Optimization, Probiotic Characteristics, and Rheological Properties of Exopolysaccharides from Lactiplantibacillus plantarum MC5
Abstract
1. Introduction
2. Results and Discussion
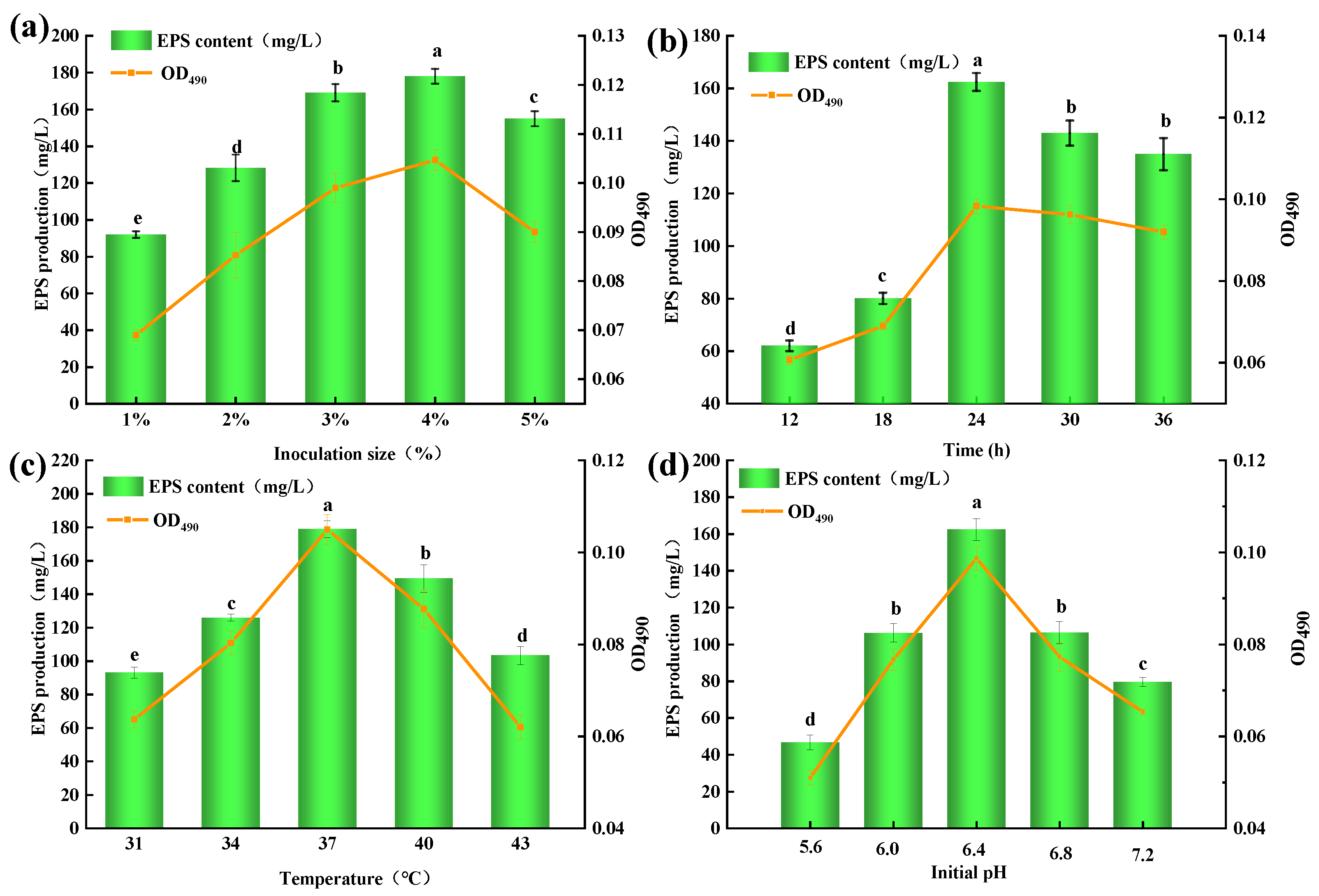
2.1. Identification of Strain MC5 and Culture Conditions Single Factor Test
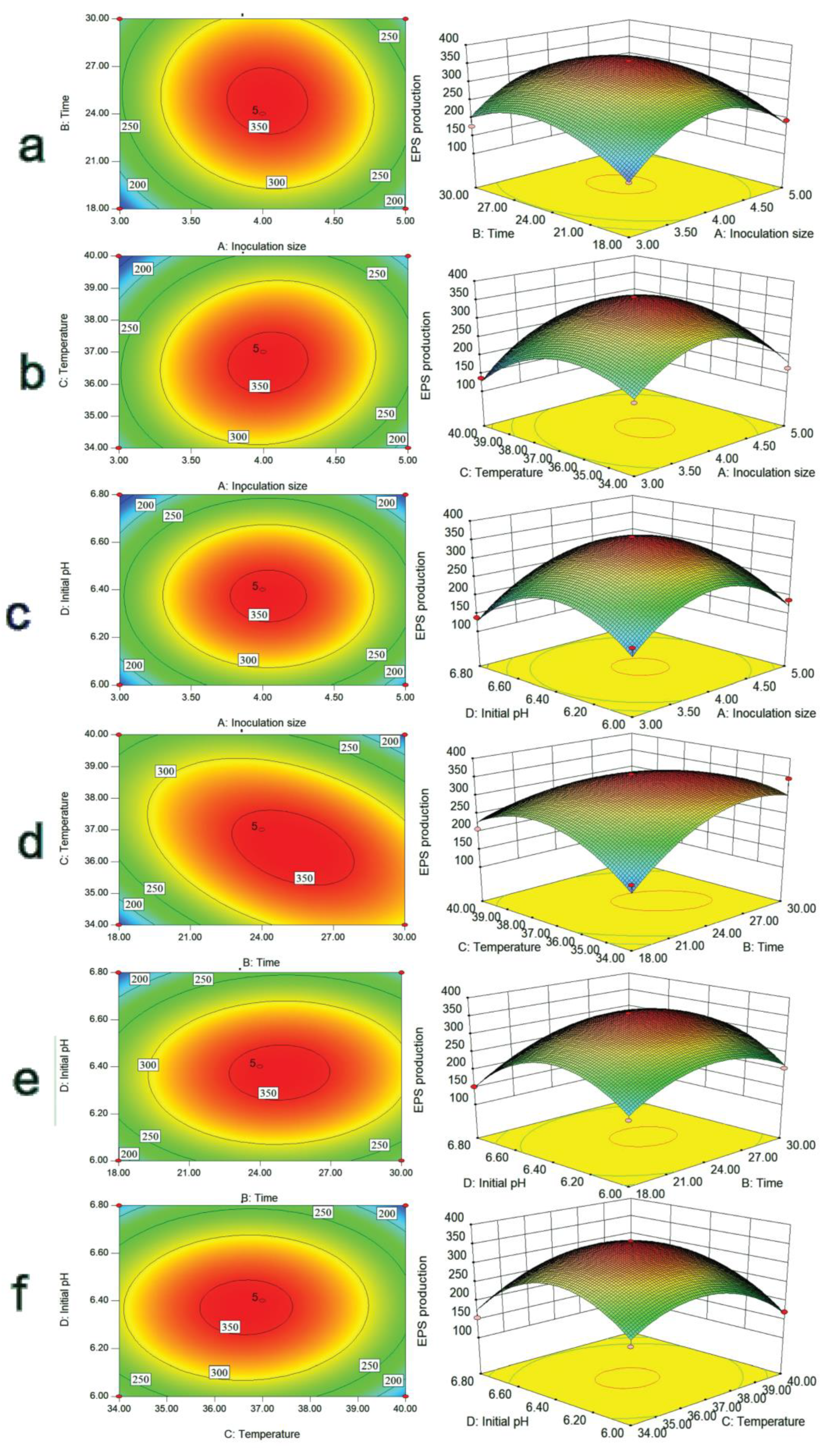
2.2. Optimization of Fermentation Conditions for EPS-MC5
2.3. Three-Dimensional Response Surfaces and Count Plots of Variables
2.4. Verification Test of EPS-MC5 Yield
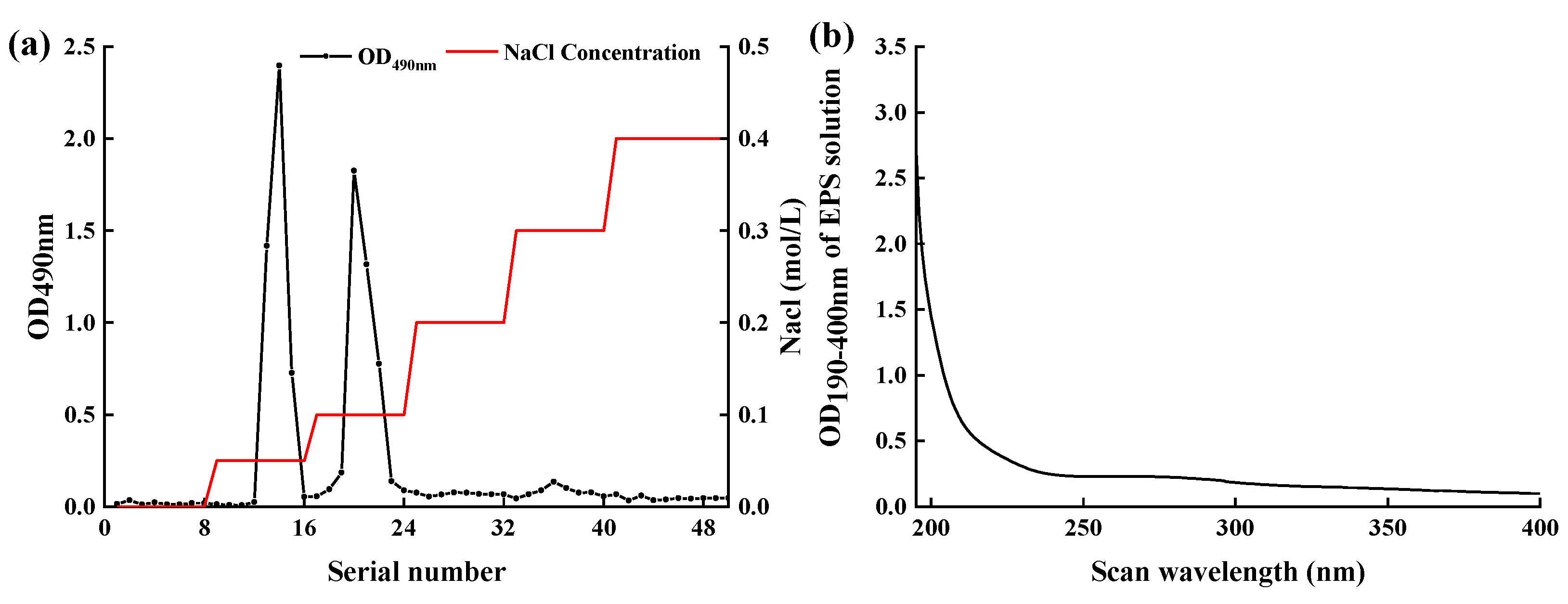
2.5. Isolation and Purification of EPS-MC5
2.6. In Vitro Resisting-Digestion Capacity of EPS-MC5 to α-Amylase and Simulated Gastrointestinal Juices
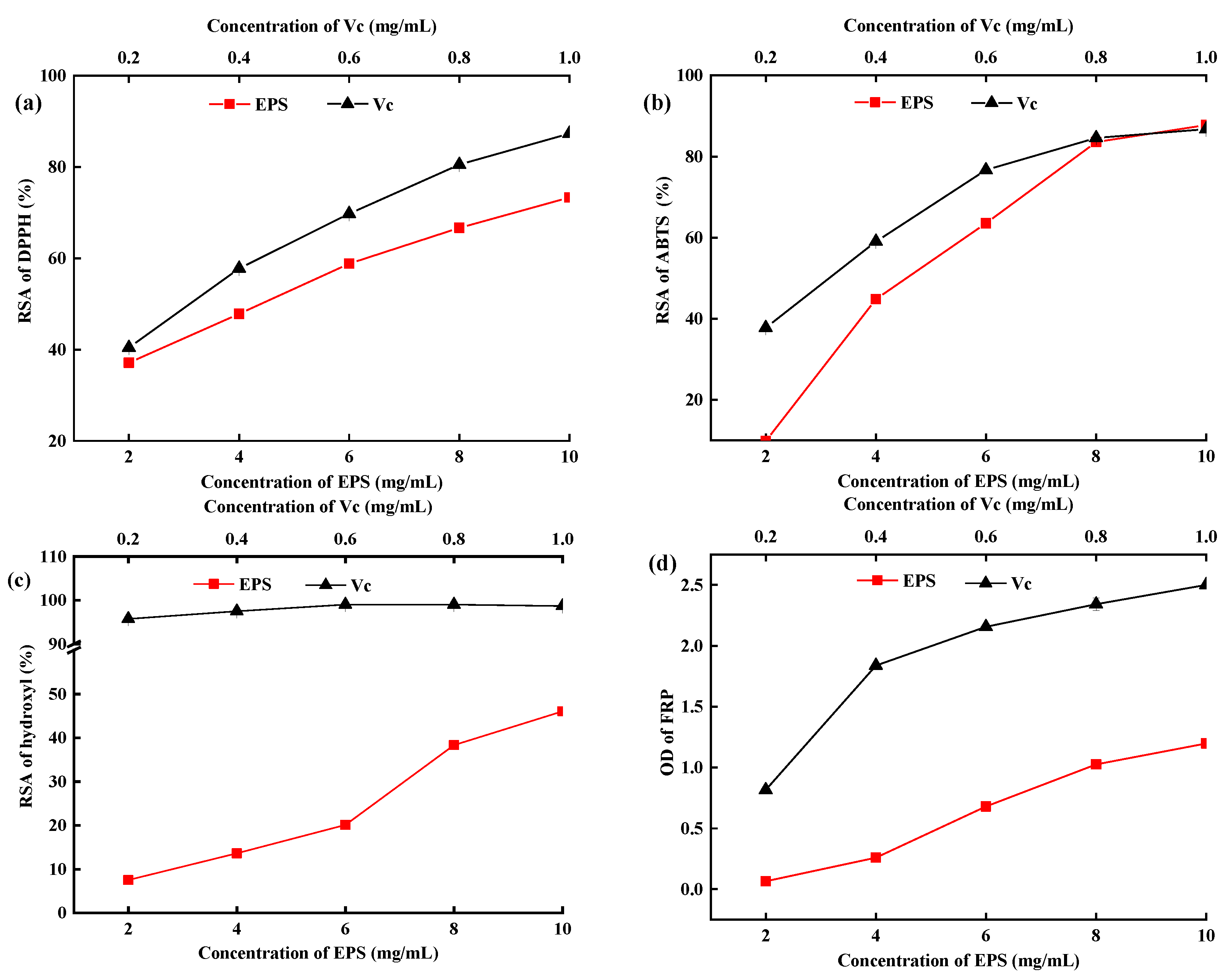
2.7. In Vitro Antioxidant Activity of EPS-MC5
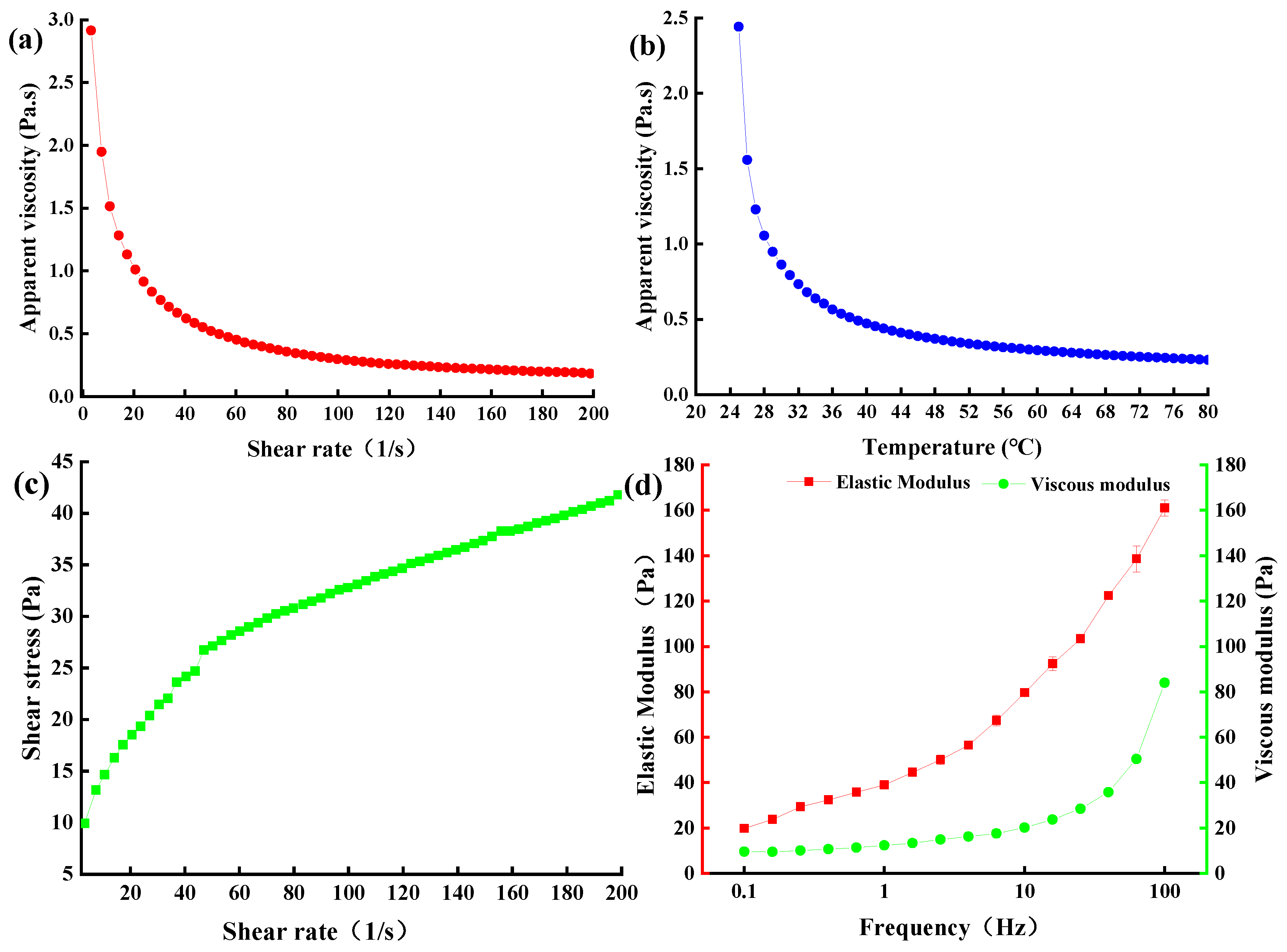
2.8. Rheological Properties of EPS-MC5
2.8.1. Apparent Viscosity of EPS-MC5
2.8.2. Viscoelastic Properties of EPS-MC5
3. Materials and Methods
3.1. Materials
3.2. Isolation and Determination of EPS-MC5
3.3. Purification of EPS-MC5
3.4. Single Factor Experiment of Culture Conditions for EPS Production from Lp. plantarum MC5
3.4.1. Effects of Inoculation Size on EPS Production from Lp. plantarum MC5
3.4.2. Effects of Culture Time on EPS Production from Lp. plantarum MC5
3.4.3. Effects of Culture Temperature on EPS Production from Lp. plantarum MC5
3.4.4. Effect of Initial pH Value on EPS Production from Lp. plantarum MC5
3.5. Optimization of Lp. plantarum MC5 EPS Culture Conditions by Response Surface
3.6. In Vitro Resisting-Digestion Capacity of EPS-MC5
3.6.1. The Resisting-Digestion Capacity of EPS-MC5 to α-Amylase (RCA)
3.6.2. The Resisting-Digestion Capacity of EPS-MC5 to Simulated Gastric Juice
3.6.3. The Resisting-Digestion Capacity of EPS-MC5 to Simulated Intestinal Juice
3.7. In Vitro Antioxidant Activity Analysis of EPS-MC5
3.7.1. The Radical Scavenging Rate (RSR) of DPPH
3.7.2. The Radical Scavenging Rate (RSR) of ABTS
3.7.3. The Radical Scavenging Rate (RSR) of Hydroxyl
3.7.4. The Ferric-Iron Reducing Power (IRP) of EPS-MC5
3.8. Analysis of Rheological Properties of EPS-MC5
3.8.1. The Preparation of the EPS-MC5 Samples
3.8.2. Apparent Viscosity and Flow Curves of EPS-MC5
3.8.3. Amplitude and Frequency Sweep Tests of EPS-MC5
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| EPS | Exopolysaccharide |
| EPS-MC5 | Exopolysaccharide production from Lp. plantarum MC5 |
| LAB | Lactic acid bacteria |
| Lp. plantarum MC5 | Lactiplantibacillus plantarum MC5 |
| RSA | Radical scavenging activity |
| DPPH• | 1,1-Diphenyl-2-picrylhydrazyl, (free radical) |
| ABTS• | 2,2’-Azinobis (3-ethylbenzothiazoline-6-sulfonic acid ammonium salt) |
| •OH | Hydroxyl (free radicals) |
| IRP | Ferric-iron reducing power |
References
- Faghfoori, Z.; Gargari, B.P.; Gharamaleki, A.S.; Bagherpour, H.; Khosroushahi, A.Y. Cellular and molecular mechanisms of probiotics effects on colorectal cancer. J. Funct. Foods 2015, 18, 463–472. [Google Scholar] [CrossRef]
- Imran, M.Y.M.; Reehana, N.; Jayaraj, K.A.; Ahamed, A.A.P.; Dhanasekaran, D.; Thajuddin, N.; Alharbi, N.S.; Muralitharan, G. Statistical optimization of exopolysaccharide production by Lactobacillus plantarum NTMI05 and NTMI20. Int. J. Biol. Macromol. 2016, 93, 731–745. [Google Scholar] [CrossRef]
- Kajala, I.; Shi, Q.; Nyyssölä, A.; Maina, N.H.; Hou, Y.; Katina, K.; Tenkanen, M.; Juvonen, R. Cloning and characterization of a Weissella confusa dextransucrase and its application in high fibre baking. PLoS ONE 2015, 10, e116418–e116437. [Google Scholar] [CrossRef]
- Dilna, S.V.; Surya, H.; Aswathy, R.G.; Varsha, K.K.; Sakthikumar, D.N.; Pandey, A.; Nampoothiri, K.M. Characterization of an exopolysaccharide with potential health-benefit properties from a probiotic Lactobacillus plantarum RJF4. LWT-Food Sci. Technol. 2015, 64, 1179–1186. [Google Scholar] [CrossRef]
- Sadishkumar, V.; Jeevaratnam, K. In vitro probiotic evaluation of potential antioxidant lactic acid bacteria isolated fromidli batter fermented with Piper betle leaves. Int. J. Food Sci. Technol. 2017, 52, 329–340. [Google Scholar] [CrossRef]
- Mayer, M.J.; D’Amato, A.; Colquhoun, I.J.; Le Gall, G.; Narbad, A. Identification of genes required for glucan exopolysaccharide production in Lactobacillus johnsonii suggests a novel biosynthesis mechanism. Appl. Environ. Microbiol. 2020, 86, e2808–e2819. [Google Scholar] [CrossRef]
- Loeffler, M.; Hilbig, J.; Velasco, L.; Weiss, J. Usage of in situ exopolysaccharide-forming lactic acid bacteria in food production: Meat products—A new field of application? Compr. Rev. Food Sci. Food Saf. 2020, 19, 2932–2954. [Google Scholar] [CrossRef]
- Macedo, M.; Lacroix, C.; Gardner, N.; Champagne, C. Effect of medium supplementation on exopolysaccharide production by Lactobacillus rhamnosus RW-9595M in whey permeate. Int. Dairy J. 2002, 12, 419–426. [Google Scholar] [CrossRef]
- Aslım, B.; Yüksekdag, Z.N.; Beyatli, Y.; Mercan, N.; Aslim, B. Exopolysaccharide production by Lactobacillus delbruckii subsp. bulgaricus and Streptococcus thermophilus strains under different growth conditions. World J. Microbiol. Biotechnol. 2005, 21, 673–677. [Google Scholar] [CrossRef]
- Bryukhanov, A.L.; Klimko, A.I.; Netrusov, A.I. Antioxidant properties of lactic acid bacteria. Microbiology 2022, 91, 463–478. [Google Scholar] [CrossRef]
- Seishima, R.; Wada, T.; Tsuchihashi, K.; Okazaki, S.; Yoshikawa, M.; Oshima, H.; Oshima, M.; Sato, T.; Hasegawa, H.; Kitagawa, Y.; et al. Ink4a/Arf-dependent loss of parietal cells induced by oxidative stress promotes CD44-dependent gastric tumorigenesis. Cancer Prev. Res. 2015, 8, 492–501. [Google Scholar] [CrossRef]
- Matthew, R.B.; Andrew, B.; Rajesh, K. Reactive oxygen species-mediated diabetic heart disease: Mechanisms and therapies. Antioxid. Redox Signal. 2022, 36, 608–630. [Google Scholar]
- Zuo, L.; Rose, B.A.; Roberts, W.J.; He, F.; Banes-Berceli, A.K. Molecular characterization of reactive oxygen species in systemic and pulmonary hypertension. Am. J. Hypertens. 2014, 27, 643–650. [Google Scholar] [CrossRef]
- Pessione, E.; Cirrincione, S. Bioactive molecules released in food by lactic acid bacteria: Encrypted peptides and biogenic amines. Front. Microbiol. 2016, 7, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Tallon, R.; Bressollier, P.; Urdaci, M.C. Isolation and characterization of two exopolysaccharides produced by Lactobacillus plantarum EP56. Res. Microbiol. 2003, 154, 705–712. [Google Scholar] [CrossRef]
- Vereecke, D.; Fichtner, E.J.; Lambert, P.Q.; Cooke, P.; Kilcrease, J.; Stamler, R.A.; Zhang, Y.; Francis, I.M.; Randall, J.J. Colonization and survival capacities underlying the multifaceted life of Rhodococcus sp. PBTS1 and PBTS2. Plant Pathol. 2021, 70, 567–583. [Google Scholar] [CrossRef]
- Degeest, B.; Vaningelgem, F.; De Vuyst, L. Microbial physiology, fermentation kinetics, and process engineering of heteropolysaccharide production by lactic acid bacteria. Int. Dairy J. 2001, 11, 747–757. [Google Scholar] [CrossRef]
- Adesulu-Dahunsi, A.T.; Sanni, A.I.; Jeyaram, K. Production, characterization and in vitro antioxidant activities of exopolysaccharide from Weissella cibaria GA44. LWT-Food Sci. Technol. 2018, 87, 432–442. [Google Scholar] [CrossRef]
- Zhao, X.F.; Liang, Q. EPS-Producing Lactobacillus plantarum MC5 as a compound starter improves rheology, texture, and antioxidant activity of yogurt during storage. Foods 2022, 11, 1660. [Google Scholar] [CrossRef] [PubMed]
- Xia, M.; Zhang, S.; Shen, L.; Yu, R.; Liu, Y.; Li, J.; Wu, X.; Chen, M.; Qiu, G.; Zeng, W. Optimization and characterization of an antioxidant exopolysaccharide produced by cupriavidus pauculus 1490. J. Polym. Environ. 2022, 30, 2077–2086. [Google Scholar] [CrossRef]
- Chaisuwan, W.; Jantanasakulwong, K.; Wangtueai, S. Microbial exopolysaccharides for immune enhancement fermentation, modifications and bioactivities. Food Biosci. 2020, 35, 100564–100601. [Google Scholar] [CrossRef]
- Jiang, G.; He, J.; Gan, L.; Li, X.; Tian, Y. Optimization of exopolysaccharides production by lactiplantibacillus pentosus B8 isolated from sichuan PAOCAI and its functional properties. Appl. Biochem. Microbiol. 2022, 58, 195–205. [Google Scholar] [CrossRef]
- Li, Y.; Liu, Y.; Cao, C.; Zhu, X.; Wang, C.; Wu, R.; Wu, J. Extraction and biological activity of exopolysaccharide produced by Leuconostoc Mesenteroides SN-8. Int. J. Biol. Macromol. 2020, 157, 36–44. [Google Scholar] [CrossRef]
- Wang, X.; Shao, C.; Liu, L.; Guo, X.; Xu, Y.; Lü, X. Optimization, partial characterization and antioxidant activity of an exopolysaccharide from Lactobacillus plantarum KX041. Int. J. Biol. Macromol. 2017, 103, 1173–1184. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Liu, C.; Li, D.; Zhao, Y.; Zhang, X.; Zeng, X.; Yang, Z.; Li, S. Antioxidant activity of an exopolysaccharide isolated from Lactobacillus plantarum C88. Int. J. Biol. Macromol. 2013, 54, 270–275. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, X.; Tian, Z.; Yang, Y.; Yang, Z. Characterization of an exopolysaccharide produced by Lactobacillus plantarum YW11 isolated from Tibet Kefir. Carbohydr. Polym. 2015, 125, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Rajoka, M.S.R.; Jin, M.; Haobin, Z.; Li, Q.; Shao, D.; Jiang, C.; Huang, Q.; Yang, H.; Shi, J.; Hussain, N. Functional characterization and biotechnological potential of exopolysaccharide produced by Lactobacillus rhamnosus strains isolated from human breast milk. LWT-Food Sci. Technol. 2018, 89, 638–647. [Google Scholar] [CrossRef]
- El-Dein, A.N.; El-Deen, A.M.N.; El-Shatoury, E.H.; Awad, G.A.; Ibrahim, M.K.; Awad, H.M.; Farid, M.A. Assessment of exopolysaccharides, bacteriocins and in vitro and in vivo hypocholesterolemic potential of some Egyptian Lactobacillus spp. Int. J. Biol. Macromol. 2021, 173, 66–78. [Google Scholar] [CrossRef] [PubMed]
- Gan, D.; Ma, L.; Jiang, C.; Xu, R.; Zeng, X. Production, preliminary characterization and antitumor activity in vitro of polysaccharides from the mycelium of Pholiota dinghuensis Bi. Carbohydr. Polym. 2011, 84, 997–1003. [Google Scholar] [CrossRef]
- Devi, P.B.; Kavitake, D.; Jayamanohar, J.; Shetty, P.H. Preferential growth stimulation of probiotic bacteria by galactan exopolysaccharide from Weissella confusa KR780676. Food Res. Int. 2021, 143, 110333–110340. [Google Scholar] [CrossRef]
- Caggianiello, G.; Kleerebezem, M.; Spano, G. Exopolysaccharides produced by lactic acid bacteria: From health-promoting benefits to stress tolerance mechanisms. Appl. Microbiol. Biotechnol. 2016, 100, 3877–3886. [Google Scholar] [CrossRef]
- Mao, Y.; Doyle, M.P.; Chen, J. Role of colanic acid exopolysaccharide in the survival of enterohaemorrhagic Escherichia coli O157:H7 in simulated gastrointestinal fluids. Lett. Appl. Microbiol. 2006, 42, 642–647. [Google Scholar] [CrossRef]
- Khalil, M.A.; Sonbol, F.I.; Al-Madboly, L.A.; Aboshady, T.A.; Alqurashi, A.S.; Ali, S.S. Exploring the therapeutic potentials of exopolysaccharides derived from lactic acid bacteria and bifidobacteria: Antioxidant, antitumor, and periodontal regeneration. Front. Microbiol. 2022, 13, 803688–803699. [Google Scholar] [CrossRef] [PubMed]
- Hao, L.; Sheng, Z.; Lu, J.; Tao, R.; Jia, S. Characterization and antioxidant activities of extracellular and intracellular polysaccharides from Fomitopsis pinicola. Carbohydr. Polym. 2016, 141, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, N.; Shoji, M.; Hoshigami, H.; Watanabe, K.; Takatsuzu, T.; Yasuda, S.; Igoshi, K.; Kinoshita, H. Antioxidant capacity of soymilk yogurt and exopolysaccharides produced by lactic acid bacteria. Biosci. Microbiota Food Health 2019, 38, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Miao, M.; Jia, X.; Jiang, B.; Wu, S.; Cui, S.W.; Li, X. Elucidating molecular structure and prebiotics properties of bioengineered α-D-glucan from Leuconostoc citreum SK24.002. Food Hydrocoll. 2016, 54, 227–233. [Google Scholar] [CrossRef]
- Xu, Y.; Cui, Y.; Yue, F.; Liu, L.; Shan, Y.; Liu, B.; Zhou, Y.; Lü, X. Exopolysaccharides produced by lactic acid bacteria and bifidobacteria structures, physiofchemical functions and applications in the food industry. Food Hydrocoll. 2019, 94, 475–499. [Google Scholar] [CrossRef]
- Li, B.; Du, P.; Smith, E.E.; Wang, S.; Jiao, Y.; Guo, L.; Huo, G.; Liu, F. In vitro and in vivo evaluation of an exopolysaccharide produced by Lactobacillus helveticus KLDS1.8701 for the alleviative effect on oxidative stress. Food Funct. 2019, 10, 1707–1717. [Google Scholar] [CrossRef]
- Zhao, D.; Liu, L.; Jiang, J.; Guo, S.; Ping, W.; Ge, J. The response surface optimization of exopolysaccharide produced by Weissella confusa XG-3 and its rheological property. Prep. Biochem. Biotechnol. 2020, 50, 1014–1022. [Google Scholar] [CrossRef]
- Ayyash, M.; Abu-Jdayil, B.; Itsaranuwat, P.; Galiwango, E.; Tamiello-Rosa, C.; Abdullah, H.; Esposito, G.; Hunashal, Y.; Obaid, R.S.; Hamed, F. Characterization, bioactivities, and rheological properties of exopolysaccharide produced by novel probiotic Lactobacillus plantarum C70 isolated from camel milk. Int. J. Biol. Macromol. 2020, 144, 938–946. [Google Scholar] [CrossRef]
- Zhou, Y.; Cui, Y.; Qu, X. Exopolysaccharides of lactic acid bacteria: Structure, bioactivity and associations: A review. Carbohydr. Polym. 2019, 207, 317–332. [Google Scholar] [CrossRef]
- Fashogbon, R.O.; Adebayo-Tayo, B.; Sanusi, J. Optimization of extracellular polysaccharide substances from lactic acid bacteria isolated from fermented dairy products. Microbiol. J. 2021, 11, 1–11. [Google Scholar] [CrossRef]
- Poespowati, T.; Mahmudi, A. Optimization of acid hydrolysis process on macroalga Ulva lactuca for reducing sugar production as feedstock of bioethanol. Int. J. Renew. Energy Res. 2018, 8, 466–475. [Google Scholar]
- Al-Sheraji, S.H.; Ismail, A.; Manap, M.Y.; Mustafa, S.; Yusof, R.M.; Hassan, F.A. Fermentation and non-digestibility of Mangifera pajang fibrous pulp and its polysaccharides. J. Funct. Foods 2012, 4, 933–940. [Google Scholar] [CrossRef]
- Vecchione, A.; Celandroni, F.; Mazzantini, D.; Senesi, S.; Lupetti, A.; Ghelardi, E. Compositional quality and potential gastrointestinal behavior of probiotic products commercialized in Italy. Front. Med. 2018, 5, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Li, E.; Yang, H.; Zou, Y.; Wang, H.; Hu, T.; Li, Q.; Liao, S. In-vitro digestion by simulated gastrointestinal juices of Lactobacillus rhamnosus cultured with mulberry oligosaccharides and subsequent fermentation with human fecal inocula. LWT-Food Sci. Technol. 2019, 101, 61–68. [Google Scholar] [CrossRef]
- Wang, K.; Niu, M.; Song, D.; Song, X.; Zhao, J.; Wu, Y.; Lu, B.; Niu, G. Preparation, partial characterization and biological activity of exopolysaccharides produced from Lactobacillus fermentum S1. J. Biosci. Bioeng. 2020, 129, 206–214. [Google Scholar] [CrossRef]
- Ayyash, M.; Abu-Jdayil, B.; Olaimat, A.; Esposito, G.; Itsaranuwat, P.; Osaili, T.; Obaid, R.S.; Kizhakkayil, J.; Liu, S.-Q. Physicochemical, bioactive and rheological properties of an exopolysaccharide produced by a probiotic Pediococcus pentosaceus M41. Carbohydr. Polym. 2020, 229, 115462–115471. [Google Scholar] [CrossRef]


| Run | Factor 1 (%) Inoculation Size | Factor 2 (h) Time | Factor 3 (°C) Temperature | Factor 4 Initial pH | EPS (mg/L) |
|---|---|---|---|---|---|
| 1 | 4 | 24 | 37 | 6.40 | 356.92 |
| 2 | 5 | 24 | 37 | 6.00 | 187.33 |
| 3 | 4 | 24 | 34 | 6.80 | 154.49 |
| 4 | 4 | 24 | 37 | 6.40 | 356.92 |
| 5 | 5 | 18 | 37 | 6.40 | 194.38 |
| 6 | 4 | 24 | 37 | 6.40 | 356.92 |
| 7 | 5 | 24 | 34 | 6.40 | 165.39 |
| 8 | 4 | 24 | 34 | 6.00 | 194.61 |
| 9 | 3 | 24 | 40 | 6.40 | 136.76 |
| 10 | 3 | 30 | 37 | 6.40 | 177.21 |
| 11 | 3 | 24 | 34 | 6.40 | 188.13 |
| 12 | 4 | 24 | 37 | 6.40 | 356.92 |
| 13 | 4 | 18 | 34 | 6.40 | 171.54 |
| 14 | 4 | 30 | 40 | 6.40 | 158.81 |
| 15 | 3 | 24 | 37 | 6.80 | 139.14 |
| 16 | 4 | 30 | 37 | 6.80 | 205.19 |
| 17 | 4 | 18 | 37 | 6.80 | 151.87 |
| 18 | 5 | 24 | 37 | 6.80 | 148.58 |
| 19 | 4 | 24 | 37 | 6.40 | 356.92 |
| 20 | 4 | 24 | 40 | 6.00 | 210.74 |
| 21 | 4 | 24 | 40 | 6.80 | 169.37 |
| 22 | 3 | 24 | 37 | 6.00 | 176.22 |
| 23 | 5 | 30 | 37 | 6.40 | 183.02 |
| 24 | 4 | 18 | 40 | 6.40 | 208.02 |
| 25 | 4 | 30 | 37 | 6.00 | 224.72 |
| 26 | 3 | 18 | 37 | 6.40 | 143.59 |
| 27 | 4 | 18 | 37 | 6.00 | 175.74 |
| 28 | 4 | 30 | 34 | 6.40 | 345.98 |
| 29 | 5 | 24 | 40 | 6.40 | 182.90 |
| Source | Sum of Squares | df | Mean Squares | F-Value | p-Value | Significance |
|---|---|---|---|---|---|---|
| Model | 1.629 × 105 | 14 | 11,638.92 | 26.34 | <0.0001 | Significant |
| A-Inoculation size | 842.53 | 1 | 842.53 | 1.91 | 0.1890 | |
| B-Time | 4400.29 | 1 | 4400.29 | 9.96 | 0.0070 | ** |
| C-Temperature | 3799.94 | 1 | 3799.94 | 8.64 | 0.0109 | * |
| D-Initial pH | 2152.58 | 1 | 2152.58 | 4.87 | 0.0445 | * |
| AB | 505.80 | 1 | 505.80 | 1.14 | 0.3028 | |
| AC | 1186.11 | 1 | 1186.11 | 2.68 | 0.1236 | |
| AD | 0.70 | 1 | 0.70 | 1.578 × 10-3 | 0.9689 | |
| BC | 12,504.83 | 1 | 12,504.83 | 28.30 | 0.0001 | ** |
| BD | 148.11 | 1 | 148.11 | 0.34 | 0.5718 | |
| CD | 87.89 | 1 | 87.89 | 0.20 | 0.6624 | |
| A2 | 72,014.98 | 1 | 72,014.98 | 162.97 | <0.0001 | ** |
| B2 | 30,161.36 | 1 | 30,161.36 | 68.26 | <0.0001 | ** |
| C2 | 41,364.46 | 1 | 41,364.46 | 93.61 | <0.0001 | ** |
| D2 | 66,101.51 | 1 | 66,101.51 | 149.59 | <0.0001 | ** |
| Residual | 6186.39 | 14 | 441.88 | |||
| Lack of fit | 6186.39 | 10 | 618.64 | |||
| Pure error | 0.000 | 4 | 0.000 | |||
| Cor total | 1.691 × 105 | 28 | ||||
| R2 | 0.9634 | |||||
| Adj-R2 | 0.9268 | |||||
| C.V. % | 10.00 |
| Groups | OD600 | EPS (mg/L) |
|---|---|---|
| Verification value | 0.077 a | 345.98 A |
| Initial value | 0.065 b | 140.34 B |
| Strains | EPS (mg/L) | DPPH | OH | Isolation Source |
|---|---|---|---|---|
| L. plantarum C88 | 69.00 | 52.23% (4000 mg/L) | 85.21% (4000 mg/L) | Chinese traditional fermented dairy Tofu [25] |
| L. plantarum YW11 | 90.00 | - | - | Kefir grains collected from Tibet [26] |
| L. plantarum EP56 | 126.40 | - | - | Corn silage [15] |
| S. thermophilus W22 | 127.00 | - | - | Village type yogurt [9] |
| L. delbruckii subsp. Bulgaricus B3 | 263.00 | - | - | |
| L. delbruckii subsp. Bulgaricus G12 | 238.00 | - | - | |
| L. rhamnosus ATCC 9595 | 352.00 | - | - | Human breast milk [27] |
| L. rhamnosus SHA114 | 461.00 | - | - | |
| L. rhamnosus SHA113 | 549.60 | - | - | |
| L. plantarum KX041 | 599.52 | 82.00% (6000 mg/L) | 82.64% (8000 mg/L) | Traditional Chinese pickle juice [24] |
| L. plantarum NTMI20 | 827.00 | 91.86% (500 mg/L) | - | Milk sources [2] |
| L. plantarum NTMI05 | 956.00 | 96.62% (500 mg/L) | - | |
| L. plantarum KU985433 | 2030.00 | 88.00% (4000 mg/L) | - | Egyptian fermented food [28] |
| L. rhamnosus RW-9595 M | 2767.00 | - | - | LAB research network culture collection [8] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, X.; Liang, Q. Optimization, Probiotic Characteristics, and Rheological Properties of Exopolysaccharides from Lactiplantibacillus plantarum MC5. Molecules 2023, 28, 2463. https://doi.org/10.3390/molecules28062463
Zhao X, Liang Q. Optimization, Probiotic Characteristics, and Rheological Properties of Exopolysaccharides from Lactiplantibacillus plantarum MC5. Molecules. 2023; 28(6):2463. https://doi.org/10.3390/molecules28062463
Chicago/Turabian StyleZhao, Xuefang, and Qi Liang. 2023. "Optimization, Probiotic Characteristics, and Rheological Properties of Exopolysaccharides from Lactiplantibacillus plantarum MC5" Molecules 28, no. 6: 2463. https://doi.org/10.3390/molecules28062463
APA StyleZhao, X., & Liang, Q. (2023). Optimization, Probiotic Characteristics, and Rheological Properties of Exopolysaccharides from Lactiplantibacillus plantarum MC5. Molecules, 28(6), 2463. https://doi.org/10.3390/molecules28062463






