1. Introduction
Phenols are toxic pollutants found in industrial wastewaters and that pose several risks to human health. Tyrosinase (EC 1.14.18.1) is a polyphenol oxidase found in several life forms, including the mushroom
Agaricus bisporus, with great potential as a biocatalyst for applications involving biomodification of phenols or bioremediation of phenol-polluted water [
1]. Kameda
et al. [
2] achieved 90% phenol removal in a synthetic phenol solution using mushroom tyrosinase. This enzyme requires molecular oxygen as a substrate, as described in
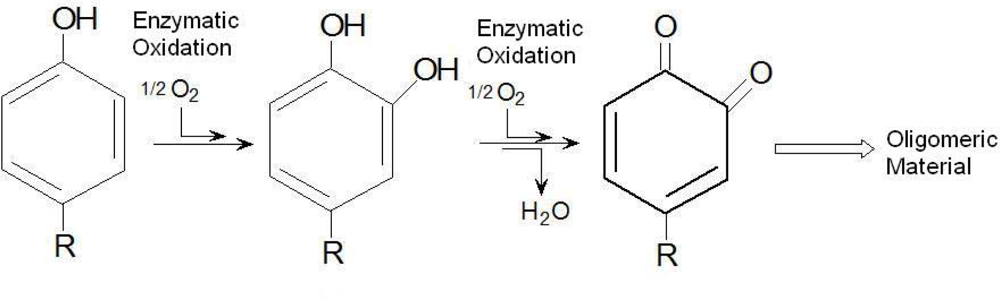
Scheme 1, which shows the enzymatic conversion of monophenols that involves their hydroxylation to form
o-hydroquinones, and then, dehydrogenation of the quinones to form
o-benzoquinones. The benzoquinones finally undergo a non-enzymatic polymerization to yield water-insoluble substances.
The low oxygen solubility in aqueous media could be a limiting factor for the enzyme-catalyzed oxidation by tyrosinase as the presence of oxygen is required, but increasing the oxygen content of the system by increasing the rate of aeration may shear inactivate the enzyme. Also elevated oxygen partial pressures may be toxic and inactivate the enzyme.
Supercritical fluids (SCFs) have been used in biocatalysis to great advantage. The first reports on the use of SCFs in biochemical reactions were published in 1985 [
3,
4]. Hammond
et al. [
3] have proved that tyrosinase could be used in SCCO
2. These preliminary studies indicated the possibility of enzymatic reactions in SCFs as the enzymes studied were able to retain their activity and stability in non-aqueous media. Since then, the use of supercritical CO
2 (SCCO
2) as a solvent for enzyme-catalyzed reactions has been a matter of considerable research interest because of its high diffusivity, low viscosity and low surface tension that can accelerate mass-transfer-limited enzymatic reactions [
5]. Moreover, the use of SCFs does not have the drawback of leaving solvent residues in the reaction products because SCF solvents are gases under atmospheric conditions. In particular, the low toxicity and reactivity of SCCO
2 make it attractive as non-aqueous reaction medium [
6], as well as the low supercritical conditions (critical temperature, Tc = 31.04 °C and critical pressure, Pc = 7.38 MPa). The most exploited property of SCCO
2 in the present work is its miscibility with other gases, such as oxygen, in the enzyme-catalyzed oxidation of phenol.
2. Results and Discussion
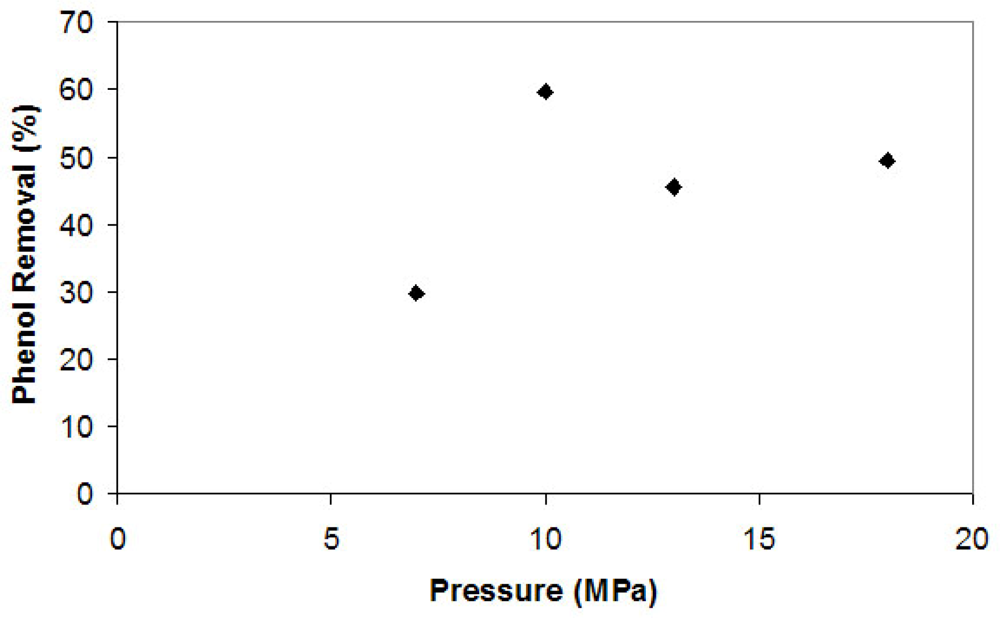
All reactions were performed in a two-phase (gas/liquid) reaction system with 210 mL of reaction medium in a 300-mL reactor. Preliminary studies were conducted in the apparatus shown in the Experimental Section at near critical conditions (35 °C, 7.00 MPa) to observe the performance of tyrosinase in the presence of CO2. After three hours, the enzymatic reaction under these conditions led to a 30% phenol removal. The presence of a limiting concentration of oxygen might be the reason for this low removal as the SCOO2 reactor is closed, avoiding the entrance of oxygen.
By raising the CO
2 partial pressure at 35 °C, it achieves supercritical conditions above 7.38 MPa, leading to a volumetric expansion in the liquid, which allows a higher solubility of other gases, such as the oxygen present in the reactor.
Figure 1 shows the results of phenol removal by tyrosinase under supercritical conditions at different pressures after a three hour reaction.
Initially, there was a raise in phenol removal as the partial pressure increased and then a slight reduction with increasing CO
2 partial pressure. There seems to be an optimum condition related to the supercritical CO
2. Probably, the results obtained were due to the solubility of both substrates and products in the SCCO
2. The solubility of water in high pressure CO
2 raises drastically at values near 10.0 MPa [
6]. Therefore, phenol removal increases in this pressure range as the amount of oxygen raises, assuming the solubility in water is constant.
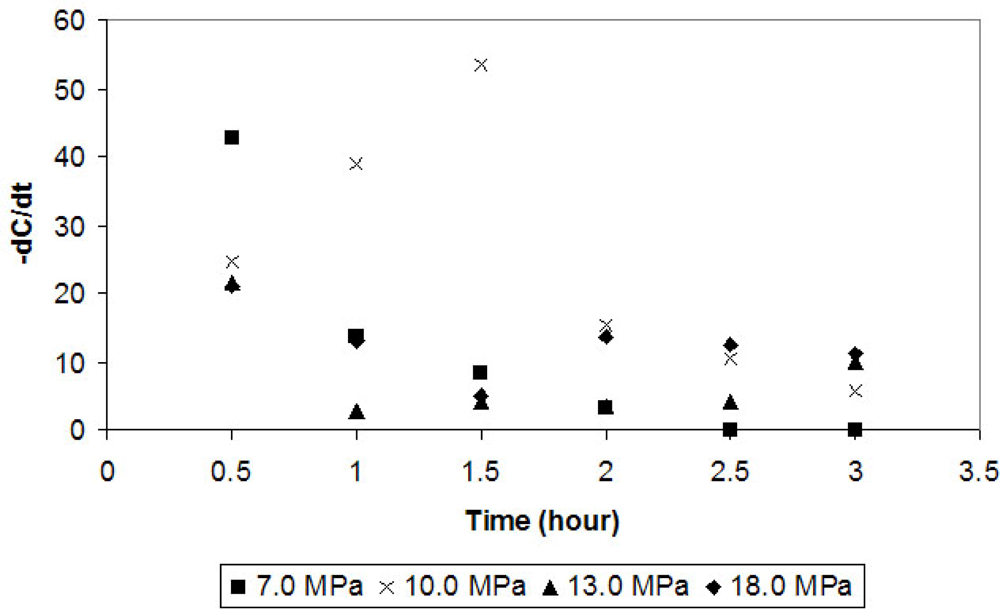
In order to study the progress of the enzymatic reaction, phenol instantaneous consumption rate (-dC/dt) was calculated. In
Figure 2 it is possible to observe that for most pressure conditions the instantaneous phenol consumption rate decreases with time. This might happen because of enzyme inhibition by the product or the consumption of the oxygen in the reactor. A major drawback in the application of tyrosinase is the reaction product inhibition (suicide inactivation) exhibited by this enzyme [
7]. Atlow
et al. [
1] used soluble tyrosinase to remove phenol from an aqueous synthetic waste solution; up to 99% conversion of the phenol was obtained, although, at higher concentrations (1.0 g/L), conversion was limited by inactivation of tyrosinase, likely by quinones formed during the reaction. Sun
et al. [
8] reported significant inactivation of soluble tyrosinase when phenol levels exceeded 0.05 g/L, likely because the quinones reacted with the free amino groups of the enzyme.
Under the best condition for phenol removal (10.0 MPa), there is a different behavior. Phenol instantaneous consumption rate raises until 1.5 hours of reaction (
Figure 2). The solubility of the product (in supercritical phase) that inactivates the enzyme must be different from the solubility of the enzyme (in supercritical phase) under this condition and, therefore, there is a greater yield of the reaction under this condition. Afterwards, there is a reduction of the phenol consumption rate, which might be due to the exhaustion of oxygen.
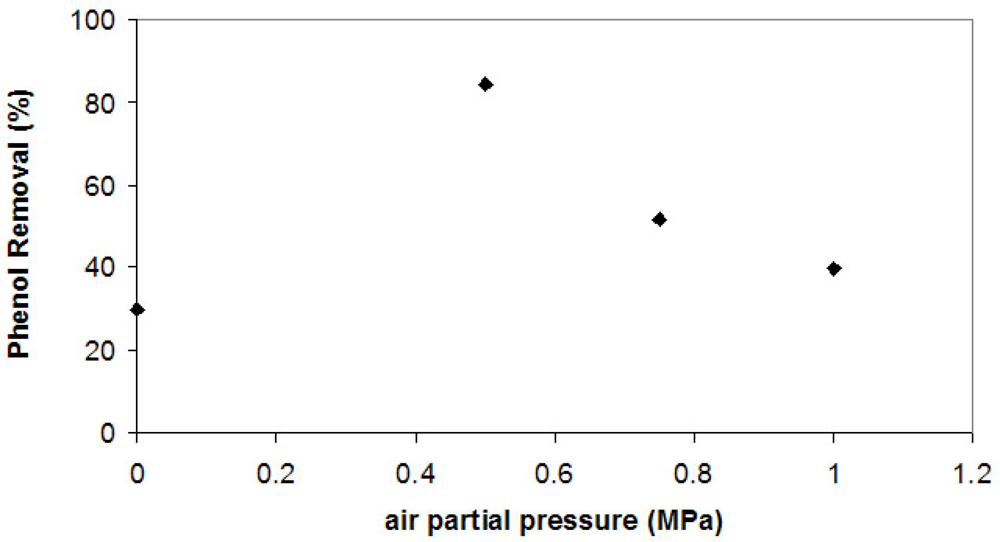
In order to raise the oxygen content of the reactor, an air compressor was added to the experimental apparatus shown in the Experimental. The air was added to the reactor after CO
2 reached a partial pressure of 7.00 MPa. Three different air partial pressures were used: 0.50, 0.75 and 1.00 MPa. The results are presented in
Figure 3.
The addition of air improved phenol removal by tyrosinase (from no air to 0.50 MPa), showing that one of the limitations of the reaction was the absence of oxygen. However, there is a maximum phenol removal with an air partial pressure of 0.50 MPa, and higher pressures reduced phenol removal.
Table 1 shows phenol instantaneous consumption rate (-dC/dt) of these experiments at the first 30 minutes of reaction. In comparison to the experiments without addition of air (
Figure 2), these rates are higher. Raising air partial pressure further (1.00 MPa), the initial phenol removal rate as well as the phenol removal diminish. Presumably, with more air available suicide inactivation becomes the limiting factor of the reaction as the phenol removal rate is slower.
3. Experimental Section
Tyrosinase crude extract was prepared from common mushrooms (
Agaricus bisporus) purchased in a local market (Rio de Janeiro, RJ, Brazil). The extraction consisted in triturating the mushrooms with different volumes of pre-cooled acetone, filtering the pulp, freezing it for 24 hours, suspending in 300 mL of distilled water, incubating the suspension overnight in a freezer, centrifuging the resultant suspension, and recovering the first extract. The suspension in water and centrifugation steps were repeated twice to obtain two more extracts [
9].
Tyrosinase activity was measured with a HACH DR/4000 UV-Vis spectrophotometer: a sample of the enzyme was added to a
l-tyrosine solution (1.2 mM) in a phosphate buffer (0.2 M, pH 6.0). A linear increase in absorbance at 280 nm was noted [
10]. Phenol was determined by a colorimetric assay [
11] based on the absorbance at 500 nm caused by the reaction between phenol, 4-aminoantipyrine and potassium ferricyanide. Phenol Removal was calculated by the equation below:
where C stands for phenol concentration, and f and i indicate final and initial, respectively.
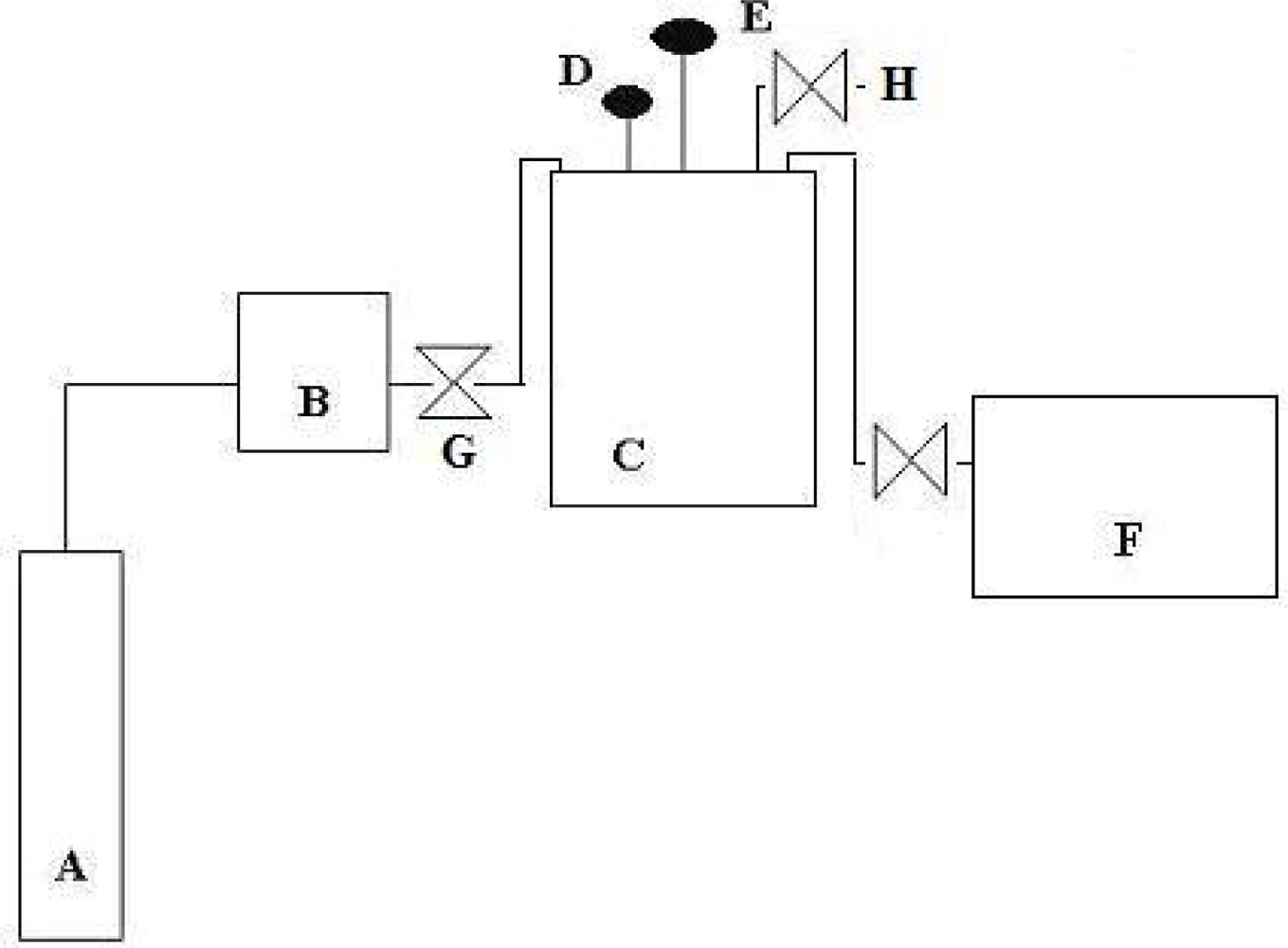
The experimental set-up, shown in
Figure 4, basically consisted of a CO
2 cylinder, a high pressure pump (ISCO 260D) and a 300-mL reactor (Autoclave Engineers, model 300BG) equipped with a mechanical stirrer (Mag-neDrive II), a heating mantle, an internal cooling loop capable of maintaining the temperature constant within 0.1°C and a pressure transducer. The enzymatic reaction involved a 210 mL-aqueous reaction mixture with 200 U/mL of tyrosinase and 100 mg/L of phenol that were placed in the reactor. The free-volume inside the reactor (90 mL) containing air, was the source of oxygen for the reaction. Afterwards, the reactor was closed, flushed and pressurized with CO
2. The substrates and enzyme were then continuously mixed with an agitation level of 1,250 rpm in order to provide a proper homogenization of the reacting mixture. The reaction time was 3 h and samples were withdrawn every 30 minutes.