N-(9-Fluorenylmethoxycarbonyl)-L-serine Amide
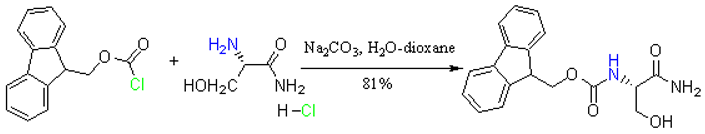
References
- Kocienski, P. Protecting Groups; Georg Thieme Verlag: Stuttgart, 1994; p. 204. [Google Scholar]
Sample availability: available from the authors and MDPI (MDPI ID 18866). |
© 2000 by the authors. All rights reserved. Molecules website www.mdpi.org/molecules/.
Share and Cite
Schmél, Z.; Kupihár, Z. N-(9-Fluorenylmethoxycarbonyl)-L-serine Amide. Molecules 2000, 5, M162. https://doi.org/10.3390/M162
Schmél Z, Kupihár Z. N-(9-Fluorenylmethoxycarbonyl)-L-serine Amide. Molecules. 2000; 5(7):M162. https://doi.org/10.3390/M162
Chicago/Turabian StyleSchmél, Zoltán, and Zoltán Kupihár. 2000. "N-(9-Fluorenylmethoxycarbonyl)-L-serine Amide" Molecules 5, no. 7: M162. https://doi.org/10.3390/M162
APA StyleSchmél, Z., & Kupihár, Z. (2000). N-(9-Fluorenylmethoxycarbonyl)-L-serine Amide. Molecules, 5(7), M162. https://doi.org/10.3390/M162



