Possible Roles of Transition Metal Cations in the Formation of Interstellar Benzene via Catalytic Acetylene Cyclotrimerization
Abstract
:1. Introduction
2. Results and Discussion
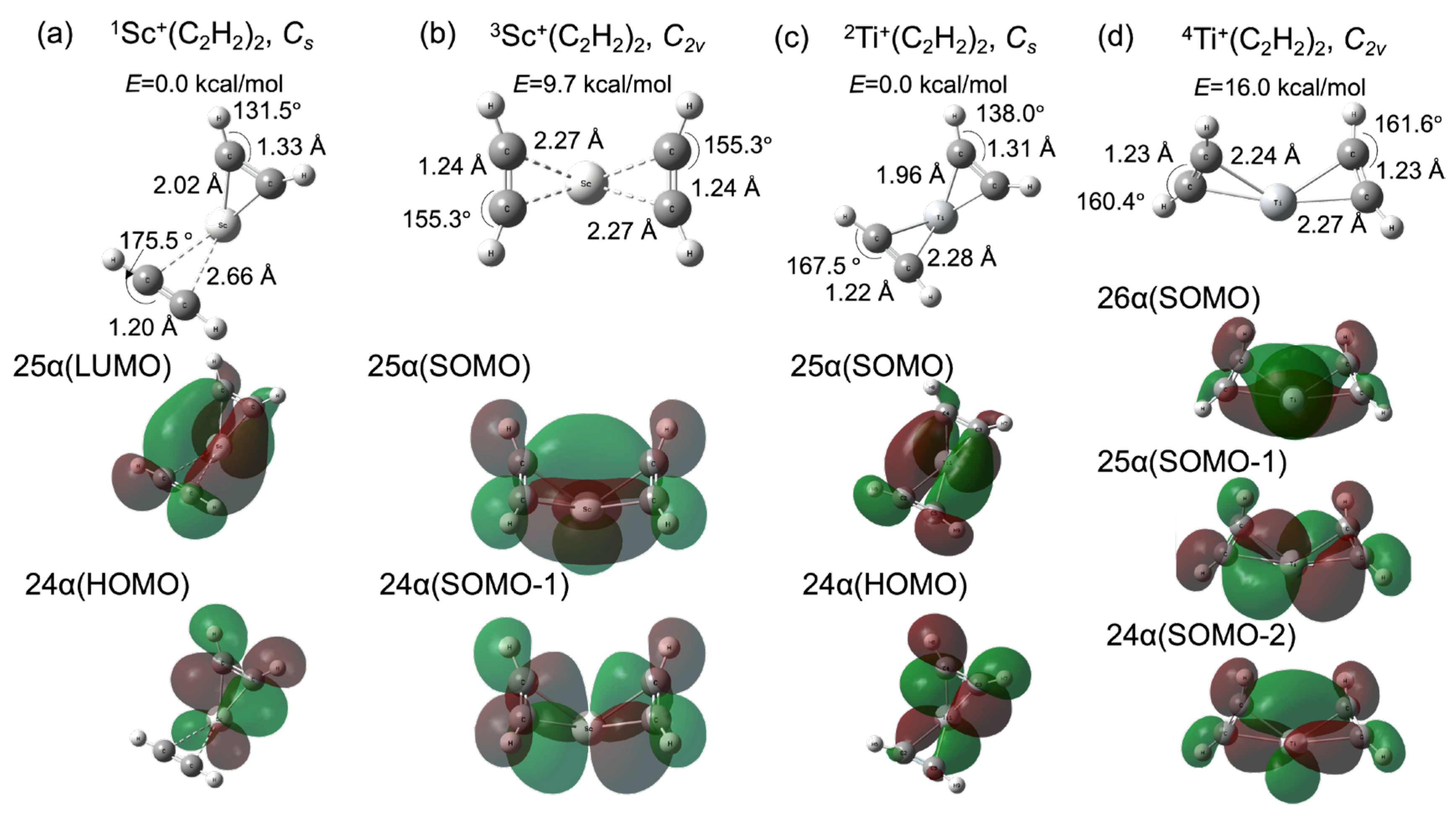
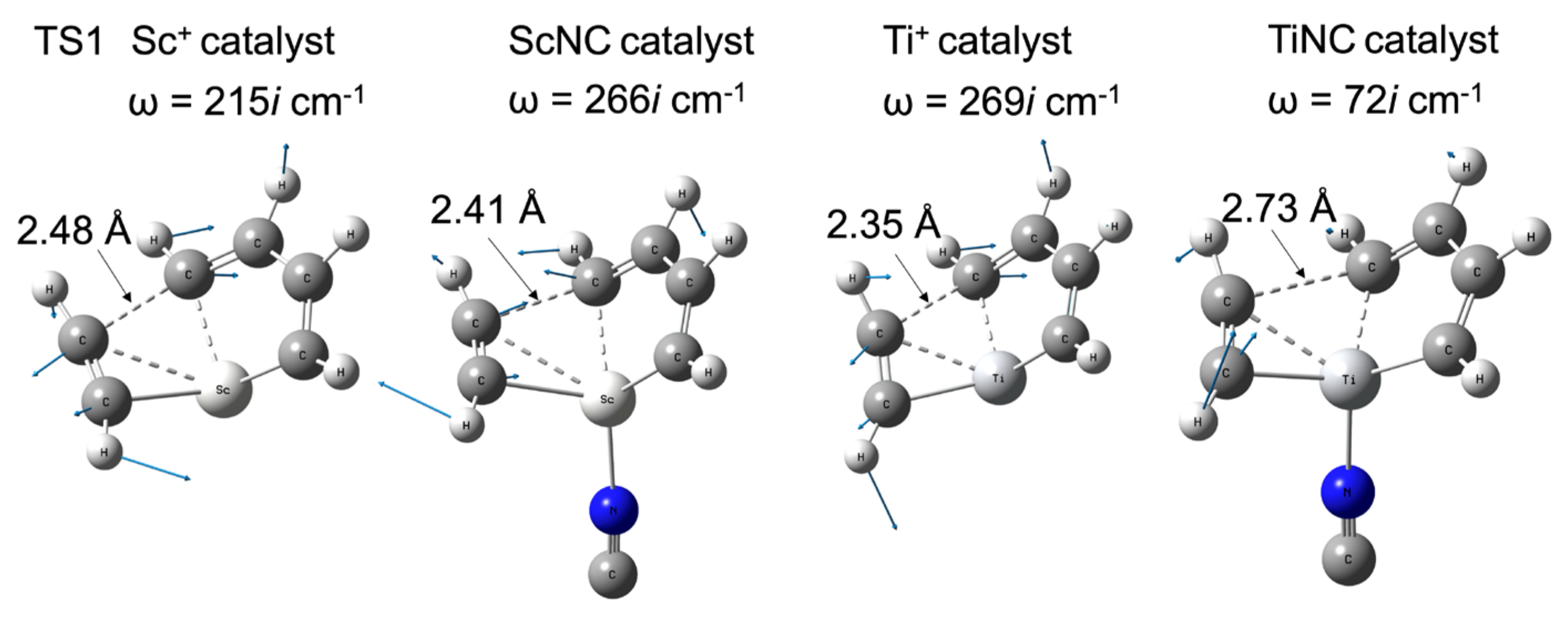
2.1. Bare Sc+ and Ti+ Catalyst Systems
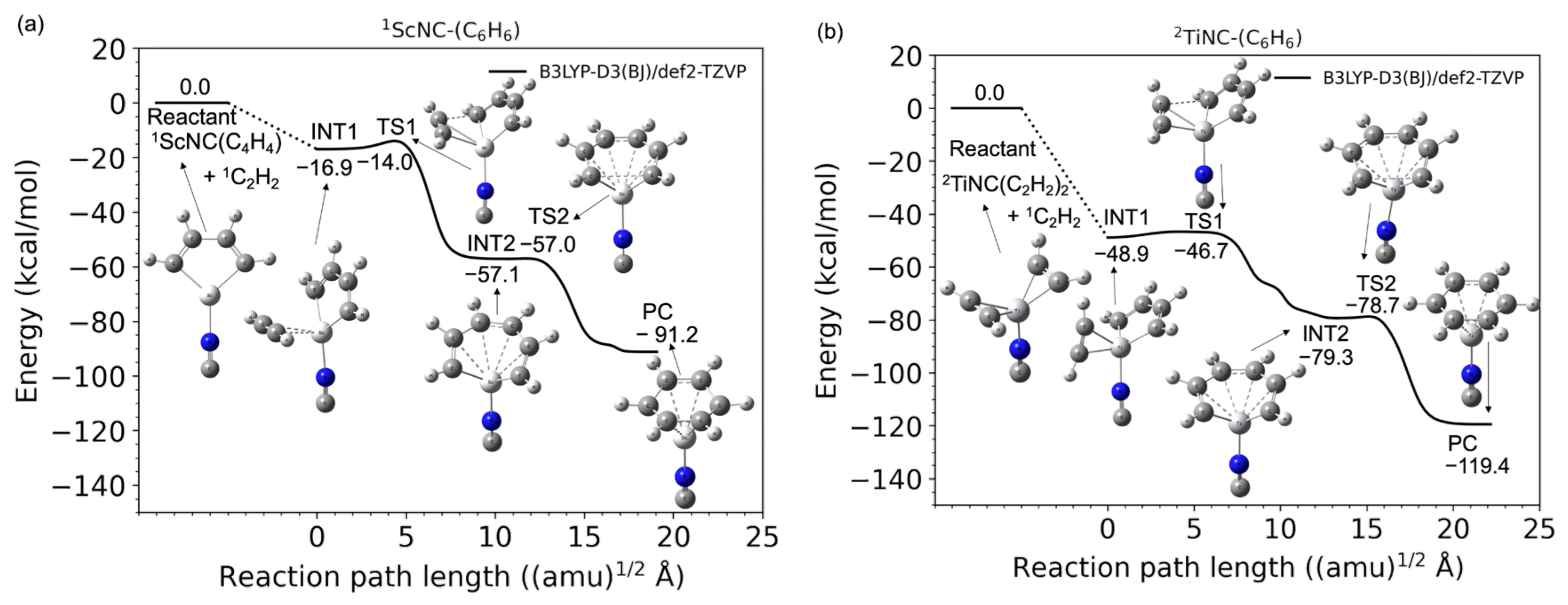
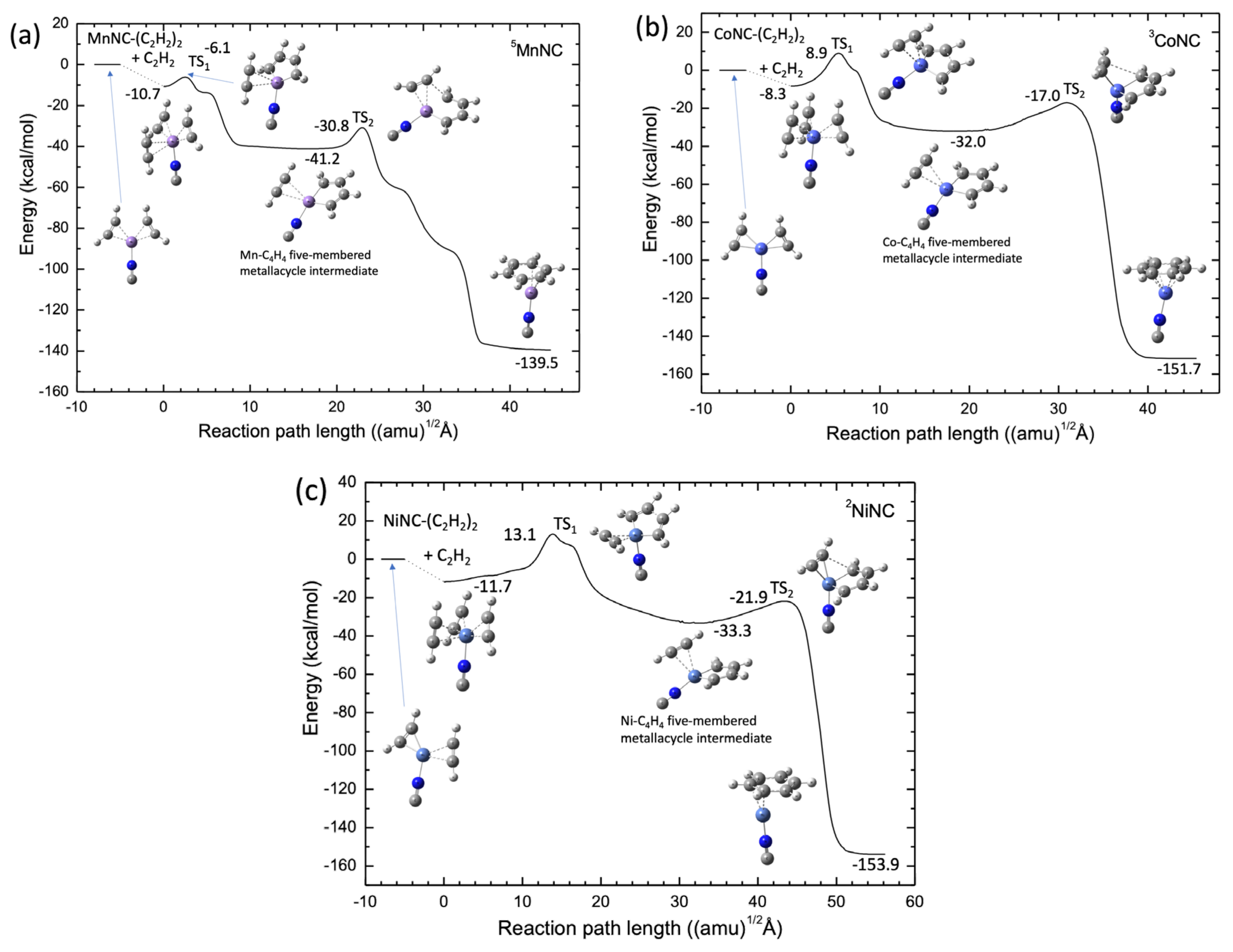
2.2. TM+NC− Catalyst System
2.3. TM+(H2O)8 Catalyst System
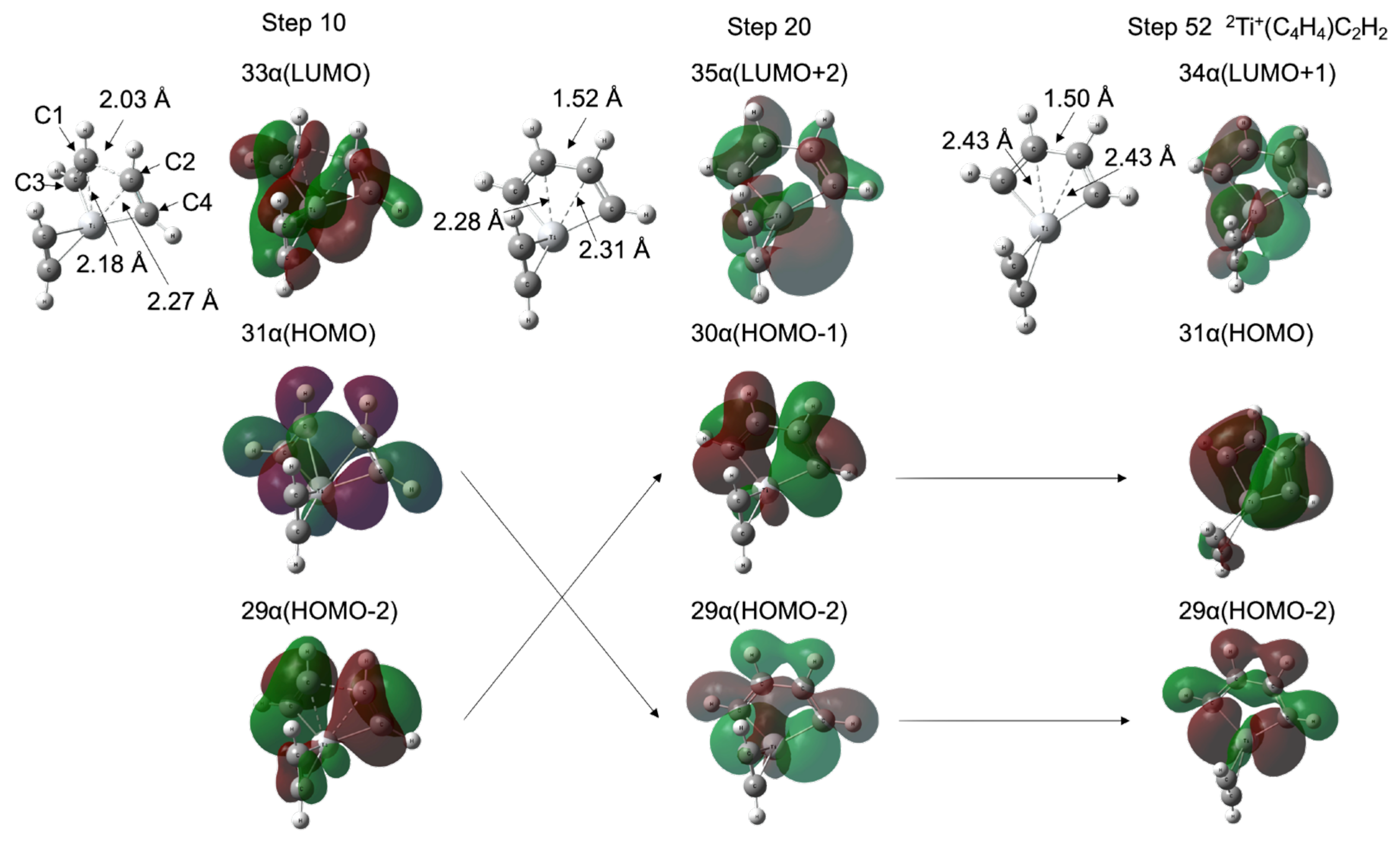
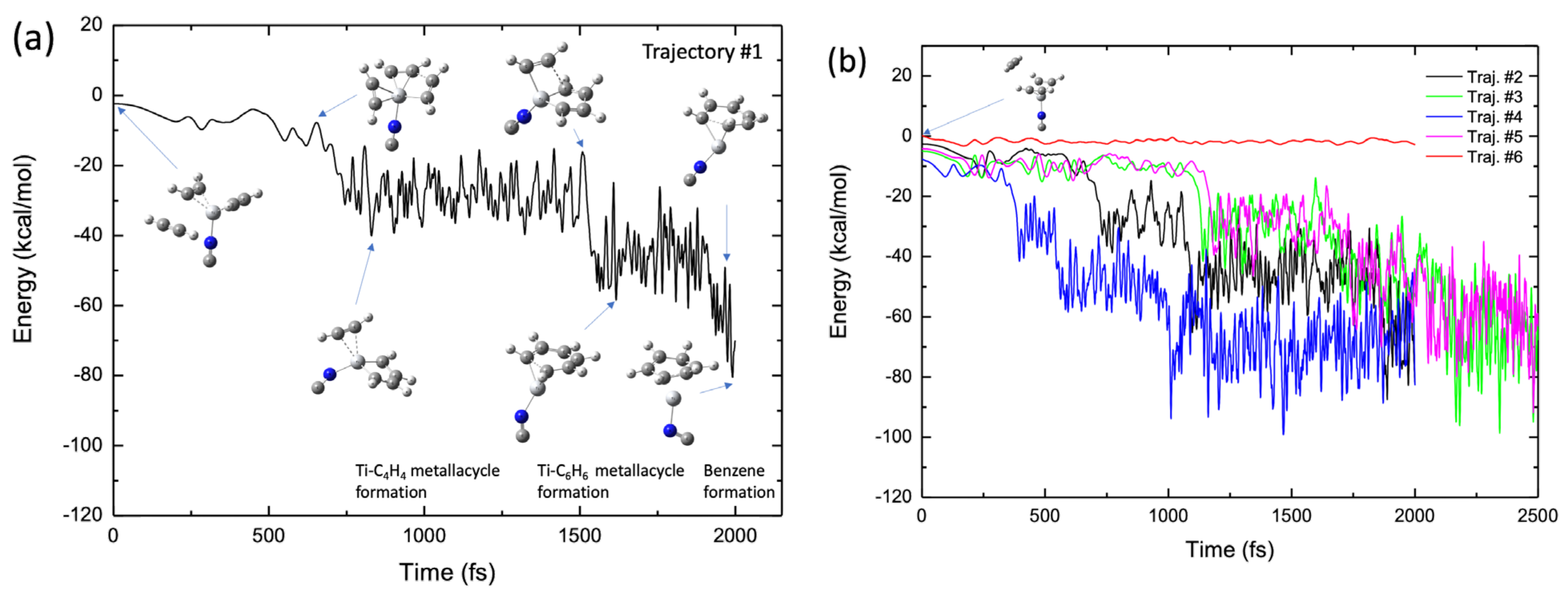
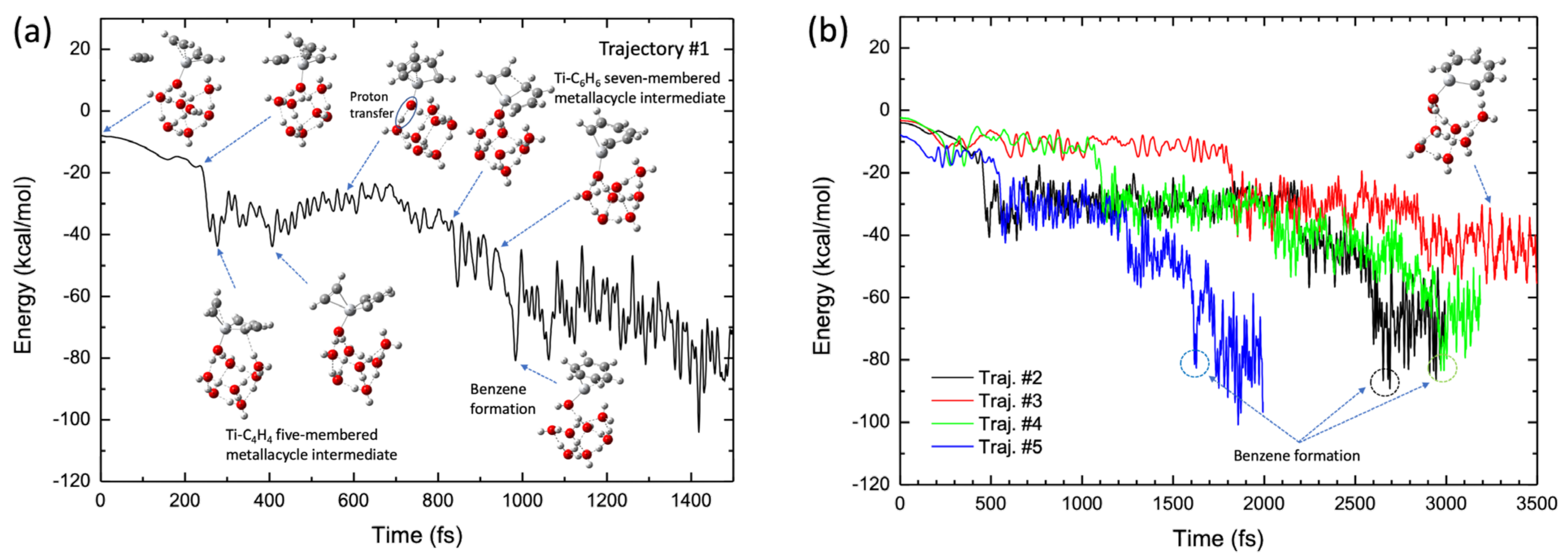
2.4. Molecular Dynamics Simulations
3. Conclusions and Future Directions
4. Computational Procedure
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tielens, A.G.G.M. Interstellar Polycyclic Aromatic Hydrocarbon Molecules. Annu. Rev. Astron. Astrophys. 2008, 46, 289–337. [Google Scholar] [CrossRef]
- Tielens, A.G.G.M. The Molecular Universe. Rev. Mod. Phys. 2013, 85, 1021–1081. [Google Scholar] [CrossRef]
- Parker, D.S.N.; Kaiser, R.I. On the Formation of Nitrogen-Substituted Polycyclic Aromatic Hydrocarbons (NPAHs) in Circumstellar and Interstellar Environments. Chem. Soc. Rev. 2017, 46, 452–463. [Google Scholar] [CrossRef] [PubMed]
- Allamandola, L.J.; Sandford, S.A.; Wopenka, B. Interstellar Polycyclic Aromatic Hydrocarbons and Carbon in Interplanetary Dust Partides and Meteorites. Science 1987, 237, 56–59. [Google Scholar] [CrossRef] [PubMed]
- Schlemmer, S.; Cook, D.J.; Harrison, J.A.; Wurfel, B.; Chapman, W.; Saykally, R.J. The Unidentified Interstellar Infrared Bands: PAHs as Carriers? Science 1994, 265, 1686–1689. [Google Scholar] [CrossRef]
- Engelbracht, C.W.; Kundurthy, P.; Gordon, K.D.; Rieke, G.H.; Kennicutt, R.C.; Smith, J.-D.T.; Regan, M.W.; Makovoz, D.; Sosey, M.; Draine, B.T.; et al. Extended Mid-Infrared Aromatic Feature Emission in M82. Astrophys. J. 2006, 642, L127–L132. [Google Scholar] [CrossRef]
- Duley, W.W. Polycyclic Aromatic Hydrocarbons, Carbon Nanoparticles and the Diffuse Interstellar Bands. Faraday Discuss. 2006, 133, 415–425. [Google Scholar] [CrossRef]
- Woods, P.M.; Millar, T.J.; Zijlstra, A.A.; Herbst, E. The Synthesis of Benzene in the Proto–Planetary Nebula CRL 618. Astrophys. J. 2002, 574, L167–L170. [Google Scholar] [CrossRef]
- Jones, B.M.; Zhang, F.; Kaiser, R.I.; Jamal, A.; Mebel, A.M.; Cordiner, M.A.; Charnley, S.B. Formation of Benzene in the Interstellar Medium. Proc. Natl. Acad. Sci. USA 2011, 108, 452–457. [Google Scholar] [CrossRef]
- Momoh, P.O.; Abrash, S.A.; Mabrouki, R.; El-Shall, M.S. Polymerization of Ionized Acetylene Clusters into Covalent Bonded Ions: Evidence for the Formation of Benzene Radical Cation. J. Am. Chem. Soc. 2006, 128, 12408–12409. [Google Scholar] [CrossRef]
- Momoh, P.O.; El-Shall, M.S. Stepwise Hydration of Ionized Acetylene Trimer. Further Evidence for the Formation of Benzene Radical Cation. Chem. Phys. Lett. 2007, 436, 25–29. [Google Scholar] [CrossRef]
- Zhao, T.Q.; Li, Q.; Liu, B.S.; Gover, R.K.E.; Sarre, P.J.; Cheung, A.S.-C. Laboratory Astrochemistry: Catalytic Conversion of Acetylene to Polycyclic Aromatic Hydrocarbons over SiC Grains. Phys. Chem. Chem. Phys. 2016, 18, 3489–3496. [Google Scholar] [CrossRef] [PubMed]
- Bach, R.D.; Wolber, G.J.; Schlegel, H.B. The Origin of the Barriers to Thermally Allowed, Six-Electron, Pericyclic Reactions: The Effect of Homo-Homo Interactions on the Trimerization of Acetylene. J. Am. Chem. Soc. 1985, 107, 2837–2841. [Google Scholar] [CrossRef]
- Houk, K.N.; Gandour, R.W.; Strozier, R.W.; Rondan, N.G.; Paquette, L.A. Barriers to Thermally Allowed Reactions and the Elusiveness of Neutral Homoaromaticity. J. Am. Chem. Soc. 1979, 101, 6797–6802. [Google Scholar] [CrossRef]
- Badger, G.M.; Lewis, G.E.; Napier, I.M. The Formation of Aromatic Hydrocarbons at High Temperatures. Part VIII. The Pyrolysis of Acetylene. J. Chem. Soc. 1960, 2825–2827. [Google Scholar] [CrossRef]
- Schore, N.E. Transition Metal-Mediated Cycloaddition Reactions of Alkynes in Organic Synthesis. Chem. Rev. 1988, 88, 1081–1119. [Google Scholar] [CrossRef]
- Lautens, M.; Klute, W.; Tam, W. Transition Metal-Mediated Cycloaddition Reactions. Chem. Rev. 1996, 96, 49–92. [Google Scholar] [CrossRef]
- Brathwaite, A.D.; Marks, J.H.; Webster, I.J.; Batchelor, A.G.; Ward, T.D.; Duncan, M.A. Coordination and Spin States in Fe+(C2H2)n Complexes Studied with Selected-Ion Infrared Spectroscopy. J. Phys. Chem. A 2022, 126, 9680–9690. [Google Scholar] [CrossRef]
- Walters, R.S.; Pillai, E.D.; Schleyer, P.V.R.; Duncan, M.A. Vibrational Spectroscopy and Structures of Ni+(C2H2)n (n = 1−4) Complexes. J. Am. Chem. Soc. 2005, 127, 17030–17042. [Google Scholar] [CrossRef]
- Walters, R.S.; Jaeger, T.D.; Duncan, M.A. Infrared Spectroscopy of Ni+(C2H2)n Complexes: Evidence for Intracluster Cyclization Reactions. J. Phys. Chem. A 2002, 106, 10482–10487. [Google Scholar] [CrossRef]
- Brathwaite, A.D.; Ward, T.B.; Walters, R.S.; Duncan, M.A. Cation−π and CH−π Interactions in the Coordination and Solvation of Cu+(Acetylene)n Complexes. J. Phys. Chem. A 2015, 119, 5658–5667. [Google Scholar] [CrossRef] [PubMed]
- Brathwaite, A.D.; Ward, T.B.; Marks, J.H.; Duncan, M.A. Coordination and Solvation in Gas-Phase Ag+(C2H2)n Complexes Studied with Selected-Ion Infrared Spectroscopy. J. Phys. Chem. A 2020, 124, 8562–8573. [Google Scholar] [CrossRef] [PubMed]
- Ward, T.B.; Brathwaite, A.D.; Duncan, M.A. Infrared Spectroscopy of Au(Acetylene)n + Complexes in the Gas Phase. Top Catal. 2018, 61, 49–61. [Google Scholar] [CrossRef]
- Marks, J.H.; Ward, T.B.; Brathwaite, A.D.; Duncan, M.A. Infrared Spectroscopy of Zn(Acetylene)n+ Complexes: Ligand Activation and Nascent Polymerization. J. Phys. Chem. A 2020, 124, 4764–4776. [Google Scholar] [CrossRef] [PubMed]
- Marks, J.H.; Ward, T.B.; Brathwaite, A.D.; Ferguson, S.; Duncan, M.A. Cyclotrimerization of Acetylene in Gas Phase V+(C2H2)n Complexes: Detection of Intermediates and Products with Infrared Spectroscopy. J. Phys. Chem. A 2019, 123, 6733–6743. [Google Scholar] [CrossRef]
- Murakami, T.; Takayanagi, T. Interstellar Benzene Formation Mechanisms via Acetylene Cyclotrimerization Catalyzed by Fe+ Attached to Water Ice Clusters: Quantum Chemistry Calculation Study. Molecules 2022, 27, 7767. [Google Scholar] [CrossRef]
- Murakami, T.; Matsumoto, N.; Takayanagi, T.; Fujihara, T. The Importance of Nuclear Dynamics in Reaction Mechanisms of Acetylene Cyclotrimerization Catalyzed by Fe+-Compounds. J. Organomet. Chem. 2023, 987–988, 122643. [Google Scholar] [CrossRef]
- Takayanagi, T. Two-State Reactivity in the Acetylene Cyclotrimerization Reaction Catalyzed by a Single Atomic Transition-Metal Ion: The Case for V+ and Fe+. Comput. Theor. Chem. 2022, 1211, 113682. [Google Scholar] [CrossRef]
- Mattila, S.; Lundqvist, P.; Gröningsson, P.; Meikle, P.; Stathakis, R.; Fransson, C.; Cannon, R. Abundances and Density Structure of the Inner Circumstellar Ring Around SN 1987A. Astrophys. J. 2010, 717, 1140–1156. [Google Scholar] [CrossRef]
- Fioroni, M. Astrochemistry of Transition Metals? The Selected Cases of [FeN]+, [FeNH]+ and [(CO)2FeN]+: Pathways toward CH3NH2 and HNCO. Phys. Chem. Chem. Phys. 2014, 16, 24312–24322. [Google Scholar] [CrossRef]
- Fioroni, M. Astro-Organometallics of Fe, Co, Ni: Stability, IR Fingerprints and Possible Locations. Comput. Theor. Chem. 2016, 1084, 196–212. [Google Scholar] [CrossRef]
- Mauron, N.; Huggins, P.J. Gas Phase Atomic Metals in the Circumstellar Envelope of IRC+10216. Astron. Astrophys. 2010, 513, A31. [Google Scholar] [CrossRef]
- Kamiński, T.; Gottlieb, C.A.; Menten, K.M.; Patel, N.A.; Young, K.H.; Brünken, S.; Müller, H.S.P.; McCarthy, M.C.; Winters, J.M.; Decin, L. Pure Rotational Spectra of TiO and TiO2 in VY Canis Majoris. Astron. Astrophys. 2013, 551, A113. [Google Scholar] [CrossRef]
- Snow, T.P., Jr.; Weiler, W.J.; Oegerle, W.R. New Observations of Interstellar Abundances and Depletions of Boron, Vanadium, Chromium, and Cobalt. Astrophys. J. 1979, 234, 506–512. [Google Scholar] [CrossRef]
- Federman, S.R.; Sheffer, Y.; Lambert, D.L.; Gilliland, R.L. Detection of Boron, Cobalt, and Other Weak Interstellar Lines toward ζ Ophiuchi. Astrophys. J. 1993, 413, L51–L54. [Google Scholar] [CrossRef]
- Hui-Bon-Hoa, A.; Alecian, G.; LeBlanc, F. Modelling of the Scandium Abundance Evolution in AmFm Stars. Astron. Astrophys. 2022, 668, A6. [Google Scholar] [CrossRef]
- Jaschek, M.; Jaschek, C. A Search for Families among Metallic-Line Stars. Z. Astrophys. 1960, 50, 155. [Google Scholar]
- Zack, L.N.; Halfen, D.T.; Ziurys, L.M. Detection of FeCN(X4Δi) in IRC+10216: A new interstellar molecule. Astrophys. J. 2011, 733, L36. [Google Scholar] [CrossRef]
- Guelin, M.; Lucas, R.; Cernicharo, J. MgNC and the Carbon-Chain Radicals in IRC +10216. Astron. Astrophys. 1993, 280, L19–L22. [Google Scholar]
- Kawaguchi, K.; Kagi, E.; Hirano, T.; Takano, S.; Saito, S. Laboratory Spectroscopy of MgNC: The First Radioastronomical Identification of Mg-Bearing Molecule. Astrophys. J. 1993, 406, L39–L42. [Google Scholar] [CrossRef]
- Ziurys, L.M.; Apponi, A.J.; Guelin, M.; Cernicharo, J. Detection of MgCN in IRC +10216: A New Metal-Bearing Free Radical. Astrophys. J. 1995, 445, L47–L50. [Google Scholar] [CrossRef]
- Hama, T.; Watanabe, N. Surface Processes on Interstellar Amorphous Solid Water: Adsorption, Diffusion, Tunneling Reactions, and Nuclear-Spin Conversion. Chem. Rev. 2013, 113, 8783–8839. [Google Scholar] [CrossRef]
- Woon, D.E. Icy Grain Mantle Surface Astrochemistry of MgNC: The Emergence of Metal Ion Catalysis Studied via Model Ice Cluster Calculations. J. Phys. Chem. A 2022, 126, 5186–5194. [Google Scholar] [CrossRef]
- Spezia, R.; Martínez-Nuñez, E.; Vazquez, S.; Hase, W.L. Theoretical and Computational Studies of Non-Equilibrium and Non-Statistical Dynamics in the Gas Phase, in the Condensed Phase and at Interfaces. Phil. Trans. R. Soc. A 2017, 375, 20170035. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Hase, W.L. Perspective: Chemical Dynamics Simulations of Non-Statistical Reaction Dynamics. Phil. Trans. R. Soc. A 2017, 375, 20160204. [Google Scholar] [CrossRef]
- Puzzarini, C. Gas-Phase Chemistry in the Interstellar Medium: The Role of Laboratory Astrochemistry. Front. Astron. Space Sci. 2022, 8, 811342. [Google Scholar] [CrossRef]
- Thripati, S.; Gautam, R.; Ramabhadran, R.O. Possible Role of Metal-Ions in the Chemistry of Prochirality and the Origin of Chirality in the Interstellar Medium. ACS Earth Space Chem. 2023, 7, 77–91. [Google Scholar] [CrossRef]
- Frenking, G.; Fröhlich, N. The Nature of the Bonding in Transition-Metal Compounds. Chem. Rev. 2000, 100, 717–774. [Google Scholar] [CrossRef]
- Albright, T.A.; Hoffmann, R.; Thibeault, J.C.; Thorn, D.L. Ethylene Complexes. Bonding, Rotational Barriers, and Conformational Preferences. J. Am. Chem. Soc. 1979, 101, 3801–3812. [Google Scholar] [CrossRef]
- Triguero, L.; Pettersson, L.G.M.; Minaev, B.; Ågren, H. Spin Uncoupling in Surface Chemisorption of Unsaturated Hydrocarbons. J. Chem. Phys. 1998, 108, 1193–1205. [Google Scholar] [CrossRef]
- Hedström, S.; Dos Santos, E.C.; Liu, C.; Chan, K.; Abild-Pedersen, F.; Pettersson, L.G.M. Spin Uncoupling in Chemisorbed OCCO and CO2: Two High-Energy Intermediates in Catalytic CO2 Reduction. J. Phys. Chem. C 2018, 122, 12251–12258. [Google Scholar] [CrossRef]
- Bondarchuk, S.V.; Minaev, B.F. Thermally Accessible Triplet State of π-Nucleophiles Does Exist. Evidence from First Principles Study of Ethylene Interaction with Copper Species. RSC Adv. 2015, 5, 11558–11569. [Google Scholar] [CrossRef]
- Linares, M.; Braida, B.; Humbel, S. Valence Bond Approach of Metal−Ligand Bonding in the Dewar−Chatt−Duncanson Model. Inorg. Chem. 2007, 46, 11390–11396. [Google Scholar] [CrossRef]
- Upton, T.H.; Rappe, A.K. A Theoretical Basis for Low Barriers in Transition-Metal Complex 2π + 2π Reactions: The Isomerization of the Dicyclopentadienyltitanium Complex Cp2TiC3H6 to Cp2TiCH2(C2H4). J. Am. Chem. Soc. 1985, 107, 1206–1218. [Google Scholar] [CrossRef]
- Knight, C.; Voth, G.A. The Curious Case of the Hydrated Proton. Acc. Chem. Res. 2012, 45, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Ruiz, J.; Ugliengo, P.; Sodupe, M.; Rimola, A. Does Fe2+ in Olivine-Based Interstellar Grains Play Any Role in the Formation of H2 ? Atomistic Insights from DFT Periodic Simulations. Chem. Commun. 2016, 52, 6873–6876. [Google Scholar] [CrossRef]
- Yang, F.; Armentrout, P.B. Periodic Trends in the Hydration Energies and Critical Sizes of Alkaline Earth and Transition Metal Dication Water Complexes. Mass Spectrom. Rev. 2023, e21830. [Google Scholar] [CrossRef]
- Liszt, H.; Lucas, R. Comparative Chemistry of Diffuse Clouds: II. CN, HCN, HNC, CH3CN & N2H+. Astron. Astrophys. 2001, 370, 576–585. [Google Scholar] [CrossRef]
- Grimme, S.; Ehrlich, S.; Goerigk, L. Effect of the Damping Function in Dispersion Corrected Density Functional Theory. J. Comput. Chem. 2011, 32, 1456–1465. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Cheeseman, J.R.; Scalmani, G.; Caricato, M.; Hratchian, H.P.; Li, X.; Barone, V.; Bloino, J.; Zheng, G.; et al. Gaussian 09, D.01; Gaussian Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Maeda, S.; Ohno, K. Global Mapping of Equilibrium and Transition Structures on Potential Energy Surfaces by the Scaled Hypersphere Search Method: Applications to Ab Initio Surfaces of Formaldehyde and Propyne Molecules. J. Phys. Chem. A 2005, 109, 5742–5753. [Google Scholar] [CrossRef]
- Ohno, K.; Maeda, S. A Scaled Hypersphere Search Method for the Topography of Reaction Pathways on the Potential Energy Surface. Chem. Phys. Lett. 2004, 384, 277–282. [Google Scholar] [CrossRef]
- Maeda, S.; Ohno, K. GRRM 11, a Program Package. Available online: https://iqce.jp/GRRM/index_e.shtml (accessed on 29 September 2021).
- Krause, C.; Werner, H.-J. Scalable Electron Correlation Methods. 6. Local Spin-Restricted Open-Shell Second-Order Møller–Plesset Perturbation Theory Using Pair Natural Orbitals: PNO-RMP2. J. Chem. Theory Comput. 2019, 15, 987–1005. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Werner, H.-J. Scalable Electron Correlation Methods. 7. Local Open-Shell Coupled-Cluster Methods Using Pair Natural Orbitals: PNO-RCCSD and PNO-UCCSD. J. Chem. Theory Comput. 2020, 16, 3135–3151. [Google Scholar] [CrossRef] [PubMed]
- Werner, H.-J.; Knowles, P.J.; Celani, P.; Györffy, W.; Hesselmann, A.; Kats, D.; Knizia, G.; Köhn, A.; Korona, T.; Kreplin, D.; et al. MOLPRO, Version 2022.2, a Package of Ab Initio Programs. Available online: https://www.molpro.net (accessed on 2 November 2023).
- Győrffy, W.; Shiozaki, T.; Knizia, G.; Werner, H.-J. Analytical Energy Gradients for Second-Order Multireference Perturbation Theory Using Density Fitting. J. Chem. Phys. 2013, 138, 104104. [Google Scholar] [CrossRef] [PubMed]
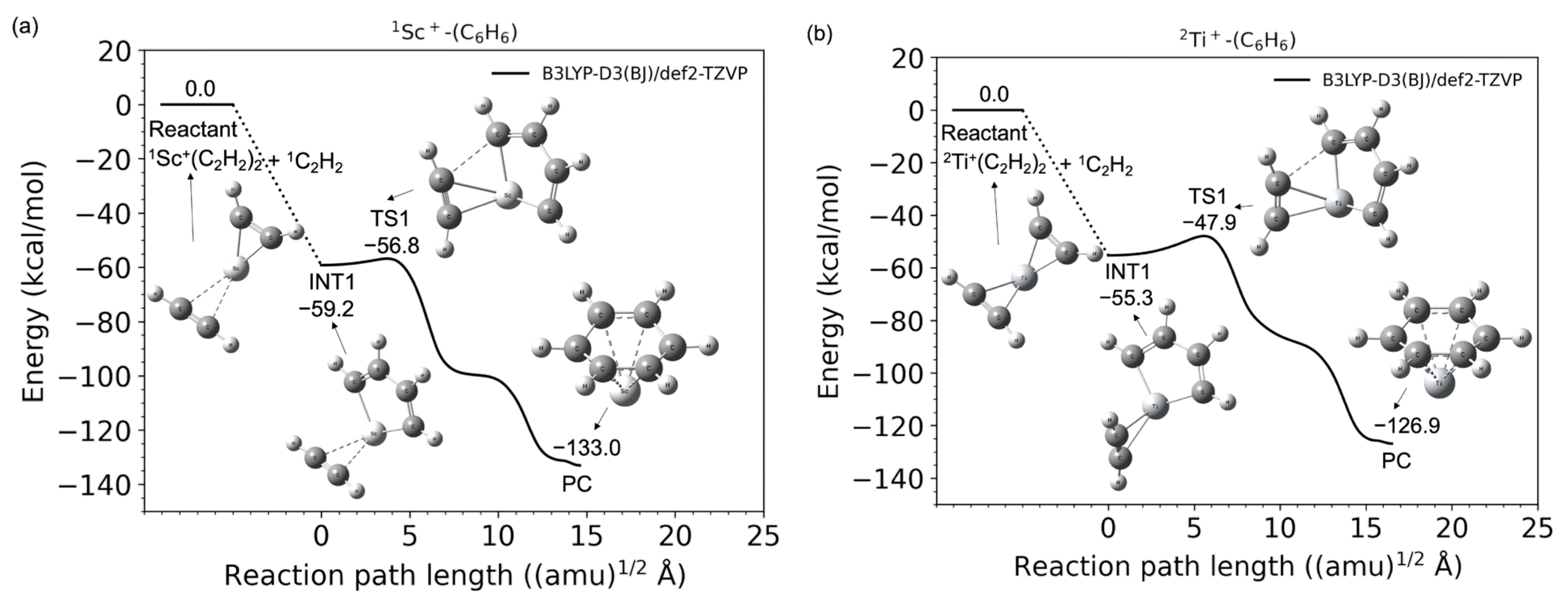


| Reactant | INT1 | TS1 | PC | Product | |
|---|---|---|---|---|---|
| Sc+-C6H6 | |||||
| Quintet | 130.5 | 80.3 | 79.5 | −24.9 | 549.9 |
| Triplet | 36.6 | 5.3 | 6.8 | −118.7 | −79.5 |
| Singlet | 0.0 | −59.2 | −56.8 | −133.0 | −41.8 |
| Ti+-C6H6 | |||||
| Sextet | 151.3 | 81.4 | 79.2 | −12.5 | 626.1 |
| Quartet | 40.6 | 2.8 | 5.0 | −90.5 | −74.3 |
| Doublet | 0.0 | −55.3 | −47.9 | −126.9 | −55.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Murakami, T.; Matsumoto, N.; Fujihara, T.; Takayanagi, T. Possible Roles of Transition Metal Cations in the Formation of Interstellar Benzene via Catalytic Acetylene Cyclotrimerization. Molecules 2023, 28, 7454. https://doi.org/10.3390/molecules28217454
Murakami T, Matsumoto N, Fujihara T, Takayanagi T. Possible Roles of Transition Metal Cations in the Formation of Interstellar Benzene via Catalytic Acetylene Cyclotrimerization. Molecules. 2023; 28(21):7454. https://doi.org/10.3390/molecules28217454
Chicago/Turabian StyleMurakami, Tatsuhiro, Naoki Matsumoto, Takashi Fujihara, and Toshiyuki Takayanagi. 2023. "Possible Roles of Transition Metal Cations in the Formation of Interstellar Benzene via Catalytic Acetylene Cyclotrimerization" Molecules 28, no. 21: 7454. https://doi.org/10.3390/molecules28217454
APA StyleMurakami, T., Matsumoto, N., Fujihara, T., & Takayanagi, T. (2023). Possible Roles of Transition Metal Cations in the Formation of Interstellar Benzene via Catalytic Acetylene Cyclotrimerization. Molecules, 28(21), 7454. https://doi.org/10.3390/molecules28217454