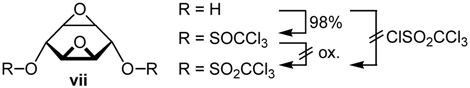Introduction
Nucleophilic substitution reactions at sp
3-carbon centers are among the most important transformations in organic chemistry. The nucleofugality of leaving groups used in organic synthesis, measured by the rates of solvolysis, covers a range of at least fourteen orders of magnitude [
1,
2]. Despite this large variety, there is still a need to improve the efficiency of many S
N2-type synthetic operations. Since halides used as (soft) alkylating reagents often tend to undergo elimination reactions on treatment with (hard) nucleophiles like e.g. alcoholates, esters of oxo-acids are widely applied alternatives. While moderately reactive phosphate esters play a crucial role in biological systems, alkyl (as also alkenyl and aryl) esters of sulfonic acids in particular are indispensable for preparative and mechanistic organic chemistry.
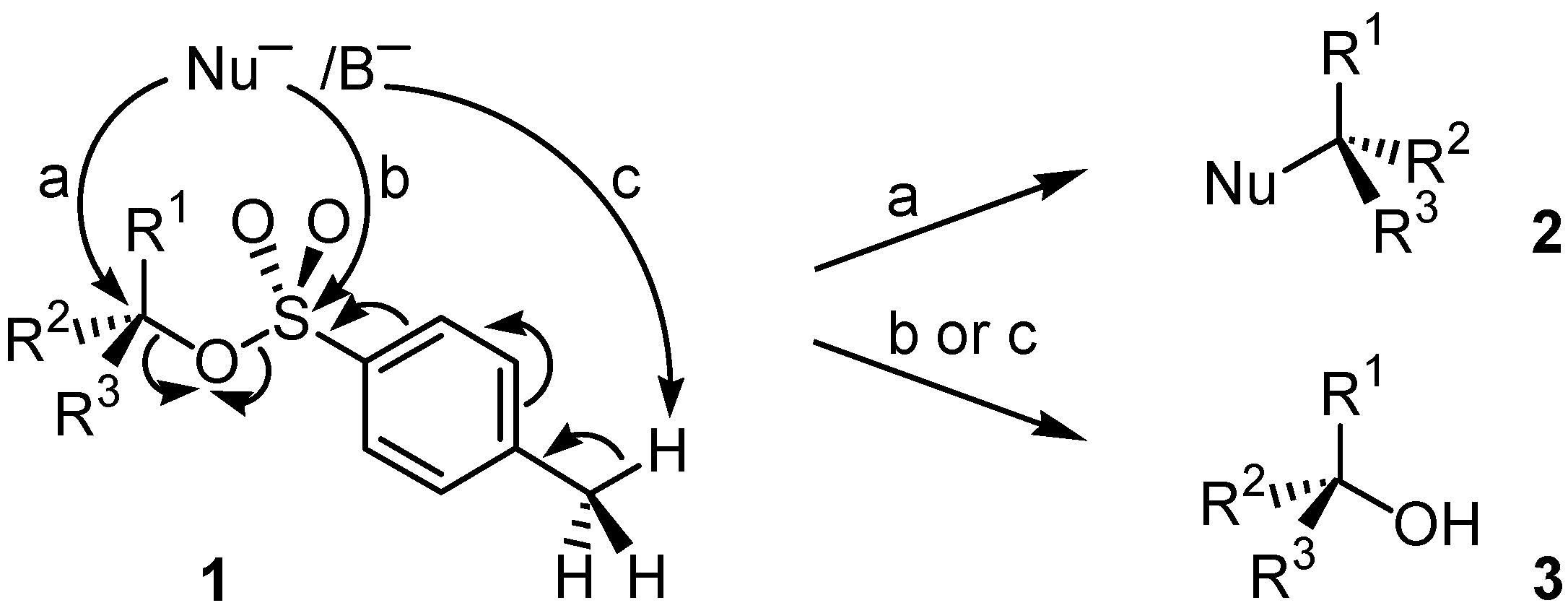
The proper choice of reagents and conditions for nucleophilic substitution reactions deserves, however, careful attention to the steric and electronic requirements of both the attacking nucleophile and the electrophile. If alkyl sulfonates are used, several side-reactions can substantially limit the value of the transformation. One major problem often encountered is unwanted sulfur-oxygen bond scission. As a typical example, S
N2 reaction by Nu¯ at the saturated carbon atom of alkyl
p-toluenesulfonates
1 (
Scheme 1, route a) has to compete with backside attack of the sulfur atom (route b) or sulfene formation if the nucleophile behaves as a strong base B¯ (route c), resulting in formation of alcohol
3 besides alkylated product
2.
Forced by the necessity to improve some demanding S
N2 substitution reactions of highly functionalized or unfavorably substituted substrates, we directed considerable efforts to the evaluation and development of more advanced sulfonyl functionalities. In this program two key aspects had to be considered: 1)
S-substitution should be retarded effectively by steric shielding, and 2) nucleofugality should be reasonably high, i.e. the reactivity of the esters should be in the range between trifluoroethanesulfonates (tresylates
4) [
3] and perfluoroalkylsulfonates (triflates
5, nonaflates
6) [
2]. The first aim could be reached with
tert-butylsulfonates
7 [
4,
5] which were, however, expected as being slightly less nucleofugic than methanesulfonates (mesylates
8) or tosylates
9. Both requirements are fulfilled by the 2,2,2-trifluoro-1,1-diphenylethanesulfonates (TDE-sulfonates
10) which can, therefore, be applied in a preparatively useful manner [
4,
5,
6]. With regard to the present paper, it is noteworthy that for the preparation of the corresponding reagent a sulfur functionality had been introduced to a fluorine containing precursor, while the opposite strategy (fluorination of a sulfur compound) was unsuccessful [
7,
8,
9,
10,
11,
12].
Regarding highly activating leaving groups, there was at that stage still a gap in the arsenal of preparatively applicable sulfonic esters between (substituted) tresylates and perfluoroalkylsulfonates. In addition to the lack of suitable reagents in this range, it is of common knowledge that the use of the latter esters is subject to certain limitations. Often problems arise from the instability of commonly used triflates
5 which result in difficulties either in storage or purity or purification of these highly reactive intermediates. Even analysis of such products deserves special attention [
13]. Moreover, the preparation of triflates is sometimes far from trivial. During the esterification of sterically congested alcohols with triflic anhydride, the formation of sulfur(IV) esters (sulfinates) has been detected, depending on the base and conditions applied [
14]. If pyridine is used as a (standard) base and solvent, pyridinium salts may be formed due to the high reactivity of triflates [
15].
With regard to the complications mentioned, we would, therefore, in this contribution like to report on some findings during our trials to prepare alkyl esters of perhalogenated sulfonic acids, in particular trichloromethyl-, fluoro-, and chlorosulfonic acid (11-13). The application of these esters in nucleophilic substitution reactions had, to our knowledge, not been studied systematically so far. When reviewing the literature we had to learn that even their preparation from the corresponding alcohols is documented only fragmentarily.
Results and Discussion
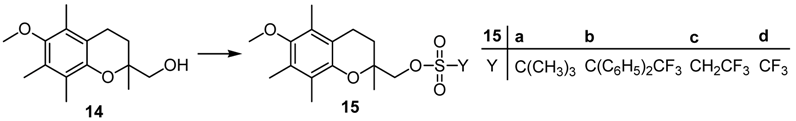
As a starting material for the preparation of representative perhalogenated esters we chose the oxa-neopentylic alcohol
14 by the following reasons:
14 is easily accessible from the commercially available carboxylic acid Trolox™. Resulting from our activities in the field of vitamin E synthesis [
16], a large variety of oxo-esters had already been prepared, most of them (including perfluoroalkylsulfonates) being easily isolable, nicely crystalline, and possessing considerable thermal stability. In addition, NMR, MS, UV, TLC, HPLC, and GC or SFC [
13] can be used as analytical methods, and samples of various structurally related by-products were available in our laboratory. Most importantly, esters
15 are ideal test systems for evaluation of the efficiency of steric shielding about the sulfonate functionality: Since the oxa-neopentylic center is not easily accessible to the nucleophile, these esters are more susceptible to sulfur attack than normal primary alkyl sulfonates. In addition, E2-elimination of the sulfonic acid is not possible, due to the lack of β-hydrogens.
Beside
tert-butylsulfonate
15a, TDE-sulfonate
15b, tresylate
15c, and triflate
15d prepared by standard and improved procedures [
2,
3,
5,
14], the nonaflate
15e was also synthesized for comparison (
Scheme 2). During preparation of
15e another limitation became apparent. In contrast to examples reported in the literature [
17], alcohol
14 did not react with (commercially available) nonafluorobutanesulfonyl fluoride under mild conditions (room temp. → refluxing CH
2Cl
2, 2,6-lutidine). After solvent exchange to 2,6-lutidine and prolonged heating (up to 90°C bath temp. for several days), alcohol
14 (59%) was re-isolated by chromatography along with decomposition products. Nonaflate
15e could be obtained in 82% yield by use of nonaflyl anhydride which had, however, first to be prepared from potassium nonafluorobutanesulfonate [
2,
18,
19] due to lack of commercial availability.
Our next aim was to gain access to trichloromethanesulfonates
11. Such esters seemed to be highly attractive electrophiles in S
N2-type reactions possessing the properties mentioned above: the three chlorine atoms should, on the one hand, strongly activate due to their powerful electron-withdrawing abilities. The trichloromethyl group on the other hand was hoped to behave like a
tert-butyl moiety in which the chlorine atoms would mimic the methyl groups of about same size, thus preventing sulfur-oxygen bond cleavage by steric hindrance. The bulkiness of the backbone should considerably influence the tendency of the sulfur(VI) atom to be attacked by a nucleophile, since this process most likely involves a trigonal bipyramidal transition state with linear arrangement of incoming nucleophile and departing leaving group, as indicated by kinetic investigations on sulfonyl chlorides [
20].
The direct esterification of hydroxy compounds with the sulfonyl chloride seems to be limited to polyhalogenated alcohols [
21,
22], while normal alkyl alcohols are unreactive [
23]. Therefore, in compliance with our expectation from earlier experiences [
24], treatment of alcohol
14 with trichloromethanesulfonyl chloride in presence of 2,6-lutidine as a base did not result in formation of trichloromethanesulfonate
15g (
Scheme 2). Instead, only starting alcohol was detected by TLC after 75 h in refluxing CH
2Cl
2. This can be interpreted as an indication for the validity of the concept of steric shielding, as explained above. The formation of chlorosulfonate
15h as the main product (36% isolated after chromatography and crystallization; 8% bissulfonate
18) under forced conditions (refluxing THF, 18 h) may be explained by attack of the sulfonyl sulfur atom by the alcohol from the less hindered side. The substitution of the CCl
3¯ group instead of chloride has its precedents in preparative applications like the haloform reaction or the generation of dichlorocarbene from methyl trichloromethanesulfinate or trichloromethanesulfonyl chloride [
25].
An alternative general strategy for the synthesis of sulfonates from alcohols is the preparation of sulfur(II) or sulfur(IV) intermediates (sulfenates, sulfinates), followed by their oxidation to the sulfur(VI) compounds. The sulfinate
16 was obtained smoothly by esterification of alcohol
14 with the sulfinyl chloride (2,6-lutidine, CH
2Cl
2, 30 min., room temp.) as a nicely crystalline material (92% after simple recrystallisation from pentane). A more rewarding transformation was the oxidation to the sulfonate: the resistance of trichloromethyl sulfinates (and structurally related compounds) towards oxidation is known, and has been applied preparatively in the almost quantitative formation of benzyl trichloromethylsulfinates from sulfenate esters and
m-chloroperbenzoic acid: Even in the presence of a large excess of reagent in refluxing chloroform, no oxidation to the sulfonates was observed [
26]. Since in a similar case [
24] even treatment with excess trifluoroperacetic acid (CH
2Cl
2, reflux) and other reagents [
27] was unsuccessful, we tested potassium permanganate as a strong oxidant.
In a first experiment, sulfinate
16 was reacted with two mole equivalents of KMnO
4 in a pyridine-water mixture at 0°C (
Scheme 2). After 15 minutes, starting material had disappeared completely, and three major products could be isolated after quenching with sodium sulfite solution, aqueous work-up, and chromatography. Alcohol
14 (20%) and the two sulfonates
15g and
15i (14% yield each) were obtained as crystalline compounds. The somewhat unexpected outcome of this oxidation reaction may be related to experiences from mechanistic studies with di- and trichloromethyl sulfenic and sulfinic derivatives: it has been stated that enhanced reactivity and participation of the trichloromethyl group in solvolytic reactions makes analogies from behavior of “normal” sulfur compounds (e.g. containing the methanesulfenyl vs. trifluoromethanesulfenyl group) questionable [
28]. Due to the only partially explored unusual reactivity of such trichloromethyl sulfur compounds, more detailed studies have to be undertaken in order to check the potential of this transformation.
Having enough material for test reactions in hand, we decided to not investigate the mechanism of that reaction and, consequently, did not optimise the conditions for preferred formation of either
15g or
15i. Although the yields of sulfonates
15g and
15i isolated are quite low, this result deserves some special comments regarding preparative applicability of such esters. It is noteworthy that the experiment described is, to the best of our knowledge, the first example for preparation of an alkyl trichloromethanesulfonate on this route starting from an alcohol. In addition to this initial aim, the unexpected formation of dichloromethanesulfonate
15i is even more remarkable: So far the chlorination of chloromethanesulfonates (prepared from chloromethanesulfonyl chloride [
29]) with N-chlorosuccinimide in hexamethylphosphoramide is reported as being the preferred method for the synthesis of aryl di- and trichloromethanesulfonates [
30]; the scope of its applicability is, however, expected to be limited by the sensitivity of functionalities contained in substrates.
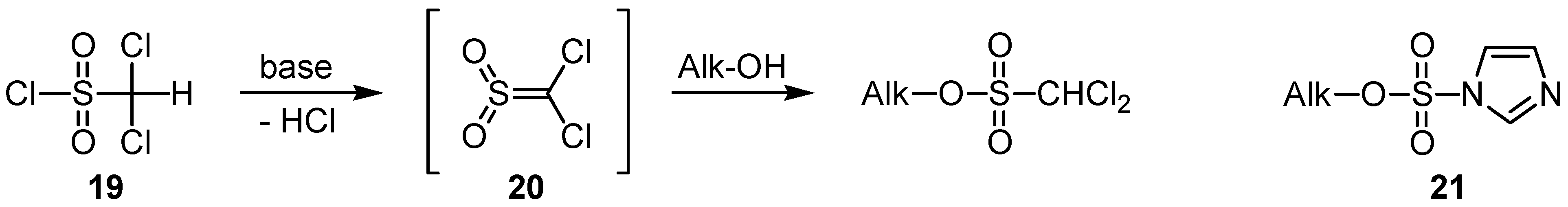
Both trichloromethanesulfonates and dichloromethanesulfonates (like e.g.
15g and
15i) should be (re-)considered as either underutilized old or promising new types of alkylating reagents, since in those esters the sulfonyl sulfur is much less prone to attack by nucleophiles than corresponding methanesulfonates or chloromethanesulfonates [
30]. In this context it is important to note that recent work has shown the (mono-) chloromethanesulfonate (monochlate) group as an extremely efficient leaving group in certain rearrangement and substitution reactions [
31]. Regarding dichloromethanesulfonates, we did not check whether their preparation is possible by direct esterification of alcohols with dichloromethanesulfonyl chloride (
19) [
32]. Most likely, this will take place via a base-induced elimination-addition process involving dichlorosulfene (
20), as outlined in
Scheme 3.
Furthermore, fluorosulfonates
12 [
33] and chlorosulfonates
13 [
34] are of particular interest for nucleophilic substitution reactions. Therefore, the preparation of sulfonates
15f and
15h was envisaged (see
Scheme 2). According to literature examples [
35], alcohol
14 was treated with sulfuryl chloride fluoride (NEt
3, CH
2Cl
2, –78°C). Surprisingly, the chlorosulfonate
15h already found accidentally during attempted synthesis of trichloroderivative
15g, was obtained in high yield. We assume that in this case the intermediately formed fluorosulfonate
15f was transformed to the chlorosulfonate
15h by substitution of fluoride by the better nucleophile Cl¯ [
36]. Remarkably, no fluoride at all could be found by microanalysis of the crystalline product isolated in 72% yield. In this regard it has to be mentioned that chlorosulfonates
13 can be obtained from alcohols and sulfuryl chloride [
34,
37]; they have also been used as
in-situ generated precursors of imidazole-1-sulfonates (imidazylates)
21 [
38]. From an industrial viewpoint, it is noteworthy that a more economical combined use of sulfur dioxide and chlorine gas as a replacement of sulfuryl chloride has been documented [
39]. The fluorosulfonate
15f could be synthesized by treatment of alcohol
14 with (toxic) fluorosulfonic anhydride (freshly prepared from fluorosulfonic acid and cyanuric chloride [
2,
40]) (2,6-lutidine, Et
2O, –78°C → room temp., 44% yield unoptimized). The material isolated contained ca. 4% of chlorosulfonate
15h, according to SFC and microanalysis, which is assumed to have its origin in chlorine-containing by-products of anhydride preparation. Ethyl ether derivative
17 (6%) was also isolated, indicating a participation of the diethyl ether solvent in this transformation.
Experimental
General
All reactions were performed under an argon atmosphere. Room temperature was 20 to 22°C. All reagents and chemicals not mentioned separately were obtained from Fluka or Aldrich, or were technical grade from Roche, and were used without further purification, if not stated individually. Nonafluorobutanesulfonic anhydride was prepared by treatment of potassium nonafluorobutane-sulfonate with sulfuric acid/oleum and subsequent dehydration of the sulfonic acid by distillation from excess phosphorus pentoxide mixed with Celite [
2,
18,
19]. Fluorosulfonic anhydride was prepared from fluorosulfonic acid and cyanuric chloride [
2,
40]. Dichloromethane was freshly distilled from CaH
2, and THF from Na/ benzophenone under argon. For transfer of solvents, reagents (if liquid), or their solutions, syringes, cannulae and rubber septa were used. The reactions were monitored by thin layer chromatography (TLC) using glass plates coated with silica gel 60 F
254, 0.25 mm thickness (Merck); spots were visualized by spraying with solutions of 2% vanillin in ethanol/water/sulfuric acid or ammonium molybdate/cerium sulfate in water/sulfuric acid, and subsequent heating. Flash chromatography was performed using silica gel 60, 0.040-0.063 mm (Merck) under max. 0.2 bar argon pressure. IR spectra were measured with a Nicolet 170-SX FT-IR instrument, values are reported in cm
‒1. NMR spectra were obtained using Bruker AC-250 and AC-250E spectrometers and were recorded at 250.1 MHz (
1H) and 235.36 MHz (
19F), respectively, for CDCl
3-solutions with tetramethylsilane (TMS) as an internal standard (
1H) or referred to CFCl
3 (
19F); chemical shifts in ppm (δ), m
c means centered multiplet, “t” structured triplet, coupling constants in Hz. EI-MS were obtained with an MS9 instrument (AEI, Manchester, GB); ionisation energy 70 eV, temp.of ion source 250°C; m/z values in %, relative to the base peak (= 100%). Gas chromatographic (GC) analyses were performed using a Dani 3800 instrument with a flame-ionisation detector (FID) and split/splitless injector, 15-m PS-086 column, carrier gas hydrogen, temperature program 50-330°C, injector temp. 40-300°C, detector temp. 330°C, values in area%. Supercritical fluid chromatography (SFC) was performed with a Lee Scientific SFC Model 600 (Dionex, Salt Lake City, UT) and a Carlo Erba model 3000 apparatus equipped with an FID operated at 375 or 325°C; SFC-grade unmodified carbon dioxide (Messer-Griesheim, Germany) was used as a mobile phase, and time split injections were performed with a Valco C14W high-pressure valve that had a 200-nL loop; injection time was 50 ms; 10-m × 50-μm i.d. and 25-m × 100-μm fused-silica capillary columns, coated with biphenyl-30 as a stationary phase, and a fused-silica restrictor (Dionex) were used; colunm temp. 100°C, if not stated otherwise; density programs 0.2-0.75 g
·mL
-1 with a linear ramp at 0.01 g
·mL
-1·min
-1 or 0.4-0.75 g
·mL
-1 with a linear ramp at 0.005 g
·mL
-1·min
-1 (cf. [
13]).
Preparation of nonafluorobutanesulfonate 15e
The magnetically stirred solution of alcohol
14 (6.01 g, 24.0 mmol) in freshly dist. CH
2Cl
2 (100 mL) contained in a 250-mL two-necked round-bottomed flask was cooled to –78°C (
14 partially crystallized out at this temp.). Then freshly dist. 2,6-lutidine (5.8 mL, 50.0 mmol) and, subsequently, nonaflyl anhydride (28.0 g, 48.1 mmol) were added in one portion. The cooling bath was removed. While warming up to room temp. the colour of the reaction mixture turned to red. At room temp. total conversion was detected by TLC [1:3 EtOAc/ hexane, R
f(
14) 0.2, R
f(
15e) 0.5]. The reaction mixture was diluted with CH
2Cl
2 and poured on ice/water. The organic layer was washed with sat. NaHCO
3 and NaCl solutions, and dried over Na
2SO
4/CaCl
2. Evaporation under reduced pressure at 20°C (rotary evaporator; caution: foaming!) gave 16.7 g crude sulfonate, which was dissolved in Et
2O (100 mL) and CH
2Cl
2 (10 mL), treated with active carbon and Speedex, filtered, and evaporated to an easy-to-stir slurry. Storage at –20°C overnight gave crystals which were washed with a small amount of Et
2O and dried under high vacuum (11.34 g). These crystals were dissolved in Et
2O (50 mL), and the solution treated with charcoal/Speedex. Dilution of the colourless solution with pentane and concentration by evaporation under reduced pressure at 20°C gave a slurry which was stored at –20°C for several hours. Suction filtration and washing with a small amount of pentane yielded 7.65 g of colourless shiny crystals, which were used for analyses. The mother liquors were collected and chromatographed (200 g SiO
2, EtOAc/ hexane 1:19) to give additional 2.82 g sulfonate; combined yield 82%. An experiment with Na
2CO
3 as a base in CH
2Cl
2 (–15 → 20°C) [
18] gave 32%
15e after chromatography.
[(RS)-3,4-Dihydro-6-methoxy-2,5,7,8-tetramethyl-2H-1-benzopyran-2-yl]methyl nonafluorobutane-sulfonate (15e): M.p. 88-89.5°C; purity 99.7% (SFC); 1H-NMR: δ 1.36 (s, Ctert-CH3, 3 H), 1.85 (mc, CH2CH2Ar, 1 H), 1.99 (mc, CH2CH2Ar, 1 H), 2.08 (s, Ar-CH3, 3 H), 2.15 (s, Ar-CH3, 3 H), 2.19 (s, Ar-CH3, 3 H), 2.66 (“t”, CH2CH2Ar, 2 H, J = 6.9), 3.63 (s, OCH3, 3 H), 4.46 (AB, CH2OSO2, 1 H, J = 10.1), 4.53 (AB, CH2OSO2, 1 H, J = 10.1); 19F-NMR: δ -126.78 (mc, CF3-CF2, 2 F), -121.86 (mc, CF3-CF2-CF2, 2 F), -111.20 (mc, CF3-CF2-CF2-CF2, 2 F), -81.18 (t, CF3, 3 F); IR (KBr): 2940, 1466, 1407, 1355, 1299, 1258, 1236, 1200, 1145, 1094, 1040, 1011, 964, 789, 736, 700, 659, 594, 507 cm-1; MS: m/z 532 (M•+, 100), 517 (M -15, 10), 233 (M -ONf, 17), 232 (M -HONf, 15), 219 (32), 217 (42), 179 (C11H15O2•+, 79); Anal. Calcd. for C19H21F9O5S (532.41): C 42.86, H 3.98, F 32.11, S 6.02, Found C 42.72, H 4.15, F 32.54, S 5.75.
Esterification of alcohol 14 with fluorosulfonic anhydride: formation of fluorosulfonate 15f and ethyl ether 17
In a 100-mL two-necked round-bottomed flask, 2,6-lutidine (1.16 mL, 10.0 mmol) was added dropwise to the solution of fluorosulfonic anhydride (1.68 g, 9.2 mmol, freshly distilled after treatment with conc. H2SO4; caution: Highly Toxic!) in Et2O (40 mL) at –78°C while stirring. Alcohol 14 (1.25 g, 5.0 mmol) dissolved in Et2O (20 mL) was then added during 5 min. (14 crystallized out partly during this operation). The cooling bath was removed. While warming up to room temperature the mixture became a white suspension. TLC [EtOAc/ hexane 1:3, Rf(14) 0.30, Rf(17) 0.80, Rf(15f) 0.58] indicated incomplete conversion. The reaction mixture was poured onto cooled (0°C) 2 N H2SO4. After separation of the phases, the organic layer was extracted with water, dried, and evaporated to dryness (rotary evaporator, reduced pressure). Filtration of the residue through a pad of silica gel gave 2.246 g of crude material which was chromatographed (470-mL SiO2 Büchi medium-pressure column, Et2O/ hexane 1:9). The first fraction contained 88 mg (6%) of oily ethyl ether 17. The second fraction gave 730 mg (44%) of sulfonate 15f (containing ca. 4% of chlorosulfonate 15h; SFC, microanalysis), which after recrystallization from a small amount of Et2O using charcoal afforded 691 mg (42%) of colourless solid.
rac-(3,4-Dihydro-6-methoxy-2,5,7,8-tetramethyl-2H-1-benzopyran-2-yl)methyl fluorosulfonate (
15f). M.p. 69-70°C; purity 95.8%
15f + 4.2%
15h (SFC at 80°C), 92.9%
15f + 3.5%
15h (SFC at 100°C; indicating thermal decomposition during analysis [
13]);
1H-NMR: δ 1.37 (s, C
tert-CH
3, 3 H), 1.86 (m
c, C
H2CH
2Ar, 1 H), 2.00 (m
c, C
H2CH
2Ar, 1 H), 2.08 (s, Ar-CH
3, 3 H), 2.15 (s, Ar-CH
3, 3 H), 2.19 (s, Ar-CH
3, 3 H), 2.66 (“t”, CH
2C
H2Ar, 2 H, J = 6.9), 3.63 (s, OCH
3, 3 H), 4.43 (A
B, CH
2OSO
2, 1 H, J = 9.9), 4.50 (
AB, CH
2OSO
2, 1 H, J = 9.9); IR (KBr): 2941, 1436, 1403, 1302, 1255, 1226, 1178, 1121, 1089, 1061, 977, 929, 880, 793, 596, 546 cm
-1; MS: 332 (M
•+, 100), 317 (M -15, 19), 217 (46), 179 (C
11H
15O
2•+, 80), 178 (66); Anal. Calcd. for C
15H
21FO
5S (332.39): C 54.20, H 6.37, F 5.72, S 9.65, Found C 54.18, H 6.47, F 5.53, S 9.55, Cl 0.51.
rac-2-(Ethoxymethyl)-3,4-dihydro-6-methoxy-2,5,7,8-tetramethyl-2H-1-benzopyran (17). Oil; purity 97.9% (GC); 1H-NMR: δ 1.19 (t, CH2-CH3, 3 H, J = 7.0), 1.28 (s, Ctert-CH3, 3 H), 1.76 (mc, CH2CH2Ar, 1 H), 1.98 (mc, CH2CH2Ar, 1 H), 2.08 (s, Ar-CH3, 3 H), 2.14 (s, Ar-CH3, 3 H), 2.18 (s, Ar-CH3, 3 H), 2.59 (“t”, CH2CH2Ar, 2 H, J = 6.8), 3.38 (AB, CH2O, 1 H, J = 9.9), 3.48 (AB, CH2O, 1 H, J = 9.9), 3.63 (s, OCH3, 3 H), 3.57 and 3.58 (2 × q, CH2-CH3, 2 H, J = 7.0); IR (film): 2975, 2932, 2863, 1457, 1404, 1375, 1330, 1287, 1255, 1160, 1117, 1012, 929, 673 cm-1; MS: 278 (M•+, 46), 219 (M -CH2OEt, 100), 179 (C11H15O2•+, 72); Anal. Calcd. for C17H26O3 (278.39): C 73.35, H 9.41, Found C 72.36, H 9.40.
Preparation of trichloromethanesulfinate 16
NEt3 (2.65 mL, 23.0 mmol) was added to the magnetically stirred solution of alcohol 14 (5.01 g, 10.0 mmol) in CH2Cl2 (100 mL), contained in a 250-mL two-necked round-bottomed flask at room temperature. Subsequently, trichloromethanesulfinyl chloride (2.78 mL, 24.0 mmol) was added dropwise (for TLC data see following procedure). The reaction mixture was diluted with CH2Cl2 and extracted twice with cooled (0°C) 2 N H2SO4. After separation of the phases, the organic layer was dried (MgSO4) and evaporated to dryness (high vacuum). The 9.12 g of crude 16 were recrystallized from 10 mL pentane (ca. –20°C, overnight, washing of crystals with cold pentane), giving 7.65 g (92%)of colourless crystals.
[(RS)-3,4-Dihydro-6-methoxy-2,5,7,8-tetramethyl-2H-1-benzopyran-2-yl]methyl (RS)-trichlorometha-nesulfinate (16). Mixture of diastereomers, ca. 1:1 (by NMR); m.p. 80-83°C; purity 99.3% (SFC); 1H-NMR: δ 1.33 and 1.35 (2 × s, Ctert-CH3, 3 H), 1.75-1.91 (m, CH2CH2Ar, 1 H), 1.92-2.11 (m, CH2CH2Ar, 1 H), 2.10 and 2.12 (2 × s, Ar-CH3, 3 H), 2.15 (s, Ar-CH3, 3 H), 2.19 (s, Ar-CH3, 3 H), 2.64 (“t”, CH2CH2Ar, 2 H, J = 6.8), 3.626 and 3.632 (2 × s, OCH3, 3 H), 4.19 (AB, CH2OSO, 1 H of diastereomer 1, J = 11.0), 4.25 (AB, CH2OSO, 1 H of diastereomer 2, J = 10.9), 4.32 (AB, CH2OSO, 1 H of diastereomer 2, J = 10.9), 4.38 (AB, CH2OSO, 1 H of diastereomer 1, J = 10.9); IR (KBr): 2994, 2939, 1459, 1403, 1304, 1254, 1187, 1115, 1088, 1061, 1041, 1009, 962, 925, 901, 827, 793, 736, 544 cm-1; MS: 414/416/418 (M•+, 8.5/8/2.5), 297 (M -CCl3, 1.3), 233 (M -SO2CCl3, 100); Anal. Calcd. for C16H21Cl3O4S (415.76): C 46.22, H 5.09, Cl 25.58, S 7.71, Found C 46.31, H 5.21, Cl 25.50, S 7.70.
Oxidation of trichloromethanesulfinate 16 with potassium permanganate: formation of sulfonates 15g and 15i
In a 250-mL round-bottomed flask, water (10 mL) was added to the solution of sulfinate 16 (2.078 g, 5.0 mmol) in pyridine (50 mL). This mixture was cooled to 0°C, and KMnO4 (1.58 g, 10.0 mmol) was added at this temperature. TLC [EtOAc/ hexane 1:3, Rf(16) 0.47, Rf(15g) 0.42, Rf(15i) 0.36, Rf(14) 0.21] showed complete disappearance of starting sulfinate within 15 minutes. The reaction was quenched by addition of a soln. of Na2S2O5 (3.80 g, 20.0 mmol) in water (20 mL) at 0°C. After short stirring at this temperature, the resulting white suspension formed was filtered. The solid material filtered off was retained for further treatment, vide infra. The clear filtrate was evaporated to dryness. The red residue was taken up in a mixture of Et2O and water. The organic phase was washed with sat. NaCl soln., and the water phase was extracted with Et2O. The combined organic phases were dried over MgSO4 and evaporated to dryness. The resulting 1.107 g of crude material were chromatographed (40 g SiO2, EtOAc/hexane 1:4, 10-mL fractions) to give a first fraction of 671 mg containing 15g and 15i, and 202 mg crystalline alcohol 14 (16%). The mixture obtained from the first fraction was separated on a Merck Lobar® column (size B, 310 × 25 mm, LiChroprep® Si 60, 40-63 μm; EtOAc/ hexane 1:19, 10-mL fractions): Successively, 301 mg (14%) trichlorosulfonate 15g and 305 mg (14%) dichloro-sulfonate 15i were eluted and obtained as colourless crystals after distilling off the solvent. The 301 mg of 15g were stirred up with hexane (2 mL) under ultrasonication, filtered off, and dried under high vacuum: 282 mg (13%) colourless 15g, which was used for obtaining the characterization data. The filtration sludge from the first filtration (after quenching of the reaction by addition of hydrogensulfite soln., see above) was stirred up with water and filtered. The 1.34 g of undissolved solids thus obtained were taken up in warm 2 N HCl (odour of SO2) and extracted with ethyl acetate. From this organic phase an additional 52 mg (4%) of alcohol 14 were obtained after evaporation under reduced pressure.
[(RS)-3,4-Dihydro-6-methoxy-2,5,7,8-tetramethyl-2H-1-benzopyran-2-yl]methyl trichloromethane-sulfonate (15g). M.p. 104-105°C; purity 100% (SFC); 1H-NMR: δ 1.39 (s, Ctert-CH3, 3 H), 1.86 (mc, CH2CH2Ar, 1 H), 2.02 (mc, CH2CH2Ar, 1 H), 2.10 (s, Ar-CH3, 3 H), 2.15 (s, Ar-CH3, 3 H), 2.19 (s, Ar-CH3, 3 H), 2.66 (“t”, CH2CH2Ar, 2 H, J = 6.9), 3.63 (s, OCH3, 3 H), 4.54 (AB, CH2OSO2, 1 H, J = 10.15), 4.60 (AB, CH2OSO2, 1 H, J = 10.15); IR (KBr): 2993, 2933, 1463, 1398, 1385, 1296, 1256, 1193, 1177, 1119, 1089, 1062, 1012, 968, 907, 866, 810, 594, 572, 515 cm-1; MS: 430/432/434 (M•+, 39/39/17), 415/417/419 (M -15, 5/5/2), 232 (M -HOSO2CCl3, 19), 219 (M- H2COSO2CCl3, 47), 217 (61), 179 (C11H15O2•+, 100); Anal. Calcd. for C16H21Cl3O5S (431.76): C 44.51, H 4.90, Cl 24.63, S 7.43, Found C 44.69, H 5.00, Cl 24.62, S 7.28.
[(RS)-3,4-Dihydro-6-methoxy-2,5,7,8-tetramethyl-2H-1-benzopyran-2-yl]methyl dichloromethane-sulfonate (15i ). M.p. 84-85°C; purity 100% (SFC); 1H-NMR: δ 1.34 (s, Ctert-CH3, 3 H), 1.83 (mc, CH2CH2Ar, 1 H), 2.03 (mc, CH2CH2Ar, 1 H), 2.09 (s, Ar-CH3, 3 H), 2.15 (s, Ar-CH3, 3 H), 2.19 (s, Ar-CH3, 3 H), 2.66 (“t”, CH2CH2Ar, 2 H, J = 6.7), 3.63 (s, OCH3, 3 H), 4.42 (AB, CH2OSO2, 1 H, J = 10.5), 4.55 (AB, CH2OSO2, 1 H, J = 10.5), 6.41 (s, CHCl2, 1 H); IR (KBr): 2992, 2940, 1467, 1402, 1370, 1304, 1258, 1193, 1174, 1120, 1094, 1062, 1004, 967, 832, 802, 781, 705, 557, 514 cm-1; MS: 396/398/400 (M•+, 51/36/8), 381/383/385 (M -15, 6/4/1), 232 (M -HOSO2CHCl2, 10), 219 (M- H2COSO2CHCl2, 30), 217 (55), 179 (C11H15O2•+, 100); Anal. Calcd. for C16H22Cl2O5S (397.31): C 48.37, H 5.58, Cl 17.85, S 8.07, Found C 48.47, H 5.56, Cl 17.76, S 8.00.
Reaction of alcohol 14 with trichloromethanesulfonyl chloride: formation of chlorosulfonate 15h and bissulfonate 18
2,6-Lutidine (2.79 mL, 24.0 mmol) was added to a solution of alcohol 14 (5.01 g, 10.0 mmol) in CH2Cl2 (100 mL). To this solution trichloromethanesulfonyl chloride (5.01 g, 3.0 mmol) was added at room temp. while stirring. After 20 min. at room temp. and 75 h at reflux only starting material could be detected by TLC, therefore the solvent was replaced by adding THF (100 mL) and subsequently distilling off CH2Cl2 and the mixture was further refluxed (bath temp. 80°C) for 18 h [TLC: EtOAc/ hexane 1:3, Rf(14) 0.24, Rf(15h) 0.44, Rf(18) 0.38]. The resulting complex reaction mixture was partitioned between Et2O and water (200 mL each). The phases were separated, and the water phase was extracted with additional Et2O (200 mL). The combined organic phases were washed with 2 N H2SO4 (100 mL), dried over MgSO4 and evaporated to dryness. The resulting 5.98 g of crude brown oil was subjected to chromatography (60 g SiO2, EtOAc/hexane 1:9) from which compounds 15h (643 mg colourless crystals, 36%, after recryst. from pentane) and 18 (108 mg colourless crystals, 8%) were collected.
Sulfuric acid bis[[(R/S)-3,4-dihydro-6-methoxy-2,5,7,8-tetramethyl-2H-1-benzopyran-2-yl]methyl] diester (18). Mixture of diastereomers, not pure; 1H-NMR: δ 1.26-1.32 (several s, Ctert-CH3, 6 H), 1.7-2.1 (m, CH2CH2Ar, 4 H), 2.04-2.25 (several s, Ar-CH3, 18 H), 2.61 (“t”, CH2CH2Ar, 4 H), 3.61-3.64 (several s, OCH3, 6 H), 4.1-4.3 (m, 4 H); MS: 562 (M•+, 5), 498 (M –SO2, 2), 482 (M –SO3, 0.1), 330 (20), 250 (35), 233 (40), 219 (73), 217 (20), 179 (C11H15O2•+, 100).