Antibacterial and Antibiofilm Effects of Allelopathic Compounds Identified in Medicago sativa L. Seedling Exudate against Escherichia coli
Abstract
1. Introduction
2. Results
2.1. Co-Cultivation of Alfalfa Seed with Barley: Impact on Germination and Growth
- The germination of barley seeds and the subsequent seedling development was massively retarded. The related delay corresponded to up to three days in comparison to the controls.
- The number of germinated seeds was much lower compared to the controls.
- The root and shoot development was significantly reduced in comparison to the controls.
2.2. Allelopathic Effect of Alfalfa Seed Exudates on Various Plant Species
2.3. Identification of the Substances Responsible for the Allelopathic Effect in Alfalfa Seeds Exudates
2.4. Allelopathic Effect of Pure Main Components of the Alfalfa Seed Exudates
2.5. Antibacterial and Antibiofilm Effects of the Alfalfa Seedling Exudate and Its Identified Compounds against Escherichia coli
2.5.1. Sensitivity of E. coli Isolates to the Tested Compounds
2.5.2. Antibiofilm Action
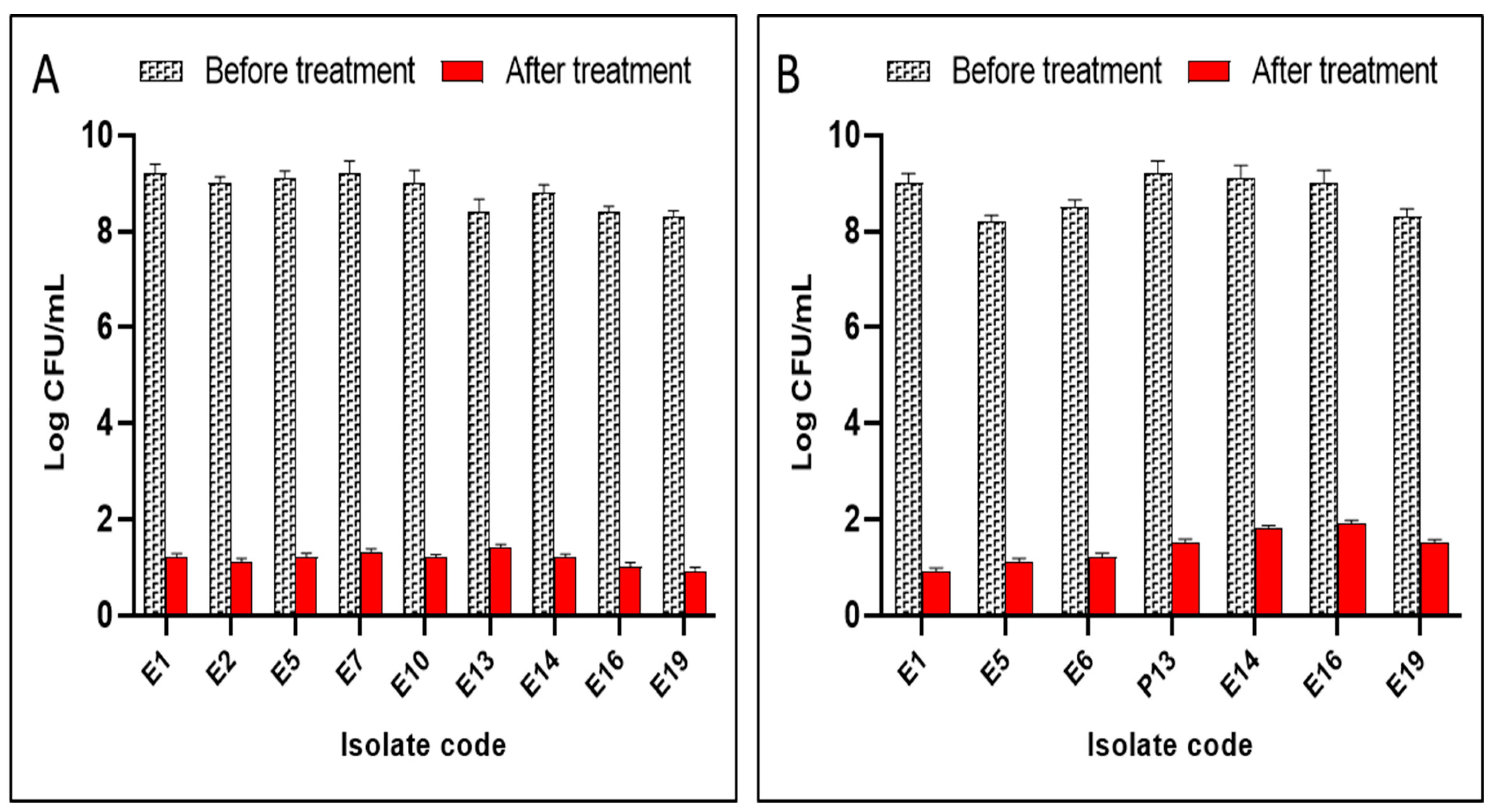
2.5.3. Count of Colony-Forming Units per Millilitre (CFU/mL)
3. Discussion
3.1. Hyperoside and Other Flavonoids
3.2. Canavanine
3.3. Uptake of the Allelochemicals
3.4. Beneficial Implications of Allelochemicals for Agriculture
3.5. Antimicrobial Activity of Allelochemicals against Pathogenic Bacteria
4. Materials and Methods
4.1. Chemicals
4.2. Co-Cultivation of Barley Seed with Alfalfa Seed
4.3. Preparation of Medicago sativa Seed Exudate
4.4. Evaluation of Allelopathic Effects of Alfalfa Seedling Exudate on Germination
4.5. HPLC Analysis of Alfalfa Exudate
4.6. Compounds Identification and Quantification Using LC-MS
4.7. Allelopathic Effect of Pure Main Components of the Alfalfa Seed Exudates
4.8. Antibacterial and Antibiofilm Effects of the Alfalfa Seedling Exudate and Its Identified Compounds against Escherichia coli
4.8.1. Bacteria
4.8.2. Antibacterial Action
4.8.3. Determination of MICs
4.8.4. Antibiofilm Action
4.8.5. Colony-Forming Units (CFU/mL) Count
4.9. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Inderjit Wardle, D.A.; Karban, R.; Callaway, R.M. The ecosystem and evolutionary contexts of allelopathy. Trends Ecol. Evol. 2011, 26, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Selmar, D.; Abouzeid, S.; Radwan, A.; Hijazin, T.; Yahyazadeh, M.; Lewerenz, L.; Nowak, M.; Kleinwächter, M. Horizontal Natural Product Transfer: A Novel Attribution in Allelopathy. In Co-Evolution of Secondary Metabolites, 1st ed.; Mérillon, J.-M., Ramawat, K.G., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 1–11. ISBN 978-3-319-76887-8. [Google Scholar]
- Cheng, F.; Cheng, Z. Research Progress on the use of Plant Allelopathy in Agriculture and the Physiological and Ecological Mechanisms of Allelopathy. Front. Plant Sci. 2015, 6, 1020. [Google Scholar] [CrossRef]
- Bowen, G.D. The toxicity of legume seed diffusates toward rhizobia and other bacteria. Plant Soil 1961, 15, 155–165. [Google Scholar] [CrossRef]
- Dharmatilake, A.J.; Bauer, W.D. Chemotaxis of Rhizobium meliloti towards Nodulation Gene-Inducing Compounds from Alfalfa Roots. Appl. Environ. Microbiol. 1992, 58, 1153–1158. [Google Scholar] [CrossRef]
- Barbour, W.M.; Hattermann, D.R.; Stacey, G. Chemotaxis of Bradyrhizobium japonicum to soybean exudates. Appl. Environ. Microbiol. 1991, 57, 2635–2639. [Google Scholar] [CrossRef] [PubMed]
- Phillips, D.A.; Joseph, C.M.; Maxwell, C.A. Trigonelline and Stachydrine Released from Alfalfa Seeds Activate NodD2 Protein in Rhizobium meliloti. Plant Physiol. 1992, 99, 1526–1531. [Google Scholar] [CrossRef]
- Phillips, D.A. Flavonoids: Plant Signals to Soil Microbes. In Phenolic Metabolism in Plants; Imprint; Stafford, H.A., Ibrahim, R.K., Eds.; Springer: Boston, MA, USA, 1992; pp. 201–231. ISBN 978-1-4613-6517-4. [Google Scholar]
- Abdul-Rahman, A.A.; Habib, S.A. Allelopathic effect of alfalfa (Medicago sativa) on bladygrass (Imperata cylindrica). J. Chem. Ecol. 1989, 15, 2289–2300. [Google Scholar] [CrossRef]
- Ells, J.E.; McSay, A.E. Allelopathic Effects of Alfalfa Plant Residues on Emergence and Growth of Cucumber Seedlings. Hort. Sci. 1991, 26, 368–370. [Google Scholar] [CrossRef]
- Miller, D.A. Allelopathic effects of alfalfa. J. Chem. Ecol. 1983, 9, 1059–1072. [Google Scholar] [CrossRef]
- Phillips, D.A.; Wery, J.; Joseph, C.M.; Jones, A.D.; Teuber, L.R. Release of Flavonoids and Betaines from Seeds of Seven Medicago Species. Crop Sci. 1995, 35, 805–808. [Google Scholar] [CrossRef]
- Chon, S.-U.; Choi, S.-K.; Jung, S.; Jang, H.-G.; Pyo, B.-S.; Kim, S.-M. Effects of alfalfa leaf extracts and phenolic allelochemicals on early seedling growth and root morphology of alfalfa and barnyard grass. Crop Prot. 2002, 21, 1077–1082. [Google Scholar] [CrossRef]
- Dornbos, D.L.; Spencer, G.F.; Miller, R.W. Medicarpin Delays Alfalfa Seed Germination and Seedling Growth. Crop Sci. 1990, 30, 162–166. [Google Scholar] [CrossRef]
- Zhang, X.-Y.; Shi, S.-L.; Li, X.-L.; Li, C.-N.; Zhang, C.-M.; A, Y.; Kang, W.-J.; Yin, G.-L. Effects of Autotoxicity on Alfalfa (Medicago sativa): Seed Germination, Oxidative Damage and Lipid Peroxidation of Seedlings. Agronomy 2021, 11, 1027. [Google Scholar] [CrossRef]
- Nielsen, K.F.; Woods, W.B.; Cuddy, T.F. The influence of the extract of some crops and soil residues on germination and growth. Can. J. Plant Sci. 1960, 40, 188–197. [Google Scholar] [CrossRef]
- Dakora, F.D.; Joseph, C.M.; Phillips, D.A. Alfalfa (Medicago sativa L.) Root Exudates Contain Isoflavonoids in the Presence of Rhizobium meliloti. Plant Physiol. 1993, 101, 819–824. [Google Scholar] [CrossRef]
- Hall, M.H.; Henderlong, P.R. Alfalfa Autotoxic Fraction Characterization and Initial Separation. Crop Sci. 1989, 29, 425. [Google Scholar] [CrossRef]
- Peters, N.K.; Long, S.R. Alfalfa Root Exudates and Compounds which Promote or Inhibit Induction of Rhizobium meliloti Nodulation Genes. Plant Physiol. 1988, 88, 396–400. [Google Scholar] [CrossRef]
- Oleszek, W.; Jurzysta, M. The allelopathic potential of alfalfa root medicagenic acid glycosides and their fate in soil environments. Plant Soil 1987, 98, 67–80. [Google Scholar] [CrossRef]
- Leoni, F.; Hazrati, H.; Fomsgaard, I.S.; Moonen, A.-C.; Kudsk, P. Determination of the Effect of Co-cultivation on the Production and Root Exudation of Flavonoids in Four Legume Species Using LC-MS/MS Analysis. J. Agric. Food Chem. 2021, 69, 9208–9219. [Google Scholar] [CrossRef] [PubMed]
- Compton, K.K.; Hildreth, S.B.; Helm, R.F.; Scharf, B.E. An Updated Perspective on Sinorhizobium meliloti Chemotaxis to Alfalfa Flavonoids. Front. Microbiol. 2020, 11, 581482. [Google Scholar] [CrossRef]
- Emmert, E.A.; Milner, J.L.; Lee, J.C.; Pulvermacher, K.L.; Olivares, H.A.; Clardy, J.; Handelsman, J. Effect of canavanine from alfalfa seeds on the population biology of bacillus cereus. Appl. Environ. Microbiol. 1998, 64, 4683–4688. [Google Scholar] [CrossRef] [PubMed]
- Hartwig, U.A.; Maxwell, C.A.; Joseph, C.M.; Phillips, D.A. Interactions among Flavonoid nod Gene Inducers Released from Alfalfa Seeds and Roots. Plant Physiol. 1989, 91, 1138–1142. [Google Scholar] [CrossRef]
- Maxwell, C.A.; Hartwig, U.A.; Joseph, C.M.; Phillips, D.A. A Chalcone and Two Related Flavonoids Released from Alfalfa Roots Induce nod Genes of Rhizobium meliloti. Plant Physiol. 1989, 91, 842–847. [Google Scholar] [CrossRef]
- Elisha, I.L.; Botha, F.S.; McGaw, L.J.; Eloff, J.N. The antibacterial activity of extracts of nine plant species with good activity against Escherichia coli against five other bacteria and cytotoxicity of extracts. BMC Complement. Altern. Med. 2017, 17, 133. [Google Scholar] [CrossRef]
- Melotto, M.; Panchal, S.; Roy, D. Plant innate immunity against human bacterial pathogens. Front. Microbiol. 2014, 5, 411. [Google Scholar] [CrossRef] [PubMed]
- Inderjit Dakshini, K.M.M.; Einhellig, F.A. (Eds.) Allelopathy: Organisms, Processes, and Applications: Symposium Papers; American Chemical Society: Washington, DC, USA, 1995; ISBN 9780841230613. [Google Scholar]
- Macías, F.A.; Molinillo, J.M.G.; Varela, R.M.; Galindo, J.C.G. Allelopathy—A natural alternative for weed control. Pest Manag. Sci. 2007, 63, 327–348. [Google Scholar] [CrossRef]
- Parvez, M.M.; Tomita-Yokotani, K.; Fujii, Y.; Konishi, T.; Iwashina, T. Effects of quercetin and its seven derivatives on the growth of Arabidopsis thaliana and Neurospora crassa. Biochem. Syst. Ecol. 2004, 32, 631–635. [Google Scholar] [CrossRef]
- Ghimire, B.K.; Ghimire, B.; Yu, C.Y.; Chung, I.-M. Allelopathic and Autotoxic Effects of Medicago sativa-Derived Allelochemicals. Plants 2019, 8, 233. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Aparicio, M.; Masi, M.; Cimmino, A.; Vilariño, S.; Evidente, A. Allelopathic Effect of Quercetin, a Flavonoid from Fagopyrum esculentum Roots in the Radicle Growth of Phelipanche ramosa: Quercetin Natural and Semisynthetic Analogues Were Used for a Structure-Activity Relationship Investigation. Plants 2021, 10, 543. [Google Scholar] [CrossRef]
- Takahashi, L.; Sert, M.A.; Kelmer-Bracht, A.M.; Bracht, A.; Ishii-Iwamoto, E.L. Effects of rutin and quercetin on mitochondrial metabolism and on ATP levels in germinating tissues of Glycine max. Plant Physiol. Biochem. 1998, 36, 495–501. [Google Scholar] [CrossRef]
- Hussain, M.I.; Reigosa Roger, M. Plant secondary metabolite rutin affects the photosynthesis and excitation energy flux responses in Arabidopsis thaliana. Allelopath. J. 2016, 38, 215–228. [Google Scholar]
- Hartwig, U.A.; Maxwell, C.A.; Joseph, C.M.; Phillips, D.A. Chrysoeriol and Luteolin Released from Alfalfa Seeds Induce nod Genes in Rhizobium meliloti. Plant Physiol. 1990, 92, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Sun, F.; Feng, W.; Qiu, X.; Liu, Y.; Yang, B.; Chen, Y.; Xia, P. Hyperoside inhibits biofilm formation of Pseudomonas aeruginosa. Exp. Ther. Med. 2017, 14, 1647–1652. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, N.; Hiradate, S.; Fujii, Y. Plant growth inhibitory activity of L-canavanine and its mode of action. J. Chem. Ecol. 2001, 27, 19–31. [Google Scholar] [CrossRef] [PubMed]
- Krasuska, U.; Andrzejczak, O.; Staszek, P.; Borucki, W.; Gniazdowska, A. Toxicity of canavanine in tomato (Solanum lycopersicum L.) roots is due to alterations in RNS, ROS and auxin levels. Plant Physiol. Biochem. 2016, 103, 84–95. [Google Scholar] [CrossRef]
- Sasamoto, H.; Mardani, H.; Sasamoto, Y.; Wasano, N.; Murashige-Baba, T.; Sato, T.; Hasegawa, A.; Fujii, Y. Evaluation of canavanine as an allelochemical in etiolated seedlings of Vicia villosa Roth: Protoplast co-culture method with digital image analysis. Vitr. Cell. Dev. Biol. Plant 2019, 55, 296–304. [Google Scholar] [CrossRef]
- Bence, A.K.; Crooks, P.A. The mechanism of L-canavanine cytotoxicity: Arginyl tRNA synthetase as a novel target for anticancer drug discovery. J. Enzyme Inhib. Med. Chem. 2003, 18, 383–394. [Google Scholar] [CrossRef]
- Mitri, C.; Soustelle, L.; Framery, B.; Bockaert, J.; Parmentier, M.-L.; Grau, Y. Plant insecticide L-canavanine repels Drosophila via the insect orphan GPCR DmX. PLoS Biol. 2009, 7, e1000147. [Google Scholar] [CrossRef]
- Scavo, A.; Abbate, C.; Mauromicale, G. Plant allelochemicals: Agronomic, nutritional and ecological relevance in the soil system. Plant Soil 2019, 442, 23–48. [Google Scholar] [CrossRef]
- Nelson, E.B. The seed microbiome: Origins, interactions, and impacts. Plant Soil 2018, 422, 7–34. [Google Scholar] [CrossRef]
- Keshavan, N.D.; Chowdhary, P.K.; Haines, D.C.; González, J.E. L-Canavanine made by Medicago sativa interferes with quorum sensing in Sinorhizobium meliloti. J. Bacteriol. 2005, 187, 8427–8436. [Google Scholar] [CrossRef] [PubMed]
- Mardani-Korrani, H.; Nakayasu, M.; Yamazaki, S.; Aoki, Y.; Kaida, R.; Motobayashi, T.; Kobayashi, M.; Ohkama-Ohtsu, N.; Oikawa, Y.; Sugiyama, A.; et al. L-Canavanine, a Root Exudate From Hairy Vetch (Vicia villosa) Drastically Affecting the Soil Microbial Community and Metabolite Pathways. Front. Microbiol. 2021, 12, 701796. [Google Scholar] [CrossRef] [PubMed]
- Barron, J.A.; Weaks, T.E. The effects of the amino acid canavanine on Pythium. Physiol. Plant Pathol. 1977, 11, 305–311. [Google Scholar] [CrossRef]
- Rosenthal, G.A.; Lambert, J.; Hoffmann, D. Canavanine incorporation into the antibacterial proteins of the fly, Phormia terranovae (Diptera), and its effect on biological activity. J. Biol. Chem. 1989, 264, 9768–9771. [Google Scholar] [CrossRef]
- Rosenthal, G.A.; Hughes, C.G.; Janzen, D.H. L-Canavanine, a Dietary Nitrogen Source for the Seed Predator Caryedes brasiliensis (Bruchidae). Science 1982, 217, 353–355. [Google Scholar] [CrossRef]
- Berg, J.M.; Tymoczko, J.L.; Stryer, L. Biochemistry, 5th ed.; Freeman, W.H., Ed.; NCBI: New York, NY, USA; Bethesda: Rockville, MD, USA, 2002; ISBN 9780716730514.
- Selmar, D.; Radwan, A.; Hijazin, T.; Abouzeid, S.; Yahyazadeh, M.; Lewerenz, L.; Kleinwächter, M.; Nowak, M. Horizontal Natural Product Transfer: Intriguing Insights into a Newly Discovered Phenomenon. J. Agric. Food Chem. 2019, 67, 8740–8745. [Google Scholar] [CrossRef]
- Pucker, B.; Selmar, D. Biochemistry and Molecular Basis of Intracellular Flavonoid Transport in Plants. Plants 2022, 11, 963. [Google Scholar] [CrossRef]
- Tegeder, M.; Rentsch, D. Uptake and partitioning of amino acids and peptides. Mol. Plant 2010, 3, 997–1011. [Google Scholar] [CrossRef]
- Hirner, B.; Fischer, W.N.; Rentsch, D.; Kwart, M.; Frommer, W.B. Developmental control of H+/amino acid permease gene expression during seed development of Arabidopsis. Plant J. 1998, 14, 535–544. [Google Scholar] [CrossRef]
- Lee, Y.-H.; Tegeder, M. Selective expression of a novel high-affinity transport system for acidic and neutral amino acids in the tapetum cells of Arabidopsis flowers. Plant J. 2004, 40, 60–74. [Google Scholar] [CrossRef]
- Svennerstam, H.; Ganeteg, U.; Bellini, C.; Näsholm, T. Comprehensive screening of Arabidopsis mutants suggests the lysine histidine transporter 1 to be involved in plant uptake of amino acids. Plant Physiol. 2007, 143, 1853–1860. [Google Scholar] [CrossRef] [PubMed]
- Paungfoo-Lonhienne, C.; Lonhienne, T.G.A.; Rentsch, D.; Robinson, N.; Christie, M.; Webb, R.I.; Gamage, H.K.; Carroll, B.J.; Schenk, P.M.; Schmidt, S. Plants can use protein as a nitrogen source without assistance from other organisms. Proc. Natl. Acad. Sci. USA 2008, 105, 4524–4529. [Google Scholar] [CrossRef]
- Komarova, N.Y.; Thor, K.; Gubler, A.; Meier, S.; Dietrich, D.; Weichert, A.; Suter Grotemeyer, M.; Tegeder, M.; Rentsch, D. AtPTR1 and AtPTR5 transport dipeptides in planta. Plant Physiol. 2008, 148, 856–869. [Google Scholar] [CrossRef] [PubMed]
- Cronin, M.T.D.; Livingstone, D.J. Predicting Chemical Toxicity and Fate; Taylor & Francis: Hoboken, NJ, USA, 2006; ISBN 9780203642627. [Google Scholar]
- Trapp, S.; Legind, C. Uptake of Organic Contaminants from Soil into Vegetables and Fruits; Springer: Berlin/Heidelberg, Germany, 2011; pp. 369–408. ISBN 978-90-481-9756-9. [Google Scholar]
- Rothwell, J.A.; Day, A.J.; Morgan, M.R.A. Experimental Determination of Octanol−Water Partition Coefficients of Quercetin and Related Flavonoids. J. Agric. Food Chem. 2005, 53, 4355–4360. [Google Scholar] [CrossRef] [PubMed]
- Behrens, C.E.; Smith, K.E.; Iancu, C.V.; Choe, J.-Y.; Dean, J.V. Transport of Anthocyanins and other Flavonoids by the Arabidopsis ATP-Binding Cassette Transporter AtABCC2. Sci. Rep. 2019, 9, 437. [Google Scholar] [CrossRef]
- Frangne, N.; Eggmann, T.; Koblischke, C.; Weissenböck, G.; Martinoia, E.; Klein, M. Flavone Glucoside Uptake into Barley Mesophyll and Arabidopsis Cell Culture Vacuoles. Energization Occurs by H+-Antiport and ATP-Binding Cassette-Type Mechanisms. Plant Physiol. 2002, 128, 726–733. [Google Scholar] [CrossRef] [PubMed]
- Klein, M.; Martinoia, E.; Hoffmann-Thoma, G.; Weissenböck, G. The ABC-like vacuolar transporter for rye mesophyll flavone glucuronides is not species-specific. Phytochemistry 2001, 56, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Hazrati, H.; Fomsgaard, I.S.; Kudsk, P. Root-Exuded Benzoxazinoids: Uptake and Translocation in Neighboring Plants. J. Agric. Food Chem. 2020, 68, 10609–10617. [Google Scholar] [CrossRef]
- Schramm, S.; Köhler, N.; Rozhon, W. Pyrrolizidine Alkaloids: Biosynthesis, Biological Activities and Occurrence in Crop Plants. Molecules 2019, 24, 498. [Google Scholar] [CrossRef]
- Nowak, M.; Wittke, C.; Lederer, I.; Klier, B.; Kleinwächter, M.; Selmar, D. Interspecific transfer of pyrrolizidine alkaloids: An unconsidered source of contaminations of phytopharmaceuticals and plant derived commodities. Food Chem. 2016, 213, 163–168. [Google Scholar] [CrossRef]
- Xuan, T.D.; Tsuzuki, E.; Uematsu, H.; Terao, H. Weed control with alfalfa pellets in transplanting rice. Weed Biol. Manag. 2001, 1, 231–235. [Google Scholar] [CrossRef]
- Xuan, T.D.; Shinkichi, T.; Khanh, T.D.; Chung, I.M. Biological control of weeds and plant pathogens in paddy rice by exploiting plant allelopathy: An overview. Crop Protection 2005, 24, 197–206. [Google Scholar] [CrossRef]
- Puig, C.G.; Gonçalves, R.F.; Valentão, P.; Andrade, P.B.; Reigosa, M.J.; Pedrol, N. The Consistency Between Phytotoxic Effects and the Dynamics of Allelochemicals Release from Eucalyptus globulus Leaves Used as Bioherbicide Green Manure. J. Chem. Ecol. 2018, 44, 658–670. [Google Scholar] [CrossRef]
- Scavo, A.; Mauromicale, G. Crop Allelopathy for Sustainable Weed Management in Agroecosystems: Knowing the Present with a View to the Future. Agronomy 2021, 11, 2104. [Google Scholar] [CrossRef]
- Zubair, H.M.; Pratley, J.E.; Sandral, G.A.; Humphries, A. Allelopathic interference of alfalfa (Medicago sativa L.) genotypes to annual ryegrass (Lolium rigidum). Available online: https://link.springer.com/article/10.1007/s10265-017-0921-9 (accessed on 23 September 2022).
- Amossé, C.; Jeuffroy, M.-H.; Celette, F.; David, C. Relay-intercropped forage legumes help to control weeds in organic grain production. Eur. J. Agron. 2013, 49, 158–167. [Google Scholar] [CrossRef]
- Teasdale, J.R. Contribution of Cover Crops to Weed Management in Sustainable Agricultural Systems. J. Prod. Agric. 1996, 9, 475–479. [Google Scholar] [CrossRef]
- El-Darier, S.M.; Zein El-Dien, M.H. Biological activity of Medicago sativa L. (alfalfa) residues on germination efficiency, growth and nutrient uptake of Lycopersicon esculentum L. (tomato) seedlings. J. Taibah Univ. Sci. 2011, 5, 7–13. [Google Scholar] [CrossRef]
- Weisberger, D.; Nichols, V.; Liebman, M. Does diversifying crop rotations suppress weeds? A meta-analysis. PLoS ONE 2019, 14, e0219847. [Google Scholar] [CrossRef]
- Wang, Q.; Wei, H.-C.; Zhou, S.-J.; Li, Y.; Zheng, T.-T.; Zhou, C.-Z.; Wan, X.-H. Hyperoside: A review on its sources, biological activities, and molecular mechanisms. Phytother. Res. 2022, 36, 2779–2802. [Google Scholar] [CrossRef]
- Roy, R.; Tiwari, M.; Donelli, G.; Tiwari, V. Strategies for combating bacterial biofilms: A focus on anti-biofilm agents and their mechanisms of action. Virulence 2018, 9, 522–554. [Google Scholar] [CrossRef]
- Mastoor, S.; Nazim, F.; Rizwan-Ul-Hasan, S.; Ahmed, K.; Khan, S.; Ali, S.N.; Abidi, S.H. Analysis of the Antimicrobial and Anti-Biofilm Activity of Natural Compounds and Their Analogues against Staphylococcus aureus Isolates. Molecules 2022, 27, 6874. [Google Scholar] [CrossRef] [PubMed]
- El-Aasr, M.; Nohara, T.; Ikeda, T.; Abu-Risha, S.E.; Elekhnawy, E.; Tawfik, H.O.; Shoeib, N.; Attia, G. LC-MS/MS metabolomics profiling of Glechoma hederacea L. methanolic extract; in vitro antimicrobial and in vivo with in silico wound healing studies on Staphylococcus aureus infected rat skin wound. Nat. Prod. Res. 2022, 9, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Latka, A.; Drulis-Kawa, Z. Advantages and limitations of microtiter biofilm assays in the model of antibiofilm activity of Klebsiella phage KP34 and its depolymerase. Sci. Rep. 2020, 10, 20338. [Google Scholar] [CrossRef]
- Attallah, N.G.M.; Al-Fakhrany, O.M.; Elekhnawy, E.; Hussein, I.A.; Shaldam, M.A.; Altwaijry, N.; Alqahtani, M.J.; Negm, W.A. Anti-Biofilm and Antibacterial Activities of Cycas media R. Br Secondary Metabolites: In Silico, In Vitro, and In Vivo Approaches. Antibiotics 2022, 11, 993. [Google Scholar] [CrossRef]
- Abdel Bar, F.M.; Alossaimi, M.A.; Elekhnawy, E.; Alzeer, M.A.A.; Abo Kamer, A.; Moglad, E.; ElNaggar, M.H. Anti-Quorum Sensing and Anti-Biofilm Activity of Pelargonium × hortorum Root Extract against Pseudomonas aeruginosa: Combinatorial Effect of Catechin and Gallic Acid. Molecules 2022, 27, 7841. [Google Scholar] [CrossRef] [PubMed]
- Elekhnawy, E.; Negm, W.A.; El-Aasr, M.; Kamer, A.A.; Alqarni, M.; Batiha, G.E.-S.; Obaidullah, A.J.; Fawzy, H.M. Histological assessment, anti-quorum sensing, and anti-biofilm activities of Dioon spinulosum extract: In vitro and in vivo approach. Sci. Rep. 2022, 12, 180. [Google Scholar] [CrossRef]



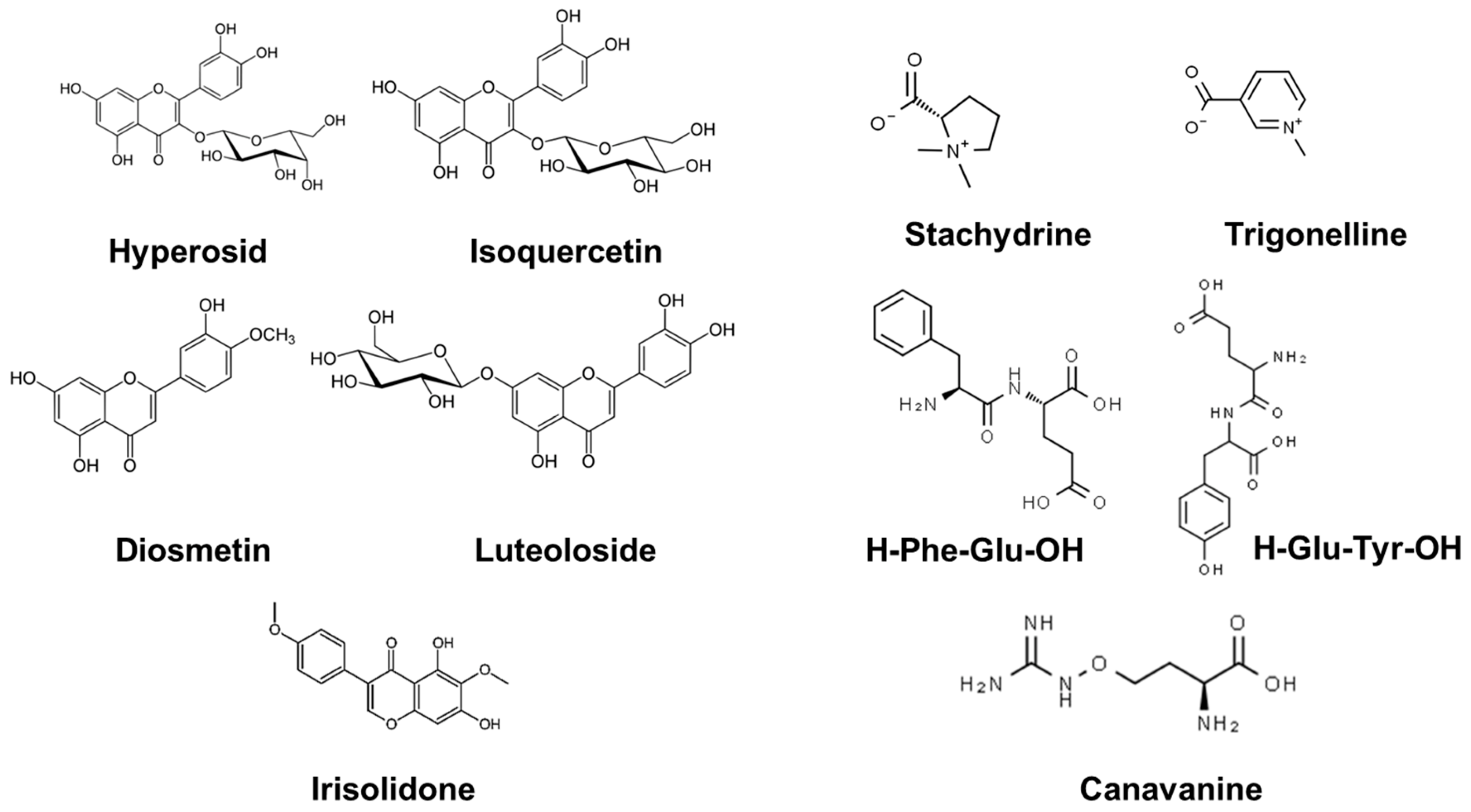


| # | Name | Rt. (min) | CCS | m/z Meas, [M + H]+ | Molecular Formula | MS/MS Fragments |
|---|---|---|---|---|---|---|
| ||||||
| 1 | Canavanine | 0.29 | 134.6 | 177.09781 | C5H12N4O3 | 76, 72, 61, 56, 44 |
| 2 | Trigonelline | 0.41 | 122.3 | 138.05495 | C7H7NO2 | 138, 92, 78, 65, 51, 39 |
| 3 | Stachydrine | 0.5 | 126.6 | 144.101905 | C7H13NO2 | 144, 102, 84, 72, 58, 44 |
| 4 | H-Glu-Tyr-OH | 2.1 | 171.9 | 311.12306 | C14H18N2O6 | 311, 248, 202, 182, 165, 147, 136, 123, 103, 91, 84 |
| 5 | H-Phe-Glu-OH | 5.6 | 169.4 | 295.1285 | C14H18N2O5 | 295, 233, 186, 166, 149, 120, 103, 84 |
| ||||||
| 6 | Quercetin 3-galactoside (hyperoside) | 17.26 | 205 | 465.1022 | C21H20O12 | 465, 303, 257, 229, 153, 85 |
| 7 | Quercetin 3-O-glucoside (isoquercetin) | 17.58 | 206.9 | 465.10222 | C21H20O12 | 465, 303, 257, 229, 153, 85 |
| 8 | Quercetin 3-O-xyloside | 18 | 182 | 435.05743 | C20H18O11 | 435, 303, 257, 229,153 |
| ||||||
| 9 | Luteoloside | 18.0 | 211.5 | 449.2856 | C21H20O11 | 449, 287, 258, 153 |
| 10 | Diosmetin isomer | 18.8 | 167.9 | 301.07006 | C16H12O6 | 301, 286, 269, 258, 229, 213, 184, 153, 124, 96 |
| ||||||
| 11 | Irisolidone isomer | 21.5 | 172.5 | 315.08569 | C17H14O6 | 315, 300, 272, 257, 229, 167, 148, 133 |
| Compound | Concentration (µg/mL) |
|---|---|
| Canavanine | 160.9 |
| Trigonelline | 54.1 |
| Stachydrine | 70.9 |
| Hyperoside | 1134 |
| Isoquercetin | 29.9 |
| Luteoloside | 1.26 |
| Diosmetin | 11.4 |
| H-Glu-Tyr-OH | 275 |
| H-Phe-Glu-OH | 14.9 |
| Isolate Code | MIC Values (µg/mL) | ||||||
|---|---|---|---|---|---|---|---|
| Total Exudate | Canavanine | Luteoloside | Hyperoside | Stachydrine | Diosmetin | Trigonelline | |
| E1 | 512 | 256 | 256 | 8 | 1024 | 1024 | 128 |
| E2 | 512 | 128 | 256 | 8 | 1024 | 2048 | 128 |
| E3 | 256 | 256 | 256 | 16 | 1024 | 1024 | 128 |
| E4 | 256 | 128 | 256 | 8 | 2048 | 1024 | 256 |
| E5 | 512 | 32 | 512 | 32 | 1024 | 1024 | 256 |
| E6 | 512 | 64 | 512 | 16 | 1024 | 1024 | 128 |
| E7 | 512 | 64 | 512 | 32 | 1024 | 2048 | 256 |
| E8 | 512 | 256 | 256 | 16 | 2048 | 1024 | 128 |
| E9 | 256 | 256 | 256 | 8 | 2048 | 1024 | 128 |
| E10 | 256 | 32 | 256 | 16 | 1024 | 2048 | 128 |
| E11 | 256 | 64 | 512 | 32 | 1024 | 1024 | 128 |
| E12 | 512 | 64 | 256 | 16 | 1024 | 1024 | 256 |
| E13 | 512 | 32 | 512 | 32 | 2048 | 1024 | 256 |
| E14 | 256 | 64 | 512 | 8 | 2048 | 2048 | 128 |
| E15 | 512 | 128 | 512 | 16 | 1024 | 1024 | 128 |
| E16 | 256 | 128 | 512 | 32 | 1024 | 2048 | 256 |
| E17 | 256 | 256 | 256 | 32 | 1024 | 1024 | 256 |
| E18 | 256 | 128 | 256 | 32 | 1024 | 1024 | 128 |
| E19 | 256 | 64 | 256 | 16 | 1024 | 1024 | 256 |
| Biofilm Formation Ability | Count of E. coli Isolates before Treatment | Count of E. coli Isolates after Treatment with | ||||||
|---|---|---|---|---|---|---|---|---|
| Total Exudate | Canavanine | Luteoloside | Hyperoside | Stachydrine | Diosmetin | Trigonelline | ||
| None forming | 2 | 3 | 4 | 3 | 4 | 2 | 2 | 2 |
| Weak | 4 | 6 | 9 | 4 | 11 | 5 | 6 | 5 |
| Moderate | 7 | 6 | 3 | 6 | 3 | 7 | 6 | 6 |
| Strong | 6 | 4 | 3 | 6 | 1 | 5 | 5 | 6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abouzeid, S.; Beutling, U.; Elekhnawy, E.; Selmar, D. Antibacterial and Antibiofilm Effects of Allelopathic Compounds Identified in Medicago sativa L. Seedling Exudate against Escherichia coli. Molecules 2023, 28, 2645. https://doi.org/10.3390/molecules28062645
Abouzeid S, Beutling U, Elekhnawy E, Selmar D. Antibacterial and Antibiofilm Effects of Allelopathic Compounds Identified in Medicago sativa L. Seedling Exudate against Escherichia coli. Molecules. 2023; 28(6):2645. https://doi.org/10.3390/molecules28062645
Chicago/Turabian StyleAbouzeid, Sara, Ulrike Beutling, Engy Elekhnawy, and Dirk Selmar. 2023. "Antibacterial and Antibiofilm Effects of Allelopathic Compounds Identified in Medicago sativa L. Seedling Exudate against Escherichia coli" Molecules 28, no. 6: 2645. https://doi.org/10.3390/molecules28062645
APA StyleAbouzeid, S., Beutling, U., Elekhnawy, E., & Selmar, D. (2023). Antibacterial and Antibiofilm Effects of Allelopathic Compounds Identified in Medicago sativa L. Seedling Exudate against Escherichia coli. Molecules, 28(6), 2645. https://doi.org/10.3390/molecules28062645






