Study of the Cytotoxic and Antifungal Activities of Neolignans 8.O.4´ and Structurally Related Compounds
Abstract
:Introduction
Experimental
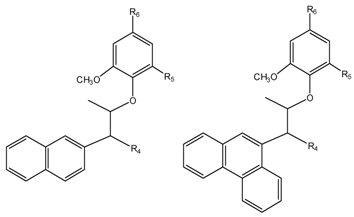
Results and Discussion
Acknowledgements
References and Notes
- J. Nat. Prod. 1997, 60, 659.
- J. of Ethnopharmacology 1998, 62, 35.
- To be published in J. Nat. Prod.
- J. Mol. Struct. (Theochem) 1999, 463, 283.
Share and Cite
Enriz, R.D.; Giannini, F.; Correche, E.; Carrasco, M.; Zacchino, S.; Matyus, P. Study of the Cytotoxic and Antifungal Activities of Neolignans 8.O.4´ and Structurally Related Compounds. Molecules 2000, 5, 576-577. https://doi.org/10.3390/50300576
Enriz RD, Giannini F, Correche E, Carrasco M, Zacchino S, Matyus P. Study of the Cytotoxic and Antifungal Activities of Neolignans 8.O.4´ and Structurally Related Compounds. Molecules. 2000; 5(3):576-577. https://doi.org/10.3390/50300576
Chicago/Turabian StyleEnriz, R. D., F. Giannini, E. Correche, M. Carrasco, S. Zacchino, and P. Matyus. 2000. "Study of the Cytotoxic and Antifungal Activities of Neolignans 8.O.4´ and Structurally Related Compounds" Molecules 5, no. 3: 576-577. https://doi.org/10.3390/50300576
APA StyleEnriz, R. D., Giannini, F., Correche, E., Carrasco, M., Zacchino, S., & Matyus, P. (2000). Study of the Cytotoxic and Antifungal Activities of Neolignans 8.O.4´ and Structurally Related Compounds. Molecules, 5(3), 576-577. https://doi.org/10.3390/50300576


