Polymer-Supported Cinchona Alkaloid-Derived Ammonium Salts as Recoverable Phase-Transfer Catalysts for the Asymmetric Synthesis of α-Amino Acids
Abstract
:Introduction
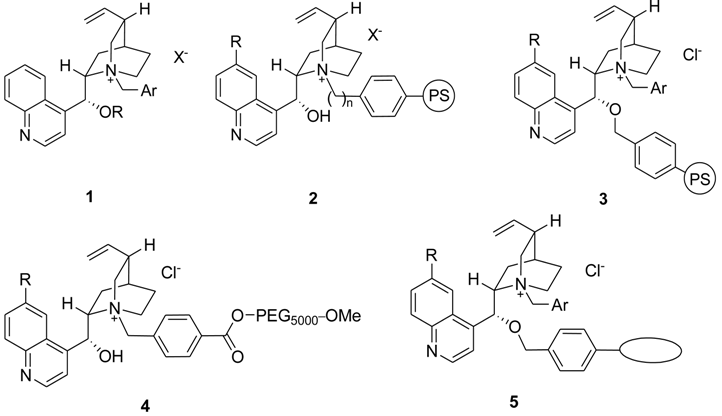
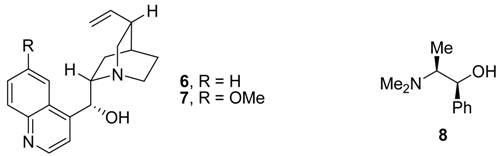
Results and Discussion


| Entry | Catalyst | Base | Solvent | T (ºC) | t (h) | Yielda (%) | eeb (%) |
|---|---|---|---|---|---|---|---|
| 1 | 2a | 25% NaOH | PhMe | 25 | 4 | 90 | 66 |
| 2 | 2a | 25% NaOH | PhMe | 0 | 17 | 90 | 90 |
| 3 | 2a | 25% KOH | PhMe:CHCl3 | -20 | 10 | 46 | 76 |
| 4 | 2a | 50% KOH | PhMe:CHCl3 | -40 | 9 | 56 | 85 |
| 5 | 2b | 25% NaOH | PhMe | 25 | 10 | 62 | 34 |
| 6 | 2b | 25% NaOH | PhMe | 0 | 140 | 23 | 50 |
| 7 | 2c | 25% NaOH | PhMe | 25 | 8 | 76 | 18 |
| 8 | 2c | 25% NaOH | PhMe | 0 | 96 | 81 | 20 |
| 9 | 9 | 25% NaOH | PhMe | 0 | 160 | 33 | 2 |
| 10 | 10a | 25% NaOH | PhMe | 25 | 1 | 96 | 44 |
| 11 | 10a | 25% NaOH | PhMe | 0 | 10 | 59 | 70 |
| 12 | 10a | 50% KOH | PhMe:CHCl3 | -20 | 18 | 71 | 69 |
| 13 | 10b | 25% NaOH | PhMe | 0 | 120 | 7 | 2 |
| 14 | 11 | 25% NaOH | PhMe | 25 | 4 | 76 | 62 |
| 15 | 11 | 25% NaOH | PhMe | 0 | 10 | 71 | 56 |
| 16 | 12 | 25% NaOH | PhMe | 25 | 12 | 78 | 56 |
| 17 | 12 | 25% NaOH | PhMe | 0 | 120 | 63 | 54 |
| 18 | 13a | 25% NaOH | PhMe | 25 | 10 | 75 | 63 |
| 19 | 13a | 25% NaOH | PhMe | 0 | 20 | 91 | 64 |
| 20 | 13a | 50% KOH | PhMe:CHCl3 | -20 | 15 | 90 | 68 |
| 21 | 13b | 25% NaOH | PhMe | 0 | 60 | 28 | 8 |
| 22 | 16 | 25% NaOH | PhMe | 25 | 2 | 92 | 59 |
| 23 | 16 | 25% NaOH | PhMe | 0 | 32 | 94 | 70 |
| 24 | 16 | 50% KOH | PhMe:CHCl3 | -20 | 3 | 86 | 74 |
| 25 | 18 | 25% NaOH | PhMe | 25 | 1 | 96 | 44 |
| 26 | 18 | 25% NaOH | PhMe | 0 | 170 | 49 | 70 |
| 27 | 18 | 50% KOH | PhMe:CHCl3 | -20 | 13 | 74 | 71 |
Conclusions
Acknowledgments
Experimental
General
General procedure for the preparation of the polymeric ammonium salts 2 and 9-13.
Polymeric ammonium salt 2a
Polymeric ammonium salt 2b
Polymeric ammonium salt 2c
Polymeric ammonium salt 9
Polymeric ammonium salt 10a
Polymeric ammonium salt 10b
Polymeric ammonium salt 11
Polymeric ammonium salt 12
Polymeric ammonium salt 13a
Polymeric ammonium salt 13b
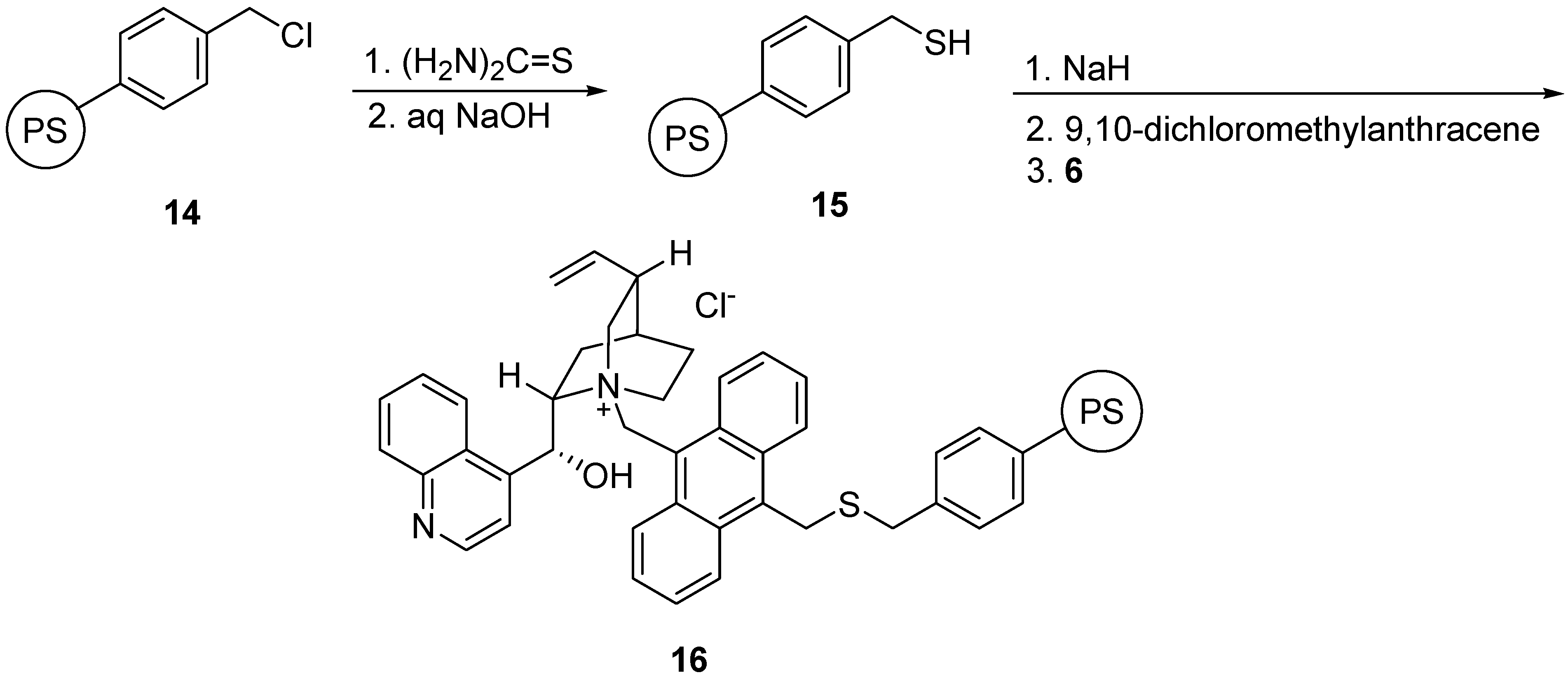
Preparation of polymeric ammonium salt 16
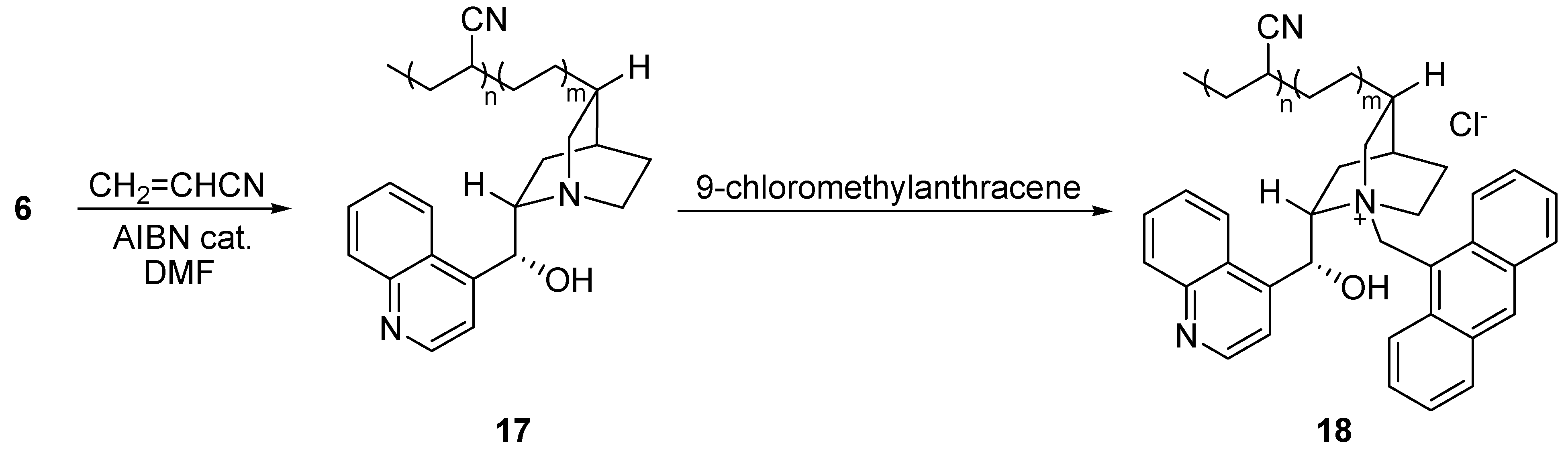
Preparation of co-polymeric ammonium salt 18 [27]
General procedure for the benzylation of 19 using the polymeric ammonium salts as PTC catalysts: Preparation of isopropyl 2-diphenylmethylenamino-3-phenylpropanoate (20).
References and Notes
- For recent reviews: Seebach, D.; Sting, A.R.; Hoffmann, M. Self-regeneration of stereocenters (SRS) - applications, limitations, and abandonment of a synthetic principle. Angew. Chem. Int. Ed. Engl. 1996, 35, 2708–2748. [Google Scholar] Wirth, T. New strategies to α-alkylated α-amino acids. Angew. Chem. Int. Ed. Engl. 1997, 36, 225–227. [Google Scholar] Cativiela, C.; Díaz-de-Villegas, M.D. Stereoselective synthesis of quaternary α-amino acids. Part 1: acyclic compounds. Tetrahedron:Asymmetry 1998, 9, 3517–3599. [Google Scholar] Cativiela, C.; Díaz-de-Villegas, M.D. Stereoselective synthesis of quaternary α-amino acids. Part 2: cyclic compounds. Tetrahedron: Asymmetry 2000, 11, 645–732. [Google Scholar] Abellán, T.; Chinchilla, R.; Galindo, N.; Guillena, G.; Nájera, C.; Sansano, J.M. Glycine and alanine imines as templates for asymmetric synthesis of α-amino acids. Eur. J. Org. Chem. 2000, 2689–2697. [Google Scholar] Nájera, C. From α-amino acids to peptides: all you need for the journey. Synlett 2002, 1388–1403. [Google Scholar]
- Phase-Transfer Catalysis; Halpern, M.E. American Chemical Society: Washington D.C., 1997. [Google Scholar]
- Kacprzak, K.; Gawronski, J. Cinchona alkaloids and their derivatives: versatile catalysts and ligands in asymmetric synthesis. Synthesis 2001, 961–998. [Google Scholar]
- O’Donnell, M.J.; Bennett, W.D.; Wu, S. The stereoselective synthesis of α-amino acids by phase-transfer catalysis. J. Am. Chem. Soc. 1989, 111, 2353–2355. [Google Scholar] Lipkowitz, K.B.; Cavanaugh, M.W.; Baker, B.; O’Donnell, M.J. Theoretical studies in molecular recognition: asymmetric induction of benzophenone imine ester enolates by the benzylcinchoninium ion. J. Org. Chem. 1991, 56, 5181–5192. [Google Scholar] O’Donnell, M.J.; Wu, S. A catalytic enantioselective synthesis of α-methyl amino acid derivatives by phase-transfer catalysis. Tetrahedron: Asymmetry 1992, 3, 591–594. [Google Scholar] O’Donnell, M.J.; Wu, S.; Huffman, J.C. A new active catalyst species for enantioselective alkylation by phase-transfer catalysis. Tetrahedron 1994, 50, 4507–4518. [Google Scholar] O’Donnell, M.J. The preparation of optically active α-amino acids from the benzophenone imines of glycine derivatives. Aldrichimica Acta 2001, 34, 3–15. [Google Scholar]
- Corey, E.J.; Xu, F.; Noe, M.C. A rational approach to catalytic enantioselective enolate alkylation using a structurally rigidified and defined chiral quaternary ammonium salt under phase transfer conditions. J. Am. Chem. Soc. 1997, 119, 12414–12415. [Google Scholar] Corey, E.J.; Noe, M.C.; Xu, F. Highly enantioselective synthesis of cyclic and functionalized α-amino acids by means of a chiral phase transfer catalyst. Tetrahedron Lett. 1998, 39, 5347–5350. [Google Scholar] Horikawa, M.; Busch-Petersen, J.; Corey, E.J. Enantioselective synthesis of α-hydroxy-α-amino acid esters by aldol coupling using a chiral quaternary ammonium salt as catalyst. Tetrahedron Lett. 1999, 40, 3843–3846. [Google Scholar]
- Lygo, B.; Wainwright, P.G. A new class of asymmetric phase-transfer catalysts derived from Cinchona alkaloids - application in the enantioselective synthesis of α-amino acids. Tetrahedron Lett. 1997, 38, 8595–8598. [Google Scholar] Lygo, B.; Crosby, J.; Peterson, J.A. Enantioselective synthesis of bis-α-amino acid esters via asymmetric phase-transfer catalysis. Tetrahedron Lett. 1999, 40, 1385–1388. [Google Scholar] Lygo, B. Enantioselective synthesis of dityrosine and isodityrosine via asymmetric phase-transfer catalysis. Tetrahedron Lett. 1999, 40, 1389–1392. [Google Scholar] Lygo, B.; Crosby, J.; Peterson, J.A. Enantioselective alkylation of alanine-derived imines using quaternary ammonium catalysts. Tetrahedron Lett. 1999, 40, 8671–8674. [Google Scholar] Lygo, B.; Crosby, J.; Lowdon, T.R.; Peterson, J.A.; Wainwright, P.G. Studies on the enantioselective synthesis of α-amino acids via asymmetric phase-transfer catalysis. Tetrahedron 2001, 57, 2403–2409. [Google Scholar] Lygo, B.; Andrews, B. I.; Crosby, J.; Peterson, J.A. Asymmetric alkylation of glycine imines using in situ generated phase-transfer catalysts. Tetrahedron Lett. 2002, 43, 8015–8018. [Google Scholar]
- O’Donnell, M.J.; Delgado, F.; Pottorf, R.S. Enantioselective solid-phase synthesis of α-amino acid derivatives. Tetrahedron 1999, 55, 6347–6362. [Google Scholar]
- Jew, S.; Jeong, B.; Yoo, M.; Huh, H.; Park, H. Synthesis and application of dimeric Cinchona alkaloid phase-transfer catalysts: α,α'-bis[O(9)-allylcinchonidinium]-o, m, or p-xylene dibromide. Chem. Commun. 2001, 1244–1245. [Google Scholar] Park, H.; Jeong, B.; Yoo, M.; Lee, J.; Park, M.; Lee, Y.; Kim, M.; Jew, J. Highly enantioselective and practical Cinchona-derived phase-transfer catalysts for the synthesis of α-amino acids. Angew. Chem. Int. Ed. 2002, 41, 3036–3038. [Google Scholar] Chinchilla, R.; Mazón, P.; Nájera, C. New dimeric anthracenyl-derived Cinchona quaternary ammonium salts as phase-transfer catalysts for the asymmetric synthesis of α-amino acids. Tetrahedron:Asymmetry 2002, 13, 927–931. [Google Scholar]
- Park, H.; Jeong, B.; Yoo, M.; Park, M.; Huh, H.; Jew, S. Trimeric Cinchona alkaloid phase-transfer catalyst: α,α',α''-tris[O(9)-allylcinchonidinium]mesitylene tribromide. Tetrahedron Lett. 2001, 42, 4645–4648. [Google Scholar]
- Mazón, P.; Chinchilla, R.; Nájera, C.; Guillena, G.; Kreiter, R.; Klein Gebbink, R.J.M.; van Koten, G. Unexpected metal base-dependent inversion of the enantioselectivity in the asymmetric synthesis of α-amino acids using phase-transfer catalysts derived from cinchonidine. Tetrahedron:Asymmetry 2002, 13, 2181–2185. [Google Scholar] Guillena, G.; Kreiter, R.; van de Coevering, R.; Klein Gebbink, R.J.M.; van Koten, G.; Mazón, P.; Chinchilla, R.; Nájera, C. Chiroptical properties and applications in PTC of new dendritic cinchonidine-derived ammonium salts. Tetrahedron:Asymmetry 2003, 14, 3705–3712. [Google Scholar]
- Ooi, T.; Kameda, M.; Maruoka, K. Molecular design of a C2-symmetric chiral phase-transfer catalyst for practical asymmetric synthesis of α-amino acids. J. Am. Chem. Soc. 1999, 121, 6519–6520. [Google Scholar] Ooi, T.; Tayama, E.; Doda, K.; Takeuchi, M.; Maruoka, K. Dramatic rate enhancement by ultrasonic irradiation in liquid-liquid phase-transfer catalytic reactions. Synlett 2000, 1500–1502. [Google Scholar] Ooi, T.; Takeuchi, M.; Kameda, M.; Maruoka, K. Practical catalytic enantioselective synthesis of α,α-dialkyl-α-amino acids by chiral phase-transfer catalysis. J. Am. Chem. Soc. 2000, 122, 5228–5229. [Google Scholar] Ooi, T.; Uematsu, Y.; Maruoka, K. Evaluation of the efficiency of the chiral quaternary ammonium salt β-Np-NAS-Br in the organic-aqueous phase-transfer alkylation of a protected glycine derivative. Adv. Synth. Catal. 2002, 344, 288–291. [Google Scholar] Ooi, T.; Uematsu, Y.; Maruoka, K. Highly enantioselective alkylation of glycine methyl and ethyl ester derivatives under phase-transfer conditions: its synthetic advantage. Tetrahedron Lett. 2004, 45, 1675–1678. [Google Scholar]
- Manabe, K. Asymmetric phase-transfer alkylation catalyzed by a chiral quaternary phosphonium salt with a multiple hydrogen-bonding site. Tetrahedron Lett. 1998, 39, 5807–5810. [Google Scholar] Manabe, K. Synthesis of novel chiral quaternary phosphonium salts with multiple hydrogen-bonding sites and their application to asymmetric phase-transfer alkylation. Tetrahedron 1998, 54, 14465–14476. [Google Scholar]
- Belokon’, Y.N.; Kotchetkov, K.A.; Churkina, T.D.; Ikonnikov, N.S.; Chesnokov, A.A.; Larionov, A.V.; Parmár, V.S.; Kumar, R.; Kagan, H.B. Asymmetric PTC C-alkylation mediated by TADDOL-novel route to enantiomerically enriched α-alkyl-α-amino acids. Tetrahedron: Asymmetry 1998, 9, 851–857. [Google Scholar] Belokon’, Y.N.; Kotchetkov, K.A.; Churkina, T.D.; Ikonnikov, N.S.; Chesnokov, A.A.; Larionov, A.V.; Singh, I.; Parmár, V.S.; Vyskocil, S.; Kagan, H.B. Asymmetric PTC C-alkylation catalyzed by chiral derivatives of tartaric acid and aminophenols. Synthesis of (R)- and (S)-α-methyl amino acids. J. Org. Chem. 2000, 65, 7041–7048. [Google Scholar] Shibuguchi, T.; Fukuta, Y.; Akachi, Y.; Sekine, A.; Oshima, T.; Shibasaki, M. Development of new asymmetric two-center catalysts in phase-transfer reactions. Tetrahedron Lett. 2002, 43, 9539–9543. [Google Scholar] Arai, S.; Tsuji, R.; Nishida, A. Phase-transfer-catalyzed asymmetric Michael reaction using newly-prepared chiral quaternary ammonium salts derived from l-tartrate. Tetrahedron Lett. 2002, 43, 9535–9537. [Google Scholar] Kita, T.; Georgieva, A.; Hashimoto, Y.; Nakata, T.; Nagasawa, K. C2-symmetric chiral pentacyclic guanidine: A phase-transfer catalyst for the asymmetric alkylation of tert-butyl glycinate Schiff base. Angew. Chem. Int. Ed. 2002, 41, 2832–2834. [Google Scholar]
- Belokon’, Y.N.; Kotchetkov, K.A.; Churkina, T.D.; Ikonnikov, N.S.; Vyskocil, S.; Kagan, H. B. Enantiomerically enriched (R)- and (S)-α -methylphenylalanine via asymmetric PTC C-alkylation catalyzed by NOBIN. Tetrahedron:Asymmetry 1999, 10, 1723–1728. [Google Scholar] Belokon’, Y.N.; Kotchetkov, K.A.; Churkina, T.D.; Ikonnikov, N.S.; Larionov, O.V.; Harutyunyan, S.R.; Vyskocil, S.; North, M.; Kagan, H.B. Highly efficient catalytic synthesis of α-amino acids under phase-transfer conditions with a novel catalyst/substrate pair. Angew Chem. Int. Ed. 2001, 40, 1948–1951. [Google Scholar] Casas, J.; Nájera, C.; Sansano, J.M.; González, J.; Saá, J.M.; Vega, M. Enantioselective synthesis of (S)-α-methylphenylalanine using (S)-BINOLAMs as new phase-transfer catalysts. Tetrahedron:Asymmetry 2001, 12, 699–702. [Google Scholar]
- Belokon’, Y.N.; North, M.; Kublitski, V.S.; Ikonnikov, N.S.; Krasik, P.E.; Maleev, V.L. Chiral salen-metal complexes as novel catalysts for asymmetric phase transfer alkylations. Tetrahedron Lett. 1999, 40, 6105–6108. [Google Scholar] Belokon’, Y.N.; Davies, R.G.; North, M. A practical asymmetric synthesis of α-methyl α-amino acids using a chiral Cu-salen complex as a phase transfer catalyst. Tetrahedron Lett. 2000, 41, 7245–7248. [Google Scholar]
- For recent reviews on polymer-supported chemistry see: Lorsbach, B. A.; Kurth, M. J. Carbon-carbon bond forming solid-phase reactions. Chem. Rev. 1999, 99, 1549–1581. [Google Scholar] James, I.W. Linkers for solid phase organic synthesis. Tetrahedron 1999, 55, 4855–4946. [Google Scholar] Shuttleworth, S.J.; Allin, S.M.; Wilson, R.D.; Nasturica, D. Functionalized polymers in organic chemistry; Part 2. Synthesis 2000, 1035–1074. [Google Scholar] Ley, S.V.; Baxendale, I.R.; Bream, R.N.; Jackson, P.S.; Leach, A.G.; Longbottom, D.A.; Nesi, M.; Scott, J.S.; Storer, R.I.; Taylor, S.J. Multi-step organic synthesis using solid-supported reagents and scavengers: a new paradigm in chemical library generation. J. Chem. Soc., Perkin Trans. 1 2000, 3815–4195. [Google Scholar] Orain, D.; Ellard, J.; Bradley, M. Protecting Groups in Solid-Phase Organic Synthesis. J. Comb. Chem. 2002, 4, 1–16. [Google Scholar] Benaglia, M.; Puglisi, A.; Cozzi, F. Polymer-Supported Organic Catalysts. Chem. Rev. 2003, 103, 3401–3429. [Google Scholar]
- Hermann, K.; Wynberg, H. Polymer-bound Cinchona alkaloids as catalysts in the Michael reaction. Helv. Chim. Acta 1977, 60, 2208–2212. [Google Scholar] Kobayashi, N.; Iwai, K. Functional polymers. 1. Poly(cinchona alkaloid-co-acrylonitrile)s. New polymer catalysts for asymmetric synthesis. J. Am. Chem. Soc. 1978, 100, 7071–7072. [Google Scholar] Hodge, P.; Khoshdel, E.; Waterhouse, J. Michael reactions catalyzed by polymer-supported quaternary ammonium salts derived from cinchona and ephedra alkaloids. J. Chem. Soc., Perkin Trans. 1 1983, 2205–2209. [Google Scholar] Hodge, P.; Khoshdel, E.; Waterhouse, J.; Fréchet, J.M.J. Michael additions catalyzed by Cinchona alkaloids bound via their vinyl groups to preformed crosslinked polymers. J. Chem. Soc., Perkin Trans. 1 1985, 2327–2331. [Google Scholar] Inagaki, M.; Hiratane, J.; Yamamoto, Y.; Oda, J. Asymmetric induction in the base-catalyzed reactions using polymer-supported quinines with spacer groups. Bull. Chem. Soc. Jpn. 1987, 60, 4121–4126. [Google Scholar] Sera, A.; Takagi, K.; Katayama, H.; Yamada, H.; Matsumoto, K. High-pressure asymmetric Michael additions of thiols, nitromethane, and methyl oxoindancarboxylate to enones. J. Org. Chem. 1988, 53, 1157–1161. [Google Scholar] Alvarez, R.; Hourdin, M.-A.; Cavé, C.; d’Angelo, J.; Chaminade, P. New polymer-supported catalysts derived from cinchona alkaloids: their use in the asymmetric Michael reaction. Tetrahedron Lett. 1999, 40, 7091–7094. [Google Scholar]
- Han, H.; Janda, K.D. Soluble Polymer-Bound Ligand-Accelerated Catalysis: Asymmetric Dihydroxylation. J. Am. Chem. Soc. 1996, 118, 7632–7633. [Google Scholar] Han, H.; Janda, K.D. A soluble polymer-bound approach to the Sharpless catalytic asymmetric dihydroxylation (AD) reaction: preparation and application of a [(DHQD)2PHAL-PEG-OMe) ligand. Tetrahedron Lett. 1997, 38, 1527–1530. [Google Scholar] Nandanan, E.; Sudalai, A.; Ravindranathan, T. New polymer supported Cinchona alkaloids for heterogeneous catalytic asymmetric dihydroxylation of olefins. Tetrahedron Lett. 1997, 38, 2577–2580. [Google Scholar] Canali, L.; Song, E.S.; Sherrington, D.C. Polymer-supported bis-Cinchona alkaloid ligands for asymmetric dihydroxylation of alkenes-a cautionary tale. Tetrahedron: Asymmetry 1998, 9, 1029–1034. [Google Scholar]
- Song, C.E.; Oh, C.R.; Lee, S.W.; Lee, S.; Canali, L.; Sherrington, D.C. Heterogeneous asymmetric aminohydroxylation of alkenes using a silica gel-supported bis-Cinchona alkaloid. Chem. Commun. 1998, 2435–2436. [Google Scholar]
- Wang, Y.; Zhengpu, Z.; Zhen, W.; Jiben, M.; Hodge, P. The asymmetric synthesis of amino acids under polymer-supported phase transfer catalytic condition. Chin. J. Polym. Sci. 1998, 16, 356–361. [Google Scholar] Zhengpu, Z.; Yongmei, W.; Zhen, W.; Hodge, P. Asymmetric synthesis of a-amino acids using polymer-supported chiral phase transfer catalysts. React. Funct. Polym. 1999, 41, 37–43. [Google Scholar] Chinchilla, R.; Mazón, P.; Nájera, C. Asymmetric synthesis of a-amino acids using polymer-supported Cinchona alkaloid-derived ammonium salts as chiral phase-transfer catalysts. Tetrahedron: Asymmetry 2000, 11, 3277–3281. [Google Scholar]
- Thierry, B.; Plaquevent, J.-C.; Cahard, D. New polymer-supported chiral phase-transfer catalysts in the asymmetric synthesis of α-amino acids: the role of a spacer. Tetrahedron: Asymmetry 2001, 12, 983–986. [Google Scholar]
- Thierry, B.; Perrard, T.; Audouard, C.; Plaquevent, J.-C.; Cahard, D. Solution- and solid-phase approaches in asymmetric phase-transfer catalysis by Cinchona alkaloid derivatives. Synthesis 2001, 1742–1746. [Google Scholar]
- Thierry, B.; Plaquevent, J.-C.; Cahard, D. Poly(ethylene glycol) supported Cinchona alkaloids as phase transfer catalysts: application to the enantioselective synthesis of α-amino acids. Tetrahedron: Asymmetry 2003, 14, 1671–1677. [Google Scholar] Danelli, T.; Annunziata, R.; Benaglia, M.; Cinquini, M.; Cozzi, F.; Tocco, G. Immobilization of catalysts derived from Cinchona alkaloids on modified poly(ethylene glycol). Tetrahedron: Asymmetry 2003, 14, 461–467. [Google Scholar]
- Chiellini, E.; Solaro, R. Stereo-ordered macromolecular matrixes bearing ammonium groups as catalysts in alkylation and carbenation reactions. J. Chem. Soc., Chem. Commun. 1977, 231–232. [Google Scholar]
- Fréchet, J.M.J.; de Smet, M.D.; Marc, D.; Farrall, M.J. Functionalization of crosslinked polystyrene resins. 2. Preparation of nucleophilic resins containing hydroxyl or thiol functionalities. Polymer 1979, 20, 675–680. [Google Scholar]
- Prepared by bubbling HCl (g) through a solution of anthracene and paraformaldehyde in dioxane: Miller, M.W.; Amidon, R.W.; Tawney, P.O. Some meso substituted anthracenes. I. 9,10-Bis-(chloromethyl)anthracene as a synthetic intermediate. J.Am. Chem. Soc. 1955, 77, 2845–2848. [Google Scholar]
- Kobayashi, N.; Iwai, K. Functional polymers. 1. Poly(cinchona alkaloid-co-acrylonitrile)s. New polymer catalysts for asymmetric synthesis. J. Am. Chem. Soc. 1978, 100, 7071–7072. [Google Scholar]
- Prepared in 80% overall yield by reaction of glycine with thionyl chloride in the presence of isopropanol (Patel, R.P.; Price, S. Synthesis of benzyl esters of α-amino acids. J. Org. Chem. 1965, 30, 3575–3576. [Google Scholar] ), followed by treatment of the crude product with benzophenone imine (O’Donnell, M.J.; Polt, R.L. A mild and efficient route to Schiff base derivatives of amino acids. J. Org. Chem. 1982, 47, 2663–2666. [Google Scholar] ).
- Oppolzer, W.; Moretti, R.; Zhou, C. Asymmetric alkylations of a sultam-derived glycine equivalent: practical preparation of enantiomerically pure α-amino acids. Helv. Chim. Acta 1994, 77, 2363–2380. [Google Scholar]
- Sample Availability: Samples of the catalysts are available from the authors.
© 2004 by MDPI (http:www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Chinchilla, R.; Mazón, P.; Nájera, C. Polymer-Supported Cinchona Alkaloid-Derived Ammonium Salts as Recoverable Phase-Transfer Catalysts for the Asymmetric Synthesis of α-Amino Acids. Molecules 2004, 9, 349-364. https://doi.org/10.3390/90500349
Chinchilla R, Mazón P, Nájera C. Polymer-Supported Cinchona Alkaloid-Derived Ammonium Salts as Recoverable Phase-Transfer Catalysts for the Asymmetric Synthesis of α-Amino Acids. Molecules. 2004; 9(5):349-364. https://doi.org/10.3390/90500349
Chicago/Turabian StyleChinchilla, Rafael, Patricia Mazón, and Carmen Nájera. 2004. "Polymer-Supported Cinchona Alkaloid-Derived Ammonium Salts as Recoverable Phase-Transfer Catalysts for the Asymmetric Synthesis of α-Amino Acids" Molecules 9, no. 5: 349-364. https://doi.org/10.3390/90500349
APA StyleChinchilla, R., Mazón, P., & Nájera, C. (2004). Polymer-Supported Cinchona Alkaloid-Derived Ammonium Salts as Recoverable Phase-Transfer Catalysts for the Asymmetric Synthesis of α-Amino Acids. Molecules, 9(5), 349-364. https://doi.org/10.3390/90500349




