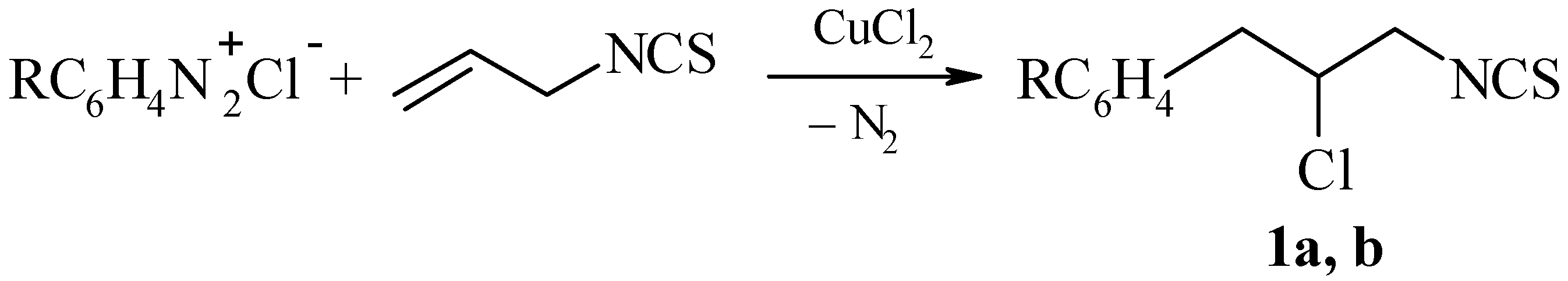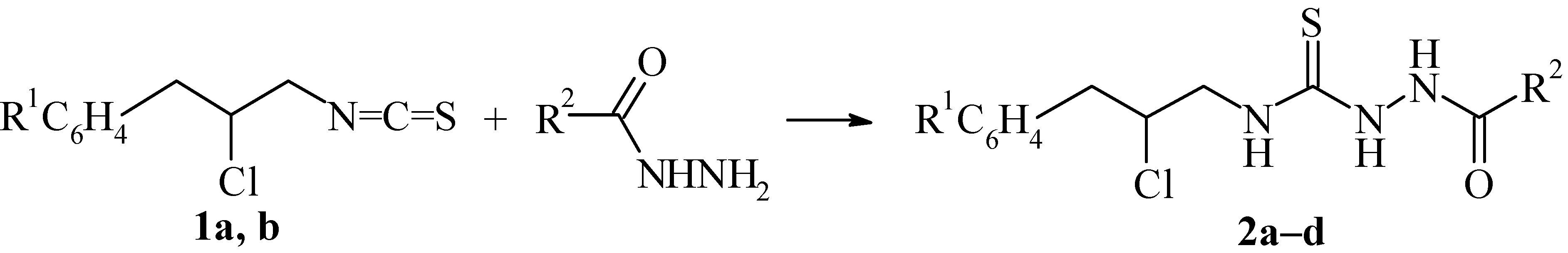General
All melting points are uncorrected.
1H-NMR spectra were obtained using a Bruker AC-200 spectrometer and were recorded at 200 MHz in DMSO-d
6. Chemical shifts are reported in ppm relative to the residual signal of the solvent. When the signals of the CH
2N or CH
2Ar groups in compounds
2a–d and
3a–d were not sharp, coupling constants were not determined. For narrow multiplets the centres of the signals is indicated. The chloroarylation of allyl isothiocyanate was carried by the method described in [
4]. The data of 3-phenyl-2-chloropropylisothiocyanate (
1a) are presented there.
3-(3-Chlorophenyl)-2-chloropropylisothiocyanate (1b); yield 27%, b.p. 108–110°C (0.5 mm), n20D 1.6066; Found: C, 48.58; H, 3.77; S, 13.25. C10H9Cl2NS requires C, 48.79; H, 3.69; S, 13.03%; 1H-NMR: δ 2.98 (dd, 1H, CH2Ar, J13 8.0 Hz, J12 14.2 Hz), 3.19 (dd, 1H, CH2Ar, J13 5.0 Hz), 3.89 (dd, 1H, CH2N, J13 5.8 Hz, J12 15.2 Hz), 4.04 (dd, 2H, CH2N, J13 4.0 Hz), 4.53 (m, 1H, CH), 7.20–7.60 (m, 4H, C6H4).
1-Acyl-4-(3-aryl-2-chloropropyl)thiosemicarbazides (2a-d). A mixture of 3-aryl-2-chloropropyl-isothiocyanate (1, 5 mmole) and the corresponding monoacylhydrazine (5 mmole) was boiled in benzene (5 mL) for 1 hour. The precipitates obtained were filtered and recrystallized from the solvent indicated.
1-Caproyl-4-(3-phenyl-2-chloropropyl)thiosemicarbazide (2a); yield 61%, m.p. 170–171°C (from dioxane); Found: C, 56.14; H, 7.22; S, 9.21; C16H24ClN3OS requires C, 56.21; H, 7.08; S, 9.38%; 1H-NMR: δ 0.90 (t, 3H, CH3), 1.24–1.36 (m, 4H, CH2CH2CH3), 1.50–1.64 (m, 2H, CH2CH2C=O), 2.22 (t, 2H, CH2C=O), 3.01(dd, 1H, CH2Ar), 3.17 (dd, 1H, CH2Ar), 3.78 (dd, 1H, CH2N), 4.03 (dd, 1H, CH2N), 4.49 (m, 1H, CH), 7.26 (m, 5H, C6H5), 10.98 (s, 1H, NHC=O).
1-Benzoyl-4-(3-phenyl-2-chloropropyl)thiosemicarbazide (2b); yield 72%, m.p. 172–173°C (from toluene); Found: C, 58.81; H, 5.32; S, 9.43; C17H18ClN3OS requires C, 58.69; H, 5.21; S, 9.22%; 1H-NMR: δ 3.05 (dd, 1H, CH2Ar), 3.19 (dd, 1H, CH2Ar), 3.77 (m, 1H, CH2N), 4.02 (m, 1H, CH2N), 4.50 (m, 1H, CH), 7.04–7.36 (m, 5H, C6H5), 7.44–7.63 (m, 3H, C6H5), 7.98–8.08 (m, 2H, C6H5 – o-H), 11.69 (s, 1H, NHC=O).
1-Caproyl-4-[3-(3-chlorophenyl)-2-chloropropyl]thiosemicarbazide (2c); yield 53%, m.p. 161–162°C (from toluene); Found: C, 50.89; H, 6.04; S, 8.64; C16H23Cl2N3OS requires C, 51.06; H, 6.16; S, 8.52%); 1H-NMR: δ 0.88 (t, 3H, CH3), 1.20–1.40 (m, 4H, CH2CH2CH3), 1.48–1.67 (m, 2H, CH2CH2C=O), 2.23 (t, 2H, CH2C=O), 3.01 (dd, 1H, CH2Ar, J13 8.6 Hz, J12 13.6 Hz), 3.19 (dd, 2H, CH2Ar, J13 7.0 Hz), 3.80 (dd, 1H, CH2N), 4.06 (dd, 1H, CH2N), 4.50 (m, 1H, CH), 7.20–7.45 (m, 4H, C6H4), 11.00 (s, 1H, NHC=O).
1-Benzoyl-4-[3-(3-chlorophenyl)-2-chloropropyl]thiosemicarbazide (2d); yield 46%, m.p. 188–189°C (from benzene); Found: C, 53.57; H, 4.31; S, 8.23; C17H17Cl2N3OS requires C, 53.41; H, 4.48; S, 8.39%; 1H-NMR: δ 3.09 (dd, 1H, CH2Ar, J13 8.0 Hz, J12 14.4 Hz), 3.21 (dd, 1H, CH2Ar, J13 6.4 Hz), 3.80 (dd, 1H, CH2N), 4.04 (dd, 1H, CH2N), 4.55 (m, 1H, CH), 7.20–7.40 (m, 4H, Ar), 7.56 (m, 3H, Ar), 8.04 (m, 2H, C6H5 – o-H), 11.64 (s, 1H, NHC=O).
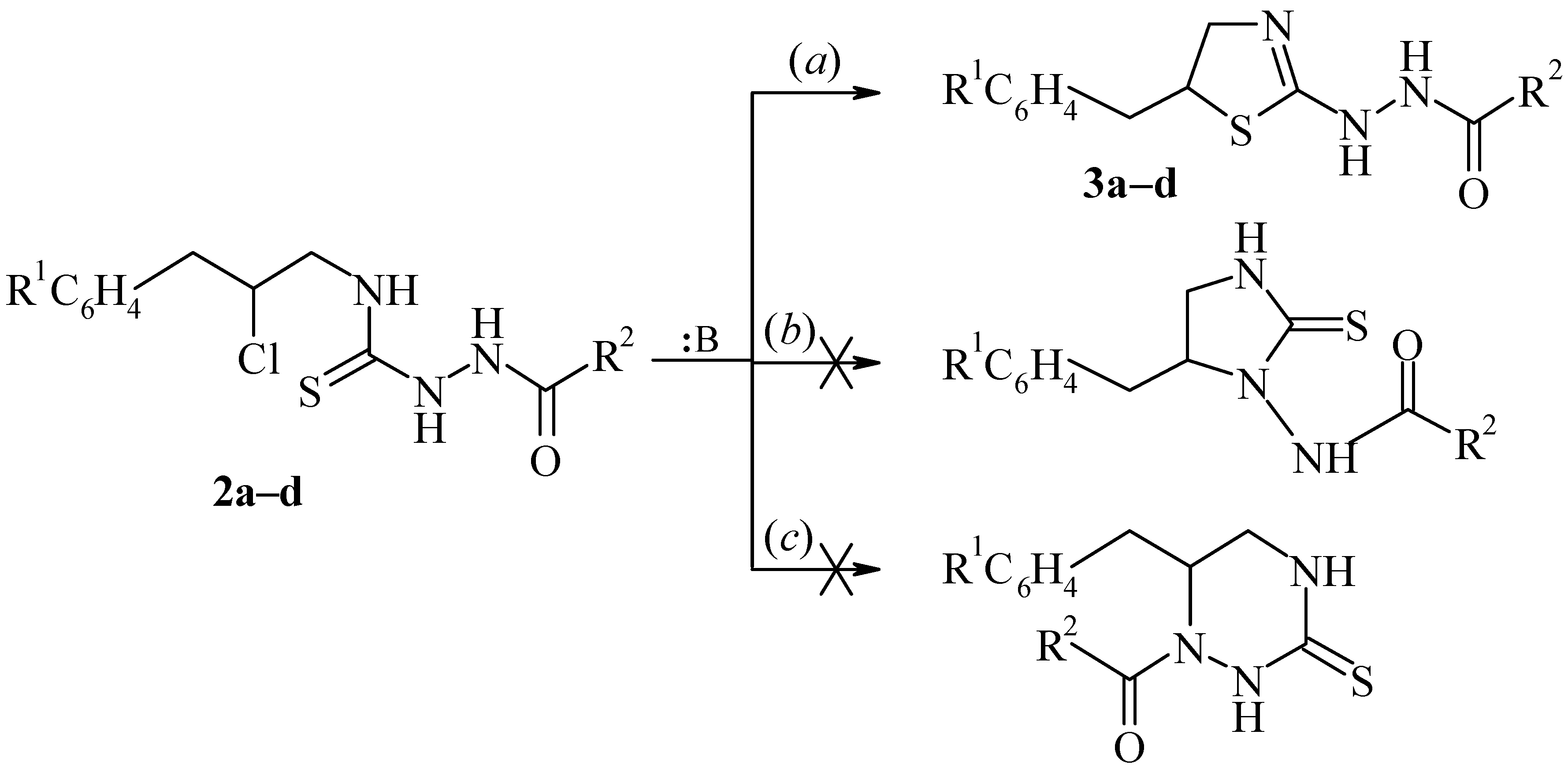
2-(2-Acylhydrazino)-5-benzyl-2-thiazolines (3a–d). Triethylamine (or N-methylmorpholine) (5 mmoles) was added to a solution of the corresponding thiosemicarbazide 2a–d (2 mmoles) in acetone (5 mL) and the mixture was boiled for 30 min. After cooling water (15 mL) was added to the mixture. The precipitate was filtered, air dried and recrystallized from the appropriate solvent.
2-(2-Caproylhydrazino)-5-benzyl-2-thiazoline (3a); yield 86%, m.p. 156–157°C (from benzene – cyclohexane, 3:1); Found: C, 62.80; H, 7.70; S, 10.61; C16H23N3OS requires C, 62.92; H, 7.59; S, 10.50%); 1H-NMR: δ 0.90 (t, 3H, CH3), 1.18–1.38 (m, 4H, CH2CH2CH3), 1.42–1.60 (m, 2H, CH2CH2C=O), 2.02 (t, 2H, CH2C=O), 2.92 (dd, 1H, CH2Ar, J13 9.2 Hz, J12 14.4 Hz), 3.04 (dd, 1H, CH2Ar, J13 6.8 Hz), 3.37 (dd, 1H, CH2N, J13 6.6 Hz, J12 8.0 Hz), 3.62 (dd, 1H, CH2N, J13 6.4 Hz), 3.94 (m, 1H, CH), 7.19–7.35 (m, 5H, C6H5).
2-(2-Benzoylhydrazino)-5-benzyl-2-thiazoline (3b); yield 56%, m.p. 143–144°C (from toluene); Found: C, 65.41; H, 5.35; S 10.11; C17H17N3OS requires C, 65.57; H, 5.50; S, 10.30%); 1H-NMR: δ 2.93 (dd, 1H, CH2Ar, J13 8.6 Hz, J12 14.0 Hz), 3.06 (dd, 1H, CH2Ar, J13 6.0 Hz), 3.40 (dd, 1H, CH2N, J13 6.4 Hz, J12 11.2 Hz), 3.66 (dd, 1H, CH2N, J13 7.0 Hz), 3.98 (m, 1H, CH), 7.10–7.33 (m, 5H, C6H5), 7.38–7.48 (m, 3H, C6H5), 7.77–7.87 (m, 2H, C6H5 – o-H).
2-(2-Caproylhydrazino)-5-(3-chlorobenzyl)-2-thiazoline (3c); yield 69%, m.p. 149–150°C (from petroleum ether – benzene, 1:5); Found: C, 56.74; H, 6.68; S, 9.33; C16H22ClN3OS requires C, 56.54; H, 6.52; S, 9.43%); 1H-NMR: δ 0.89 (t, 3H, CH3), 1.21–1.37 (m, 4H, CH2CH2CH3), 1.45–1.60 (m, 2H, CH2CH2C=O), 2.03 (t, 2H, CH2C=O), 2.88 (dd, 1H, CH2Ar, J13 8.2 Hz, J12 13.6 Hz), 3.05 (dd, 1H, CH2Ar, J13 6.4 Hz), 3.38 (dd, 1H, CH2N, J13 5.4 Hz, J12 10.4 Hz), 3.63 (dd, 2H, CH2N, J13 6.6 Hz), 3.97 (m, 1H, CH), 7.16–7.36 (m, 4H, C6H4).
2-(2-Benzoylhydrazino)-5-(3-chlorobenzyl)-2-thiazoline (3d); yield 56%, m.p. 176–177°C (from toluene); Found : C, 59.21; H, 4.44; S, 9.37; C17H16ClN3OS requires C, 59.04; H, 4.66; S, 9.27%); 1H-NMR: δ 2.92 (dd, 1H, CH2Ar, J13 8.0 Hz, J12 14.4 Hz), 3.08 (dd, 2H, CH2Ar, J13 6.2 Hz), 3.41 (dd, 1H, CH2N, J13 5.6 Hz, J12 10.2 Hz), 3.66 (dd, 1H, CH2N, J13 5.8 Hz), 4.01 (m, 1H, CH), 7.15–7.50 (m, 7H, Ar), 7.78–7.88 (m, 2H, C6H5 – o-H).