The Importance of Keto-Enol Forms of Arylpropanoids Acting as Antifungal Compounds
Abstract
:Introduction
- 1-
- An exhaustive conformational and electronic study of this series using different levels of theory.
- 2-
- A correlation study between antifungal activity and computed parameters (ΔE of enolization and net atomic charges).
- 3-
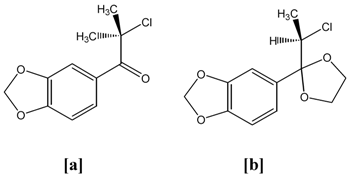
- Synthesis and evaluation of antifungal activity of compounds [a] and [b] to corroborate the results obtained on steps 1 and 2.
Experimental
Chemistry
Calculation Methods
Results and Discussion
Acknowledgements
References and Notes
- Zacchino, S.; et al. J. Nat. Prod. 1997, 60, 659.
- Zacchino, S.; et al. J. of Ethnopharmacol. 1998, 62, 35.
- S. Zacchino. J. Nat. Prod. (in press).
- Rodríguez, A. M.; et al. J. Mol. Struct. (Theochem) 1999, 463, 283.
Share and Cite
Giannini, F.; Devia, C.; Rodríguez, A.; Enriz, R.; Suvire, F.; Baldoni, H.; Furlan, R.; Zacchino, S. The Importance of Keto-Enol Forms of Arylpropanoids Acting as Antifungal Compounds. Molecules 2000, 5, 580-582. https://doi.org/10.3390/50300580
Giannini F, Devia C, Rodríguez A, Enriz R, Suvire F, Baldoni H, Furlan R, Zacchino S. The Importance of Keto-Enol Forms of Arylpropanoids Acting as Antifungal Compounds. Molecules. 2000; 5(3):580-582. https://doi.org/10.3390/50300580
Chicago/Turabian StyleGiannini, F., C. Devia, A. Rodríguez, R. Enriz, F. Suvire, H. Baldoni, R. Furlan, and S. Zacchino. 2000. "The Importance of Keto-Enol Forms of Arylpropanoids Acting as Antifungal Compounds" Molecules 5, no. 3: 580-582. https://doi.org/10.3390/50300580
APA StyleGiannini, F., Devia, C., Rodríguez, A., Enriz, R., Suvire, F., Baldoni, H., Furlan, R., & Zacchino, S. (2000). The Importance of Keto-Enol Forms of Arylpropanoids Acting as Antifungal Compounds. Molecules, 5(3), 580-582. https://doi.org/10.3390/50300580


