Insights into Antioxidant Activity and Trace Element Distribution of Aqueous Extract of Silybum marianum Seeds
Abstract
1. Introduction
2. Results and Discussions
2.1. Analysis of Trace Elements in Decoction of SM Seeds
2.1.1. Separation Performance of Silymarin Flavonoids
2.1.2. Element Concentrations in Fractionated Eluents
2.2. Analysis of Antioxidant Activity of Aqueous Extract from SM Seeds
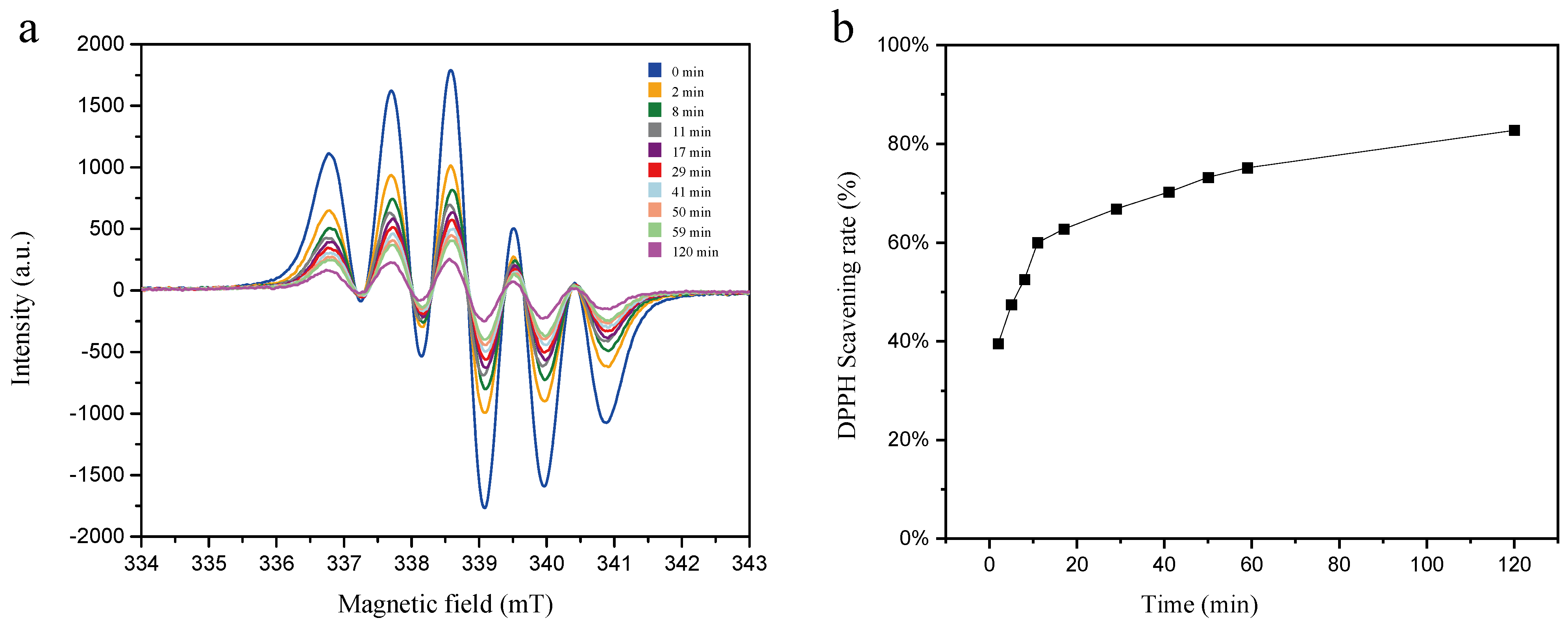
2.2.1. DPPH Radical Scavenging Ability of the SM Extract
2.2.2. •OH Radical Scavenging Ability of the SM Extract
3. Materials and Methods
3.1. Materials and Reagents
3.2. Preparation of Boiling Water Decoction and Extract of SM Seeds
3.3. Apparatus
3.4. Determination of Inorganic Elements in Fractionated Eluents of Boiling Water Decoction of SM Seeds (SM Decoction)
3.4.1. RP-HPLC Separation of Silymarin Flavonoids
3.4.2. ICP-MS Analysis of Elements in the HPLC Fractionated Eluents
3.5. Measurement of Antioxidant Activity of Boiling Water Extract from SM Seeds (SM Extract)
3.5.1. Time-Dependence of DPPH Radical Scavenging Activity by SM Extracts
3.5.2. Dependence of DPPH Radical Scavenging Activity on SM Extract Concentration
3.5.3. Time-Dependence of •OH Radical Scavenging Activity by SM Extract
3.5.4. Dependence of •OH Radical Scavenging Activity on SM Extract Concentration
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| SM | Silybum marianum |
| TXF | Taxifolin |
| SCN | Silycristin |
| SDN | Silydianin |
| SBNA | Silybin A |
| SBNB | Silybin B |
| ISBNA | Isosilybin A |
| ISBNB | Isosilybin B |
| DPPH | 2,2-Diphenyl-1-picrylhydrazyl |
| DMPO | 5,5-Dimethyl-1-pyrroline N-oxide |
| TCM | Traditional Chinese medicine |
| RP-HPLC | Reversed-phase high-performance liquid chromatography |
| EPR | Electron paramagnetic resonance spectroscopy |
| ICP-MS | Inductively coupled plasma mass spectrometry (ICP-MS) |
| ICP-AES | Inductively coupled plasma atomic emission spectrometry |
| HPLC | High-performance liquid chromatography |
| LC-MS | Liquid chromatography–mass spectrometry |
References
- Flora, K.; Hahn, M.; Rosen, H.; Benner, K. Milk thistle (Silybum marianum) for the therapy of liver disease. Am. J. Gastroenterol. 1998, 93, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Abenavoli, L.; Capasso, R.; Milic, N.; Capasso, F. Milk thistle in liver diseases: Past, present, future. Phytother. Res. 2010, 24, 1423–1432. [Google Scholar] [CrossRef]
- Lee, D.Y.W.; Liu, Y. Molecular structure and stereochemistry of silybin A, silybin B, isosilybin A, and isosilybin B, isolated from Silybum marianum (milk thistle). J. Nat. Prod. 2003, 66, 1171–1174. [Google Scholar] [CrossRef]
- Fehér, J.; Lengyel, G. Silymarin in the prevention and treatment of liver diseases and primary liver cancer. Curr. Pharm. Biotechnol. 2012, 13, 210–217. [Google Scholar] [CrossRef]
- Ahmad, N.; Abbasi, B.H.; Fazal, H. Evaluation of antioxidant activity and its association with plant development in Silybum marianum L. Ind. Crop. Prod. 2013, 49, 164–168. [Google Scholar] [CrossRef]
- Köksal, E.; Gülçin, I.; Beyza, S.; Sarikaya, Ö.; Bursal, E. In vitro antioxidant activity of silymarin. J. Enzym. Inhib. Med. Chem. 2009, 24, 395–405. [Google Scholar] [CrossRef] [PubMed]
- Akkaya, H.; Yilmaz, O. Antioxidant capacity and radical scavenging activity of Silybum marianum and Ceratonia siliqua. Ekoloji 2012, 21, 9–16. [Google Scholar] [CrossRef]
- Javeed, A.; Ahmed, M.; Sajid, A.R.; Sikandar, A.; Aslam, M.; Hassan, T.U.; Samiullah; Nazir, Z.; Ji, M.; Li, C. Comparative assessment of phytoconstituents, antioxidant activity and chemical analysis of different parts of milk thistle Silybum marianum L. Molecules 2022, 27, 2641. [Google Scholar] [CrossRef]
- Iqbal, T.; Aslam, T.; Zulfiqar, S.; Faisal, N.; Rehman, A.U.; Aslam, S.; Rehman, R. Exploring Silybum marianum L. seeds from Pakistan for its antibacterial, antioxidant, antidiabetic activities, and phytochemical analysis. Nat. Prod. Res. 2025. Online. [Google Scholar] [CrossRef]
- Bhattacharya, S. Phytotherapeutic properties of milk thistle seeds: An overview. J. Adv. Pharm. Educ. Res. 2011, 1, 6–79. [Google Scholar] [CrossRef]
- Kazazis, C.E.; Evangelopoulos, A.A.; Kollas, A.; Vallianou, N.G. The therapeutic potential of milk thistle in diabetes. Rev. Diabet. Stud. 2014, 11, 167–174. [Google Scholar] [CrossRef]
- Qin, N.B.; Jia, C.C.; Xu, J.; Li, D.H.; Xu, F.X.; Bai, J.; Li, Z.L.; Hua, H.M. New amides from seeds of Silybum marianum with potential antioxidant and antidiabetic activities. Fitoterapia 2017, 119, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Azadpour, M.; Farajollahi, M.M.; Varzi, A.M.; Hashemzadeh, P.; Mahmoudvand, H.; Barati, M. Extraction, chemical composition, antioxidant property, and in-vitro anticancer activity of Silymarin from Silybum marianum on KB and A549 cell lines. Curr. Drug Discov. Technol. 2021, 18, 511–517. [Google Scholar] [CrossRef]
- Kim, J.; Han, S.H.; Kim, N.K.; Tran, G.H.; Shim, J.; Chin, J.H.; Kim, J.H.; Lee, S. Antioxidant activities and silymarin content of Silybum marianum using different extraction methods. J. Appl. Biol. Chem. 2024, 67, 397–406. [Google Scholar] [CrossRef]
- Aldayel, M.F. Potential antibacterial and antioxidant inhibitory activities of Silybum marianum mediated biosynthesised He-Ne laser. Saudi J. Biol. Sci. 2023, 30, 103795. [Google Scholar] [CrossRef]
- Lekmine, S.; Benslama, O.; Ola, M.S.; Touzout, N.; Moussa, H.; Tahraoui, H.; Hafsa, H.; Zhang, J.; Amrane, A. Preliminary data on Silybum marianum metabolites: Comprehensive characterization, antioxidant, antidiabetic, antimicrobial activities, LC-MS/MS profiling, and predicted ADMET analysis. Metabolites 2025, 15, 13. [Google Scholar] [CrossRef]
- Viktorova, J.; Stranska-Zachariasova, M.; Fenclova, M.; Vitek, L.; Hajslova, J.; Kren, V.; Ruml, T. Complex evaluation of antioxidant capacity of milk thistle dietary supplements. Antioxidants 2019, 8, 317. [Google Scholar] [CrossRef]
- Serce, A.; Toptanci, B.C.; Tanrikut, S.E.; Altas, S.; Kizil, G.; Kizil, S.; Kizil, M. Assessment of the antioxidant activity of Silybum marianum seed extract and its protective effect against DNA oxidation, protein damage and lipid peroxidation. Food Technol. Biotech. 2016, 54, 455–461. [Google Scholar] [CrossRef]
- Wallace, S.N.; Carrier, D.J.; Clausen, E.C. Batch solvent extraction of flavonolignans from milk thistle (Silybum marianum L. Gaertner). Phytochem. Anal. 2005, 16, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, N.; Itoh, N.; Yamasaki, K. Absolute quantification of pure free radical reagents by combination of the effective magnetic moment method and quantitative electron paramagnetic resonance method. Anal. Chem. 2022, 94, 12595–12603. [Google Scholar] [CrossRef] [PubMed]
- Sahu, I.D.; Lorigan, G.A. Electron paramagnetic resonance as a tool for studying membrane proteins. Biomolecules 2020, 10, 763. [Google Scholar] [CrossRef] [PubMed]
- Morales, N.P.; Sirijaroonwong, S.; Yamanont, P.; Phisalaphong, C. Electron paramagnetic resonance study of the free radical scavenging capacity of curcumin and its demethoxy and hydrogenated derivatives. Biol. Pharma. Bull. 2015, 38, 1478–1483. [Google Scholar] [CrossRef] [PubMed]
- Barriga-González, G.; Aguilera-Venegas, B.; Folch-Cano, C.; Pérez-Cruz, F.; Olea-Azar, C. Electron spin resonance as a powerful tool for studying antioxidants and radicals. Curr. Med. Chem. 2013, 20, 4731–4743. [Google Scholar] [CrossRef]
- Zhao, Y.; Chen, B.; Yao, S. Simultaneous determination of abietane-type diterpenes, flavonolignans and phenolic compounds in compound preparations of Silybum marianum and Salvia miltiorrhiza by HPLC-DAD-ESI MS. J. Pharm. Biomed. 2005, 38, 564–570. [Google Scholar] [CrossRef]
- Cai, X.L.; Li, D.N.; Qiao, J.Q.; Lian, H.Z.; Wang, S.K. Determination of silymarin flavonoids by HPLC and LC-MS and investigation of extraction rate of silymarin in Silybum marianum fruits by boiling water. Asian J. Chem. 2009, 21, 63–74. [Google Scholar]
- He, W.M.; Xu, M.D.; Yang, Q.; Zhang, S.J.; Wen, X.L.; Mao, G.N. Nutritional compositions of Silybum marianum Gaertn seed oil and its hypolipidemic effect in rats. Acta Nutr. Sin. 1996, 18, 163–167. [Google Scholar]
- Tzen, J.T.C.; Jinn, T.R.; Chen, Y.C.; Li, F.Y.; Cheng, F.C.; Shi, L.S.; She, K.H.; Chen, B.C.M.; Hsieh, V.; Tu, M.L. Magnesium lithospermate B possesses inhibitory activity on Na+,K+-ATPase and neuroprotective effects against ischemic stroke. Acta Pharmacol. Sin. 2007, 28, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Selvaraj, S.; Krishnaswamy, S.; Devashya, V.; Sethuraman, S.; Krishnan, U.M. Flavonoid–metal Ion complexes: A novel class of therapeutic agents. Med. Res. Rev. 2014, 34, 677–702. [Google Scholar] [CrossRef]
- Khater, M.; Ravishankar, D.; Greco, F.; Osborn, H.M. Metal complexes of flavonoids: Their synthesis, characterization and enhanced antioxidant and anticancer activities. Future Med. Chem. 2019, 11, 2845–2867. [Google Scholar] [CrossRef]
- Šimunková, M.; Biela, M.; Štekláč, M.; Hlinčík, A.; Klein, E.; Malček, M. Cu(II) complexes of flavonoids in solution: Impact of the Cu(II) ion on the antioxidant and DNA-intercalating properties. J. Mol. Liq. 2022, 359, 119230. [Google Scholar] [CrossRef]
- Halevas, E.; Mavroidi, B.; Zahariou, G.; Pelecanou, M.; Hatzidimitriou, A.G. Structurally characterized copper complexes of flavonoid naringenin with enhanced radical scavenging activity. Inorg. Chim. Acta 2023, 546, 121325. [Google Scholar] [CrossRef]
- Ikeda, N.E.A.; Novak, E.M.; Maria, D.A.; Velosa, A.S.; Pereira, R.M.S. Synthesis, characterization and biological evaluation of Rutin-zinc(II) flavonoid-metal complex. Chem.-Biol. Interact. 2015, 239, 184–191. [Google Scholar] [CrossRef]
- Chen, H.; Liang, P.; Hu, B.; Zhao, L.; Sun, D.H.; Wang, X.R. The application of inductively coupled plasma atomic emission spectrometry/mass spectrometry in the trace elements and speciation analysis of traditional Chinese medicine. Spectrosc. Spect. Anal. 2002, 22, 1019–1024. [Google Scholar]
- Mira, L.; Fernandez, M.T.; Santos, M.; Rocha, R.; Florêncio, M.H.; Jennings, K.R. Interactions of flavonoids with iron and copper ions: A mechanism for their antioxidant activity. Free Radic. Res. 2002, 36, 1199–1208. [Google Scholar] [CrossRef] [PubMed]
- Samsonowicz, M.; Regulska, E. Spectroscopic study of molecular structure, antioxidant activity and biological effects of metal hydroxyflavonol complexes. Spectrochim. Acta A 2017, 173, 757–771. [Google Scholar] [CrossRef]
- Perron, N.R.; Brumaghim, J.L. A review of the antioxidant mechanisms of polyphenol compounds related to iron binding. Cell Biochem. Biophys. 2009, 53, 75–100. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.L.; Chen, Y.J.; Dai, L.M.; Lian, H.Z. Study of inorganic elements in Silybum marianum: Distribution and extraction rate. Spectrosc. Spect. Anal. 2006, 26, 2125–2129. [Google Scholar]
- Yoshikawa, T.; You, F. Oxidative stress and bioregulation. Int. J. Mol. Sci. 2024, 25, 3360. [Google Scholar] [CrossRef]
- Sharebiani, H.; Mokaram, M.; Mirghani, M.; Fazeli, B.; Stanek, A. The effects of antioxidant supplementation on the pathologic mechanisms of metabolic syndrome and cardiovascular disease development. Nutrients 2024, 16, 1641. [Google Scholar] [CrossRef]
- Quan, L.; Zhou, E.C.; Guo, X.W.; Yin, G. Antioxidant activity of herbal medicine Pyrrosia lingua evaluated by electron paramagnetic resonance spectroscopy. Anal. Methods 2025, 17, 476–484. [Google Scholar] [CrossRef]




| Fraction Number | As | Cd | Co | Cr | Cu | Fe | Mn | Mo | Zn |
|---|---|---|---|---|---|---|---|---|---|
| First | 0.03 (0.02) | 0.10 (0.02) | 0.23 (0.00) | 2.71 (0.23) | 46.3 (0.38) | 16.1 (2.04) | 6.19 (0.26) | 1.49 (0.01) | 27.3 (1.56) |
| Second | ND | ND | 0.07 (0.00) | 0.74 (0.03) | 11.7 (0.66) | 15.4 (2.25) | 4.10 (0.53) | 0.18 (0.00) | 6.73 (0.92) |
| Third | 0.04 (0.01) | ND | ND | ND | 4.53 (0.64) | ND | 2.01 (0.18) | 0.09 (0.02) | ND |
| Fourth | ND | 0.21 (0.01) | ND | 2.54 (0.33) | 6.03 (0.56) | 15.8 (2.49) | 3.92 (0.19) | ND | 7.91 (1.06) |
| Fifth | ND | 0.35 (0.01) | 0.14 (0.01) | ND | 1.78 (0.24) | ND | 1.87 (0.05) | ND | 5.05 (0.98) |
| Sixth | ND | 0.06 (0.00) | 0.07 (0.00) | 0.08 (0.04) | 3.40 (0.24) | ND | 2.15 (0.16) | 0.17 (0.01) | ND |
| Seventh | ND | 0.08 (0.01) | ND | 0.06 (0.01) | 3.80 (0.11) | 5.80 (2.30) | 1.47 (0.08) | 0.21 (0.01) | ND |
| Eighth | 0.21 (0.00) | 0.06 (0.00) | ND | ND | 2.28 (0.06) | ND | 1.82 (0.13) | ND | 2.44 (0.17) |
| Ninth | ND | 0.12 (0.00) | ND | 0.22 (0.06) | 2.07 (0.19) | 10.5 (2.27) | 1.80 (0.25) | ND | ND |
| Total concentration in digestion solution of HPLC effluent (ng/mL) | 0.28 | 0.98 | 0.51 | 6.35 | 81.9 | 63.6 | 25.3 | 2.14 | 49.4 |
| Total amount in 200 μL decoction (ng) | 0.84 | 2.94 | 1.53 | 19.0 | 246 | 191 | 75.9 | 6.42 | 148 |
| Total concentration in 30 mL decoction (ng/mL) | 4.20 | 14.7 | 7.65 | 95.0 | 1230 | 955 | 380 | 32.1 | 740 |
| Fraction Number | Compound | Time Range (min) | Fraction Number | Compound | Time Range (min) |
|---|---|---|---|---|---|
| First | — | 0.0–2.7 | Sixth | SDN | 19.0–24.5 |
| Second | — | 2.7–4.7 | Seventh | — | 24.5–30.3 |
| Third | — | 4.7–7.0 | Eighth | SBN | 30.3–38.0 |
| Fourth | TXF | 7.0–15.0 | Ninth | ISBN | 38.0–45.0 |
| Fifth | SCN | 15.0–19.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Quan, L.; Wang, Y.-X.; Cai, X.-L.; Zhou, E.-C.; Guo, X.-W.; Chen, Y.-J.; Lian, H.-Z. Insights into Antioxidant Activity and Trace Element Distribution of Aqueous Extract of Silybum marianum Seeds. Molecules 2026, 31, 1034. https://doi.org/10.3390/molecules31061034
Quan L, Wang Y-X, Cai X-L, Zhou E-C, Guo X-W, Chen Y-J, Lian H-Z. Insights into Antioxidant Activity and Trace Element Distribution of Aqueous Extract of Silybum marianum Seeds. Molecules. 2026; 31(6):1034. https://doi.org/10.3390/molecules31061034
Chicago/Turabian StyleQuan, Li, Yi-Xiao Wang, Xiu-Lan Cai, En-Chao Zhou, Xue-Wen Guo, Yi-Jun Chen, and Hong-Zhen Lian. 2026. "Insights into Antioxidant Activity and Trace Element Distribution of Aqueous Extract of Silybum marianum Seeds" Molecules 31, no. 6: 1034. https://doi.org/10.3390/molecules31061034
APA StyleQuan, L., Wang, Y.-X., Cai, X.-L., Zhou, E.-C., Guo, X.-W., Chen, Y.-J., & Lian, H.-Z. (2026). Insights into Antioxidant Activity and Trace Element Distribution of Aqueous Extract of Silybum marianum Seeds. Molecules, 31(6), 1034. https://doi.org/10.3390/molecules31061034






