Xanthone Dimers in Angiosperms, Fungi, Lichens: Comprehensive Review of Their Sources, Structures, and Pharmacological Properties
Abstract
1. Introduction
2. Distribution
3. Structural Characteristics and Classification
3.1. Dimer
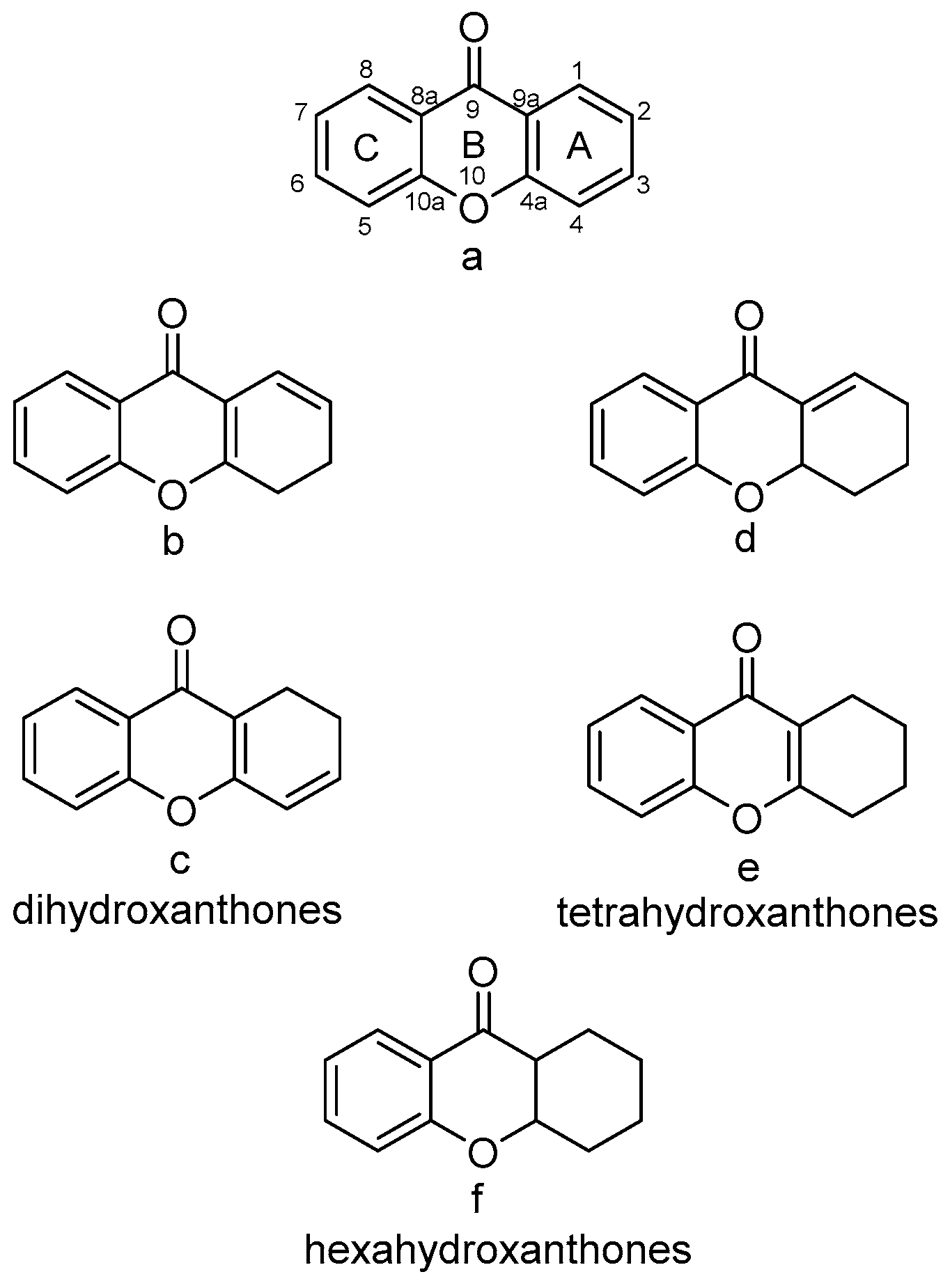
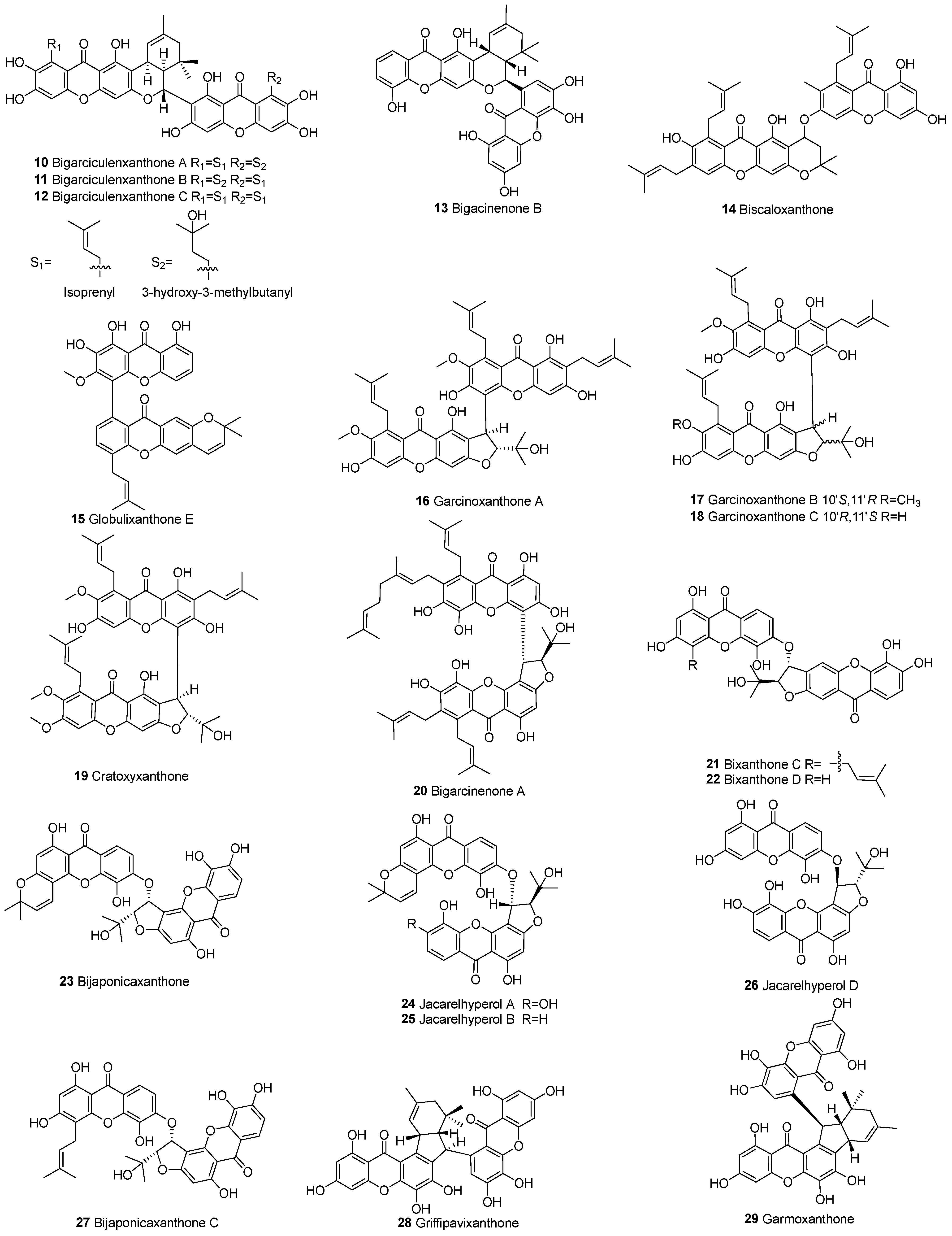
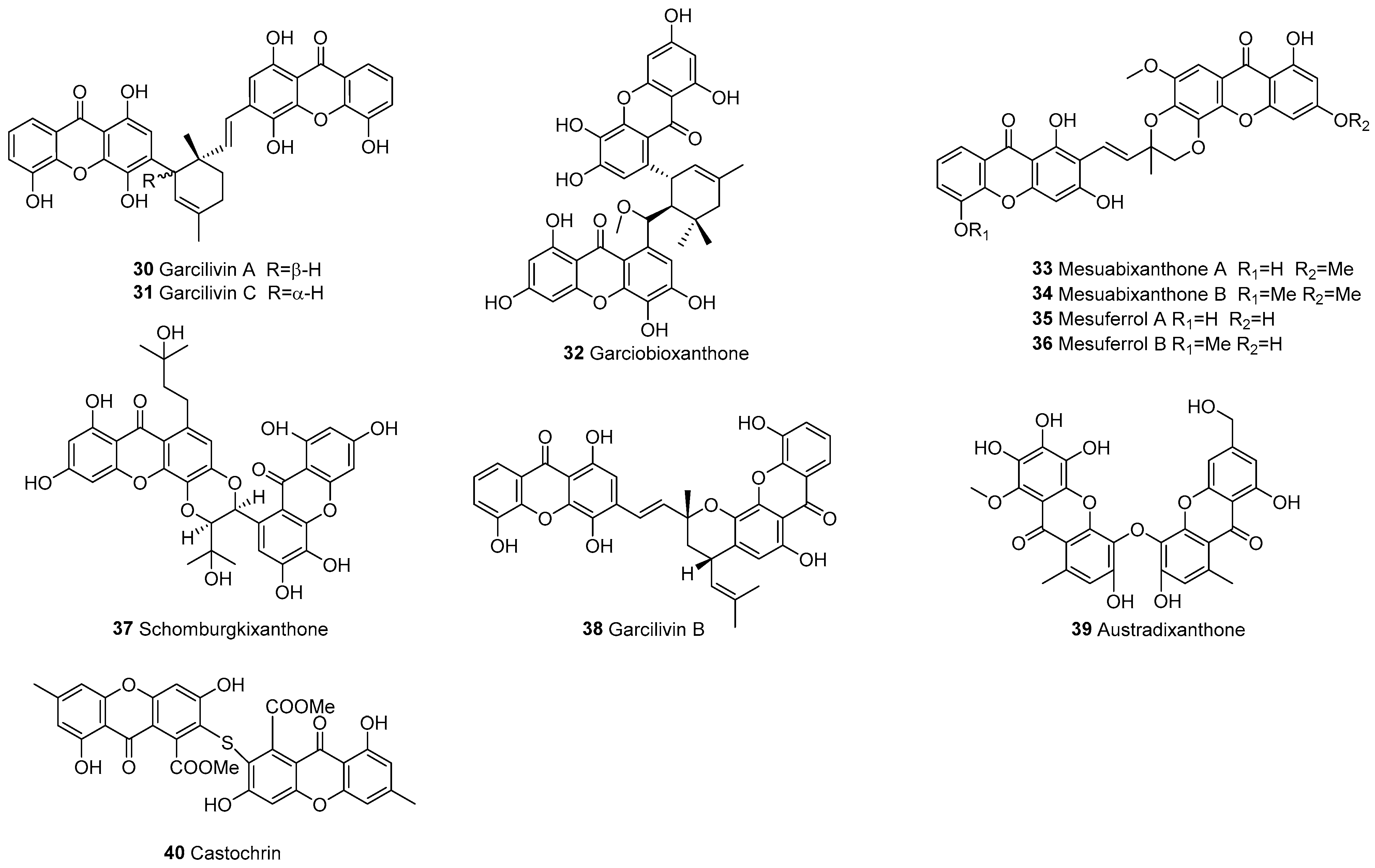
3.1.1. Xanthone–Xanthone Dimers (a–a)
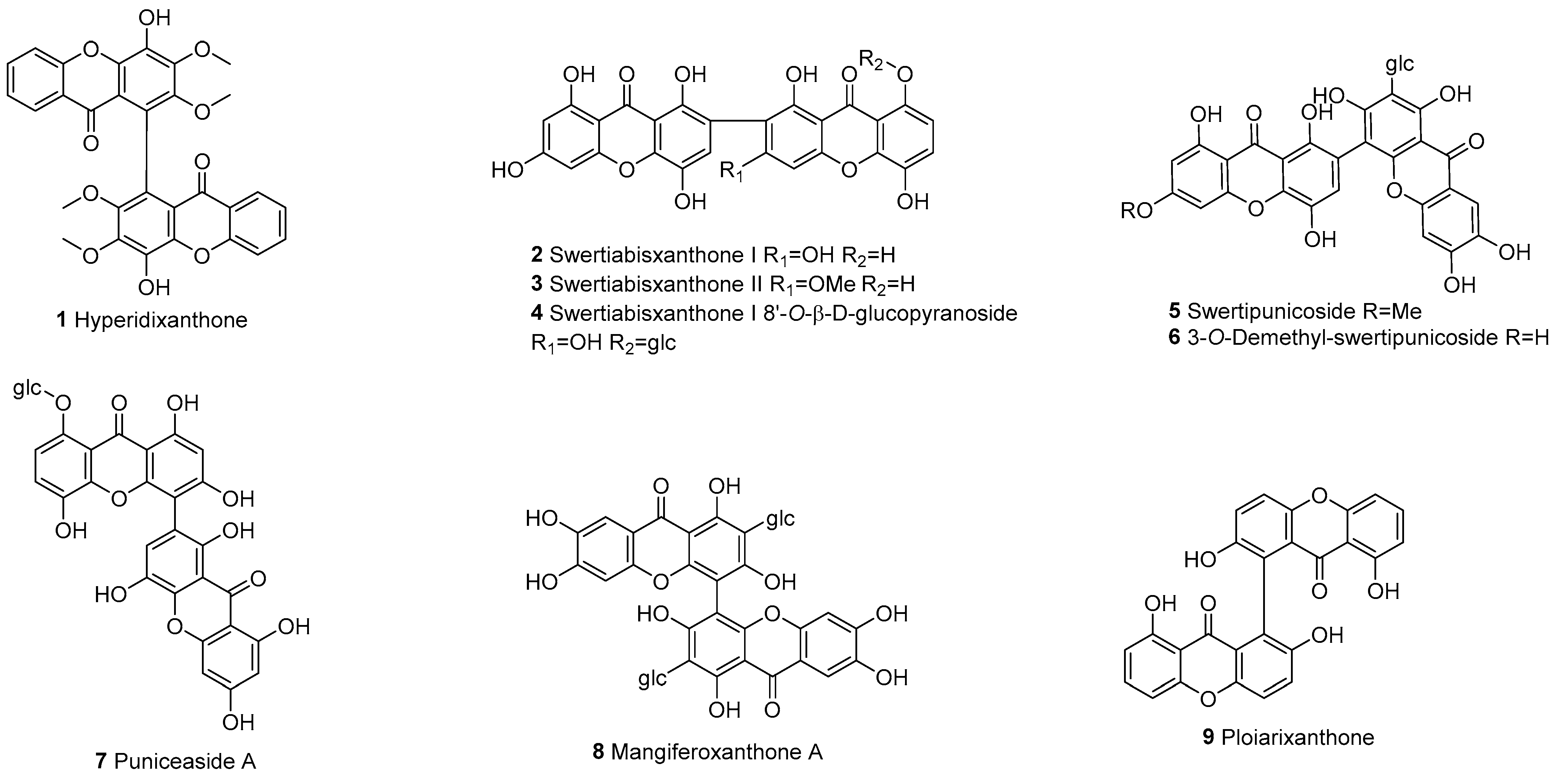
3.1.2. Xanthone–Tetrahydroxanthone Dimers (a–e)
3.1.3. Dimeric Dihydroxanthones (b–b)
3.1.4. Dihydroxanthone–Tetrahydroxanthone Dimers (b–d or c–e)
3.1.5. Dimeric Tetrahydroxanthones (d–d or e–e)
3.1.6. Tetrahydroxanthone (d)–Hexahydroxanthone (f) Dimers
3.1.7. Dimeric Hexahydroxanthones (f–f)
3.2. Heterodimers
3.2.1. Xanthone–Flavone Heterodimers
3.2.2. Xanthonelignans
3.2.3. Xanthone–Benzophenone Heterodimers
3.2.4. Xanthone–Chromanone Heterodimers
3.2.5. Dimeric Xanthone Derivatives (Chromanone–Chromanone Dimers)
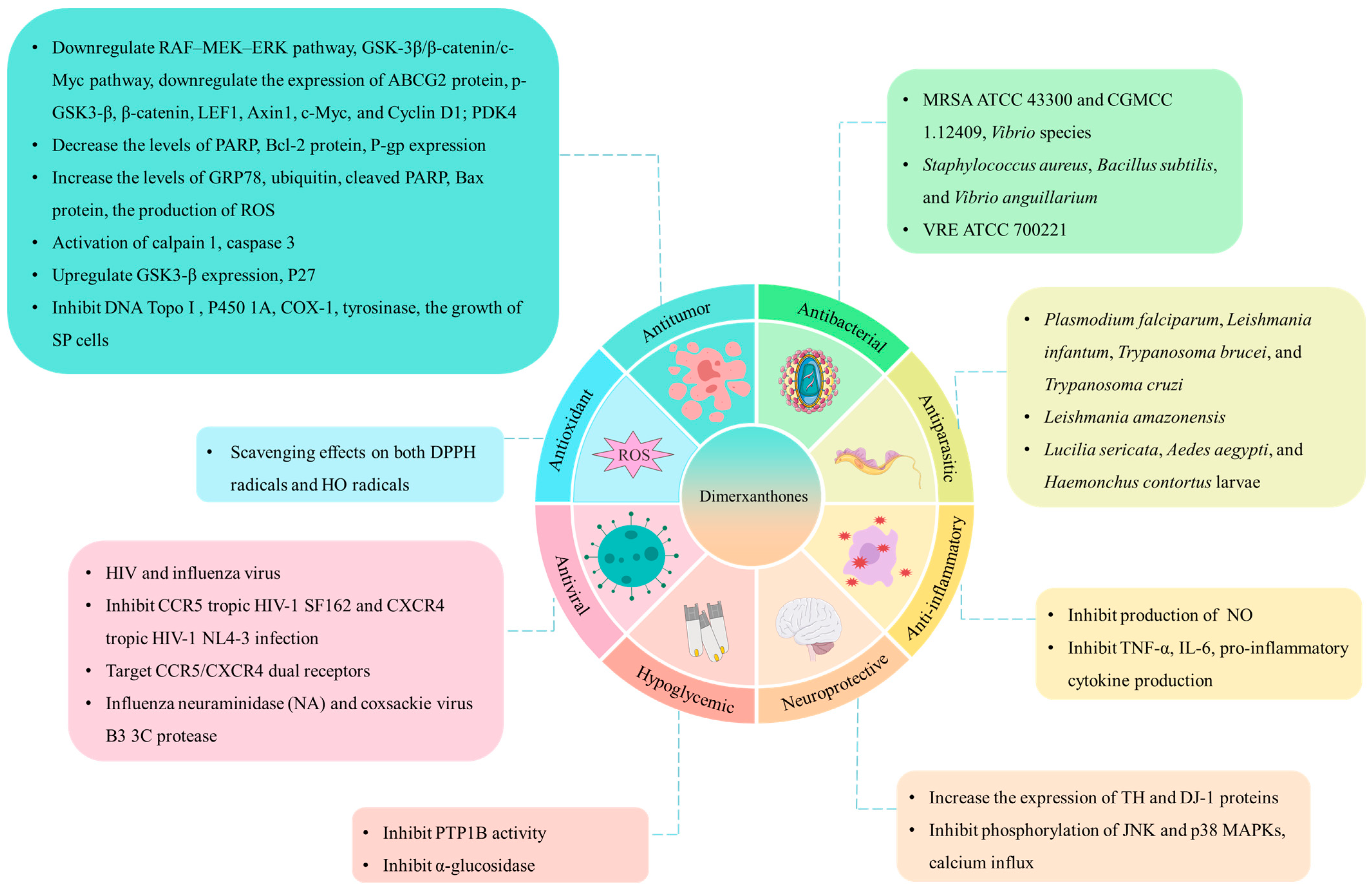
4. Pharmacology Effects
4.1. Antitumor Activity
4.2. Antibacterial Activity
4.3. Antioxidant Activity
4.4. Anti-Inflammatory Activity
4.5. Neuroprotective Effects
4.6. Hypoglycemic Effects
4.7. Antiviral Activity
4.8. Antiparasitic Activity
4.9. Other Activities
5. Conclusions and Prospects
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- El-Seedi, H.R.; Ibrahim, H.; Yosri, N.; Ibrahim, M.A.A.; Hegazy, M.-E.F.; Setzer, W.N.; Guo, Z.; Zou, X.; Refaey, M.S.; Salem, S.E.; et al. Naturally occurring xanthones; biological activities, chemical profiles and in silico drug discovery. Curr. Med. Chem. 2023, 31, 62–101. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.Y.; Sun, S.F.; Yi, C.; Yang, C.-Y.; Chen, K.-L.; Zhang, X.-W.; Liu, Y.-B. Muyocoxanthones O–S: Undescribed xanthones with antioxidative damage bioactivity to cardiomyocytes from the endophytic fungus Muyocopron laterale. Phytochemistry 2023, 209, 113625. [Google Scholar] [CrossRef]
- Mohammad, N.A.; Abang Zaidel, D.N.; Muhamad, I.I.; Hamid, M.A.; Yaakob, H.; Jusoh, Y.M.M. Optimization of the antioxidant-rich xanthone extract from mangosteen (Garcinia mangostana L.) pericarp via microwave-assisted extraction. Heliyon 2019, 5, e02571. [Google Scholar] [CrossRef]
- Park, S.Y.; Lee, J.H.; Ko, S.Y.; Kim, N.; Kim, S.Y.; Lee, J.C. Antimicrobial activity of alpha-mangostin against Staphylococcus species from companion animals in vitro and therapeutic potential of alpha-mangostin in skin diseases caused by S. pseudintermedius. Front. Cell Infect. Microbiol. 2023, 13, 1203663. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.J.; Song, M.R.; Liu, Y.; Yang, S.; Chen, S.; Kang, J.; Shen, J.; Zhu, K. Rational Design of Natural Xanthones Against Gram-negative Bacteria. Adv. Sci. 2025, e2411923. [Google Scholar] [CrossRef]
- Tran, T.T.T.; Le, P.M.; Nguyen, T.K.A.; Hoang, T.M.N.; Do, T.Q.A.; Martel, A.L.; Lewicky, J.D.; Klem, A.; Le, H.-T. Novel human STING activation by hydrated-prenylated xanthones from Garcinia cowa. J. Pharm. Pharmacol. 2023, 75, 1058–1065. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Mansouripour, A.; Xi, Z.; Zhang, L.; Xu, G.; Zhou, H.; Xu, H. Nujiangexanthone A inhibits cervical cancer cell proliferation by promoting mitophagy. Molecules 2021, 26, 2858. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Wang, N.; Tian, X.Y.; Su, J.; Qin, Y.; He, R.; He, X. The protective effect of mangiferin on formaldehyde-induced HT22 cell damage and cognitive impairment. Pharmaceutics 2023, 15, 1568. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.L.; Chen, Y.C.; Xiong, L.A.; Huang, Q.Y.; Gong, T.T.; Chen, Y.; Ma, L.F.; Fang, L.; Zhan, Z.J. Discovery of phenylcarbamoyl xanthone derivatives as potent neuroprotective agents for treating ischemic stroke. Eur. J. Med. Chem. 2023, 251, 115251. [Google Scholar] [CrossRef]
- Zhong, Y.M.; Xu, Y.Y.; Tan, Y.Z.; Zhang, X.; Wang, R.; Chen, D.; Wang, Z.; Zhong, X. Lipidomics of the erythrocyte membrane and network pharmacology to explore the mechanism of mangiferin from Anemarrhenae rhizoma in treating type 2 diabetes mellitus rats. J. Pharm. Biomed. Anal. 2023, 230, 115386. [Google Scholar] [CrossRef]
- Wang, L.P.; Fu, W.W.; Tan, H.S.; Hong, Z.; Xu, H. Chemistry of xanthones isolated from Garcinia species in China. World Chin. Med. 2016, 11, 1154–1170. [Google Scholar]
- Qin, T.; Porco, J.A. Total syntheses of secalonic acids A and D. Angew. Chem. Int. Ed. 2014, 53, 3107–3110. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.D.; Guo, Z.M.; Wang, J.B.; Hu, G.; Su, Y.; Chen, L.; Lv, Q.; Yu, H.; Qin, J.; Xu, W. Dicerandrol B: A natural xanthone dimer induces apoptosis in cervical cancer HeLa cells through the endoplasmic reticulum stress and mitochondrial damage. Oncol. Targets Ther. 2019, 12, 1185–1193. [Google Scholar] [CrossRef]
- Ma, Y.C.; Wang, Y.; Song, B. Griffipavixanthone induces apoptosis of human breast cancer MCF-7 cells in vitro. Breast Cancer 2019, 26, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Ding, Z.J.; Lao, Y.Z.; Zhang, H.; Fu, W.; Zhu, L.; Tan, H.; Xu, H. Griffipavixanthone, a dimeric xanthone extracted from edible plants, inhibits tumor metastasis and proliferation via downregulation of the RAF pathway in esophageal cancer. Oncotarget 2016, 7, 1826–1837. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Yu, G.; Kurtan, T.; Mandi, A.; Peng, J.; Mo, X.; Liu, M.; Li, H.; Sun, X.; Li, J.; et al. Versixanthones A–F, cytotoxic xanthone–chromanone dimers from the marine-derived fungus Aspergillus versicolor HDN1009. J. Nat. Prod. 2015, 78, 2691–2698. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.L.; Han, J.H.; Zhang, X.W.; Xu, W.; Yang, J.; Song, F. Investigation on the chemical constituents of the marine-derived fungus strain Aspergillus brunneoviolaceus MF180246. Nat. Prod. Res. 2022, 38, 1369–1374. [Google Scholar] [CrossRef] [PubMed]
- Zang, Z.; Yang, W.; Cui, H.; Cai, R.; Li, C.; Zou, G.; Wang, B.; She, Z. Two antimicrobial heterodimeric tetrahydroxanthones with a 7,7′-Linkage from mangrove endophytic fungus Aspergillus flavus QQYZ. Molecules 2022, 27, 2691. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Shui, H.; Zhang, M.; Zeng, Y.; Zheng, M.; Zhu, K.K.; Wang, S.B.; Bi, H.; Hong, K.; Cai, Y.S. Aculeaxanthones A–E, new xanthones from the marine-derived fungus Aspergillus aculeatinus WHUF0198. Front. Microbiol. 2023, 14, 1138830. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.Y.; Liao, Y.Y.; Huang, X.M.; Tang, C.; Cai, P. A novel xanthone dimer derivative with antibacterial activity isolated from the bark of Garcinia mangostana. Nat. Prod. Res. 2018, 32, 1769–1774. [Google Scholar] [CrossRef]
- Merza, J.; Aumond, M.C.; Rondeau, D.; Dumontet, V.; Le Ray, A.-M.; Séraphin, D.; Richomme, P. Prenylated xanthones and tocotrienols from Garcinia virgata. Phytochemistry 2004, 65, 2915–2920. [Google Scholar] [CrossRef] [PubMed]
- Zhong, F.F.; Chen, Y.; Yang, G.Z. Chemical constituents from the bark of Garcinia xanthochymus and their 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical-scavenging activities. Helv. Chim. Acta 2008, 9, 1695–1703. [Google Scholar] [CrossRef]
- Du, X.G.; Wang, W.W.; Zhang, S.P.; Pu, X.-P.; Zhang, Q.-Y.; Ye, M.; Zhao, Y.-Y.; Wang, B.-R.; Khan, I.A.; Guo, D.-A. Neuroprotective xanthone glycosides from Swertia punicea. J. Nat. Prod. 2010, 73, 1422–1426. [Google Scholar] [CrossRef]
- Zhang, S.P.; Du, X.G.; Pu, X.P. 3-O-demethylswertipunicoside protects against oxidative toxicity in PC12 cells. Biol. Pharm. Bull. 2010, 33, 1529–1533. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shi, J.M.; Huang, H.J.; Qiu, S.X.; Feng, S.-X.; Li, X.-E. Griffipavixanthone from Garcinia oblongifolia champ induces cell apoptosis in human non-small-cell lung cancer H520 cells in vitro. Molecules 2014, 19, 1422–1431. [Google Scholar] [CrossRef] [PubMed]
- Oriola, A.O.; Kar, P. Naturally occurring xanthones and their biological implications. Molecules 2024, 29, 4241. [Google Scholar] [CrossRef] [PubMed]
- Wezeman, T.; Bräse, S.; Masters, K.S. Xanthone dimers: A compound family which is both common and privileged. Nat. Prod. Rep. 2015, 32, 6–28. [Google Scholar] [CrossRef] [PubMed]
- Masters, K.S.; Brase, S. Xanthones from fungi, lichens, and bacteria: The natural products and their synthesis. Chem. Rev. 2012, 112, 3717–3776. [Google Scholar] [CrossRef]
- Wang, W.; Zeng, Y.H.; Osman, K.; Shinde, K.; Rahman, M.; Gibbons, S.; Mu, Q. Norlignans, acylphloroglucinols, and a dimeric xanthone from Hypericum chinense. J. Nat. Prod. 2010, 73, 1815–1820. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.M.; Liu, Y.L.; Blaskó, G.; Cordell, G.A. Swertiabisxanthone-I from Swertia macrosperma. Phytochemistry 1989, 28, 3569–3571. [Google Scholar] [CrossRef]
- Yan, Y.S.; Li, Y.X.; Sa, K.R.; Sun, D.; Li, H.; Chen, L. Xanthones and phenylpropanoids from the whole herb of Swertia pseudochinensis and their anti-inflammatory activity. Chem. Biodivers. 2023, 20, e202201040. [Google Scholar] [CrossRef]
- Urbain, A.; Marston, A.; Grilo, L.S.; Bravo, J.; Purev, O.; Purevsuren, B.; Batsuren, D.; Reist, M.; Carrupt, P.A.; Hostettmann, K. Xanthones from Gentianella amarellas sp. acuta with acetylcholinesterase and monoamine oxidase inhibitory activities. J. Nat. Prod. 2008, 71, 895–897. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.N.; Hou, C.Y.; Liu, Y.L.; Lin, L.-Z.; Gil, R.R.; Cordell, G.A. Swertifrancheside, an HIV-reverse transcriptase inhibitor and the first flavone-xanthone dimer, from Swertia franchetiana. J. Nat. Prod. 1994, 57, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Mageed, W.M.; Bayoumi, S.A.; Chen, C.X.; Vavricka, C.J.; Li, L.; Malik, A.; Dai, H.; Song, F.; Wang, L.; Zhang, J.; et al. Benzophenone C-glucosides and gallotannins from mango tree stem bark with broad-spectrum anti-viral activity. Bioorganic Med. Chem. 2014, 22, 2236–2243. [Google Scholar] [CrossRef] [PubMed]
- Bennett, G.J.; Hiok-Huang, L.; Timothy, K.L. Novel metabolites from Ploiarium alternifolium, a bixanthone and two anthraquinonylxanthones. Tetrahedron Lett. 1990, 31, 751–754. [Google Scholar] [CrossRef]
- Shi, F.Z.; Fang, Y.D.; Fan, M.; Jiang, X.-J.; Wang, S.; Wei, G.-Z. Cytotoxic depsidones and xanthones from Garcinia esculenta Y. H. Li. Fitoterapia 2024, 172, 105779. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Fan, H.; Yang, G.Z.; Jiang, Y.; Zhong, F.; He, H. Two unusual xanthones from the bark of Garcinia xanthochymus. Helv. Chim. Acta 2011, 94, 662–668. [Google Scholar] [CrossRef]
- Taher, M.; Salleh, W.M.N.H.W.; Alkhamaiseh, S.I.; Ahmad, F.; Rezali, M.F.; Susanti, D.; Hasan, C.M. A new xanthone dimer and cytotoxicity from the stem bark of Calophyllum canum. Z. Naturforschung C 2021, 76, 87–91. [Google Scholar] [CrossRef]
- Nkengfack, A.E.; Mkounga, P.; Meyer, M.; Fomum, Z.T.; Bodo, B. Globulixanthones C, D and E: Three prenylated xanthones with antimicrobial properties from the root bark of Symphonia globulifera. Phytochemistry 2002, 61, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.Y.; Li, D.; Wang, A.Q.; Dong, Z.; Yin, S.; Zhang, Q.; Ye, Y.; Li, L.; Lin, L. Nitric oxide inhibitory xanthones from the pericarps of Garcinia mangostana. Phytochemistry 2016, 131, 115–123. [Google Scholar] [CrossRef]
- Sia, G.L.; Bennett, G.J.; Harrison, L.J.; Sim, K.Y. Minor xanthone from the bark of Cratoxylum cochinchinense. Phytochemistry 1995, 6, 1521–1528. [Google Scholar] [CrossRef]
- Zhu, L.L.; Fu, W.W.; Watanabe, S.; Shao, Y.-N.; Tan, H.-S.; Zhang, H.; Tan, C.-H.; Xiu, Y.-F.; Norimoto, H.; Xu, H.-X. Xanthine oxidase inhibitors from Garcinia esculenta twigs. Planta Medica 2014, 80, 1721–1726. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.L.; Wang, S.P.; Du, L.J.; Yang, J.-S.; Xiao, P.-G. Xanthones from Hypericum japonicum and H. henryi. Phytochemistry 1998, 49, 1395–1402. [Google Scholar] [CrossRef]
- Ishiguro, K.; Nagata, S.; Oku, H.; Yamaki, M. Bisxanthones from Hypericum japonicum: Inhibitors of PAF-induced hypotension. Planta Medica 2002, 68, 258–261. [Google Scholar] [CrossRef]
- Fu, P.; Zhang, W.D.; Li, T.Z.; Liu, R.H.; Li, H.L.; Zhang, W.; Chen, H.S. A new bisxanthone from Hypericum japonicum Thunb.ex Murray. Chin. Chem. Lett. 2005, 16, 771–773. [Google Scholar]
- Triyasa, K.S.; Diantini, A.; Barliana, M.I. A review of herbal medicine-based phytochemical of Garcinia as molecular therapy for breast cancer. Drug Des. Dev. Ther. 2022, 16, 3573–3588. [Google Scholar] [CrossRef]
- Mbwambo, Z.H.; Kapingu, M.C.; Moshi, M.J.; Machumi, F.; Apers, S.; Cos, P.; Ferreira, D.; Marais, J.P.J.; Berghe, D.V.; Maes, L.; et al. Antiparasitic activity of some xanthones and biflavonoids from the root bark of Garcinia livingstonei. J. Nat. Prod. 2006, 69, 369–372. [Google Scholar] [CrossRef] [PubMed]
- Sordat-Diserens, I.; Hamburger, M.; Rogers, C.; Hostettmannt, K. Dimeric xanthones from Garcinia livingstonei. Phytochemistry 1992, 31, 3589–3593. [Google Scholar] [CrossRef]
- Feng, S.; Jiang, Y.; Li, J.; Qiu, S.; Chen, T. A new bixanthone derivative from the bark of Garcinia oblongifolia. Nat. Prod. Res. 2014, 28, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Gray, A.I.; Waterman, P.G. Mesuabixanthone-A and Mesuabixanthone-B: Novel bis-xanthones from the stem bark of Mesua ferrea (Guttiferae). Nat. Prod. Lett. 1993, 1, 53–58. [Google Scholar] [CrossRef]
- Linum, M.; Tosa, H.; Tanaka, T.; Riswan, S. Two new dimeric xanthones in Mesua ferrea. Heterocycles 1996, 43, 1999–2008. [Google Scholar]
- Lien Do, T.M.; Duong, T.H.; Nguyen, V.K.; Phuwapraisirisan, P.; Doungwichitrkul, T.; Niamnont, N.; Jarupinthusophon, S.; Sichaem, J. Schomburgkixanthone, a novel bixanthone from the twigs of Garcinia schomburgkiana. Nat. Prod. Res. 2021, 35, 3613–3618. [Google Scholar] [CrossRef] [PubMed]
- Ebrahim, W.; El-Neketi, M.; Lewald, L.I.; Orfali, R.S.; Lin, W.; Rehberg, N.; Kalscheuer, R.; Daletos, G.; Proksch, P. Metabolites from the fungal endophyte Aspergillus austroafricanus in Axenic culture and in fungal-bacterial mixed cultures. J. Nat. Prod. 2016, 79, 914–922. [Google Scholar] [CrossRef]
- Cai, S.; King, J.B.; Du, L.; Powell, D.R.; Cichewicz, R.H. Bioactive sulfur-containing sulochrin dimers and other metabolites from an Alternaria sp. isolate from a Hawaiian soil sample. J. Nat. Prod. 2014, 77, 2280–2287. [Google Scholar] [CrossRef]
- Li, S.J.; Jiao, F.W.; Li, W.; Zhang, X.; Yan, W.; Jiao, R.H. Cytotoxic xanthone derivatives from the mangrove-derived endophytic fungus Peniophora incarnata Z4. J. Nat. Prod. 2020, 83, 2976–2982. [Google Scholar] [CrossRef] [PubMed]
- Cai, G.; Hu, X.; Zhang, R.; Wang, J.; Fang, X.; Pang, X.; Bai, J.; Zhang, T.; Zhang, T.; Lv, H.; et al. Subplenones A–J: Dimeric xanthones with antibacterial activity from the endophytic fungus Subplenodomus sp. CPCC 401465. J. Nat. Prod. 2023, 86, 2474–2486. [Google Scholar] [CrossRef] [PubMed]
- Shim, S.H.; Baltrusaitis, J.; Gloer, J.B.; Wicklow, D.T. Phomalevones A–C: Dimeric and pseudodimeric polyketides from a fungicolous Hawaiian isolate of Phoma sp. (Cucurbitariaceae). J. Nat. Prod. 2011, 74, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.W.; Wu, X.Y.; Zhao, Z.Y.; Huang, Z.-Q.; Lei, X.-S.; Yang, G.-X.; Li, J.; Xiong, J.; Hu, J.-F. Terricoxanthones A–E, unprecedented dihydropyran-containing dimeric xanthones from the endophytic fungus Neurospora terricola HDF-Br-2 associated with the vulnerable conifer Pseudotsuga gaussenii. Phytochemistry 2024, 219, 113963. [Google Scholar] [CrossRef]
- Guan, J.; Zhang, P.P.; Wang, X.H.; Guo, Y.-T.; Zhang, Z.-J.; Li, P.; Lin, L.-P. Structure-guided discovery of diverse cytotoxic dimeric xanthones/chromanones from Penicillium chrysogenum C-7-2-1 and their interconversion properties. J. Nat. Prod. 2024, 87, 238–251. [Google Scholar] [CrossRef]
- Rotinsulu, H.; Yamazaki, H.; Miura, T.; Chiba, S.; Wewengkang, D.S.; A Sumilat, D.; Namikoshi, M. A 2,4′-linked tetrahydroxanthone dimer with protein tyrosine phosphatase 1B inhibitory activity from the Okinawan freshwater Aspergillus sp. J. Antibiot. 2017, 70, 967–969. [Google Scholar] [CrossRef] [PubMed]
- El-Elimat, T.; Figueroa, M.; Raja, H.A.; Graf, T.N.; Swanson, S.M.; Falkinham, J.O.; Wani, M.C.; Pearce, C.J.; Oberlies, N.H. Biosynthetically distinct cytotoxic polyketides from Setophoma terrestris. Eur. J. Org. Chem. 2015, 2015, 109–121. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, H.; Ukai, K.; Namikoshi, M. Asperdichrome, an unusual dimer of tetrahydroxanthone through an ether bond, with protein tyrosine phosphatase 1B inhibitory activity, from the Okinawan freshwater Aspergillus sp. TPU1343. Tetrahedron Lett. 2016, 57, 732–735. [Google Scholar] [CrossRef]
- Ganapathy, D.; Reiner, J.R.; Loffler, L.E.; Ma, L.; Gnanaprakasam, B.; Niepötter, B.; Koehne, I.; Tietze, L.F. Enantioselective total synthesis of secalonic Acid E. Chemistry 2015, 21, 16807–16810. [Google Scholar] [CrossRef]
- Franck, B.; Baumann, G.; Ohnsorge, U. Ergochrome, an unusual complete group of dimer dyes from Claviceps purpurea. Tetrahedron Lett. 1965, 6, 2031–2037. [Google Scholar] [CrossRef] [PubMed]
- Valdomir, G.; Senthilkumar, S.; Ganapathy, D.; Zhang, Y.; Tietze, L.F. Enantioselective total synthesis of blennolide H and phomopsis-H76 A and determination of their structure. Chemistry 2018, 24, 8760–8763. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Yi, Z.W.; Wang, Y.Q.; Zhang, Q.; Zhong, T.; Qiu, Y.; Wu, Z.; Tang, X. Differential proteomic analysis of HL60 cells treated with secalonic acid F reveals caspase 3-induced cleavage of Rho GDP dissociation inhibitor 2. Oncol. Rep. 2012, 28, 2016–2022. [Google Scholar] [CrossRef]
- Andersen, R.; Büchi, G.; Kobbe, B.; Demain, A.L. Secalonic acids D and F are toxic metabolites of Aspergillus aculeatus. J. Org. Chem. 1977, 42, 352–353. [Google Scholar] [CrossRef]
- Peng, X.P.; Sun, F.S.; Li, G.; Wang, C.; Zhang, Y.; Wu, C.; Zhang, C.; Sun, Y.; Wu, S.; Zhang, Y.; et al. New xanthones with antiagricultural fungal pathogen activities from the endophytic fungus Diaporthe goulteri L17. J. Agric. Food Chem. 2021, 69, 11216–11224. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Luo, Y.F.; Zhang, M.; Dai, J.-G.; Wang, W.-J.; Wu, J. Three xanthone dimers from the Thai mangrove endophytic fungus Phomopsis sp. xy21. J. Asian Nat. Prod. Res. 2018, 20, 217–226. [Google Scholar] [CrossRef]
- Rukachaisirikul, V.; Sommart, U.; Phongpaichit, S.; Sakayaroj, J.; Kirtikara, K. Metabolites from the endophytic fungus Phomopsis sp. PSU-D15. Phytochemistry 2008, 69, 783–787. [Google Scholar] [CrossRef]
- Ding, B.; Yuan, J.; Huang, X.S.; Wen, W.; Zhu, X.; Liu, Y.; Li, H.; Lu, Y.; He, L.; Tan, H.; et al. New dimeric members of the phomoxanthone family: Phomolactonexanthones A, B and deacetylphomoxanthone C isolated from the fungus Phomopsis sp. Mar. Drugs 2013, 11, 4961–4972. [Google Scholar] [CrossRef] [PubMed]
- Wagenaar, M.M.; Clardy, J. Dicerandrols, new antibiotic and cytotoxic dimers produced by the fungus Phomopsis longicolla isolated from an endangered Mint. J. Nat. Prod. 2001, 64, 1006–1009. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.D.; Feng, T.; Xu, J. Mangrove endophytic fungi-derived dicerandrol A and its inhibitory effects and preliminary mechanism on HepG2 cells. Chin. J. Antibiot. 2022, 47, 481–487. [Google Scholar]
- Ramos, G.D.C.; Silva-Silva, J.V.; Watanabe, L.A.; Siqueira, J.E.d.S.; Almeida-Souza, F.; Calabrese, K.S.; Marinho, A.M.D.R.; Marinho, P.S.B.; de Oliveira, A.S. Phomoxanthone A, compound of endophytic fungi Paecilomyces sp. and its potential antimicrobial and antiparasitic. Antibiotics 2022, 11, 1332. [Google Scholar] [CrossRef]
- Rezanka, T.; Sigler, K. Hirtusneanoside, an unsymmetrical dimeric tetrahydroxanthone from the lichen Usnea hirta. J. Nat. Prod. 2007, 70, 1487–1491. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Qi, X.; Mo, X.; Yu, G.; Wang, Q.; Zhu, T.; Gu, Q.; Liu, M.; Li, J.; Li, D. Structure-based discovery of cytotoxic dimeric tetrahydroxanthones as potential topoisomerase I inhibitors from a marine-derived fungus. Eur. J. Med. Chem. 2018, 148, 268–278. [Google Scholar] [CrossRef] [PubMed]
- Isaka, M.; Palasarn, S.; Kocharin, K.; Saenboonrueng, J. A cytotoxic xanthone dimer from the entomopathogenic fungus Aschersonia sp. BCC 8401. J. Nat. Prod. 2005, 68, 945–946. [Google Scholar] [CrossRef] [PubMed]
- Chutrakul, C.; Boonruangprapa, T.; Suvannakad, R.; Isaka, M.; Sirithunya, P.; Toojinda, T.; Kirtikara, K. Ascherxanthone B from Aschersonia luteola, a new antifungal compound active against rice blast pathogen Magnaporthe grisea. J. Appl. Microbiol. 2009, 107, 1624–1631. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.Y.; Yi, C.; Sun, S.F.; Li, Y.; Liu, Y.-B. Anti-inflammatory dimeric tetrahydroxanthones from an endophytic Muyocopron laterale. J. Nat. Prod. 2022, 85, 148–161. [Google Scholar] [CrossRef]
- Stewart, M.; Capon, R.J.; White, J.M.; Lacey, E.; Tennant, S.; Gill, J.H.; Shaddock, M.P. Rugulotrosins A and B: two new antibacterial metabolites from an Australian isolate of a Penicillium sp. J. Nat. Prod. 2004, 67, 728–730. [Google Scholar] [CrossRef]
- Ondeyka, J.G.; Dombrowski, A.W.; Polishook, J.P.; Felcetto, T.; Shoop, W.L.; Guan, Z.; Singh, S.B. Isolation and insecticidal/anthelmintic activity of xanthonol, a novel bis-xanthone, from a non-sporulating fungal species. J. Antibiot. 2006, 59, 288–292. [Google Scholar] [CrossRef]
- Greco, C.; Mattos-Shipley, K.D.; Bailey, A.M.; Mulholland, N.P.; Vincent, J.L.; Willis, C.L.; Cox, R.J.; Simpson, T.J. Structure revision of cryptosporioptides and determination of the genetic basis for dimeric xanthone biosynthesis in fungi. Chem. Sci. 2019, 10, 2930–2939. [Google Scholar] [CrossRef] [PubMed]
- Wei, P.Y.; Liu, L.X.; Liu, T.; Chen, C.; Luo, D.Q.; Shi, B.Z. Three new pigment protein tyrosine phosphatases inhibitors from the insect parasite fungus Cordyceps gracilioides: Terreusinone A, pinophilin C and cryptosporioptide A. Molecules 2015, 20, 5825–5834. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.H.; Li, H.X.; Wu, P.; Xu, L.; Yuan, Y.; Wei, X. Bioactive polyhydroxanthones from Penicillium purpurogenum. J. Nat. Prod. 2020, 83, 1480–1487. [Google Scholar] [CrossRef] [PubMed]
- Qin, T.; Iwata, T.; Ransom, T.T.; Beutler, J.A.; Porco, J.J.A. Syntheses of dimeric tetrahydroxanthones with varied linkages: Investigation of “shapeshifting” properties. J. Am. Chem. Soc. 2015, 137, 15225–15233. [Google Scholar] [CrossRef] [PubMed]
- Koolen, H.H.; Menezes, L.S.; Souza, M.P.; Silva, F.; Almeida, F.G.; de Souza, A.Q.; Nepel, A.; Barison, A.; Silva, F.H.; Evangelista, D.E.; et al. Talaroxanthone, a novel xanthone dimer from the endophytic fungus talaromyces sp. associated with Duguetia stelechantha (diels) R. E. Fries (Article). J. Braz. Chem. Soc. 2013, 5, 880–883. [Google Scholar]
- Isaka, M.; Jaturapat, A.; Rukseree, K.; Danwisetkanjana, K.; Tanticharoen, M.; Thebtaranonth, Y. Phomoxanthones A and B, novel xanthone dimers from the endophytic fungus Phomopsis Species. J. Nat. Prod. 2001, 64, 1015–1018. [Google Scholar] [CrossRef] [PubMed]
- Shiono, Y.; Sasaki, T.; Shibuya, F.; Yasuda, Y.; Koseki, T.; Supratman, U. Isolation of a phomoxanthone A derivative, a new metabolite of tetrahydroxanthone, from a Phomopsis sp. isolated from the mangrove, Rhizhopora mucronata. Nat. Prod. Commun. 2013, 8, 1735–1737. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.W.; Chen, D.D.; Li, Q.; Feng, T.; Xu, J. Metabolomics-guided discovery of new dimeric xanthones from co-cultures of mangrove endophytic fungi Phomopsis asparagi DHS-48 and Phomopsis sp. DHS-11. Mar. Drugs 2024, 22, 102. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Li, Y.P.; Li, X.X.; Lu, Z.H.; Zheng, Q.H.; Liu, Q.Y. Isolation of 4,4′-bond secalonic acid D from the marine-derived fungus Penicillium oxalicum with inhibitory property against hepatocellular carcinoma. J. Antibiot. 2019, 72, 34–44. [Google Scholar] [CrossRef]
- Tan, S.Y.; Yang, B.; Liu, J.; Xun, T.; Liu, Y.; Zhou, X. Penicillixanthone A, a marine-derived dual-coreceptor antagonist as anti-HIV-1 agent. Nat. Prod. Res. 2019, 33, 1467–1471. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Zhao, Z.Y.; Dong, Q.; Gao, X.; Xu, H.; Yang, R.; Qin, J.C.; Luo, D. Anti-tumor activity and underlying mechanism of phomoxanthone B in MCF7 cells. Anti-Cancer Agents Med. Chem. 2021, 21, 1825–1834. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.G.; McMillin, D.W.; Tamayo, G.; Delmore, J.; Mitsiades, C.S.; Clardy, J. Inhibition of tumor cells interacting with stromal cells by xanthones isolated from a Costa Rican Penicillium sp. J. Nat. Prod. 2012, 75, 793–797. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, Y.; Gotfredsen, C.H.; Larsen, T.O. Genetic characterization of neosartorin biosynthesis provides insight into heterodimeric natural product generation. Org. Lett. 2018, 20, 7197–7200. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.K.; Nguyen-Si, H.V.; Devi, A.P.; Poonsukkho, P.; Sangvichien, E.; Tran, T.-N.; Yusuke, H.; Mitsunaga, T.; Chavasiri, W. Eumitrins F-H: Three new xanthone dimers from the lichen Usnea baileyi and their biological activities. Nat. Prod. Res. 2023, 37, 1480–1490. [Google Scholar] [CrossRef]
- Nguyen, V.; Genta-Jouve, G.; Duong, T.; Beniddir, M.A.; Gallard, J.-F.; Ferron, S.; Boustie, J.; Mouray, E.; Grellier, P.; Chavasiri, W.; et al. Eumitrins C-E: Structurally diverse xanthone dimers from the vietnamese lichen Usnea baileyi. Fitoterapia 2020, 141, 104449. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.M.; Takeda, N.; Iitaka, Y.; Sankawa, V.; Shibata, S. The structures of eumitrins A1, A2 and B: The yellow pigments of the lichen, Usnea bayleyi (Stirt.) Zahlbr. Tetrahedron 1973, 29, 519–528. [Google Scholar] [CrossRef]
- Wang, F.; Jiang, J.; Hu, S.; Hao, X.; Cai, Y.S.; Ye, Y.; Ma, H.; Sun, W.; Cheng, L.; Huang, C.; et al. Nidulaxanthone A, a xanthone dimer with a heptacyclic 6/6/6/6/6/6/6 ring system from Aspergillus sp. F029. Org. Chem. Front. 2020, 7, 953–959. [Google Scholar] [CrossRef]
- Zhang, Y.; Fu, P.; Zhang, Y.; Xu, Y.; Zhang, C.; Liu, X.; Che, Y. Cladoxanthones A and B, Xanthone-derived metabolites with a spiro [cyclopentane-1,2′-[3,9a]ethanoxanthene]-2,4′,9′,11′-tetraone skeleton from a Cladosporium sp. J. Nat. Prod. 2022, 85, 2541–2546. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh, S.K.; Mishra, P.D.; Kulkarni-Almeida, A.; Verekar, S.; Sahoo, M.R.; Periyasamy, G.; Goswami, H.; Khanna, A.; Balakrishnan, A.; Vishwakarma, R. Anti-inflammatory and anticancer activity of ergoflavin isolated from an endophytic fungus. Chem. Biodivers. 2009, 6, 784–789. [Google Scholar] [CrossRef] [PubMed]
- Castelão, J.F., Jr.; Gottlieb, O.R.; De Lima, R.A.; Mesquita, A.A.; Gottliebb, H.E.; Wenkert, E. Xanthonolignoids from Kielmeyera and Caraipa species—13C NMR spectroscopy of xanthones. Phytochemistry 1977, 16, 735–740. [Google Scholar] [CrossRef]
- Abou-shoer, M.; Habib, A.Z.; Chang, C.J.; Cassady, J.M. Seven xanthonolignoids from Psorospermum febrifugum. Phytochemistry 1989, 28, 2483–2487. [Google Scholar] [CrossRef]
- Iinuma, M.; Tosa, H.; Ito, T.; Tanaka, T.; Riswan, S. Three new benzophenone-xanthone dimers from the root of Garcinia dulcis. Chem. Pharm. Bull. 1996, 44, 1744–1747. [Google Scholar] [CrossRef][Green Version]
- Chen, L.; Bi, Y.X.; Li, Y.P.; Bi, Y.-X.; Li, Y.-P.; Li, X.-X.; Liu, Q.-Y.; Ying, M.-G.; Zheng, Q.-H. Secalonic Acids H and I, two new secondary metabolites from the marine-derived fungus Penicillium oxalicum. Heterocycles 2017, 94, 1766–1774. [Google Scholar]
- Schüffler, A.; Liermann, J.C.; Kolshorn, H.; Opatz, T.; Anke, H. Isolation, structure elucidation, and biological evaluation of the unusual heterodimer chrysoxanthone from the ascomycete IBWF11-95A. Tetrahedron Lett. 2009, 50, 4813–4815. [Google Scholar] [CrossRef]
- Zhang, W.; Krohn, K.; Flörke, U.; Pescitelli, G.; Di Bari, L.; Antus, S.; Kurtán, T.; Rheinheimer, J.; Draeger, S.; Schulz, B. New mono- and dimeric members of the secalonic acid Family: Blennolides A–G isolated from the fungus Blennoria sp. Chemistry 2008, 14, 4913–4923. [Google Scholar] [CrossRef]
- Nguyen, V.K.; Dong, P.S.; Nguyen-Si, H.V.; Sangvichien, E.; Tran, T.-N.; Hoang, L.-T.; Dao, M.-T.; Nguyen, H.; Phan, H.-V.; Yusuke, H.; et al. Eumitrins I-K: Three new xanthone dimers from the lichen Usnea baileyi. J. Nat. Med. 2023, 77, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Aberhart, D.J.; De Mayo, P. Mould metabolites—V: The constitution of ergoxanthin. Tetrahedron 1966, 22, 2359–2366. [Google Scholar] [CrossRef]
- Yang, J.X.; Xu, F.; Huang, C.H.; Li, J.; She, Z.; Pei, Z.; Lin, Y. Metabolites from the mangrove endophytic fungus Phomopsis sp. (#zsu-H76). Eur. J. Org. Chem. 2010, 2010, 3692–3695. [Google Scholar]
- Pontius, A.; Krick, A.; Mesry, R.; Kehraus, S.; Foegen, S.E.; Müller, M.; Klimo, K.; Gerhäuser, C.; König, G.M. Monodictyochromes A and B, dimeric xanthone derivatives from the marine algicolous fungus Monodictys putredinis. J. Nat. Prod. 2008, 71, 1793–1799. [Google Scholar] [CrossRef]
- Xu, Y.J.; Cao, S.G.; Wu, X.H.; Lai, Y.-H.; Tan, B.; Pereira, J.; Goh, S.; Venkatraman, G.; Harrison, L.J.; Sim, K.-Y. Griffipavixanthone, a novel cytotoxic bixanthone from Garcinia griffithii and G. pavifolia. Tetrahedron Lett. 1998, 39, 9103–9106. [Google Scholar] [CrossRef]
- Zhao, F.N.; Niu, Q.; Xiao, D.; Xu, H.-N.; Wang, H.-X.; Bi, R.-L.; He, H.-P.; Jiang, Z.-Y. A new xanthone from hulls of Garcinia mangostana and its cytotoxic activity. China J. Chin. Mater. Medica 2023, 48, 5817–5821. [Google Scholar]
- Zhai, A.F.; Zhang, Y.; Zhu, X.N.; Liang, J.; Wang, X.; Lin, Y.; Chen, R. Secalonic acid A reduced colchicine cytotoxicity through suppression of JNK, p38 MAPKs and calcium influx. Neurochem. Int. 2011, 58, 85–91. [Google Scholar] [CrossRef]
- Zhai, A.F.; Zhu, X.N.; Wang, X.L.; Chen, R.; Wang, H. Secalonic acid A protects dopaminergic neurons from 1-methyl-4-phenylpyridinium (MPP (+))-induced cell death via the mitochondrial apoptotic pathway. Eur. J. Pharmacol. 2013, 713, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Dominique, S.; Alex, P.G.; Christiane, E.Y.; Dodehe, Y.; Adèle, K.N. Diversity of endophytic fungi isolated from the bark of Ceiba pentandra (L.) Gaertn., (Bombacaceae) and antibacterial potential of Secalonic Acid A produced by Diaporthe searlei EC321. Chem. Biodivers. 2023, 20, e202301010. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.Y.; Tao, L.Y.; Liang, Y.J.; Yan, Y.Y.; Dai, C.L.; Xia, X.K.; She, Z.G.; Lin, Y.C.; Fu, L.W. Secalonic acid D induced leukemia cell apoptosis and cell cycle arrest of G (1) with involvement of GSK-3beta/beta-catenin/c-Myc pathway. Cell Cycle 2009, 8, 2444–2450. [Google Scholar] [CrossRef] [PubMed]
- Hong, R. Secalonic acid D as a novel DNA topoisomerase I inhibitor from marine lichen-derived fungus Gliocladium sp. T31. Pharm. Biol. 2011, 49, 796–799. [Google Scholar] [CrossRef]
- Hu, Y.P.; Tao, L.Y.; Wang, F.; Zhang, J.Y.; Liang, Y.J.; Fu, L.W. Secalonic acid D reduced the percentage of side populations by down-regulating the expression of ABCG2. Biochem. Pharmacol. 2013, 85, 1619–1625. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Huang, L.Y.; Tao, L.Y.; Zhang, J.; Wang, F.; Zhang, X.; Fu, L. Secalonic acid D induces cell apoptosis in both sensitive and ABCG2-overexpressing multidrug resistant cancer cells through upregulating c-Jun expression. Acta Pharm. Sin. B 2019, 9, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, P.; Tang, X.X.; Yi, Z.W.; Qiu, Y.K.; Wu, Z. Two new compounds from marine-derived fungus Penicillium sp. F11. J. Asian Nat. Prod. Res. 2012, 14, 197–203. [Google Scholar] [CrossRef]
- Lim, C.; Kim, J.; Choi, J.N.; Ponnusamy, K.; Jeon, Y.; Kim, S.-U.; Kim, J.G.; Lee, C. Identification, fermentation, and bioactivity against Xanthomonas oryzae of antimicrobial metabolites isolated from Phomopsis longicolla S1B4. J. Microbiol. Biotechnol. 2010, 20, 494–500. [Google Scholar] [PubMed]
- Zhou, D.D.; Chen, D.D.; Wu, J.W.; Feng, T.; Liu, P.; Xu, J. Dicerandrol C suppresses proliferation and induces apoptosis of HepG2 and Hela cancer cells by inhibiting Wnt/β-catenin signaling pathway. Mar. Drugs 2024, 22, 278. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.X.; Xing, S.P.; Wei, X.X.; Lu, J.; Zhao, G.; Ma, X.; Dai, Z.; Liang, X.; Huang, W.; Liu, Y.; et al. 12-O-deacetyl-phomoxanthone A inhibits ovarian tumor growth and metastasis by downregulating PDK4. Biomed. Pharmacother. 2024, 175, 116736. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Liu, Y.; Wei, X.; Yang, C.; Lu, J.; Yan, S.; Ma, X.; Cheng, X.; You, Z.; Ding, Y.; et al. Identification of penexanthone A as a novel chemosensitizer to induce ferroptosis by targeting Nrf2 in human colorectal cancer cells. Mar. Drugs 2024, 22, 357. [Google Scholar] [CrossRef]
- Carlton, W.W.; Tuite, J.; Mislivec, P. Investigations of the toxic effects in mice of certain species of Penicillium. Toxicol. Appl. Pharmacol. 1968, 13, 372–387. [Google Scholar] [CrossRef]


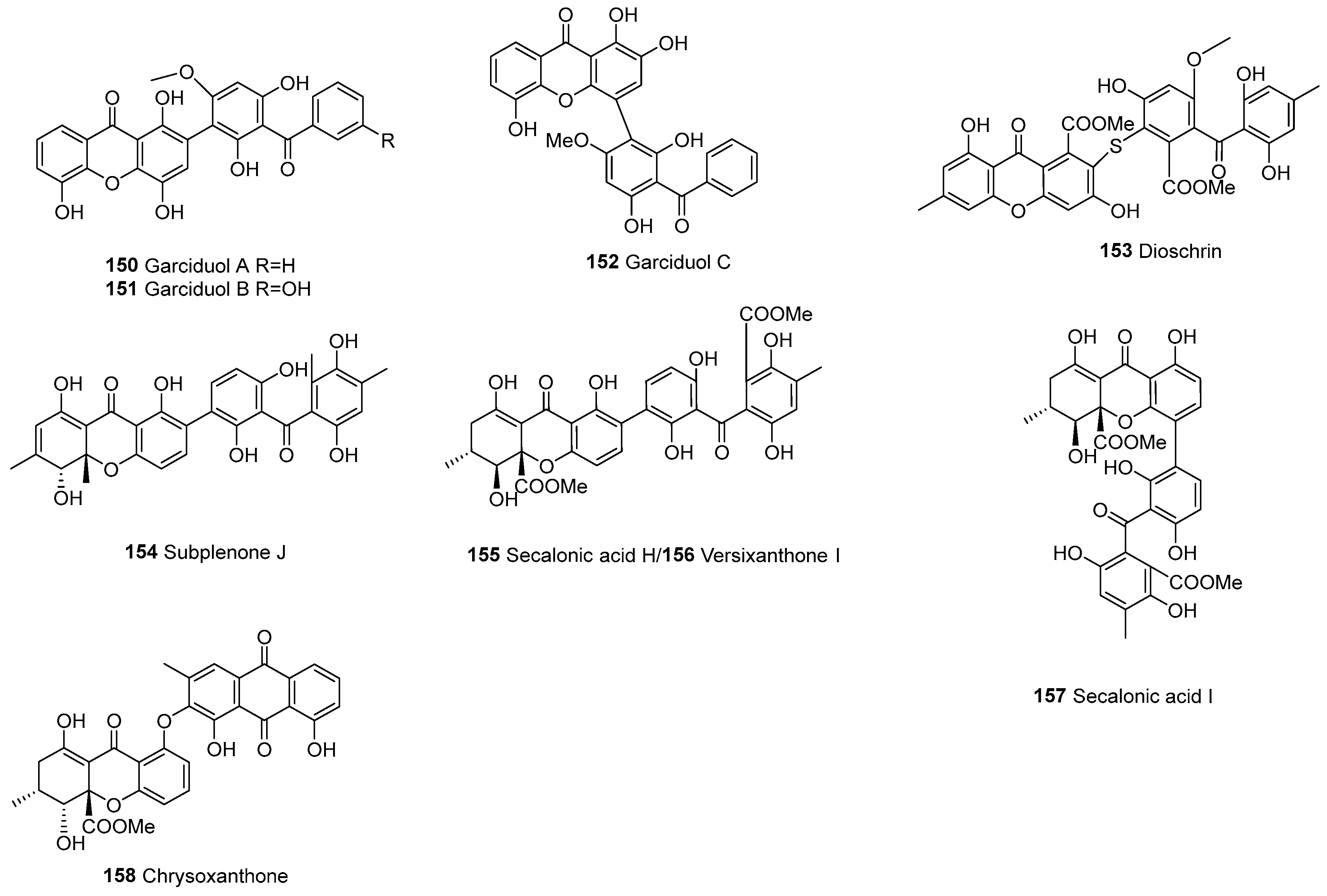

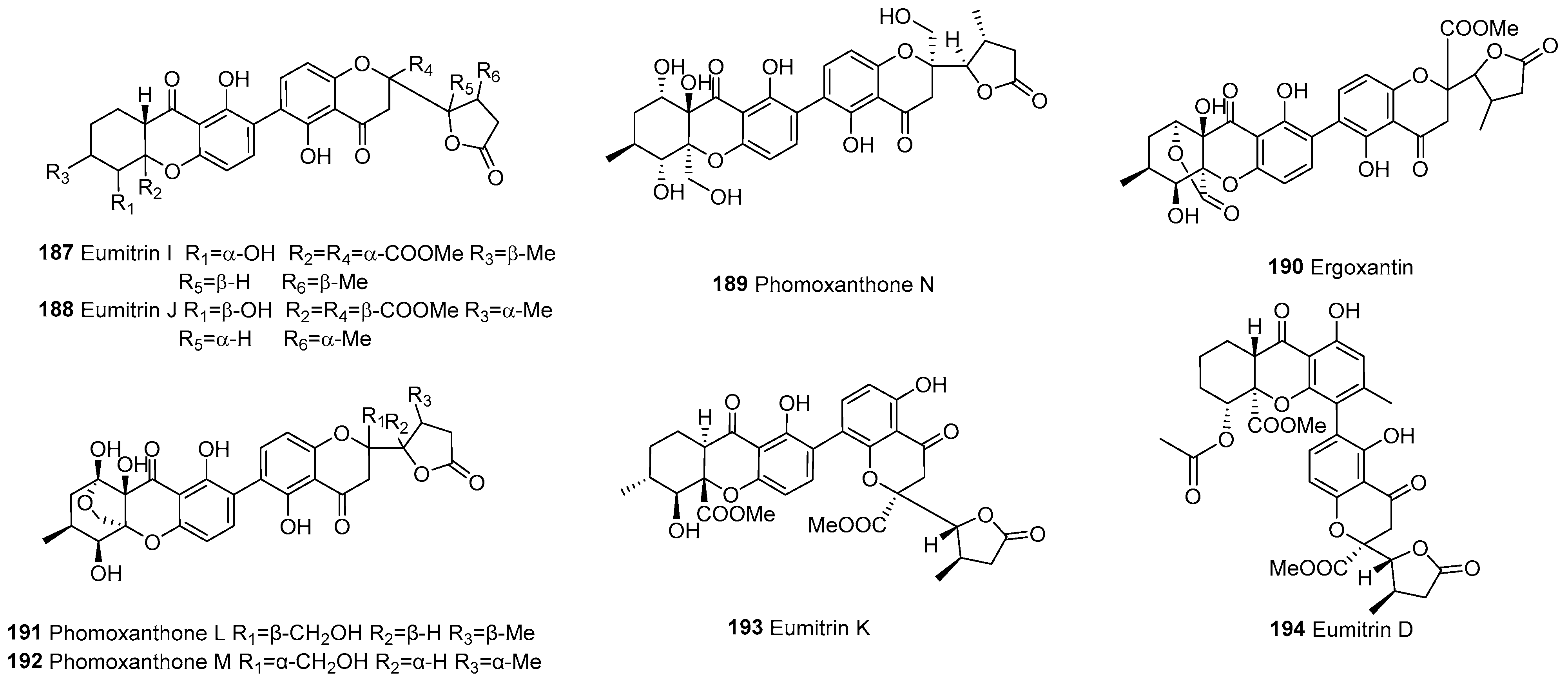
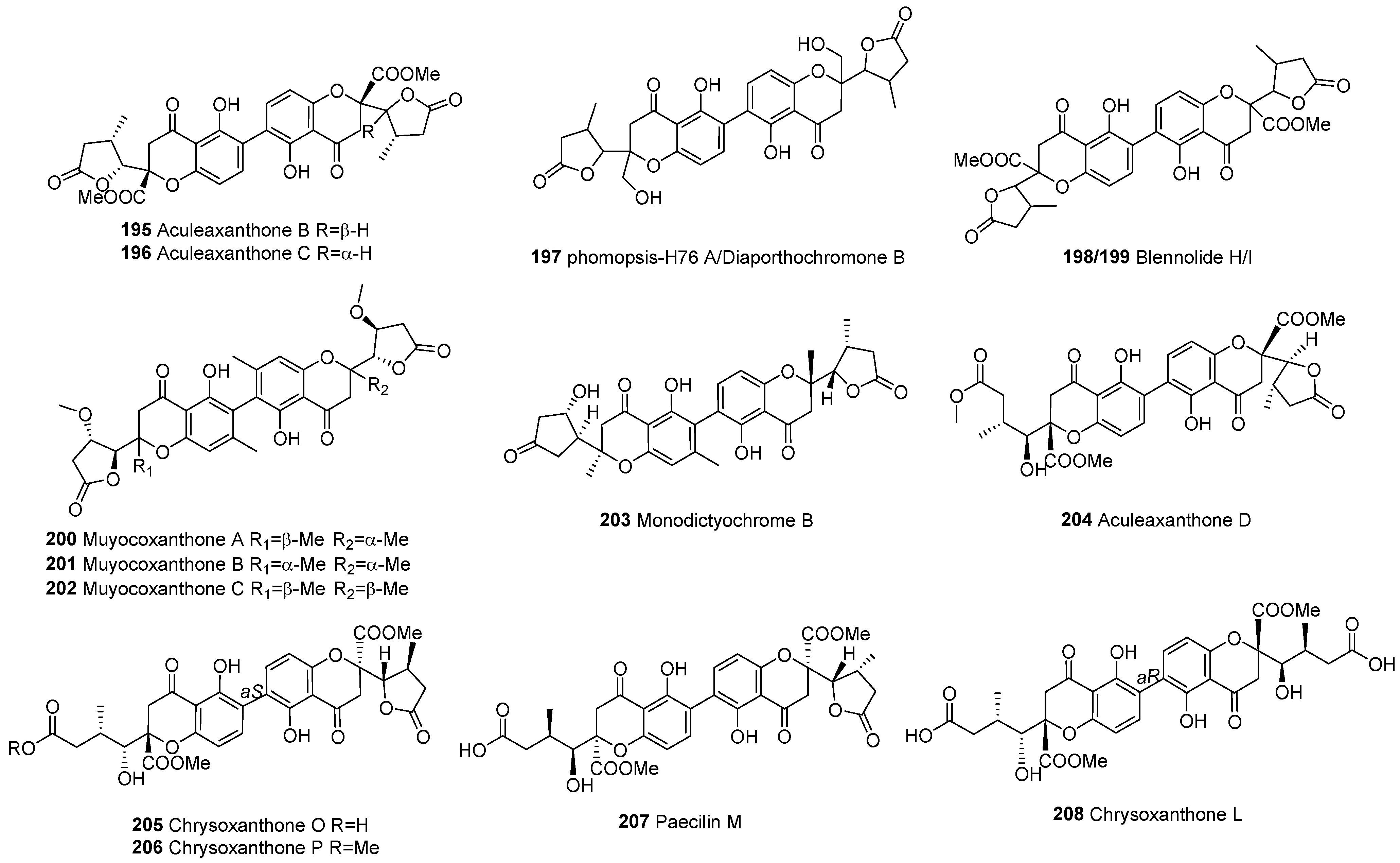
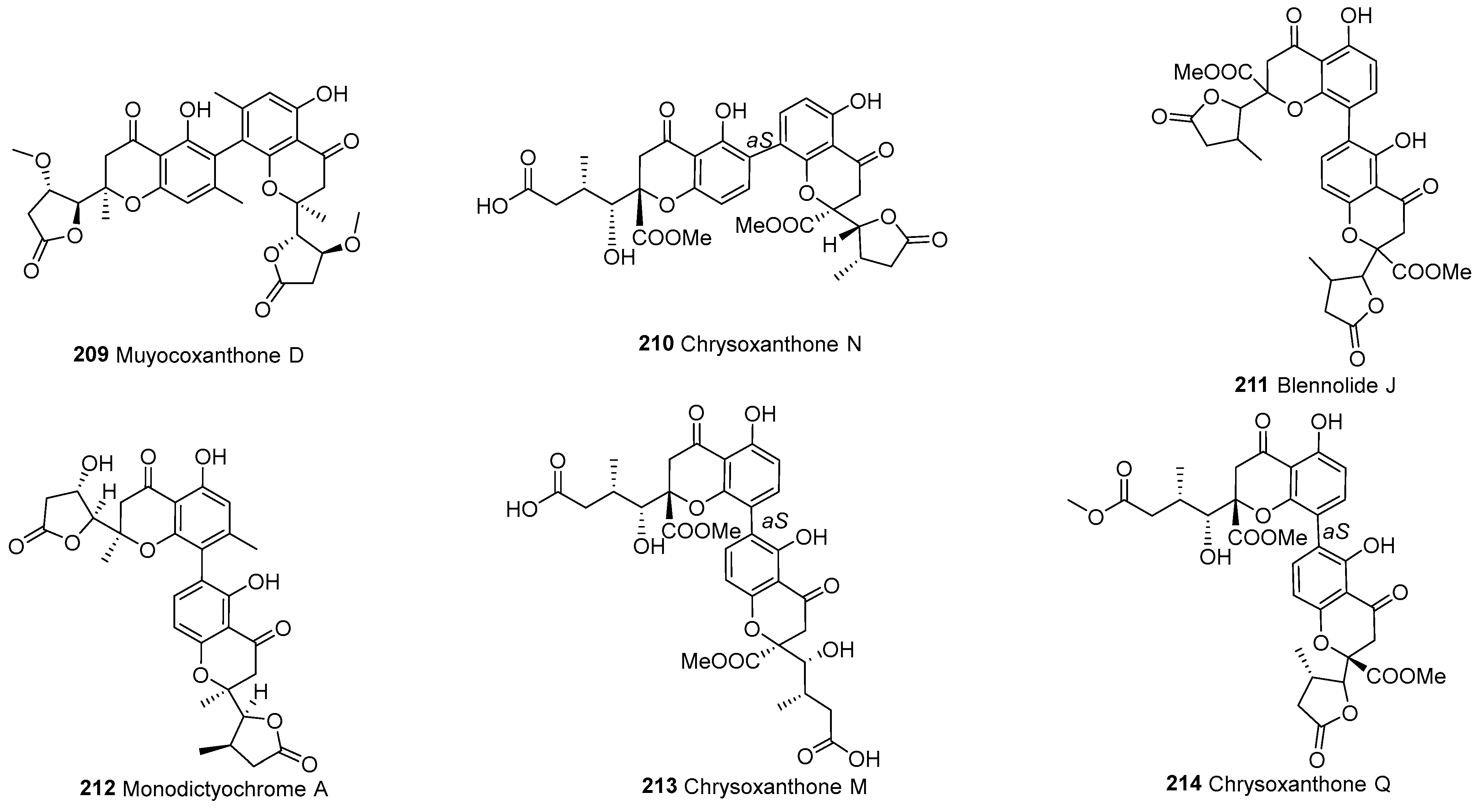
| Division | Class | Family | Genus | Number of Species |
|---|---|---|---|---|
| Plant | ||||
| Angiosperm | Dicotyledoneae | Clusiaceae | Garcinia, Mesua, Symphonia, Cratoxylum, Caraipa | 13 |
| Hypericaceae | Hypericum | 4 | ||
| Calophyllaceae | Calophyllum | 1 | ||
| Anacardiaceae | Mangifera | 1 | ||
| Gentianaceae | Swertia, Gentianella | 4 | ||
| Lichen | Basidiolichenes | Ramalinaceae | Usnea | 2 |
| Fungi | ||||
| Ascomycota | Eurotiomycetes | Trichocomaceae | Aspergillus, Talaromyces | 14 |
| Sordariomycetes | Diaporthaceae | Diaporthe, Aschersonia | 2 | |
| Sordariaceae | Neurospora | 1 | ||
| Leotiomycetes | Dermateaceae | Cryptosporiopsis | 1 | |
| Ascomycetes | Leptosphaeriaceae | Subplenodomus | 1 | |
| Dothideomycetes | Muyocopronaceae | Muyocopron | 1 | |
| Blennoriaceae | Blennoria. | 1 | ||
| Phaeosphaeriaceae | Setophoma | 1 | ||
| Ascomycotina | Plectomycetes | Eurotiaceae | Penicillium | 8 |
| Pyrenomycetes | Clavicipitaceae | Claviceps | 1 | |
| Basidiomycota | Agaricomycetes | Peniophoraceae | Peniophora | 1 |
| Deuteromycotina | Coelomycetes | Sphaeropsidales | Phoma, Phomopsis | 9 |
| Incertae sedis | Pyrenochaeta | 1 | ||
| Dothidea | Dermateaceae | Alternaria, Cladosporium | 2 | |
| Hyphomycetes | Tubeufiaceae | Monodictys | 1 | |
| Moniliaceae | Paecilomyces | 2 | ||
| Other fungi | Ascomycete IBWF11-95A | 1 | ||
| PM0651480 (isolated as an endophytic fungus from the leaves of Mimosops elengil) | 1 | |||
| Non-sporulating fungus, MF6460 (isolated from leaf litter of Manilkara bidentata) | 1 | |||
| Mangrove endophytic fungus No. ZSU44 | 1 | |||
| Number | Xanthone Type | Monomer Type | Subtype | Compound Name | Molecular Formula | Source (Plant Part) | Ref. |
|---|---|---|---|---|---|---|---|
| 1 | Xanthone (a) | a–a | C1–C1’ | Hyperidixanthone | C30H22O10 | Hypericum chinense (entire plant) | [29] |
| 2 | C2–C2’ | Swertiabixanthone I | C32H23O17 | Swertia franchetiana (entire plant) | [30] | ||
| 3 | Swertiabixanthone II | C27H16O12 | Swertia pseudochinensis (entire plant) | [31] | |||
| 4 | Swertiabisxanthone I 8′-O-D-glucopyranoside | C32H24O17 | Swertia punicea, Gentianella amarella ssp. acuta (entire plant) | [23,32] | |||
| 5 | C2–C4’ | Swertipunicoside | C33H26O17 | S. franchetiana (entire plant) | [33] | ||
| 6 | 3-O-demethylswertipunicoside | C32H24O17 | S. franchetiana (entire plant) | [23] | |||
| 7 | C4–C2’ | Puniceaside A | C32H24O17 | S. punicea (entire plant) | [23] | ||
| 8 | C4–C4’ | Mangiferoxanthone A | C38H34O22 | Mangifera indica (stem bark) | [34] | ||
| 9 | C8–C8’ | Ploiarixanthone | C26H14O9 | Ploiartium alternifolium (branches) | [35] | ||
| 10 | Complex | Bigarciculenxanthone A | C46H46O13 | Garcinia esculenta (twigs and leaves) | [36] | ||
| 11 | Bigarciculenxanthone B | C46H46O13 | G. esculenta (twigs and leaves) | [36] | |||
| 12 | Bigarciculenxanthone C | C46H44O12 | G. esculenta (twigs and leaves) | [36] | |||
| 13 | Bigarcinenone B | C36H28O11 | Garcinia xanthochymus (bark) | [37] | |||
| 14 | Biscaloxanthone | C47H48O10 | Calophyllum canum (stem bark) | [38] | |||
| 15 | Globulixanthone E | C37H30O9 | Symphonia globulifera (root bark) | [39] | |||
| 16 | Garcinoxanthone A | C48H50O13 | Garcinia mangostana (pericarp) | [40] | |||
| 17 | Garcinoxanthone B | C48H50O13 | G. mangostana (pericarp) | [40] | |||
| 18 | Garcinoxanthone C | C48H47O13 | G. mangostana (pericarp) | [40] | |||
| 19 | Cratoxyxanthone | C48H50O13 | G. mangostana (bark) | [41] | |||
| 20 | Bigarcinenone A | C56H62O13 | G. xanthochymus (bark) | [22,42] | |||
| 21 | Bixanthone C | C31H21O12 | Hypericum japonicum (entire plant) | [27] | |||
| 22 | Bixanthone D | C36H29O12 | H. japonicum (entire plant) | [27] | |||
| 23 | Bijaponicaxanthone | C36H28O13 | H. japonicum, Hypericum henryi (aerial parts) | [43] | |||
| 24 | Jacarelhyperol A | C36H28O13 | H. japonicum (aerial parts) | [44] | |||
| 25 | Jacarelhyperol B | C36H28O12 | H. japonicum (aerial parts) | [44] | |||
| 26 | Jacarelhyperol D | C31H21O13 | H. japonicum (aerial parts) | [45] | |||
| 27 | Bijaponicaxanthone C | C36H30O13 | H. japonicum, Hypericum riparium (entire plant) | [45] | |||
| 28 | Griffipavixanthone | C36H28O12 | Garcinia griffithii, Garcinia pavifolia (bark), G. esculenta (twigs and leaves), Garcinia oblongifolia (bark) | [14,15,25,46] | |||
| 29 | Garmoxanthone | C36H28O12 | G. mangostana (pericarp) | [20] | |||
| 30 | Garcilivin A | C36H28O10 | Garcinia livingstonei (bark) | [47,48] | |||
| 31 | Garcilivin C | C36H28O10 | G. livingstonei (bark) | [47,48] | |||
| 32 | Garciobioxanthone | C36H32O13 | G. oblongifolia (bark) | [49] | |||
| 33 | Mesuabixanthone A | C33H24O12 | Mesua ferrea (stem bark) | [50] | |||
| 34 | Mesuabixanthone B | C34H26O12 | M. ferrea (stem bark) | [50] | |||
| 35 | Mesuferrol A | C32H22O12 | M. ferrea (bark) | [51] | |||
| 36 | Mesuferrol B | C33H24O12 | M. ferrea (bark) | [51] | |||
| 37 | Schomburgkixanthone | C36H34O14 | Garcinia schomburgkiana (twigs) | [52] | |||
| 38 | Garcilivin B | C36H28O10 | G. livingstonei (bark) | [48] | |||
| 39 | a–a | C–O–C | Austradixanthone | C30H22O13 | Aspergillus austroafricanus | [53] | |
| 40 | a–a | C–S–C | Castochrin | C32H21O12 | Alternaria sp. | [54] | |
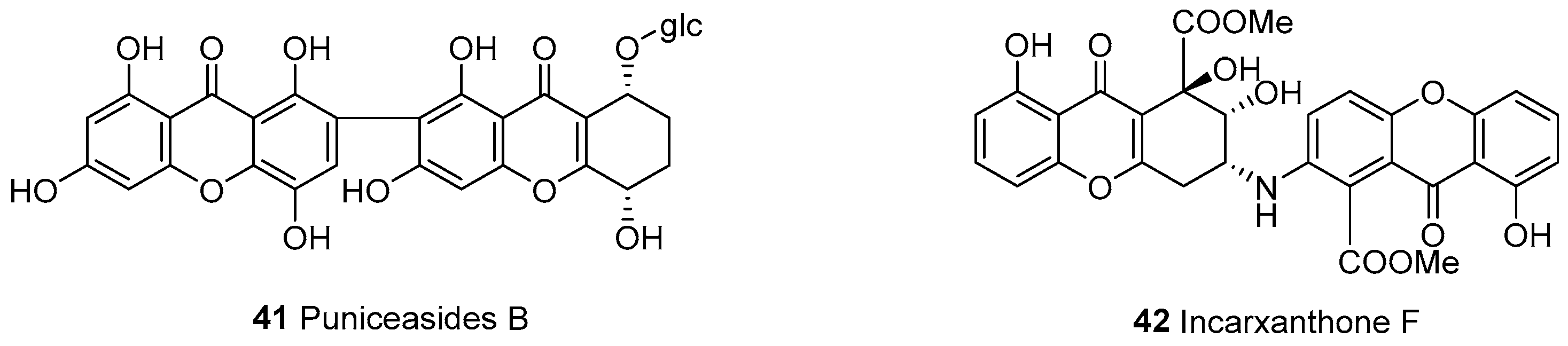
| 41 | Xanthone (a)-tetrahydroxanthone (e) | a–e | C2–C2’ | Puniceaside B | C32H28O17 | S. punicea (entire plant) | [23] |
| 42 | a–e | C–NH–C | Incarxanthone F | C30H23NO12 | Peniophora incarnata Z4 | [55] | |
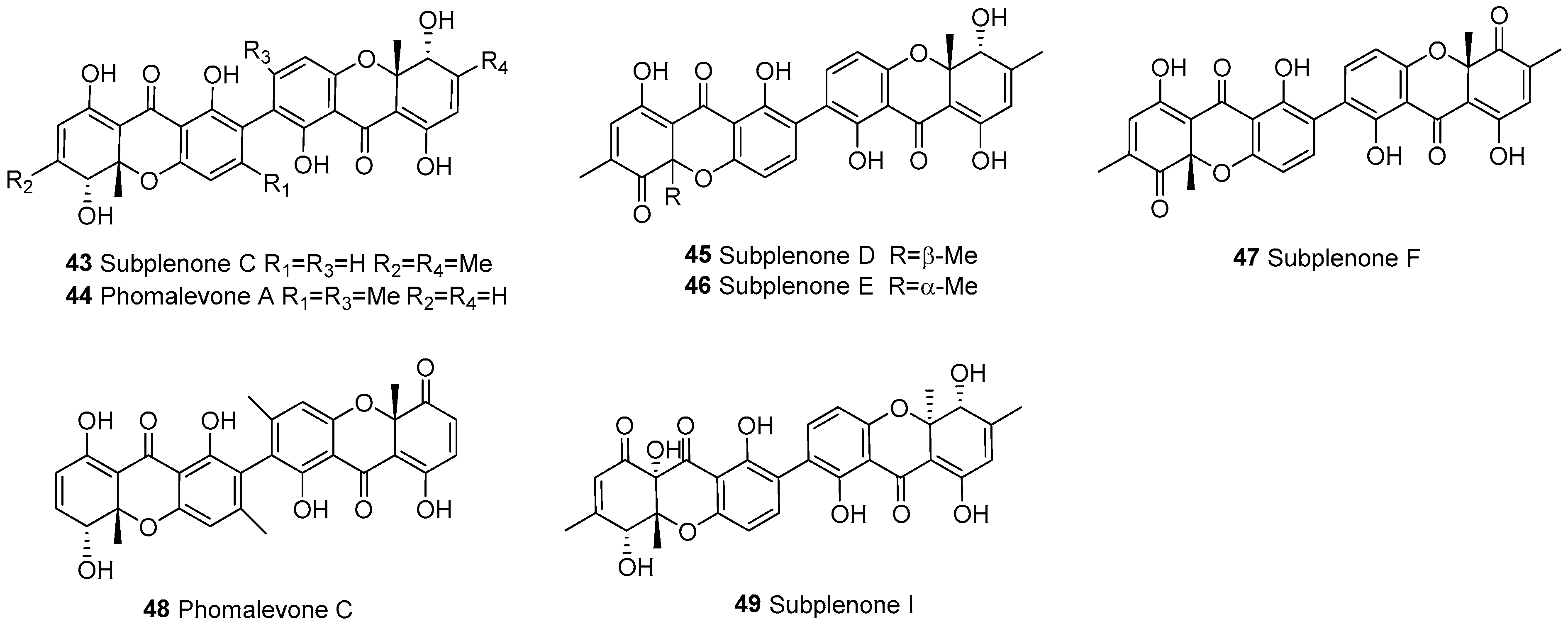
| 43 | Dihydroxanthones (b/c) | b–b | C2–C2’ | Subplenone C | C30H26O10 | Subplenodomus sp. CPCC 401465 | [56] |
| 44 | C2–C2’ | Phomalevone A | C30H26O10 | Phoma sp. | [57] | ||
| 45 | C2–C2’ | Subplenone D | C30H24O10 | Subplenodomus sp. CPCC 401465 | [56] | ||
| 46 | C2–C2’ | Subplenone E | C30H24O10 | Subplenodomus sp. CPCC 401465 | [56] | ||
| 47 | C2–C2’ | Subplenone F | C30H22O10 | Subplenodomus sp. CPCC 401465 | [56] | ||
| 48 | C2–C2’ | Phomalevone C | C30H24O10 | Phoma sp. | [57] | ||
| 49 | C2–C2’ | Subplenone I | C30H26O11 | Subplenodomus sp. CPCC 401465 | [56] | ||
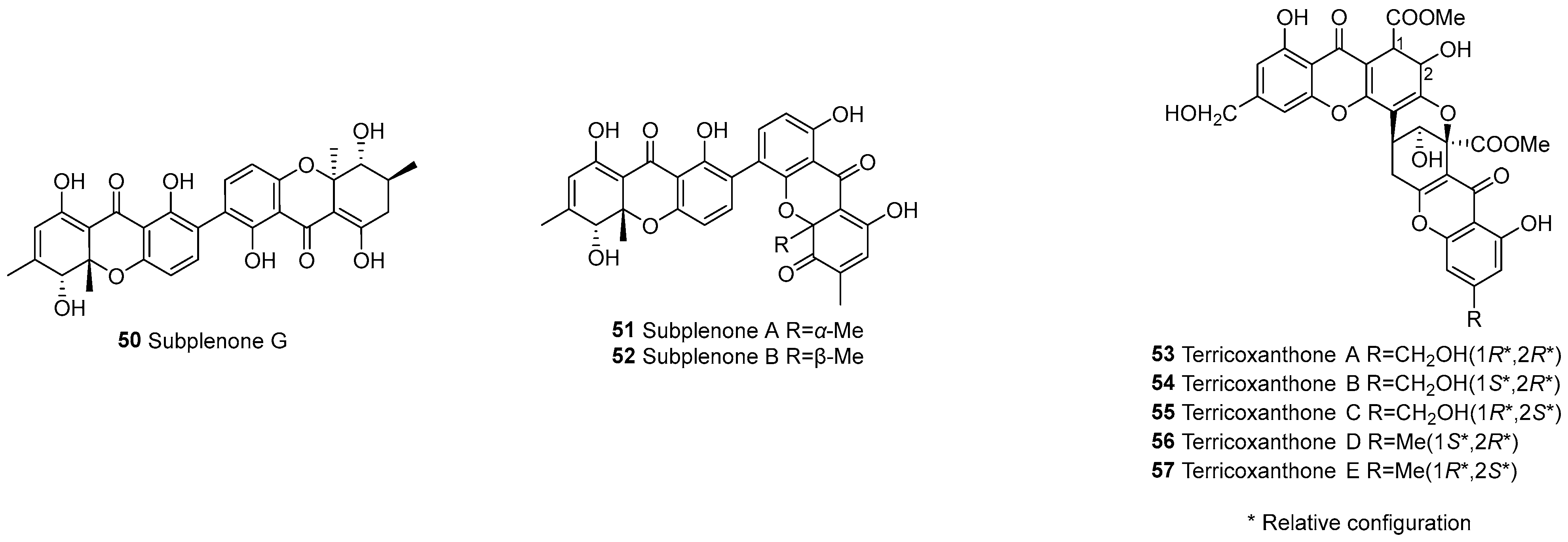
| 50 | b–d | C2–C2’ | Subplenone G | C30H28O10 | Subplenodomus sp. CPCC 401465 | [56] | |
| 51 | C2–C4’ | Subplenone A | C30H24O10 | Subplenodomus sp. CPCC 401465 | [56] | ||
| 52 | C2–C4’ | Subplenone B | C30H24O10 | Subplenodomus sp. CPCC 401465 | [56] | ||
| 53 | c–e | 3,4-epoxy | Terricoxanthone A | C32H26O15 | Neurospora terricola HDF-Br-2 | [58] | |
| 54 | 3,4-epoxy | Terricoxanthone B | C32H26O15 | N. terricola HDF-Br-2 | [58] | ||
| 55 | 3,4-epoxy | Terricoxanthone C | C32H26O15 | N. terricola HDF-Br-2 | [58] | ||
| 56 | 3,4-epoxy | Terricoxanthone D | C32H26O14 | N. terricola HDF-Br-2 | [58] | ||
| 57 | 3,4-epoxy | Terricoxanthone E | C32H26O14 | N. terricola HDF-Br-2 | [58] | ||
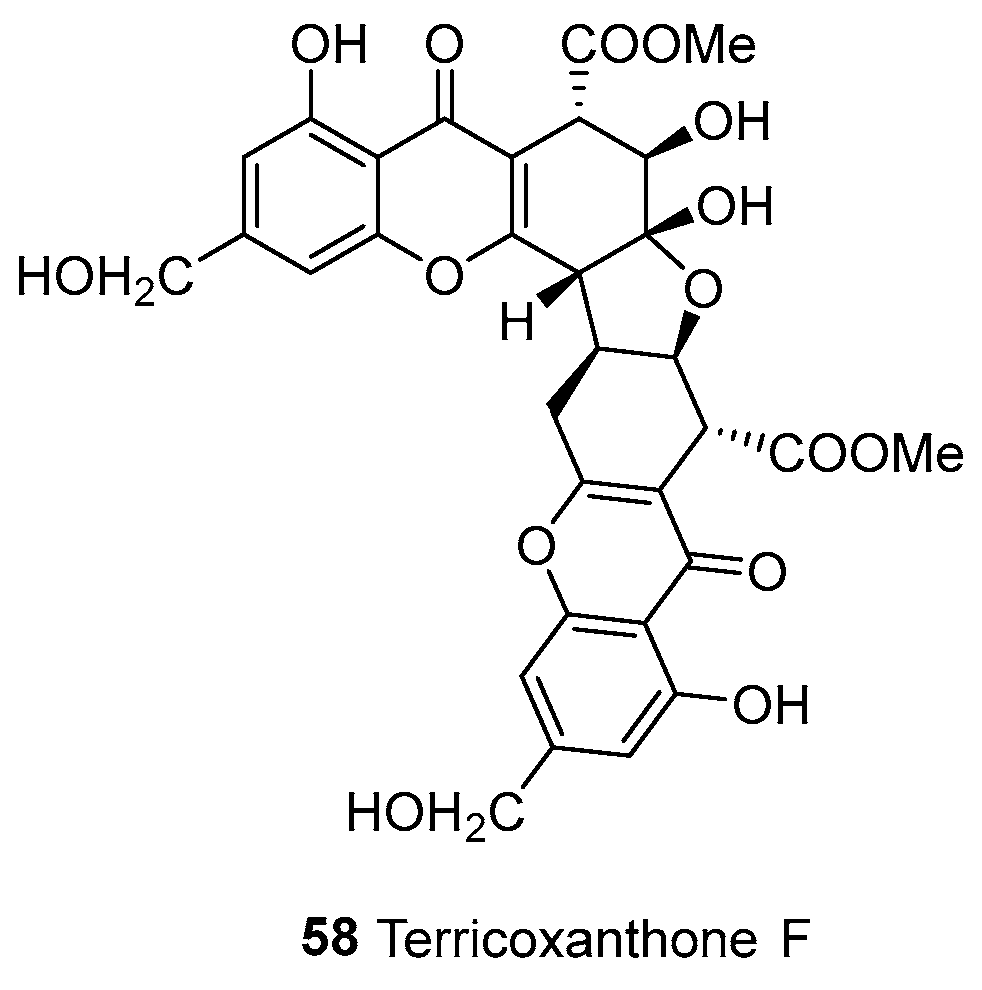
| 58 | Tetrahydroxanthones (d/e) | e–e | Complex | Terricoxanthone F | C32H28O15 | N. terricola HDF-Br-2 | [58] |
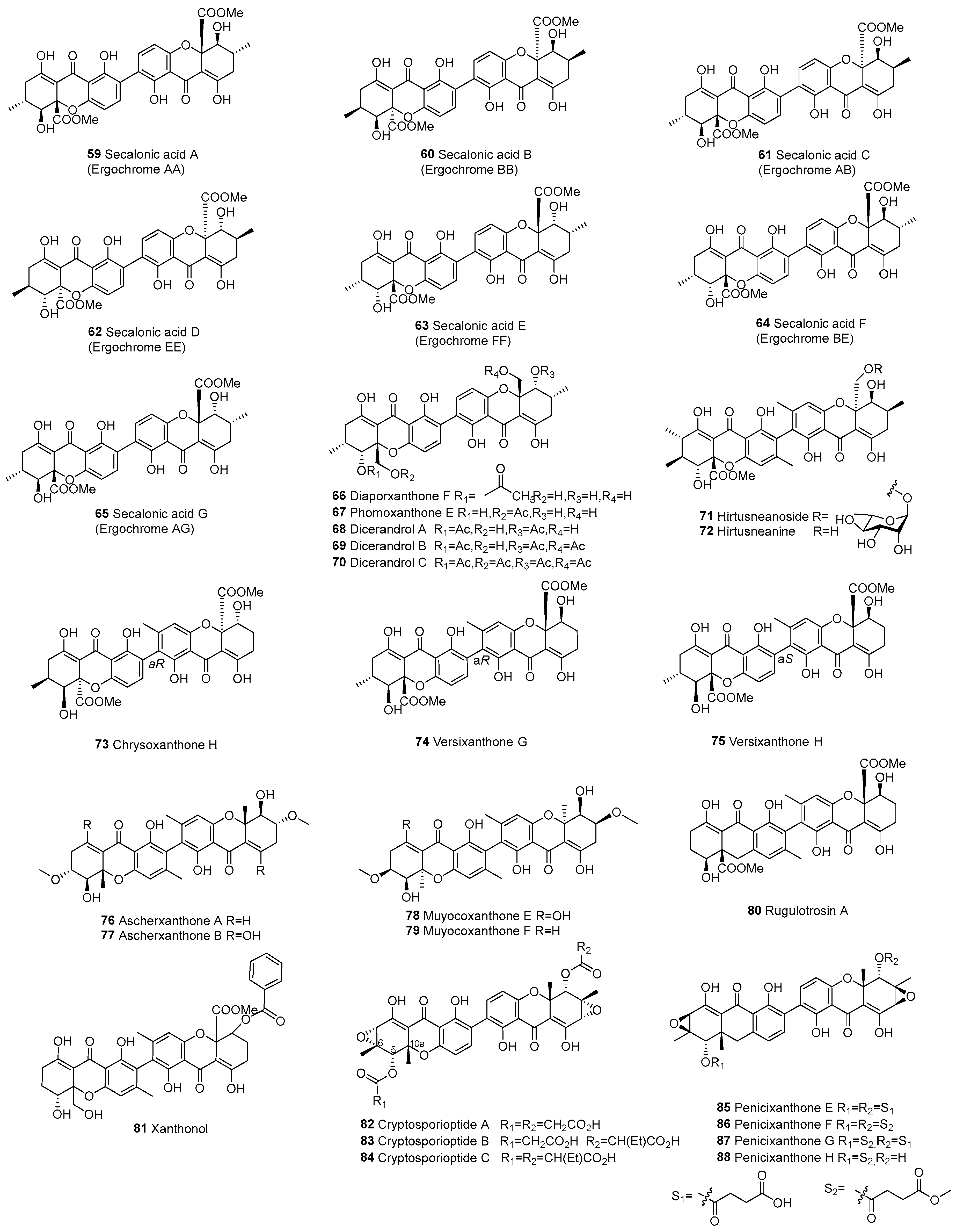
| 59 | d–d | C2–C2’ | Secaionic acid A (Ergochrome AA) | C32H30O14 | Pyrenochaeta terrestris, Penicillium chrysogenum C-7-2-1 | [59,60,61] | |
| 60 | C2–C2’ | Secaionic acid B (Ergochrome BB) | C32H30O14 | Claviceps purpurea | [12,62,63] | ||
| 61 | C2–C2’ | Secaionic acid C (Ergochrome AB) | C32H30O14 | C. purpurea | [64] | ||
| 62 | C2–C2’ | Secalonic acid D (Ergochrome EE) | C32H30O14 | Penicillium sp. F11, Penicillium oxalicum, Aspergillus aculeatus, Aspergillus aculeatinus WHUF0198, Aspergillus sp. TPU1343, C. purpurea the mangrove endophytic fungus No. ZSU44 | [12,16,17,19,62] | ||
| 63 | C2–C2’ | Secalonic acid E (Ergochrome FF) | C32H30O14 | C. purpurea, P. terrestris | [60,61,63,65] | ||
| 64 | C2–C2’ | Secalonic acid F (Ergochrome BE) | C32H30O14 | Aspergillus sp. TPU1343, P. chrysogenum C-7-2-1, A. aculeatus | [17,59,60,62,66,67] | ||
| 65 | C2–C2’ | Secalonic acid G (Ergochrome AG) | C32H30O14 | P. terrestris | [61] | ||
| 66 | C2–C2’ | Diaporxanthone F | C32H32O13 | Diaporthe goulteri L17 | [68] | ||
| 67 | C2–C2’ | Phomoxanthone E | C34H34O14 | Phomopsis sp. xy21; D. goulteri L17 | [68,69] | ||
| 68 | C2–C2’ | Dicerandrol A | C34H34O14 | Phomopsis longicolla, Phomopsis sp. xy21, Phomopsis sp. PSU-D15; Phomopsis asparagi DHS-48 and Phomopsis sp. DHS-11 | [70,71,72,73] | ||
| 69 | C2–C2’ | Dicerandrol B | C36H36O15 | P. longicolla, Phomopsis sp. xy21; Paecilomyces sp, Paecilomyces sp. EJC01.1 | [13,68,71,72,74] | ||
| 70 | C2–C2’ | Dicerandrol C | C38H38O16 | Phomopsis sp. xy21; P. asparagi DHS-48 and Phomopsis sp. DHS-11 | [71,72] | ||
| 71 | C2–C2’ | Hirtusneanoside | C40H46O17 | Usnea hirta (whole thallus) | [75] | ||
| 72 | C2–C2’ | Hirtusneanine | C34H36O13 | U. hirta (whole thallus) | [75] | ||
| 73 | C2–C2’ | Chrysoxanthone H | C32H30O14 | P. chrysogenum C-7-2-1 | [59] | ||
| 74 | C2–C2’ | Versixanthone G | C32H31O14 | Aspergillus versicolor HDN1009 | [76] | ||
| 75 | C2–C2’ | Versixanthone H | C32H31O14 | A. versicolor HDN1009 | [76] | ||
| 76 | C2–C2’ | Ascherxanthone A | C32H34O10 | Aschersonia sp. BCC 8401 | [77] | ||
| 77 | C2–C2’ | Ascherxanthone B | C32H34O12 | Aschersonia luteola BCC 8774 | [78] | ||
| 78 | C2–C2’ | Muyocoxanthone E | C32H34O12 | Muyocopron laterale | [79] | ||
| 79 | C2–C2’ | Muyocoxanthone F | C32H34O12 | M. laterale | [79] | ||
| 80 | C2–C2’ | Rugulotrosin A | C32H30O14 | Penicillium sp. | [12,80] | ||
| 81 | C2–C2’ | Xanthonol | C38H34O14 | Non-sporulating fungus, MF6460 (isolated from leaf litter of Manilkara bidentata) | [81] | ||
| 82 | C2–C2’ | Cryptosporioptide A | C38H30O18 | Cryptosporiopsis sp. 8999 | [82,83] | ||
| 83 | C2–C2’ | Cryptosporioptide B | C38H34O18 | Cryptosporiopsis sp. 8999 | [82] | ||
| 84 | C2–C2’ | Cryptosporioptide C | C40H38O18 | Cryptosporiopsis sp. 8999 | [82] | ||
| 85 | C2–C2’ | Penicixanthone E | C38H34O18 | Penicillium purpurogenum SC0070 | [84] | ||
| 86 | C2–C2’ | Penicixanthone F | C40H38O18 | P. purpurogenum SC0070 | [84] | ||
| 87 | C2–C2’ | Penicixanthone G | C39H36O18 | P. purpurogenum SC0070 | [84] | ||
| 88 | C2–C2’ | Penicixanthone H | C35H32O15 | P. purpurogenum SC0070 | [84] | ||
| 89 | C2–C2’ | Aflaxanthone A | C30H30O11 | Aspergillus flavus QQYZ | [18] | ||
| 90 | C2–C2’ | Aflaxanthone B | C30H30O11 | A. flavus QQYZ | [18] | ||
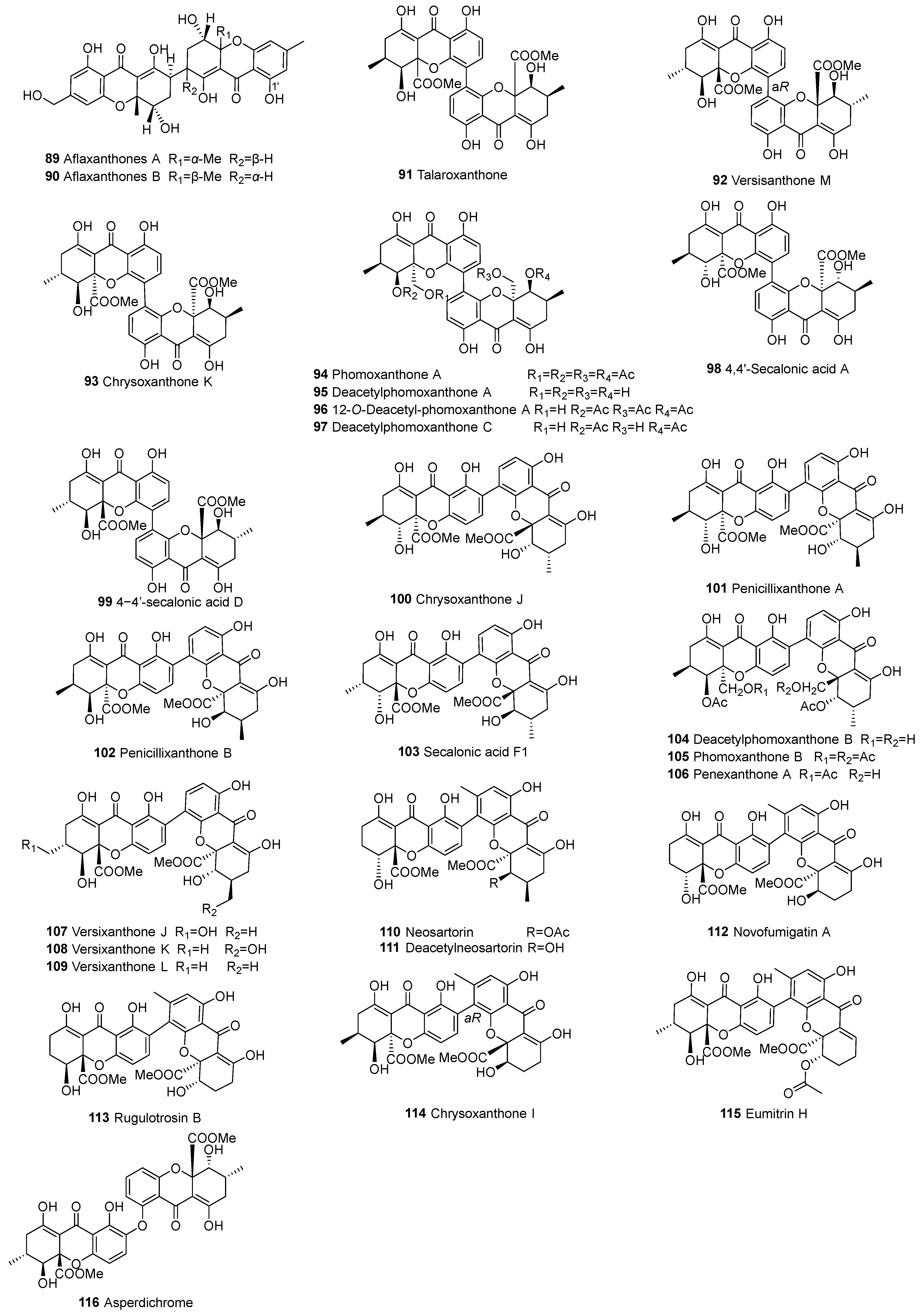
| 91 | d–d | C4–C4’ | Talaroxanthone (4-4’-secalonic acid E) | C32H30O14 | Talaromyces sp. | [85,86] | |
| 92 | C4–C4’ | Versixanthone M | C32H30O14 | A. versicolor HDN1009 | [76] | ||
| 93 | C4–C4’ | Chrysoxanthone K | C32H30O14 | P. chrysogenum C-7-2-1 | [59] | ||
| 94 | C4–C4’ | Phomoxanthone A | C38H38O16 | Phomopsis sp. BCC 1323, y254 and IM 41-1; Paecilomyces sp. EJC01.1, D. goulteri L17 | [68,74,87,88] | ||
| 95 | C4–C4’ | Deacetylphomoxanthone A | C30H30O12 | Phomopsis sp. BCC 1323 | [87] | ||
| 96 | C4–C4’ | 12-O-deacetylphomoxanthone A | C36H36O15 | P. asparagi DHS-48 and Phomopsis sp. DHS-11; Phomopsis sp. IM 41-1 | [68,88,89] | ||
| 97 | C4–C4’ | Deacetylphomoxanthone C | C34H33O14 | D. goulteri L17, HNY29-2B | [68,71] | ||
| 98 | C4–C4’ | 4-4′-secalonic acid A | C32H30O14 | P. chrysogenum C-7-2-1 | [59,85] | ||
| 99 | C4–C4’ | 4-4’-secalonic acid D | C32H30O14 | A. aculeatinus WHUF0198 | [19,90] | ||
| 100 | d–d | C2–C4’ | Chrysoxanthone J | C32H30O14 | P. chrysogenum C-7-2-1 | [59] | |
| 101 | C2–C4’ | Penicillixanthone A (2-4′-linked SAA) | C32H31O14 | Aspergillus niger, Aspergillus fumigates; P. chrysogenum C-7-2-1 | [17,59,60,91] | ||
| 102 | C2–C4’ | Penicillixanthone B | C32H31O14 | Setophoma terrestris MSX45109 | [60,61] | ||
| 103 | C2–C4’ | Secalonic acid F1 | C32H30O14 | Aspergillus sp. TPU1343, P. chrysogenum C-7-2-1 | [17,59,60] | ||
| 104 | C2–C4’ | Deacetylphomoxanthone B | C34H34O14 | Phomopsis sp. BCC 1323, Phomopsis sp. PSU-D15 | [70,71] | ||
| 105 | C2–C4’ | Phomoxanthone B | C38H38O16 | Phomopsis sp. BCC 1323 and xy21 | [70,74,87,92] | ||
| 106 | C2–C4’ | Penexanthone A | C36H36O15 | Penicillium sp. CR1642D, D. goulteri L17 | [68,71,93] | ||
| 107 | C2–C4’ | Versixanthone J | C32H31O15 | A. versicolor HDN1009 | [76] | ||
| 108 | C2–C4’ | Versixanthone K | C32H31O15 | A. versicolor HDN1009 | [76] | ||
| 109 | C2–C4’ | Versixanthone L | C32H30O14 | A. versicolor HDN1009 | [76] | ||
| 110 | C2–C4’ | Neosartorin | C34H32O15 | Aspergillus novofumigatus | [80,94] | ||
| 111 | C2–C4’ | Deacetylneosartorin | C32H28O14 | A. novofumigatus | [94] | ||
| 112 | C2–C4’ | Novofumigatin A | C30H26O14 | A. novofumigatus | [94] | ||
| 113 | C2–C4’ | Rugulotrosin B | C32H30O14 | Penicillium sp. | [80] | ||
| 114 | C2–C4’ | Chrysoxanthone I | C32H30O14 | P. chrysogenum C-7-2-1 | [59] | ||
| 115 | C2–C4’ | Eumitrin H | C34H34O14 | Usnea baileyi (whole thallus) | [95] | ||
| 116 | d–d | C–O–C | Asperdichrome | C32H34O14 | Aspergillus sp. TPU1343 | [62] | |
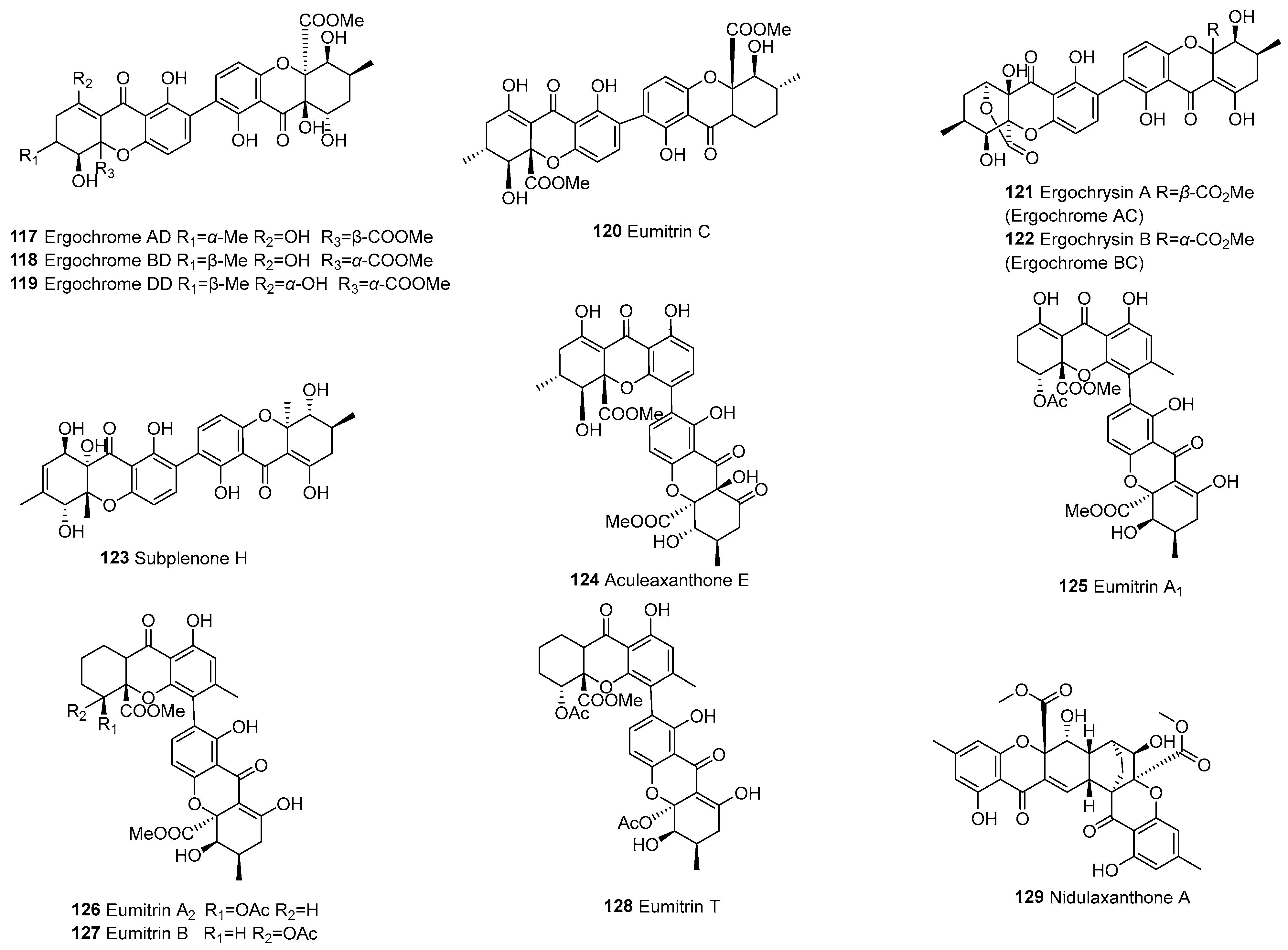
| 117 | Tetrahydroxanthone (d) and hexahydroxanthone (f) | d–f | C2–C2’ | Ergochrome AD | C32H32O15 | C. purpurea | [64] |
| 118 | C2–C2’ | Ergochrome BD | C32H32O15 | C. purpurea | [64] | ||
| 119 | C2–C2’ | Ergochrome DD | C31H28O14 | C. purpurea | [64] | ||
| 120 | C2–C2’ | Eumitrin C | C32H32O13 | U. baileyi (whole thallus) | [96] | ||
| 121 | C2–C2’ | Ergochrysin A (Ergochrome AC) | C31H28O14 | C. purpurea | [64] | ||
| 122 | C2–C2’ | Ergochrysin B (Ergochrome BC) | C31H28O14 | C. purpurea | [64] | ||
| 123 | C2–C2’ | Subplenone H | C30H28O11 | Subplenodomus sp. CPCC 401465 | [56] | ||
| 124 | C4–C2’ | Aculeaxanthone E | C32H30O15 | A. aculeatinus WHUF0198 | [18] | ||
| 125 | C4–C2’ | Eumitrin A1 | C34H32O15 | U. baileyi (whole thallus) | [97] | ||
| 126 | C4–C2’ | Eumitrin A2 | C34H34O14 | U. baileyi (whole thallus) | [97] | ||
| 127 | C4–C2’ | Eumitrin B | C34H34O14 | U. baileyi (whole thallus) | [97] | ||
| 128 | C4–C2’ | Eumitrin T | C34H34O14 | U. baileyi (whole thallus) | [27] | ||
| 129 | d–f | 2,3-ring | Nidulaxanthone A | C32H28O12 | Aspergillus sp. F029 | [98] | |
| 130 | Hexahydroxanthones (f) | f–f | C2–C2’ | Eumitrin F | C32H34O12 | U. baileyi (whole thallus) | [95] |
| 131 | C2–C2’ | Eumitrin G | C32H34O12 | U. baileyi (whole thallus) | [95] | ||
| 132 | C2–C2’ | Phomoxanthone C | C30H34O14 | Phomopsis sp. | [69] | ||
| 133 | C2–C2’ | Phomoxanthone D | C30H32O14 | Phomopsis sp. xy21, P. asparagi DHS-48 and Phomopsis sp. DHS-11 | [69,89] | ||
| 134 | C2–C2’ | Cladoxanthone B | C37H34O12 | Cladosporium sp. | [99] | ||
| 135 | C2–C2’ | Ergochrome CD | C31H30O15 | C. purpurea | [64] | ||
| 136 | C2–C2’ | Ergoflavin (Ergochrome CC) | C32H34O16 | C. purpurea, PM0651480 (isolated as an endophytic fungus from the leaves of Mimosops elengi) | [64,100] | ||
| 137 | f–f | C2–C4’ | Eumitrin E | C33H31O15 | U. baileyi (whole thallus) | [96] | |
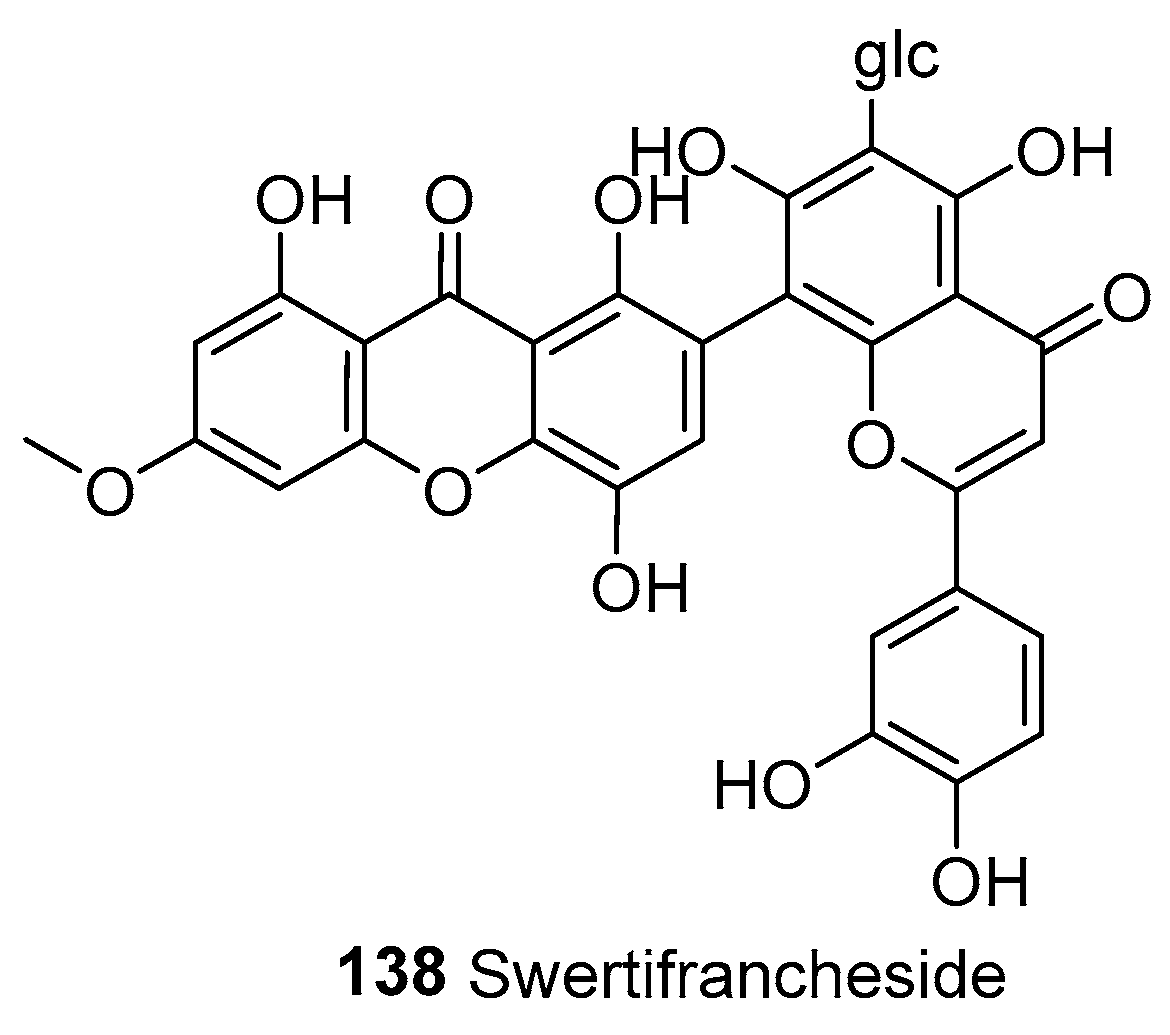
| 138 | Heterodimers | Xanthone–flavone | Swertifrancheside | C35H29O17 | S. franchetiana (entire plant) | [33] | |
| 139 | Xanthonelignans | Cadensin A | C24H20O9 | Caraipa densiflora | [101] | ||
| 140 | Cadensin B | C25H22O10 | C. densiflora | [101] | |||
| 141 | Kielcorin | C24H20O8 | Psorospermum febrifugum (root) | [102] | |||
| 142 | Cadensin D | C25H22O9 | P. febrifugum (root) | [102] | |||
| 143 | Isocadensin D | C25H22O9 | P. febrifugum (root) | [102] | |||
| 144 | Isodensin D monoacetate | C27H24O10 | P. febrifugum (root) | [102] | |||
| 145 | Cadensin F | C26H24O10 | P. febrifugum (root) | [102] | |||
| 146 | 6-Hydroxyisocadensin F | C26H24O11 | P. febrifugum (root) | [102] | |||
| 147 | Cadensin G | C24H20O10 | P. febrifugum (root) | [102] | |||
| 148 | 5’-Demethoxycadensin G | C23H18O9 | Cratoxylum cochinchinense (bark) | [41] | |||
| 149 | (±) Esculentin A | C24H20O10 | G. esculenta (branches) | [42] | |||
| 150 | Heterodimers | Xanthone– benzophenones | Garciduol A | C27H18O9 | Garcinia duicis (root) | [103] | |
| 151 | Garciduol B | C27H18O10 | G. duicis (root) | [103] | |||
| 152 | Garciduol C | C27H18O9 | G. duicis (root) | [103] | |||
| 153 | C–S–C | Dioschrin | C33H26O13S | Alternaria sp. | [54] | ||
| 154 | Subplenone J | C30H26O10 | Subplenodomus sp. CPCC 401465 | [56] | |||
| 155 | Secalonic acid H | C32H28O14 | Aspergillus brunneoviolaceus MF180246, P. oxalicurn | [17,104] | |||
| 156 | Versixanthone I | C32H28O14 | A. versicolor HDN1009 | [76] | |||
| 157 | Secalonic acid I | C32H28O14 | P. oxalicurn | [104] | |||
| 158 | C–O–C | Chrysoxanthone | C32H30O14 | The ascomycete IBWF11-95A; Aspergillus sp. TPU1343 | [62,105] | ||
| 159 | d–chromanone | C2–C2’ | Chrysoxanthone B | C32H30O14 | A. aculeatinus WHUF0198 | [19] | |
| 160 | C2–C2’ | Chrysoxanthone C | C32H30O14 | A. aculeatinus WHUF0198, A. brunneoviolaceus MF180246 | [17,19] | ||
| 161 | C2–C2’ | (-)-Blennolide G | C32H30O10 | Blennoria sp. | [54] | ||
| 162 | C2–C2’ | Blennolide G | C32H30O10 | Blennoria sp. | [106] | ||
| 163 | C2–C2’ | Muyocoxanthone G | C32H34O11 | M. laterale | [79] | ||
| 164 | C2–C2’ | Muyocoxanthone H | C32H34O12 | M. laterale | [79] | ||
| 165 | C2–C2’ | Muyocoxanthone J | C32H34O11 | M. laterale | [79] | ||
| 166 | C2–C2’ | Muyocoxanthone K | C32H34O11 | M. laterale | [79] | ||
| 167 | C2–C2’ | Versixanthone D | C32H30O14 | A. versicolor HDN1009 | [16] | ||
| 168 | C2–C2’ | Versixanthone F | C33H34O15 | A. versicolor HDN1009 | [16] | ||
| 169 | C2–C2’ | Chrysoxanthone E | C32H30O14 | P. chrysogenum C-7-2-1 | [59] | ||
| 170 | C2–C2’ | Diaporxanthone A | C32H32O13 | D. goulteri L17 | [68] | ||
| 171 | C2–C2’ | Diaporxanthone B | C32H32O13 | D. goulteri L17 | [68] | ||
| 172 | C2–C2’ | Phomolactonexanthone B | C34H34O14 | D. goulteri L17, Phomopsis sp. HNY29-2B | [71] | ||
| 173 | C2–C2’ | Diaporxanthone C | C36H38O16 | D. goulteri L17 | [68] | ||
| 174 | C2–C2’ | Diaporxanthone D | C36H38O16 | D. goulteri L17 | [68] | ||
| 175 | C4–C4’ | Versixanthone B | C32H30O14 | A. versicolor HDN1009 | [16] | ||
| 176 | C4–C4’ | Diaporxanthone E | C32H32O13 | D. goulteri L17 | [68] | ||
| 177 | C2–C4’ | Chrysoxanthone D | C32H30O14 | P. chrysogenum C-7-2-1 | [59] | ||
| 178 | C2–C4’ | Versixanthone C | C32H30O14 | A. versicolor HDN1009 | [16] | ||
| 179 | C2–C4’ | Phomolactonexanthone A | C34H33O14 | D. goulteri L17, Phomopsis sp. HNY29-2B | [68,71] | ||
| 180 | C2–C4’ | Muyocoxanthone I | C32H34O12 | M. laterale | [79] | ||
| 181 | C2–C4’ | Muyocoxanthone L | C32H34O11 | M. laterale | [79] | ||
| 182 | C4–C2’ | Aculeaxanthone A | C32H30O14 | A. aculeatinus WHUF0198 | [19] | ||
| 183 | C4–C2’ | Chrysoxanthone F | C32H30O14 | P. chrysogenum C-7-2-1 | [59] | ||
| 184 | C4–C2’ | Chrysoxanthone G | C32H30O14 | P. chrysogenum C-7-2-1 | [59] | ||
| 185 | C4–C2’ | Versixanthone A | C32H30O14 | A. versicolor HDN1009 | [16] | ||
| 186 | C4–C2’ | Versixanthone E | C33H34O15 | A. versicolor HDN1009 | [16] | ||
| 187 | f–chromanone | C2–C2’ | Eumitrin I | C32H32O13 | U. baileyi (whole thallus) | [107] | |
| 188 | C2–C2’ | Eumitrin J | C32H32O13 | U. baileyi (whole thallus) | [107] | ||
| 189 | C2–C2’ | Phomoxanthone N | C30H30O13 | P. asparagi DHS-48 and Phomopsis sp. DHS-11 | [89] | ||
| 190 | C2–C2’ | Ergoxantin | C31H28O14 | Portuguese ergot drug | [108] | ||
| 191 | C2–C2’ | Phomoxanthone L | C30H30O13 | P. asparagi DHS-48 and Phomopsis sp. DHS-11 | [89] | ||
| 192 | C2–C2’ | Phomoxanthone M | C30H30O13 | P. asparagi DHS-48 and Phomopsis sp. DHS-11 | [89] | ||
| 193 | C2–C4’ | Eumitrin K | C32H32O13 | U. baileyi (whole thallus) | [107] | ||
| 194 | C4–C2’ | Eumitrin D | C34H34O14 | U. baileyi (whole thallus) | [96] | ||
| 195 | chromanones | C2–C2’ | Aculeaxanthone B | C32H30O14 | A. aculeatinus WHUF0198 | [19] | |
| 196 | C2–C2’ | Aculeaxanthone C | C32H30O14 | A. aculeatinus WHUF0198 | [19] | ||
| 197 | C2–C2’ | Phomopsis-H76 A/Diaporthochromone B | C30H30O12 | P. asparagi DHS-48 and Phomopsis sp. DHS-11, Phomopsis sp. (#zsu-H76) | [65,89,109] | ||
| 198 | C2–C2’ | Blennolide H | C32H30O14 | Alternaria sp. | [54] | ||
| 199 | C2–C2’ | Blennolide I | C32H30O14 | Blennoria sp. | [54] | ||
| 200 | C2–C2’ | Muyocoxanthone A | C32H34O12 | M. laterale | [79] | ||
| 201 | C2–C2’ | Muyocoxanthone B | C32H34O12 | M. laterale | [79] | ||
| 202 | C2–C2’ | Muyocoxanthone C | C32H34O12 | M. laterale | [79] | ||
| 203 | C2–C2’ | Monodictyochrome B | C30H30O11 | Monodictys putredinis | [110] | ||
| 204 | C2–C2’ | Aculeaxanthone D | C33H34O15 | A. aculeatinus WHUF0198 | [18] | ||
| 205 | C2–C2’ | Chrysoxanthone O | C32H32O15 | P. chrysogenum C-7-2-1 | [59] | ||
| 206 | C2–C2’ | Chrysoxanthone P | C32H34O15 | P. chrysogenum C-7-2-1 | [59] | ||
| 207 | C2–C2’ | Paecilins M | C32H34O15 | P. chrysogenum C-7-2-1 | [59] | ||
| 208 | C2–C2’ | Chrysoxanthone L | C32H34O16 | P. chrysogenum C-7-2-1 | [59] | ||
| 209 | C2–C4’ | Muyocoxanthone D | C32H34O12 | M. laterale | [79] | ||
| 210 | C2–C4’ | Chrysoxanthone N | C32H32O15 | P. chrysogenum C-7-2-1 | [59] | ||
| 211 | C4–C2’ | Blennolide J | C32H30O14 | Blennoria sp. | [54] | ||
| 212 | C4–C2’ | Monodictyochrome A | C30H30O11 | M. putredinis | [110] | ||
| 213 | C4–C2’ | Chrysoxanthone M | C32H34O16 | P. chrysogenum C-7-2-1 | [59] | ||
| 214 | C4–C2’ | Chrysoxanthone Q | C32H34O15 | P. chrysogenum C-7-2-1 | [59] |
| Compounds | Biological Activities | Testing Subjects | Outcome | Effects/Mechanisms | Ref. |
|---|---|---|---|---|---|
| Swertiabisxanthone-I 8′-O-D-glucopyranoside (4) | Neuroprotective | H2O2-induced PC12 cell | Cell viability up to 157.8% ± 6.0 (at a concentration of 25 μg/mL) | — | [23] |
| Swertipunicoside (5) | Antiviral | Inhibitor of HIV reverse transcriptase | ED50 = 3.0 μg/mL | — | [33] |
| 3-O-Demethylswertipunicoside (6) | Neuroprotective | H2O2-induced PC12 cell | Cell viability up to 123.0% ± 5.6 (at a concentration of 25 μg/mL) | Increase the protein expression of both tyrosine hydroxylase (TH) and DJ-1 | [23,24] |
| Mangiferoxanthone A (8) | Anti-influenza | Influenza neuraminidase (NA) | 55.8% inhibition | — | [34] |
| Antiviral | Coxsackie virus B3 3C protease | 46.1% inhibition at 100 μM | [34] | ||
| Bigarciculenxanthone A (10) | Cytotoxicity | Myeloid leukemia HL-60, lung cancer A-549, hepatocellular carcinoma SMMC-7721, breast cancer MDA-MB-231, colon cancer SW480 cells | IC50 = 15.2–22.9 μM | — | [36] |
| Bigarciculenxanthone B (11) | Cytotoxicity | HL-60, A-549, SMMC-7721, MDA-MB-231, SW480 cells | IC50 = 17.8–29.9 μM | — | [36] |
| Bigarcinenone B (13) | Antioxidative | 1,1-diphenyl-2-picrylhydrazyl (DPPH) free-radical-scavenging method | IC50 = 20.14 μM | — | [37] |
| Luminol-H2O2-CoII-EDTA luminescence system | IC50 = 2.85 μM | — | [37] | ||
| Biscaloxanthone (14) | Cytotoxicity | Human lung carcinoma (A549), breast cancer (MCF-7), cervical cancer (C33A), and normal rat fibroblast (3T3L1) | IC50 > 50 μM | — | [38] |
| Globulixanthone E (15) | Antimicrobial | Staphylococcus aureus (ATCC 6538) Bacillus subtilis (ATCC 6633) Vibrio anguillarium (ATCC 19264) | MIC = 3.12–5.56 μg/mL | — | [39] |
| Garcinoxanthone B (17) | Anti-inflammation | Lipopolysaccharide (LPS)-stimulated RAW264.7 cells | IC50 = 11.3 ± 1.7 μM | Inhibit production of NO | [40] |
| Garcinoxanthone C (18) | Anti-inflammation | As above | IC50 = 18.0 ± 1.8 μM | Inhibit production of NO | [40] |
| Cratoxyxanthone (19) | Cytotoxicity | K562, HeLa cells | IC50 = 17.1–39.8 μg/mL | — | [112] |
| Bigarcinenone A (20) | Antioxidative | DPPH radical-scavenging test | IC50 = 9.2 μM | — | [22] |
| Bixanthone C (21) | Treat hepatic brosis | — | — | — | [27] |
| Bixanthone D (22) | Treat hepatic brosis | — | — | — | [27] |
| Jacarelhyperol A (24) | Anti-PAF | Male ddY mice (SPF grade), 6 weeks old | — | Inhibit PAF-induced hypotension in vivo | [44] |
| Jacarelhyperol B (25) | Anti-PAF | As above | — | As above | [44] |
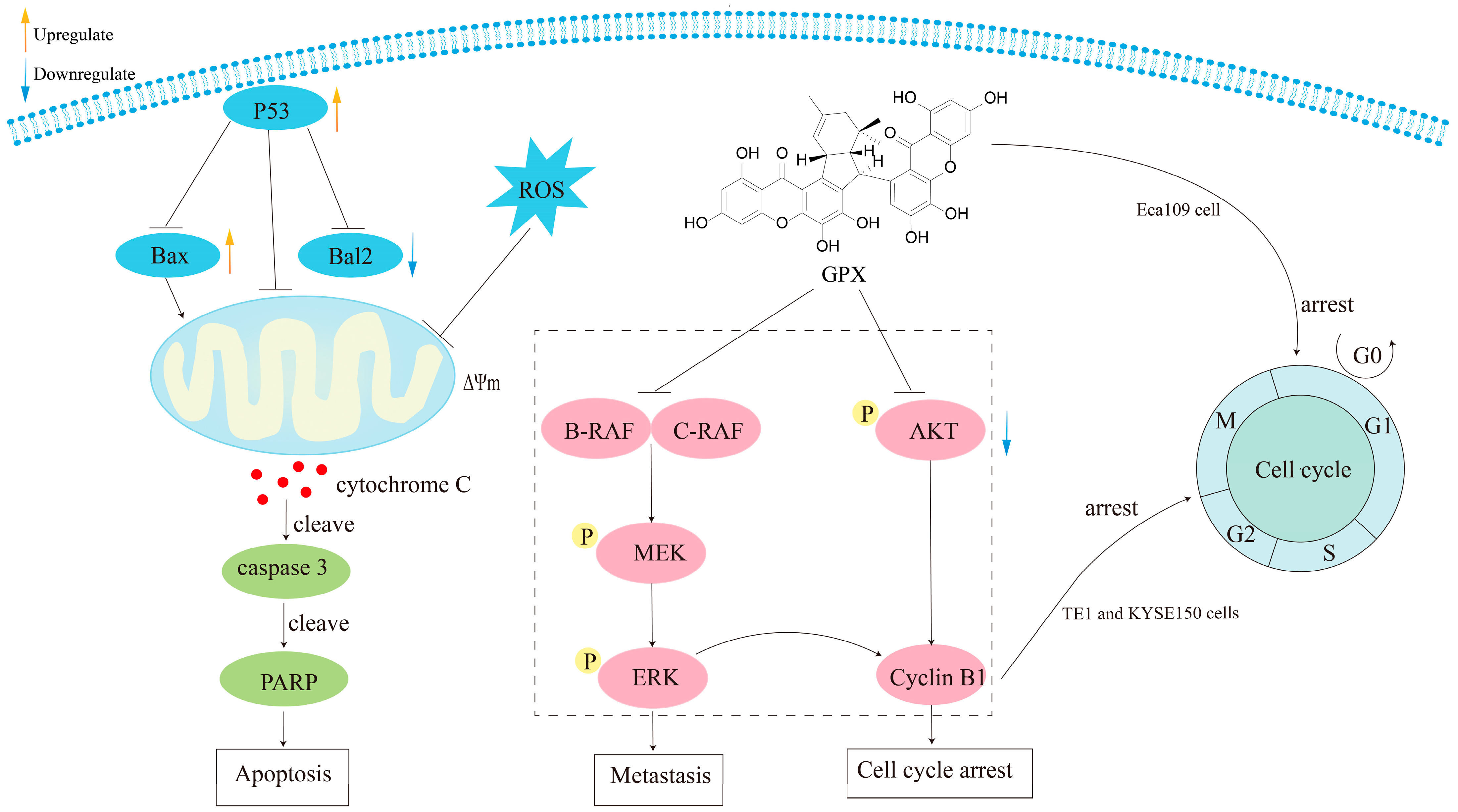
| Griffipavixanthone (28) | Anticancer | Human non-small-cell lung cancer H520 cell | IC50 = 3.03 ± 0.21 μM | Induce cell apoptosis through mitochondrial apoptotic pathway accompanying with ROS production | [25] |
| Human esophageal cancer cell line TE1 and KYSE150 cells | — | Inhibit cell migration and invasion; render cell proliferation and induce G2/M cell cycle arrest; inhibit tumor metastasis and proliferation via downregulating RAF–MEK–ERK pathway | [15] | ||
| Human breast cancer cells MCF-7 and T-47D | IC50, 48h = 9.64 ± 0.12 μM IC50, 48h = 10.21 ± 0.38 μM | Increase the mRNA and protein expression level of p53 and its target genes, upregulate p53 and Bax expression, suppress Bcl-2 expression, induce MCF-7 cell apoptosis | [14] | ||
| Hypoglycemic | Sucrase | IC50 = 4.58 μM | Inhibit sucrase | [52] | |
| XO inhibitors | Xanthine oxidase | IC50 = 6.3 μM | Inhibit XO | [42] | |
| Garmoxanthone (29) | Antibacterial | MRSA (ATCC 43300) MRSA (CGMCC1.12409) | MIC = 3.9 μg/mL | — | [20] |
| Vibrio rotiferianus (MCCC E385), V. vulnificus (MCCC E1758), V. campbellii (MCCC E333) | MIC = 15.6–31.2 μg/mL | — | [20] | ||
| Garcilivin A (30) | Cytotoxicity | MRC-5 cells | IC50 = 2.0 μM | — | [47] |
| Antiparasitic | Trypanosoma brucei brucei Trypanosoma cruzi Plasmodium falciparum | IC50 = 0.4 μM IC50 = 4.0 μM IC50 = 6.7 μM | — | [47] | |
| Garcilivin C (31) | Cytotoxicity | MRC-5 cells | IC50 = 52.3 μM | — | [47] |
| Antiparasitic | T. cruzi | IC50 = 7.7 μM | — | [47] | |
| Schomburgkixanthone (37) | Hypoglycemic | In vitro inhibition rat intestinal a-glucosidase (maltase, sucrase) | IC50 = 0.79 μΜ IC50 = 0.81 μΜ | — | [52] |
| Puniceaside B (41) | Neuroprotective | H2O2-induced PC12 cell | Cell viability 98.1% ± 6.8 at a concentration of 25 μg/mL | — | [23] |
| Subplenone C (43) | Antibacterial | Methicillin-resistant S. aureus ATCC 43300 and 700698, S. aureus ATCC 29213 MSSA, vancomycin-resistant Enterococcus faecium ATCC 700221, and Staphylococcus epidermidis ATCC 12228 MSSE | MIC = 1.0–4.0 μg/mL | — | [56] |
| Phomalevone A (44) | Antibacterial | B. subtilis (ATCC 6051) and S. aureus (ATCC 29213) | Inhibitory zones of 36 and 23 mm | — | [57] |
| Subplenone D (45) | Antibacterial | Methicillin-resistant S. aureus ATCC 43300 and 700698, S. aureus ATCC 29213 MSSA, vancomycin-resistant E. faecalis ATCC 51299, vancomycin-resistant Enterococcus faecium ATCC 700221, E. faecalis ATCC 29212 VSE, and S. epidermidis ATCC 12228 MSSE | MIC = 0.125–4.0 μg/mL | — | [56] |
| Subplenone E (46) | Antibacterial | As above | MIC = 0.125–2.0 μg/mL | — | [56] |
| Subplenone F (47) | Antibacterial | As above | MIC = 0.25–4.0 μg/mL | — | [56] |
| Phomalevone C (48) | Antibacterial | B. subtilis (ATCC 6051) and S. aureus (ATCC 29213) | Inhibitory zones of 34 and 22 mm | — | [57] |
| Antifungal | Aspergillus flavus (NRRL 6541) and Fusarium verticillioides (NRRL 25457) | Inhibition zones of 10 mm IC50 = 4 μg/mL against A. flavus, MIC = 10 μg/mL against F. verticillioides | — | [57] | |
| Subplenone I (49) | Antibacterial | Methicillin-resistant S. aureus ATCC 43300 and 700698, S. aureus ATCC 29213 MSSA, vancomycin-resistant E. faecalis ATCC 51299, vancomycin-resistant E. faecium ATCC 700221, E. faecalis ATCC 29212 VSE, and S. epidermidis ATCC 12228 MSSE | MIC = 0.5–16 μg/mL | — | [56] |
| Subplenone G (50) | Antibacterial | As above | MIC = 0.125–4.0 μg/mL | — | [56] |
| Subplenone A (51) | Antibacterial | As above | MIC = 0.125–2.0 μg/mL | — | [56] |
| Subplenone B (52) | Antibacterial | As above | MIC = 0.25–4.0 μg/mL | — | [56] |
| Terricoxanthone F (58) | Antibacterial | Candida Albicans | MIC = 16 μg/mL | — | [58] |
| Secaionic acid A (59) | Cytotoxicity | H23 human non-small-cell lung carcinoma cells | IC50 = 2.6 μM | — | [59] |
| MDA-MB-435 (melanoma) and SW-620 (colon) cancer cell lines | IC50 = 0.16 μM IC50 = 0.41 μM | — | [61] | ||
| Antibacterial | Gram-positive bacterium Micrococcus luteus and S. aureus | MIC = 38 μg/mL MIC = 75 μg/mL | — | [61] | |
| Neuroprotective (reduced colchicine cytotoxicity) | Newborn Sprague–Dawley rats | SAA at doses of 3 and 10 μM | Inhibit phosphorylation of JNK and p38 MAPKs, calcium influx, and the activation of caspase-3 | [113] | |
| Neuroprotective | Pregnant Sprague–Dawley rats (14–16 days) and male C57BL/6J mice (8–10 weeks old, 20–22 g) | SAA at doses of 0.15 μg/kg and 0.75 μg/kg | Inhibit the phosphorylation of JNK and p38 MAPK, downregulate Bax expression, and suppress caspase-3 activation | [114] | |
| Antibacterial | All multidrug-resistant bacterial strains Escherichia coli 942, E. coli 4814, S. aureus 931, S. aureus 934, S. aureus MRSA 1872, and K. pneumonia 815 | MIC = 4.7–37.5 μg/mL | — | [115] | |
| Secaionic acid B (60) | Antifungal | Microbotryum violaceum | 13 mm (Agar diffusion assays) | — | [106] |
| Antialgal | Chlorella fusca | 5 mm (Agar diffusion assays) | — | [106] | |
| Antimicrobial | Gram-positive (Bacillus megaterium) and Gram-negative (E. coli) bacteria | 0 mm and 15 mm (Agar diffusion assays) | — | [106] | |
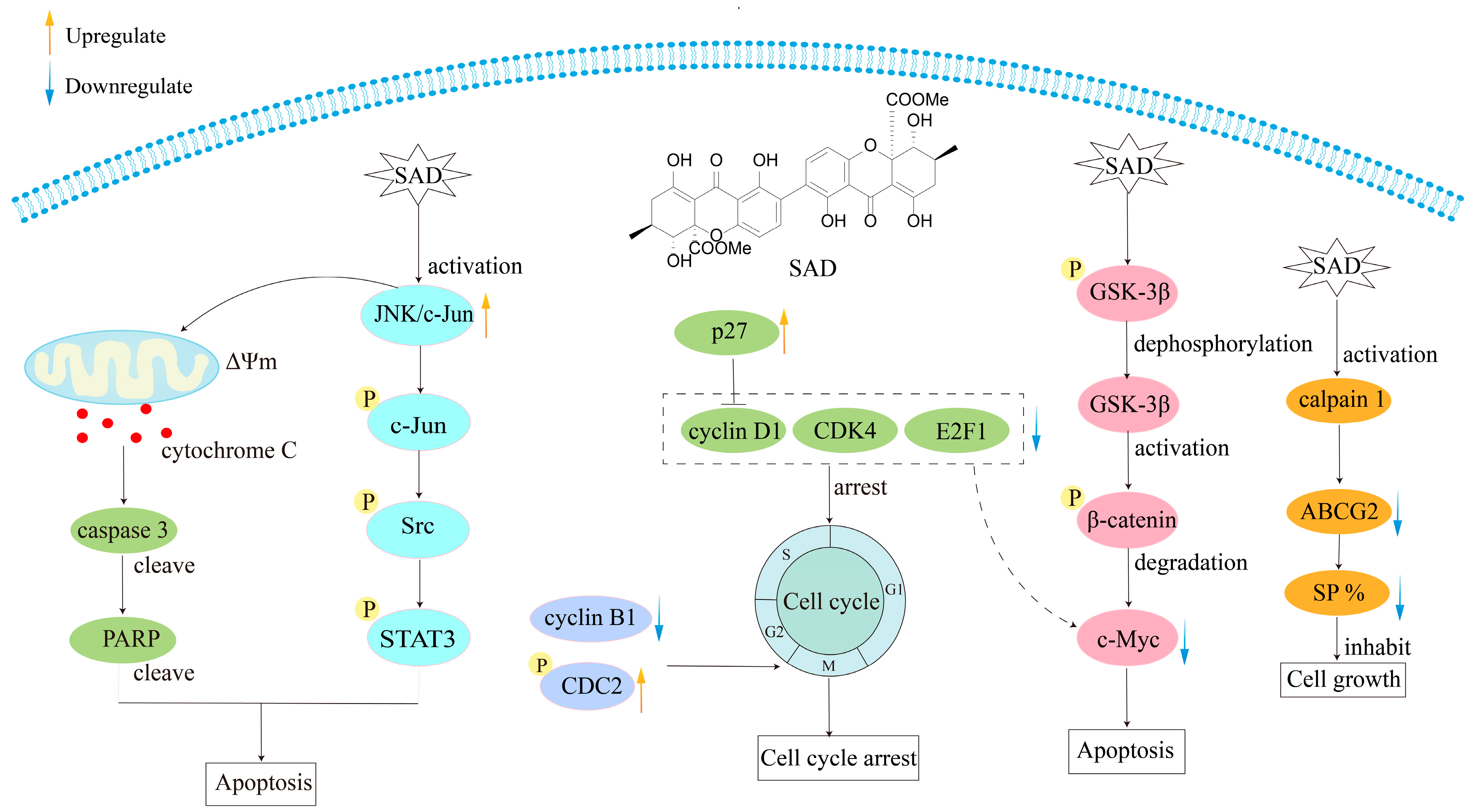
| Secalonic acid D (62) | Cytotoxicity | HL60, K562 cells | IC50 = 0.38 μmol/L IC50 = 0.43 μmol/L | Induce leukemia cell apoptosis and cell cycle arrest of G1 with involvement of GSK-3β/β-catenin/c-Myc pathway | [116] |
| BGC-823, SKHEP, SGC-7901, HeLa, HGC-27, A549, EC9706, SKMES-1, KYSE450, SPC-A1, CNE1, 95D, CNE2, Jeko-1, SW620, Raji, SW480, U937, LOVO, A375, HuH-7, PLC/PRF/5 1, HFF, H22 | IC50, Average = 1.353 μg/mL | — | [90] | ||
| Anticancer | Plasmid substrate, pBR32 | MIC = 0.4 μM | Inhibit the binding of Topo I to DNA | [117] | |
| Anticancer | Human oral epidermoid carcinoma cell line KB and its vincristine-selected derivative ABCB1-overexpressing cell line KBv200, human breast carcinoma cell line MCF-7 and its doxorubicin-selected derivative ABCB1-overexpressing cell line MCF-7/Adr, human epidermoid carcinoma cell line KB-3-1 and its doxorubicin-selected derivative ABCC1-overexpressing cell line CA120, human colon carcinoma cell line S1 and its mitoxantrone-selected derivative ABCG2-overexpressing cell line S1-M1-80, human lung carcinoma cell lines A549, GLC82, and H460 | IC50 = 0.080–0.308 μmol/L | Downregulate the expression of ABCG2 protein by activation of calpain 1, inhibit the growth of SP cells, and decrease the percentage of SP cells | [118] | |
| Cytotoxicity | Human colon carcinoma cell line S1, non-small-cell lung cancer cell line H460, MCF-7, and their corresponding mitoxantrone-selected derivative ABCG2-overexpressing cell lines S1-MI-80, H460/MX20, doxorubicin-selected cell line MCF-7/ADR, human normal colon epithelial cells (NCM460), and human umbilical vein endothelial cells (HUVEC) | IC50 = 3.8–27 μmol/L | Induce cancer cell death through c-Jun/Src/STAT3 signaling axis by inhibiting the proteasome-dependent degradation of c-Jun in both sensitive cells and ABCG2-mediated MDR cells | [119] | |
| Secalonic acid E (63) | Cytotoxicity | SW-620 (colon) | IC50 = 19.12 μM | — | [61] |
| Antibacterial | Gram-positive bacterium M. luteus | MIC = 36 μg/mL | — | [61] | |
| Secalonic acid F (64) | Cytotoxicity | HL60 cells | IC50 = 4 μg/ml | Induce RhoGDI 2 differential expression, caspase 3 activation, and RhoGDI 2 cleavage | [66] |
| HT1080, Cne2, and Bel7402 cell lines | IC50 = 11.43–16.6 μmol/L | — | [120] | ||
| Hypoglycemic | Protein tyrosine phosphatase 1B (PTP1B) | IC50 = 9.6 μM | Inhibit PTP1B | [62] | |
| Secalonic acid G (65) | Antibacterial | Gram-positive bacterium M. luteus and S. aureus | MIC = 5 μg/mL MIC = 39 μg/mL | — | [61] |
| Cytotoxicity | MDA-MB-435 (melanoma) and SW-620 (colon) cancer cell lines | IC50 = 3.27 μM IC50 = 3.67 μM | — | [61] | |
| Diaporxanthone F (66) | Antifungal | Colletotrichum musae (ACCC 31244) | Minimum dosages 2.5 μg/scrip | — | [68] |
| Dicerandrol A (68) | Cytotoxicity | Human breast MDA-MB-435, human colon HCT-116, human lung Calu-3, and human liver Huh7 HCT-116 and A549 cells | IC50 < 10 μM IC100 = 7.0 μg/mL | — | [71,72] |
| Cytotoxicity | HepG2 cell | IC50 = 4.83 ± 0.22 μmol/L | — | [73,89] | |
| Antimicrobial | Xanthomonas oryzae KACC 10331, Gram-positive bacteria (S. aureus KCTC 1916, B. subtilis KCTC, Clavibacter michiganesis KACC 20122), yeast (C. albicans) | MIC = 0.125–8 μg/mL | — | [121] | |
| Antimicrobial | S. aureus and B. subtilis | 10.8 mm and 11.0 mm (zones of inhibition resulting from 300 μg/disk) | — | [72] | |
| Dicerandrol B (69) | Antimicrobial | S. aureus and B. subtilis | 8.5 mm and 9.5 mm (zones of inhibition resulting from 300 μg/disk) | — | [72] |
| Cytotoxicity | MDA-MB-435, HCT-116, Calu-3, and Huh7 cells HCT-116 and A549 cells | IC50 < 10 μM IC100 = 1.8 μg/mL | — | [71,72] | |
| Anticancer | Human cervical cancer HeLa cells | IC50, 24h = 7.13 μg/mL IC50, 48h = 3.00 μg/mL IC50, 96h = 1.84 μg/mL | Inhibit HeLa cell viability and induce G2/M cell cycle arrest, increase the levels of GRP78, ubiquitin, cleaved PARP, and Bax protein, decrease the levels of PARP and Bcl-2 protein, increase the Bax/Bcl-2 ratio, increase the production of ROS | [13] | |
| Antimicrobial | X. oryzae KACC 10331 | MIC = 16 μg/mL | — | [121] | |
| Dicerandrol C (70) | Cytotoxicity | HCT-116 and A549 cells | IC100 = 7.0 μg/mL IC100 = 1.8 μg/mL | — | [72] |
| Antimicrobial | S. aureus and B. subtilis | 7.0 mm and 8.0 mm (zones of inhibition resulting from 300 μg/disk) | — | [72] | |
| Antimicrobial | X. oryzae KACC 10331 | MIC > 16 μg/mL | — | [121] | |
| Anticancer | HepG2 and HeLa cancer cells | IC50, 48h = 4.17 ± 0.49 μM IC50, 48h = 5.18 ± 0.56 μM | Downregulate the transcription level of β-catenin-stimulated Wnt target gene and the expression of related proteins, including p-GSK3-β, β-catenin, LEF1, Axin1, c-Myc, and CyclinD1; and upregulate GSK3-β expression | [122] | |
| Hirtusneanoside (71) | Antimicrobial | Gram-positive bacteria S. aureus and B. subtilis | LD50 = 0.0034 μM LD50 = 0.0140 μM | — | [75] |
| Chrysoxanthone H (73) | Cytotoxicity | H23 human non-small-cell lung carcinoma cells | IC50 = 6.9 μM | — | [59] |
| Versixanthone G (74) | Anticancer | Human leukemia cell lines HL-60 and K562, lung cancer cell line A549, non-small-cell lung cancer cell H1975, human gastric cancer cell line MGC803, human embryonic kidney HEK293, human ovarian carcinoma cell line HO-8910, and human colon cancer cell line HCT-116 | IC50 = 4.6–20.9 μM | Inhibit Topo I, arresting the cell cycle at the G2/M phase | [76] |
| Versixanthone H (75) | Anticancer | HL-60, K562, A549, H1975, MGC803, HEK293, HO-8910, HCT-116 | IC50 = 5.3–22.1 μM | Inhibit Topo I | [76] |
| Ascherxanthone A (76) | Antiparasitic | P. falciparum K1 | IC50 = 0.20 μg/mL | — | [77] |
| Cytotoxicity | African green monkey kidney fibroblast (Vero) Human epidermoid carcinoma cells (KB) Human breast cancer cells (BC) Human lung cancer cells (NCI-H187) | IC50 = 0.16–1.7 μg/mL | — | [77] | |
| Ascherxanthone B (76) | Antifungal | Magnaporthe grisea | IC90 = 0.58 μg/mL | — | [78] |
| Muyocoxanthone F (79) | Anti-inflammation | LPS-stimulated RAW264.7 cells | IC50 = 1.3 μM | Inhibit production of NO | [79] |
| Rugulotrosin A (80) | Antibacterial | Gram-positive (Enterococcus faecalis, B. cereus, S. aureus) | LD99 = 1.6 μg/mL LD99 = 3.1 μg/mL LD99 = 200 μg/mL | — | [80] |
| Xanthonol (81) | Insecticidal and anthelmintic activities | Lucilia sericata, Aedes aegypti, Haemonchus contortus | LD99 = 33 μg/mL LD99 = 8 μg/mL LD99 = 50 μg/mL | — | [81] |
| Penicixanthone G (87) | Cytotoxicity | Human carcinoma A549, HeLa, and HepG2 cells | IC50 = 0.3–0.6 μM | — | [84] |
| Antibacterial | S. aureus and the methicillin-resistant strain MRSA | MIC = 0.4 μg/mL | — | [84] | |
| Aflaxanthone A (89) | Antibacterial | Pathogenic bacteria (methicillin-resistant S. aureus A7983, B. subtilis ATCC 6633) | MIC = 12.5 μM MIC = 25 μM | — | [18] |
| Antifungal | C. albicans ATCC 10231 Fusarium oxysporum Penicillium italicum Collettrichum musae Colletotrichum gloeosporioides | MIC = 12.5 μM MIC = 12.5 μM MIC = 50 μM MIC = 25 μM MIC = 3.13 μM | — | [18] | |
| Aflaxanthone B (90) | Antibacterial | B. subtilis ATCC 6633 | MIC = 25 μM | — | [18] |
| Antifungal | C. albicans ATCC 10231, F. oxysporum, C. musae, and C. gloeosporioide | MIC = 12.5–25 μM | — | [18] | |
| Versixanthone M (92) | Cytotoxicity | HL-60, K562, A549, H1975, MGC803, HO-8910, HCT-116 | IC50 = 0.4–11.7 μM | — | [76] |
| Chrysoxanthone K (93) | Cytotoxicity | H23 human non-small-cell lung carcinoma cells | IC50 = 3.9 μM | — | [59] |
| Phomoxanthone A (94) | Cytotoxicity | Two cancer cell lines (KB, BC-1) and Vero cells | IC50 = 0.51–1.4 μg/mL | — | [87] |
| Antitubercular | Mycobacterium tuberculosis (H37Ra strain) | IC50 = 0.5 μg/mL | — | [87] | |
| Antimalarial | P. falciparum (K1, multidrug-resistant strain) | IC50 = 0.11 μg/mL | — | [87] | |
| Antimicrobial | B. subtilis | MIC = 7.81 μg/mL | — | [74] | |
| 12-O-Deacetylphomoxanthone A (96) | Cytotoxicity | HepG2, HeLa cells | IC50 = 12.06 ± 0.55 μM IC50 = 20.36 ± 1.99 μM | — | [89] |
| Anticancer | Human ovarian cancer (OC) cells (SKOV-3 and ES-2) | IC50 = 6.08 μM IC50 = 3.8 μM | Downregulate PDK4 | [123] | |
| 4-4’-secalonic acid D (99) | Cytotoxicity | BGC-823, SKHEP, SGC-7901, HeLa, HGC-27, A549, EC9706, SKMES-1, KYSE450, SPC-A1, CNE1, 95D, CNE2, Jeko-1, SW620, Raji, SW480, U937, LOVO, A375, HuH-7, PLC/PRF/5 1, HFF, H22 | IC50, Average = 1.026 μg/mL | — | [90] |
| Chrysoxanthone J (100) | Cytotoxicity | H23 human non-small-cell lung carcinoma cells | IC50 = 6.4 μM | — | [59] |
| Penicillixanthone A (101) | Anti-HIV-1 | TZM-bl cells CCR5-tropic HIV-1 SF162 and CXCR4-tropic HIV-1 NL4-3 | IC50 = 0.36 μM IC50 = 0.26 μM | — | [91] |
| Cytotoxicity | MDA-MB-435 (melanoma) and SW-620 (colon) cancer cell lines | IC50 = 0.18 μM IC50 = 0.21 μM | — | [61] | |
| Antimicrobial | Gram-positive bacterium M. luteus and S. aureus | MIC = 46 μg/mL MIC = 93 μg/mL | — | [61] | |
| Antibacterial | S. aureus | MIC = 6.25 μg/mL | — | [17] | |
| Penicillixanthone B (102) | Antimicrobial | Gram-positive bacterium M. luteus and S. aureus | MIC = 15 μg/mL MIC = 59 μg/mL | — | [61] |
| Cytotoxicity | MDA-MB-435 (melanoma) and SW-620 (colon) cancer cell lines | IC50 = 5.20 μM IC50 = 5.55 μM | — | [61] | |
| Secalonic acid F1 (103) | Antibacterial | S. aureus | MIC = 25 μg/mL | — | [17] |
| Deacetylphomoxanthone B (104) | Antimicrobial | X. oryzae KACC 10331 | MIC = 4 μg/mL | — | [121] |
| Cytotoxicity | MDA-MB-435, HCT-116, Calu-3, and Huh7 cells | IC50 < 10 μM | — | [71] | |
| Phomoxanthone B (105) | Cytotoxicity | MCF7 cells A549 cells 830 cells | IC50, 72h = 0.45 ± 0.12 μM IC50, 72h = 8.01 ± 0.15 μM IC50, 72h = 2.96 ± 1.04 μM | Induce apoptosis, cell cycle arrest at the G2/M phase, inhibit the migration and invasion | [92] |
| Cytotoxicity | Two cancer cell lines (KB, BC-1) and Vero cells | IC50 = 0.7–4.1 μg/mL | — | [87] | |
| Antimalarial | P. falciparum (K1, multidrug-resistant strain) | IC50 = 0.33 μg/mL | — | [87] | |
| Antitubercular | M. tuberculosis (H37Ra strain) | MIC = 6.25 μg/mL | — | [87] | |
| Penexanthone A (106) | Cytotoxicity | MDA-MB-435, HCT-116, Calu-3, and Huh7 cells | IC50 < 10 μM | — | [71] |
| Anticancer | Human colorectal cancer cells (HCT116, SKOV3, MCF-7, A549, and PC3) | IC50 = 1.27–7.08 μg/mL | Enhance the sensitivity of colorectal cancer (CRC) to CDDP and induce ferroptosis by targeting Nrf2 inhibition | [124] | |
| Versixanthone J (107) | Cytotoxicity | HL-60 cells | IC50 = 47.3 μM | — | [76] |
| Versixanthone K (108) | Anticancer | HO-8910, HEK293 cells | IC50 = 49.5 μM | Inhibit Topo I | [76] |
| Versixanthone L (109) | Cytotoxicity | HL-60, K562, A549, MGC803, HO-8910, HCT-116 cells | IC50 = 0.5–1.6 μM | — | [76] |
| Neosartorin (110) | Antibacterial | MRSA MB5393 | MIC = 128 μg/mL | — | [94] |
| Deacetylneosartorin (111) | Antibacterial | MRSA | MIC = 64 μg/mL | — | [94] |
| Rugulotrosin B (113) | Antibacterial | B. subtilis | LD99 = 25 μg/mL | — | [80] |
| Eumitrin H (115) | Antibacterial | E. coli ATCC25922, Pseudomonas aeruginosa ATCC27853, S. aureus ATCC25923, C. albicans TISTR | IC50 = 125–250 μg/mL | [95] | |
| Antitumor | Tyrosinase | IC50 > 200 μM | Inhibit tyrosinase | [95] | |
| Hypoglycemic | α-glucosidase | IC50 = 64.2 ± 0.51 μM | Inhibit α-glucosidase | [95] | |
| Asperdichrome (116) | Hypoglycemic | PTP1B | IC50 = 6.0 μM | Inhibit PTP1B | [62] |
| Antibacterial | S. aureus | MIC = 25 μg/mL | [17] | ||
| Eumitrin C (120) | Antiparasitic | P. falciparum FcB1 | IC50 = 96.5 ± 3.5 μM | — | [96] |
| Subplenone H (123) | Antibacterial | Methicillin-resistant S. aureus ATCC 43300 and 700698, S. aureus ATCC 29213 MSSA, vancomycin-resistant E. faecalis ATCC 51299, vancomycin-resistant E. faecium ATCC 700221, E. faecalis ATCC 29212 VSE, and S. epidermidis ATCC 12228 MSSE | MIC = 0.125–2.0 μg/mL | — | [56] |
| Nidulaxanthone A (129) | Cytotoxicity | HepG2 SW1990, MCF-7, HCT116, and LO2 cells | IC50 = 21.9 μM IC50 > 30 μM | — | [98] |
| Eumitrin F (130) | Antibacterial Antitumor | E. coli ATCC25922, P. aeruginosa ATCC27853, S. aureus ATCC25923, B. subtilis ATCC6633, C. albicans TISTR | IC50 = 62.5–500 μM | — | [95] |
| Eumitrin G (131) | Antibacterial | As above | IC50 = 62.5–500 μM | — | [95] |
| Phomoxanthone D (133) | Immunosuppressive | The proliferation of ConA-induced (T-cell) and LPS-induced (B-cell) murine splenic lymphocytes | IC50 = 44.84 ± 1.26 μM IC50 = 77.76 ± 1.47 μM | — | [89] |
| Cladoxanthone B (134) | Cytotoxicity | MB49 (sensitive mouse bladder carcinoma cells), J82 (human bladder carcinoma cells), 4T1 (mouse breast carcinoma cells), and Huh7 (human hepatocellular carcinoma cells) | IC50 = 24.7–46.4 μM | — | [99] |
| Ergoflavin (136) | Anti-inflammation | Human TNF-a and IL-6 THP-1 cells and human PBMCs | IC50 = 1.9 ± 0.1 μM IC50 = 1.2 ± 0.3 μM | Inhibit TNF-α and IL-6 | [100] |
| Cytotoxicity | ACHN, H460, Panc1, HCT116, and Calu1 cancer cell lines | IC50 = 1.0 –8. 45 μM | — | [100] | |
| Swertifrancheside (138) | Antiviral | Inhibitor of HIV reverse transcriptase | Inhibitory activity of 99.8% at 200 μg/mL (ED50 = 30.9 μg/mL) | — | [33] |
| Subplenone J (154) | Antibacterial | Methicillin-resistant S. aureus ATCC 43300 and 700698, S. aureus ATCC 29213 MSSA, vancomycin-resistant E. faecalis ATCC 51299, vancomycin-resistant E. faecium ATCC 700221, E. faecalis ATCC 29212 VSE, and S. epidermidis ATCC 12228 MSSE | MIC = 0.125–4.0 μg/mL | — | [56] |
| Secalonic acid H (155) | Antibacterial | S. aureus | MIC = 25 μg/mL | — | [17] |
| Versixanthone I (156) | Cytotoxicity | HL-60 cells | IC50 = 27.8 μM | — | [76] |
| Chrysoxanthone (158) | Antimicrobial | Arthrobacter citreus, Penicillium notatum Bacillus brevis, Corynebacterium insidiosum, Aspergillus ochraceus | MIC = 2.5–20 μg/mL | — | [105] |
| Cytotoxicity | Jurkat, L-1210, Colo-320, and HeLa-S3 cells | IC50 > 50 μg/mL | — | [105] | |
| Chrysoxanthone C (160) | Antibacterial | S. aureus | MIC = 50 μg/mL | — | [17] |
| Muyocoxanthone K (166) | Anti-inflammation | LPS-stimulated RAW264.7 cells | IC50 = 5.1 μM | Inhibit production of NO | [79] |
| Versixanthone D (167) | Cytotoxicity | HL-60, K562, A549, H1975, 803, HO8910, and HCT-116 cells | IC50 = 3.1–13.9 μM | — | [16] |
| Versixanthone F (168) | Cytotoxicity | HL-60, K562, A549, HO8910, and HCT-116 cells | IC50 = 0.7–20.8 μM | — | [16] |
| Diaporxanthone A (170) | Antifungal | Nectria sp. | Minimum dosages 10 μg/scrip | — | [68] |
| Diaporxanthone D (174) | Cytotoxicity | A2870 human ovarian cancer, HepG2 human liver cancer, EC109 human esophagus cancer, PC3 human prostate cancer, A549 human lung adenocarcinoma cancer cell lines, and HBE human bronchial epithelial cell lines | IC50 = 1.66–8.4 μM | — | [68] |
| Versixanthone B (175) | Cytotoxicity | HL-60, 803 cells | IC50 = 9.9–21.6 μM | — | [16] |
| Versixanthone C (178) | Cytotoxicity | HL-60, K562, H1975 cells | IC50 = 7.8–25.6 μM | — | [16] |
| Versixanthone A (185) | Cytotoxicity | HL-60, K562, H1975, HO8910 cells | IC50 = 2.6–11.2 μM | — | [16] |
| Versixanthone E (186) | Cytotoxicity | HL-60, K562, H1975, 803, HO8910 cells | IC50 = 1.6–11.1 μM | — | [16] |
| Anticancer | Topoisomerase I | — | Inhibit Topo I | [16] | |
| Phomoxanthone N (189) | Immunosuppressive | The proliferation of ConA-induced (T-cell) and LPS-induced (B-cell) murine splenic lymphocytes | IC50 = 75.75 ± 1.78 μM IC50 = 102.65 ± 1.38 μM | — | [89] |
| Phomoxanthone L (191) | Immunosuppressive | As above | IC50 = 55.53 ± 0.93 μM IC50 = 89.27 ± 2.25 μM | — | [89] |
| Phomoxanthone M (192) | Immunosuppressive | As above | IC50 = 60.25 ± 1.58 μM IC50 = 87.66 ± 2.76 μM | — | [89] |
| Eumitrin D (194) | Antiparasitic | P. falciparum FcB1 | IC50 = 73.0 ± 1.0 μM | — | [96] |
| Cytotoxicity | Huh7 (differential hepatocellular carcinoma), Caco 2 (differentiating colorectal adenocarcinoma), PC-3 (prostate carcinoma), MCF-7 cells | IC50 = 35 μM IC50 = 44 μM IC50 = 42 μM IC50 = 12 μM | — | [96] | |
| Phomopsis-H76 A (197) | Proangiogenic | Zebrafish embryos | — | Accelerated the growth of sub-intestinal vessel plexus (SIV) branch markedly | [109] |
| Muyocoxanthone B (201) | Anti-inflammation | LPS-stimulated RAW264.7 cells | IC50 = 5.2 μM | Inhibit production of NO | [79] |
| Monodictyochrome B (203) | Anticancer | Cyclooxygenase-1 (COX-1) and aromatase enzymatic | IC50 = 7.5 μM | Inhibit cytochrome P450 enzymes | [110] |
| Monodictyochrome A (212) | Anticancer | As above | IC50 = 5.3 μM | As above | [110] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, F.; Fan, M.; Li, H.; Li, S.; Wang, S. Xanthone Dimers in Angiosperms, Fungi, Lichens: Comprehensive Review of Their Sources, Structures, and Pharmacological Properties. Molecules 2025, 30, 967. https://doi.org/10.3390/molecules30040967
Shi F, Fan M, Li H, Li S, Wang S. Xanthone Dimers in Angiosperms, Fungi, Lichens: Comprehensive Review of Their Sources, Structures, and Pharmacological Properties. Molecules. 2025; 30(4):967. https://doi.org/10.3390/molecules30040967
Chicago/Turabian StyleShi, Fengzhi, Min Fan, Haifeng Li, Shiwei Li, and Shuang Wang. 2025. "Xanthone Dimers in Angiosperms, Fungi, Lichens: Comprehensive Review of Their Sources, Structures, and Pharmacological Properties" Molecules 30, no. 4: 967. https://doi.org/10.3390/molecules30040967
APA StyleShi, F., Fan, M., Li, H., Li, S., & Wang, S. (2025). Xanthone Dimers in Angiosperms, Fungi, Lichens: Comprehensive Review of Their Sources, Structures, and Pharmacological Properties. Molecules, 30(4), 967. https://doi.org/10.3390/molecules30040967







