Exploration of Raw Pigmented-Fleshed Sweet Potatoes Volatile Organic Compounds and the Precursors
Abstract
1. Introduction
2. Results
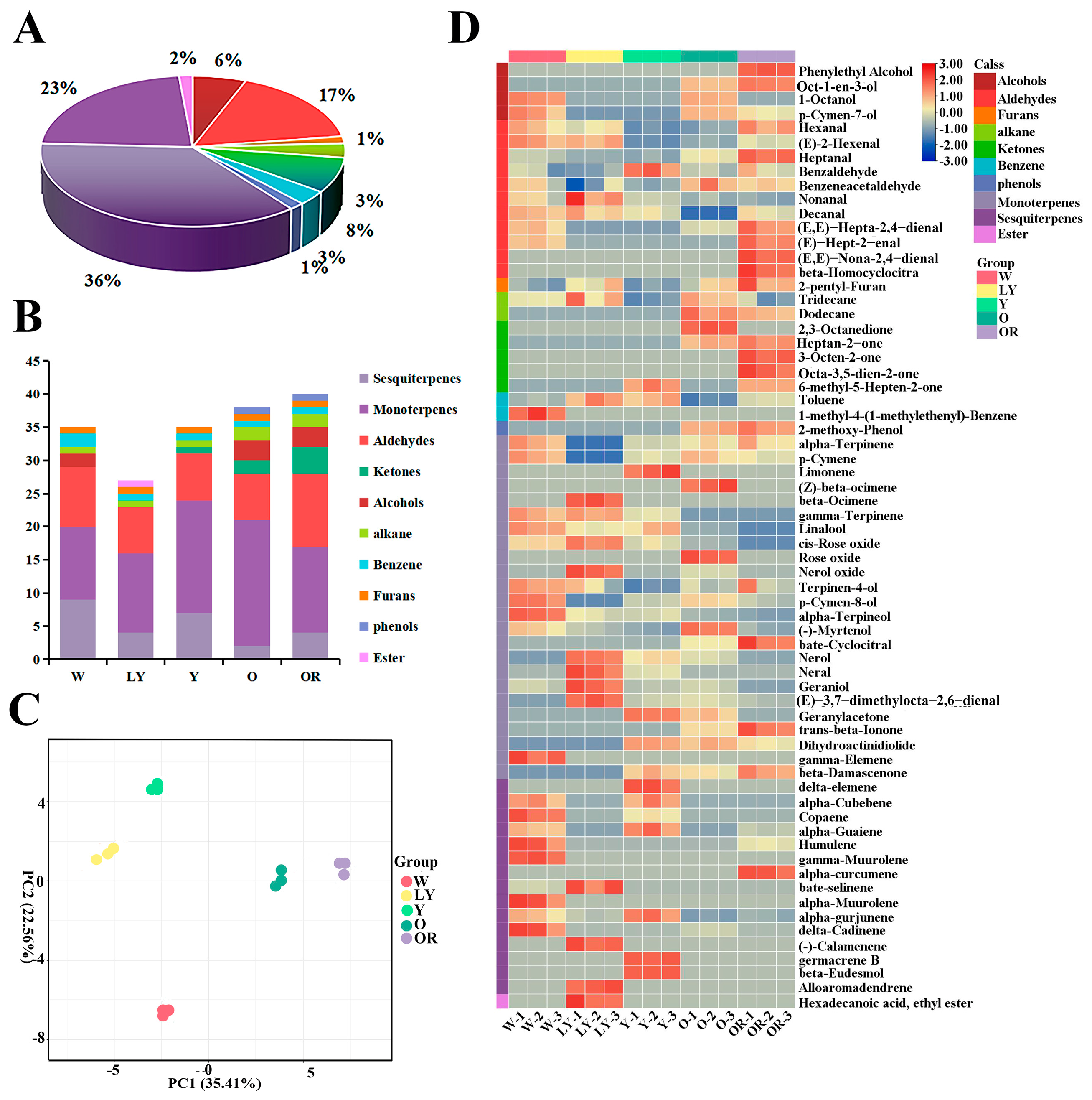
2.1. Analysis of Volatile Organic Compounds for Five Sweet Potato Cultivars
2.2. Analysis of Odor Activity Value of Five Sweet Potato Cultivars
2.3. Correlation Analysis of Aroma and Content of Starch, Soluble Sugar, and Carotenoid in Five Sweet Potato Cultivars
2.4. Correlation Analysis of Aroma with Amino Acids Contents in Five Sweet Potato Cultivars
2.5. Correlation Analysis of Aroma with Fatty Acids Contents in Five Sweet Potato Cultivars
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Determination of Volatile Organic Compounds by HS-SPME/GC-MS
4.3. Relative Odor Activity Value Analysis
4.4. Determination of Total Starch
4.5. Determination of Soluble Sugar Content
4.6. Determination of Amino Acids by HPLC-MS/MS
4.7. Determination of Fatty Acids by GC-MS
4.8. Data Processing and Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Laveriano-Santos, E.P.; López-Yerena, A.; Jaime-Rodríguez, C.; González-Coria, J.; Lamuela-Raventós, R.M.; Vallverdú-Queralt, A.; Romanyà, J.; Pérez, M. Sweet Potato Is Not Simply an Abundant Food Crop: A Comprehensive Review of Its Phyto-chemical Constituents, Biological Activities, and the Effects of Processing. Antioxidants 2022, 11, 1648. [Google Scholar] [CrossRef]
- Qin, Y.; Naumovski, N.; Ranadheera, C.S.; D’Cunha, N.M. Nutrition-related health outcomes of sweet potato (Ipomoea batatas) consumption: A systematic review. Food Biosci. 2022, 50, 102208. [Google Scholar] [CrossRef]
- Jia, R.; Zhang, R.; Gangurde, S.S.; Tang, C.; Jiang, B.; Li, G.; Wang, Z. Integrated analysis of carotenoid metabolites and transcriptome identifies key genes controlling carotenoid compositions and content in sweetpotato tuberous roots (Ipomoea batatas L.). Front. Plant Sci. 2022, 13, 993682. [Google Scholar] [CrossRef]
- Huang, J.; Qin, Y.; Xie, Z.; Wang, P.; Zhao, Z.; Huang, X.; Chen, Q.; Huang, Z.; Chen, Y.; Gao, A. Combined transcriptome and metabolome analysis reveal that the white and yellow mango pulp colors are associated with carotenoid and flavonoid accumulation, and phytohormone signaling. Genomics 2023, 115, 110675. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, R. Marketing Healthy Food in an African City: Consumer Motivations for Adopting Orange-Fleshed Sweet Potato in Maputo, Mozambique. Food Nutr. Bull. 2021, 42, 361–377. [Google Scholar] [CrossRef] [PubMed]
- Drapal, M.; Fraser, P.D. Determination of carotenoids in sweet potato (Ipomoea batatas L., Lam) tubers: Implications for accurate provitamin A determination in staple sturdy tuber crops. Phytochemistry 2019, 167, 112102. [Google Scholar] [CrossRef] [PubMed]
- Mostafa, S.; Wang, Y.; Zeng, W.; Jin, B. Floral Scents and Fruit Aromas: Functions, Compositions, Biosynthesis, and Regulation. Front. Plant Sci. 2022, 13, 860157. [Google Scholar] [CrossRef] [PubMed]
- Jin, W.; Zhang, Z.; Zhao, S.; Liu, J.; Gao, R.; Jiang, P. Characterization of volatile organic compounds of different pigmented rice after puffing based on gas chroma-tography-ion migration spectrometry and chemometrics. Food Res. Int. 2023, 169, 112879. [Google Scholar] [CrossRef]
- Zhou, Y.; Gao, S.; Wei, J.; Chen, X.; Zhu, S.; Zhou, X. Systematical construction of rice flavor types based on HS-SPME-GC–MS and sensory evaluation. Food Chem. 2023, 413, 135604. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Chen, Y.; Zhang, J.; Wang, Z.; Qi, K.; Li, H.; Tian, R.; Wu, X.; Qiao, X.; Zhang, S.; et al. Comparative analysis of volatile aromatic compounds from a wide range of pear (Pyrus L.) germplasm resources based on HS-SPME with GC–MS. Food Chem. 2023, 418, 135963. [Google Scholar] [CrossRef]
- Wang, D.; Duan, C.Q.; Shi, Y.; Zhu, B.Q.; Javed, H.U.; Wang, J. Free and glycosidically bound volatile compounds in sun-dried raisins made from different fragrance intensi-ties grape varieties using a validated HS-SPME with GC-MS method. Food Chem. 2017, 228, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Wang, M.; Wu, M.; Li, X.; Liu, H.; Niu, N.; Li, S.; Chen, L. Volatile organic compounds (VOCs) from plants: From release to detection. TrAC Trends Anal. Chem. 2023, 158, 116872. [Google Scholar] [CrossRef]
- Dudareva, N.; Klempien, A.; Muhlemann, J.K.; Kaplan, I. Biosynthesis, function and metabolic engineering of plant volatile organic compounds. New Phytol. 2013, 198, 16–32. [Google Scholar] [CrossRef]
- Wang, M.-S.; Fan, M.; Zheng, A.-R.; Wei, C.-K.; Liu, D.-H.; Thaku, K.; Wei, Z.-J. Characterization of a fermented dairy, sour cream: Lipolysis and the release profile of flavor compounds. Food Chem. 2023, 423, 136299. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Zhan, J.; Tang, X.; Li, T.; Duan, S. Characterization and identification of pork flavor compounds and their precursors in Chinese indigenous pig breeds by volatile profiling and multivariate analysis. Food Chem. 2022, 385, 132543. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Fu, J.J.; Mao, J.L.; Dong, X.P.; Chen, Y.W. Lipidomics reveals the relationship between lipid oxidation and flavor formation of basic amnio acids par-ticipated Low-Sodium cured large yellow croaker. Food Chem. 2023, 429, 136888. [Google Scholar] [CrossRef]
- Ni, Z.; Yang, Y.; Zhang, Y.; Hu, Q.; Lin, J.; Lin, H.; Hao, Z.; Wang, Y.; Zhou, J.; Sun, Y. Dynamic change of the carotenoid metabolic pathway profile during oolong tea processing with supplementary LED light. Food Res. Int. 2023, 169, 112839. [Google Scholar] [CrossRef]
- Shi, J.; Wu, W.; Zhang, Y.; Baldermann, S.; Peng, Q.; Wang, J.; Xu, L.; Yang, G.; Fu, J.; Lv, H.; et al. Comprehensive analysis of carotenoids constituents in purple-coloured leaves and carotenoid-derived aroma differences after processing into green, black, and white tea. LWT 2023, 173, 114286. [Google Scholar] [CrossRef]
- Zhang, R.; Chen, H.; Chen, Y.; Tang, C.; Jiang, B.; Wang, Z. Impact of different cooking methods on the flavor and chemical profile of yellow-fleshed table-stock sweet-potatoes (Ipomoea batatas L.). Food Chem. X 2023, 17, 100542. [Google Scholar] [CrossRef]
- Mayobre, C.; Pereira, L.; Eltahiri, A.; Bar, E.; Lewinsohn, E.; Garcia-Mas, J.; Pujol, M. Genetic dissection of aroma biosynthesis in melon and its relationship with climacteric ripening. Food Chem. 2021, 353, 129484. [Google Scholar] [CrossRef]
- Chen, X.; Fedrizzi, B.; Kilmartin, P.A.; Quek, S.Y. Development of volatile organic compounds and their glycosylated precursors in tamarillo (Solanum beta-ceum Cav.) during fruit ripening: A prediction of biochemical pathway. Food Chem. 2021, 339, 128046. [Google Scholar] [CrossRef] [PubMed]
- Mrudulakumari, V.U.; Lee, E.Y. Flavonoids, terpenoids, and polyketide antibiotics: Role of glycosylation and biocatalytic tactics in engineering glycosylation. Biotechnol. Adv. 2020, 41, 107550. [Google Scholar] [CrossRef] [PubMed]
- Werner, N.; Ramirez-Sarmiento, C.A.; Agosin, E. Protein engineering of carotenoid cleavage dioxygenases to optimize beta-ionone biosynthesis in yeast cell factories. Food Chem. 2019, 299, 125089. [Google Scholar] [CrossRef]
- Maoz, I.; Lewinsohn, E.; Gonda, I. Amino acids metabolism as a source for aroma volatiles biosynthesis. Curr. Opin. Plant Biol. 2022, 67, 102221. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, F.J.; Zamora, R. Formation of phenylacetic acid and benzaldehyde by degradation of phenylalanine in the pres-ence of lipid hydroperoxides: New routes in the amino acid degradation pathways initiated by lipid oxidation products. Food Chem. X 2019, 2, 100037. [Google Scholar] [CrossRef]
- Yao, Y.; Zhang, R.; Jia, R.; Deng, Y.; Wang, Z. Impact of different cooking methods on the chemical profile of orange-fleshed sweet potato (Ipomoea batatas L.). LWT 2023, 173, 114288. [Google Scholar] [CrossRef]
- Irshad, A.; Guo, H.; Ur Rehman, S.; Gu, J.; Wang, C.; Xiong, H.; Xie, Y.; Zhao, S.; Liu, L. Screening of Induced Mutants Led to the Identification of Starch Biosynthetic Genes Associated with Im-proved Resistant Starch in Wheat. Int. J. Mol. Sci. 2022, 23, 10741. [Google Scholar] [CrossRef]
- Maness, N. Extraction and analysis of soluble carbohydrates. Methods Mol. Biol. 2010, 639, 341–370. [Google Scholar]
- Kovács, Z.; Simon-Sarkadi, L.; Sovány, C.; Kirsch, K.; Galiba, G.; Kocsy, G. Differential effects of cold acclimation and abscisic acid on free amino acid composition in wheat. Plant Sci. 2011, 180, 61–68. [Google Scholar] [CrossRef]
- Li, S.-S.; Yuan, R.-Y.; Chen, L.-G.; Wang, L.-S.; Hao, X.-H.; Wang, L.-J.; Zheng, X.-C.; Du, H. Systematic qualitative and quantitative assessment of fatty acids in the seeds of 60 tree peony (Paeonia section Moutan DC.) cultivars by GC–MS. Food Chem. 2015, 173, 133–140. [Google Scholar] [CrossRef]
- Jiang, X.; Zhang, R.; Yao, Y.; Yang, Y.; Wang, B.; Wang, Z. Effect of cooking methods on metabolites of deep purple-fleshed sweetpotato. Food Chem. 2023, 429, 136931. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Zhang, M.; Ye, J.; Qian, M.; Li, X.; Zhao, W.; Bai, W. HS-SPME-GCMS and OAV analyses of characteristic volatile flavour compounds in salt-baked drumstick. LWT 2022, 170, 114041. [Google Scholar] [CrossRef]
- Eisele, T.A.; Semon, M.J. Best estimated aroma and taste detection threshold for guaiaicol in water and ap-plejuice. J. Food Sci. 2005, 70, S267–S269. [Google Scholar] [CrossRef]
- Fang, X.; Liu, Y.; Xiao, J.; Ma, C.; Huang, Y. GC–MS and LC-MS/MS metabolomics revealed dynamic changes of volatile and non-volatile compounds during withering process of black tea. Food Chem. 2023, 410, 135396. [Google Scholar] [CrossRef]
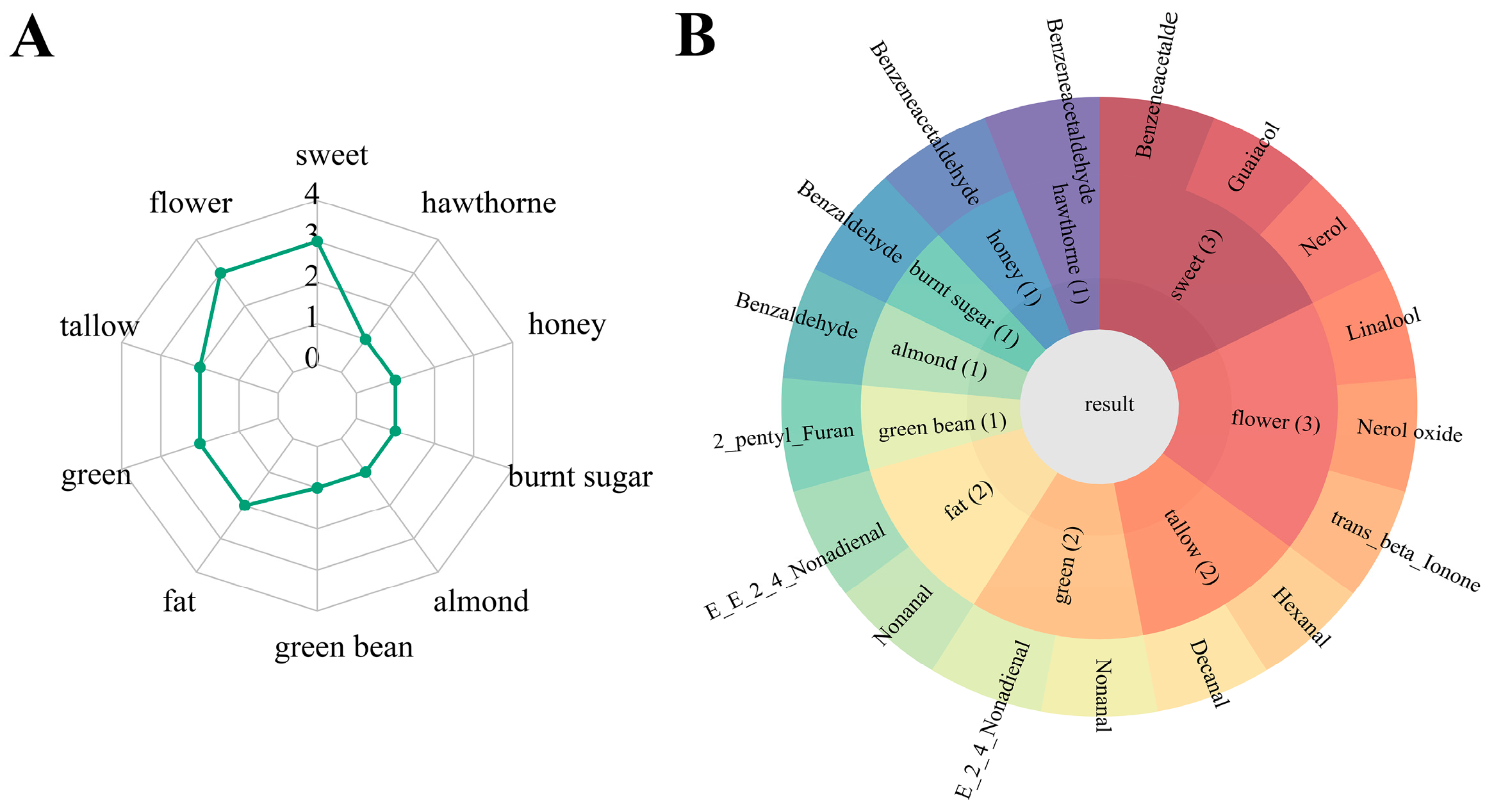

| Aroma Compounds | Class | Threshold (mg/kg) a | Description b | W | LY | Y | O | OR |
|---|---|---|---|---|---|---|---|---|
| Oct-1-en-3-ol | aldehydes | 0.002 | mushroom | 9.11 | 14.18 | |||
| Hexanal | aldehydes | 0.0045 | grass, tallow, fat | 19.15 | 15.28 | 7.09 | 8.89 | 22.77 |
| Benzaldehyde | aldehydes | 0.05 | almond, burnt sugar | 1.22 | 1.08 | 2.06 | 1.25 | 1.52 |
| Benzeneacetaldehyde | aldehydes | 0.04 | hawthorne, honey, sweet | 1.72 | 1.34 | 1.40 | 1.99 | 1.82 |
| Nonanal | aldehydes | 0.0035 | fat, citrus, green | 6.46 | 10.02 | 5.25 | 4.27 | 3.80 |
| Decanal | aldehydes | 0.003 | soap, orange peel, tallow | 10.37 | 10.00 | 7.08 | 7.40 | |
| (E,E)-nona-2,4-dienal | aldehydes | 0.00006 | fat, wax, green | 41.10 | ||||
| 2-pentyl-Furan | furan | 0.0048 | Green bean, butter | 3.88 | 7.05 | 3.51 | 6.71 | 9.59 |
| Guaiacol | phenols | 0.00017 | smoke, sweet, medicine | 95.90 | 114.44 | |||
| Linalool | terpenoids | 0.0015 | flower, lavender | 40.37 | 21.92 | 32.15 | 7.31 | |
| Nerol oxide | terpenoids | 0.007 | oil, flower | 35.85 | 4.46 | |||
| Nerol | terpenoids | 0.049 | sweet | 9.96 | 3.57 | 2.06 | 0.28 | |
| Geraniol | terpenoids | 0.0075 | rose, geranium | 8.64 | 49.92 | 10.99 | 15.01 | 2.50 |
| trans-β-Ionone | terpenoids | 0.000461 | seaweed, violet, flower, raspberry | 70.73 | 175.63 | |||
| alpha-gurjunene | terpenoids | 0.001 | wood, balsamic | 47.95 | 19.04 | 72.06 | 5.62 | 11.23 |
| W | LY | Y | O | OR | |
|---|---|---|---|---|---|
| Aspartic acid | 398.07 ± 0.06 ab | 378.58 ± 0.05 cd | 422.54 ± 0.03 ab | 426.98 ± 0.01 a | 388.80 ± 0.02 bc |
| Glutamic acid | 368.55 ± 0.06 b | 246.36 ± 0.05 d | 407.38 ± 0.03 a | 193.08 ± 0.01 e | 286.10 ± 0.03 c |
| Asparagine | 443.71 ± 0.02 a | 211.26 ± 0.07 c | 176.04 ± 0.04 d | 78.61 ± 0.02 e | 244.05 ± 0.03 b |
| Glutamine | 139.88 ± 0.02 a | 100.32 ± 0.06 b | 61.54 ± 0.05 c | 50.01 ± 0.01 d | 105.8 ± 0.04 b |
| Phenylalanine | 62.15 ± 0.02 d | 72.16 ± 0.06 bc | 126.82 ± 0.02 a | 67.95 ± 0.01 c | 75.93 ± 0.01 b |
| Tyrosine | 16.99 ± 0.02 d | 35.34 ± 0.06 a | 27.30 ± 0.03 b | 18.44 ± 0.02 d | 24.25 ± 0.01 c |
| Tryptophan | 11.94 ± 0.01 b | 19.11 ± 0.06 a | 10.90 ± 0.04 bc | 8.18 ± 0.01 d | 9.73 ± 0.03 c |
| Threonine | 58.53 ± 0.03 b | 37.86 ± 0.06 c | 108.83 ± 0.03 a | 33.47 ± 0.01 cd | 32.04 ± 0.01 d |
| Valine | 24.81 ± 0.04 bc | 32.46 ± 0.08 a | 25.64 ± 0.03 b | 19.42 ± 0.02 d | 22.56 ± 0.01 c |
| Methionine | 20.51 ± 0.01 a | 16.53 ± 0.07 b | 20.15 ± 0.03 a | 11.49 ± 0.02 d | 15.09 ± 0.01 c |
| Isoleucine | 16.21 ± 0.03 b | 24.95 ± 0.08 a | 15.38 ± 0.02 b | 12.65 ± 0.02 c | 14.21 ± 0.01 bc |
| Leucine | 18.37 ± 0.03 b | 21.98 ± 0.07 a | 16.10 ± 0.04 c | 11.48 ± 0.02 d | 15.48 ± 0.01 c |
| Lysine | 6.38 ± 0.07 b | 5.96 ± 0.03 b | 9.06 ± 0.04 a | 4.95 ± 0.03 c | 8.93 ± 0.01 a |
| Histidine | 19.73 ± 0.49 a | 15.08 ± 0.027 b | 12.27 ± 0.44 c | 8.69 ± 0.08 d | 12.94 ± 0.34 c |
| Serine | 93.75 ± 0.03 a | 79.69 ± 0.06 b | 81.01 ± 0.03 b | 57.60 ± 0.01 c | 62.48 ± 0.01 c |
| Glycine | 14.81 ± 0.02 a | 13.85 ± 0.09 a | 9.7 ± 0.03 b | 8.96 ± 0.07 b | 9.67 ± 0.01 b |
| Arginine | 25.75 ± 0.02 a | 20.46 ± 0.07 b | 18.05 ± 0.05 c | 10.74 ± 0.06 d | 27.71 ± 0.01 a |
| Alanine | 33.99 ± 0.05 b | 30.29 ± 0.08 c | 34.25 ± 0.03 b | 19.57 ± 0.02 d | 40.36 ± 0.01 a |
| Gamma-Aminobutyric acid | 5.34 ± 0.07 d | 8.88 ± 0.06 b | 6.92 ± 0.02 c | 5.43 ± 0.04 d | 24.21 ± 0.05 a |
| Proline | 14.32 ± 0.03 b | 19.74 ± 0.07 a | 14.69 ± 0.03 b | 9.01 ± 0.02 c | 18.76 ± 0.01 a |
| Total | 1793.78 | 1390.86 | 1604.58 | 1056.73 | 1439.10 |
| W | LY | Y | O | OR | |
|---|---|---|---|---|---|
| C10:0 | 8.88 × 10−3 ± 0.015 b | 0.88 × 10−3 ± 0.03 e | 16.87 × 10−3 ± 0.02 a | 1.44 × 10−3 ± 0.02 d | 2.81 × 10−3 ± 0.03 c |
| C12:0 | 7.89 × 10−2 ± 0.01 b | 1.33 × 10−2 ± 0.03 d | 9.77 × 10−2 ± 0.02 a | 1.56 × 10−2 ± 0.02 d | 4.21 ± 0.03 c |
| C14:0 | 2.53 × 10−2 ± 0.02 c | 1.94 × 10−2 ± 0.03 e | 4.54 × 10−2 ± 0.02 a | 0.02 × 10−2 ± 0.02 d | 0.03 ± 0.04 b |
| C15:0 | 9.84 × 10−3 ± 0.06 c | 5.33 × 10−3 ± 0.03 e | 12.83 × 10−3 ± 0.06 a | 0.01 ± 0.02 d | 0.01 ± 0.01 b |
| C14:1 | 2.51 × 10−3 ± 0.02 b | 3.22 × 10−3 ± 0.05 b | 6.22 × 10−3 ± 0.12 a | 2.91 × 10−3 ± 0.06 b | 3.37 × 10−3 ± 0.06 b |
| C16:0 | 1.83 ± 0.01 c | 1.56 ± 0.02 e | 2.75 ± 0.02 a | 1.70 ± 0.01 d | 2.27 ± 0.03 b |
| C17:0 | 6.80 × 10−3 ± 0.01 c | 6.85 × 10−3 ± 0.04 c | 11.66 × 10−3 ± 0.03 a | 6.66 × 10−3 ± 0.04 c | 8.08 × 10−3 ± 0.06 b |
| C18:0 | 1.12 ± 0.013 c | 0.93 ± 0.03 e | 1.66 ± 0.02 a | 1.03 ± 0.01 d | 1.42 ± 0.03 b |
| C18:1n9c | 0.02 ± 0.03 d | 0.03 ± 0.03 bc | 0.08 ± 0.05 a | 0.03 ± 0.04 cd | 0.04 ± 0.16 b |
| C18:2n6c | 0.61 ± 0.01 c | 0.55 ± 0.03 d | 0.87 ± 0.03 a | 0.48 ± 0.02 e | 0.79 ± 0.04 b |
| C18:3n3 | 0.02 ± 0.01 c | 0.02 ± 0.03 c | 0.04 ± 0.05 a | 0.02 ± 0.02 c | 0.03 ± 0.05 b |
| C18:3n6 | 1.86 × 10−3 ± 0.02 c | 1.61 × 10−3 ± 0.07 cd | 4.04 × 10−3 ± 0.04 a | 1.43 × 10−3 ± 0.10 d | 2.21 × 10−3 ± 0.05 b |
| C20:0 | 2.78 × 10−3 ± 0.04 c | 5.28 × 10−3 ± 0.08 b | 8.98 × 10−3 ± 0.03 a | 2.25 × 10−3 ± 0.07 c | 5.15 × 10−3 ± 0.04 b |
| C20:1 | 0.09 ± 0.019 d | 0.10 ± 0.03 c | 0.15 ± 0.03 b | 0.07 ± 0.02 e | 0.17 ± 0.04 a |
| C20:2 | 2.63 × 10−3 ± 0.04 c | 3.07 × 10−3 ± 0.02 bc | 5.27 × 10−3 ± 0.11 a | 2.47 × 10−3 ± 0.05 c | 3.71 × 10−3 ± 0.06 b |
| C20:3n6 | 0.75 × 10−3 ± 0.04 d | 1.22 × 10−3 ± 0.03 c | 2.93 × 10−3 ± 0.06 a | 0.94 × 10−3 ± 0.03 d | 1.53 × 10−3 ± 0.02 b |
| C20:5n3 | 8.96 × 10−3 ± 0.01 c | 9.29 × 10−3 ± 0.03 c | 17.83 × 10−3 ± 0.07 a | 6.86 × 10−3 ± 0.04 d | 12.68 × 10−3 ± 0.02 b |
| C21:0 | 0.01 ± 0.02 c | 0.01 ± 0.02 c | 0.03 ± 0.06 a | 0.01 ± 0.03 d | 0.02 ± 0.06 b |
| C22:0 | 0.26 ± 0.02 c | 0.28 ± 0.02 c | 0.60 ± 0.02 a | 0.12 ± 0.02 d | 0.40 ± 0.04 b |
| C22:1n9 | 4.12 × 10−3 ± 0.01 c | 4.18 × 10−3 ± 0.02 c | 9.39 × 10−3 ± 0.06 a | 3.86 × 10−3 ± 0.06 c | 0.01 ± 0.05 b |
| C22:2 | 3.15 × 10−3 ± 0.03 d | 4.15 × 10−3 ± 0.01 c | 8.67 × 10−3 ± 0.03 a | 2.05 × 10−3 ± 0.03 e | 5.72 × 10−3 ± 0.03 b |
| C22:6n3 | 0.01 ± 0.03 c | 0.01 ± 0.02 c | 0.01 ± 0.07 a | 0.01 ± 0.04 c | 0.01 ± 0.05 b |
| C23:0 | 0.23 × 10−3 ± 0.08 c | 0.43 × 10−3 ± 0.01 bc | 1.41 × 10−3 ± 0.15 a | 5.14 × 10−3 ± 0.07 b | 0.31 × 10−3 ± 0.06 bc |
| C24:0 | 2.90 × 10−3 ± 0.01 d | 3.61 × 10−3 ± 0.01 c | 6.74 × 10−3 ± 0.03 a | 1.75 × 10−3 ± 0.07 e | 4.67 × 10−3 ± 0.04 b |
| C24:1 | 0.26 × 10−3 ± 0.08 c | 0.26 × 10−3 ± 0.12 c | 1.51 × 10−3 ± 0.05 a | 0.43 × 10−3 ± 0.18 b | 0.16 × 10−3 ± 0.36 c |
| Total | 4.13 | 3.57 | 6.45 | 3.55 | 5.30 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yao, Y.; Zhang, R.; Jia, R.; Yao, Z.; Qiao, Y.; Wang, Z. Exploration of Raw Pigmented-Fleshed Sweet Potatoes Volatile Organic Compounds and the Precursors. Molecules 2024, 29, 606. https://doi.org/10.3390/molecules29030606
Yao Y, Zhang R, Jia R, Yao Z, Qiao Y, Wang Z. Exploration of Raw Pigmented-Fleshed Sweet Potatoes Volatile Organic Compounds and the Precursors. Molecules. 2024; 29(3):606. https://doi.org/10.3390/molecules29030606
Chicago/Turabian StyleYao, Yanqiang, Rong Zhang, Ruixue Jia, Zhufang Yao, Yake Qiao, and Zhangying Wang. 2024. "Exploration of Raw Pigmented-Fleshed Sweet Potatoes Volatile Organic Compounds and the Precursors" Molecules 29, no. 3: 606. https://doi.org/10.3390/molecules29030606
APA StyleYao, Y., Zhang, R., Jia, R., Yao, Z., Qiao, Y., & Wang, Z. (2024). Exploration of Raw Pigmented-Fleshed Sweet Potatoes Volatile Organic Compounds and the Precursors. Molecules, 29(3), 606. https://doi.org/10.3390/molecules29030606





