Small Structural Changes in Chili-Derived Capsaicin Resulting in Nonivamide Analogs of Significantly Improved Cytotoxicity and Good Tumor/Non-Tumor Cell Selectivity
Abstract
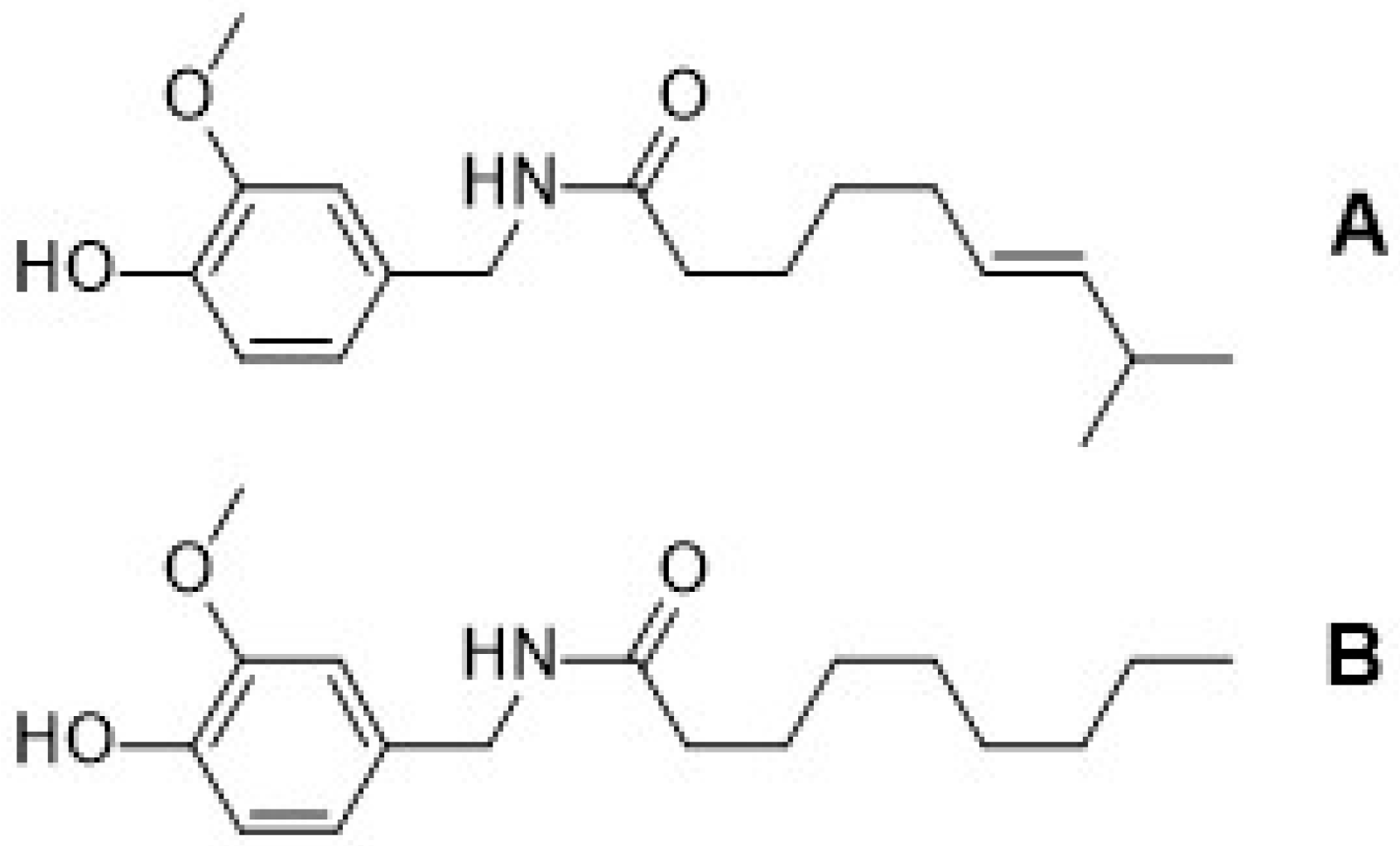
1. Introduction
2. Results
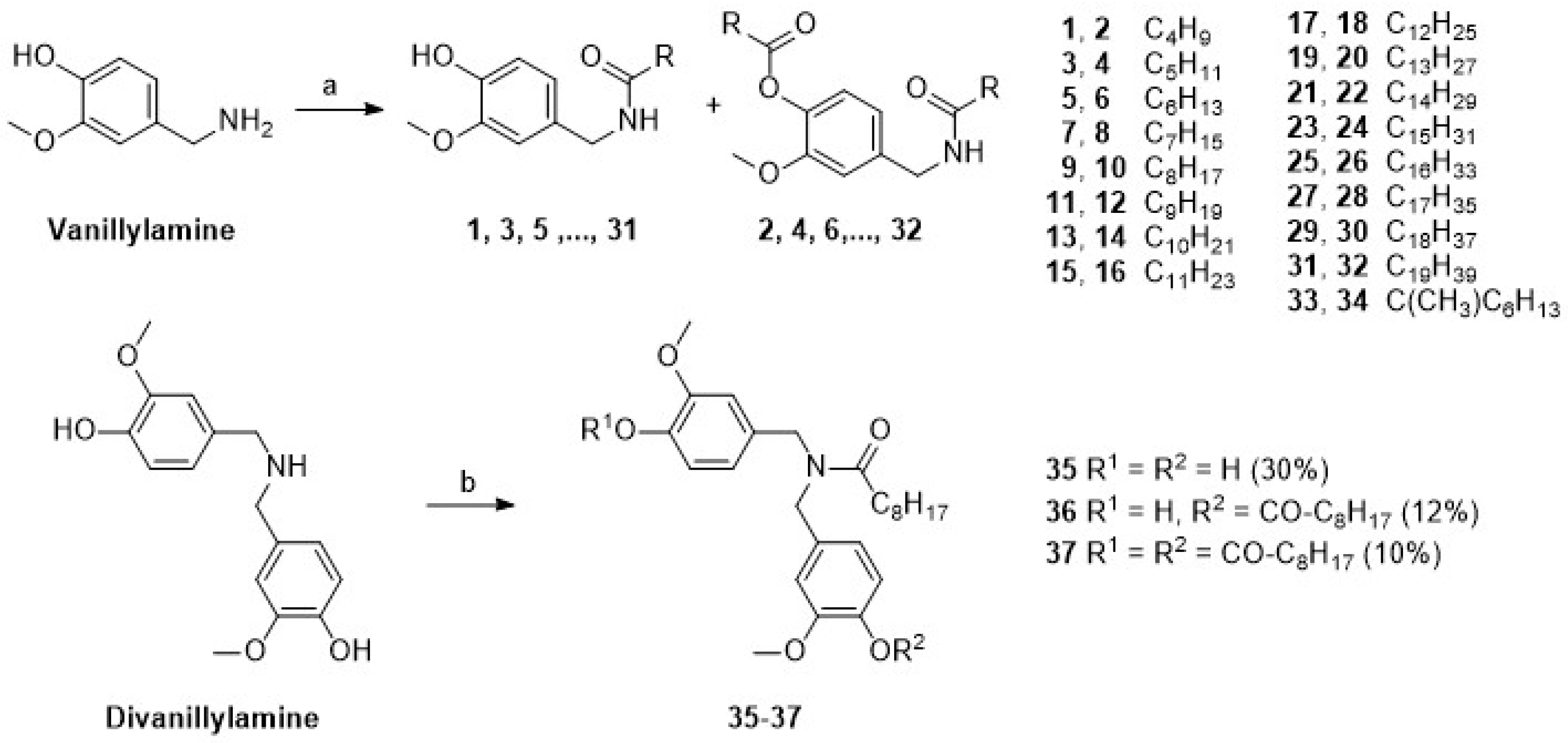
2.1. Synthesis
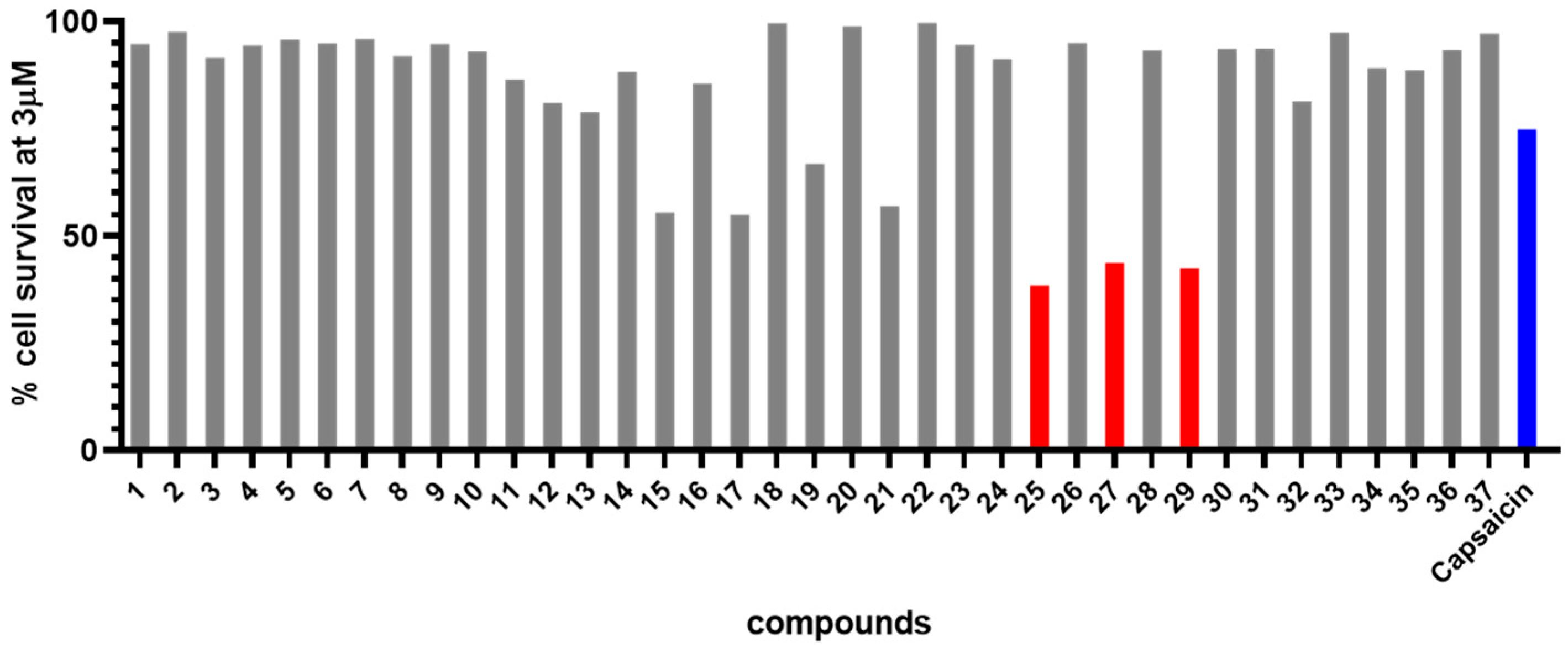
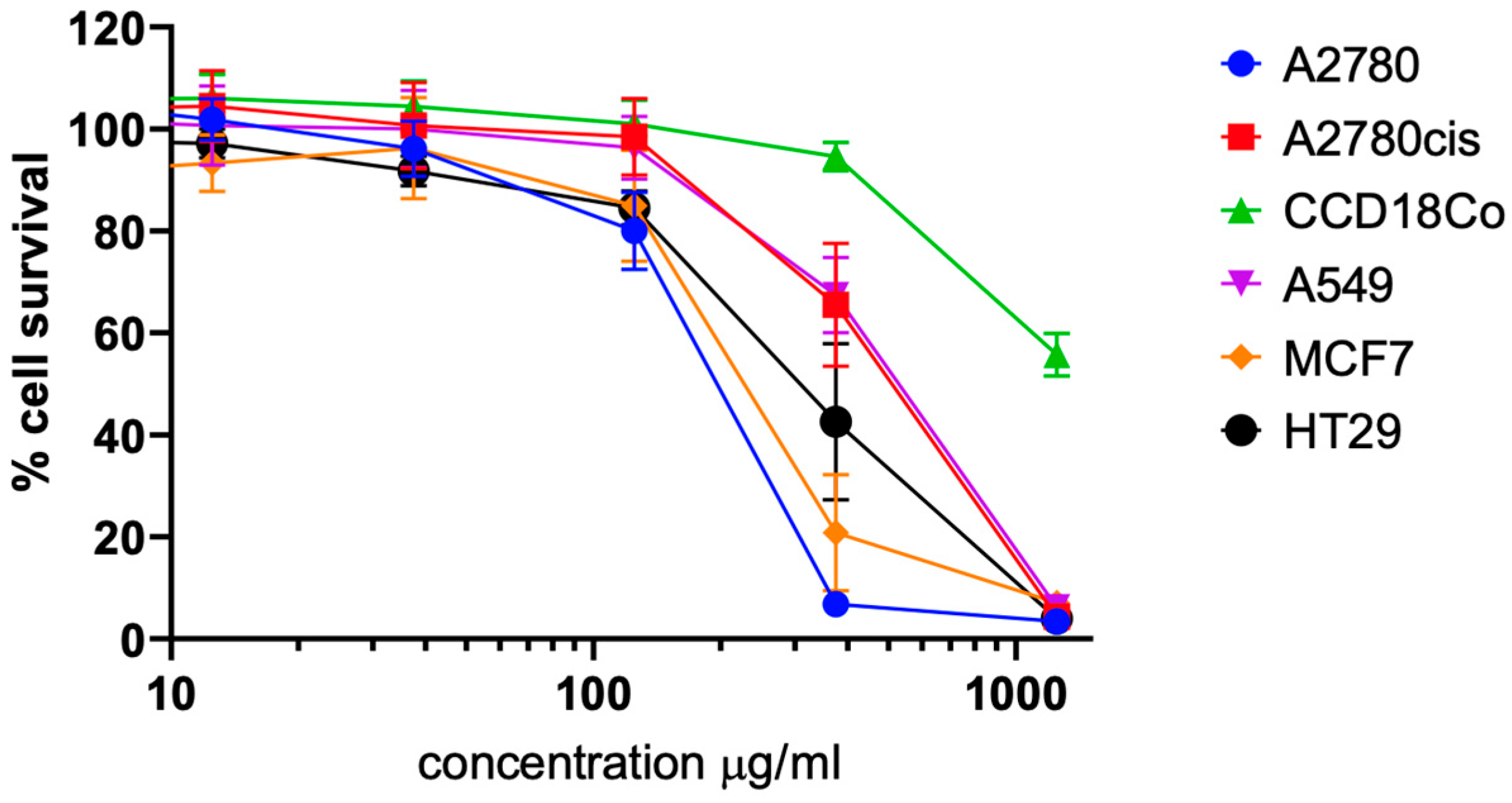
2.2. Biological Evaluation
3. Discussion
4. Materials and Methods
4.1. General
4.2. Synthesis of the Amides 1–37
4.2.1. N-[(4-Hydroxy-3-methoxy)benzyl]pentanamide (1) and 2-methoxy-4-[(pentanoylamino)methyl]phenyl pentanoate (2)
4.2.2. N-[(4-Hydroxy-3-methoxy)benzyl]hexanamide (3) and 2-methoxy-4-[(hexanoylamino)methyl]phenyl hexanoate (4)
4.2.3. N-[(4-Hydroxy-3-methoxy)benzyl]heptanamide (5) and 2-methoxy-4-[(heptanoylamino)methyl]phenyl heptanoate (6)
4.2.4. N-[(4-Hydroxy-3-methoxy)benzyl]octanamide (7) and 2-methoxy-4-[(octanoylamino)methyl]phenyl octanoate (8)
4.2.5. N-[(4-Hydroxy-3-methoxy)benzyl]nonanamide (9) and 2-methoxy-4-[(nonanoylamino)methyl]phenyl nonanoate (10)
4.2.6. N-[(4-Hydroxy-3-methoxy)benzyl]decanamide (11) and 2-methoxy-4-[(decanoylamino)methyl]phenyl decanoate (12)
4.2.7. N-[(4-Hydroxy-3-methoxy)benzyl]undecanamide (13) and 22-methoxy-4-[(undecanoylamino)methyl]phenyl undecanoate (14)
4.2.8. N-[(4-Hydroxy-3-methoxy)benzyl]dodecanamide (15) and 2-methoxy-4-[(dodecanoylamino)methyl]phenyl dodecanoate (16)
4.2.9. N-[(4-Hydroxy-3-methoxy)benzyl]tridecanamide (17) and 2-methoxy-4-[(tridecanoylamino)methyl]phenyl tridecanoate (18)
4.2.10. N-[(4-Hydroxy-3-methoxy)benzyl]tetradecanamide (19) and 2-methoxy-4-[(tetradecanoylamino)methyl]phenyl tetradecanoate (20)
4.2.11. N-[(4-Hydroxy-3-methoxy)benzyl]pentadecanamide (21) and 2-methoxy-4-[(pentadecanoylamino)methyl]phenyl pentadecanoate (22)
4.2.12. N-[(4-Hydroxy-3-methoxy)benzyl]hexadecanamide (23) and 2-methoxy-4-[(hexadecanoylamino)methyl]phenyl hexadecanoate (24)
4.2.13. N-[(4-Hydroxy-3-methoxy)benzyl]heptadecanamide (25) and 2-methoxy-4-[(heptadecanoylamino)methyl]phenyl heptadecanoate (26)
4.2.14. N-[(4-Hydroxy-3-methoxy)benzyl]octadecanamide (27) and 2-methoxy-4-[(octadecanoylamino)methyl]phenyl octadecanoate (28)
4.2.15. N-[(4-Hydroxy-3-methoxy)benzyl]nonadecanamide (29) and 2-methoxy-4-[(nonadecanoylamino)methyl]phenyl nonadecanoate (30)
4.2.16. N-[(4-Hydroxy-3-methoxy)benzyl]icosanamide (31) and 2-methoxy-4-[(icosanoylamino)methyl]phenyl icosanoate (32)
4.2.17. N,N-bis(4-hydroxy-3-methoxybenzyl)nonanamide (33), 4-{[4-hydroxy-3-methoxybenzyl)(nonanoyl)amino]methyl}-2-methoxyphenyl nonanoate (34) and (nonanoylimino) bis(methylene-2-methoxy-4,1-phenylene) dinonanoate (35)
4.2.18. N-[(4-Hydroxy-3-methoxyphenyl)methyl]-2-methyloctanamide (36) and 4-{[(2-methyloctanoyl)amino]methyl}-2-(methyloxy)phenyl 2-methyloctanoate (37)
4.3. Capsaicin
4.4. Preparation of the Chili Extract
4.5. SRB Assay
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Perry, L.; Dickau, R.; Zarrillo, S.; Holst, I.; Pearsall, D.M.; Piperno, D.R.; Berman, M.J.; Cooke, R.G.; Rademaker, K.; Ranere, A.J.; et al. Starch Fossils and the Domestication and Dispersal of Chili Peppers (Capsicum spp. L.) in the Americas. Science 2007, 315, 986–988. [Google Scholar] [CrossRef]
- Govindarajan, V.S. Capsicum-production, technology, chemistry, and quality. Part 1: History, botany, cultivation, and primary processing. CRC Crit. Rev. Food Sci. Nutr. 1985, 22, 109–176. [Google Scholar] [CrossRef]
- Buchholz, C.F. Chemische Untersuchungen der trockenen, reifen spanischen Pfeffers. Alm. Oder Taschenb. Für Scheidekünste Apoth. 1816, 37, 1–30. [Google Scholar]
- Tresh, J.C. Capsicine: An alkaloid said to be contained in Capsicum fruit: Does it exist? Pharm. J. Transact. 1876, 6, 941–942. [Google Scholar]
- Buchheim, R. Fructus Capsici. Vierteljahresschr. Für Prakt. Pharm. 1872, 4, 507–521. [Google Scholar]
- Buchheim, R. Ueber die “scharfen Stoffe”. Arch. Heilkd. 1873, 13, 1–42. [Google Scholar]
- Högyes, E. Adatok a Capsicum annuum (paprika) alkatrészeinek élettani hatásához. Bull. Med. Sci. Assoc. 1877, 2, 51–56. [Google Scholar]
- Bennett, D.J.; Kirby, G.W. Constitution and biosynthesis of capsaicin. J. Chem. Soc. C 1968, 442–446. [Google Scholar] [CrossRef]
- Caterina, M.J.; Schumacher, M.A.; Tominaga, M.; Rosen, T.A.; Levine, J.D.; Julius, D. The capsaicin receptor: A heat-activated ion channel in the pain pathway. Nature 1997, 389, 816–824. [Google Scholar] [CrossRef]
- Bley, K.; Boorman, G.; Mohammad, B.; McKenzie, D.; Babbar, S. A comprehensive review of the carcinogenic and anticarcinogenic potential of capsaicin. Toxicol. Pathol. 2012, 40, 847–873. [Google Scholar] [CrossRef]
- Lujan-Mendez, F.; Roldan-Padron, O.; Castro-Ruiz, J.E.; Lopez-Martinez, J.; Garcia-Gasca, T. Capsaicinoids and Their Effects on Cancer: The “Double-Edged Sword” Postulate from the Molecular Scale. Cells 2023, 12, 2573. [Google Scholar] [CrossRef]
- Qiao, S.; Li, W.; Tsubouchi, R.; Haneda, M.; Murakami, K.; Yoshino, M. Involvement of peroxynitrite in capsaicin-induced apoptosis of C6 glioma cells. Neurosci. Res. 2005, 51, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, A.M.; Sanchez, M.G.; Malagarie-Cazenave, S.; Olea, N.; Diaz-Laviada, I. Induction of apoptosis in prostate tumor PC-3 cells and inhibition of xenograft prostate tumor growth by the vanilloid capsaicin. Apoptosis 2006, 11, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Athanasiou, A.; Smith, P.A.; Vakilpour, S.; Kumaran, N.M.; Turner, A.E.; Bagiokou, D.; Layfield, R.; Ray, D.E.; Westwell, A.D.; Alexander, S.P.H.; et al. Vanilloid receptor agonists and antagonists are mitochondrial inhibitors: How vanilloids cause non-vanilloid receptor mediated cell death. Biochem. Biophys. Res. Commun. 2007, 354, 50–55. [Google Scholar] [CrossRef]
- Kim, C.S.; Park, W.H.; Park, J.Y.; Kang, J.H.; Kim, M.O.; Kawada, T.; Yoo, H.; Han, I.S.; Yu, R. Capsaicin, a spicy component of hot pepper, induces apoptosis by activation of the peroxisome proliferator-activated receptor γ in HT-29 human colon cancer cells. J. Med. Food 2004, 7, 267–273. [Google Scholar] [CrossRef]
- Surh, Y.-J.; Lee, S.S. Capsaicin, a double-edged sword: Toxicity, metabolism, and chemopreventive potential. Life Sci. 1995, 56, 1845. [Google Scholar] [CrossRef] [PubMed]
- Surh, Y.J.; Lee, S.S. Capsaicin in hot chili pepper: Carcinogen, co-carcinogen or anticarcinogen? Food Chem. Toxicol. 1996, 34, 313–316. [Google Scholar] [CrossRef]
- Archer, V.E.; Jones, D.W. Capsaicin pepper, cancer and ethnicity. Med. Hypotheses 2002, 59, 450–457. [Google Scholar] [CrossRef]
- Lopez-Carrillo, L.; Camargo, M.C.; Schneider, B.G.; Sicinschi, L.A.; Hernandez-Ramirez, R.U.; Correa, P.; Cebrian, M.E. Capsaicin consumption, Helicobacter pylori CagA status and IL1B-31C > T genotypes: A host and environment interaction in gastric cancer. Food Chem. Toxicol. 2012, 50, 2118–2122. [Google Scholar] [CrossRef]
- Pabalan, N.; Jarjanazi, H.; Ozcelik, H. The Impact of Capsaicin Intake on Risk of Developing Gastric Cancers: A Meta-Analysis. J. Gastrointest. Cancer 2014, 45, 334–341. [Google Scholar] [CrossRef]
- Akagi, A.; Sano, N.; Uehara, H.; Minami, T.; Otsuka, H.; Izumi, K. Non-carcinogenicity of capsaicinoids in B6C3F1 mice. Food Chem. Toxicol. 1998, 36, 1065–1071. [Google Scholar] [CrossRef] [PubMed]
- Adetunji, T.L.; Olawale, F.; Olisah, C.; Adetunji, A.E.; Aremu, A.O. Capsaicin: A two decade systematic review of global research output and recent advances against human cancer. Front. Oncol. 1922, 12, 908487. [Google Scholar] [CrossRef]
- Zhang, B.; Zhao, J.; Wang, Z.; Guo, P.; Liu, A.; Du, G. Identification of multi-target anti-AD chemical constituents from traditional Chinese medicine formulae by integrating virtual screening and in vitro validation. Front. Pharmacol. 2021, 12, 709607. [Google Scholar] [CrossRef]
- Radhakrishna, G.K.; Ammunje, D.N.; Kunjiappan, S.; Ravi, K.; Vellingiri, S.; Ramesh, S.H.; Almeida, S.D.; Sireesha, G.; Ramesh, S.; Al-Qahtani, S.; et al. A Comprehensive Review of Capsaicin and Its Role in Cancer Prevention and Treatment. Drug Res. 2024, 74, 195–207. [Google Scholar] [CrossRef]
- Brown, K.C.; Modi, K.J.; Light, R.S.; Cox, A.J.; Long, T.E.; Gadepalli, R.S.; Rimoldi, J.M.; Miles, S.L.; Rankin, G.; Valentovic, M.; et al. Anticancer Activity of Region B Capsaicin Analogs. J. Med. Chem. 2023, 66, 4294–4323. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2001, 71, 209–249. [Google Scholar] [CrossRef]
- Holohan, C.; van Schoeybroek, S.; Longley, D.B.; Johnston, P.G. Cancer drug resistance: An evolving paradigm. Nat. Rev. Cancer 2013, 13, 714–726. [Google Scholar] [CrossRef] [PubMed]
- Heise, N.V.; Quast, J.; Csuk, R. Revisiting capsaicin and nonivamide: Their analogs exert strong inhibitory activity against cholinesterases. Eur. J. Med. Chem. Rep. 2024, 12, 100200. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, H.; Li, R.; Sterling, K.; Song, W. Amyloid β-based therapy for Alzheimer’s disease: Challenges, successes and future. Signal Transduct. Target. Ther. 2023, 8, 248. [Google Scholar] [CrossRef] [PubMed]
- Drosky, P.; Siehl, H.-U.; Zeller, K.-P.; Sieler, J.; Berger, S.; Sicker, D. Sharp, sharper, Capsaicin. Chem. Unserer Zeit 2015, 49, 114–122. [Google Scholar] [CrossRef]
- Olguín-Rojas, J.A.; Fayos, O.; Vázquez-León, L.A.; Ferreiro-González, M.; Rodríguez-Jimenes, G.D.; Palma, M.; Garcés-Claver, A.; Barbero, G.F. Progression of the Total and Individual Capsaicinoids Content in the Fruits of Three Different Cultivars of Capsicum chinense Jacq. Agronomy 2019, 9, 141. [Google Scholar] [CrossRef]
- Attuquayefio, V.K.; Buckle, K.A. Rapid sample preparation method for HPLC analysis of capsaicinoids in capsicum fruits and oleoresins. J. Agric. Food Chem. 1987, 35, 777–779. [Google Scholar] [CrossRef]
- Garces-Claver, A.; Arnedo-Andres, M.S.; Abadia, J.; Gil-Ortega, R.; Alvarez-Fernandez, A. Determination of Capsaicin and Dihydrocapsaicin in Capsicum Fruits by Liquid Chromatography-Electrospray/Time-of-Flight Mass Spectrometry. J. Agric. Food Chem. 2006, 54, 9303–9311. [Google Scholar] [CrossRef]
- Thompson, R.Q.; Phinney, K.W.; Sander, L.C.; Welch, M.J. Reversed-phase liquid chromatography and argentation chromatography of the minor capsaicinoids. Anal. Bioanal. Chem. 2005, 381, 1432–1440. [Google Scholar] [CrossRef]
- Migglautsch, A.K.; Willim, M.; Schweda, B.; Glieder, A.; Breinbauer, R.; Winkler, M. Aliphatic hydroxylation and epoxidation of capsaicin by cytochrome P450 CYP505X. Tetrahedron 2018, 74, 6199–6204. [Google Scholar] [CrossRef]
- Heise, N.; Becker, S.; Mueller, T.; Bache, M.; Csuk, R.; Güttler, A. Mitochondria-Targeting 1,5-Diazacyclooctane-Spacered Triterpene Rhodamine Conjugates Exhibit Cytotoxicity at Sub-Nanomolar Concentration against Breast Cancer Cells. Int. J. Mol. Sci. 2023, 24, 10695. [Google Scholar] [CrossRef] [PubMed]
- Heise, N.; Lehmann, F.; Csuk, R.; Mueller, T. Targeted theranostics: Near-infrared triterpenoic acid-rhodamine conjugates as prerequisites for precise cancer diagnosis and therapy. Eur. J. Med. Chem. 2023, 259, 115663. [Google Scholar] [CrossRef] [PubMed]

| Comp. | A2780 | A2780cis | A549 | MCF7 | HT29 | CCD18Co | SI |
|---|---|---|---|---|---|---|---|
| 25 | 2.8 | >100 | 60.5 | 38.4 | 78.5 | >100 | >35.7 |
| 27 | 3.4 | 76.4 | 50.3 | 32.6 | 42.4 | >100 | >29.0 |
| 29 | 3.0 | 59.5 | 55.3 | 15.9 | 68.4 | >100 | >33.4 |
| A | 15.9 | 84.3 | 70.3 | 42.1 | 51.0 | >100 | >6.3 |
| Cell Line | Extract (μg/mL) | Capsaicin A (μg/mL) |
|---|---|---|
| A2780 | 183.4 | 4.9 |
| A2780cis | 460.9 | 25.8 |
| A549 | 484.5 | 21.5 |
| MCF7 | 232.4 | 12.9 |
| HT29 | 309.9 | 15.6 |
| CCD18Co | >1250 | >30.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Heise, N.V.; Csuk, R.; Mueller, T. Small Structural Changes in Chili-Derived Capsaicin Resulting in Nonivamide Analogs of Significantly Improved Cytotoxicity and Good Tumor/Non-Tumor Cell Selectivity. Molecules 2025, 30, 3488. https://doi.org/10.3390/molecules30173488
Heise NV, Csuk R, Mueller T. Small Structural Changes in Chili-Derived Capsaicin Resulting in Nonivamide Analogs of Significantly Improved Cytotoxicity and Good Tumor/Non-Tumor Cell Selectivity. Molecules. 2025; 30(17):3488. https://doi.org/10.3390/molecules30173488
Chicago/Turabian StyleHeise, Niels V., René Csuk, and Thomas Mueller. 2025. "Small Structural Changes in Chili-Derived Capsaicin Resulting in Nonivamide Analogs of Significantly Improved Cytotoxicity and Good Tumor/Non-Tumor Cell Selectivity" Molecules 30, no. 17: 3488. https://doi.org/10.3390/molecules30173488
APA StyleHeise, N. V., Csuk, R., & Mueller, T. (2025). Small Structural Changes in Chili-Derived Capsaicin Resulting in Nonivamide Analogs of Significantly Improved Cytotoxicity and Good Tumor/Non-Tumor Cell Selectivity. Molecules, 30(17), 3488. https://doi.org/10.3390/molecules30173488









