Fluoride-Mediated Synthesis of Co(OH)F and Electronic Structure Optimization for Enhanced Water Oxidation Performance
Abstract
1. Introduction
2. Result and Discussion
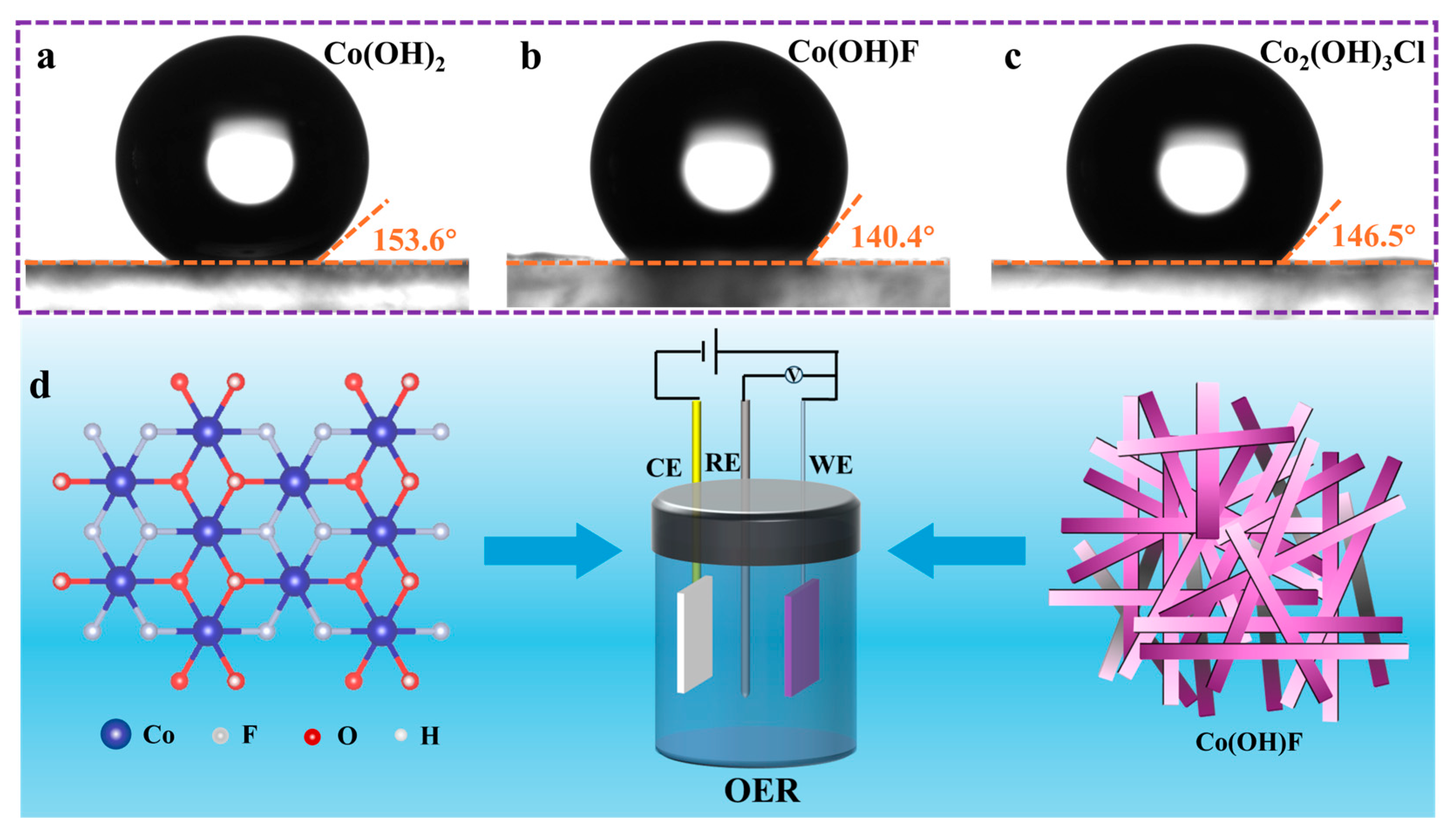
2.1. Morphology and Structure Characterization
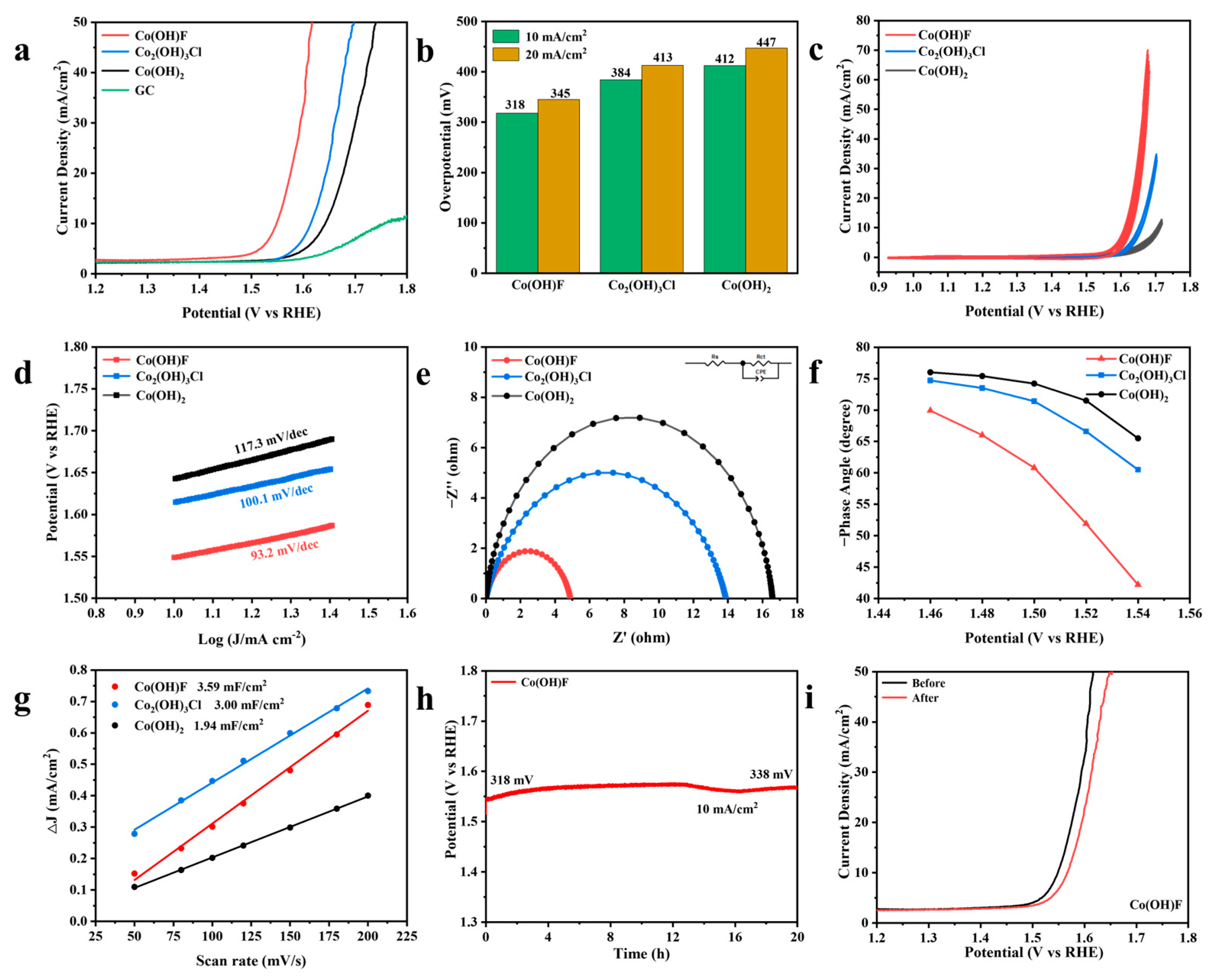
2.2. Electrochemical Performance
3. Experimental Section
3.1. Materials and Chemicals
3.2. Preparation of Electrode Materials
3.3. Characterization
3.4. Electrochemical Measurements
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, X.-P.; Wang, H.-Y.; Zheng, H.; Zhang, W.; Cao, R. O–O bond formation mechanisms during the oxygen evolution reaction over synthetic molecular catalysts. Chin. J. Catal. 2021, 42, 1253–1268. [Google Scholar] [CrossRef]
- Deng, B.; Yu, G.; Zhao, W.; Long, Y.; Yang, C.; Du, P.; He, X.; Zhang, Z.; Huang, K.; Li, X.; et al. A self-circulating pathway for the oxygen evolution reaction. Energy Environ. Sci. 2023, 16, 5210–5219. [Google Scholar] [CrossRef]
- Moysiadou, A.; Lee, S.; Hsu, C.S.; Chen, H.M.; Hu, X. Mechanism of Oxygen Evolution Catalyzed by Cobalt Oxyhydroxide: Cobalt Superoxide Species as a Key Intermediate and Dioxygen Release as a Rate-Determining Step. J. Am. Chem. Soc. 2020, 142, 11901–11914. [Google Scholar] [CrossRef] [PubMed]
- Fabbri, E.; Schmidt, T.J. Oxygen Evolution Reaction—The Enigma in Water Electrolysis. ACS Catal. 2018, 8, 9765–9774. [Google Scholar] [CrossRef]
- Liang, Q.; Brocks, G.; Bieberle-Hütter, A. Oxygen evolution reaction (OER) mechanism under alkaline and acidic conditions. J. Phys. Energy 2021, 3, 026001. [Google Scholar] [CrossRef]
- Xu, X.; Pan, Y.; Ge, L.; Chen, Y.; Mao, X.; Guan, D.; Li, M.; Zhong, Y.; Hu, Z.; Peterson, V.K.; et al. High-Performance Perovskite Composite Electrocatalysts Enabled by Controllable Interface Engineering. Small 2021, 17, 2101573. [Google Scholar] [CrossRef]
- Plevová, M.; Hnát, J.; Bouzek, K. Electrocatalysts for the oxygen evolution reaction in alkaline and neutral media. A comparative review. J. Power Sources 2021, 507, 230072. [Google Scholar] [CrossRef]
- Mohammed-Ibrahim, J. A review on NiFe-based electrocatalysts for efficient alkaline oxygen evolution reaction. J. Power Sources 2020, 448, 227375. [Google Scholar] [CrossRef]
- Zhang, H.; Luo, Y.; Chu, P.K.; Liu, Q.; Liu, X.; Zhang, S.; Luo, J.; Wang, X.; Hu, G. Recent advances in non-noble metal-based bifunctional electrocatalysts for overall seawater splitting. J. Alloys Compd. 2022, 922, 166113. [Google Scholar] [CrossRef]
- Jamadar, A.S.; Sutar, R.; Patil, S.; Khandekar, R.; Yadav, J.B. Progress in metal oxide-based electrocatalysts for sustainable water splitting. Mater. Rep. Energy 2024, 4, 100283. [Google Scholar] [CrossRef]
- Gupta, S.; Fernandes, R.; Patel, R.; Spreitzer, M.; Patel, N. A review of cobalt-based catalysts for sustainable energy and environmental applications. Appl. Catal. A Gen. 2023, 661, 119254. [Google Scholar] [CrossRef]
- Zhang, R.; van Straaten, G.; di Palma, V.; Zafeiropoulos, G.; van de Sanden, M.C.M.; Kessels, W.M.M.; Tsampas, M.N.; Creatore, M. Electrochemical Activation of Atomic Layer-Deposited Cobalt Phosphate Electrocatalysts for Water Oxidation. ACS Catal. 2021, 11, 2774–2785. [Google Scholar] [CrossRef]
- Cui, H.; Zhao, Y.; Ren, W.; Wang, M.; Liu, Y. Large scale selective synthesis of α-Co(OH)2 and β-Co(OH)2 nanosheets through a fluoride ions mediated phase transformation process. J. Alloys Compd. 2013, 562, 33–37. [Google Scholar] [CrossRef]
- Zhang, X.; Zhong, H.; Zhang, Q.; Zhang, Q.; Wu, C.; Yu, J.; Ma, Y.; An, H.; Wang, H.; Zou, Y.; et al. High-spin Co3+ in cobalt oxyhydroxide for efficient water oxidation. Nat. Commun. 2024, 15, 1383. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Budiyanto, E.; Tuysuz, H. Principles of Water Electrolysis and Recent Progress in Cobalt-, Nickel-, and Iron-Based Oxides for the Oxygen Evolution Reaction. Angew. Chem. Int. Ed. Engl. 2022, 61, e202103824. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.H.; Han, M.H.; Ko, Y.J.; Min, B.K.; Chae, K.H.; Oh, H.S. Electrode reconstruction strategy for oxygen evolution reaction: Maintaining Fe-CoOOH phase with intermediate-spin state during electrolysis. Nat. Commun. 2022, 13, 605. [Google Scholar] [CrossRef]
- Wan, S.; Qi, J.; Zhang, W.; Wang, W.; Zhang, S.; Liu, K.; Zheng, H.; Sun, J.; Wang, S.; Cao, R. Hierarchical Co(OH)F Superstructure Built by Low-Dimensional Substructures for Electrocatalytic Water Oxidation. Adv. Mater. 2017, 29, 1700286. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Li, Y.; Zhang, Z.; Tang, W.; Zhao, J.; Yang, T.; Wu, Z.; Bao, W. Co(OH)F/Ni(OH)2@FeOOH core–shell heterostructure as a high-efficiency electrocatalyst with strong electron interactions towards boosting the oxygen evolution reaction. Int. J. Hydrogen Energy 2024, 51, 890–897. [Google Scholar] [CrossRef]
- Liang, R.; Zhang, B.; Du, Y.; Han, X.; Li, S.; Xu, P. Understanding the Anion Effect of Basic Cobalt Salts for the Electrocatalytic Oxygen Evolution Reaction. ACS Catal. 2023, 13, 8821–8829. [Google Scholar] [CrossRef]
- Chen, P.; Zhou, T.; Wang, S.; Zhang, N.; Tong, Y.; Ju, H.; Chu, W.; Wu, C.; Xie, Y. Dynamic Migration of Surface Fluorine Anions on Cobalt-Based Materials to Achieve Enhanced Oxygen Evolution Catalysis. Angew. Chem. Int. Ed. 2018, 57, 15471–15475. [Google Scholar] [CrossRef]
- Dong, Q.; Zhong, Q.; Zhou, J.; Li, Y.; Wang, Y.; Cai, J.; Yu, S.; He, X.; Zhang, S. Construction of Heterostructured Ni3S2@V-NiFe(III) LDH for Enhanced OER Performance. Molecules 2024, 29, 6018. [Google Scholar] [CrossRef] [PubMed]
- Lin, Q.; Guo, D.; Zhou, L.; Yang, L.; Jin, H.; Li, J.; Fang, G.; Chen, X.; Wang, S. Tuning the Interface of Co1−xS/Co(OH)F by Atomic Replacement Strategy toward High-Performance Electrocatalytic Oxygen Evolution. ACS Nano 2022, 16, 15460–15470. [Google Scholar] [CrossRef]
- Zha, Q.; Xu, W.; Li, X.; Ni, Y. Chlorine-doped alpha-Co(OH)2 hollow nano-dodecahedrons prepared by a ZIF-67 self-sacrificing template route and enhanced OER catalytic activity. Dalton Trans. 2019, 48, 12127–12136. [Google Scholar] [CrossRef]
- Kou, Y.; Liu, J.; Li, Y.; Qu, S.; Ma, C.; Song, Z.; Han, X.; Deng, Y.; Hu, W.; Zhong, C. Electrochemical Oxidation of Chlorine-Doped Co(OH)2 Nanosheet Arrays on Carbon Cloth as a Bifunctional Oxygen Electrode. ACS Appl. Mater. Interfaces 2018, 10, 796–805. [Google Scholar] [CrossRef]
- Meng, X.; Deng, D. A new approach to facilely synthesize crystalline Co2(OH)3Cl microstructures in an eggshell reactor system. CrystEngComm 2017, 19, 2953–2959. [Google Scholar] [CrossRef]
- Ben Yahia, H.; Shikano, M.; Tabuchi, M.; Kobayashi, H.; Avdeev, M.; Tan, T.T.; Liu, S.; Ling, C.D. Synthesis and characterization of the crystal and magnetic structures and properties of the hydroxyfluorides Fe(OH)F and Co(OH)F. Inorg. Chem. 2014, 53, 365–374. [Google Scholar] [CrossRef]
- Jiu, H.; Wei, H.; Wang, C.; Che, S.; Guo, Z.; Han, Y.; Xu, Q.; Yu, X.; Zhang, L. Construction of Co(OH)F/Ni(OH)2@Fe(OH)3 core-shell heterojunction on nickel foam for efficient oxygen evolution. Int. J. Hydrogen Energy 2022, 47, 33719–33727. [Google Scholar] [CrossRef]
- Liang, Z.; Yang, Z.; Huang, Z.; Qi, J.; Chen, M.; Zhang, W.; Zheng, H.; Sun, J.; Cao, R. Novel insight into the epitaxial growth mechanism of six-fold symmetrical β-Co(OH)2/Co(OH)F hierarchical hexagrams and their water oxidation activity. Electrochim. Acta 2018, 271, 526–536. [Google Scholar] [CrossRef]
- Zhao, X.; Li, M.; Liang, J.; Li, L. Improving the electrocatalytic oxygen evolution byin situconstructing 1D Co9S8/Co(OH)F heterointerfaces. New J. Chem. 2022, 46, 23060–23065. [Google Scholar] [CrossRef]
- Peng, Y.; Wang, P.; Li, J.; Wu, J.; Lin, F.; Zhang, D. Optimizing the chlorine evolution reaction performance of Co(OH)2 catalyst for enhanced antifouling ability. J. Ind. Eng. Chem. 2023, 118, 91–100. [Google Scholar] [CrossRef]
- Gao, P.; Zeng, Y.; Tang, P.; Wang, Z.; Yang, J.; Hu, A.; Liu, J. Understanding the Synergistic Effects and Structural Evolution of Co(OH)2 and Co3O4 toward Boosting Electrochemical Charge Storage. Adv. Funct. Mater. 2021, 32, 2108644. [Google Scholar] [CrossRef]
- Zheng, W.; Zheng, X.-R.; Lu, Q.; Cao, Y.-H.; Wang, Y.; Fu, H.-P.; Zhang, J.-F.; Deng, Y.-D.; Hu, W.-B. Spontaneous anchoring Cl into α-Co(OH)2 as efficient and stable oxygen reduction electrocatalysts for seawater battery. Rare Met. 2024, 43, 3074–3083. [Google Scholar] [CrossRef]
- Liu, Q.; Liao, X.; Tang, Y.; Wang, J.; Lv, X.; Pan, X.; Lu, R.; Zhao, Y.; Yu, X.-Y.; Wu, H.B. Low-coordinated cobalt arrays for efficient hydrazine electrooxidation. Energy Environ. Sci. 2022, 15, 3246–3256. [Google Scholar] [CrossRef]
- Zhou, S.; Jang, H.; Qin, Q.; Li, Z.; Kim, M.G.; Li, C.; Liu, X.; Cho, J. Three-dimensional hierarchical Co(OH)F nanosheet arrays decorated by single-atom Ru for boosting oxygen evolution reaction. Sci. China Mater. 2020, 64, 1408–1417. [Google Scholar] [CrossRef]
- Fang, D.; He, F.; Xie, J.; Xue, L. Calibration of Binding Energy Positions with C1s for XPS Results. J. Wuhan Univ. Technol.-Mater. Sci. Ed. 2020, 35, 711–718. [Google Scholar] [CrossRef]
- Chen, X.; Wang, X.; Fang, D. A review on C1s XPS-spectra for some kinds of carbon materials. Fuller. Nanotub. Carbon Nanostruct. 2020, 28, 1048–1058. [Google Scholar] [CrossRef]
- Jiang, H.; He, Q.; Li, X.; Su, X.; Zhang, Y.; Chen, S.; Zhang, S.; Zhang, G.; Jiang, J.; Luo, Y.; et al. Tracking Structural Self-Reconstruction and Identifying True Active Sites toward Cobalt Oxychloride Precatalyst of Oxygen Evolution Reaction. Adv. Mater. 2019, 31, 1805127. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Sun, F.; Zhou, S.; Hu, W.; Zhang, H.; Dong, J.; Jiang, Z.; Zhao, J.; Li, J.; Yan, W.; et al. Atomic-level insight into super-efficient electrocatalytic oxygen evolution on iron and vanadium co-doped nickel (oxy)hydroxide. Nat. Commun. 2018, 9, 2885. [Google Scholar] [CrossRef]
- Cai, J.; He, X.; Dong, Q.; Li, Y.; Su, P.; Qu, D.; Liu, J.; Lu, Y.; Jin, Q.; Sun, Z. Employing shielding effect of intercalated cinnamate anion in NiFe LDH for stable and efficient seawater oxidation. Surf. Interfaces 2024, 51, 104772. [Google Scholar] [CrossRef]
- Zhang, R.; Cheng, L.; Wang, Z.; Kong, F.; Tsegazab, Y.; Lv, W.; Wang, W. Ni3S2-Co9S8 heterostructure nanowires supported on Ni foam as highly efficient and stable electrocatalyst for oxygen evolution reaction. Appl. Surf. Sci. 2020, 526, 146753. [Google Scholar] [CrossRef]
- Anantharaj, S.; Kundu, S. Do the Evaluation Parameters Reflect Intrinsic Activity of Electrocatalysts in Electrochemical Water Splitting? ACS Energy Lett. 2019, 4, 1260–1264. [Google Scholar] [CrossRef]
- Cossar, E.; Houache, M.S.E.; Zhang, Z.; Baranova, E.A. Comparison of electrochemical active surface area methods for various nickel nanostructures. J. Electroanal. Chem. 2020, 870, 114246. [Google Scholar] [CrossRef]
- Kim, B.K.; Kim, M.J.; Kim, J.J. Impact of Surface Hydrophilicity on Electrochemical Water Splitting. ACS Appl. Mater. Interfaces 2021, 13, 11940–11947. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.J.; Jiang, L. Design and Creation of Superwetting/Antiwetting Surfaces. Adv. Mater. 2006, 18, 3063–3078. [Google Scholar] [CrossRef]
- Zhu, G.; Zhao, S.; Yu, Y.; Fan, X.; Liu, K.; Quan, X.; Liu, Y. Tuning Local Proton Concentration and *OOH Intermediate Generation for Efficient Acidic H2O2 Electrosynthesis at Ampere-Level Current Density. Angew. Chem. Int. Ed. Engl. 2025, 64, e202503626. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dong, Q.; Li, Y.; Liu, J.; Wen, Y.; Wang, J.; Mo, H.; Jin, Q.; Zhang, S.; He, X. Fluoride-Mediated Synthesis of Co(OH)F and Electronic Structure Optimization for Enhanced Water Oxidation Performance. Molecules 2025, 30, 3529. https://doi.org/10.3390/molecules30173529
Dong Q, Li Y, Liu J, Wen Y, Wang J, Mo H, Jin Q, Zhang S, He X. Fluoride-Mediated Synthesis of Co(OH)F and Electronic Structure Optimization for Enhanced Water Oxidation Performance. Molecules. 2025; 30(17):3529. https://doi.org/10.3390/molecules30173529
Chicago/Turabian StyleDong, Qianqian, Yuhao Li, Jihao Liu, Yaru Wen, Junjie Wang, Haining Mo, Qianqian Jin, Shaohui Zhang, and Xiong He. 2025. "Fluoride-Mediated Synthesis of Co(OH)F and Electronic Structure Optimization for Enhanced Water Oxidation Performance" Molecules 30, no. 17: 3529. https://doi.org/10.3390/molecules30173529
APA StyleDong, Q., Li, Y., Liu, J., Wen, Y., Wang, J., Mo, H., Jin, Q., Zhang, S., & He, X. (2025). Fluoride-Mediated Synthesis of Co(OH)F and Electronic Structure Optimization for Enhanced Water Oxidation Performance. Molecules, 30(17), 3529. https://doi.org/10.3390/molecules30173529




