Introduction
Many natural compounds posses a lactone ring in their structure [
1,
2]. Some lactones, particularly those with α,β-unsaturation, have properties of pharmacological interest, such as cardiac glycosides, sesquiterpene lactones, coumarins, polyketide-macrolide antibiotic, kawa styrylpyrones and some cytotoxic lignans [
3,
4,
5].
Some years ago, we isolated several polyhydroxylated lactones from
Passiflora foetida resin [
6], although the proposed structure was recently revised as a consequence of a stereospecific synthesis [
7]. In our search for antiparasite compounds derived from Colombian flora, we have now studied the leishmanicidal effects of passifloricin A and several hemisynthetic analogues and we report the structure of a new compound.
Results and Discussion
Structure of compounds
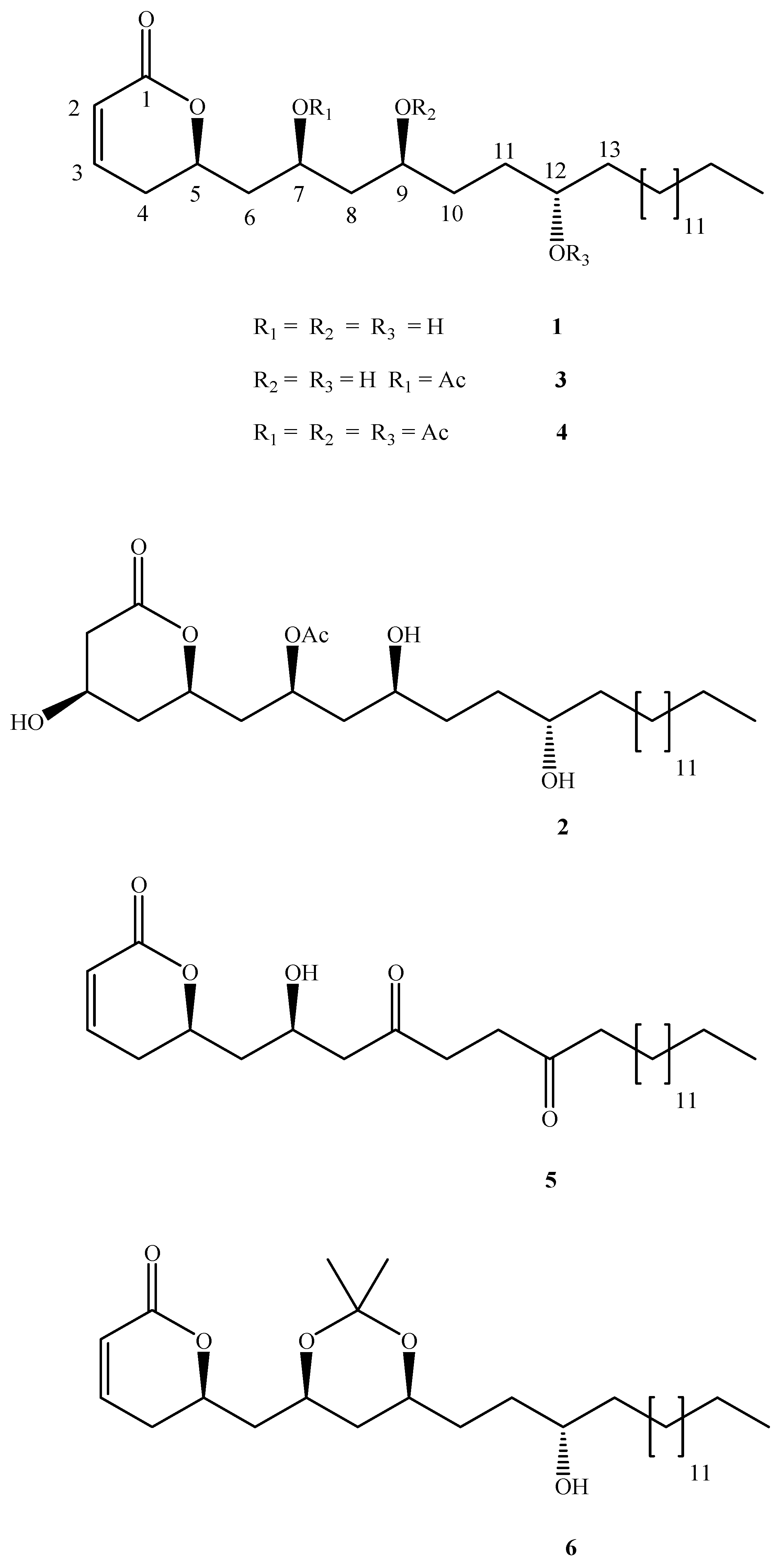
From the ethanolic resin extract and through repeated chromatography purification the known compounds
1-3 were isolated (
Figure 1).
Figure 1.
Structures of passifloricin A (1) and several derivatives
Figure 1.
Structures of passifloricin A (1) and several derivatives
Compounds 1 and 2 correspond to passifloricin A and C, respectively, while compound 3 is reported by first time. Its structure was elucidated as follows: the 1H- and 13C-NMR spectra were essentially similar to those of passifloricin A, in that they display two olefinic signals at δ 5.96 and δ 6.83, correlated to signals at δ 121.39 and δ 144.85 in a HMQC experiment. Long range coupling between these protons and the lactone carbonyl group at δ 163.95 was observed. In addition the signal at δ 6.83 showed correlations with the signal at δ 75.30, corresponding to lactone ring closure (C-5).
The DEPT experiment revealed four oxymethine carbon atoms at δ 69.18, δ 75.21, δ 75.30 and δ 78.65, assigned to C-7-, C-8, C-5 and C-12, according to COSY, HMQC and HMBC experiments. In addition an acetoxyl group was detected by signals at δ 2.03 (3H) in the 1H-NMR and a singlet and quartet at δ 170.75 and δ 21.18 in the 13C-NMR. The position of the acetoxyl group was assigned to C‑7, because of the long range correlation between the methyl group protons and the oxymethine carbon at δ 69.18; this signal was downfield with respect to that seen in passifloricin A.
Compounds
4-6 were prepared from passifloricin A through peracetylation, oxidation and isopropylidene derivatization. The
1H-NMR of compound
4 showed two singlet signals at δ 2.04 and δ 2.06 (3H and 6H respectively); the
13C-NMR spectra displayed signals at 170.61, 170.76 and 170.88 ppm, assignable to acetyl carbonyl groups. On the other hand, the
1H-NMR spectra of compound
5 lacks the signals at 3.68 and 3.98 ppm, but the
13C-NMR spectra contains two other signals at δ 210.2 and δ 218.4, corresponding to ketone carbonyl groups. The position of these functional groups was assigned on the basis of HMQC and HMBC experiments. Finally, JMOD of compound
6 showed two quaternary signals at δ 20.29 and δ 30.55 ppm and a singlet at δ 99.09, assignable to the acetonide group and typical of a syn configuration between hydroxyl groups. This compound had been already reported by us previously [
7].
Leishmanicidal activity
In vitro assays (
Table 1) showed a relationship between structure and leishmanicidal activity. Thus, passifloricin A (
1) was the more active compound (LD
50= 0.2 y ED
50 = 0.5), while changes in the hydroxyl substituents (compounds
3-6) cause slight decreases in the activity but the toxicity was reduced too. Activity against
Leishmania amastigotes practically disappears when passifloricin A is transformed into a Michael adduct (compound
2). Although antiparasitical activity levels were adequate, it would still be necessary to further transform the structures of the passifloricins to obtain a selectivity index (IS) near to a value of 20 for any such derivatives to be of practical use. Similarly the respective contributions of the lactone and hydroxyl groups should be established, since these may interact with a target through intra- or intermolecular H‑bonding.
Table 1.
In vitro activity of passifloricins
Table 1.
In vitro activity of passifloricins
| Compound | ED50 | LD50 | SIED50/LD50 |
|---|
| X | s | X | s |
|---|
| 1 | 0.5 | 0.1 | 0.2 | 0.02 | 2.9 |
| 2 | 24.0 | 0.8 | 10.3 | 0.05 | 2.3 |
| 3 | 2.8 | 0.1 | 0.6 | 0.01 | 4.4 |
| 4 | 3.5 | 0.1 | 0.6 | 0.02 | 5.9 |
| 5 | 15.0 | 2.0 | 3.5 | 0.1 | 4.2 |
| 6 | 2.6 | 0.1 | 1.4 | 0.0 | 1.9 |
| Glucantime | 6.7 | 1.5 | 416.4 | 0.2 | 59.6 |
Experimental
General
1H- and 13C-NMR spectra were recorded as CDCl3 solutions on a 300 MHz Bruker instrument. Chemical shifts (δ) are expressed in ppm with the protonated solvent peak as reference; coupling constants (J) are given in Hertz (Hz). Optical rotations were obtained (Na-D line) using a cell (1.5 mL) with 1dm path length on a SR-6 (Polyscience) polarimeter. UV spectra were recorded on a Genesis II instrument. Infrared spectra (IR) were recorded on a Perkin-Elmer RXI (FT-IR) apparatus. TLC were carried out on silica gel 60 F-254 aluminium sheets (Merck), followed by detection at 254 nm and with universal reagent. Column chromatography was performed using silica gel 60 (70-200 μm) (Merck).
Plant Material
Aerial parts of Passiflora foetida var. Hispida (Killip, E.P) were collected in Santa Fe de Antioquia (Colombia). Voucher specimens are deposited in the Herbario Universidad de Antioquia (HUA).
Extraction, isolation and reactions
Compounds
1 and
2 were obtained as previously reported [
7]; compound
3 was purified by fractionation on silica gel using mixtures of n-hexane-EtOAC (100% n-hexane to 100% EtOAC gradient). The last fraction was chromatographed with CH
2Cl
2-EtOAC (95:5). The
δ-lactone of (7S)-acetoxy-(2Z,5R,9S,12S)-trihydroxyhexacos-2-enoic acid (
3, 30.0 mg) was thus obtained as an amorphous white powder; m.p 66-68 °C; UV λ
max (CHCl
3, nm) 253; IR (KBr) ν (cm
–1): 3448, 2925, 2853, 1738, 1376, 1240; [α]
20 = + 29.48 (c = 0.42, CHCl
3);
1H-NMR: δ 6.83 (H
3,
dddd,
J = 9.74, 6.01, 2.69, 2.43) 5.96 ( H
2,
dd,
J = 9.77, 1.69) 5.13 (H
7,
m) 4.50 (H
5,
m) 3.92 (H
12,
t,
J = 6.03) 3.86 (H
9,
t,
J = 6.12) 2.52 (H
4,
dddd,
J = 18.39, 5.75, 5.57, 4.97) 2.18 – 2.41 (H
4`,
m) 2.05 - 2.18 (H
6,
m) 2.03 (3H,
s) 1.90 - 2.01 (H
6` and H
10,
m) 1.70 – 1.78 (H
8,
m) 1.32 – 1.53 (H
11 y H
13,
m) 1.11 – 1.31 (24H,
m) 0.82 (H
26,
t,
J = 6.28);
13C-NMR: δ 14.11 (C
26) 21.18 (CH
3) 22.68 (CH
2) 26.21 (CH
2) 29.27 (CH
2) 29.35 (C
4) 29.63 – 29.74 (CH
2) 31.92 (C
10) 32.61 (C
11) 35.95 (C
13) 40.16 (C
6) 41.03 (C
8) 69.18 (C
7) 75.21 (C
9) 75.30 (C
5) 78.65 (C
12) 121.39 (C
2) 144.86 (C
3) 163.95 (C
1=O) and 170.75 (C=O).
δ-Lactone of (7S,9S,12S)-triacetoxy-(2Z,5R)-hydroxyhexacos-2-enoic acid (4)
To a solution of passifloricin A (15 mg, 0.033 mmol) in pyridine was added an excess of acetic anhydride and the mixture was stirred at 25°C for 12h. After quenching with 1.0M aq. HCl (10 mL) the mixture was extracted with ethyl acetate (3 x 10 mL), the organic solution was washed with 1.0M NaHCO3 solution, dried over Na2SO4, filtered and evaporated. Purification of the residue by column chromatography (CH2Cl2 elution) gave compound 4 (17 mg, 88 %) as an amorphous white powder; m.p. 38–40 ºC; UV λmax (CHCl3, nm) 241; IR (KBr) ν (cm–1): 2918, 2855, 1732, 1240; [α]20 = + 27.82 (c = 0.34, CHCl3); 1H-NMR: δ 6.85 (H3, dddd, J = 9.77, 5.83, 3.10, 2.56) 6.01 (H2, dd, J = 9.82, 1.67) 5.01 – 5.09 (H7, m) 4.86 - 4.90 (H9, m) 4.82 (H12, t, J = 5.81) 4.48 (H5, ddd, J = 10.81, 6.52, 6.32) 2.97 (H6´, ddd, J = 7.56, 6.32, 3.58) 2.43 (H4, dt, J = 4.93) 2.33 (H4´, dt, J = 2.49) 2.09 – 2.20 (H6, m) 2.06 (6H, s) 2.04 (3H, s) 1.77 – 1.92 (H8, m) 1.31 – 1.62 (H10, H11 y H13, m) 1.15 – 1.30 (24H, m) 0.85 (H26, t, J = 6.3); 13C-NMR: δ 14.10 (C26) 21.16 (3CH3) 22.69 (CH2) 25.31 (CH2) 29.23 (CH2) 29.36 (CH2) 29.51 – 30.17 (8CH2) 31.94 (CH2) 34.11 (CH2) 38.93 (C8) 38.01 (C6) 67.85 (C7) 70.83 (C9) 73.91 (C12) 74.93 (C5) 121.44 (C2) 144.68 (C3) 163.72 (C1 = O) and 170.88 (C = O).
δ-Lactone of (9, 12)-dioxo-(2Z, 5R,7R)- dihidroxyhexacos-2-enoic acid (5)
To a solution of passifloricin A (15 mg, 0.033 mmol) in CH2Cl2 was added PDC (77 mg, 0.205 mmol) and 4Å molecules sieves (50 mg) and the mixture was stirred at 25°C for 6h. After quenching with 5% aq. HCl (10 mL) the mixture was extracted with ethyl acetate (3 x 10 mL), the organic solution was washed with saturated NaHCO3 solution, dried over Na2SO4, filtered and evaporated. Purification of the residue by column chromatography (CH2Cl2 elution) gave compound 5 (12 mg, 81 %) as an amorphous white powder; m.p. 70–72 ºC; UV λmax (CHCl3, nm) 241; IR (KBr) ν (cm‑1): 3420, 2910, 1691; [α]20 = + 25.33 (c = 0.38, CHCl3); 1H-NMR: δ 6.89 (H3, dt, J = 4.40) 6.03 (H2, dt, J = 1.83) 4.73 (H5, ddd, J = 12.89, 6.39, 2.46) 4.32 (H7, m) 2.05 (H6, dddd, J = 14.35, 8.08, 6.65, 6.42) 2.69 – 2.78 (H8, H10 and H11, m) 2.41 – 2.48 (H4 and H13, m) 1.50 – 1.61 (H14, m) 1.15 – 1.38 (24H, m) 0.85 (H26, t, J = 6.44); 13C-NMR: δ 14.11 (C26) 22.71 (CH2) 23.93 (C14) 29.20 (C13) 29.23 – 29.67 (9CH2) 31.95 (CH2) 36.21 (C11) 36.88 (C10) 40.74 (C6) 42.73 (C4) 49.31 (C8) 64.78 (C7) 75.56 (C5) 121.36 (C2) 145.17 (C3) 184.8 (C1=O) 210.2 (C9 = O) and 218.4 (C12 = O)
Leishmanicidal Assays
Leishmania panamensis promastigotes, M/HOM/87/UA140 strain, were grown at 28°C in Scheneider´s Drosophila medium (Sigma) supplemented with 10% (v/v) heat-inactivated fetal calf serum (FCS). After 48h of growth, U-937 cells [
8,
9] were washed twice with Dubelcco´s phosphate buffer saline (DPBS) (Gibco BRL). 100.000 cells/mL were exposed to stationary phase growth promastigotes at a ratio of 25 parasites/cell. The range of concentrations varied between 0.1 and 10 μg/mL, aseptically solubilized in DMSO. For each compound, three independent experiments were performed in triplicate. The percentage of infection was calculated by dividing the number of infected cells obtained in the presence of each compound by the number of infected cells obtained in the absence of treatment. Glucantime was used as the positive control. Results were expressed as ED
50 which was calculated by Probit analysis. The ED
50 of glucantime, used as the standard drug in these assays, was 6.7 μg/mL.
Cytotoxicity
Cytotoxicity was evaluated by the enzymatic micromethod on human promonocytic U-937 cells. Briefly, U-937 cells were introduced into 96-well flat-bottomed microtiter plates at a density of 5000 cells/100 μL. The cultures were incubated for 96h in the presence of various compound concentrations (3-100 μg/mL). The optical density at 570 nm was measured using an ELISA plate reader (Bio Rad). Cells cultivated in the absence of treatment but maintained under the same conditions were used as control. Three independent experiments were performed in triplicate for the determination of cytotoxicity of each compound. Results were expressed as LD50 and calculated by Probit analysis.