Structural Characterization and Anti-Gout Activity of a Novel Acidic Sanghuangporus vaninii Polysaccharide
Abstract
1. Introduction
2. Results
2.1. Extraction and Purification
2.2. Chemical Composition Assay
2.3. FT-IR Assay
2.4. Methylation Assay
2.5. NMR Assay
2.6. Cell
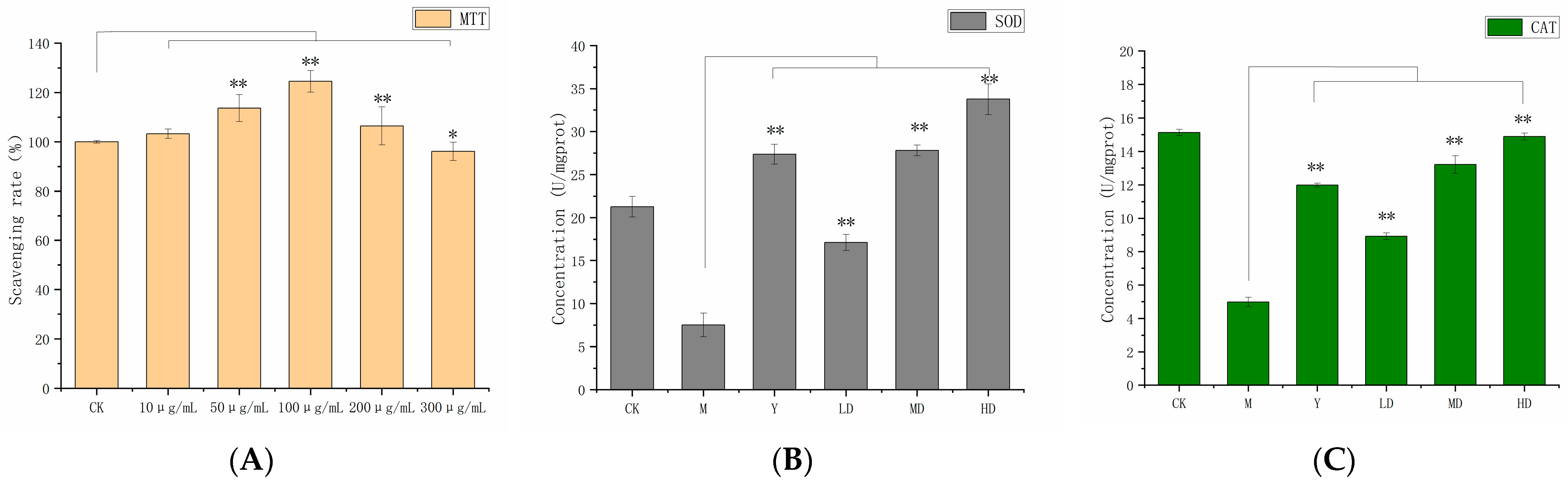
2.6.1. Cell Viability Assay
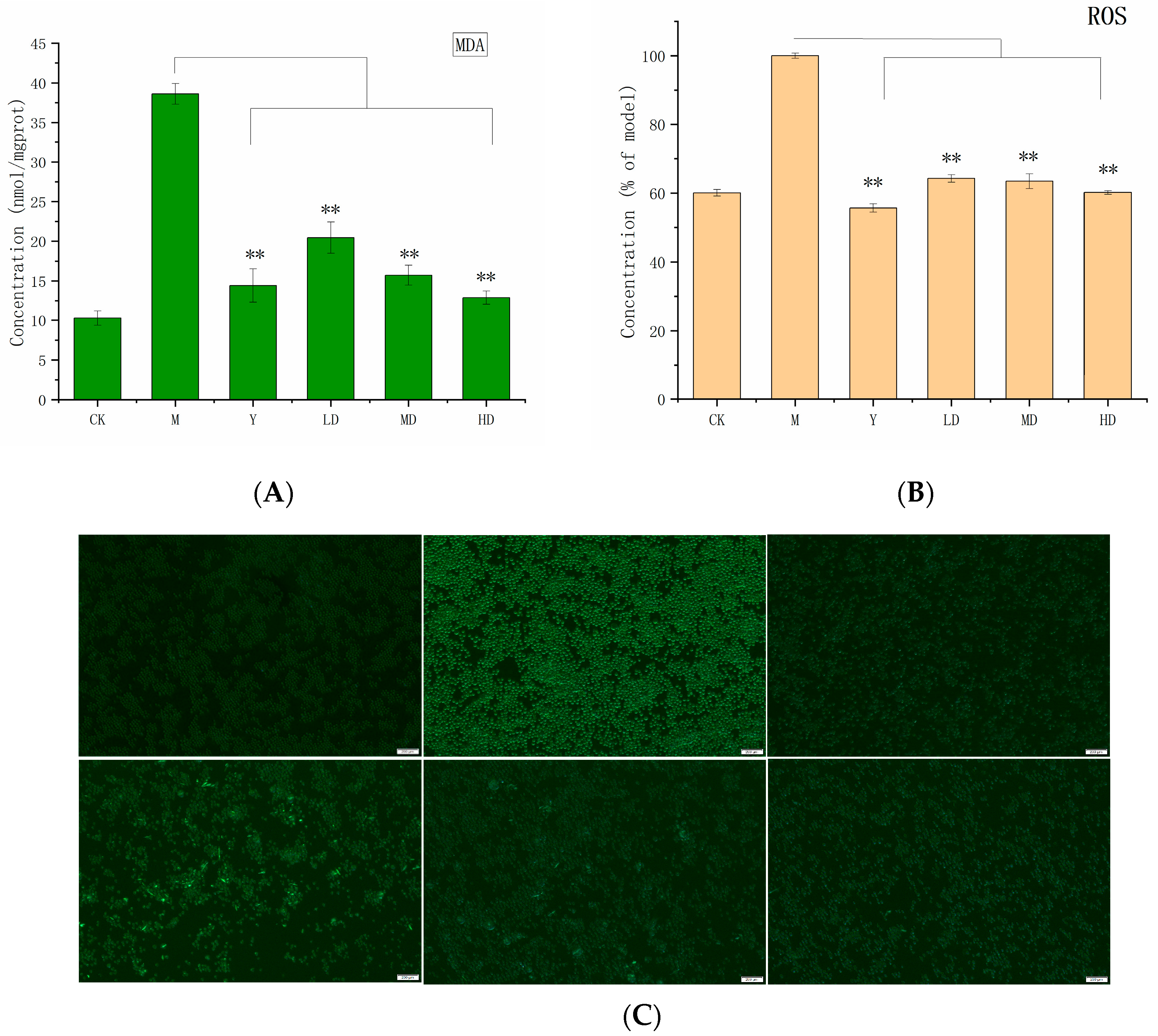
2.6.2. Antioxidant Activity Assay
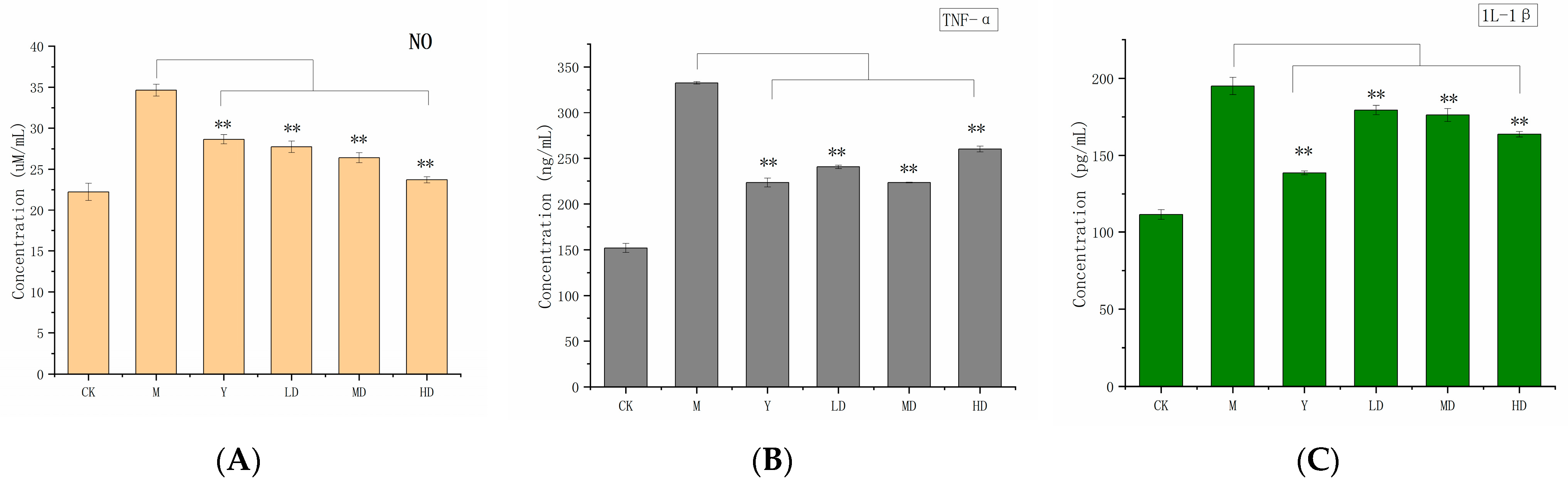
2.6.3. Anti-Inflammatory Activity
3. Discussion
4. Materials and Methods
4.1. Materials and Reagents
4.2. Polysaccharide Extraction
4.3. Separation and Purification
4.4. Structural Characterization
4.4.1. Chemical Composition Analysis
4.4.2. Molecular Weight Analysis
4.4.3. Monosaccharide Composition Analysis
4.4.4. Fourier Transform Infrared (FT-IR) Spectroscopy
4.4.5. Nuclear Magnetic Resonance (NMR) Spectroscopy
4.4.6. Methylation
4.5. Cell
4.5.1. Cell Culture
4.5.2. Cell Viability
4.5.3. Cell Experiment Groups and Treatments
4.5.4. Antioxidant Activity
4.5.5. Anti-Inflammatory Activity
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| MSU | Monosodium urate |
| 1L-1β | Interleukin-1β |
| ROS | Reactive oxygen species |
| XO | Xanthine oxidase |
| FBS | Fetal bovine serum |
| MTT | Methylthiazolyldiphenyl-tetrazolium |
| NO | Nitric oxide |
| ELISA | Enzyme-linked immunosorbent assay |
| HPGPC | High-performance gel permeation chromatography |
| RID | Refractive index detector |
| PMP | 1-phenyl-3-methyl-5-pyrazolone |
| TFA | Trifluoroacetic acid |
| D2O | Deuterium oxide |
| GC-MS | Gas chromatography–mass spectrometry |
| SOD | Superoxide dismutase |
| MDA | Malondialdehyde |
| CAT | Catalase |
| TNF-α | Tumor necrosis factor-α |
| SD | Standard deviation |
| ANOVA | Analysis of variance |
| PMAA | Partially methylated alditol acetate |
References
- Cross, M.; Ong, K.L.; Culbreth, G.T.; Steinmetz, J.D.; Cousin, E.; Lenox, H.; Kopec, J.A. Global, regional, and national burden of gout, 1990–2020, and projections to 2050: A systematic analysis of the Global Burden of Disease Study 2021. Lancet Rheumatol. 2024, 6, e507–e517. [Google Scholar] [CrossRef]
- Ge, X.; Zhang, Y.; Fang, R.L.; Zhao, J.J.; Huang, J.Y. Exploring the inhibition mechanism of interleukin-1-beta in gouty arthritis by polygonum cuspidatum using network pharmacology and molecular docking: A review. Medicine 2023, 102, e34396. [Google Scholar] [CrossRef]
- Zhao, P.; Hu, H.Z.; Chen, X.T.; Jiang, Q.Y.; Yu, X.Z.; Cen, X.L.; Lin, S.Q.; Mai, S.Q.; Pang, W.L.; Chen, J.X.; et al. Mild hyperthermia enhanced synergistic uric acid degradation and multiple ROS elimination for an effective acute gout therapy. J. Nanobiotechnol. 2024, 22, 275. [Google Scholar] [CrossRef]
- Kimura, Y.; Tsukui, D.; Kono, H. Uric Acid in Inflammation and the Pathogenesis of Atherosclerosis. Int. J. Mol. Sci. 2021, 22, 12394. [Google Scholar] [CrossRef]
- Zhang, X.; An, S.Y.; Zhou, L.Y.; Bao, Y.H. Comparison of Morphology, Components, and Activity of Four Species of Sanghuangporus Mushrooms (Agaricomycetes). Int. J. Med. Mushrooms 2025, 27, 45–65. [Google Scholar] [CrossRef]
- Wan, X.L.; Jin, X.; Wu, X.M.; Yang, X.; Lin, D.M.; Li, C.T.; Fu, Y.P.; Liu, Y.; Liu, X.Z.; Lv, J.H.; et al. Structural characterisation and antitumor activity against non-small cell lung cancer of polysaccharides from Sanghuangporus vaninii. Carbohydr. Polym. 2022, 276, 118798. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.L.; Xie, T.C.; Wang, Z.X.; Lin, C.; Li, Z.C.; Huo, J.; Li, Y.; Liu, C.; Gao, J.M.; Qi, J. Distinguishing Sanghuangporus from sanghuang-related fungi: A comparative and phylogenetic analysis based on mitogenomes. Appl. Microbiol. Biotechnol. 2024, 108, 423. [Google Scholar] [CrossRef] [PubMed]
- Song, J.L.; Wang, Z.W.; Chi, Y.; Zhang, Y.; Fang, C.Y.; Shu, Y.T.; Cui, J.; Bai, H.L.; Wang, J. Anti-gout activity and the interaction mechanisms between Sanghuangporus vaninii active components and xanthine oxidase. Bioorg. Chem. 2023, 133, 106394. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.C.; Chen, R.B.; Tang, C.H.; Li, L.Q.; Yan, J.K.; Zhang, H.A. Polysaccharide extracted from cultivated Sanghuangporous vaninii spores using three-phase partitioning with enzyme/ultrasound pretreatment: Physicochemical characteristics and its biological activity in vitro. Int. J. Biol. Macromol. 2023, 253, 126622. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.X.; Chen, X.F.; Gong, P. Classification, structure and mechanism of antiviral polysaccharides derived from edible and medicinal fungus. Int. J. Biol. Macromol. 2021, 183, 1753–1773. [Google Scholar] [CrossRef]
- Xia, Y.J.; Yang, C.Y.; Liu, X.F.; Wang, G.Q.; Xiong, Z.Q.; Song, X.; Yang, Y.J.; Zhang, H.; Ai, L.Z. Enhancement of triterpene production via in situ extractive fermentation of Sanghuangporus vaninii YC-1. Biotechnol. Appl. Biochem. 2022, 69, 2561–2572. [Google Scholar] [CrossRef]
- Liu, J.Z.; Song, J.Y.; Gao, F.S.; Chen, W.J.; Zong, Y.; Li, J.M.; He, Z.M.; Du, R. Extraction, Purification, and Structural Characterization of Polysaccharides from Sanghuangporus vaninii with Anti-Inflammatory Activity. Molecules 2023, 28, 6081. [Google Scholar] [CrossRef]
- Qu, Y.D.; Yang, H.X.; Li, S.Y.; Li, L.Z.; Li, Y.; Wang, D. The involvement of Th1 cell differentiation in the anti-tumor effect of purified polysaccharide from Sanghuangporus vaninii in colorectal cancer via multi-omics analysis. Int. J. Biol. Macromol. 2023, 237, 123927. [Google Scholar] [CrossRef]
- Sun, Z.; Li, Z.; Tan, Y.Y.; Wang, X.X.; Wang, C.X.; Dong, M.Y.; Liu, H.H.; Chen, H.; Li, Y.; Li, L.Z.; et al. Anti-Gouty Arthritis and Anti-Hyperuricemia Properties of Sanghuangporus vaninii and Inonotus hispidus in Rodent Models. Nutrients 2022, 14, 4421. [Google Scholar] [CrossRef]
- Yin, C.M.; Li, Y.H.; Li, J.T.; Fan, X.Z.; Yao, F.; Shi, D.F.; Cheng, Y.Q.; Liu, M.F.; Lu, Q.; Gao, H. Gastrointestinal digestion, probiotic fermentation behaviors and immunomodulatory effects of polysaccharides from Sanghuangporus vaninii. Int. J. Biol. Macromol. 2022, 223, 606–617. [Google Scholar] [CrossRef]
- Zhou, X.B.; Shi, Q.Y.; Li, J.H.; Quan, S.L.; Zhang, X.Y.; Gu, L.L.; Li, H.X.; Ju, Y.; Hu, M.; Li, Q. Medicinal fungus Phellinus igniarius alleviates gout in vitro by modulating TLR4/NF-kB/NLRP3 signaling. Front. Pharmacol. 2022, 13, 1011406. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Tao, Y.F.; Wang, X.S.; Gan, Y.H.; Zeng, Y.T.; Li, S.S.; Zhu, Q. Aqueous extract of Phellinus igniarius ameliorates hyperuricemia and renal injury in adenine/potassium oxonate-treated mice. Biomed. Pharmacother. 2024, 177, 116859. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.M.; Qiu, Z.C.; Li, L.Y.; Vidyarthi, S.K.; Zheng, Z.J.; Zhang, R.T. Structural characterization and antioxidant activities of one neutral polysaccharide and three acid polysaccharides from Ziziphus jujuba cv. Hamidazao: A comparison. Carbohydr. Polym. 2021, 261, 117879. [Google Scholar] [CrossRef] [PubMed]
- Ren, G.M.; Xu, L.M.; Lu, T.Y.; Yin, J.S. Structural characterization and antiviral activity of lentinan from Lentinus edodes mycelia against infectious hematopoietic necrosis virus. Int. J. Biol. Macromol. 2018, 115, 1202–1210. [Google Scholar] [CrossRef] [PubMed]
- Sims, I.M.; Carnachan, S.M.; Bell, T.J.; Hinkley, S.F.R. Methylation analysis of polysaccharides: Technical advice. Carbohydr. Polym. 2018, 188, 1–7. [Google Scholar] [CrossRef]
- Li, Y.X.; Sheng, Y.; Lu, X.C.; Guo, X.; Xu, G.Y.; Han, X.; An, L.P.; Du, P.G. Isolation and purification of acidic polysaccharides from Agaricus blazei Murill and evaluation of their lipid-lowering mechanism. Int. J. Biol. Macromol. 2020, 157, 276–287. [Google Scholar] [CrossRef]
- Li, H.P.; Feng, Y.B.; Sun, W.X.; Kong, Y.; Jia, L. Antioxidation, anti-inflammation and anti-fibrosis effect of phosphorylated polysaccharides from Pleurotus djamor mycelia on adenine-induced chronic renal failure mice. Int. J. Biol. Macromol. 2021, 170, 652–663. [Google Scholar] [CrossRef]
- Toto, A.; Wild, P.; Graille, M.; Turcu, V.; Crézé, C.; Hemmendinger, M.; Sauvain, J.J.; Bergamaschi, E.; Canu, I.G.; Hopf, N.B. Urinary Malondialdehyde (MDA) Concentrations in the General Population-A Systematic Literature Review and Meta-Analysis. Toxics 2022, 10, 160. [Google Scholar] [CrossRef]
- Shen, M.; Chen, X.; Huang, L.; Yu, Q.; Chen, Y.; Xie, J. Sulfated Mesona chinensis benth polysaccharide enhance the anti-inflammatory activities of cyclophosphamide-treated mice. J. Funct. Foods 2021, 76, 104321. [Google Scholar] [CrossRef]
- Hou, C.Y.; Chen, L.L.; Yang, L.Z.; Ji, X.L. An insight into anti-inflammatory effects of natural polysaccharides. Int. J. Biol. Macromol. 2020, 153, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.G.; Tu, S.J.; Huang, C.M.; Chen, Y.C.; Chiang, H.S.; Lee, Y.T.; Yen, J.C.; Lin, C.L.; Chung, C.C.; Liu, T.C.; et al. Single-cell RNA sequencing of immune cells in patients with acute gout. Sci. Rep. 2022, 12, 22130. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.F.; Song, T.T.; Chen, J.F.; Lv, G.Y. Recovery of a hypolipidemic polysaccharide from artificially cultivated Sanghuangporus vaninii with an effective method. Front. Nutr. 2023, 9, 1095556. [Google Scholar] [CrossRef]
- Wang, H.; Ma, J.X.; Zhou, M.; Si, J.; Cui, B.K. Current advances and potential trends of the polysaccharides derived from medicinal mushrooms sanghuang. Front. Microbiol. 2022, 13, 965934. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Xue, Q.; Wang, H.; Jin, R.; Liu, X.; Li, L.; Liu, Y.; Wang, D.; Hu, M. Structural characterization and osteogenic potential of polysaccharide purified from Sanghuangporus vaninii. Carbohydr. Polym. 2025, 367, 124018. [Google Scholar] [CrossRef]
- Ji, X.L.; Guo, J.H.; Ding, D.Q.; Gao, J.; Hao, L.R.; Guo, X.D.; Liu, Y.Q. Structural characterization and antioxidant activity of a novel high-molecular-weight polysaccharide from Ziziphus Jujuba cv. Muzao. J. Food Meas. Charact. 2022, 16, 2191–2200. [Google Scholar] [CrossRef]
- Lee, Q.C.; Xue, Z.X.; Luo, Y.J.; Lin, Y.P.; Lai, M.Y.; Xu, H.Y.; Liu, B.; Zheng, M.F.; Lv, F.; Zeng, F. Low molecular weight polysaccharide of Tremella fuciformis exhibits stronger antioxidant and immunomodulatory activities than high molecular weight polysaccharide. Int. J. Biol. Macromol. 2024, 281, 136097. [Google Scholar] [CrossRef]
- Chen, X.; Ni, L.; Fu, X.; Wang, L.; Duan, D.; Huang, L.; Xu, J.; Gao, X. Molecular mechanism of anti-inflammatory activities of a novel sulfated galactofucan from Saccharina japonica. Mar. Drugs 2021, 19, 430. [Google Scholar] [CrossRef]
- Liu, H.M.; Liu, X.Y.; Yan, Y.Y.; Gao, J.H.; Qin, Z.; Wang, X.D. Structural properties and antioxidant activities of polysaccharides isolated from sunflower meal after oil extraction. Arab. J. Chem. 2021, 14, 103420. [Google Scholar] [CrossRef]
- Guo, Q.; Zhao, L.Y.; Zhu, Y.H.; Wu, J.; Hao, C.T.; Song, S.; Shi, W. Optimization of culture medium for Sanghuangporus vaninii and a study on its therapeutic effects on gout. Biomed. Pharmacother. 2021, 135, 111194. [Google Scholar] [CrossRef]
- Liu, J.Z.; Song, J.Y.; Chen, W.J.; Sun, L.; Zhao, Y.; Zong, Y.; He, Z.M.; Du, R. Assessment of cytotoxicity, acute, subacute toxicities and antioxidant activities (in vitro) of Sanghuangporus vaninii crude polysaccharide. J. Ethnopharmacol. 2024, 319, 117284. [Google Scholar] [CrossRef]
- Zeng, D.Y.; Yin, C.Y.; Wei, H.N.; Li, Y.Y.; Yang, Y.Q.; Nie, H.M.; Pan, Y.S.; Xu, R.Y.; Tai, Y.; Du, J.Y.; et al. Activation of Nrf2 antioxidant signaling alleviates gout arthritis pain and inflammation. Biomed. Pharmacother. 2024, 170, 115957. [Google Scholar] [CrossRef]
- Chelombitko, M.A. Role of Reactive Oxygen Species in Inflammation: A Minireview. Moscow Univ. Biol. Sci. Bull. 2018, 73, 242–246. [Google Scholar] [CrossRef]
- Chen, Y.; Shi, J.F.; Wu, Y.Z.; Guo, Z.Y.; Li, S.H.; Li, W.P.; Wu, Z.H.; Wang, H.J.; Jiang, H.F.; Jiang, Z.Y. NADH Photosynthesis System with Affordable Electron Supply and Inhibited NADH Oxidation. Angew. Chem. Int. Edit. 2023, 62, e202310238. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.F.; Lv, G.Y.; Shen, M.; Song, T.T.; Juan, P. Protective effect of polysaccharide from the artificially cultivated Sanghuangporus vaninii against H2O2-induced toxicity in vitro and in zebrafish models. Arab. J. Chem. 2023, 16, 105115. [Google Scholar]
- Xie, Y.J.; Tao, M.X.; Yan, X.D.; Fan, X.X.; Bayoude, A.; Lu, Y.; Zhao, S.L.; Yu, B.Y.; Li, R.S. Acidic polysaccharide CP-2 from Dioscoreae Rhizoma ameliorated acute alcoholic liver injury through the gut-liver axis and AMPK/PPAR pathway. Int. J. Biol. Macromol. 2025, 310, 143145. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Liu, H.M.; Zhai, Y.; Li, Y.; Zhu, X.; Zhang, W. Laminarin protects against hydrogen peroxide-induced oxidative damage in MRC-5 cells possibly via regulating NRF2. PeerJ 2017, 5, e3642. [Google Scholar] [CrossRef]
- Sun, X.; Yang, L.; Sun, H.; Sun, Y.; Wei, S.Y.; Han, Y.; Wang, W.K.; Kong, L.; Wang, X.J. Traditional Chinese Medicine (TCM) and related active compounds in the treatment of gout: Regulation of signaling pathways and urate transporters. Front. Pharmacol. 2023, 14, 1275974. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.X.; Wang, Q.L.; Deng, W.W.; Sun, C.Y.; Wei, Q.Y.; Adu-Frimpong, M.; Shi, J.X.; Yu, J.N.; Xu, X.M. Anti-hyperuricemic and anti-gouty arthritis activities of polysaccharide purified from Lonicera japonica in model rats. Int. J. Biol. Macromol. 2019, 123, 801–809. [Google Scholar] [CrossRef]
- Wang, Y.; Lin, Z.J.; Zhang, B.; Jiang, Z.X.; Guo, F.F.; Yang, T. Cichorium intybus L. Extract Suppresses Experimental Gout by Inhibiting the NF-κB and NLRP3 Signalling Pathways. Int. J. Mol. Sci. 2019, 20, 4921. [Google Scholar] [CrossRef]
- Yue, F.F.; Zhang, J.R.; Xu, J.X.; Niu, T.F.; Lü, X.; Liu, M.S. Effects of monosaccharide composition on quantitative analysis of total sugar content by phenol-sulfuric acid method. Front. Nutr. 2022, 9, 963318. [Google Scholar] [CrossRef] [PubMed]
- Filgueiras, M.F.; Borges, E.M. Quick and Cheap Colorimetric Quantification of Proteins Using 96-Well-Plate Images. J. Chem. Educ. 2022, 99, 1778–1787. [Google Scholar] [CrossRef]
- Zheng, L.; Ma, Y.H.; Zhang, Y.J.; Meng, Q.J.; Yang, J.H.; Wang, B.L.; Liu, Q.G.; Cai, L.; Gong, W.L.; Yang, Y.; et al. Increased antioxidant activity and improved structural characterization of sulfuric acid-treated stepwise degraded polysaccharides from Pholiota nameko PN-01. Int. J. Biol. Macromol. 2021, 166, 1220–1229. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Liu, J.X.; Wang, X.; Hu, H.W.; Zhang, Y.R.; Liu, T.T.; Zhao, H. Structure characterization and antioxidant activity of carboxymethylated polysaccharide from Pholiota nameko. J. Food Biochem. 2022, 46, e14121. [Google Scholar] [CrossRef]
- Ji, X.L.; Yan, Y.Z.; Hou, C.Y.; Shi, M.M.; Liu, Y.Q. Structural characterization of a galacturonic acid-rich polysaccharide from Ziziphus jujuba cv. Muzao. Int. J. Biol. Macromol. 2020, 147, 844–852. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, T.T.; Wang, X.; Zhou, L.Y.; Qi, J.; An, S.Y. Structural characterization, antioxidant activity and anti-inflammatory of the phosphorylated polysaccharide from Pholiota nameko. Front. Nutr. 2022, 9, 976552. [Google Scholar] [CrossRef]
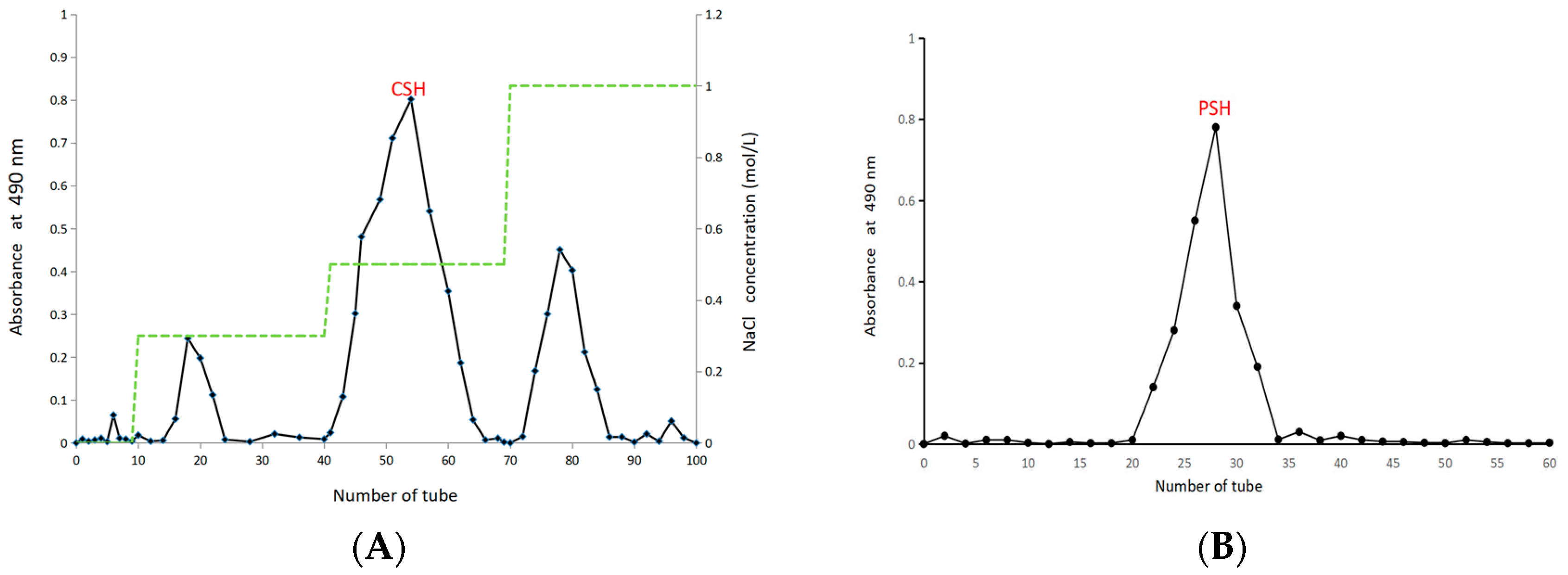
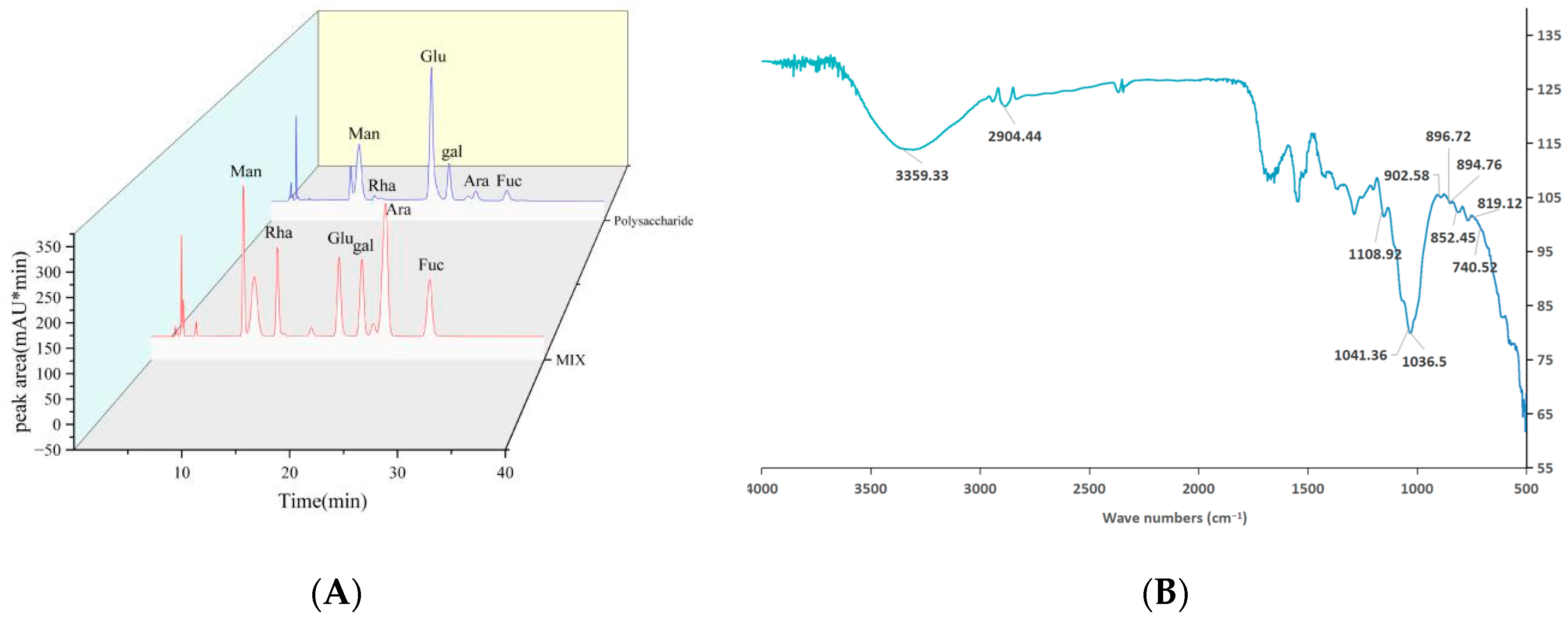
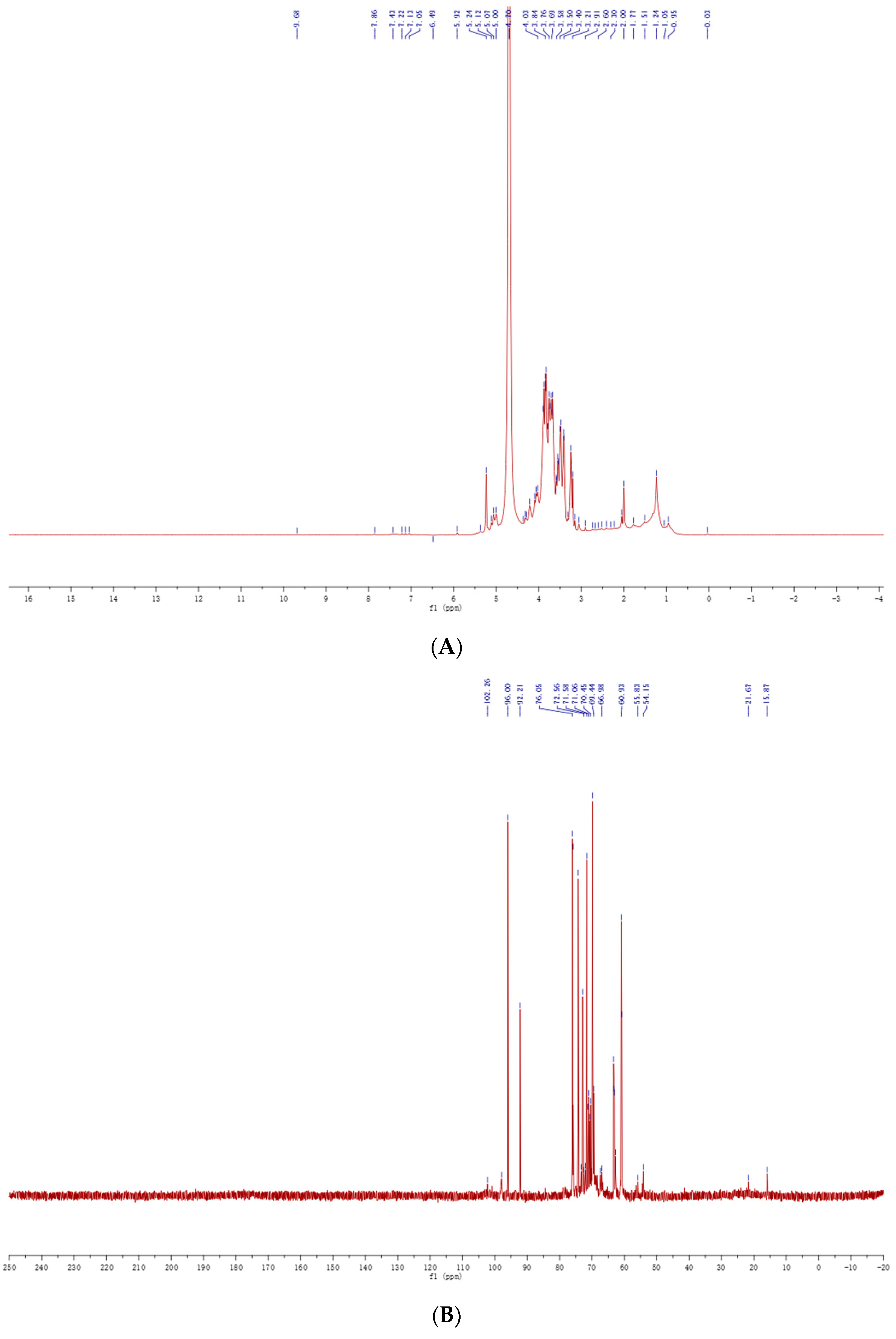
| Fraction | Content | Monosaccharide | Content (%, Area) |
|---|---|---|---|
| Total sugar (%) | 93.94 ± 3.55 | Man | 7.6 |
| Protein (%) | 1.09 ± 0.32 | Rha | 1.6 |
| Uronic acid (%) | 17.05 ± 2.71 | Glc | 66.5 |
| Gal | 16.7 | ||
| Molecular weight (Da) | 5.25 × 104 | Ara | 1.7 |
| Fuc | 5.9 |
| Number | Methylation Product | Linkage Pattern | Area Ratio (%) |
|---|---|---|---|
| 1 | 2,3,5,6-penta-O-acetyl-4-O-methyl-D-glucitol | →3,6)-Glcp-(2→ | 12.4 |
| 2 | 3,5,6-tetra-O-acetyl-2,4-di-O-methyl-D-glucitol | →4)-Glcp-(2→ | 43.5 |
| 3 | 5-di-O-acetyl-2,3,4,6-tetra-O-methyl-D-glucitol | Glcp-(1→ | 11.2 |
| 4 | 3,5-tri-O-acetyl-2,4,6-tri-O-methyl-D-galactitol/ | →3)-Galp-(1→ | 16.4 |
| 5 | 5-di-O-acetyl-2,3,4-tri-O-methyl-D-arabinitol | Araf-(1→ | 2.1 |
| 6 | 2,3,4,5-penta-O-acetyl-D-arabinitol | →3,5)-Araf-(2→ | 1.7 |
| 7 | 3,4,5-tetra-O-acetyl-6-deoxy-2-O-methyl-L-galactitol | →4)-Fucp-(3→ | 5.5 |
| 8 | 2,5-tri-O-acetyl-6-deoxy-3,4-di-O-methyl-L-mannitol | →2)-Rhap-(1→ | 2.1 |
| 9 | 5,6-tri-O-acetyl-2-(acetylmethylamino)-2-deoxy-3,4-di-O-methyl-D-mannitol | →6)-Manp-(1→ | 5.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; An, S.; Zhou, L.; Chen, C.; Yang, X. Structural Characterization and Anti-Gout Activity of a Novel Acidic Sanghuangporus vaninii Polysaccharide. Molecules 2025, 30, 3536. https://doi.org/10.3390/molecules30173536
Zhang X, An S, Zhou L, Chen C, Yang X. Structural Characterization and Anti-Gout Activity of a Novel Acidic Sanghuangporus vaninii Polysaccharide. Molecules. 2025; 30(17):3536. https://doi.org/10.3390/molecules30173536
Chicago/Turabian StyleZhang, Xu, Siyu An, Lanying Zhou, Chen Chen, and Xue Yang. 2025. "Structural Characterization and Anti-Gout Activity of a Novel Acidic Sanghuangporus vaninii Polysaccharide" Molecules 30, no. 17: 3536. https://doi.org/10.3390/molecules30173536
APA StyleZhang, X., An, S., Zhou, L., Chen, C., & Yang, X. (2025). Structural Characterization and Anti-Gout Activity of a Novel Acidic Sanghuangporus vaninii Polysaccharide. Molecules, 30(17), 3536. https://doi.org/10.3390/molecules30173536






