Utility of Sulphones in Heterocyclic Synthesis: Synthesis of Some Pyridine, Chromene and Thiophene Derivatives
Abstract
:Introduction
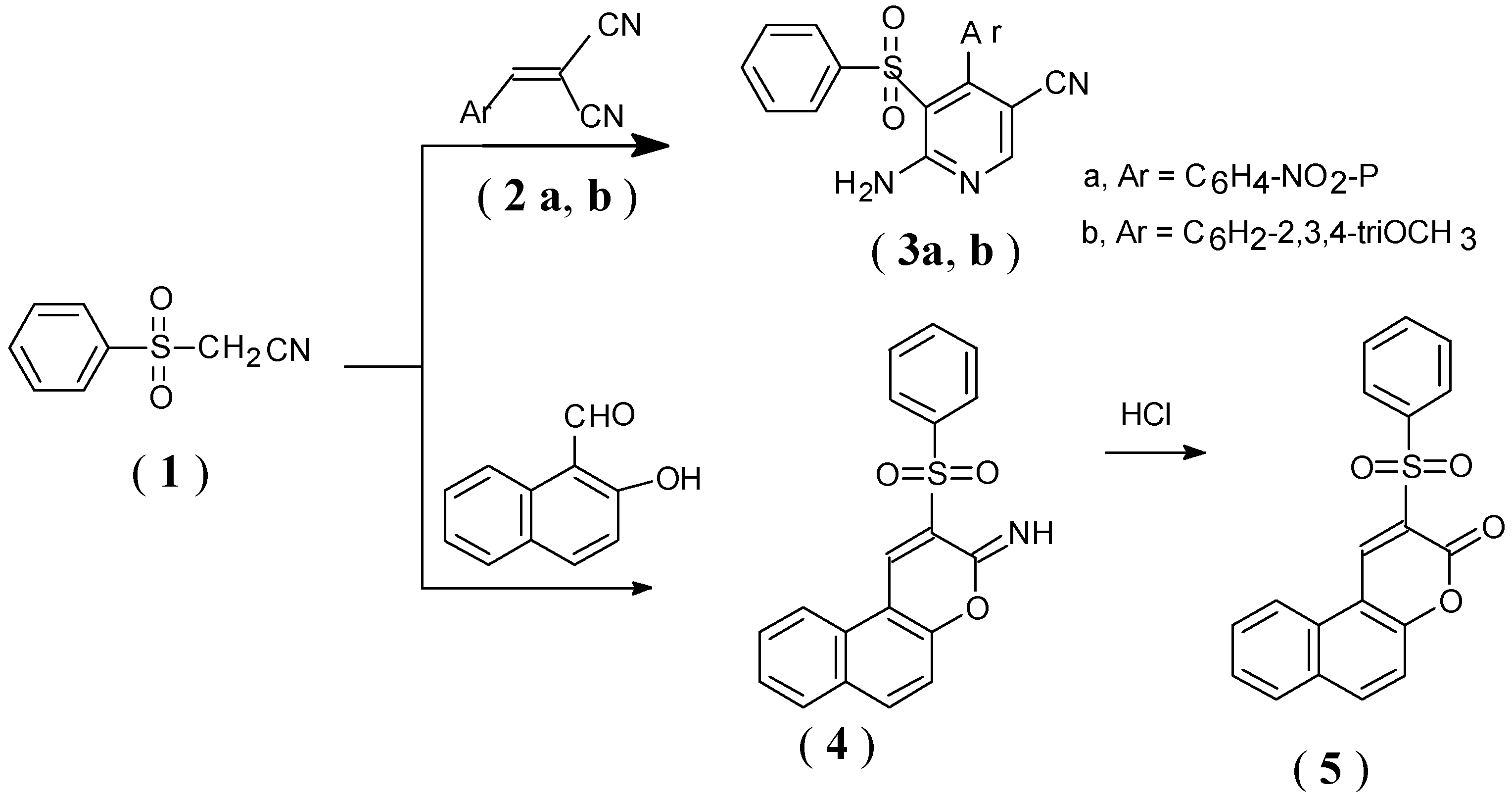
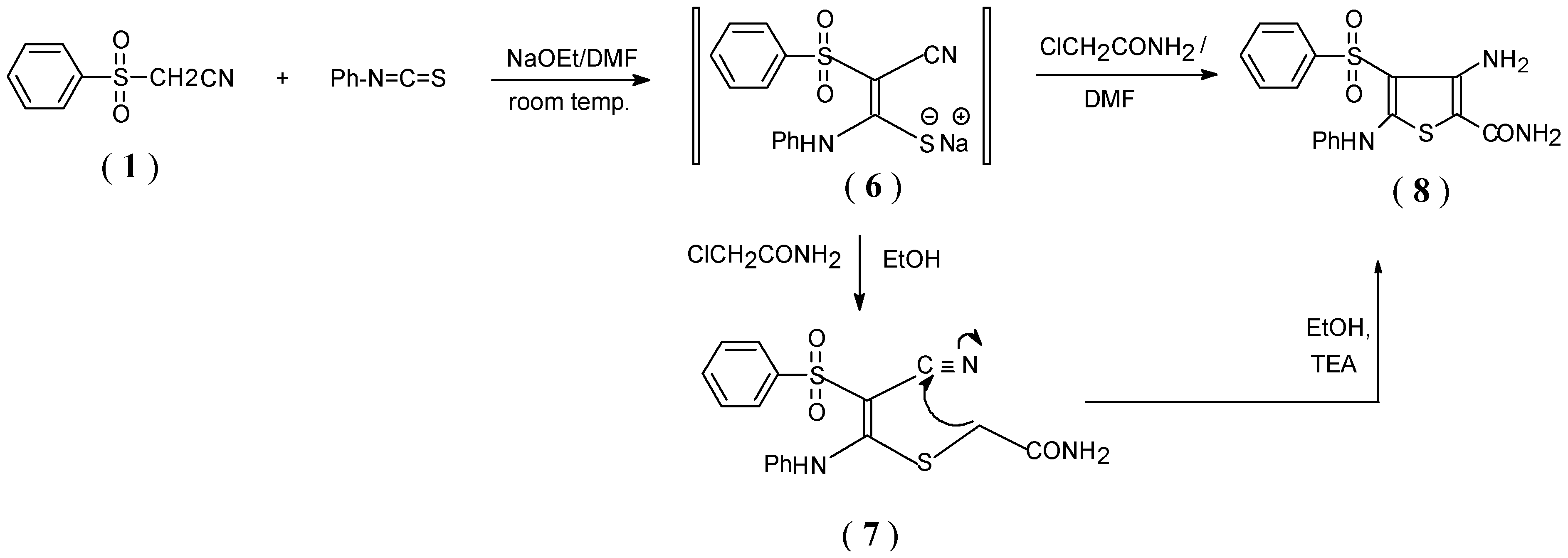
Results and Discussion

Conclusion
Experimental
General
Synthesis of pyridine derivatives (3a,b). General procedure
Synthesis of 2- imino-3-phenylsulfonyl-2H-benzo[ f ]chromene (4)
Synthesis of 3-phenylsulfonyl-2H-benzo[ f ]-2-chromenone (5)
Preparation of compound (6)
Synthesis of the acyclic intermediates 7, 11, 13 and 15. General procedure
Synthesis of thiophene derivatives (10)
Synthesis of thiophene derivatives ( 8, 12, 14 and 16 )
Method A
Method B
References and Notes
- Kaliszan, R.; Milczarska, B.; Lega, B. J. Pharmacol. Pharm. 1978, 30(4), 585, (C.A. 1979, 90, 197398y).
- Allen, M. P.; Gottlieb, D. Appl. Microbiol. 1970, 20(6), 919.
- Fadda, A.A.; Refat, Hala M.; Biehl (Eds.) J. Org. Chem. 1995, 60, 1985.
- Fadda, A.A.; Refat, Hala M.; Biehl (Eds.) Heterocycles 1995, 41, 289.
- Balasubramanian; et al. J. Chem. Soc. 1955, 3296. Hussien, A.H.; Elghandour, A.H.H.; Ramiz, M.M.M.; Elnagdi, M.H. Synthesis 1989, 775. Dressler, H.; Graham, J. E. J. Org. Chem. 1967, 32(4), 985. [CrossRef]
- Fadda, A.A.; Refat, Hala M.; El-Zemaity, M. T.; Biehl (Eds.) Heterocycles 1996, 43, 23.
- Fadda, A. A.; Amer, F. A.; Zaki, M. E. A. Phosphorus, Sulfur and Silicon 1999. accepted.
- Fadda, A.A.; Refat, Hala M.; Zaki, M. E. A. Synthetic Communications 1999. accepted.
- Fadda, A. A.; Abdelrazek, F. M. Z. Naturforsch. 1986, 41b, 499.
- Fadda, A.A.; Refat, Hala M. Synthetic Communications 1999. accepted.
- Fuentes, L.; Vaquero, J.; Ardid, M. I.; Castillo, J. Del; Soto, L. Jose. Synthesis 1999, 9, 768.
- Rao, R. B.; Singh, S. R. J. Indian Chem. Soc. 1973, 50, 492.
- Bhargava, P. N.; Sharma, S. C. Bull. Chem. Soc., Jpn. 1962, 35, 1926.
- Mallick, S. K.; Martin, A. R.; Lingard, R. G. J. Med. Chem. 1971, 14, 528.
- Singh, S. R. J. Indian Chem. Soc. 1975, 52, 734.
- Hantzsch, A.; Weber, H. J. Ber. Dtsch. Chem. Ges. 1887, 20, 3118. [CrossRef]
- Mehta, M. R.; Teivedi, J. P. Indian J. Chem. Sect. B 1990, 29(12), 1146.
- Samples Availability: available from the authors.
| No. | mp° C | Yld% | Mol. formula | Analysis Calcd. ( Found %) | Characterization | ||
|---|---|---|---|---|---|---|---|
| C | H | N | |||||
| 3a | 180 | 78 | C19H12N4O4S | 56.71 | 3.14 | 14.71 | IR: 3350-3450 (NH2), 2220 (CN), |
| (380.38) | (56.84) | (3.15) | (14.73) | 1610cm-1 (C=N); 1 H-NMR (CDCl3) δ | |||
| 6.5 (s, 2H, NH2 exchangeable with | |||||||
| D2O,), 7.8-8.4 (m, 9H, Ar-H), 8.5 (s, | |||||||
| 1H, o-Py-H); MS: (m/z) 380 M+. | |||||||
| 3b | 125 | 83 | C21H19N3O5S | 59.10 | 4.43 | 9.87 | IR: 3350-3450 (NH2), 2220 (CN), |
| (425.47) | (59.29) | (4.47) | (9.88) | 1610cm-1 (C=N); 1 H-NMR (CDCl3) δ | |||
| 3.8 (s, 3H, OCH3), 3.9 (s, 3H, OCH3), | |||||||
| 4.0 (s, 3H, OCH3), 7.1 (d, 2H, NH2, | |||||||
| exchangeable with D2O), 7.7-8.0 (m, | |||||||
| 6H, Ar-H), 8.5 (s, 1H, o-Py-H); MS: | |||||||
| (m/z) 425 M+ . | |||||||
| 4 | 225 | 90 | C19H13NO3S | 68.00 | 3.84 | 4.15 | IR: 3150 (NH), 1650 (C=N), 1561cm- |
| (335.38) | (68.05) | (3.88) | (4.17) | 1 (Ph); 1 H-NMR (CDCl3) δ 7.3-8.5 | |||
| (m, 11H, Ar-H), 8.8 (s, 1H, NH ex- | |||||||
| changeable with D2O), 9.2 (s, 1H, C4- | |||||||
| H); MS: (m/z) 335 M+ . | |||||||
| 5 | 267 | 98 | C19H12O4S | 67.69 | 3.56 | IR: 1700 (γ-Lactone), 1555cm-1 (Ph); | |
| (336.37) | (67.85) | (3.57) | 1 H-NMR (CDCl3) δ 7.55-8.25 (m, | ||||
| 9H, Ar-H), 8.4 (d, J=8.1 Hz, 1H, C9- | |||||||
| H), 8.45 (d, J=8.1 Hz,1H, C10-H), 9.2 | |||||||
| (s, 1H, C4-H); MS: (m/z) 336 M+ . | |||||||
| 7 | 219 | 85 | C17H15N3O3S2 | 54.55 | 4.00 | 11.24 | IR: 3340 (NH2), 3150(NH), 2220 |
| (373.46) | (54.69) | (4.02) | (11.26) | (CN), 1680cm-1 (C=O, amidic); 1H- | |||
| NMR (CDCl3) δ 3.7(s, 2H, CH2), 6.2 | |||||||
| (br, 2H, CONH2 exchangeable with | |||||||
| D2O), 7.2-7.98 (m, 10H, Ar-H), 9.25 | |||||||
| (s, 1H, NH exchangeable with D2O); | |||||||
| MS: (m/z) 373 M+ . | |||||||
| 8 | 145 | 62 | C17H15N3O3S2 | 45.51 | 4.01 | 11.23 | IR: 3450 (NH2), 3150(NH), 1661 |
| (373.46) | (45.50) | (4.02) | (11.26) | (CO), 1550 (Ph), 1270, 1180cm-1 | |||
| (SO2); 1 H-NMR (CDCl3) δ 5.5 (br, | |||||||
| 2H, NH2 exchangeable with D2O), 6.2 | |||||||
| (br, 2H, CONH2 exchangeable with | |||||||
| D2O), 7.4-8.0 (m, 10H, Ar-H), 8.6 (s, | |||||||
| 1H, NH exchangeable with D2O); | |||||||
| MS: (m/z) 373 M+ | |||||||
| 10 | 154 | 88 | C23H18N2O3S2 | 63.50 | 4.12 | 6.43 | IR: 3463 (NH2), 3194(NH), 1720 |
| (434.54) | (63.59) | (4.14) | (6.45) | (CO), 1587cm-1 (Ph); 1 H-NMR | |||
| (CDCl3) δ 6.5 (br, 2H, NH2 ex- | |||||||
| changeable with D2O), 7.3-8.1 (m, | |||||||
| 15H, Ar-H), 9.22 (s, 1H, NH ex- | |||||||
| changeable with D2O); MS: (m/z) | |||||||
| 434 M+ . | |||||||
| 11 | 190 | 94 | C18H16N2O3S2 | 58.00 | 4.29 | 7.50 | IR: 3287(NH), 2220 (CN), 1730 |
| (372.47) | (58.06) | (4.30) | (7.52) | (CO), 1603 cm-1 (Ph); 1 H-NMR | |||
| (CDCl3) δ 2.3 (s, 3H, CH3), 3.7 (s, | |||||||
| 2H, CH2), 7.16-7.97 (m, 10H, Ar-H), | |||||||
| 9.25 (s, 1H, NH exchangeable with | |||||||
| D2O); MS: (m/z) 372 M+ . | |||||||
| 12 | 165 | 88 | C18H16N2O3S2 | 58.10 | 4.28 | 7.51 | IR: 3456 (NH2), 3288 (NH), 1730 |
| (372.47) | (58.06) | (4.30) | (7.52) | (CO), 1603cm-1 (Ph); 1 H-NMR | |||
| (CDCl3) δ 2.14 (s, 3H, CH3), 5.3 (br, | |||||||
| 2H, NH2 exchangeable with D2O), | |||||||
| 7.26-7.96 (m, 10H, Ar-H), 9.25 (s, | |||||||
| 1H, NH exchangeable with D2O); | |||||||
| MS: (m/z) 372 M+ | |||||||
| 13 | 188 | 80 | C17H13N3O2S2 | 57.33 | 3.65 | 11.82 | IR: 3200 (NH), 2220, 2195 (two CN), |
| (355.44) | (57.46) | (3.66) | (11.83) | 1603 cm-1 (Ph); 1 H-NMR (CDCl3) δ | |||
| 3.23 (s, 2H, CH2), 7.12-7.87 (m, 10H, | |||||||
| Ar-H), 9.25 (s, 1H, NH exchangeable | |||||||
| with D2O); MS: (m/z) 355M+ . | |||||||
| 14 | 158 | 66 | C17H13N3O2S2 | 57.29 | 3.63 | 11.81 | IR: 3456 (NH2), 3266 (NH), 2220 |
| (355.44) | (57.46) | (3.66) | (11.83) | (CN), 1603cm-1 (Ph); 1 H-NMR | |||
| (CDCl3) δ 6.5 (br, 2H, NH2 ex- | |||||||
| changeable with D2O), 7.31-7.96 (m, | |||||||
| 10H, Ar-H), 8.7 (s, 1H, NH ex- | |||||||
| changeable with D2O); MS: (m/z) | |||||||
| 355M+ . | |||||||
| 15 | 248 | 93 | C19H18N2O4S2 | 56.66 | 4.46 | 6.95 | IR: 3278(NH), 2220 (CN), 1730 cm-1 |
| (402.49) | (56.71) | (4.47) | (6.96) | (CO); 1 H-NMR (CDCl3) δ 1.3 (t, | |||
| J=2.5 Hz, 3H, CH3), 3.7 (s, 2H, CH2), | |||||||
| 4.3 (q, J=2.5Hz, 2H, CH2), 7.5-8.56 | |||||||
| (m, 10H, Ar-H), 9.25 (s, 1H, NH ex- | |||||||
| changeable with D2O); MS: (m/z) | |||||||
| 402 M+ . | |||||||
| 16 | 152 | 86 | C19H18N2O4S2 | 56.70 | 4.45 | 6.94 | IR: 3448 (NH2), 3243(NH), 1730 |
| (402.50) | (56.71) | (4.47) | (6.96) | (CO), 1603cm-1 (Ph); 1 H-NMR | |||
| (CDCl3) δ 1.26 (t, J= 2.5 Hz, 3H, | |||||||
| CH3), 4.22 (q, J=2.5Hz, 2H, CH2), | |||||||
| 6.55 (br, 2H, NH2 exchangeable with | |||||||
| D2O), 7.18-7.96 (m, 10H, Ar-H), 9.25 | |||||||
| (s, 1H, NH exchangeable with D2O); | |||||||
| MS: (m/z) 402 M+ . | |||||||
© 2000 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Fadda, A.A.; Refat, H.M.; Zaki, M.E.A. Utility of Sulphones in Heterocyclic Synthesis: Synthesis of Some Pyridine, Chromene and Thiophene Derivatives. Molecules 2000, 5, 701-709. https://doi.org/10.3390/50500701
Fadda AA, Refat HM, Zaki MEA. Utility of Sulphones in Heterocyclic Synthesis: Synthesis of Some Pyridine, Chromene and Thiophene Derivatives. Molecules. 2000; 5(5):701-709. https://doi.org/10.3390/50500701
Chicago/Turabian StyleFadda, A. A., Hala M. Refat, and M. E. A. Zaki. 2000. "Utility of Sulphones in Heterocyclic Synthesis: Synthesis of Some Pyridine, Chromene and Thiophene Derivatives" Molecules 5, no. 5: 701-709. https://doi.org/10.3390/50500701




