Lipid-Lowering Effects of Ethyl 2-Phenacyl-3-aryl-1H-pyrrole- 4-carboxylates in Rodents
Abstract
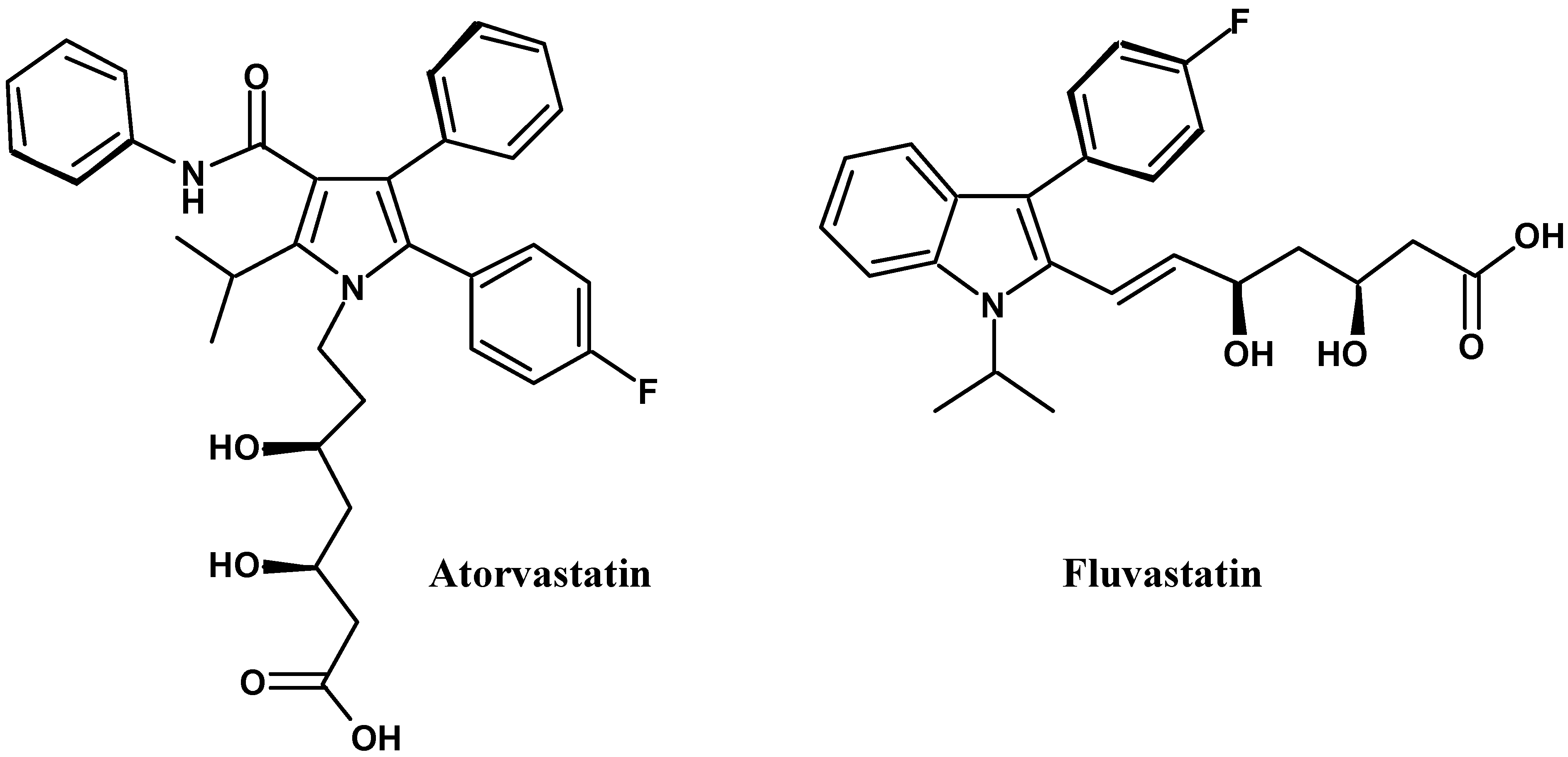
:Introduction
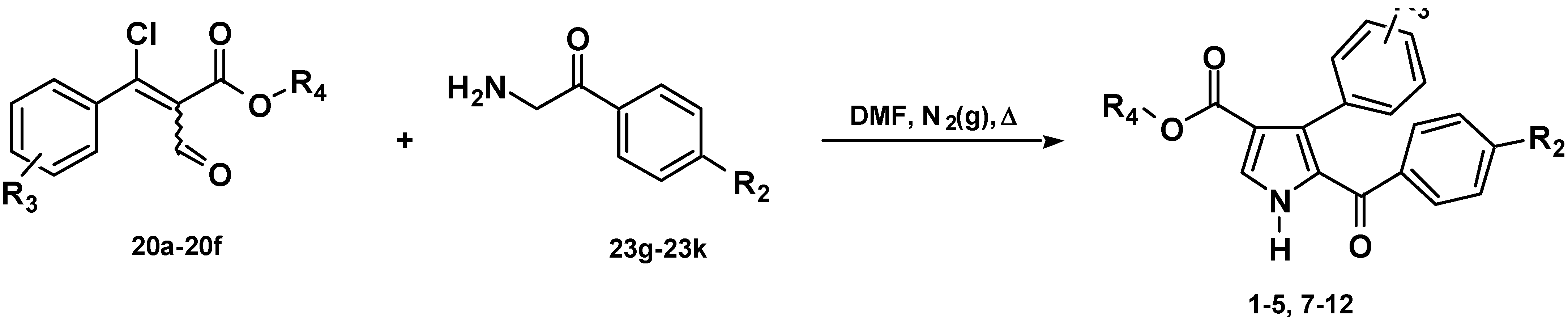
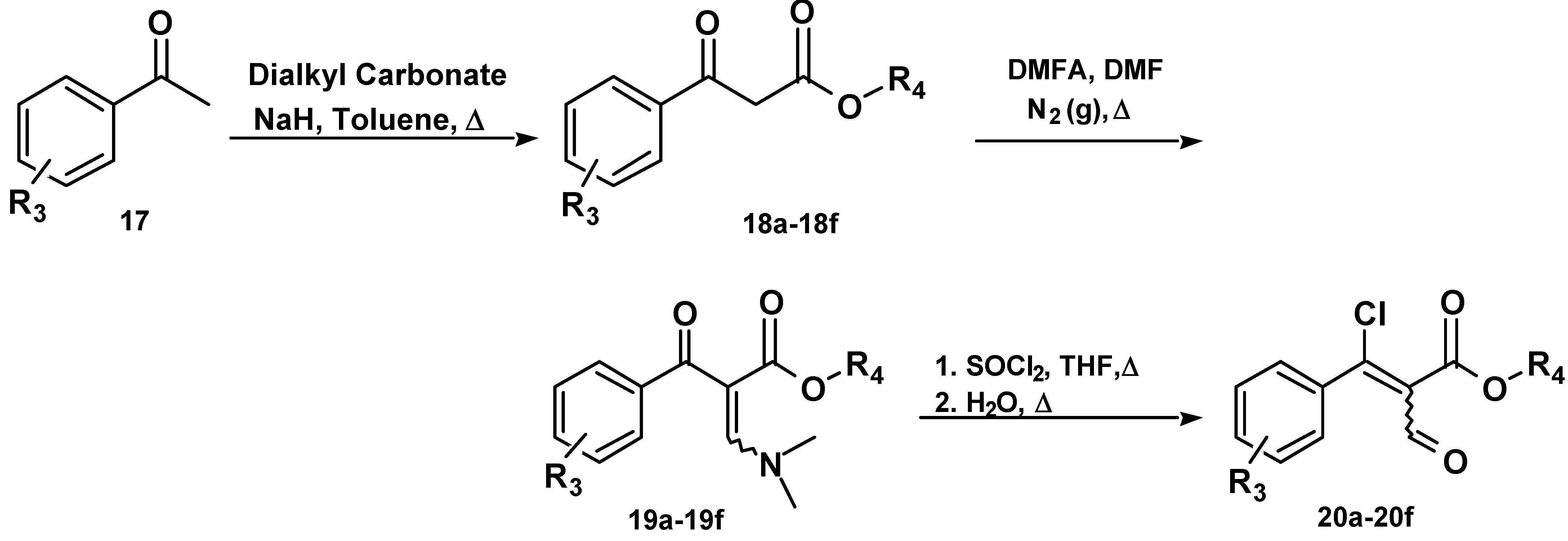
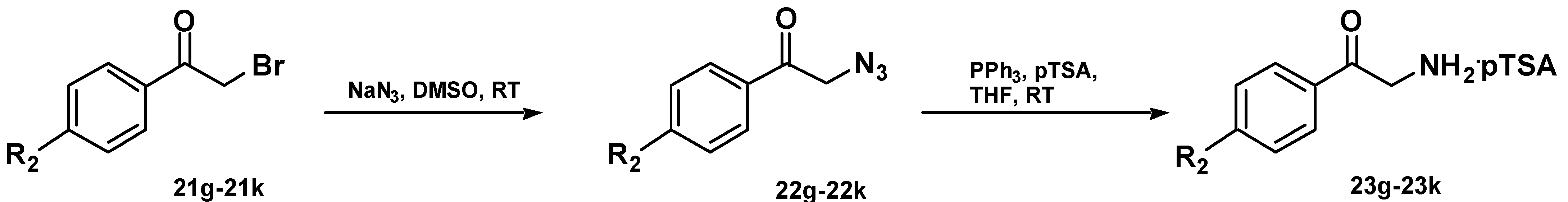
Chemistry
Results
| Compound | Ra- | Rb- | Rc- | Rd- |
|---|---|---|---|---|
| 1 | H- | 4'-Cl-C6H4- | 4'-Cl-C6H4- | H3CH2COOC- |
| 2 | H- | C6H5- | 4'-Br-C6H4- | H3CH2COOC- |
| 3 | H- | C6H5- | 3'-Cl-C6H4- | H3CH2COOC- |
| 4 | H- | 4'-F-C6H4- | 4'-CH3-C6H4- | H3CH2COOC- |
| 5 | H- | 4'-Cl-C6H4- | 4'-CH3-C6H4- | H3CH2COOC- |
| 6 | H3C- | CH3CH2O- | H- | 4'-Cl-C6H4- |
| 7 | H- | 4'-F-C6H4- | 4'-F-C6H4- | H3CH2COOC- |
| 8 | H- | 4'-CH3O-C6H4- | 4'-Cl-C6H4- | H3CH2COOC- |
| 9 | H- | 4'-Cl-C6H4- | 4'-F-C6H4- | H3CH2COOC- |
| 10 | H- | 4'-CH3-C6H4- | 4'-CH3O-C6H4- | H3COOC- |
| 11 | H- | 4'-CH3O-C6H4- | 4'-CH3O-C6H4- | H3COOC- |
| 12 | H- | 4'-Cl-C6H4- | 4'-CH3O-C6H4- | H3COOC- |
| 13 | H- | 4'-CH3O-C6H4- | 4'-CH3O-C6H4- | 4'-CH3O-C6H4- |
| 14 | H- | CH3O- | 3',4'-(CH3O)2-C6H4- | 3',4'-(CH3O)2-C6H4- |
| 15 | H3C- | CH3CH2O- | 3',4'-(CH3O)2-C6H4- | 3',4'-(CH3O)2-C6H4- |
| N=6 Compound | (g/rat/day) Food Consumption | Percent of Control (X±SD) | |||
| Day 7 | Day 14 | ||||
| Cholesterol | Triglycerides | Cholesterol | Triglycerides | ||
| 1 | 19.13 | 98±5 | 93±18 | 68±16* | 147±7* |
| 2 | 20.60 | 120±24 | 85±13 | 104±7 | 159±8* |
| Atorvastatina | 19.03 | 110±14 | 92±28 | 69±6* | 119±5* |
| Gemfibrozilb | 17.47 | 122±5 | 56±62* | 116±17 | 138±2* |
| Controlc | 20.06 | 100±14d | 100±7e | 100±8f | 100±6g |
| a Dosed at 8 mg/kg/day | e 94 mg/dL serum triglycerides | ||||
| b Dosed at 90 mg/kg/day | f 46 mg/dL total serum cholesterol | ||||
| c 1% CMC | g 56 mg/dL serum triglycerides | ||||
| N=6 | Percent of Control (X±SD) | |
| Compound | Cholesterol | Triglycerides |
| Liver | ||
| 1 | 187±19* | 106±20 |
| 2 | 197±19* | 100±40 |
| Atorvastatina | 169±19* | 166±35 |
| Gemfibrozilb | 107±18 | 96±16 |
| Controlc | 100±24d | 100±35e |
| Small intestine | ||
| 1 | 48±13 | 127±24 |
| 2 | 33±17 | 115±20 |
| Atorvastatina | 49±15 | 55±18* |
| Gemfibrozilb | 75±16 | 81±28 |
| Controlc | 100±35f | 100±19g |
| Aorta | ||
| 1 | 61±12* | 15±41* |
| 2 | 126±18 | 30±28* |
| Atorvastatina | 105±17 | 56±23* |
| Gemfibrozilb | 106±13 | 29±22* |
| Controlc | 100±15h | 100±14i |
| Feces | ||
| 1 | 123±18 | 84±20 |
| 2 | 94±10 | 92±16 |
| Atorvastatina | 178±54 | 85±16 |
| Gemfibrozilb | 106±13 | 98±23 |
| Controlc | 100±22j | 100±24k |
| a Dosed at 8 mg/kg/day | g 12.78 mg/g (wt) | |
| b Dosed at 90 mg/kg/day | h 2.97 mg/dL (wt) | |
| c 1% CMC | I 13.85 mg/dL (wt) | |
| d 3.17 mg/g (wet tissue) | j 7.92 mg/dL (wt) | |
| e 3.31 mg/dL (wt) | k 9.44 mg/dL (wt) | |
| Percent of Control (X±SD) | ||||
| N=6 | Day 7 | Day14 | ||
| Compound | Cholesterol | Triglycerides | Cholesterol | Triglycerides |
| 3 | 68±11* | 28±29* | 72±8* | 59±37 |
| 4 | 69±9* | 54±5* | 66±11* | 113±43 |
| 5 | 73±8* | 99±8 | 87±8 | 184±28 |
| 6 | 97±7 | 62±7* | 82±9* | 109±20 |
| 7 | 67±15* | 56±24* | 76±14 | 61±30* |
| 8 | 79±14* | 91±31 | 91±13 | 53±23* |
| 9 | 95±8 | 58±14* | 91±18 | 52±23* |
| 10 | 93±10 | 70±19 | 98±17 | 60±21* |
| 11 | 89±9 | 76±26 | 90±17 | 77±30 |
| 12 | 102±12* | 67±11* | 103±12 | 67±31 |
| 13 a | 80±13* | 49±17* | 76±8* | 65±28* |
| 14 a | 62±19* | 55±20* | 66±14* | 62±23* |
| 15 a | 75±9* | 83±14 | 89±3* | 76±24 |
| Atorvastatinb | 77±6* | 66±11* | 81±17 | 84±33 |
| Gemfibrozilc | 62±11* | 43±21* | 57±13* | 82±15 |
| Niacind | 97±18 | 70±8 | 93±24 | 102±21 |
| Controle | 100±14f | 100±7g | 100±8h | 100±6i |
| a Dosed at 4 mg/kg/day, IP | f 188 mg/dL total serum cholesterol | |||
| b Dosed at 8 mg/kg/day, IP | g 231 mg/dL serum triglycerides | |||
| c Dosed at 90 mg/kg/day, IP | h 164 mg/dL total serum cholesterol | |||
| d Dosed at 12.5 mg/kg/day, IP | i 179 mg/dL serum triglycerides | |||
Discussion
| N=6 | Percent of Control (X±SD) | |
| Compound | LDL-Cholesterol | HDL-Cholesterol |
| 3 | - | 102±20 |
| 4 | - | 112±24 |
| 5 | - | 109±21 |
| 6 | - | 130±12 |
| 7 | 47±21* | 63±53 |
| 8 | 65±40* | 56±38 |
| 9 | 53±31 | 283±10* |
| 10 | 106±5 | 46±31* |
| 11 | 47±15* | 96±63 |
| 12 | 47±25* | 203±21 |
| 13a | 39±34* | 82±31 |
| 14a | 114±21 | 148±4 |
| 15a | 156±17 | 250±11 |
| Atorvastatinb | - | 178±34 |
| Gemfibrozilc | - | 192±8* |
| Niacind | - | 152±19 |
| Controle | 100±15f | 100±14g |
| a Dosed at 4 mg/kg/day, IP | e 1% CMC, PO | |
| b Dosed at 8 mg/kg/day, IP | f 86 mg/dL LDL-Cholesterol | |
| c Dosed at 90 mg/kg/day, IP | g 63 mg/dL HDL-Cholesterol | |
Conclusions
Experimental
General:
Chemistry
Hypolipidemic Studies.
Statistical Analysis.
Acknowledgements
References
- Evans, M.A.; Smith, D.C.; Holub, J.M.; Argenti, A.; Hoff, M.; Dalglish, G.A.; Wilson, D.L.; Taylor, B.M.; Berkowitz, J.D.; Burnham, B.S.; Krumpe, K.; Gupton, J.T.; Scarlett, T.C.; Durham, R.; Hall, I.H. Synthesis and Cytotoxicity of Substituted Ethyl 2-phenacyl-3-phenylpyrrole-4-carboxylates. Arch. Pharm. Pharm. Med. Chem. 2003, 336, 181–190. [Google Scholar]
- Burnham, B.S.; Gupton, J.T.; Krumpe, K.E.; Webb, T.; Shuford, J.; Bowers, B.; Warren, A.E.; Barnes, C.; Hall, I.H. Cytotoxicity of Substituted Alkyl-3,4-bis(4-methoxyphenyl)pyrrole-2-carboxylates in L1210 Lymphoid Leukemia Cells. Arch. Pharm. Pharm. Med. Chem. 1998, 331, 337–341. [Google Scholar]
- Gupton, J.T.; Burnham, B.S.; Byrd, B.D.; Krumpe, K.E.; Stokes, C.; Shuford, J.; Winkle, S.; Webb, T.; Warren, A.E.; Barnes, C.; Henry, J.; Hall, I.H. The Cytotoxicity and Mode of Action of 2,3,4-Trisubstituted Pyrroles and Related Derivatives in Human Tmolt4 Leukemia Cells. Pharmazie 1999, 54, 691–697. [Google Scholar]
- Gupton, J.T.; Burnham, B.S.; Krumpe, K.E.; Du, K.; Sikorski, J.A.; Warren, A.E.; Barnes, C.; Hall, I.H. Synthesis and Cytotoxicity of 2,4-Disubstituted and 2,3,4-Trisubstituted Brominated Pyrroles in Murine and Human Cultured Tumor Cells. Arch. Pharm. Pharm. Med. Chem. 2000, 333, 3–9. [Google Scholar]
- Procopiou, P.A.; Draper, C.D.; Hutson, J.L.; Inglis, G.G.; Ross, B.C.; Watson, N.S. Inhibitors of cholesterol biosynthesis. 2. 3,5-Dihydroxy-7-(N-pyrrolyl)-6-heptenoates, a novel series of HMG-CoA reductase inhibitors. J. Med. Chem. 1993, 36, 3658–3662. [Google Scholar]
- Roth, B.D.; Blankley, C.J.; Chucholowski, A.W.; Ferguson, E.; Hoefle, M.L.; Ortwine, D.F.; Newton, R.S.; Sekerke, C.S.; Sliskovic, D.R.; Stratton, C.D.; Wilson, M.W. Inhibitors of cholesterol biosynthesis. 3. Tetrahydro-4-hydroxy-6-[2-(1H-pyrrol-1-yl)ethyl]-2H-pyran-2-one inhibitors of HMG-CoA reductase. 2. Effects of introducing substituents at positions three and four of the pyrrole nucleus. J. Med. Chem. 1991, 34, 357–366. [Google Scholar]
- Jendralla, H.; Baader, E.; Bartmann, W.; Beck, G.; Bergmann, A.; Granzer, E.; von Kerekjarto, B.; Kesseler, K.; Krause, R.; Schubert, W.; Wess, G. Synthesis and biological activity of new HMG-CoA reductase inhibitors. 2. Derivatives of 7-(1H-pyrrol-3-yl)-substituted-3,5-dihydroxy-hept-6(E)-enoic (-heptanoic) acids. J. Med. Chem. 1990, 33, 61–70. [Google Scholar]
- Roth, BD; Ortwine, DF; Hoefle, ML; Stratton, CD; Sliskovic, DR; Wilson, MW; Newton, RS. Inhibitors of cholesterol biosynthesis. 1. trans-6-(2-pyrrol-1-ylethyl)-4-hydroxypyran-2-ones, a novel series of HMG-CoA reductase inhibitors. 1. Effects of structural modifications at the 2- and 5-positions of the pyrrole nucleus. J. Med. Chem. 1990, 33, 21–31. [Google Scholar]
- Bocan, T.M.; Ferguson, E.; McNally, W.; Uhlendorf , P.D.; Bak Mueller, S.; Dehart, P.; Sliskovic, D.R.; Roth, B.D.; Krause, B.R.; Newton, R.S. Hepatic and nonhepatic sterol synthesis and tissue distribution following administration of a liver selective HMG-CoA reductase inhibitor, CI-981: comparison with selected HMG-CoA reductase inhibitors. Biochim. Biophys Acta 1992, 1123, 133–144. [Google Scholar]
- Watanabe, M.; Koike, H.; Ishiba, T.; Okada, T.; Seo, S.; Hirai, K. Synthesis and biological activity of methanesulfonamide pyrimidine- and N-methanesulfonyl pyrrole-substituted 3,5-dihydroxy-6-heptenoates, a novel series of HMG-CoA reductase inhibitors. Bioorg. Med. Chem. 1997, 5, 437–444. [Google Scholar]
- Corsini, A.; Bellosta, S.; Baetta, R.; Fumagalli, R.; Paoletti, R.; Bernini, F. New insights into the pharmacodynamic and pharmacokinetic properties of statins. Pharmcol. Ther. 1999, 84, 413–428. [Google Scholar]
- McKenney, J.M. Dyslipiemias. In Applied Therapeutics, 7th ed.; Koda-Kimble, M.A., Young, L.Y., Eds.; Lippincott Williams and Wilkins: Philadelphia, PA, 2001; pp. 11-1 to 11-43. [Google Scholar]
- Takemoto, M.; Liao, J.K. Pleiotropic effects of 3-hydroxy-3-methylglutaryl coenzyme a reductase inhibitors. Arterioscler. Thromb. Vasc. Biol. 2001, 21, 1712–1719. [Google Scholar]
- Shovman, O.; Levy, Y.; Gilburd, B.; Shoenfeld, Y. Antiinflammatory and immunomodulatory properties of statins. Immunol. Res. 2002, 25, 271–285. [Google Scholar]
- Lefer, D.J. Statins as potent antiinflammatory drugs. Circulation 2002, 106, 2041–2042. [Google Scholar]
- Leung, B.P.; Sattar, N.; Crilly, A.; Prach, M.; McCarey, D.W.; Payne, H.; Madhok, R.; Campbell, C.; Gracie, J.A.; Liew, F.Y.; McInnes, I.B. A novel anti-inflammatory role for simvastatin in inflammatory arthritis. J. Immunol. 2003, 170, 1524–1530. [Google Scholar]
- Vega, G.L.; Weiner, M.F.; Lipton, A.M.; Von Bergmann, K.; Lutjohann, D.; Moore, C.; Svetlik, D. Reduction in Levels of 24S-Hydroxycholesterol by Statin Treatment in Patients with Alzheimer Disease. Arch. Neurol. 2003, 60, 510–515. [Google Scholar]
- Crisby, M.; Carlson, L.A.; Winblad, B. Statins in the Prevention and Treatment of Alzheimer’s Disease. Alzheimer Dis. Assoc. Disord. 2002, 16, 131–136. [Google Scholar]
- Austen, B.; Christodoulou, G.; Terry, J.E. Relationship Between Cholesterol Levels, Statins and Alzheimer’s Disease in the Human Population. J. Nutr. Health Aging 2002, 6, 377–382. [Google Scholar]
- Wolozin, B.; Kellman, W.; Rousseau, P.; Celesia, G.G.; Siegel, G. Decreased Prevalence of Alzheimer Disease Associated with 3-Hydroxy-3-methylglutaryl Coenzyme A Reductase Inhibitors. Arch Neurol. 2000, 57, 1439–1443. [Google Scholar]
- Kaneko, I.; Hazama-Shimda, y; Endo, A. Inhibitory effects on lipid metabolism in cultured cells of ML-236B, a potent inhibitor of 3-hydroxy-3-methylglutaryl-coenzyme-A reductase. Eur J. Biochem 1978, 87, 313–321. [Google Scholar]
- Burnham, B.S.; Sood, A.; Tomasz, J.; Powell, W.J.; Spielvogel, B.F.; Chen, S.Y.; Hall, I.H. The Hypolipidemic Activity of Boronated Nucleosides in Male Mice and Rats. Metal-Based Drugs 1996, 3, 173–183. [Google Scholar]
- Hall, I.H.; Burnham, B.S.; Elkins, A.; Sood, A.; Powell, W.; Tomasz, J.; Spielvogel, B.F. Boronated Pyrimidines and Purines as Cytotoxic, Hypolipidemic and Anti-inflammatory Agents. Metal-Based Drugs 1996, 3, 155–160. [Google Scholar]
- Burnham, B.S.; Chen, S.Y.; Sood, A.; Spielvogel, B.F.; Hall, I.H. Synthesis and Cytotoxicity of Amine-Borane Adducts of Cyclohexylamines and Toluidines. Pharmazie 1995, 50, 779–783. [Google Scholar]
- Burnham, B.S.; Chen, S.Y.; Sood, A.; Spielvogel, B.F.; Hall, I.H. Hypolipidemic Activity of Amine-Borane Adducts of Cyclohexylamine and Toluidine in Rodents. Metal-Based Drugs 1995, 2, 221–232. [Google Scholar]
- Hall, I.H.; Burnham, B.S; Rajendran, K.G.; Chen, S.Y.; Sood, A.; Spielvogel, B.F.; Shaw, B.R. Hypolipidemic Activity of Boronated Nucleosides and Nucleotides in Rodents. Biomed Pharmacother. 1993, 47, 79–87. [Google Scholar]
- Sood, A.; Spielvogel, B.F.; Shaw, B.R.; Carlton, L.; Burnham, B.S.; Hall, E.; Hall, I.H. The Synthesis and Antineoplastic Activity of 2'-Deoxynucleoside-cyanoboranes in Murine and Human Cultured Cells. Anti-Cancer Research 1992, 12, 335–344. [Google Scholar]
- Sood, C.; Sood, A.; Spielvogel, B.F.; Yousef, J.; Burnham, B.; Hall, I. Synthesis and Antineoplastic Activity of Some Cyano-, Carboxy-, Carbomethoxy-, and Carbamoylborane Adducts of Heterocyclic Amines. J Pharm. Sci. 1991, 80, 1133–1140. [Google Scholar]
- Hall, I.H.; Hall, E.S.; Wong, O.T. The antineoplastic activity of 2,3-dihydrophthalazine-1 ,4-dione and N-butyl-2,3-dihydrophthalazine-I,4-dione in human and murine tumor cells. Anti-Cancer Drugs 1992, 3, 55. [Google Scholar]
- Hall, I.H.; Lee, K.H.; Starnes, C.O.; Muraoka, 0.; Sumida, Y.; Waddell, T.G. Antihyperlipidemic activity of sesquiterpene lactones and related compounds. J. Pharm. Sci. 1980, 69, 694. [Google Scholar]
- Simlot, R.; Izydore, R.A.; Wong, O.T.; Hall, I.H. Synthesis and hypolipidemic activity of 4-substituted l-acyl-1 ,2,4-triazolidine-3,5-dione in rodents. J. Pharmacol Sci. 1993, 82, 408–415. [Google Scholar]
- Gupton, J.T.; Krumpe, K.E.; Burnham, B.S.; Webb, T.; Shuford, R.J.; Sikorski, J. The Application of Vinylogous Imminium Salt Derivatives to a Regiocontrolled and Efficient Relay Synthesis of Lukianol A and Related Marine Natural Products. Tetrahedron 1999, 55, 14515–14522. [Google Scholar]
- Gupton, J.T.; Krumpe, K.E.; Burnham, B.S.; Dwornik, K.A.; Petrich, S.A.; Du, X.K.; Bruce, M.A.; Vu, P.; Vargas, M.; Keertikar, K.M.; Hosein, K.N.; Jones, C.R.; Sikorski, J.A. The Application of Disubstituted Vinylogous Iminium Salts and Related Synthons to the Regiocontrolled Preparation of Unsymmetrical 2,3,4-Trisubstituted Pyrroles. Tetrahedron 1998, 45, 5075–5088. [Google Scholar]
- Gordon, A.J.; Ford, R.A. The Chemist’s Companion: A Handbook of Practical Data, Techniques, and References; John Wiley and Sons: New York, 1972; pp. 429–436. [Google Scholar]
- Gupton, J.T.; Krolikowski, D.A.; Yu, R.H.; Sikorski, J.A.; Riesinger, S.W. Application of 2-substituted vinamidinium salts to the synthesis of 2,4-disubstituted pyrroles. J. Org. Chem 1990, 55, 4735–4740. [Google Scholar]
- Gupton, J.T.; Clough, S.C.; Miller, R.B.; Lukens, J.R.; Henry, C.A.; Kanters, R.P.F.; James, A.; Sikorski, J.A. The application of vinylogous iminium salt derivatives to the synthesis of Ningalin B hexamethyl ether. Tetrahedron 2003, 59, 207–215. [Google Scholar]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar]
- Sample availability: Contact the authors.
© 2004 by MDPI (http:www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Holub, J.M.; O'Toole-Colin, K.; Getzel, A.; Argenti, A.; Evans, M.A.; Smith, D.C.; Dalglish, G.A.; Rifat, S.; Wilson, D.L.; Taylor, B.M.; et al. Lipid-Lowering Effects of Ethyl 2-Phenacyl-3-aryl-1H-pyrrole- 4-carboxylates in Rodents. Molecules 2004, 9, 134-157. https://doi.org/10.3390/90300134
Holub JM, O'Toole-Colin K, Getzel A, Argenti A, Evans MA, Smith DC, Dalglish GA, Rifat S, Wilson DL, Taylor BM, et al. Lipid-Lowering Effects of Ethyl 2-Phenacyl-3-aryl-1H-pyrrole- 4-carboxylates in Rodents. Molecules. 2004; 9(3):134-157. https://doi.org/10.3390/90300134
Chicago/Turabian StyleHolub, Justin M., Kathy O'Toole-Colin, Adam Getzel, Anthony Argenti, Michael A. Evans, Daniel C. Smith, Gerard A. Dalglish, Shahzad Rifat, Donna L. Wilson, Brett M. Taylor, and et al. 2004. "Lipid-Lowering Effects of Ethyl 2-Phenacyl-3-aryl-1H-pyrrole- 4-carboxylates in Rodents" Molecules 9, no. 3: 134-157. https://doi.org/10.3390/90300134
APA StyleHolub, J. M., O'Toole-Colin, K., Getzel, A., Argenti, A., Evans, M. A., Smith, D. C., Dalglish, G. A., Rifat, S., Wilson, D. L., Taylor, B. M., Miott, U., Glersaye, J., Lam, K. S., McCranor, B. J., Berkowitz, J. D., Miller, R. B., Lukens, J. R., Krumpe, K., Gupton, J. T., & Burnham, B. S. (2004). Lipid-Lowering Effects of Ethyl 2-Phenacyl-3-aryl-1H-pyrrole- 4-carboxylates in Rodents. Molecules, 9(3), 134-157. https://doi.org/10.3390/90300134