Tracking Lead: Potentiometric Tools and Technologies for a Toxic Element
Abstract
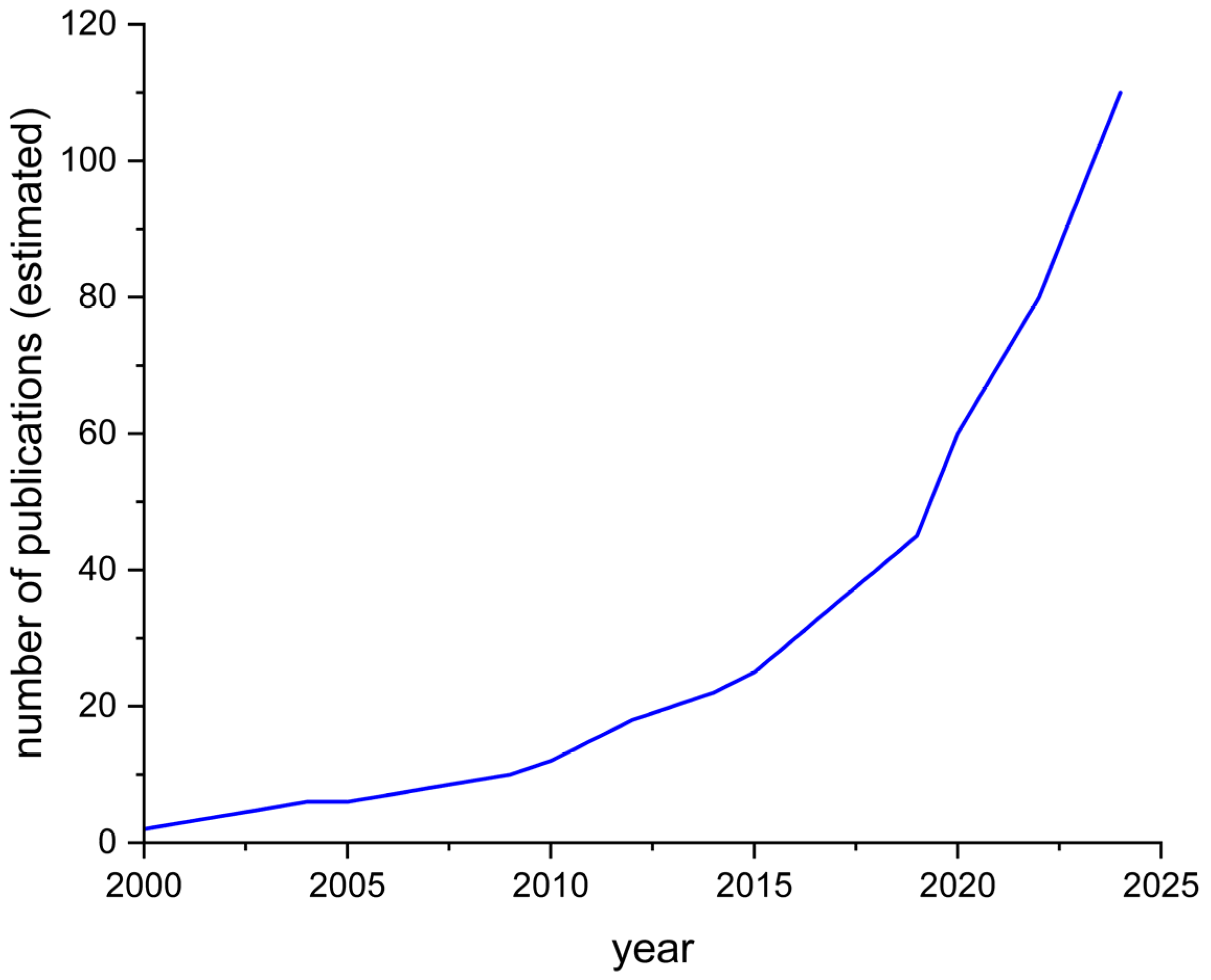
1. Introduction
2. Toxicity of Lead
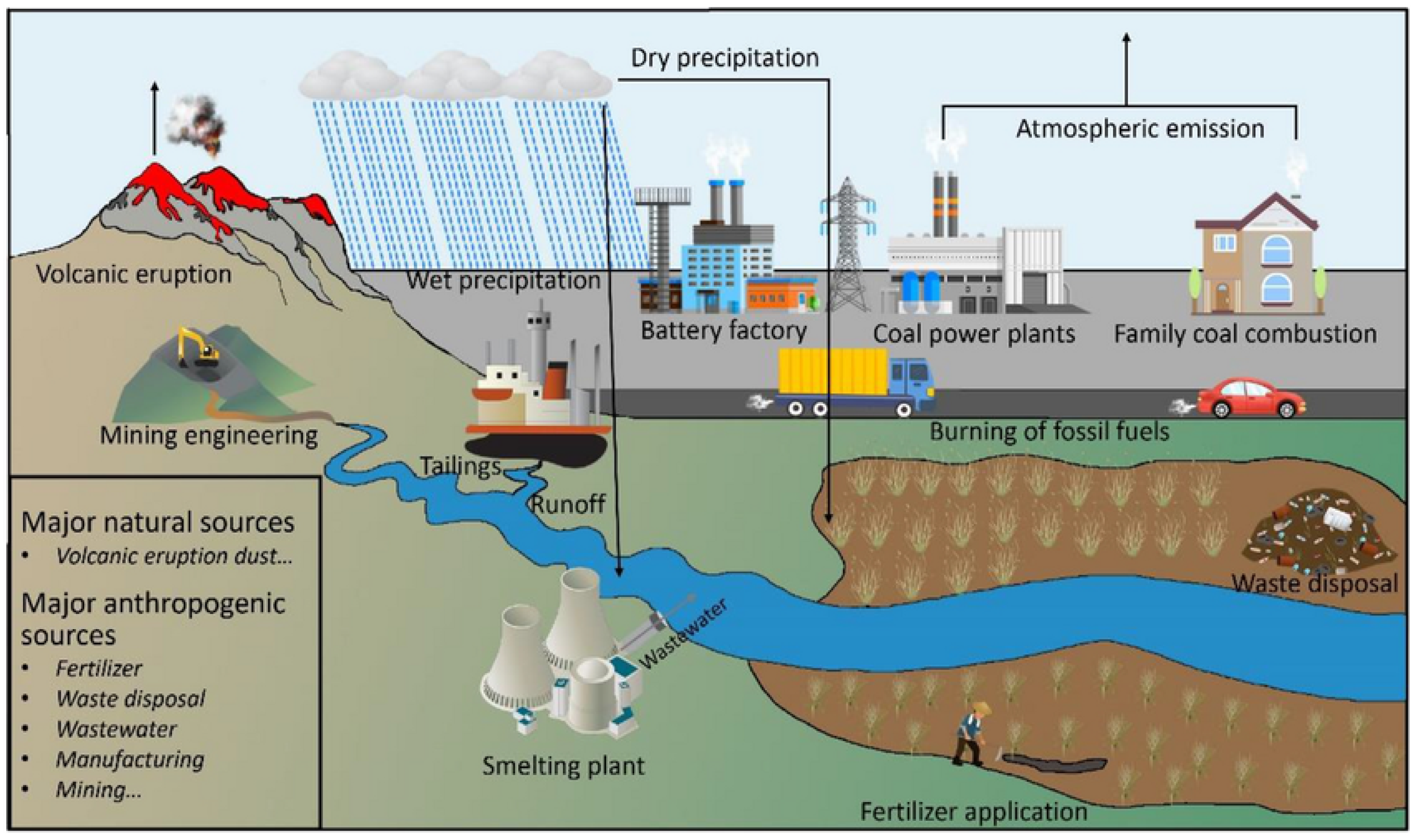
2.1. Sources of Lead Pollution
2.2. Effects on Environment
2.3. Effects on Human
3. Background of Potentiometric Determination of Metal Ions
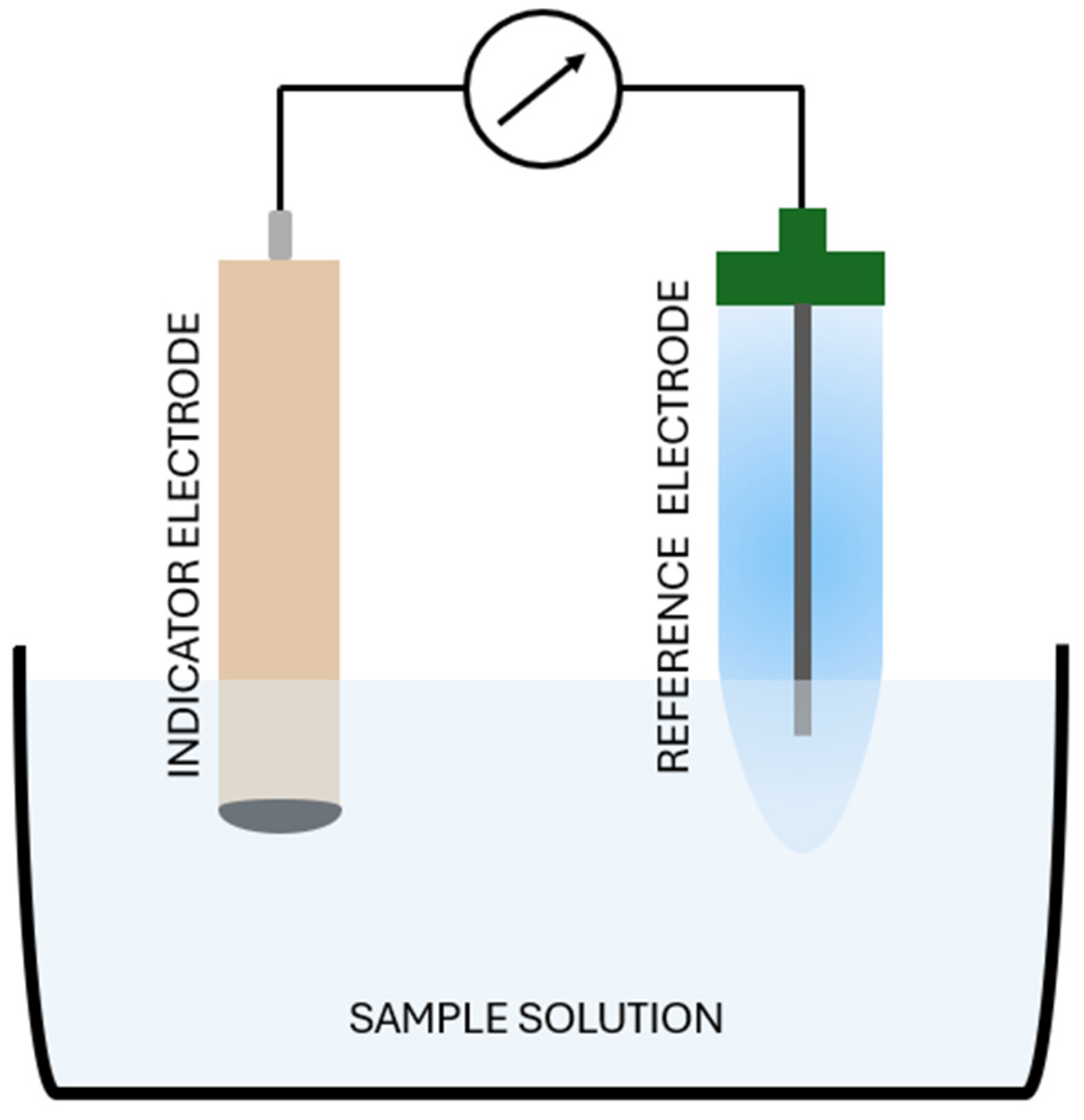
3.1. Potentiometry Basics
3.2. Ion-Selective Electrodes
3.3. Potentiometric Titration
4. Ion-Selective Electrodes for Lead Detection
5. Lead-Selective Electrode Design Solutions
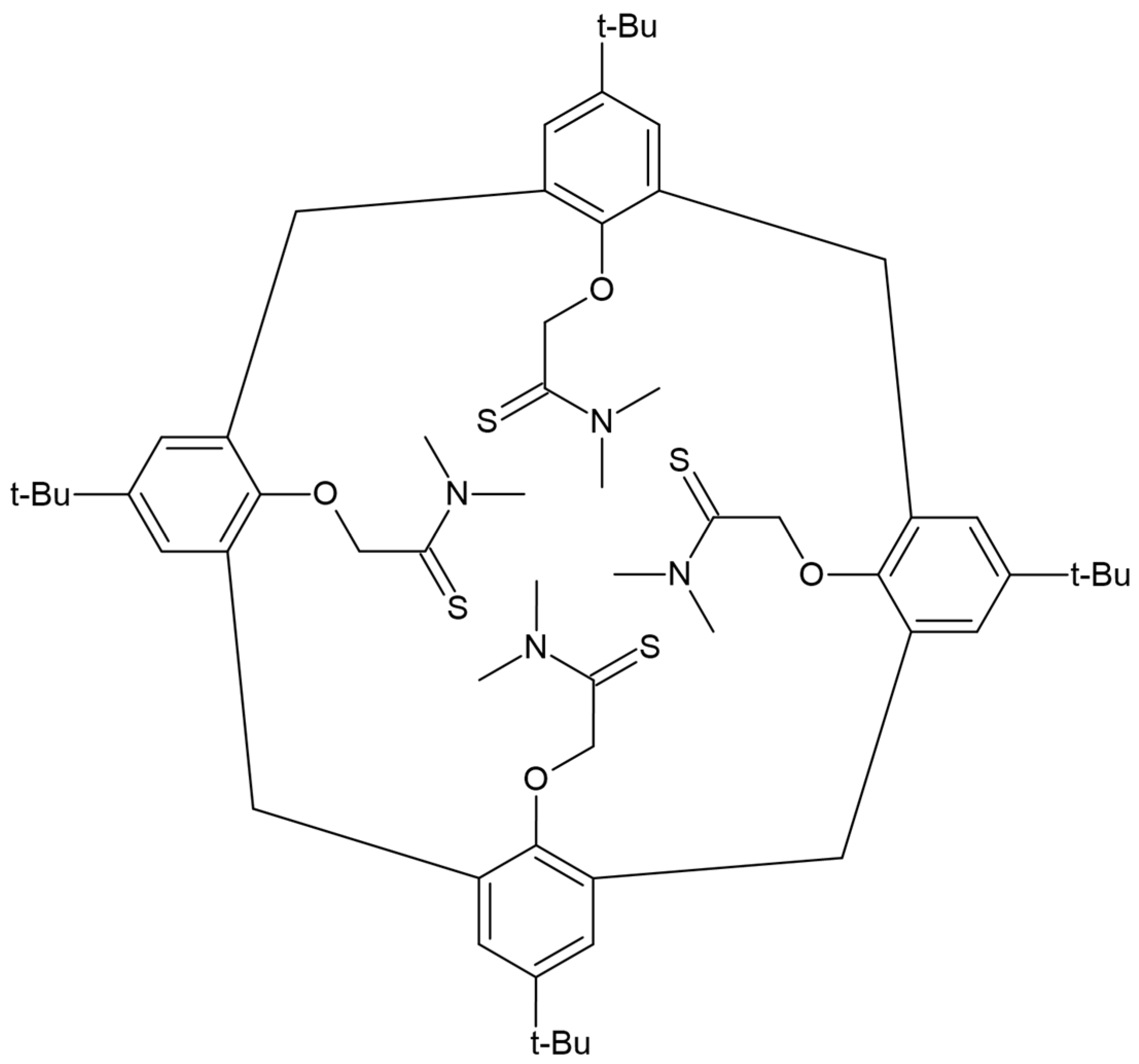
5.1. Electrode with Internal Solution
5.2. Carbon Paste Electrode
5.3. Solid-Contact Electrodes
6. Discussion
7. Current Limitations and Future Development
7.1. Limitations
7.2. Opportunities for Future Development
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AAS | Atomic Absorption Spectrometry |
| ASV | Anodic Stripping Voltammetry |
| BBPA | Bis(1-butylpentyl) adipate |
| BMImCl | 1-butyl-3-methylimidazolium chloride |
| BP C60 | Bimodal Pore C60 |
| CNF | Carbon Nanofibers |
| CPE | Carbon Paste Electrode |
| DMImCl | 1-decyl-3-methylimidazolium chloride |
| DOS | Dioctyl Sebacate |
| EMF | Electromotive Force |
| EMImCl | 1-ethyl-3-methylimidazolium chloride |
| ETH 500 | tetradodecylammonium tetrakis-(4-chlorophenyl)borate |
| GC | Glassy Carbon |
| GFAAS | Graphite Furnace Atomic Absorption Spectrometry |
| HMImCl | 1-hexyl-3-methylimidazolium chloride |
| HPIMN | Novel Schiff Base Ligand Ionophore |
| HMImPF6 | 1-hexyl-3-methylimidazolium hexafluorophosphate |
| ICP MS | Inductively Coupled Plasma Mass Spectrometry |
| ISE | Ion-Selective Electrode |
| KTpClPB | Potassium-tetrakis-(4 chlorophenyl)borate |
| LoD | Limit of Detection |
| MIP | Molecularly Imprinted Polymer |
| NaTFPB | Sodium trakis[3,5-bis(trifluoro-methyl)phenyl]borate |
| NC | Nanocomposite |
| NIOSH | National Institute for Occupational Safety and Health |
| NMImCl | 1-benzy-3-methylimidazolium chloride |
| o NPOE | 2-nitrophenyl Octyl Ether |
| PAMTB | Propyl 2-(acrylamidomethyl)-3,4,5-trihydroxy benzoate |
| PANI | Polyaniline |
| PEDOT | Poly(3,4-ethylenedioxythiophene) |
| PFOA | Perfluorooctanoic Acid |
| PU | Polyurethane |
| PVC | Polyvinyl Chloride |
| PSS | Poly(styrene sulfonate) |
| TDI | Toluene Diisocyanate |
| TBP | Tributyl phosphate |
| THF | Tetrahydrofuran |
| TMTDS | Tetramethyl Thiuram Disulfide |
| TCP | Tricresylphosphate |
| WHO | World Health Organization |
References
- Sanders, T.; Liu, Y.; Buchner, V.; Tchounwou, P.B. Neurotoxic Effects and Biomarkers of Lead Exposure: A Review. Rev. Environ. Health 2009, 24, 15–45. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Wang, P.-Y.; Buchter, G. Ion-Selective Electrodes for Detection of Lead (II) in Drinking Water: A Mini-Review. Environments 2018, 5, 95. [Google Scholar] [CrossRef]
- Wani, A.L.; Ara, A.; Usmani, J.A. Lead toxicity: A review. Interdiscip. Toxicol. 2015, 8, 55–64. [Google Scholar] [CrossRef]
- Boskabady, M.; Marefati, N.; Farkhondeh, T.; Shakeri, F.; Farshbaf, A.; Boskabady, M.H. The effect of environmental lead exposure on human health and the contribution of inflammatory mechanisms, a review. Environ. Int. 2018, 120, 404–420. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Villalva, A.; Marcela, R.-L.; Nelly, L.-V.; Patricia, B.-N.; Guadalupe, M.-R.; Brenda, C.-T.; Eugenia, C.-V.M.; Martha, U.-C.; Isabel, G.-P.; Fortoul, T. Lead systemic toxicity: A persistent problem for health. Toxicology 2025, 515, 154163. [Google Scholar] [CrossRef]
- Collin, M.S.; Venkatraman, S.K.; Vijayakumar, N.; Kanimozhi, V.; Arbaaz, S.M.; Stacey, R.G.S.; Anusha, J.; Choudhary, R.; Lvov, V.; Tovar, G.I.; et al. Bioaccumulation of lead (Pb) and its effects on human: A review. J. Hazard. Mater. Adv. 2022, 7, 100094. [Google Scholar] [CrossRef]
- Zhao, G.; Tran, T.-T.; Modha, S.; Sedki, M.; Myung, N.V.; Jassby, D.; Mulchandani, A. Multiplexed Anodic Stripping Voltammetry Detection of Heavy Metals in Water Using Nanocomposites Modified Screen-Printed Electrodes Integrated With a 3D-Printed Flow Cell. Front. Chem. 2022, 10, 815805. [Google Scholar] [CrossRef]
- Grabarczyk, M.; Wawruch, A. Screen-Printed Carbon Electrode Modified with Carbon Nanotubes and Copper Film as a Simple Tool for Determination of Trace Concentrations of Lead Ions. Membranes 2024, 14, 53. [Google Scholar] [CrossRef]
- Wardak, C. A highly selective lead-sensitive electrode with solid contact based on ionic liquid. J. Hazard. Mater. 2011, 186, 1131–1135. [Google Scholar] [CrossRef]
- Özbek, O.; Gezegen, H.; Cetin, A.; Isildak, Ö. A Potentiometric Sensor for the Determination of Pb(II) Ions in Different Environmental Samples. ChemistrySelect 2022, 7, e202202494. [Google Scholar] [CrossRef]
- Jasiński, A.; Guziński, M.; Lisak, G.; Bobacka, J.; Bocheńska, M. Solid-contact lead(II) ion-selective electrodes for potentiometric determination of lead(II) in presence of high concentrations of Na(I), Cu(II), Cd(II), Zn(II), Ca(II) and Mg(II). Sens. Actuators B Chem. 2015, 218, 25–30. [Google Scholar] [CrossRef]
- Guziński, M.; Lisak, G.; Kupis, J.; Jasiński, A.; Bocheńska, M. Lead(II)-selective ionophores for ion-selective electrodes: A review. Anal. Chim. Acta 2013, 791, 1–12. [Google Scholar] [CrossRef]
- Duffus, J.H. ‘Heavy metals’ a meaningless term? (IUPAC Technical Report). Pure Appl. Chem. 2002, 74, 793–807. [Google Scholar] [CrossRef]
- Agrawal, J.; Sherameti, I.; Varma, A. Detoxification of Heavy Metals: State of Art. In Detoxification of Heavy Metals; Springer: Berlin/Heidelberg, Germany, 2011; pp. 1–34. [Google Scholar] [CrossRef]
- Numan, A.; Gill, A.A.; Rafique, S.; Guduri, M.; Zhan, Y.; Maddiboyina, B.; Li, L.; Singh, S.; Dang, N.N. Rationally engineered nanosensors: A novel strategy for the detection of heavy metal ions in the environment. J. Hazard. Mater. 2021, 409, 124493. [Google Scholar] [CrossRef] [PubMed]
- Flegal, A.R.; Scelfo, G.M. LEAD|Properties and Determination. In Encyclopedia of Food Sciences and Nutrition; Elsevier: Amsterdam, The Netherlands, 2003; pp. 3477–3482. [Google Scholar] [CrossRef]
- Kumar, A.; Kumar, A.; Cabral-Pinto, M.; Chaturvedi, A.K.; Shabnam, A.A.; Subrahmanyam, G.; Mondal, R.; Gupta, D.K.; Malyan, S.K.; Kumar, S.S.; et al. Lead Toxicity: Health Hazards, Influence on Food Chain, and Sustainable Remediation Approaches. Int. J. Environ. Res. Public Health 2020, 17, 2179. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-W.; Choi, H.; Hwang, U.-K.; Kang, J.-C.; Kang, Y.J.; Kim, K.I.; Kim, J.-H. Toxic effects of lead exposure on bioaccumulation, oxidative stress, neurotoxicity, and immune responses in fish: A review. Environ. Toxicol. Pharmacol. 2019, 68, 101–108. [Google Scholar] [CrossRef]
- Shan, B.; Hao, R.; Zhang, J.; Li, J.; Ye, Y.; Lu, A. Microbial remediation mechanisms and applications for lead-contaminated environments. World J. Microbiol. Biotechnol. 2023, 39, 38. [Google Scholar] [CrossRef]
- Sawut, R.; Kasim, N.; Maihemuti, B.; Hu, L.; Abliz, A.; Abdujappar, A.; Kurban, M. Pollution characteristics and health risk assessment of heavy metals in the vegetable bases of northwest China. Sci. Total Environ. 2018, 642, 864–878. [Google Scholar] [CrossRef]
- Showqi, I.; Lone, F.A.; Naikoo, M. Preliminary assessment of heavy metals in water, sediment and macrophyte (Lemna minor) collected from Anchar Lake, Kashmir, India. Appl. Water Sci. 2018, 8, 80. [Google Scholar] [CrossRef]
- Kumar, V. Heavy Metals Accumulation in Soil and Agricultural Crops Grown in the Province of Asahi India Glass Ltd., Haridwar (Uttarakhand), India. Adv. Crop Sci. Technol. 2015, 4, 203. [Google Scholar] [CrossRef]
- Hassan, M. Potential Health Risk of Heavy Metals Accumulation in Vegetables Irrigated with Polluted River. 2018. Available online: https://www.researchgate.net/publication/322936970 (accessed on 15 June 2025).
- Chaoua, S.; Boussaa, S.; El Gharmali, A.; Boumezzough, A. Impact of irrigation with wastewater on accumulation of heavy metals in soil and crops in the region of Marrakech in Morocco. J. Saudi Soc. Agric. Sci. 2019, 18, 429–436. [Google Scholar] [CrossRef]
- Angrand, R.C.; Collins, G.; Landrigan, P.J.; Thomas, V.M. Relation of blood lead levels and lead in gasoline: An updated systematic review. Environ. Health 2022, 21, 138. [Google Scholar] [CrossRef] [PubMed]
- Nigam, A.; Sharma, N.; Tripathy, S.; Kumar, M. Development of semiconductor based heavy metal ion sensors for water analysis: A review. Sens. Actuators A Phys. 2021, 330, 112879. [Google Scholar] [CrossRef]
- An, Y.-J.; Kim, Y.-M.; Kwon, T.-I.; Jeong, S.-W. Combined effect of copper, cadmium, and lead upon Cucumis sativus growth and bioaccumulation. Sci. Total Environ. 2004, 326, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Vangronsveld, J.; Clijsters, H. Toxic Effects of Metals. In Plants and the Chemical Elements; Wiley: Hoboken, NJ, USA, 1994; pp. 149–177. [Google Scholar] [CrossRef]
- Dahmani-Muller, H.; van Oort, F.; Gélie, B.; Balabane, M. Strategies of heavy metal uptake by three plant species growing near a metal smelter. Environ. Pollut. 2000, 109, 231–238. [Google Scholar] [CrossRef]
- Chakraborty, S.; Upare, R.; Das, S. Mitigation of lead-induced soil toxicity and plant growth promotion by phosphate-solubilizing bacterium Enterobacter hormaechei KR2215 isolated from mangrove rhizosphere. J. Environ. Sci. 2025, in press. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, K.; Ming, C.; Tian, J.; Teng, H.; Xu, Z.; He, J.; Liu, F.; Zhou, Y.; Xu, J.; et al. Lead toxicity in Nicotiana tabacum L.: Damage antioxidant system and disturb plant metabolism. Ecotoxicol. Environ. Saf. 2025, 291, 117837. [Google Scholar] [CrossRef]
- Ruley, A.T.; Sharma, N.C.; Sahi, S.V.; Singh, S.R.; Sajwan, K.S. Effects of lead and chelators on growth, photosynthetic activity and Pb uptake in Sesbania drummondii grown in soil. Environ. Pollut. 2006, 144, 11–18. [Google Scholar] [CrossRef]
- Sahi, S.V.; Barlow, R.; Bryant, N.; Andersland, J.; Sahi, S. Lead Hyperaccumulation by Sesbania drummondii. 2000. Available online: https://www.researchgate.net/publication/265154356 (accessed on 15 June 2025).
- MacFarlane, G.R.; Burchett, M.D. Toxicity, growth and accumulation relationships of copper, lead and zinc in the grey mangrove Avicennia marina (Forsk.) Vierh. Mar. Environ. Res. 2002, 54, 65–84. [Google Scholar] [CrossRef]
- Gumpu, M.B.; Sethuraman, S.; Krishnan, U.M.; Rayappan, J.B.B. A review on detection of heavy metal ions in water—An electrochemical approach. Sens. Actuators B Chem. 2015, 213, 515–533. [Google Scholar] [CrossRef]
- Liu, C.; Bai, R.; Ly, Q.S. Selective removal of copper and lead ions by diethylenetriamine-functionalized adsorbent: Behaviors and mechanisms. Water Res. 2008, 42, 1511–1522. [Google Scholar] [CrossRef]
- Gwaltney-Brant, S.M. Heavy Metals. In Haschek and Rousseaux’s Handbook of Toxicologic Pathology; Elsevier: Amsterdam, The Netherlands, 2013; pp. 1315–1347. [Google Scholar] [CrossRef]
- Mason, L.H.; Harp, J.P.; Han, D.Y. Pb Neurotoxicity: Neuropsychological Effects of Lead Toxicity. Biomed. Res. Int. 2014, 2014, 840547. [Google Scholar] [CrossRef]
- Pretsch, E. The new wave of ion-selective electrodes. TrAC Trends Anal. Chem. 2007, 26, 46–51. [Google Scholar] [CrossRef]
- Bakker, E.; Pretsch, E. Modern Potentiometry. Angew. Chem. Int. Ed. 2007, 46, 5660–5668. [Google Scholar] [CrossRef] [PubMed]
- Zdrachek, E.; Bakker, E. Potentiometric Sensing. Anal. Chem. 2019, 91, 2–26. [Google Scholar] [CrossRef] [PubMed]
- Buck, R.P.; Lindner, E. Peer Reviewed: Tracing the History of Selective Ion Sensors. Anal. Chem. 2001, 73, 88A–97A. [Google Scholar] [CrossRef]
- Bakker, E. Encyclopedia of analytical Science. In Encyclopedia of Analytical Science, 3rd ed.; Elsevier: Oxford, UK, 2019; pp. 231–251. [Google Scholar]
- Huang, M.-R.; Rao, X.-W.; Li, X.-G.; Ding, Y.-B. Lead ion-selective electrodes based on polyphenylenediamine as unique solid ionophores. Talanta 2011, 85, 1575–1584. [Google Scholar] [CrossRef]
- Shamsipur, M.; Ganjali, M.R.; Rouhollahi, A. Lead-Selective Membrane Potentiometric Sensor Based on an 18-Membered Thiacrown Derivative. Anal. Sci. 2001, 17, 935–938. [Google Scholar] [CrossRef]
- Clarke, B.L.; Wooten, L.A.; Compton, K.G. Potentiometric Titration in Non-Aqueous Solutions. Ind. Eng. Chem. Anal. Ed. 1931, 3, 321–323. [Google Scholar] [CrossRef]
- Mohan, C.; Sharma, K.; Chandra, S. Lead (II)-Selective Potentiometric Sensor Based on Thiophene-2-Aldehyde Thiosemicarbazone (TATS) Schiff Base in PVC Matrix. Anal. Bioanal. Electrochem. 2022, 14, 860–870. [Google Scholar]
- Leaflet: Manual for Ion-Selective Electrodes. Available online: https://www.metrohm.com/pl_pl/products/8/1098/81098042.html (accessed on 20 August 2025).
- Lindner, E.; Toth, K.; Pungor, E. Lead-selective neutral carrier based liquid membrane electrode. Anal. Chem. 1984, 56, 1127–1131. [Google Scholar] [CrossRef]
- Didina, S.E.; Mitnik, L.L.; Koshmina, N.V.; Grekovich, A.L.; Mikhelson, K.N. Lead-selective electrodes based on liquid ion-exchangers. Sens. Actuators B Chem. 1994, 19, 396–399. [Google Scholar] [CrossRef]
- Kivalo, P.; Virtanen, R.; Wickström, K.; Wilson, M.; Pungor, E.; Horvai, G.; Tóth, K. An Evaluation Of Some Commercial Lead(II)-Selective Electrodes. Anal. Chim. Acta 1976, 87, 401–409. [Google Scholar] [CrossRef]
- Ion-Selective Electrode, Pb. Available online: https://www.metrohm.com/pl_pl/products/6/0502/60502170.html (accessed on 23 July 2025).
- Thermo Scientific™ Orion™ Ion Selective Electrodes. Available online: https://assets.thermofisher.com/TFS-Assets/LPD/brochures/Orion-Ion-Selective-Electrodes-Brochure.pdf (accessed on 23 July 2025).
- RoyChoudhury, P.; Majumdar, S.; Sarkar, S.; Kundu, B.; Sahoo, G.C. Performance investigation of Pb(II) removal by synthesized hydroxyapatite based ceramic ultrafiltration membrane: Bench scale study. Chem. Eng. J. 2019, 355, 510–519. [Google Scholar] [CrossRef]
- Cadogan, A.; Gao, Z.; Lewenstam, A.; Ivaska, A.; Diamond, D. All-solid-state sodium-selective electrode based on a calixarene ionophore in a poly(vinyl chloride) membrane with a polypyrrole solid contact. Anal. Chem. 1992, 64, 2496–2501. [Google Scholar] [CrossRef]
- Lindner, E.; Pendley, B.D. A tutorial on the application of ion-selective electrode potentiometry: An analytical method with unique qualities, unexplored opportunities and potential pitfalls; Tutorial. Anal. Chim. Acta 2013, 762, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Bakker, E.; Bühlmann, P.; Pretsch, E. Carrier-Based Ion-Selective Electrodes and Bulk Optodes. 1. General Characteristics. Chem. Rev. 1997, 97, 3083–3132. [Google Scholar] [CrossRef]
- Ceresa, A.; Bakker, E.; Hattendorf, B.; Günther, D.; Pretsch, E. Potentiometric Polymeric Membrane Electrodes for Measurement of Environmental Samples at Trace Levels: New Requirements for Selectivities and Measuring Protocols, and Comparison with ICPMS. Anal. Chem. 2001, 73, 343–351. [Google Scholar] [CrossRef]
- Yu, S.; Li, F.; Yin, T.; Liu, Y.; Pan, D.; Qin, W. A solid-contact Pb2+-selective electrode using poly(2-methoxy-5-(2′-ethylhexyloxy)-p-phenylene vinylene) as ion-to-electron transducer. Anal. Chim. Acta 2011, 702, 195–198. [Google Scholar] [CrossRef]
- Malinowska, E. Lead-selective membrane electrodes based on neutral carriers. Part I. Acyclic Amides oxamides. Analyst 1990, 115, 1085–1087. [Google Scholar] [CrossRef]
- Jeong, T.; Lee, H.; Jeong, D.; Jeon, S. A lead(II)-selective PVC membrane based on a Schiff base complex of N,N′-bis(salicylidene)-2,6-pyridinediamine. Talanta 2005, 65, 543–548. [Google Scholar] [CrossRef]
- Hasse, W.; Ahlers, B.; Reinbold, J.; Cammann, K. PbOH+ -selective membrane electrode based on crown ethers. Sens. Actuators B Chem. 1994, 19, 383–386. [Google Scholar] [CrossRef]
- Bakker, E. Selectivity of carrier-based ion-selective electrodes: Is the problem solved? TrAC Trends Anal. Chem. 1997, 16, 252–260. [Google Scholar] [CrossRef]
- Nisah, K.; Rahmi, R.; Ramli, M.; Idroes, R.; Alva, S.; Iqhrammullah, M.; Safitri, E. Optimization of Castor Oil-Based Ion Selective Electrode (ISE) with Active Agent 1,10-Phenanthroline for Aqueous Pb2+ Analysis. Membranes 2022, 12, 987. [Google Scholar] [CrossRef] [PubMed]
- Adams, R.N. Carbon Paste Electrodes. Anal. Chem. 1958, 30, 1576. [Google Scholar] [CrossRef]
- Vytřas, K.; Svancara, I.; Metelka, R. Carbon paste electrodes in electroanalytical chemistry. J. Serbian Chem. Soc. 2009, 74, 1021–1033. [Google Scholar] [CrossRef]
- Mishra, D.; Krause, A.; Sahni, H.S.; Chatterjee, S. Multi-walled carbon nanotube-functional ionophore based composite potentiometric sensor for selective detection of lead in water. Diam. Relat. Mater. 2023, 137, 110156. [Google Scholar] [CrossRef]
- Hussein, K.M.; Omar, M.M.; Mohamed, G.G.; EL-Sanafery, S.S. Lead determination by potentiometric method using a new modified carbon paste electrode in real food samples. Microchem. J. 2025, 208, 112533. [Google Scholar] [CrossRef]
- Gao, L.; Tian, Y.; Gao, W.; Xu, G. Recent Developments and Challenges in Solid-Contact Ion-Selective Electrodes. Sensors 2024, 24, 4289. [Google Scholar] [CrossRef]
- Cheong, Y.H.; Ge, L.; Lisak, G. Highly reproducible solid contact ion selective electrodes: Emerging opportunities for potentiometry—A review. Anal. Chim. Acta 2021, 1162, 338304. [Google Scholar] [CrossRef]
- Wardak, C.; Pietrzak, K.; Morawska, K.; Grabarczyk, M. Ion-Selective Electrodes with Solid Contact Based on Composite Materials: A Review. Sensors 2023, 23, 5839. [Google Scholar] [CrossRef] [PubMed]
- Wardak, C. Improved Lead Sensing Using a Solid-Contact Ion-Selective Electrode with Polymeric Membrane Modified with Carbon Nanofibers and Ionic Liquid Nanocomposite. Materials 2023, 16, 1003. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yin, T.; Qin, W. An all-solid-state polymeric membrane Pb2+-selective electrode with bimodal pore C60 as solid contact. Anal. Chim. Acta 2015, 876, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, X.; Zeng, X.; Waterhouse, G.I.; Jiang, X.; Zhang, Z.; Yu, L. Antifouling improvement in Pb2+ ion selective electrodes by using an environmentally friendly capsaicin derivative. Talanta 2023, 258, 124436. [Google Scholar] [CrossRef]
- Wang, X.; Yu, L.; Li, F.; Zhang, G.; Zhou, W.; Jiang, X. Synthesis of amide derivatives containing capsaicin and their antioxidant and antibacterial activities. J. Food Biochem. 2019, 43, e13061. [Google Scholar] [CrossRef]
- Wang, X.; Jiang, X.; Yu, L. Performance assessment of capsaicin derivatives containing amide groups used as active substances for antifouling coatings. Prog. Org. Coat. 2021, 160, 106515. [Google Scholar] [CrossRef]
- Upadhyay, K.; Viramgami, A.; Balachandar, R.; Pagdhune, A.; Shaikh, I.; Sivaperumal, P. Development and validation of Graphite Furnace Atomic Absorption Spectrometry method and its application for clinical evaluation of blood lead levels among occupationally exposed lead smelting plant workers. Anal. Sci. 2023, 39, 517–526. [Google Scholar] [CrossRef]
- Thermo Scientific iCAP TQ ICP-MS: Typical Limits of Detection. Available online: https://assets.thermofisher.com/TFS-Assets/CMD/Application-Notes/TN-43287-ICP-MS-iCAP-TQ-Typical-Limits-Detection-TN43287-EN.pdf (accessed on 20 August 2025).
- Lead in Drinking Water. Available online: https://www.metrohm.com/pl_pl/applications/application-notes/aa-v-101-200/an-v-214.html (accessed on 20 August 2025).
- Lead by Portable Ultrasonic Extraction/ASV. Available online: https://www.cdc.gov/niosh/docs/2014-151/pdfs/methods/7701.pdf (accessed on 20 August 2025).
- Ruiz-Gonzalez, A. Ion-Selective Electrodes in the Food Industry: Development Trends in the Potentiometric Determination of Ionic Pollutants. Electrochem 2024, 5, 178–212. [Google Scholar] [CrossRef]
- Golcs, Á.; Horváth, V.; Huszthy, P.; Tóth, T. Fast Potentiometric Analysis of Lead in Aqueous Medium under Competitive Conditions Using an Acridono-Crown Ether Neutral Ionophore. Sensors 2018, 18, 1407. [Google Scholar] [CrossRef]
- Qin, W.; Liang, R.; Fu, X.; Wang, Q.; Yin, T.; Song, W. Trace-Level Potentiometric Detection in the Presence of a High Electrolyte Background. Anal. Chem. 2012, 84, 10509–10513. [Google Scholar] [CrossRef]
- Sisodia, N.; McGuinness, K.L.; Wadhawan, J.D.; Lawrence, N.S. In situ recalibration of ion selective electrodes. Sens. Diagn. 2022, 1, 134–138. [Google Scholar] [CrossRef]
- Liu, Y.; Gao, Y.; Yan, R.; Huang, H.; Wang, P. Disposable Multi-Walled Carbon Nanotubes-Based Plasticizer-Free Solid-Contact Pb2+-Selective Electrodes with a Sub-PPB Detection Limit. Sensors 2019, 19, 2550. [Google Scholar] [CrossRef] [PubMed]
- Fathy, M.A.; Bühlmann, P. Next-Generation Potentiometric Sensors: A Review of Flexible and Wearable Technologies. Biosensors 2025, 15, 51. [Google Scholar] [CrossRef]
- Kamel, A.H.; El-Naggar, A.M.; Argig, A.A.A. Response characteristics of lead-selective membrane sensors based on a newly synthesized quinoxaline derivatives as neutral carrier ionophores. Ionics 2017, 23, 3497–3506. [Google Scholar] [CrossRef]
- Lu, Z.; Yang, S.; Yang, Q.; Luo, S.; Liu, C.; Tang, Y. A glassy carbon electrode modified with graphene, gold nanoparticles and chitosan for ultrasensitive determination of lead(II). Microchim. Acta 2013, 180, 555–562. [Google Scholar] [CrossRef]
- Zhang, L.; Wei, Z.; Liu, P.; Wei, H.; Ma, D. Performance Comparison of Solid Lead Ion Electrodes with Different Carbon-Based Nanomaterials as Electron-Ion Exchangers. Sensors 2021, 21, 1663. [Google Scholar] [CrossRef]
- Khan, A.A.P.; Khan, A.; Rahman, M.M.; Asiri, A.M.; Oves, M. Lead sensors development and antimicrobial activities based on graphene oxide/carbon nanotube/poly(O-toluidine) nanocomposite. Int. J. Biol. Macromol. 2016, 89, 198–205. [Google Scholar] [CrossRef]
- Stelmach, E.; Maksymiuk, K.; Michalska, A. Dual Sensitivity─Potentiometric and Fluorimetric─Ion-Selective Membranes. Anal. Chem. 2021, 93, 14737–14742. [Google Scholar] [CrossRef]
- Kudr, J.; Nguyen, H.V.; Gumulec, J.; Nejdl, L.; Blazkova, I.; Ruttkay-Nedecky, B.; Hynek, D.; Kynicky, J.; Adam, V.; Kizek, R. Simultaneous Automatic Electrochemical Detection of Zinc, Cadmium, Copper and Lead Ions in Environmental Samples Using a Thin-Film Mercury Electrode and an Artificial Neural Network. Sensors 2014, 15, 592–610. [Google Scholar] [CrossRef]
- Ma, X.; Armas, S.M.; Soliman, M.; Lytle, D.A.; Chumbimuni-Torres, K.Y.; Tetard, L.; Lee, W.H. In Situ Monitoring of Pb2+ Leaching from the Galvanic Joint Surface in a Prepared Chlorinated Drinking Water. Environ. Sci. Technol. 2018, 52, 2126–2133. [Google Scholar] [CrossRef]
- Fan, Y.; Xu, Z.; Huang, Y.; Wang, T.; Zheng, S.; DePasquale, A.; Brüeckner, C.; Lei, Y.; Li, B. Long-term continuous and real-time in situ monitoring of Pb(II) toxic contaminants in wastewater using solid-state ion selective membrane (S-ISM) Pb and pH auto-correction assembly. J. Hazard. Mater. 2020, 400, 123299. [Google Scholar] [CrossRef]
- Darestani-Farahani, M.; Montealegre, I.M.; Gilavan, M.T.; Kirby, T.; Selvaganapathy, P.R.; Kruse, P. A highly sensitive ion-selective chemiresistive sensor for online monitoring of lead ions in water. Analyst 2024, 149, 2915–2924. [Google Scholar] [CrossRef]

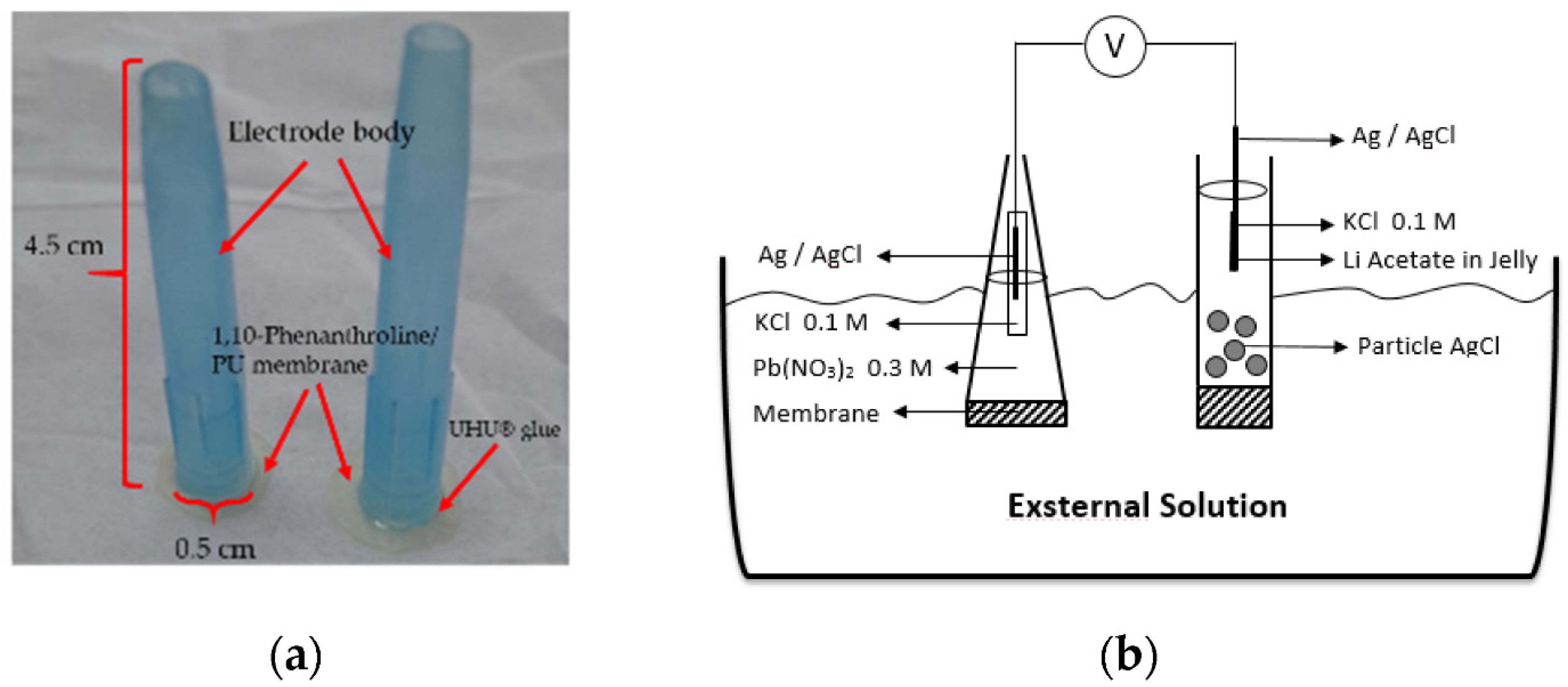
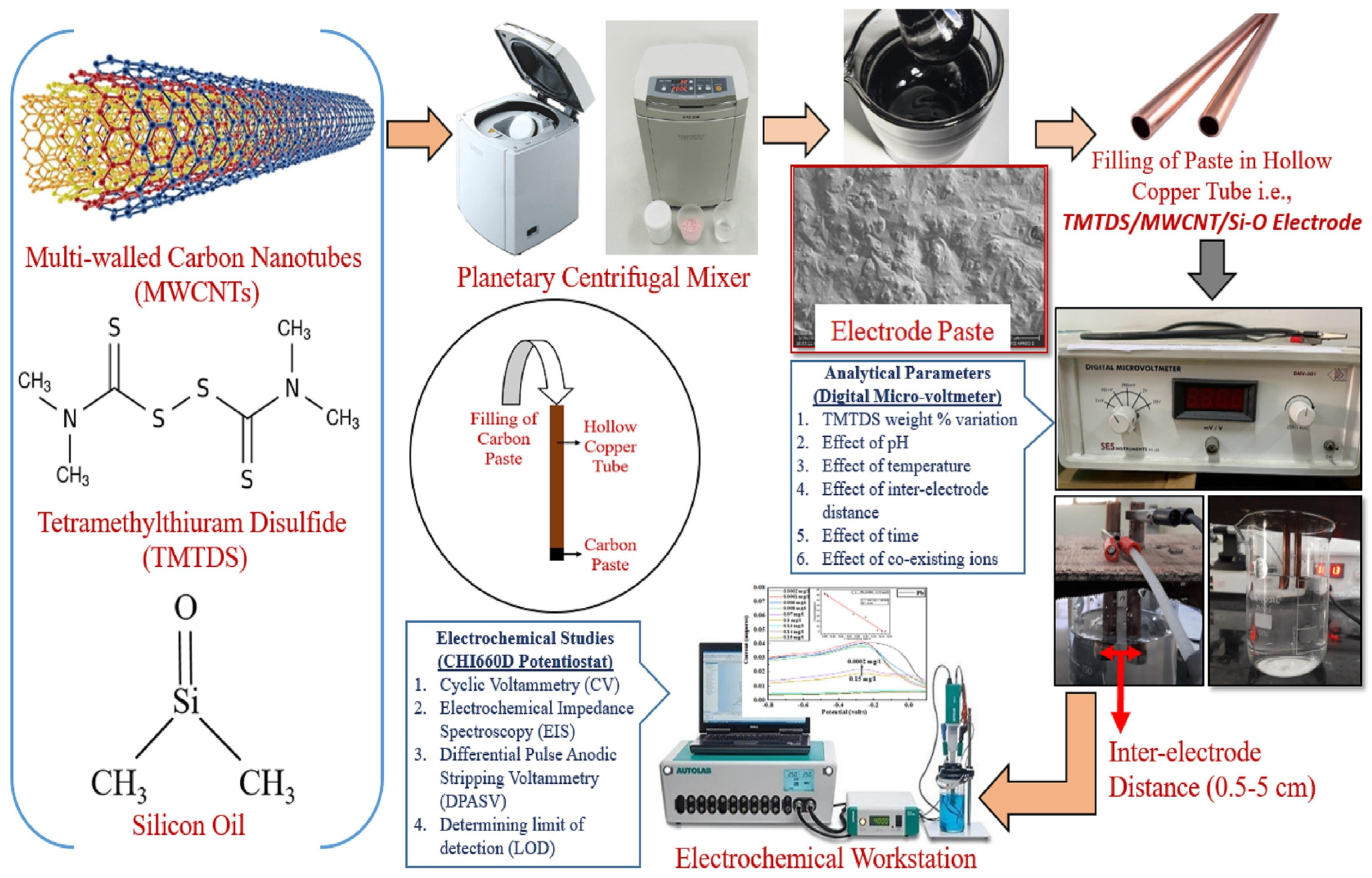
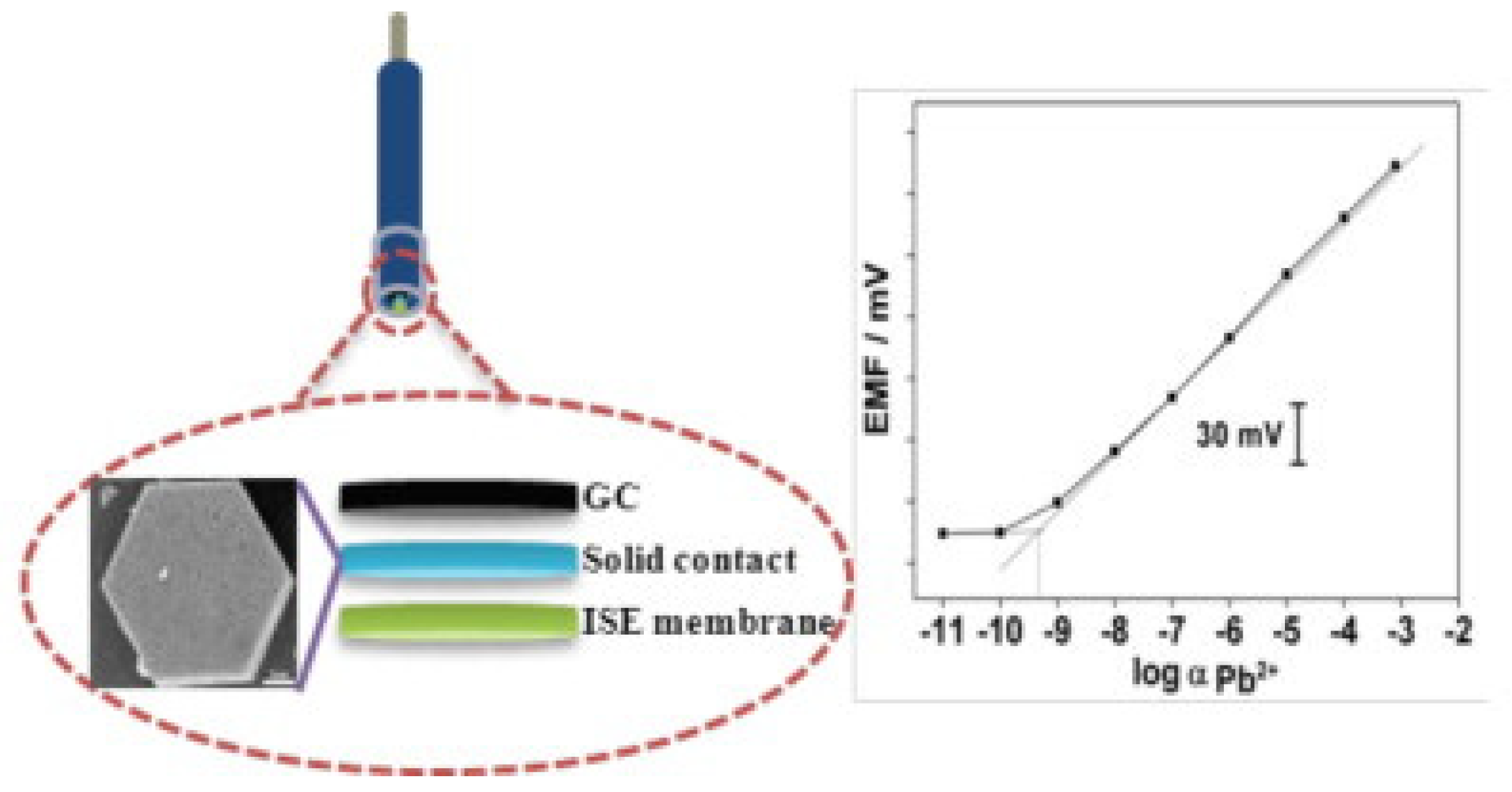
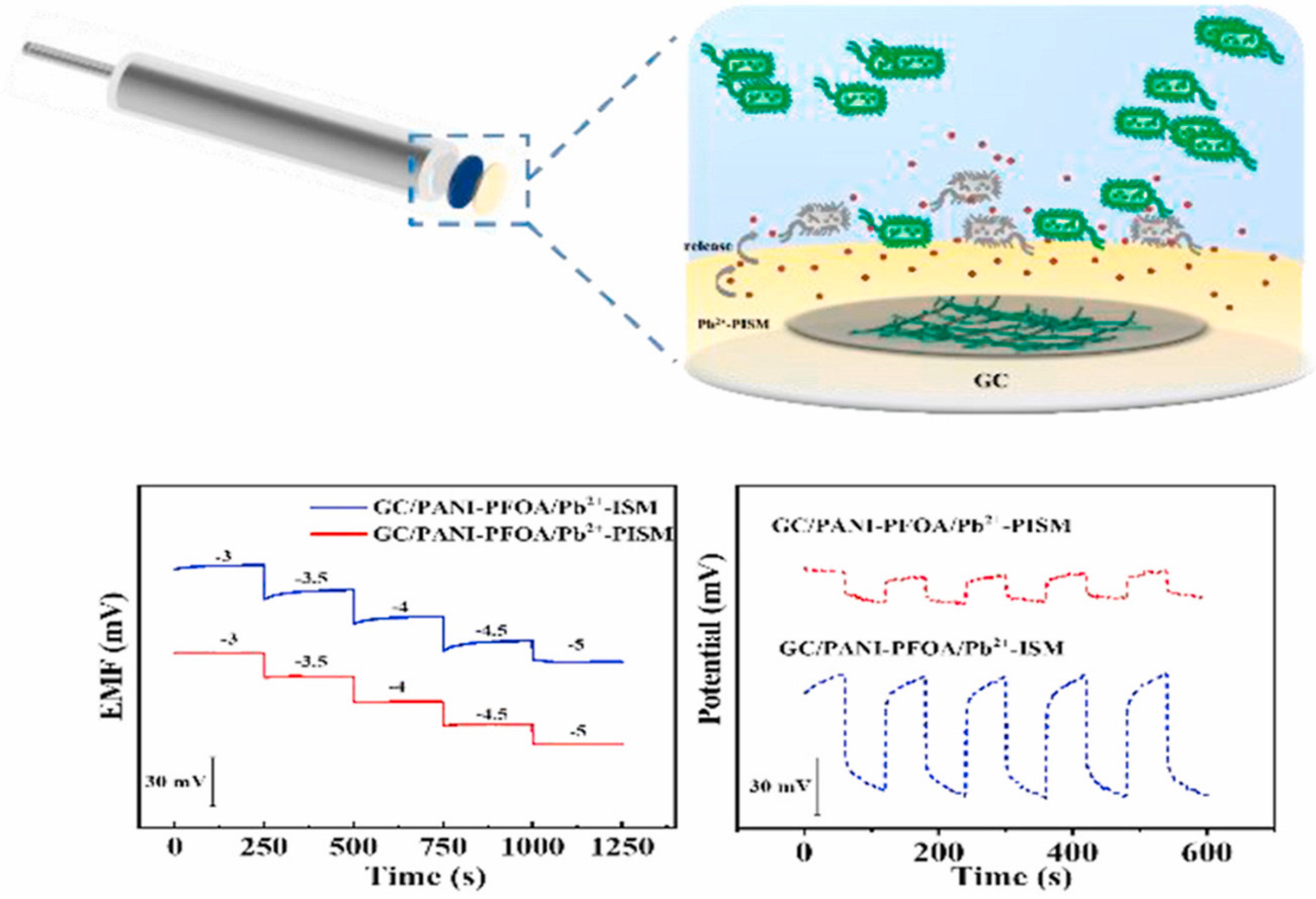
| Electrode Design | Limit of Detection [M] | Linear Range [M] | Slope [mV/dec] | Working pH Range | Sample Type for Lead Detection | Recovery Rates [%] | Reference |
|---|---|---|---|---|---|---|---|
| castor oil-based electrode with internal solution | 10−10 | 1.0 × 10−10–1.0 × 10−5 | 27.25 | 7–8 | wastewater | N/R | [64] |
| composite carbon paste electrode | 1.5 × 10−7 | 9.7 × 10−10–7.2 × 10−7 | N/R | 1–5 | water | N/R | [67] |
| graphite paste electrode with TCP as plasticizer | 3.33 × 10−5 | 1.0 × 10−5–1.0 × 10−2 | 29.50 ± 0.40 | 3–8 | food samples | 97.6–101.2 | [68] |
| graphite paste electrode with o-NPOE as plasticizer | 3.33 × 10−6 | 1.0 × 10−6–1.0 × 10−2 | 29.90 ± 0.56 | 3–9 | food samples | 97.3–101.8 | |
| solid-contact electrode modified with ionic liquids | 4.3 × 10−9 | 1.0 × 10−8–1.0 × 10−1 | 29.8 | 3.5–7.3 | tap, river and wastewater | 92.7–104.8 | [9] |
| solid-contact electrode modified with NC | 6.0 × 10−9 | 1.0 × 10−8–1.0 × 10−2 | 31.5 | 3.1–7.6 | wastewater | N/R | [72] |
| BP-C60-based solid-contact electrode | 5.0 × 10−10 | 1.0 × 10−9–1.0 × 10−3 | 28.8 ± 1.2 | N/R | tap water | N/R | [73] |
| PANI–PFOA solid-contact electrode with PAMTB additive | 1.9 × 10−7 | 1.0 × 10–6–1.0 × 10−3 | 28.5 ± 0.8 | N/R | seawater | 89.5–110.9 | [74] |
| Method | Average Detection Limit | Use Case/Professional Application | Reference |
|---|---|---|---|
| Graphite Furnace AAS (GFAAS) | 1.6 µg/L in blood | Clinical diagnostics (biomonitoring of Pb exposure) | [77] |
| ICP-MS (iCAP TQ) | 1 ng/L | High-sensitivity environmental and quality control analysis | [78] |
| Portable ASV (scTRACE Gold) | 0.2 µg/L | Field monitoring of drinking water | [79] |
| Ultrasonic Extraction + ASV (NIOSH 7701) | 0.05 µg per air sample | Occupational/environmental health monitoring (air filters) | [80] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Drużyńska, M.; Lenar, N.; Paczosa-Bator, B. Tracking Lead: Potentiometric Tools and Technologies for a Toxic Element. Molecules 2025, 30, 3492. https://doi.org/10.3390/molecules30173492
Drużyńska M, Lenar N, Paczosa-Bator B. Tracking Lead: Potentiometric Tools and Technologies for a Toxic Element. Molecules. 2025; 30(17):3492. https://doi.org/10.3390/molecules30173492
Chicago/Turabian StyleDrużyńska, Martyna, Nikola Lenar, and Beata Paczosa-Bator. 2025. "Tracking Lead: Potentiometric Tools and Technologies for a Toxic Element" Molecules 30, no. 17: 3492. https://doi.org/10.3390/molecules30173492
APA StyleDrużyńska, M., Lenar, N., & Paczosa-Bator, B. (2025). Tracking Lead: Potentiometric Tools and Technologies for a Toxic Element. Molecules, 30(17), 3492. https://doi.org/10.3390/molecules30173492







