Synthesis of 1-(2'-O-Methyl-ß-D-ribofuranosyl)-1H-imidazo[4,5-d]pyridazine-4,7(5H,6H)-dione: An Attractive Building Block for Antisense and Triple-helical Applications
Abstract
:Introduction
Results and Discussion
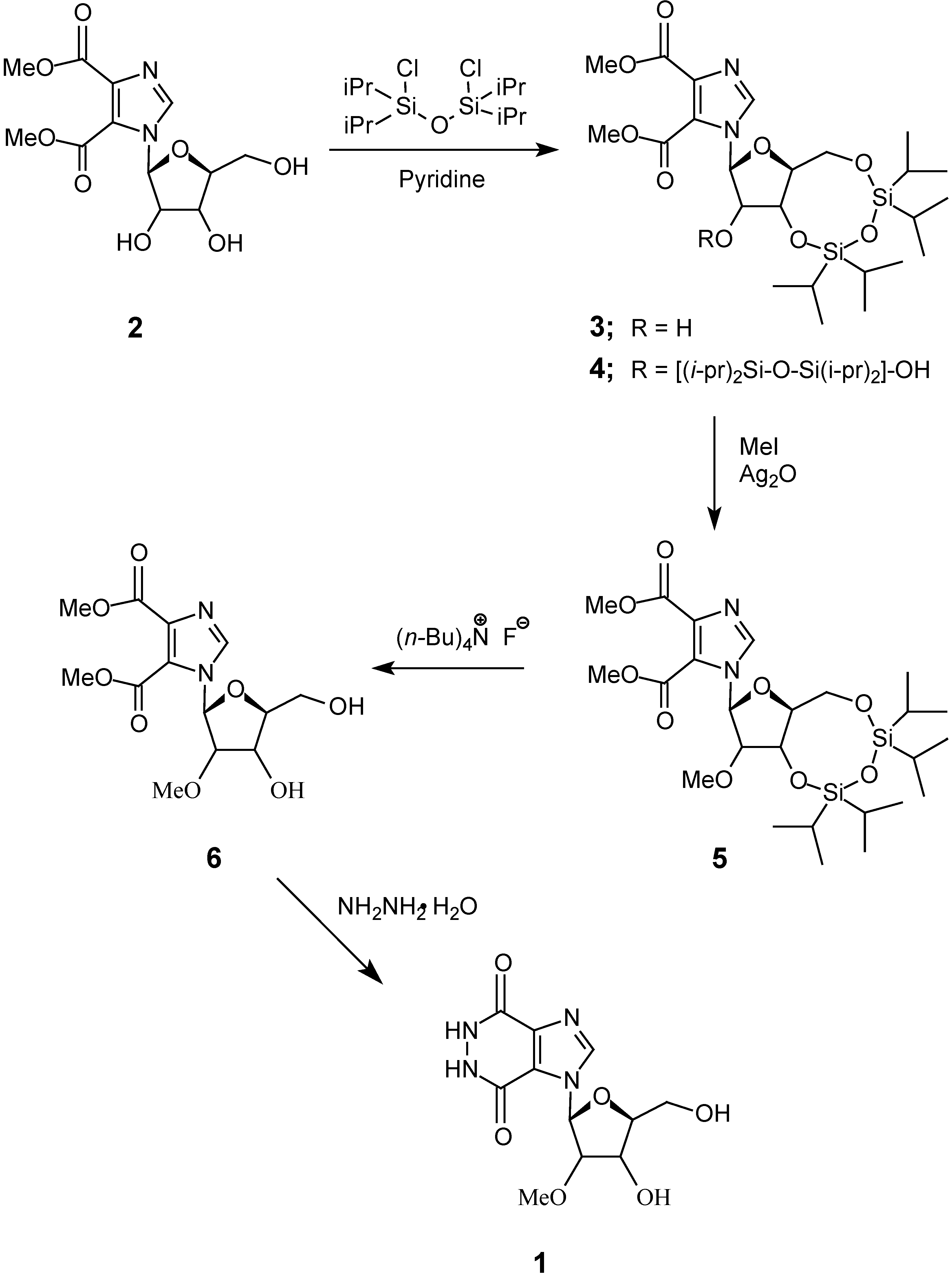
Experimental
General
Acknowledgments
References
- Ushijima, K.; Ishibashi, T.; Yamakawa, H.; Tsukahara, S.; Takai, K.; Maruyama, T.; Takaku, H. Biochemistry 1999, 38, 6570.
- Wan, M. S.; Fell, P. L.; Akhtar, S. Antisense Nucleic Acid Drug Dev. 1998, 8, 309.
- Hou, Y. M.; Gamper, H. B. Biochemistry 1996, 35, 15340.
- Lubini, P.; Zurcher, W.; Egli, M. Chem. Biol. 1994, 1, 39.
- Lesnik, E. A.; Guinosso, C. J.; Kawasaki, A. M.; Sasmor, H.; Zounes, M.; Cummins, L. L.; Ecker, D. J.; Cook, P. D.; Freier, S. M. Biochemistry 1993, 32, 7832.
- Wyss, P. C.; Fischer, U. Helv. Chim. Acta 1978, 61, 3149. Cook, P. D.; Dea, P.; Robins, R. K. J. Heterocyclic Chem. 1978, 15, 1. Cook, P. D.; Robins, R. K. Nucleic Acid Chemistry, Part 1; Townsend, L. B, Tipson, R. S., Eds.; John Wiley, Inc.: New York, 1978; p. 211. [Google Scholar] Fischer, U.; Wyss, P. C. Fischer, U.; Wyss, P. C. Tapiero, C.; Imbach, J. L.; Panzicka, R. P.; Townsend, L. B. J. Carbohyd. Nucleosides Nucleotides 1976, 3, 191.
- Markiewicz, Wojciech T. J. Chem. Res. (S) 1979, 1, 24.
- Chen, H. –M.; Hosmane, R. S. Molecules 2000, 5, 1187.
- Sample Availability: Available from authors
© 2001 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Chen, H.-M.; Hosmane, R.S. Synthesis of 1-(2'-O-Methyl-ß-D-ribofuranosyl)-1H-imidazo[4,5-d]pyridazine-4,7(5H,6H)-dione: An Attractive Building Block for Antisense and Triple-helical Applications. Molecules 2001, 6, 203-207. https://doi.org/10.3390/60300203
Chen H-M, Hosmane RS. Synthesis of 1-(2'-O-Methyl-ß-D-ribofuranosyl)-1H-imidazo[4,5-d]pyridazine-4,7(5H,6H)-dione: An Attractive Building Block for Antisense and Triple-helical Applications. Molecules. 2001; 6(3):203-207. https://doi.org/10.3390/60300203
Chicago/Turabian StyleChen, Huan-Ming, and Ramachandra S. Hosmane. 2001. "Synthesis of 1-(2'-O-Methyl-ß-D-ribofuranosyl)-1H-imidazo[4,5-d]pyridazine-4,7(5H,6H)-dione: An Attractive Building Block for Antisense and Triple-helical Applications" Molecules 6, no. 3: 203-207. https://doi.org/10.3390/60300203
APA StyleChen, H.-M., & Hosmane, R. S. (2001). Synthesis of 1-(2'-O-Methyl-ß-D-ribofuranosyl)-1H-imidazo[4,5-d]pyridazine-4,7(5H,6H)-dione: An Attractive Building Block for Antisense and Triple-helical Applications. Molecules, 6(3), 203-207. https://doi.org/10.3390/60300203



