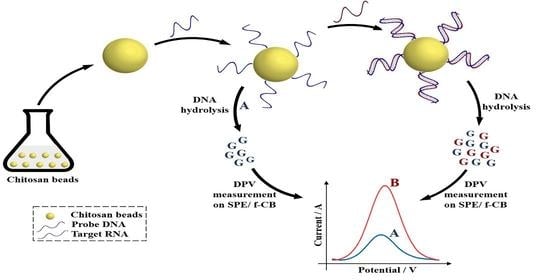
Development of a New Route for the Immobilization of Unmodified Single-Stranded DNA on Chitosan Beads and Detection of Released Guanine after Hydrolysis
Abstract
1. Introduction
2. Results and Discussion
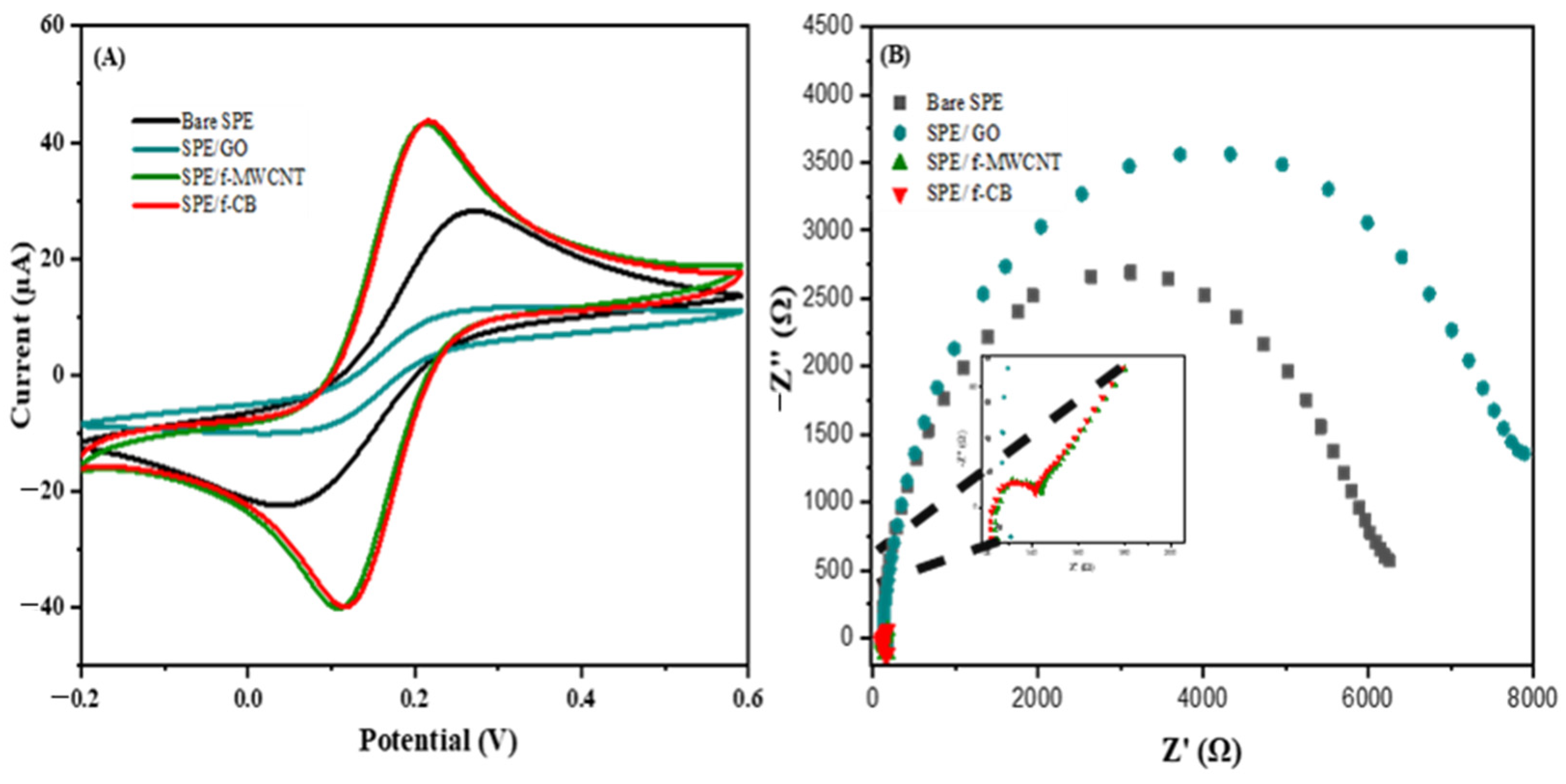
2.1. Electrochemical Characterization of Nanomaterial-Modified SPE
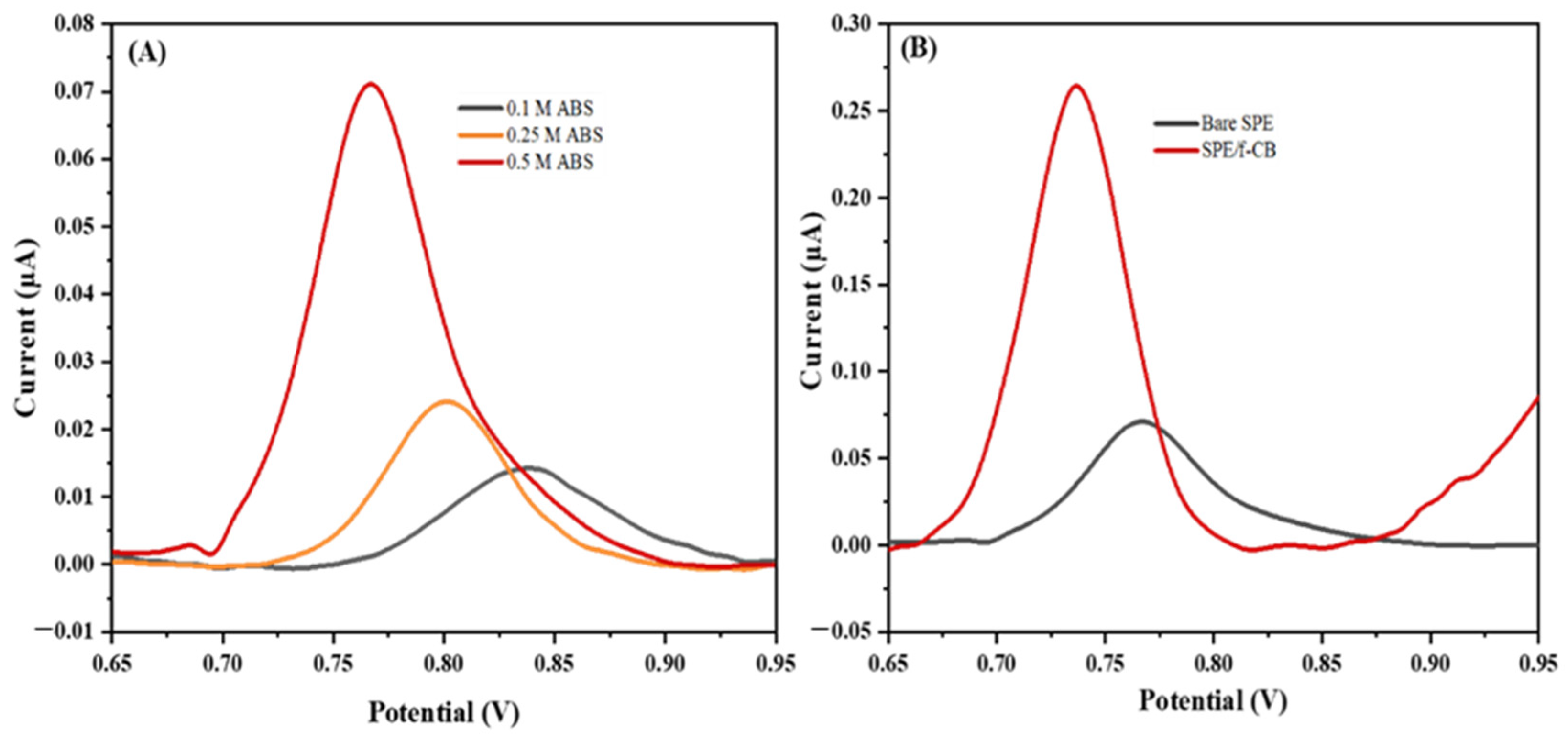
2.2. Effect of Buffer Concentration and Modification of SPE with f-CB in the Electrochemical Oxidation of Guanine
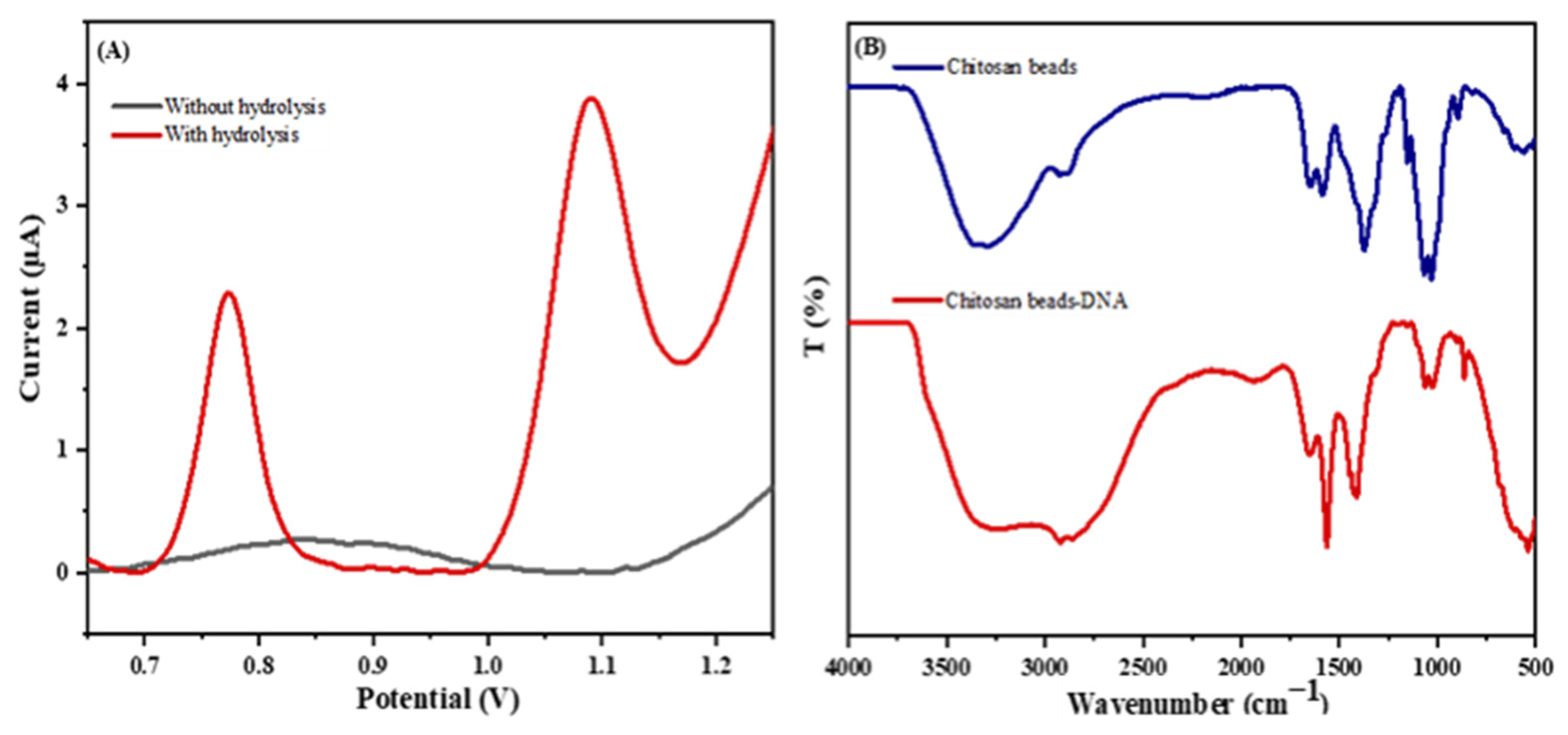
2.3. DNA Hydrolysis
2.4. Optimization of the Concentration of DNA Capture Probe
2.5. Electrochemical Detection of miRNA-222
2.6. Repeatability
2.7. Selectivity Study
2.8. MiRNA-222 Analysis in Real Sample
3. Materials and Methods
3.1. Chemicals
3.2. Apparatus
3.3. Preparation of Functionalized Carbon Black
3.4. Preparation of Nanomaterial Dispersions
3.5. SPE Modification by Nanomaterials
3.6. Electrochemical Measurements
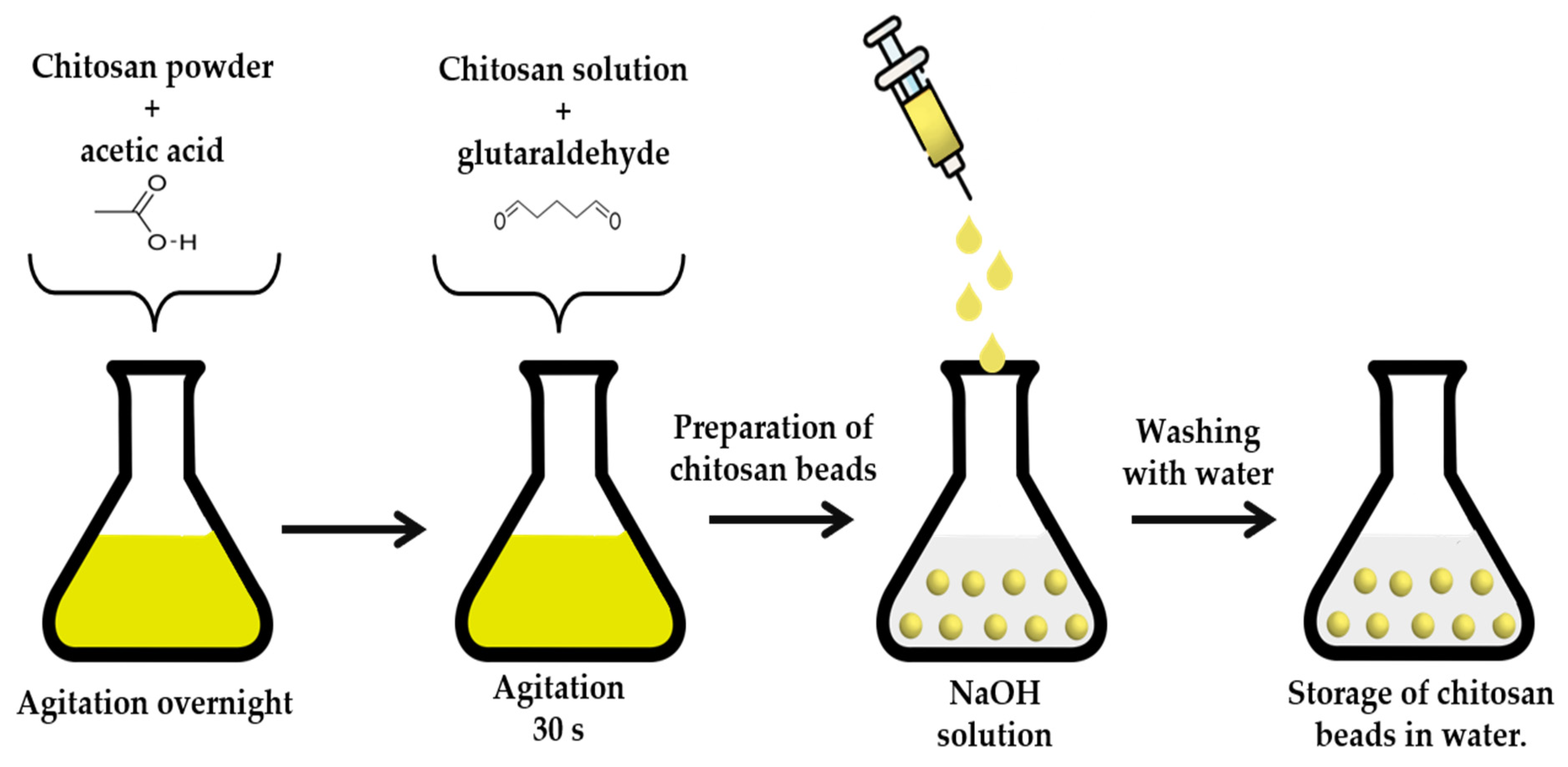
3.7. Preparation of Chitosan Beads
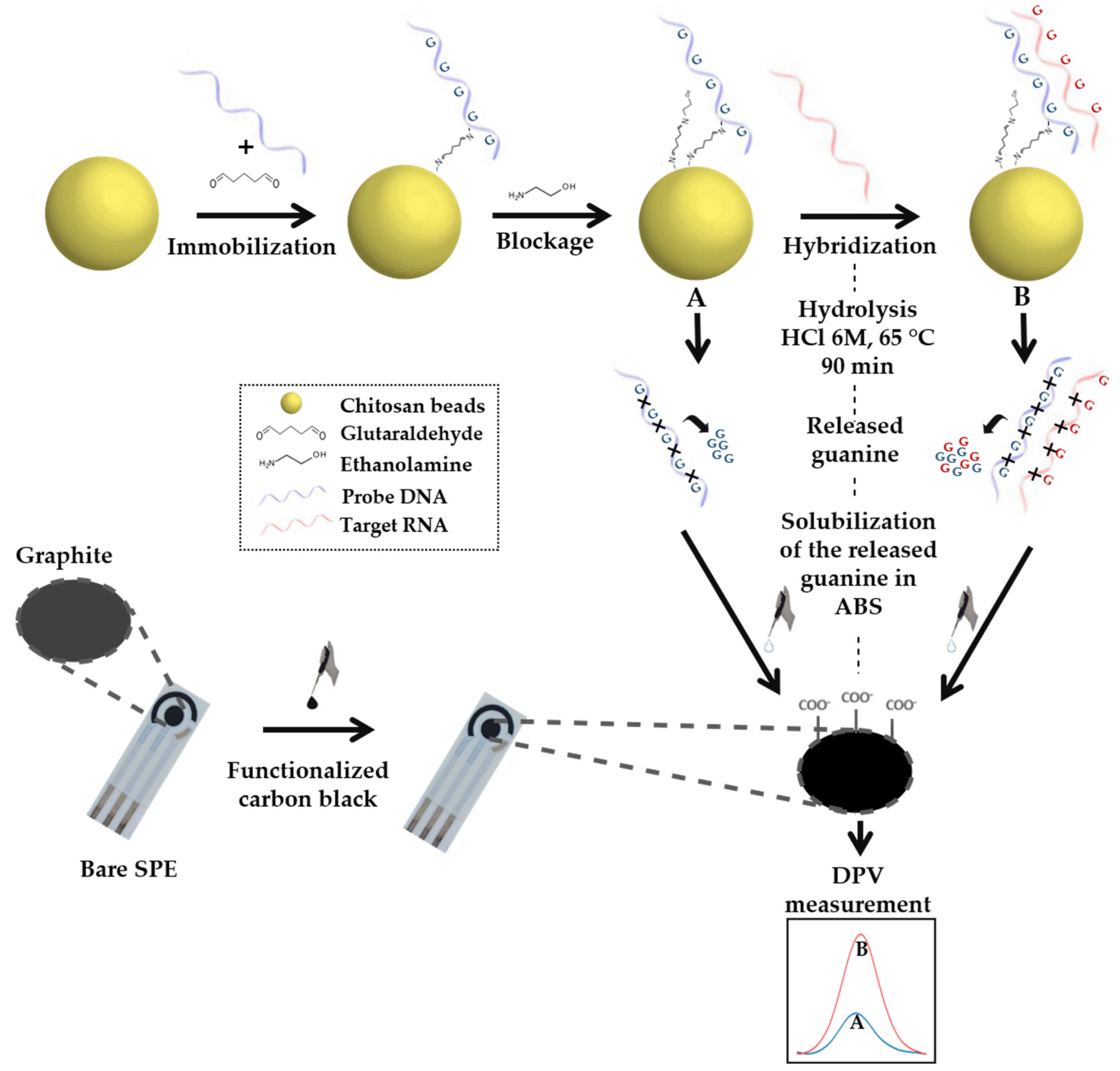
3.8. Single-Stranded DNA Immobilization
3.9. Hybridization of miRNA
3.10. Hydrolysis of Nucleic Acid Compounds and Liberation of Guanine
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Karrat, A.; Lamaoui, A.; Amine, A.; Palacios-Santander, J.M.; Cubillana-Aguilera, L. Applications of chitosan in molecularly and ion imprinted polymers. Chem. Afr. 2020, 3, 513–533. [Google Scholar] [CrossRef]
- Lima, M.; Teixeira-Santos, R.; Gomes, L.C.; Faria, S.I.; Valcarcel, J.; Vázquez, J.A.; Cerqueira, M.A.; Pastrana, L.; Bourbon, A.I.; Mergulhão, F.J. Development of chitosan-based surfaces to prevent single-and dual-species biofilms of Staphylococcus aureus and Pseudomonas aeruginosa. Molecules 2021, 26, 4378. [Google Scholar] [CrossRef]
- Zargar, V.; Asghari, M.; Dashti, A. A review on chitin and chitosan polymers: Structure, chemistry, solubility, derivatives, and applications. ChemBioEng Rev. 2015, 2, 204–226. [Google Scholar] [CrossRef]
- Carreno-Gomez, B.; Duncan, R. Evaluation of the biological properties of soluble chitosan and chitosan microspheres. Int. J. Pharm. 1997, 148, 231–240. [Google Scholar] [CrossRef]
- Dutta, P.; Tripathi, S.; Mehrotra, G.; Dutta, J. Perspectives for chitosan based antimicrobial films in food applications. Food Chem. 2009, 114, 1173–1182. [Google Scholar] [CrossRef]
- Yang, T.-C.; Chou, C.-C.; Li, C.-F. Preparation, water solubility and rheological property of the N-alkylated mono or disaccharide chitosan derivatives. Food Res. Int. 2002, 35, 707–713. [Google Scholar] [CrossRef]
- Yu, W.; Tang, Y.; Sang, Y.; Liu, W.; Wang, S.; Wang, X. Preparation of a carboxylated single-walled carbon-nanotube-chitosan functional layer and its application to a molecularly imprinted electrochemical sensor to quantify semicarbazide. Food Chem. 2020, 333, 127524. [Google Scholar] [CrossRef]
- Lou, B.-S.; Rajaji, U.; Chen, S.-M.; Chen, T.-W. A simple sonochemical assisted synthesis of NiMoO4/chitosan nanocomposite for electrochemical sensing of amlodipine in pharmaceutical and serum samples. Ultrason. Sonochem. 2020, 64, 104827. [Google Scholar] [CrossRef]
- Aranaz, I.; Acosta, N.; Civera, C.; Elorza, B.; Mingo, J.; Castro, C.; Gandía, M.D.L.L.; Heras Caballero, A. Cosmetics and cosmeceutical applications of chitin, chitosan and their derivatives. Polymers 2018, 10, 213. [Google Scholar] [CrossRef]
- Xing, K.; Zhu, X.; Peng, X.; Qin, S. Chitosan antimicrobial and eliciting properties for pest control in agriculture: A review. Agron. Sustain. Dev. 2015, 35, 569–588. [Google Scholar] [CrossRef]
- Rao, D.; Sheng, Q.; Zheng, J. Preparation of flower-like Pt nanoparticles decorated chitosan-grafted graphene oxide and its electrocatalysis of hydrazine. Sens. Actuators B Chem. 2016, 236, 192–200. [Google Scholar] [CrossRef]
- Lu, J.; Cui, D.; Li, H.; Zhang, Y.; Liu, S. Cytochrome P450 bienzymes assembled on Au/chitosan/reduced graphene oxide nanosheets for electrochemically-driven drug cascade metabolism. Electrochim. Acta 2015, 165, 36–44. [Google Scholar] [CrossRef]
- Yadav, A.K.; Dhiman, T.K.; Lakshmi, G.; Berlina, A.N.; Solanki, P.R. A highly sensitive label-free amperometric biosensor for norfloxacin detection based on chitosan-yttria nanocomposite. Int. J. Biol. Macromol. 2020, 151, 566–575. [Google Scholar] [CrossRef]
- Xu, B.; Zheng, D.; Qiu, W.; Gao, F.; Jiang, S.; Wang, Q. An ultrasensitive DNA biosensor based on covalent immobilization of probe DNA on fern leaf-like α-Fe2O3 and chitosan Hybrid film using terephthalaldehyde as arm-linker. Biosens. Bioelectron. 2015, 72, 175–181. [Google Scholar] [CrossRef]
- Karrat, A.; Amine, A. Recent advances in chitosan-based electrochemical sensors and biosensors. Arab. J. Chem. Environ. Res. 2020, 7, 66–93. [Google Scholar]
- Giraldo, J.D.; Rivas, B.L. Direct ionization and solubility of chitosan in aqueous solutions with acetic acid. Polym. Bull. 2021, 78, 1465–1488. [Google Scholar] [CrossRef]
- Gayathri, S.B.; Kamaraj, P.; Arthanareeswari, M.; Devikala, S. DNA nanostructures based biosensor for the determination of aromatic compounds. Biosens. Bioelectron. 2015, 72, 191–196. [Google Scholar] [CrossRef]
- Rashid, J.I.A.; Yusof, N.A. The strategies of DNA immobilization and hybridization detection mechanism in the construction of electrochemical DNA sensor: A review. Sens. Bio-Sens. Res. 2017, 16, 19–31. [Google Scholar] [CrossRef]
- Yammouri, G.; Mohammadi, H.; Amine, A. A highly sensitive electrochemical biosensor based on carbon black and gold nanoparticles modified pencil graphite electrode for microRNA-21 detection. Chem. Afr. 2019, 2, 291–300. [Google Scholar] [CrossRef]
- Chen, Y.-X.; Wu, X.; Huang, K.-J. A sandwich-type electrochemical biosensing platform for microRNA-21 detection using carbon sphere-MoS2 and catalyzed hairpin assembly for signal amplification. Sens. Actuators B Chem. 2018, 270, 179–186. [Google Scholar] [CrossRef]
- El Aamri, M.; Mohammadi, H.; Amine, A. Development of a Novel Electrochemical Sensor Based on Functionalized Carbon Black for the Detection of Guanine Released from DNA Hydrolysis. Electroanalysis 2022, 35, e202100613. [Google Scholar] [CrossRef]
- El Aamri, M.; Mohammadi, H.; Amine, A. Novel label-free colorimetric and electrochemical detection for MiRNA-21 based on the complexation of molybdate with phosphate. Microchem. J. 2022, 182, 107851. [Google Scholar] [CrossRef]
- Yola, M.L.; Eren, T.; Atar, N. A novel and sensitive electrochemical DNA biosensor based on Fe@ Au nanoparticles decorated graphene oxide. Electrochim. Acta 2014, 125, 38–47. [Google Scholar] [CrossRef]
- Lin, X.-H.; Wu, P.; Chen, W.; Zhang, Y.-F.; Xia, X.-H. Electrochemical DNA biosensor for the detection of short DNA species of Chronic Myelogenous Leukemia by using methylene blue. Talanta 2007, 72, 468–471. [Google Scholar] [CrossRef]
- Mohammadi, H.; Yammouri, G.; Amine, A. Current advances in electrochemical genosensors for detecting microRNA cancer markers. Curr. Opin. Electrochem. 2019, 16, 96–105. [Google Scholar] [CrossRef]
- Akanda, M.R.; Sohail, M.; Aziz, M.A.; Kawde, A.N. Recent advances in nanomaterial-modified pencil graphite electrodes for electroanalysis. Electroanalysis 2016, 28, 408–424. [Google Scholar] [CrossRef]
- Gallay, P.; López Mujica, M.; Bollo, S.; Rivas, G. Genosensing Applications of Glassy Carbon Electrodes Modified with Multi-Walled Carbon Nanotubes Non-Covalently Functionalized with Polyarginine. Micromachines 2022, 13, 1978. [Google Scholar] [CrossRef] [PubMed]
- Moustakim, H.; Mohammadi, H.; Amine, A. Electrochemical DNA Biosensor Based on Immobilization of a Non-Modified ssDNA Using Phosphoramidate-Bonding Strategy and Pencil Graphite Electrode Modified with AuNPs/CB and Self-Assembled Cysteamine Monolayer. Sensors 2022, 22, 9420. [Google Scholar] [CrossRef] [PubMed]
- Arduini, F.; Cinti, S.; Mazzaracchio, V.; Scognamiglio, V.; Amine, A.; Moscone, D. Carbon black as an outstanding and affordable nanomaterial for electrochemical (bio) sensor design. Biosens. Bioelectron. 2020, 156, 112033. [Google Scholar] [CrossRef]
- Elanthamilan, E.; Sriram, B.; Rajkumar, S.; Dhaneshwaran, C.; Nagaraj, N.; Merlin, J.P.; Vijayan, A.; Wang, S.-F. Couroupita guianansis dead flower derived porous activated carbon as efficient supercapacitor electrode material. Mater. Res. Bull. 2019, 112, 390–398. [Google Scholar] [CrossRef]
- Aamri, M.E.; Mohammadi, H.; Amine, A. Based Colorimetric Detection of miRNA-21 Using Pre-Activated Nylon Membrane and Peroxidase-Mimetic Activity of Cysteamine-Capped Gold Nanoparticles. Biosensors 2023, 13, 74. [Google Scholar] [CrossRef] [PubMed]
- Du, P.; Li, H.; Mei, Z.; Liu, S. Electrochemical DNA biosensor for the detection of DNA hybridization with the amplification of Au nanoparticles and CdS nanoparticles. Bioelectrochemistry 2009, 75, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Pothipor, C.; Aroonyadet, N.; Bamrungsap, S.; Jakmunee, J.; Ounnunkad, K. A highly sensitive electrochemical microRNA-21 biosensor based on intercalating methylene blue signal amplification and a highly dispersed gold nanoparticles/graphene/polypyrrole composite. Analyst 2021, 146, 2679–2688. [Google Scholar] [CrossRef]
- Gai, P.; Gu, C.; Li, H.; Sun, X.; Li, F. Ultrasensitive ratiometric homogeneous electrochemical microRNA biosensing via target-triggered Ru (III) release and redox recycling. Anal. Chem. 2017, 89, 12293–12298. [Google Scholar] [CrossRef] [PubMed]
- Azab, S.M.; Elhakim, H.K.; Fekry, A.M. The strategy of nanoparticles and the flavone chrysin to quantify miRNA-let 7a in zepto-molar level: Its application as tumor marker. J. Mol. Struct. 2019, 1196, 647–652. [Google Scholar] [CrossRef]
- Wang, P.; Wu, H.; Dai, Z.; Zou, X. Simultaneous detection of guanine, adenine, thymine and cytosine at choline monolayer supported multiwalled carbon nanotubes film. Biosens. Bioelectron. 2011, 26, 3339–3345. [Google Scholar] [CrossRef]
- Bettazzi, F.; Hamid-Asl, E.; Esposito, C.L.; Quintavalle, C.; Formisano, N.; Laschi, S.; Catuogno, S.; Iaboni, M.; Marrazza, G.; Mascini, M. Electrochemical detection of miRNA-222 by use of a magnetic bead-based bioassay. Anal. Bioanal. Chem. 2013, 405, 1025–1034. [Google Scholar] [CrossRef]
- Chiticaru, E.A.; Pilan, L.; Ioniţă, M. Electrochemical Detection Platform Based on RGO Functionalized with Diazonium Salt for DNA Hybridization. Biosensors 2022, 12, 39. [Google Scholar] [CrossRef]
- Phua, J.L.; Teh, P.L.; Yeoh, C.K.; Voon, C.H. Functionalized carbon black in epoxy composites: Effect of single-and dual-matrix systems. Polym. Bull. 2022, 79, 5437–5455. [Google Scholar] [CrossRef]
- Jang, Y.H.; Goddard, W.A.; Noyes, K.T.; Sowers, L.C.; Hwang, S.; Chung, D.S. pKa Values of Guanine in Water: Density Functional Theory Calculations Combined with Poisson−Boltzmann Continuum−Solvation Model. J. Phys. Chem. B 2003, 107, 344–357. [Google Scholar] [CrossRef]
- Arduini, F.; Di Nardo, F.; Amine, A.; Micheli, L.; Palleschi, G.; Moscone, D. Carbon black-modified screen-printed electrodes as electroanalytical tools. Electroanalysis 2012, 24, 743–751. [Google Scholar] [CrossRef]
- Liu, S.; Su, W.; Li, Y.; Zhang, L.; Ding, X. Manufacturing of an electrochemical biosensing platform based on hybrid DNA hydrogel: Taking lung cancer-specific miR-21 as an example. Biosens. Bioelectron. 2018, 103, 1–5. [Google Scholar] [CrossRef]
- Zhao, H.; Qu, Y.; Yuan, F.; Quan, X. A visible and label-free colorimetric sensor for miRNA-21 detection based on peroxidase-like activity of graphene/gold-nanoparticle hybrids. Anal. Methods 2016, 8, 2005–2012. [Google Scholar] [CrossRef]
- Nascimento, G.A.; Souza, E.V.; Campos-Ferreira, D.S.; Arruda, M.S.; Castelletti, C.H.; Wanderley, M.S.; Ekert, M.H.; Bruneska, D.; Lima-Filho, J.L. Electrochemical DNA biosensor for bovine papillomavirus detection using polymeric film on screen-printed electrode. Biosens. Bioelectron. 2012, 38, 61–66. [Google Scholar] [CrossRef]
- Chung, D.-J.; Kim, K.-C.; Choi, S.-H. Electrochemical DNA biosensor based on avidin–biotin conjugation for influenza virus (type A) detection. Appl. Surf. Sci. 2011, 257, 9390–9396. [Google Scholar] [CrossRef]
- Marrazza, G.; Chianella, I.; Mascini, M. Disposable DNA electrochemical sensor for hybridization detection. Biosens. Bioelectron. 1999, 14, 43–51. [Google Scholar] [CrossRef]
- Erdem, A.; Papakonstantinou, P.; Murphy, H.; McMullan, M.; Karadeniz, H.; Sharma, S. Streptavidin modified carbon nanotube based graphite electrode for label-free sequence specific dna detection. Electroanal. Int. J. Devoted Fundam. Pract. Asp. Electroanal. 2010, 22, 611–617. [Google Scholar] [CrossRef]
- Ramulu, T.; Venu, R.; Sinha, B.; Lim, B.; Jeon, S.; Yoon, S.; Kim, C. Nanowires array modified electrode for enhanced electrochemical detection of nucleic acid. Biosens. Bioelectron. 2013, 40, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zhang, B.; Lin, X.; Weng, W. Hybridization biosensor based on the covalent immobilization of probe DNA on chitosan–mutiwalled carbon nanotubes nanocomposite by using glutaraldehyde as an arm linker. Sens. Actuators B Chem. 2011, 156, 599–605. [Google Scholar] [CrossRef]
- Barreiro-Iglesias, R.; Coronilla, R.; Concheiro, A.; Alvarez-Lorenzo, C. Preparation of chitosan beads by simultaneous cross-linking/insolubilisation in basic pH: Rheological optimisation and drug loading/release behaviour. Eur. J. Pharm. Sci. 2005, 24, 77–84. [Google Scholar] [CrossRef]



| Platform | Strategies | Detection Method | Solid Substrate | Limit of Detection | Reference |
|---|---|---|---|---|---|
| PET | Adsorption of ferrocene-DNA | Current reduction of ferrocene tags by CV and DPV. | ITO | 5 × 10−9 M | [42] |
| Graphene/AuNP hybrid | Adsorption of unmodified DNA | Catalytic activity of hybrids with a concomitant colorless-to-blue color change. | In solution | 3.2 × 10−9 M | [43] |
| Polylysine-chitosan | Adsorption of unmodified DNA | Decrease of the signal of methylene blue by DPV | Au | 4.4 × 10−9 M | [44] |
| Ar-COOH/Avidin | Avidin interaction with biotinylated DNA | Decrease of current value after hybridization using CV | GCE | 8.51 × 10−14 M | [45] |
| Graphite-Avidin | Avidin interaction with biotinylated DNA | Daunomycin hydrochloride as an indicator of hybridization reaction (DPV) | SPE | 1 µg/mL | [46] |
| MWCNT-STR | Avidin interaction with biotinylated DNA | Guanine oxidation using DPV | PGE | 1.45 × 10−7 M | [47] |
| AuNWs | Covalent interaction with SH-DNA | Oxidation signal of methylene blue accumulation measured using DPV | AuNWs electrode | 6.78 × 10−9 M | [48] |
| Chitosan-MWCNTs/GTD | Covalent interaction with NH2-DNA | Hybridization capacity monitored by EIS | GCE | 8.5 × 10−14 M | [49] |
| RGO/Ar-COOH/EDC/NHS | Covalent interaction with NH2-DNA | Hybridization capacity monitored by EIS | SPE | 1 × 10−9 M | [38] |
| Chitosan beads/GTD | Covalent interaction with unmodified DNA | DPV oxidation of released guanines | SPE | 0.2 × 10−9 M | This work |
| Added miRNA (nM) | Detected (nM) | Recovery (%) | RSD % |
|---|---|---|---|
| 1 | 0.99 | 99.0 | 2.55 |
| 10 | 01.3 | 103.0 | 3.51 |
| 1000 | 986 | 98.6 | 2.02 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chahri, I.; Karrat, A.; Mohammadi, H.; Amine, A. Development of a New Route for the Immobilization of Unmodified Single-Stranded DNA on Chitosan Beads and Detection of Released Guanine after Hydrolysis. Molecules 2023, 28, 2088. https://doi.org/10.3390/molecules28052088
Chahri I, Karrat A, Mohammadi H, Amine A. Development of a New Route for the Immobilization of Unmodified Single-Stranded DNA on Chitosan Beads and Detection of Released Guanine after Hydrolysis. Molecules. 2023; 28(5):2088. https://doi.org/10.3390/molecules28052088
Chicago/Turabian StyleChahri, Ikram, Abdelhafid Karrat, Hasna Mohammadi, and Aziz Amine. 2023. "Development of a New Route for the Immobilization of Unmodified Single-Stranded DNA on Chitosan Beads and Detection of Released Guanine after Hydrolysis" Molecules 28, no. 5: 2088. https://doi.org/10.3390/molecules28052088
APA StyleChahri, I., Karrat, A., Mohammadi, H., & Amine, A. (2023). Development of a New Route for the Immobilization of Unmodified Single-Stranded DNA on Chitosan Beads and Detection of Released Guanine after Hydrolysis. Molecules, 28(5), 2088. https://doi.org/10.3390/molecules28052088