Double-Cabin Galvanic Cell-Synthesizing Nanoporous, Flower-like, Pb-Containing Pd–Au Nanoparticles for Nonenzymatic Formaldehyde Sensor
Abstract
1. Introduction
2. Results and Discussion
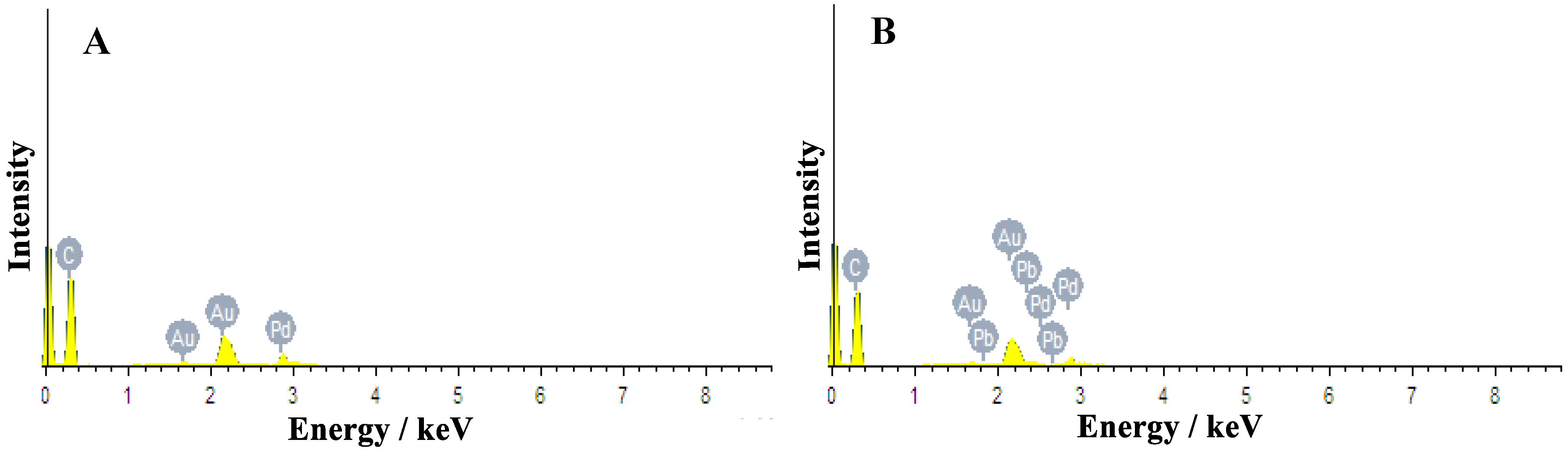
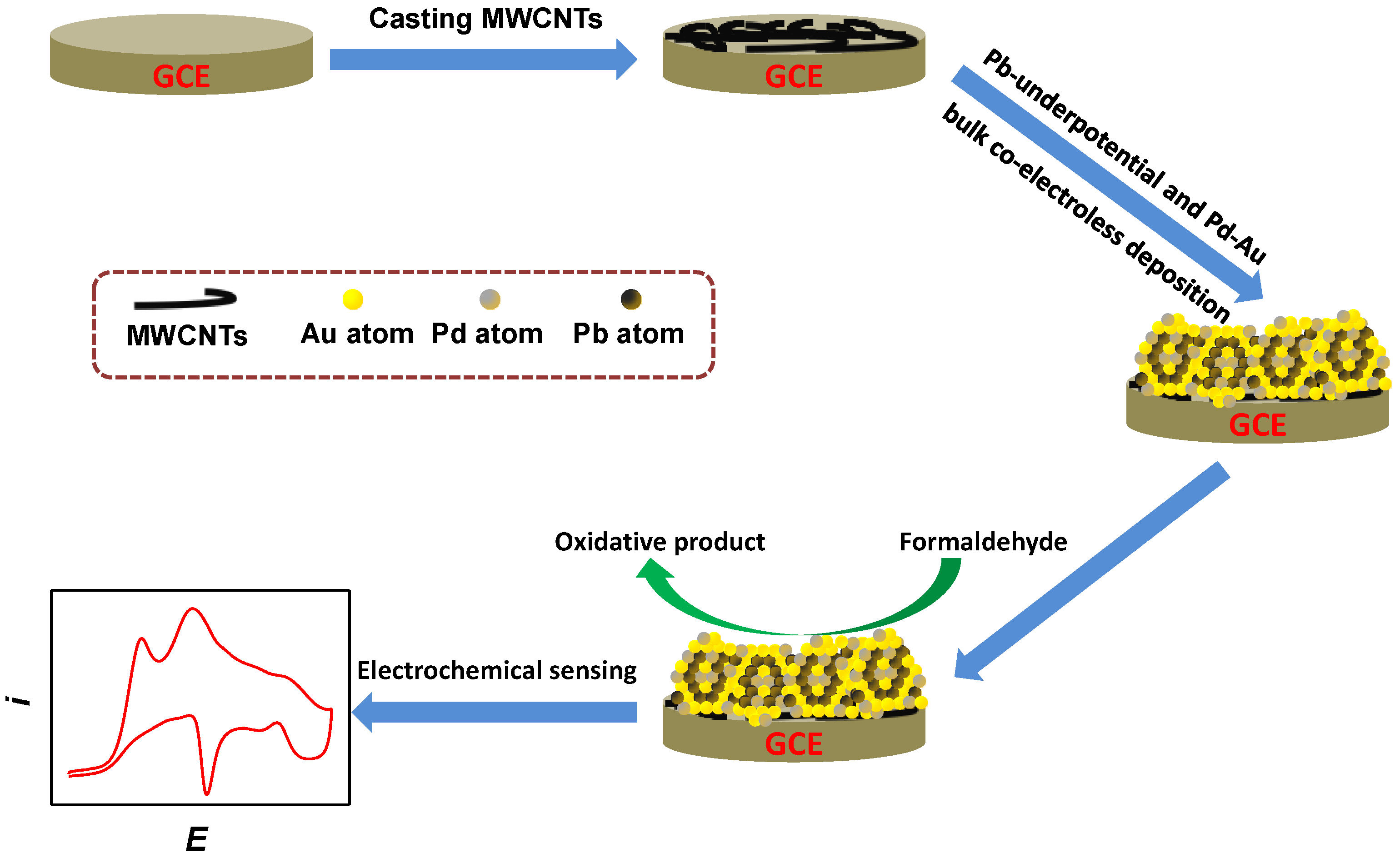
2.1. Fabrication and Characterization of Pb-Containing Pd–Au Nanoparticles
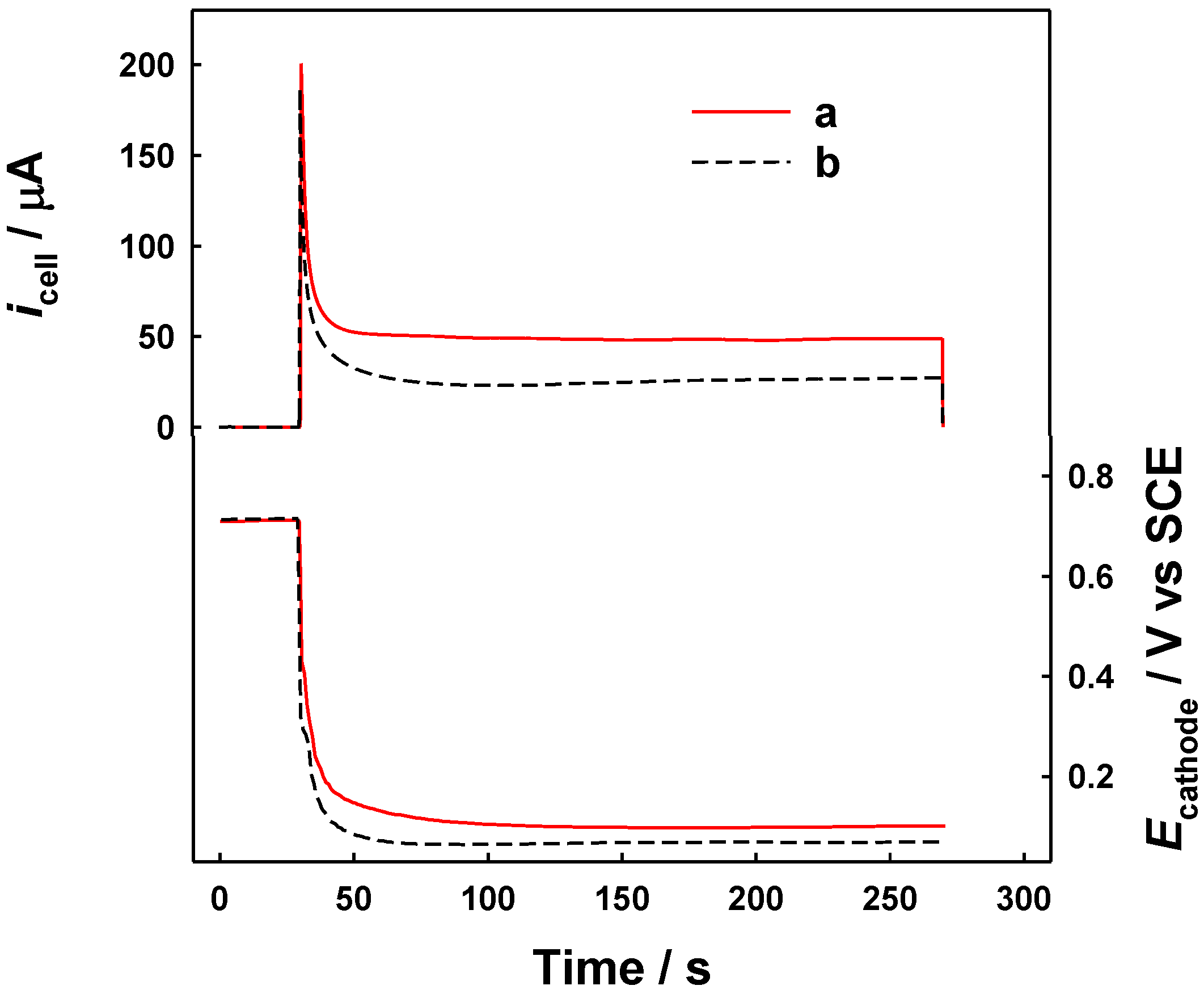
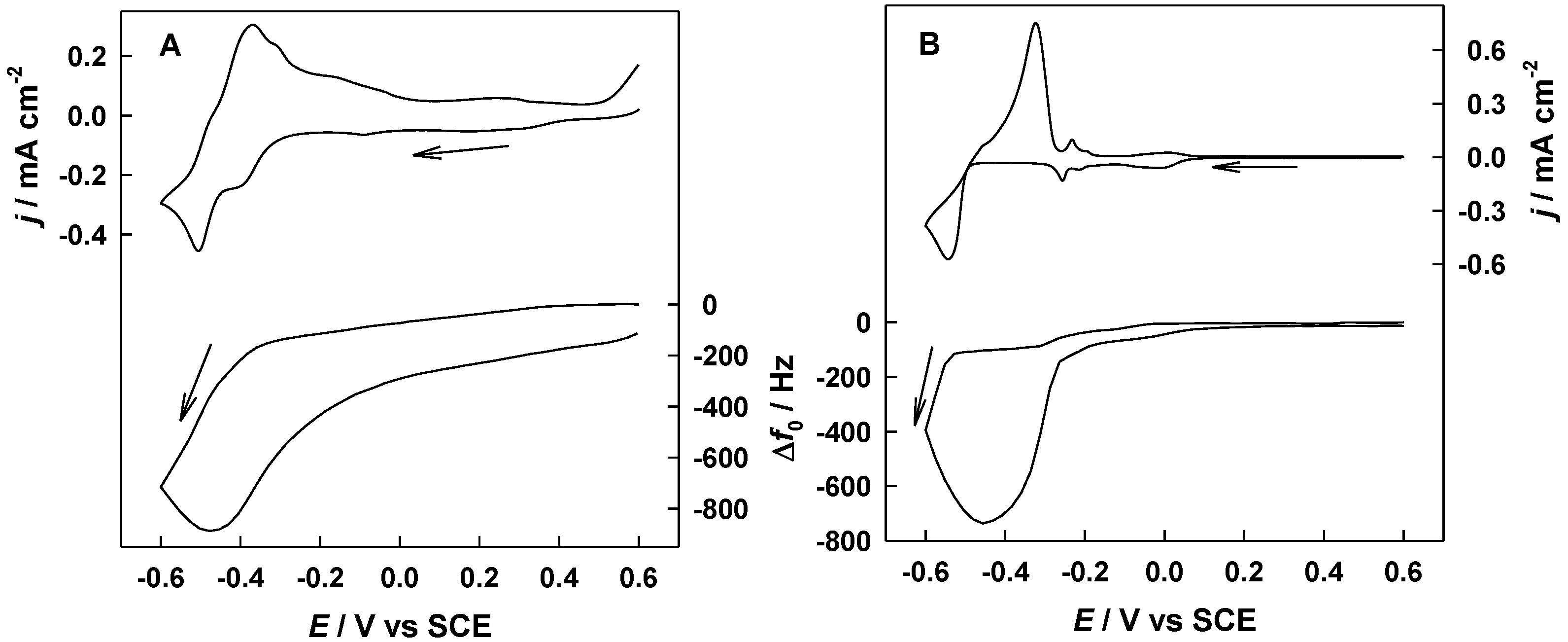
2.2. Electrochemical Study of the Evolution of Cathodic Deposition of Pb during the GRR Process
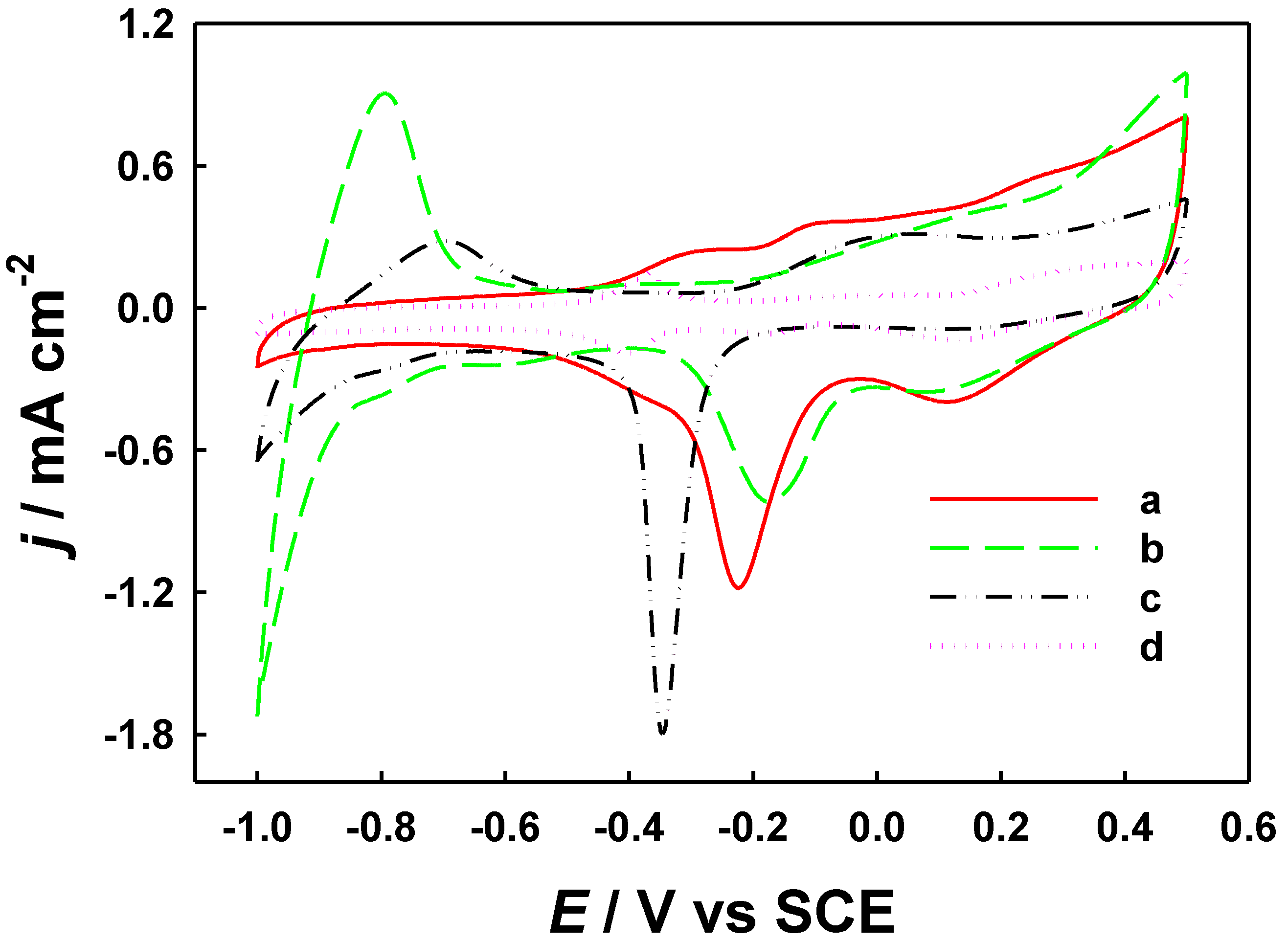
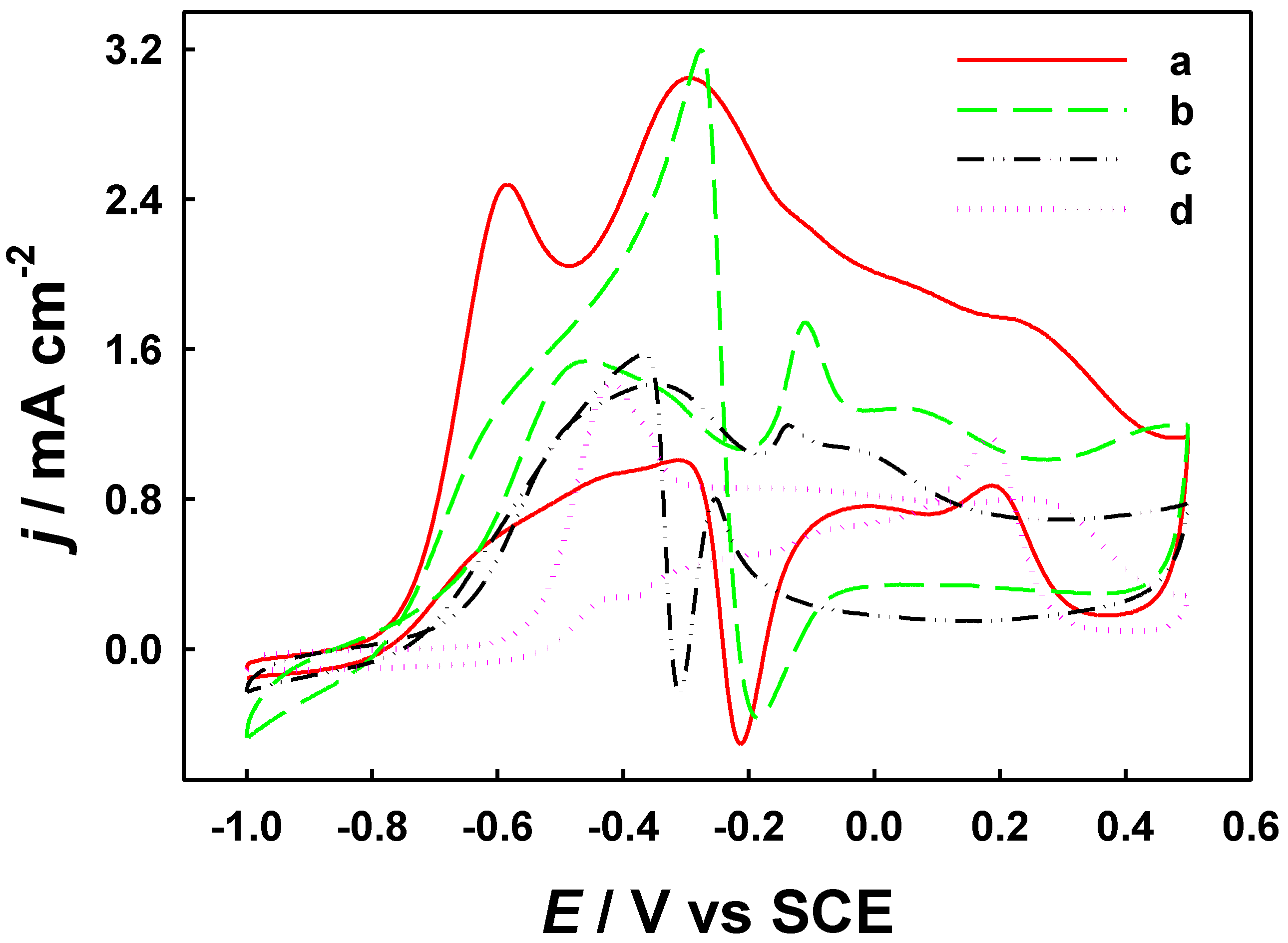
2.3. Electrochemical Behaviors and Electrocatalytic Responses to Formaldehyde on Modified Electrodes
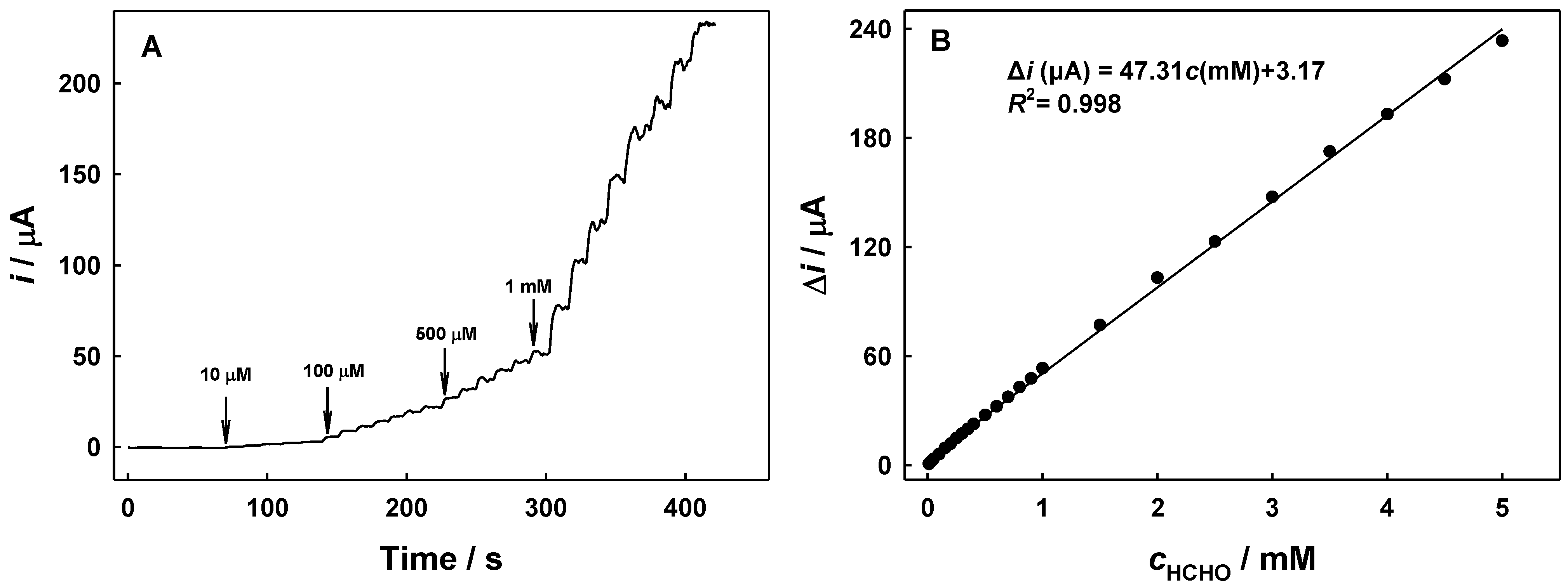
2.4. Amperometric Sensing of Formaldehyde
2.5. Interference Tests, Repeatability, and Stability of the Sensor
2.6. Real-Sample Analysis
3. Materials and Methods
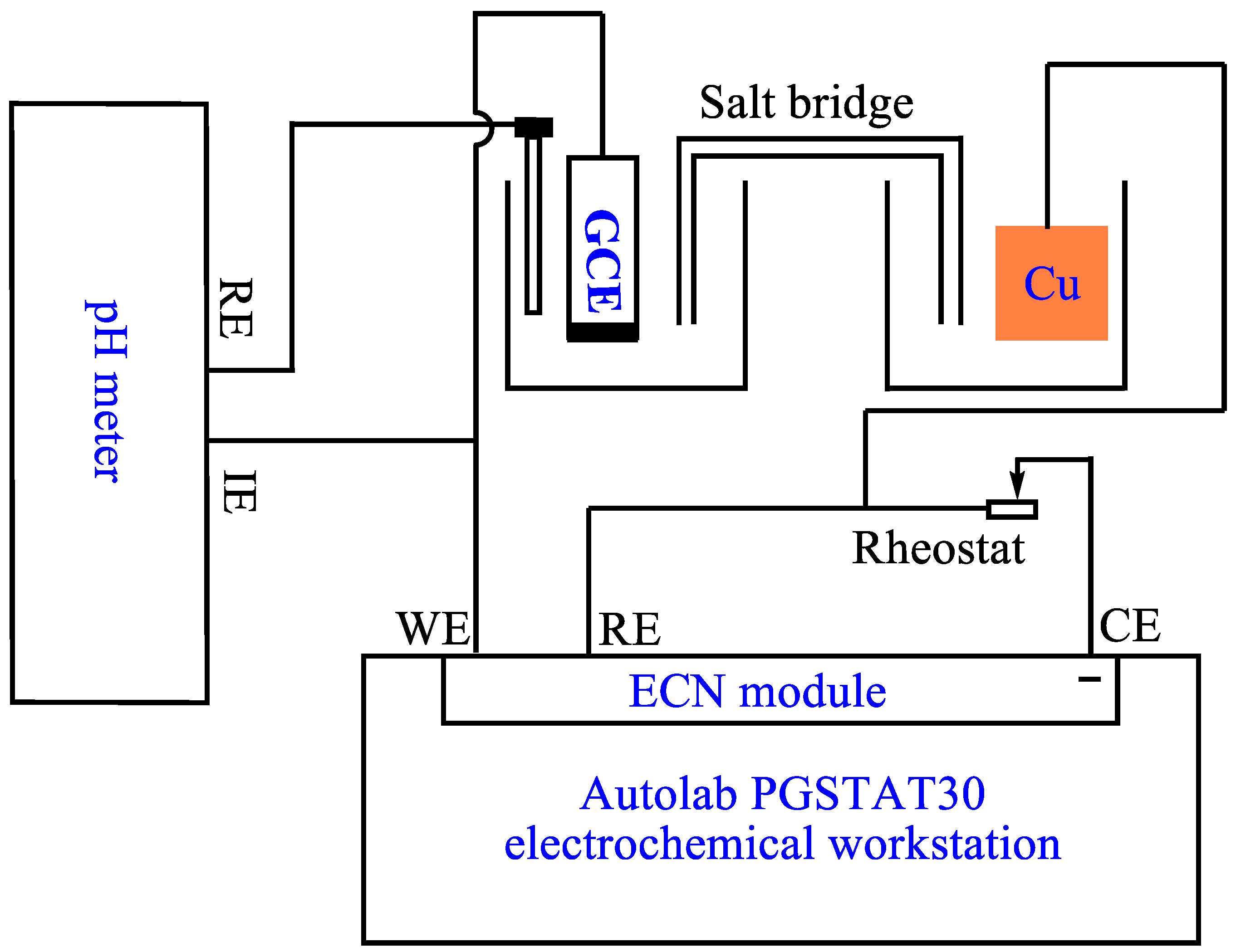
3.1. Instrumentation and Reagents
3.2. DCGC Synthesis of Pb-Containing Pd–Au Nanoparticles on MWCNTs/GCE
3.3. Electrochemical Characterization of the Modified Electrodes and Nonenzymatic Formaldehyde Sensor
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wu, X.; Li, Z.; Tao, J.; Zhao, J.; Xie, Y.; Zhao, S. Efficient removal of formaldehyde from wastewater by Cu defect induction of dendritic snowflake Cu7S4 Fenton-like catalysts. J. Environ. Chem. Eng. 2024, 12, 111982. [Google Scholar] [CrossRef]
- Wang, H.; Dong, X.; Tang, R.; Li, J.; Sun, Y.; Wang, Z.; Kim, K.-H.; Dong, F. Selective breakage of C-H bonds in the key oxidation intermediates of gaseous formaldehyde on self-doped CaSn(OH)6 cubes for safe and efficient photocatalysis. Appl. Catal. B-Environ. Energy 2020, 277, 119214. [Google Scholar] [CrossRef]
- Kazmi, B.; Shareef, R.; Noman, S.; Saeed, S.; Zehra, T.; Masood, Z.; Albasher, G.; Juchelková, D. Towards greener approach: Techno-economic insights into formaldehyde bio production from a hybrid pine and mustard biomass combination. Process. Saf. Environ. 2024, 186, 969–979. [Google Scholar] [CrossRef]
- Zhang, H.; Zheng, Z.; Yu, T.; Liu, C.; Qian, H.; Li, J. Seasonal and diurnal patterns of outdoor formaldehyde and impacts on indoor environments and health. Environ. Res. 2022, 205, 112550. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.-Q.; Zhang, Z.-Q. Microwave-assisted on-line derivatization for sensitive flow injection fluorometric determination of formaldehyde in some foods. Talanta 2009, 80, 242–245. [Google Scholar] [CrossRef] [PubMed]
- Lavilla, I.; Cabaleiro, N.; Pena, F.; Calle, I.d.l.; Bendicho, C. Ultrasound-assisted emulsification microextraction with simultaneous derivatization coupled to fibre optics-based cuvetteless UV-vis micro-spectrophotometry for formaldehyde determination in cosmetic samples. Anal. Chim. Acta 2010, 674, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, F.; Careri, M.; Musci, M.; Mangia, A. Fish and food safety: Determination of formaldehyde in 12 fish species by SPME extraction and GC-MS analysis. Food Chem. 2007, 100, 1049–1053. [Google Scholar] [CrossRef]
- Miralles, P.; Chisvert, A.; Alonso, M.J.; Hernandorena, S.; Salvador, A. Determination of free formaldehyde in cosmetics containing formaldehyde-releasing preservatives by reversed-phase dispersive liquid-liquid microextraction and liquid chromatography with post-column derivatization. J. Chromatogr. A 2018, 1543, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yu, Y.; Zhang, C.; Song, N.; Guo, Z.; Liang, M. Highly sensitive and selective detection of formaldehyde via bio-electrocatalysis over aldehyde dehydrogenase. Anal. Chem. 2022, 94, 15827–15831. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Hao, Y.; Huang, L.; Luo, Y.; Chen, S.; Xu, M.; Chen, W. Recent advances in electrochemical sensors for formaldehyde. Molecules 2024, 29, 327. [Google Scholar] [CrossRef]
- Rahman, M.M. Efficient formaldehyde sensor development based on Cu-codoped ZnO nanomaterial by an electrochemical approach. Sens. Actuators B Chem. 2020, 305, 127541. [Google Scholar] [CrossRef]
- Shimomura, T.; Itoh, T.; Sumiya, T.; Mizukami, F.; Ono, M. Electrochemical biosensor for the detection of formaldehyde based on enzyme immobilization in mesoporous silica materials. Sens. Actuators B Chem. 2008, 135, 268–275. [Google Scholar] [CrossRef]
- He, J.; Xu, X.; Sun, H.; Miao, T.; Li, M.; Zhou, S.; Zhou, W. Participation of lattice oxygen in perovskite oxide as a highly sensitive sensor for p-phenylenediamine detection. Molecules 2023, 28, 1122. [Google Scholar] [CrossRef]
- He, J.; Xu, X.; Li, M.; Zhou, S.; Zhou, W. Recent advances in perovskite oxides for non-enzymatic electrochemical sensors: A review. Anal. Chim. Acta 2023, 1251, 341007. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudian, M.R.; Basirun, W.J.; Woi, P.M.; Hazarkhani, H.; Alias, Y.B. Voltammetric sensing of formaldehyde by using a nanocomposite prepared by reductive deposition of palladium and platinum on polypyrrole-coated nitrogen-doped reduced graphene oxide. Microchim. Acta 2019, 186, 369. [Google Scholar] [CrossRef]
- Safavi, A.; Farjami, F. Electrochemical design of ultrathin palladium coated gold nanoparticles as nanostructured catalyst for amperometric detection of formaldehyde. Electroanalysis 2011, 23, 1842–1848. [Google Scholar] [CrossRef]
- Zhou, S.; McIlwrath, K.; Jackson, G.; Eichhorn, B. Enhanced CO tolerance for hydrogen activation in Au-Pt dendritic. J. Am. Chem. Soc. 2006, 128, 1780–1781. [Google Scholar] [CrossRef]
- Zhang, J.; Lv, F.; Li, Z.; Jiang, G.; Tan, M.; Yuan, M.; Zhang, Q.; Cao, Y.; Zheng, H.; Zhang, L.; et al. Cr-doped Pd metallene endows a practical formaldehyde sensor new limit and high selectivity. Adv. Mater. 2021, 34, 2105276. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, M.; Cai, Z.; Chen, M.; Cheng, F. A novel electrochemical sensor for formaldehyde based on palladium nanowire arrays electrode in alkaline media. Electrochim. Acta 2012, 68, 172–177. [Google Scholar] [CrossRef]
- Kaviana, S.; Azizi, S.N.; Ghasemi, S. Novel bimetallic nanoporous Pd-Cu-SBA-16/CPE as a highly sensitive sensor for determination of formaldehyde. J. Electroanal. Chem. 2017, 799, 308–314. [Google Scholar] [CrossRef]
- Sarin, S.; Zhu, H.; Jaatinen, E.; Xiao, Q.; Liu, H.; Jia, J.; Chen, C.; Zhao, J. Enhancing catalytic performance of palladium in gold and palladium alloy nanoparticles for organic synthesis reactions through visible light irradiation at ambient temperatures. J. Am. Chem. Soc. 2013, 135, 5793–5801. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zhang, J.; Kuang, Q.; Xie, S.; Jiang, Z.; Xie, Z.; Zheng, L. Cu2+-assisted synthesis of hexoctahedral Au-Pd alloy nanocrystals with high-index facets. J. Am. Chem. Soc. 2011, 133, 17114–17117. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.W.; Kim, N.H.; Lee, K.Y.; Kwon, K.; Kim, M.; Han, S.W. Synthesis and characterization of flower-shaped porous Au-Pd alloy nanoparticles. J. Phys. Chem. C 2008, 112, 6717–6722. [Google Scholar] [CrossRef]
- Hong, J.W.; Kim, D.; Lee, Y.W.; Kim, M.; Kang, S.W.; Han, S.W. Atomic-distribution-dependent electrocatalytic activity of Au-Pd bimetallic nanocrystal. Angew. Chem. Int. Ed. 2011, 50, 8876–8880. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Bliznakov, S.; Liu, Z.; Fang, J.; Dimitrov, N. Composition-dependent electrocatalytic activity of Pt-Cu nanocube catalysts for formic acid oxidation. Angew. Chem. Int. Ed. 2010, 49, 1282–1285. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Guo, S.; Zhai, Y.; Dong, S. Layer-by-layer self-assembly for constructing a graphene/platinum nanoparticle three-dimensional hybrid nanostructure using ionic liquid as a linker. Langmuir 2010, 26, 7614–7618. [Google Scholar] [CrossRef] [PubMed]
- Hwang, S.J.; Yoo, S.J.; Jang, S.; Lim, T.-H.; Hong, S.A.; Kim, S.-K. Ternary Pt-Fe-Co alloy electrocatalysts prepared by electrodeposition: Elucidating the roles of Fe and Co in the oxygen reduction reaction. J. Phys. Chem. C 2011, 115, 2483–2488. [Google Scholar] [CrossRef]
- Ulapane, S.B.; Kamathewatta, N.J.B.; Borkowski, A.K.; Steuart, S.J.; Berrie, C.L. Periodic silver and gold nanodot array fabrication on nanosphere lithography-based patterns using electroless deposition. J. Phys. Chem. C 2020, 124, 15646–15655. [Google Scholar] [CrossRef]
- Zhang, C.; Luo, L.; Luo, J.; Evans, D.G.; Sun, X. A process-analysis microsystem based on density gradient centrifugation and its application in the study of the galvanic replacement mechanism of Ag nanoplates with HAuCl4. Chem. Commun. 2012, 48, 7241–7243. [Google Scholar] [CrossRef]
- Qu, L.; Dai, L.; Osawa, E. Shape/size-controlled syntheses of metal nanoparticles for site-selective modification of carbon nanotubes. J. Am. Chem. Soc. 2006, 128, 5523–5532. [Google Scholar] [CrossRef]
- Personick, M.L.; Langille, M.R.; Zhang, J.; Mirkin, C.A. Shape control of gold nanoparticles by silver underpotential deposition. Nano Lett. 2011, 11, 3394–3398. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Henry, J.B.; Fortgang, P.; Henig, J.; Plumeré, N.; Bandarenka, A.S. In depth analysis of complex interfacial processes: In situ electrochemical characterization of deposition of atomic layers of Cu, Pb and Te on Pd electrodes. RSC Adv. 2012, 2, 10994–11006. [Google Scholar] [CrossRef]
- Shimazu, K.; Kawaguchi, T.; Isomura, T. Construction of mixed mercaptopropionic acid/alkanethiol monolayers of controlled composition by structural control of a gold substrate with underpotentially deposited lead atoms. J. Am. Chem. Soc. 2002, 124, 652–661. [Google Scholar] [CrossRef] [PubMed]
- Bagchi, J.; Bhattacharya, S.K. The effect of composition of Ni-supported Pt-Ru binary anode catalysts on ethanol oxidation for fuel cells. J. Power Sources 2007, 163, 661–670. [Google Scholar] [CrossRef]
- Safavi, A.; Maleki, N.; Farjami, F.; Farjami, E. Electrocatalytic oxidation of formaldehyde on palladium nanoparticles electrodeposited on carbon ionic liquid composite electrode. J. Electroanal. Chem. 2009, 626, 75–79. [Google Scholar] [CrossRef]
- Wang, Y.; Nguyen, T.S.; Liu, X.; Wang, X. Novel palladium-lead (Pd–Pb/C) bimetallic catalysts for electrooxidation of ethanol in alkaline media. J. Power Sources 2010, 195, 2619–2622. [Google Scholar] [CrossRef]
- Xi, H.; Chen, X.; Cao, Y.; Xu, J.; Ye, C.; Deng, D.; Zhang, J.; Huang, G. Electrochemical determination of formaldehyde via reduced AuNPs@PPy composites modified electrode. Microchem. J. 2020, 156, 104846. [Google Scholar] [CrossRef]
- Nachaki, E.O.; Ndangili, P.M.; Naumih, N.M.; Masika, E. Nickel-palladium-based electrochemical sensor for quantitative detection of formaldehyde. Chemistryselect 2018, 3, 384–392. [Google Scholar] [CrossRef]
- Trivedi, D.; Crosse, J.; Tanti, J.; Cass, A.J.; Toghill, K.E. The electrochemical determination of formaldehyde in aqueous media using nickel modified electrodes. Sens. Actuators B Chem. 2018, 270, 298–303. [Google Scholar] [CrossRef]
- Soleh, A.; Saisahas, K.; Promsuwan, K.; Saichanapan, J.; Thavarungkul, P.; Kanatharana, P.; Meng, L.; Mak, W.C.; Limbut, W. A wireless smartphone-based “tap-and-detect” formaldehyde sensor with disposable nano-palladium grafted laser-induced graphene (nanoPd@LIG) electrodes. Talanta 2023, 254, 124169. [Google Scholar] [CrossRef]
- Xie, Q.; Wang, J.; Zhou, A.; Zhang, Y.; Liu, H.; Xu, Z.; Yuan, Y.; Deng, M.; Yao, S. A study of depletion layer effects on equivalent circuit parameters using an electrochemical quartz crystal impedance system. Anal. Chem. 1999, 71, 4649–4656. [Google Scholar] [CrossRef]
- Tan, Y.; Xie, Q.; Huang, J.; Duan, W.; Ma, M.; Yao, S. Study on glucose biofuel cells using an electrochemical noise device. Electroanalysis. Electroanalysis 2008, 14, 1599–1606. [Google Scholar] [CrossRef]
- Wang, T.; Fu, Y.; Chai, L.; Chao, L.; Bu, L.; Meng, Y.; Chen, C.; Ma, M.; Xie, Q.; Yao, S. Filling carbon nanotubes with prussian blue nanoparticles of high peroxidase-like catalytic activity for colorimetric chemo- and biosensing. Chem.-Eur. J. 2014, 20, 2623–2630. [Google Scholar] [CrossRef] [PubMed]


| Modified Electrode | Linear Range (mM) | LOD (μM) | References |
|---|---|---|---|
| Au@PPy composites-Glassy carbon | 0.4−2.4 | 20 | [37] |
| Ni-Pd/GCE | 0.01–1.0 | 5.4 | [38] |
| Pd-Cu-SBA-16/CPE | 1.79–121.86 | 16 | [20] |
| Pd2Pt1 PPy/N-rGONC/GCE | 0.01−0.9 | 27 | [15] |
| Ni-GCE | 0.01−1.0 | 6 | [39] |
| Cr-Pdene/C | 0.001–0.005 | 1.0 | [18] |
| CILE/Au/Pd | 0.015–1.4, 1.4–56.7 | 3.0 | [16] |
| nanoPd@LIG | 0.01–4.0 | 6.4 | [40] |
| Pb-Pd3Au1/MWCNTs/GCE | 0.01–5.0 | 0.89 | This work |
| Sample | Added (μM) | Found (μM) | Recovery (%) | RSD (%) |
|---|---|---|---|---|
| 100 | 102.8 | 102.8 | 5.4 | |
| Tap Water | 200 | 201.1 | 100.6 | 3.9 |
| 400 | 402.9 | 100.7 | 3.4 | |
| 100 | 108.1 | 108.1 | 5.5 | |
| Lake Water | 200 | 189.4 | 94.7 | 4.1 |
| 400 | 388.3 | 97.1 | 3.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, Z.; Tang, Z.; Chao, L. Double-Cabin Galvanic Cell-Synthesizing Nanoporous, Flower-like, Pb-Containing Pd–Au Nanoparticles for Nonenzymatic Formaldehyde Sensor. Molecules 2024, 29, 2772. https://doi.org/10.3390/molecules29122772
Huang Z, Tang Z, Chao L. Double-Cabin Galvanic Cell-Synthesizing Nanoporous, Flower-like, Pb-Containing Pd–Au Nanoparticles for Nonenzymatic Formaldehyde Sensor. Molecules. 2024; 29(12):2772. https://doi.org/10.3390/molecules29122772
Chicago/Turabian StyleHuang, Zhao, Zhongsen Tang, and Long Chao. 2024. "Double-Cabin Galvanic Cell-Synthesizing Nanoporous, Flower-like, Pb-Containing Pd–Au Nanoparticles for Nonenzymatic Formaldehyde Sensor" Molecules 29, no. 12: 2772. https://doi.org/10.3390/molecules29122772
APA StyleHuang, Z., Tang, Z., & Chao, L. (2024). Double-Cabin Galvanic Cell-Synthesizing Nanoporous, Flower-like, Pb-Containing Pd–Au Nanoparticles for Nonenzymatic Formaldehyde Sensor. Molecules, 29(12), 2772. https://doi.org/10.3390/molecules29122772






