2.1. Characterization of the Adsorbent
The SEM images of TBC and CTAB@TBC are presented in
Figure 1. As shown in
Figure 1a–h, TBC exhibits a well-defined, orderly structure with discernible regular pore arrangements. In contrast, CTAB@TBC displays a looser and more porous morphology, characterized by increased structural complexity and irregular pore distribution. CTAB modification significantly enhances the biochar’s surface roughness, transforming the originally smooth surface into one covered with numerous fine granular particles. This observation confirms that CTAB has been effectively loaded onto the biochar surface [
9]. As the CTAB content increases, the surface roughness of TBC increases, and CTAB aggregation becomes more pronounced. The morphological transformation, marked by particle deposition and increased roughness, may create additional active or adsorption sites, thereby improving the material’s adsorption performance and related properties.
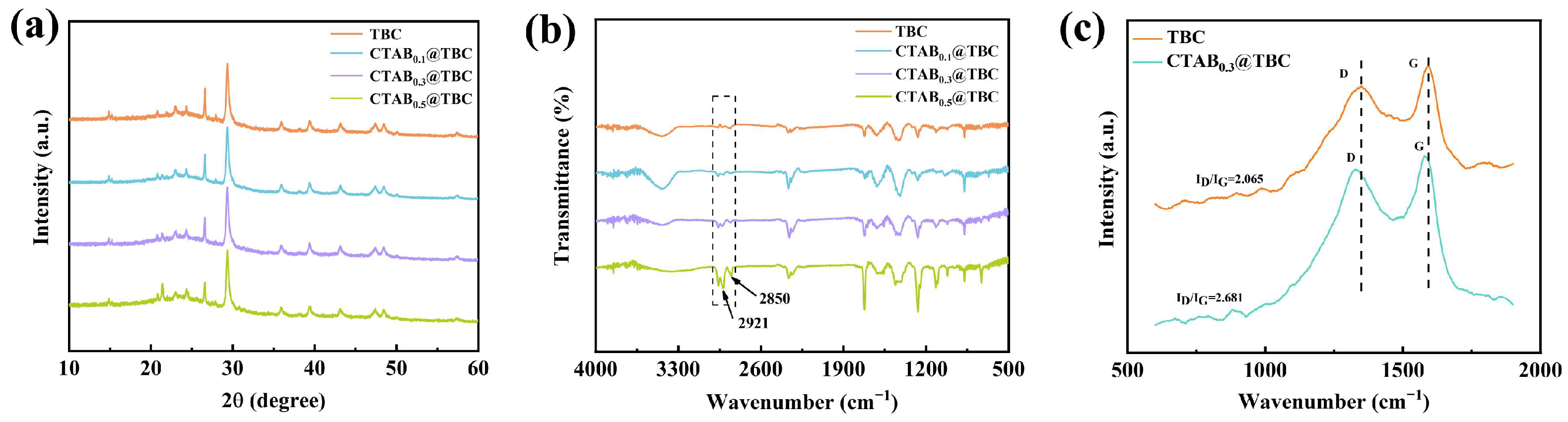
The XRD patterns of TBC and CTAB@TBC are illustrated in
Figure 2a. All samples exhibit nearly identical diffraction patterns, indicating that the CTAB surface modification does not affect the crystalline structure of TBC. The XRD data reveal diffraction peaks that suggest the presence of amorphous carbon in TBC. A broad peak observed at approximately 23.4° is attributed to amorphous carbon, while the weak diffraction peaks observed at 20.7°, 22.9°, and 43.2° correspond to the characteristic diffraction of the graphite-like carbon’s microcrystalline structure. The diffraction peaks at 29.3°, 35.9°, 39.3°, 43.1°, 47.3°, and 48.4° correspond to the standard characteristic peaks of calcite-type calcium carbonate (CaCO
3), confirming the presence of calcium carbonate in the sample [
5,
22].
Figure 2b shows the FTIR spectra of TBC and CTAB@TBC. Comparative analysis of their spectral features indicates that both samples exhibit distinct vibrational absorption bands. Notably, the FTIR spectrum of CTAB@TBC displays two additional characteristic peaks at 2850 cm
−1 and 2921 cm
−1, which are absent in the TBC spectrum. These peaks correspond to the symmetric and asymmetric stretching vibrations of methylene (–CH
2–) groups, respectively, arising from CTAB molecules immobilized on the TBC surface [
12,
14], the intensity of the –CH
2– stretching vibration peak increases significantly with increasing CTAB proportion. This observation provides clear spectroscopic evidence for the successful incorporation of CTAB into the TBC matrix.
Figure 2c displays the Raman spectra of TBC and CTAB
0.3@TBC, featuring characteristic D (1345 cm
−1) and G (1593 cm
−1) bands. The D band is linked to structural disorder and localized defects in graphite, originating from the vibrational modes of sp
3-hybridized carbon atoms. In contrast, the G band corresponds to the stretching vibrations of sp
2-hybridized carbon atoms within graphene’s two-dimensional hexagonal lattice [
30]. The intensity ratio of the D band to the G band (ID/IG) indicates the relative levels of structural defects and graphitic ordering in biochar. The ID/IG values for TBC and CTAB
0.3@TBC were 2.065 and 2.681, respectively, demonstrating increased structural disorder in CTAB
0.3@TBC that facilitates the adsorption process. CTAB
0.3@TBC exhibits the highest degree of structural disorder among the materials studied.
The TGA and DTG curves of TBC and CTAB
0.3@TBC are presented in
Figure 3a,b. For TBC, an initial mass loss of 5.94% at sub-10 °C temperatures is attributed to the vaporization of physically adsorbed moisture. A slow mass reduction of 12.81% between 100 °C and 630 °C is attributed to the decomposition of organic components such as hemicellulose, cellulose, and lignin remaining in the biochar. Above 630 °C, an 8.87% mass loss indicates the slow reformation and graphitization of highly stable aromatic carbon structures, as well as the decomposition of a small amount of inorganic carbonates [
31]. The thermal decomposition trends of CTAB
0.3@TBC and TBC are similar. Below 100 °C, a mass loss of 4.98% occurs due to the volatilization of physically adsorbed water. Between 100 °C and 650 °C, the mass loss is 16.74%, primarily caused by the thermal decomposition of CTAB and organic substances such as cellulose and lignin remaining in the biochar. Above 650 °C, a mass loss of approximately 9.80% suggests the gradual decomposition and structural reorganization of the highly stable aromatic carbon framework. Additionally, A characteristic peak at 411.5 °C in the DTG profile of CTAB
0.3@TBC is attributed to the thermal degradation of CTAB [
30].
Figure 3c,d present the N
2 adsorption–desorption isotherms and pore size distribution profiles of TBC and CTAB
0.3@TBC. Both TBC and CTAB
0.3@TBC exhibit mesoporous structures (2–50 nm) featuring slit-shaped or irregularly stacked pores, as evidenced by Type IV adsorption isotherms with H3 hysteresis loops. Quantitative analysis reveals that the specific surface area of the CTAB-modified TBC decreased from 57.39 m
2/g to 21.93 m
2/g, whereas the average pore diameter increased from 17.27 nm to 43.62 nm. This change is primarily attributed to the adsorption of CTAB, a long-chain cationic surfactant, onto the biochar surface through electrostatic interactions, hydrophobic effects, and van der Waals forces. During this process, CTAB molecules also enter certain micropores and mesopores, partially blocking the pore channels and thereby reducing the accessible surface area. CTAB modification led to an overall reduction in the N
2 adsorption capacity of CTAB
0.3@TBC, particularly in the high P/P
0 region. This phenomenon indicates a decrease in total pore volume, which is attributed to the partial pore blockage caused by the intrusion of CTAB molecules into the pore structure. These observations confirm the successful modification of the biochar by CTAB [
22].
2.2. Adsorption Experiment
2.2.1. Performance Screening of Different Adsorption Materials
The adsorption performance of the synthesized material CTAB@TBC toward XO was evaluated by comparative experiments, as shown in
Figure 4. Pristine TBC exhibited a removal efficiency of 50.9% and an adsorption capacity of 84.83 mg/g. As the CTAB content increased, both the removal efficiency and adsorption capacity of CTAB@TBC were enhanced. Specifically, the removal efficiency increased from 77.90% for CTAB
0.1@TBC to 90.07% for CTAB
0.3@TBC, while the adsorption capacity rose from 129.83 to 150.11 mg/g. However, further increases in CTAB content did not lead to additional improvement, and CTAB
0.5@TBC showed no significant advantage over CTAB
0.3@TBC. These results indicate that an appropriate CTAB loading can significantly enhance the adsorption performance of TBC. Therefore, CTAB
0.3@TBC was selected for subsequent in-depth studies.
2.2.2. Impact of Initial pH on Adsorption
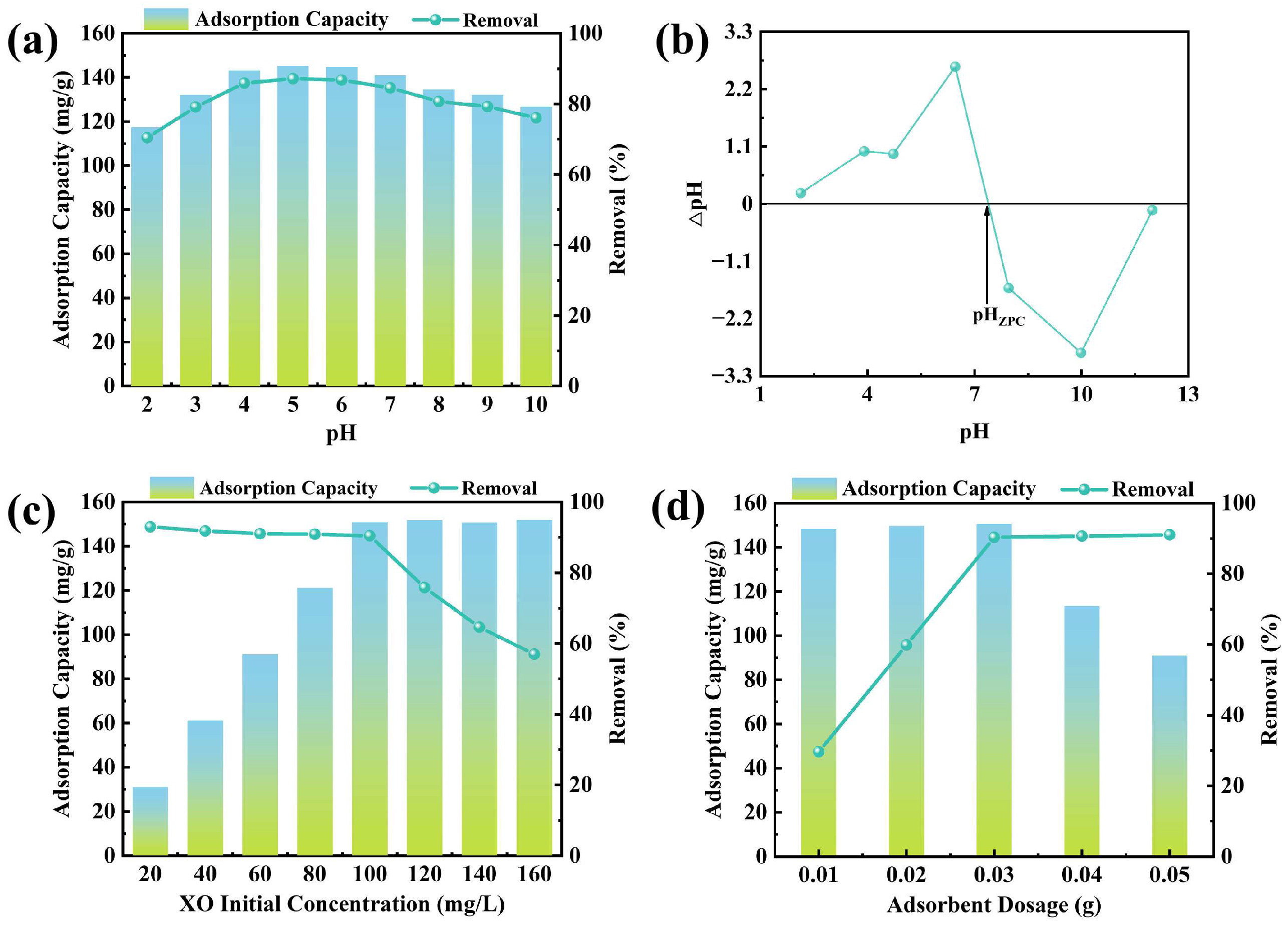
The impact of the solution’s initial pH on XO adsorption by CTAB@TBC was evaluated (
Figure 5a). Adsorption capacity and removal efficiency exhibited clear pH dependence: both increased across pH 2.0–5.0 and then declined progressively over the range 5.0–10.0. This phenomenon arises from the combined influence of the surface charge of CTAB@TBC and the pH-dependent dissociation behavior of XO. As the pH changes in aqueous solution, XO transitions among various ionic species (e.g., H
4XO
+, H
4XO, H
3XO
−, HXO
3−, XO
4−). These variations indicate that adsorption is governed not only by electrostatic interactions but also by potential chemical interactions. Overall, CTAB@TBC exhibits an effective pH range of 4.0 to 6.0 for XO adsorption. The adsorption mechanism can be clarified by examining the zero-point charge (ZPC) of CTAB@TBC, which was determined to be 7.39 (
Figure 5b). When the solution pH is lower than this value, the CTAB@TBC surface becomes positively charged, which favors the adsorption of anionic XO. Conversely, at pH values above pH
ZPC, the surface becomes negatively charged and the electrostatic attraction between the surface and XO is consequently weakened, leading to reduced adsorption [
1]. Based on the above analysis, the pH value of the adsorption solution was adjusted to 6.0 to ensure the best adsorption effect.
2.2.3. Impact of CTAB@TBC Dosage on Adsorption
The optimal CTAB@TBC dosage was determined by evaluating how different application levels influenced the adsorption capacity and removal efficiency of XO. As depicted in
Figure 5c, raising the CTAB@TBC dosage from 0.01 g to 0.03 g markedly improved the removal efficiency of XO, increasing it from 29.67% to 90.32%. Beyond 0.03 g, however, the efficiency remained essentially unchanged, indicating a saturation point. When the dosage was increased from 0.03 g to 0.05 g, the adsorption capacity decreased significantly, from 150.54 mg/g to 91.05 mg/g. This decrease can be attributed to the limited availability of dye molecules at higher adsorbent concentrations, which prevents the full utilization of adsorption sites [
22]. Therefore, when adsorption equilibrium is reached at the solid–liquid interface, the adsorption capacity per unit mass of the adsorbent decreases.
2.2.4. Impact of XO’s Initial Concentration on Adsorption
The initial concentration of XO within the reaction system plays a critical role in determining the adsorption performance of the synthesized adsorbent. As shown in
Figure 5d, varying the starting XO concentration markedly influences the adsorption behavior of CTAB@TBC. As the initial concentration of XO increased from 20 to 160 mg/L, the equilibrium adsorption capacity increased from 30.99 to 150.80 mg/g and then approached a plateau. This behavior is attributed to the progressive saturation of active sites on the adsorbent surface. At higher XO concentrations, the larger concentration gradient provides a stronger driving force for mass transfer, thereby accelerating the diffusion of XO molecules toward the adsorbent surface [
12,
19]. However, when the initial XO concentration exceeds 100 mg/L, the dye removal efficiency significantly decreases. Based on this trend, 100 mg/L can be identified as the optimal initial XO concentration for achieving maximum adsorption performance.
2.2.5. Impact of Co-Existing Ions on Adsorption Process
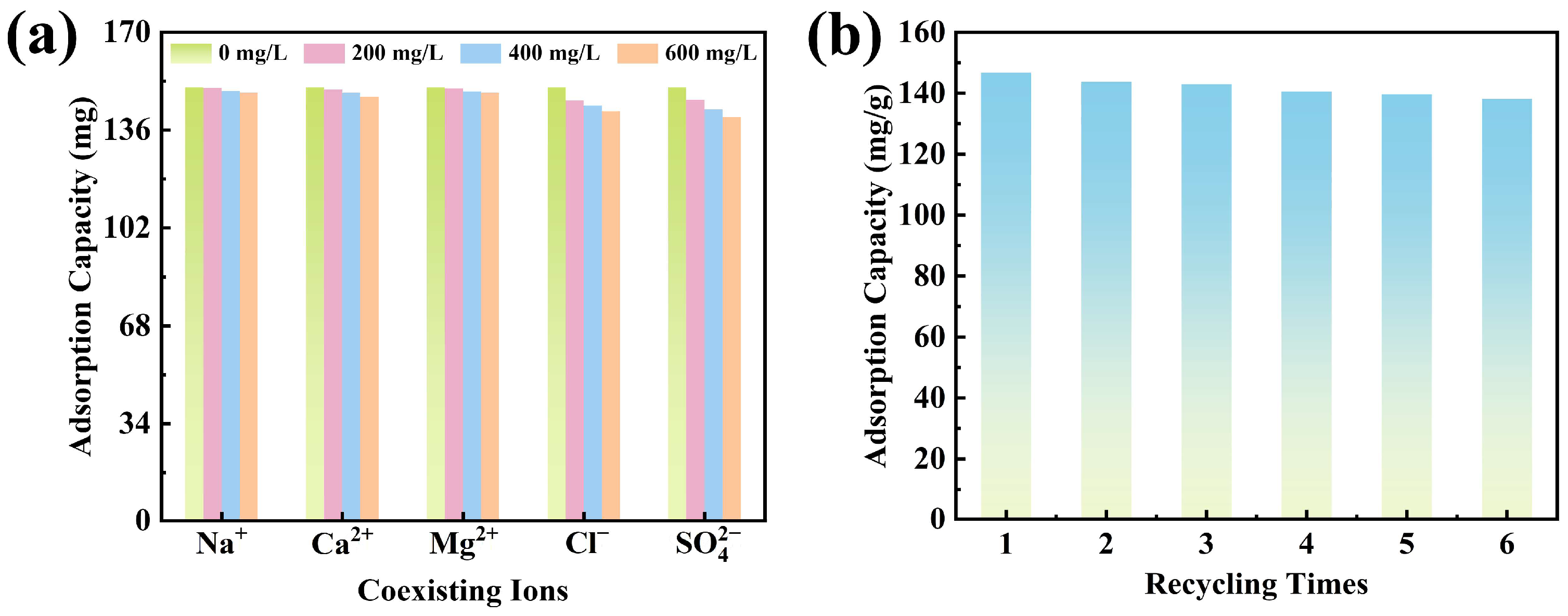
To evaluate the stability and anti-interference performance of CTAB@TBC toward XO adsorption under practical application conditions, a competitive ion coexistence experiment was conducted. As illustrated in
Figure 6a, elevating the levels of coexisting cations produced only a minimal influence on the adsorption of XO by CTAB@TBC. This minimal impact is attributed to the presence of metal ions in aqueous solution primarily as positively charged species or hydrated M(OH)
x complexes, which cannot effectively compete with anionic dye molecules for the available adsorption sites on CTAB@TBC [
32]. In contrast, the presence of anions reduced XO uptake by vying for the positively charged sites available on CTAB@TBC, which also weakened the electrostatic forces responsible for adsorption. Experimental findings revealed a competitive sequence of Cl
− < SO
42−. The monovalent chloride ion, having relatively low charge density, shows weak competitiveness, whereas sulfate, a divalent anion with a high charge density, which results in stronger electrostatic attraction. Consequently, its inhibitory effect on related processes is also more pronounced [
22].
2.2.6. Reusability of CTAB@TBC
Evaluating the cyclic stability of the CTAB@TBC adsorbent during XO removal is crucial for assessing its practical applicability and long-term effectiveness. For regeneration, the adsorbent was desorbed with 0.1 mol/L NaOH solution after each adsorption experiment. This adsorption–desorption cycle was repeated six times, and the resulting recyclability performance is presented in
Figure 6b.
Across successive adsorption–desorption cycles, CTAB@TBC showed a slight reduction in its adsorption capacity for XO. After six adsorption–desorption cycles, the adsorption capacity of CTAB@TBC decreased from 150.82 to 138.12 mg/g, retaining 91.58% of its initial value. This reduction in adsorption efficiency can be attributed to the partial loss of CTAB from the interlayer structure of TBC during desorption, which decreases the number of available active sites. Nevertheless, CTAB@TBC maintained a high adsorption capacity after six cycles, demonstrating good regenerability and promising potential for reuse in practical applications.
To validate the practical applicability of CTAB@TBC, its technical and economic performance was benchmarked against commercial sorbents (e.g., activated carbon, ion-exchange resins) widely used for XO removal. Technically, CTAB@TBC exhibits a superior adsorption capacity (150.82 mg/g) and maintains exceptional cyclic stability, retaining 91.58% of its initial capacity after six cycles. Economically, CTAB@TBC offers a competitive advantage owing to low-cost precursors (TBC and CTAB) and a straightforward synthesis protocol, collectively reducing production costs compared to advanced functional sorbents. In contrast, activated carbon requires energy-intensive activation and regeneration, while ion-exchange resins entail higher synthesis expenses and inferior cyclic stability for anionic dyes such as XO.
After CTAB-modified biochar reaches adsorption saturation, incineration treatment can facilitate the complete mineralization of adsorbed organic dyes, thus effectively eliminating the risk of secondary pollution. Meanwhile, this process enables the recovery of valuable components from incineration ash and the energy utilization of high-carbon substrates, thereby constructing an integrated closed-loop process of “adsorption-incineration-resource recovery”. This study provides a sustainable strategy for dye wastewater treatment and the high-value utilization of biomass resources.
2.3. Adsorption Kinetics
A comprehensive understanding of adsorption kinetics is essential for elucidating the mechanisms governing the interactions between XO molecules and the CTAB@TBC surface. To systematically investigate the time-dependent behavior of these interactions, batch adsorption experiments were conducted at a constant temperature of 25 °C with an initial XO concentration of 100 mg/L. The kinetic data were analyzed using pseudo–first-order and pseudo–second-order models, as well as an intraparticle diffusion model, to elucidate the mechanisms that control the adsorption rate. To explore the kinetic mechanism of the adsorption process, this study employed the pseudo-first-order kinetic model (Equation (1)) and pseudo-second-order kinetic model (Equation (2)), respectively, to characterize the potentially involved physical and chemical adsorption behaviors. In addition, to further elucidate the mass transfer characteristics of XO molecules within the CTAB@TBC material, the intra-particle diffusion model (Equation (3)) was employed to analyze the internal diffusion and any concomitant proton transfer. Subsequently, the experimental data were fitted nonlinearly using the kinetic Equations (1)–(3) [
19,
20,
22].
where q
t and q
e represent the adsorption capacity at contact time t and at adsorption equilibrium, respectively (mg/g). k
1, k
2 and k
i refer to the rate constants of the pseudo-first-order kinetic model (1/min), the pseudo-second-order kinetic model (1/min) and the intraparticle diffusion adsorption model (mg/g·min0.5), respectively, t refers to the adsorption time in the adsorption process (min), C is the boundary layer effect constant in the intraparticle diffusion model (mg/g).
As illustrated in
Figure 7, the pseudo-first-order kinetic, pseudo-second-order kinetic, and intra-particle diffusion models were used to analyze the kinetic behavior of the adsorption process. The rate constants k
1, k
2, and k
i for each model were determined through linear regression. The relevant parameters were calculated according to Equations (3)–(5) and summarized in
Table 1. The analysis of goodness of fit (R
2) and the evaluation results of deviations between the calculated and measured values indicated that the pseudo-second-order kinetic model had a significantly better ability to describe the experimental data than the pseudo-first-order model, suggesting that the adsorption process was more consistent with the pseudo-second-order reaction mechanism. Furthermore, the kinetic parameters suggest that XO adsorption onto CTAB@TBC adheres to a pseudo-second-order kinetic model. The close correspondence between the calculated and experimental values indicates that chemisorption is likely the primary rate-controlling step, involving valence-based interactions through electron sharing or exchange between the CTAB-modified TBC surface and XO molecules [
20,
22].
Figure 7b and
Table 1 display the fitted curves and corresponding parameters of the intraparticle diffusion model, offering insight into contaminant-transport behavior and enabling identification of the rate-controlling steps in the adsorption process. The adsorption process can be divided into three sequential phases: Phase I (0–10 min) is characterized by a fast adsorption step mainly controlled by film-layer diffusion, during which plentiful reactive groups (such as amino groups) together with the porous framework of CTAB@TBC promote effective XO adsorption through electrostatic attraction. Phase II (10–40 min) represents the period in which intraparticle diffusion becomes the predominant mechanism, during which XO gradually moves into the mesoporous framework of CTAB@TBC. The modest decline in diffusion rate indicates that adsorption continues, supported by the adsorbent’s internal pore structure. Phase III (>60 min) marks the onset of adsorption equilibrium, during which the uptake rate declines sharply as the internal active sites become nearly saturated. The considerable intercept (C) observed in the diffusion graph indicates strong boundary-layer resistance and restricted pore accessibility, both of which hinder additional adsorption. Overall, these findings show that CTAB@TBC makes full use of its accessible surface area and pore structure during the adsorption process [
19].
When intraparticle diffusion becomes the rate-limiting step, the overall adsorption rate is primarily governed by the mass transfer of pollutants within the composite porous network structure of the CTAB@TBC adsorbent. The experimental data exhibit a strong fit with the intraparticle diffusion model, suggesting that this mass transfer mechanism plays a dominant role in regulating the adsorption performance of CTAB@TBC. Further analysis, combining the pseudo-second-order kinetic model and the intraparticle diffusion model, reveals that the adsorption behavior of XO onto CTAB@TBC follows a dual mechanism involving both chemisorption and diffusion control. Specifically, chemisorption is represented by the chemical interactions between the adsorbate and the surface functional groups of the adsorbent, while diffusion control reflects the mass transfer resistance encountered by the adsorbate molecules as they migrate through the porous medium. The synergistic effect of the above mechanisms not only endows CTAB@TBC with excellent adsorption capacity, especially high removal efficiency under low-concentration pollutant conditions, but also highlights its potential for practical water treatment applications [
19].
2.4. Adsorption Isotherms
The adsorption isotherm describes the relationship between the equilibrium concentration of XO in solution and the amount adsorbed by CTAB@TBC at a given temperature. The analysis of this isotherm is essential for elucidating the partitioning behavior of dye molecules between the liquid phase and the adsorbent, and it provides a deeper understanding of the adsorption mechanism, surface properties, and binding strength of modified TBC. To gain a deeper understanding of how XO is removed, the interactions between the modified TBC and the solution were further analyzed through isotherm modeling. Batch adsorption experiments were conducted at pH 6 with varying initial concentrations of XO. To evaluate the adsorption data, the Langmuir and Freundlich isotherm models were employed, each offering complementary insight into the fundamental adsorption behavior. The Langmuir model (Equation (4)) assumes that adsorption occurs as a single molecular layer on a homogeneous surface, while the Freundlich model (Equation (5)) serves as an empirical description of adsorption taking place on non-uniform surfaces with reversible binding of molecules [
3,
14].
where K
L denotes the Langmuir constant related to adsorption property (L/mg), K
F represents Freundlich constant indicative of adsorption capacity (mg/g), q
e is the adsorption capacity at equilibrium (mg/g), and C
e denotes the equilibrium concentration of XO that persists in the solution (mg/L).
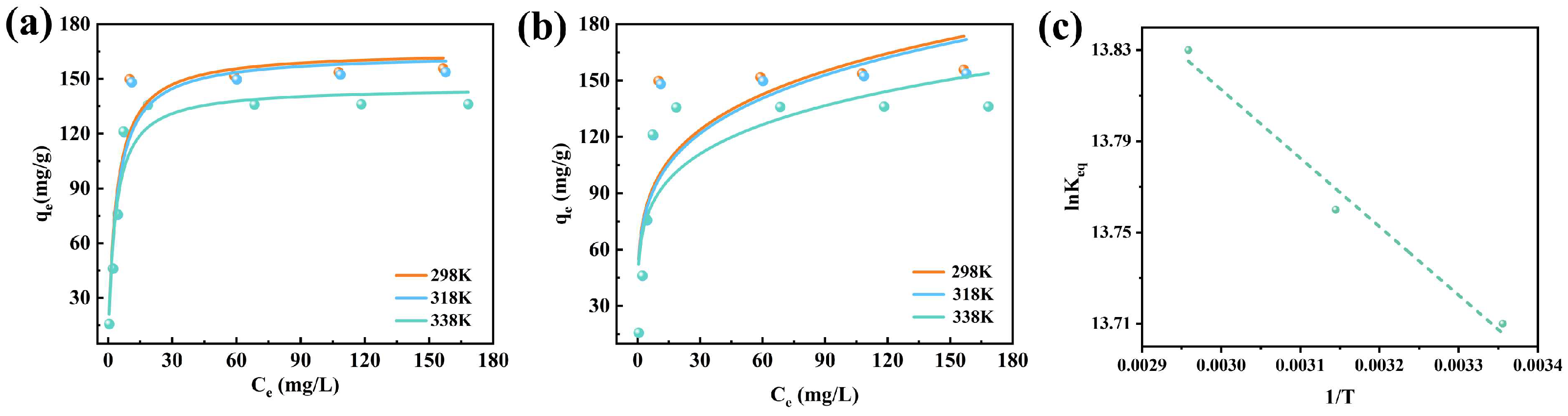
Figure 8 presents the fitting results of the adsorption behavior of XO on CTAB@TBC according to the Langmuir and Freundlich isotherm models. The corresponding model parameters and coefficients of determination (R
2) are provided in
Table 2. The Langmuir model fitted the experimental results better than the Freundlich model, indicating the uniform adsorption of XO dye molecules on the active sites of CTAB@TBC [
14], which implies the formation of monolayer adsorption with a limited capacity due to the finite active sites. A relatively large K
L value (in the unified measurement unit system) reflects a high affinity between CTAB@TBC and XO, endowing the adsorbent with a high adsorption capacity even at low XO concentrations. This type of adsorption is typically irreversible and forms a single molecular layer. The good fitting of experimental data to the Langmuir model verifies this monolayer adsorption mechanism, demonstrating that CTAB@TBC has uniformly distributed surface active sites that only allow monolayer adsorption of XO molecules [
3].
2.5. Thermodynamics of Adsorption
To investigate the thermodynamic mechanism underlying the adsorption process, this study applied the Langmuir model, which provides the best fit for the adsorption isotherm of XO onto CTAB@TBC, to determine the relevant thermodynamic parameters, including the Gibbs free energy change (ΔG), enthalpy change (ΔH), and entropy change (ΔS). These values were calculated using Equations (6)–(8), following the procedures outlined in previous studies [
12,
33].
where K
eq denotes the equilibrium constant for adsorption, R refers to the universal gas constant (8.314 J/mol·K), T indicates for the absolute temperature in Kelvin, M
XO is the molar weight of XO (mg/mol).
The adsorption thermodynamic curve, presented in
Figure 8c, demonstrates that the adsorption behavior follows the Van’t Hoff model. Based on the fitted line, the ΔH and ΔS were derived from its slope and intercept. A complete set of thermodynamic parameters is provided in
Table 3. The positive ΔH indicates that the adsorption of XO onto CTAB@TBC is an endothermic process, while the negative ΔG confirms that the adsorption proceeds spontaneously under the experimental conditions. In addition, the further decrease in ΔG with increasing temperature suggests that higher temperatures enhance the spontaneity of the adsorption process [
19]. The positive value of ΔS indicates that the solid–liquid interface becomes more disordered as XO is adsorbed [
12]. Taken together, these findings demonstrate that the adsorption of XO on CTAB@TBC occurs spontaneously, driven by combined enthalpic and entropic contributions that affirm its thermodynamic favorability under the examined conditions.
2.6. Adsorption Mechanism of XO on CTAB@TBC
Based on the analyses of adsorption kinetics (
Section 2.3) and adsorption isotherms (
Section 2.4), the adsorption of XO onto CTAB@TBC primarily follows a monolayer physicochemical process dominated by chemical adsorption. This process also involves physical mechanisms, including surface adsorption, intraparticle diffusion, and electrostatic interactions. The numerous active sites on the CTAB@TBC surface promote the adsorption of XO molecules, which mainly arises from van der Waals and other intermolecular forces between the material surface and XO molecules. TBC possesses a well-developed mesoporous structure (average pore diameter 29.60 nm), which is substantially larger than the Stokes radius (0.129 nm) and the hydrated radius (0.335 nm) of XO, allowing XO molecules to freely access the pores [
34,
35]. CTAB modification imparts a positive surface charge to TBC, facilitating efficient adsorption of anionic XO via electrostatic attraction. Furthermore, lower temperatures favor physical adsorption. The synergistic effect of these mechanisms enables CTAB@TBC to retain high adsorption performance at low temperatures.
Then, the adsorption mechanism was investigated by comparing the FT-IR and XPS characteristics before and after XO adsorption, with a systematic assessment of the contributions of physical adsorption, electrostatic attraction, hydrophobic interaction, and complexation.
As shown in
Figure 9a, the broad O–H stretching vibration peak at 3417 cm
−1 is attributed to the –OH or –SO
3− groups of XO interacting with hydrogen atoms on the surface of CTAB@TBC via hydrogen bonding, which represents a typical physical adsorption interaction. Additionally, the decreased intensity of the C–H stretching vibration peaks of CTAB (2919.6 and 2850.2 cm
−1) suggests a conformational change in the alkyl chain induced by XO adsorption, reflecting hydrophobic interactions between the alkyl tails of CTAB and the aromatic rings of XO. The peak at 1620 cm
−1 corresponds to π–π stacking between the C=C bonds in the XO benzene ring and the aromatic ring of biochar, another physical adsorption force that enhances the binding affinity. The peak at 1116.5 cm
−1 is associated with the S=O stretching vibration of the sulfonic acid group in XO, confirming the presence of XO on the material surface [
12,
36]. These FT-IR results collectively indicate that both physical adsorption (hydrogen bonding, π–π stacking) and hydrophobic interactions contribute to the initial adsorption stage.
The XPS spectra of CTAB@TBC and CTAB@TBC with XO adsorbed (
Figure 9b–e) further reveal the multi-stage nature of the adsorption process. As shown in
Figure 9b, the appearance of a new S 2p characteristic peak (corresponding to the SO
3− group in XO) and the disappearance of the Br 3d peak (corresponding to Br
− in CTAB) confirm an ion exchange process, which is a form of electrostatic attraction. This ion exchange represents a dominant chemical interaction in the intermediate stage of adsorption. The C 1s spectrum exhibited no significant changes, indicating that the carbon backbone did not participate in the adsorption process, which supports that the main interactions occur at the functional groups rather than the carbon skeleton [
36]. In the O 1s spectrum, the increased proportion of the C=O component (from 35.47% to 38.88%) after XO adsorption suggests the formation of coordinate bonds between the carboxyl groups of XO and the surface sites of CTAB@TBC, indicating a contribution from complexation. The decreased proportion of C–O (from 39.88% to 37.34%) reflects the consumption of surface hydroxyl groups to form hydrogen bonds with XO, further verifying the role of physical adsorption. The slight decrease in adsorbed water (from 24.65% to 23.78%) implies dehydration during adsorption, which is consistent with the strengthening of surface interactions [
12]. In the N 1s spectrum, the positive shift in binding energies of –NH
2 (from 399.98 eV to 400.18 eV) and –NH
3+ (from 402.64 eV to 402.97 eV) indicates a reduction in electron cloud density around nitrogen atoms, which is attributed to electrostatic attraction between the quaternary ammonium groups of CTAB and the sulfonic acid groups of XO [
35,
36]. This electrostatic interaction is a dominant force in the later stage of adsorption, stabilizing the adsorbed XO molecules.
Overall, the adsorption process of XO onto CTAB@TBC involves multiple synergistic mechanisms: In the initial stage, physical adsorption (hydrogen bonding, π–π stacking) and hydrophobic interactions dominate, driving the rapid attachment of XO to the material surface. In the intermediate stage, ion exchange (a form of electrostatic attraction) becomes prominent, facilitating the replacement of Br− by SO3− and enhancing the binding strength. In the final stage, electrostatic attraction and complexation stabilize the adsorbed XO, with the quaternary ammonium groups and surface carboxyl groups playing key roles.
Based on a comprehensive assessment of the characterization results and experimental data, we put forward a reasonable explanation of how XO adsorbs onto CTAB@TBC, as depicted in
Figure 10.