Synthesis of Some Aldoxime Derivatives of 4H-Pyran-4-ones
Abstract
:Introduction
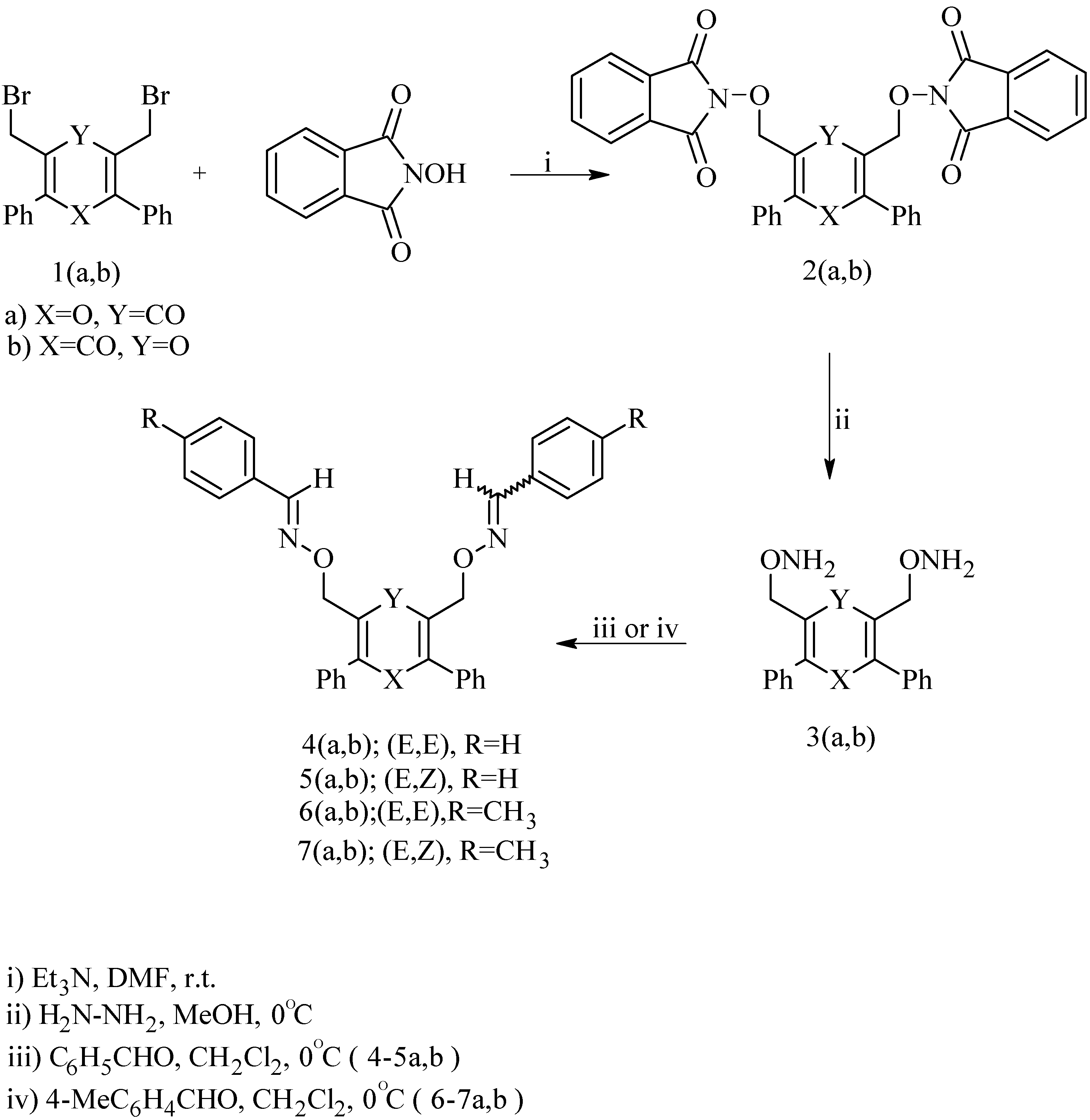
Results and Discussion
Conclusions
Experimental
General
General procedure for preparation of di(N-phthalimidoxymethyl) pyranones 2a,b.
General procedure for preparation of di(aminoxymethyl) pyranones 3a,b.
General procedure for preparation of aldoxime derivatives of 4H-pyran-4-ones 4-7a,b.
References and Notes
- Moriguchi, T.; Matsuura, H.; Itakura, Y.; Katsuki, H.; Saito, H.; Nishiyama, N. Allixin, a phytoalexin produced by garlic, and its analogs as novel exogenous substances with neurotrophic activity. Life Sci. 1997, 61, 1413–1420. [Google Scholar]
- Murrary, R. D. H. Naturally occurring oxygen-ring compounds. Aromat. Heteroaromat. Chem. 1978, 5, 472–501. [Google Scholar]
- Clark, B. P.; Ross, W. J.; Todd, A. 6-Substituted pyranone compounds for pharmaceutical uses. Ger Offen 3,012,584; [Chem. Abstr., 1981, 94, P121322f],
- Clark, B. P.; Ross, W. J.; Todd, A. 5-Substituted pyranone compounds for pharmaceutical uses. Ger Offen 3,012,597; [Chem. Abstr., 1981, 94, P83945b],
- Shahrisa, A.; Tabrizi, R.; Ahsani, H. R. A novel method for the synthesis of 4H-pyran-4-one derivatives. Org. Prep. & Proced. Int. 2000, 32, 47–55. [Google Scholar]
- Shahrisa, A.; Tabrizi, R. Synthesis and characterization of new macrocyclic polyether-diester ligands containing a 4H-pyran-4-one subcyclic unit. Iran. J. Chem. & Chem. Eng. 1999, 18, 91–97. [Google Scholar]
- Yamamura, S.; Nishiyama, S. Synthetic studies on polypropionate-derived 4-pyrone-containing marine natural products. Bull. Chem. Soc. Jpn. 1997, 70, 2025–2037. [Google Scholar]
- Naito, T.; Kobayashi, H.; Ogura, H.; Zama, K. Preparation of pyranone compounds as intermediate for cephalosporin derivatives. Jpn. Kokai Tokkyo Koho JP 04 91,088; [Chem. Abstr., 1992, 117, 131069m],
- Allowhn, J.; Brumm, M.; Frenking, G.; Hornivius, M.; Massa, W.; Steubert, F. W.; Wocadlo, S. Synthese und konformationsanalysen von pyranophanonen und pyrylophanium-verbinung mit intraannularen substituenten. J. Prakt. Chem. 1993, 335, 503–514. [Google Scholar]
- Massa, W.; Schween, M.; Steuber, F. W.; Wocadlo, S. Konformationsanalysen von pyrolytisch erzeugten [2.2] phanen des 4-pyranons. Chem. Ber. 1990, 123, 1119–1128. [Google Scholar]
- Girard, A. Organic Synthesis Collective; John Wiley & Sons: Chichester (U.K.), 1943; Vol. 2, p. 86. [Google Scholar]
- Sample Availability: Available from the authors.
© 2002 by MDPI (http://www.mdpi.org). Reproduction is permitted for non commercial purposes.
Share and Cite
Abrishami, F.; Teimuri-Mofrad, R.; Bayat, Y.; Shahrisa, A. Synthesis of Some Aldoxime Derivatives of 4H-Pyran-4-ones. Molecules 2002, 7, 239-244. https://doi.org/10.3390/70200239
Abrishami F, Teimuri-Mofrad R, Bayat Y, Shahrisa A. Synthesis of Some Aldoxime Derivatives of 4H-Pyran-4-ones. Molecules. 2002; 7(2):239-244. https://doi.org/10.3390/70200239
Chicago/Turabian StyleAbrishami, Fateme, Reza Teimuri-Mofrad, Yadollah Bayat, and Aziz Shahrisa. 2002. "Synthesis of Some Aldoxime Derivatives of 4H-Pyran-4-ones" Molecules 7, no. 2: 239-244. https://doi.org/10.3390/70200239
APA StyleAbrishami, F., Teimuri-Mofrad, R., Bayat, Y., & Shahrisa, A. (2002). Synthesis of Some Aldoxime Derivatives of 4H-Pyran-4-ones. Molecules, 7(2), 239-244. https://doi.org/10.3390/70200239



