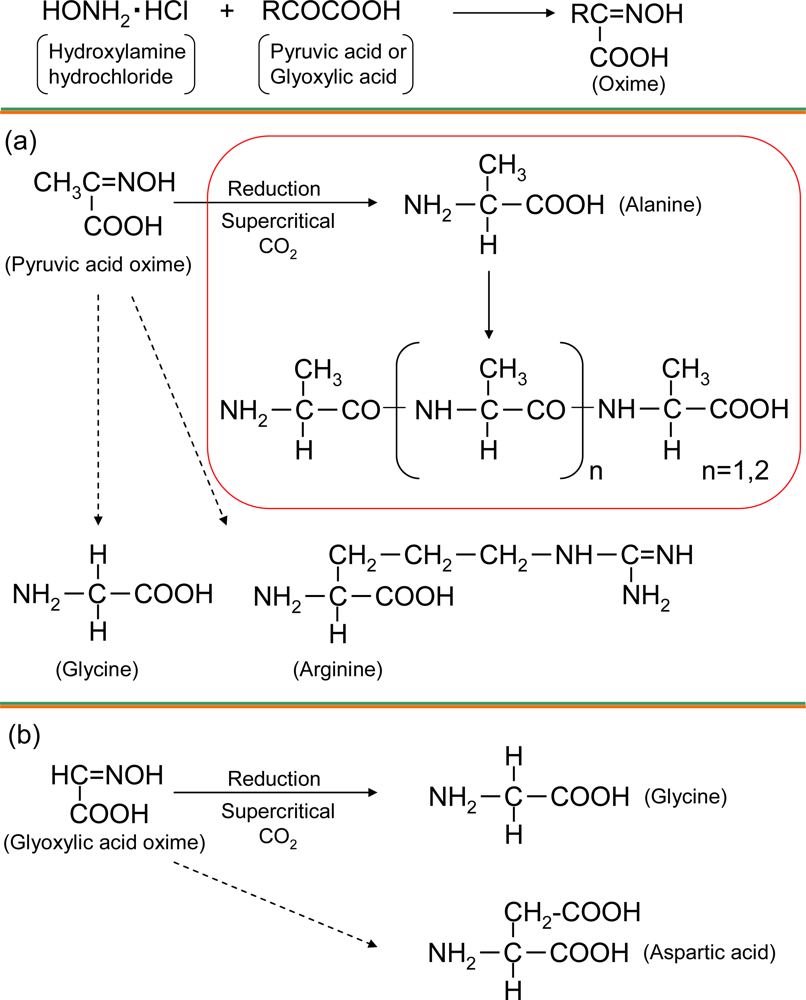
2.1. Amino acid synthesis in supercritical carbon dioxide
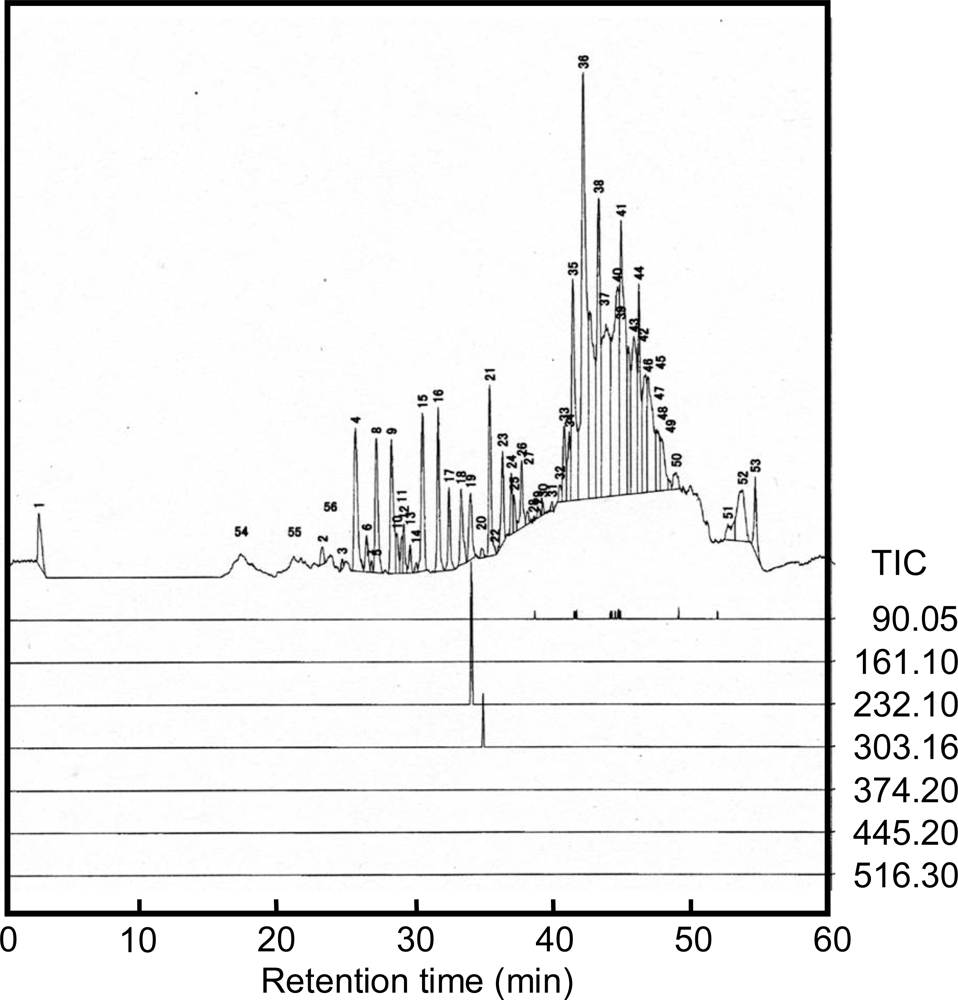
2.2. Analysis of alanine polymer
3. Experimental Section
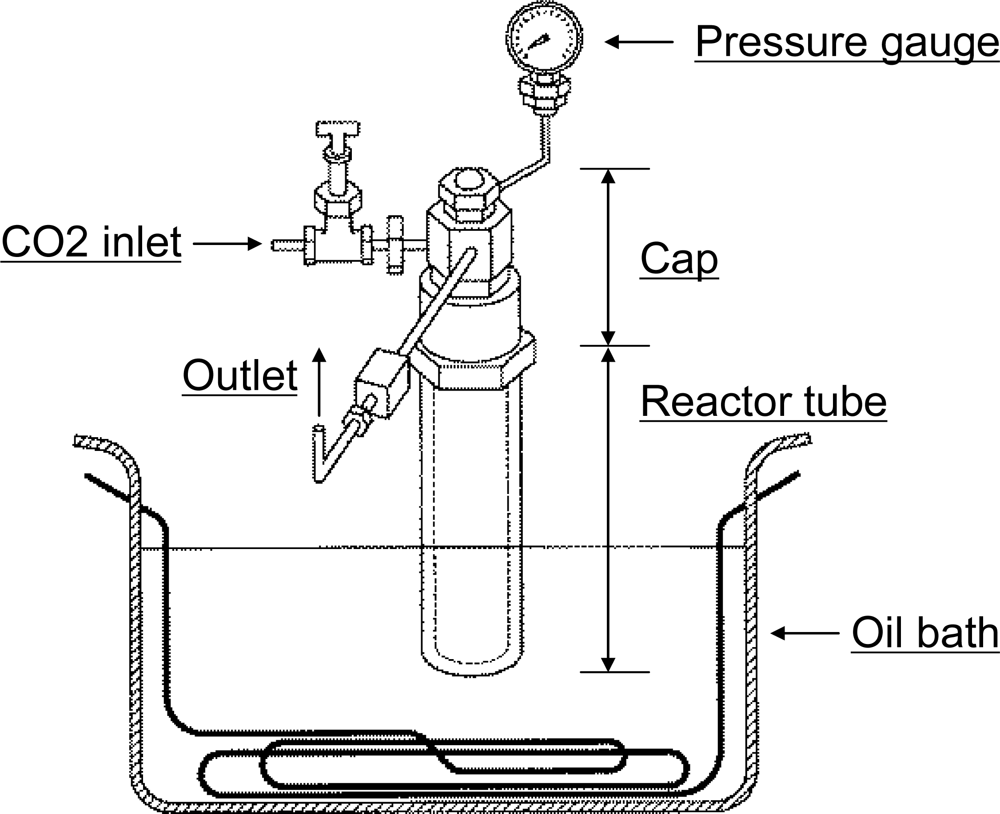
3.1. CO2 reaction conditions
3.2. Amino-acid analysis
3.3. LC-MS analysis
4. Conclusions
Acknowledgments
References and Notes
- Simoneit, BRT. Prebiotic organic synthesis under hydrothermal conditions: an overview. Adv. Space Res 2004, 33, 88–94. [Google Scholar]
- Miller, SL. A production of amino acids under possible primitive Earth conditions. Science 1953, 117, 528–529. [Google Scholar]
- Markhinin, EK; Podkletnov, NE. The phenomenon of formation of prebiological compounds in volcanic processes. Orig. Life 1977, 3, 225–235. [Google Scholar]
- Charlou, J; Donval, J. Hydrothermal methane venting between 12oN and 6oN along the Mid-Atlantic Ridge. J. Geophys. Res 1993, 98, 9625–9642. [Google Scholar]
- Gunter, BD. C1-C4 hydrocarbons in hydrothermal gases. Geochim. Cosmochim. Acta 1978, 42, 137–139. [Google Scholar]
- Simoneit, BRT; Kawka, OE; Brault, M. Origin of gasses and condensates in the Guaymas Basin hydrothermal system (Gulf of California). Chem. Geol 1988, 71, 169–182. [Google Scholar]
- Welhan, JA. Origins of methane in hydrothermal systems. Chem. Geol 1988, 71, 183–198. [Google Scholar]
- Welhan, JA; Lupton, JE. Light hydrocarbon gasses in Guaymas Basin hydrothermal fluids: Themogenic versus abiogenic origin. Am. Assoc. Petrol. Geol. Bull 1987, 71, 215–223. [Google Scholar]
- Furukawa, Y; Siekine, T; Oba, M; Kakegawa, T; Nakazawa, H. Biomolecule formation by oceanic impacts on early Earth. Nature Geosci 2009, 2, 62–66. [Google Scholar]
- Gelpi, E; Han, L; Nooner, DW; Oro, J. Closed system Fischer Tropsch synthesis over meteoric iron, iron ore and nickel iron alloy. Geochim. Cosmochim. Acta 1970, 34, 965–979. [Google Scholar]
- Studier, MH; Hayatsu, R; Anders, E. Origin of organic matter in early solar system (I. Hydrocarbons). Geochim. Cosmochim. Acta 1968, 32, 151–173. [Google Scholar]
- Kasting, JF. Bolide impacts and the oxidation of carbon in the Earth's early atmosphere. Orig. Life Evol. Biosphere 1989, 20, 199–231. [Google Scholar]
- Kobayashi, K; Tsuchiya, M; Oshima, T; Yanagawa, H. Abiotic synthesis of amino acids and imidazol by proton irradiation of simulated primitive Earth's early atmosphere. Orig. Life Evol. Biosphere 1990, 20, 99–109. [Google Scholar]
- McKay, DS; Gibson, EK; Thomas-Keprta, KL; Vali, H; Romanek, CS; Clemett, SJ; Chillier, XDF; Maechling, CR; Zare, RN. Search for Past Life on Mars: Possibble Relic Biogenic Activity in Martian Meteorite ALH84001. Science 1996, 273, 924–930. [Google Scholar]
- Sagan, C; Toon, OB; Gierasch, PJ. Climatic Change on Mars. Science 1973, 181, 1045–1049. [Google Scholar]
- Harder, H; Christensen, UR. A one-plume model of martian mantle convection. Nature 1996, 380, 507–509. [Google Scholar]
- Cody, GD; Boctor, NZ; Filley, TR; Hazen, RM; Scott, JH; Sharma, A; Yoder, HS, Jr. Primordial synthesis of carbonylated iron-sulfur clusters and the synthesis of pyruvate. Science 2000, 289, 1337–1340. [Google Scholar]
- Sokolskaya, A. Glycine and alanine synthesis from formaldehyde and hydroxylamine in the field of ultrasound waves. Orig. Life 1976, 7, 183–185. [Google Scholar]
- Hamlin, KE, Jr; Hartung, WH. The synthesis of α-amino acids from substituted acetoacetic esters. J. Biol. Chem 1942, 145, 349–357. [Google Scholar]
- Borszeky, K; Mallat, T; Aeschiman, R; Schweizer, WB; Baiker, A. Enantioselective Hydrogenation of Pyruvic Acid Oxime to Alanine on Pd/Alumina. J. Catalysis 1996, 161, 451–458. [Google Scholar]
- Ikehara, K. Origins of gene, genetic code, protein, and life. Viva Origino 2001, 29, 66–85. [Google Scholar]
- Johnson, AP; Cleaves, HJ; Dwokin, JP; Glavin, DP; Lazcano, A; Bada, JL. The miller volcanic spark discharge experiment. Science 2008, 322, 404. [Google Scholar]
- Goto, T; Futamura, Y; Yamaguchi, Y; Yamamoto, K. Condensation reaction of amino acids under hydrothermal conditions with adiabatic expansion cooling. J. Chem. Eng. Jpn 2005, 38, 295–299. [Google Scholar]
- Bixon, M; Scheraga, HA; Lifson, S. Effect of hydrophobic binding on the stability of poly-l-alanine helixes in water. Biopolymers 1963, 1, 419–429. [Google Scholar]
- Aleman, C; Roca, R; Luque, FJ; Orozco, M. Helical Preferences of alanine, glycine, and aminoisobutyric homopeptides. Proteins 1997, 28, 83–93. [Google Scholar]
- Miick, SM; Martinez, GV; Fiori, WR; Todd, AP; Millhouser, GL. Short alanine-based peptides may form 3(10)-helices and not alpha-helices in aqueous solution. Nature 1992, 359, 653–655. [Google Scholar]
- Fiori, WR; Miick, SM; Millhouser, GL. Increasing sequence length favors alpha-helix over 3(10)-helix in alanine-based peptides: Evidence for a length dependent structural transition. Biochemistry 1993, 32, 11957–11962. [Google Scholar]
- Hagiwara, M; Mitsui, H; Machi, S; Kagiya, T. Liquid carbon dioxide as a solvent for the radiation polymerization of ethylene. J. Polym. Sci. A-Polym. Chem 1968, 6, 603–608. [Google Scholar]
- Hagiwara, M; Mitsui, H; Machi, S; Kagiya, T. Kinetics of the γ-radiation-induced polymerization of ethylene in liquid carbon dioxide. J. Polym. Sci. A-Polym. Chem 1968, 6, 609–621. [Google Scholar]

| Primary Reactant | Temp. (ºC) | Products (μmol) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Before hydrolysis reaction | After hydrolysis reaction | ||||||||
| Ala | Gly | Asp | Arg | Ala | Gly | Asp | Arg | ||
| Pyruvic acid | 60 | 60.7 | 0.99 | — | — | 256.2 | 1.5 | — | 0.32 |
| 31 | 4.1 | 2.2 | — | — | 70.2 | 3.4 | — | — | |
| Glyoxylic acid | 60 | — | 866.3 | 2.5 | — | — | 819.2 | 1.8 | — |
| 31 | — | 183.3 | — | — | — | 209.2 | — | — | |
| Alanine monomer | Dimer | Trimer | Tetramer | Pentamer | Hexamer | Heptamer | |
|---|---|---|---|---|---|---|---|
| Calculated [M+H]+ | 90.05 | 161.09 | 232.13 | 303.16 | 374.20 | 445.24 | 516.28 |
| Retention time(min) | suspended | — | 33.7 | 34.6 | — | — | — |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fujioka, K.; Futamura, Y.; Shiohara, T.; Hoshino, A.; Kanaya, F.; Manome, Y.; Yamamoto, K. Amino Acid Synthesis in a Supercritical Carbon Dioxide - Water System. Int. J. Mol. Sci. 2009, 10, 2722-2732. https://doi.org/10.3390/ijms10062722
Fujioka K, Futamura Y, Shiohara T, Hoshino A, Kanaya F, Manome Y, Yamamoto K. Amino Acid Synthesis in a Supercritical Carbon Dioxide - Water System. International Journal of Molecular Sciences. 2009; 10(6):2722-2732. https://doi.org/10.3390/ijms10062722
Chicago/Turabian StyleFujioka, Kouki, Yasuhiro Futamura, Tomoo Shiohara, Akiyoshi Hoshino, Fumihide Kanaya, Yoshinobu Manome, and Kenji Yamamoto. 2009. "Amino Acid Synthesis in a Supercritical Carbon Dioxide - Water System" International Journal of Molecular Sciences 10, no. 6: 2722-2732. https://doi.org/10.3390/ijms10062722
APA StyleFujioka, K., Futamura, Y., Shiohara, T., Hoshino, A., Kanaya, F., Manome, Y., & Yamamoto, K. (2009). Amino Acid Synthesis in a Supercritical Carbon Dioxide - Water System. International Journal of Molecular Sciences, 10(6), 2722-2732. https://doi.org/10.3390/ijms10062722