Molecular Simulation of Coal Molecular Diffusion Properties in Chicheng Coal Mine
Abstract
:1. Introduction
2. Results and Discussion
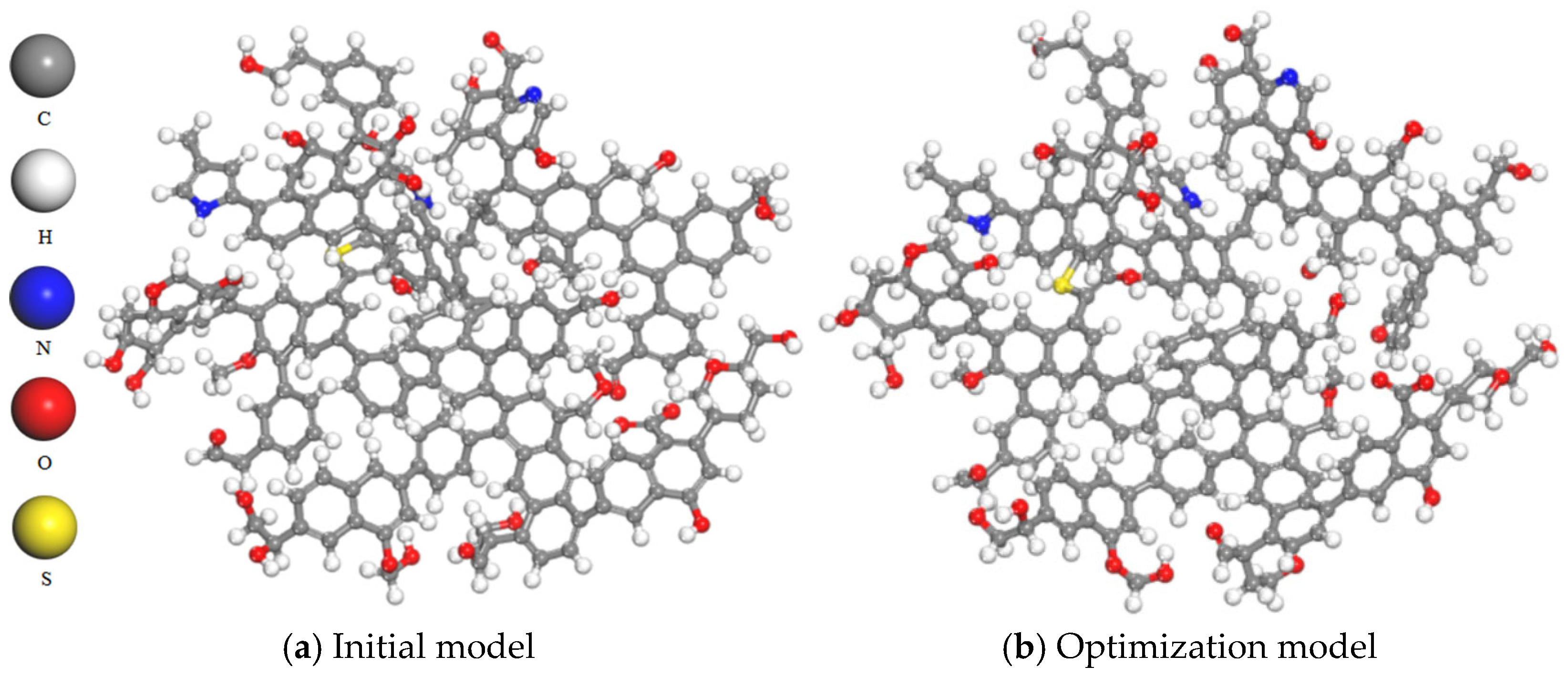
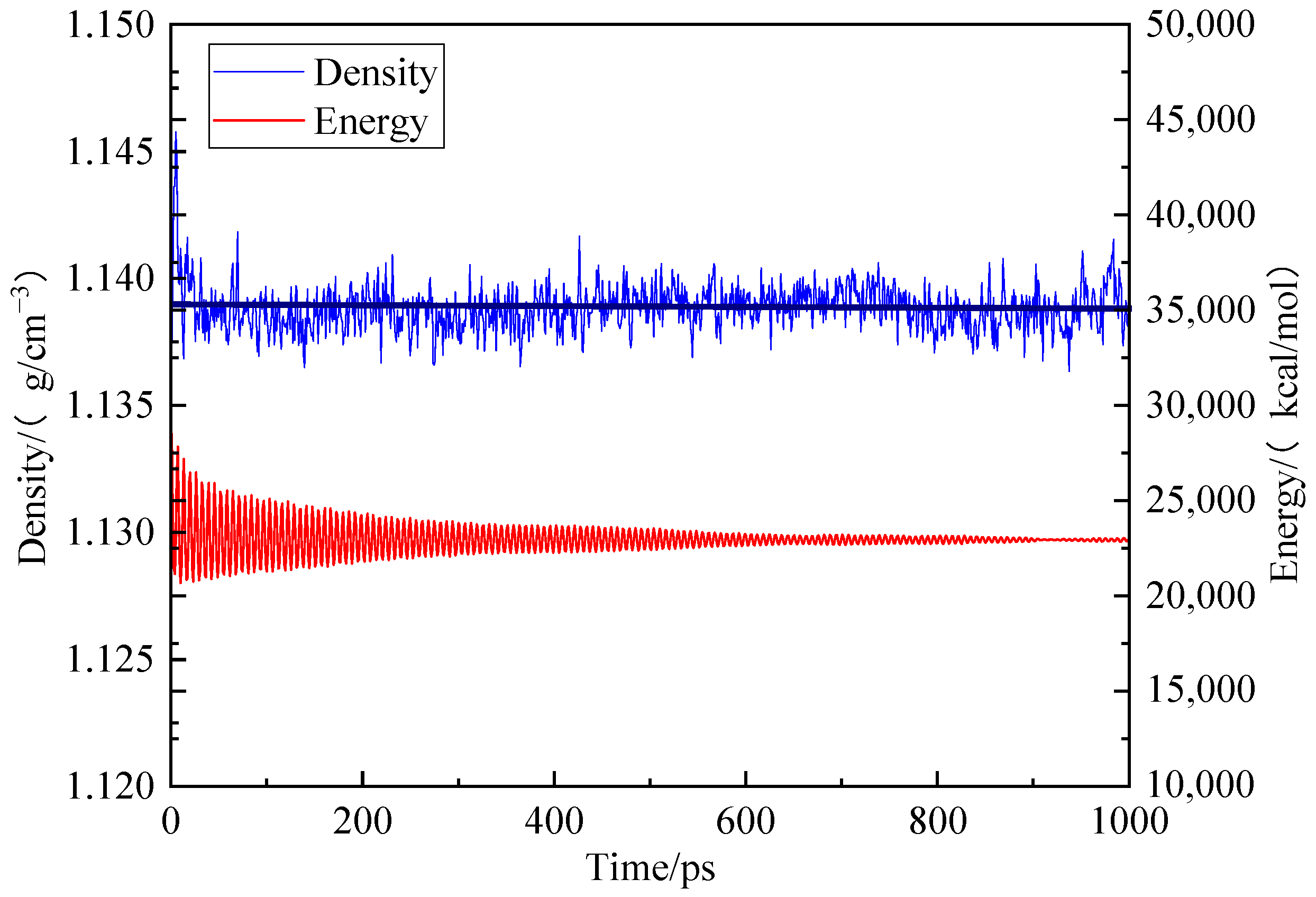
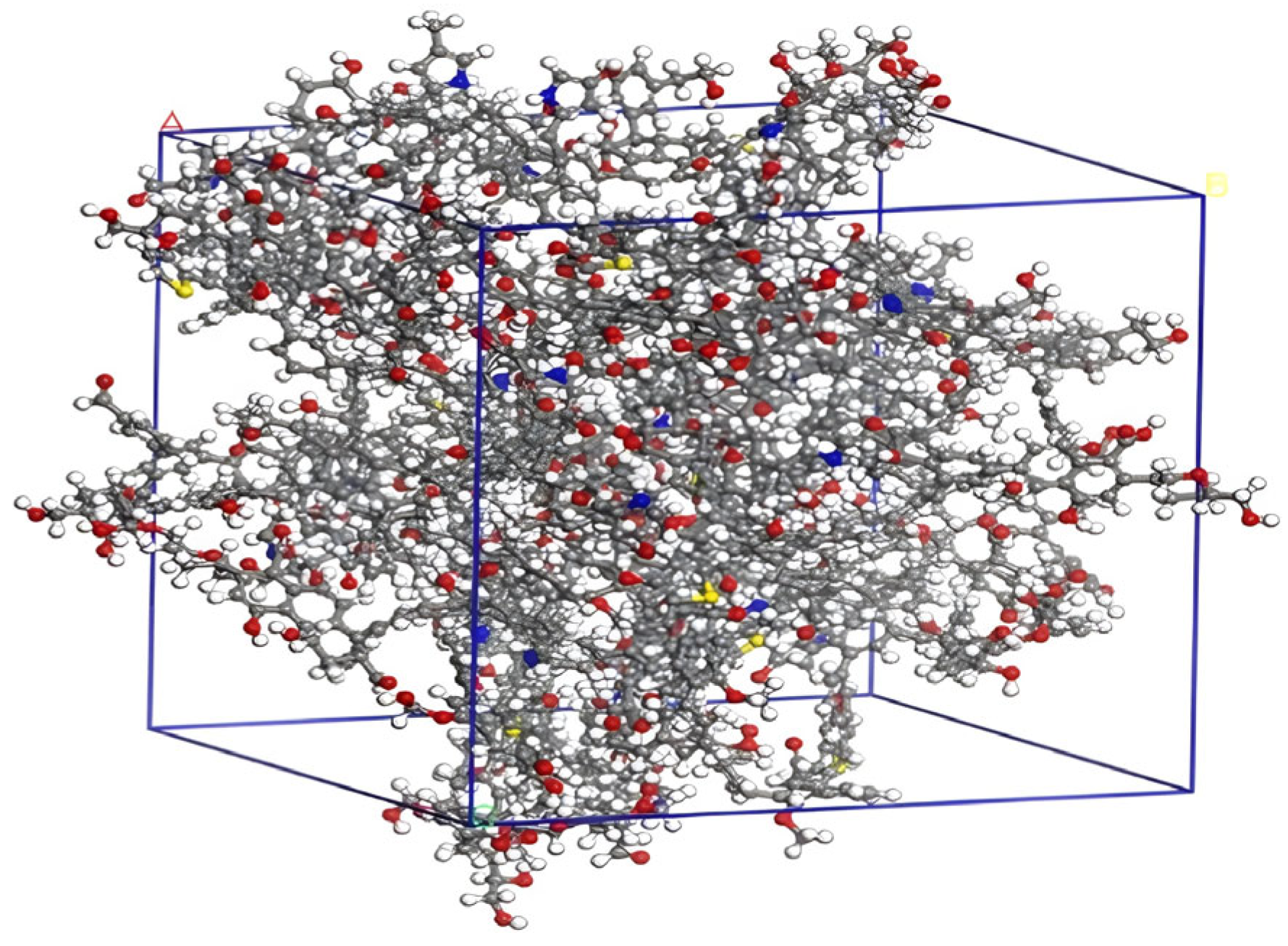
2.1. Structural Characterization and Construction of Macromolecular Structure of Coal
2.2. Theoretical Formula of Gas Diffusion Characteristics of Coal
3. Materials and Methods
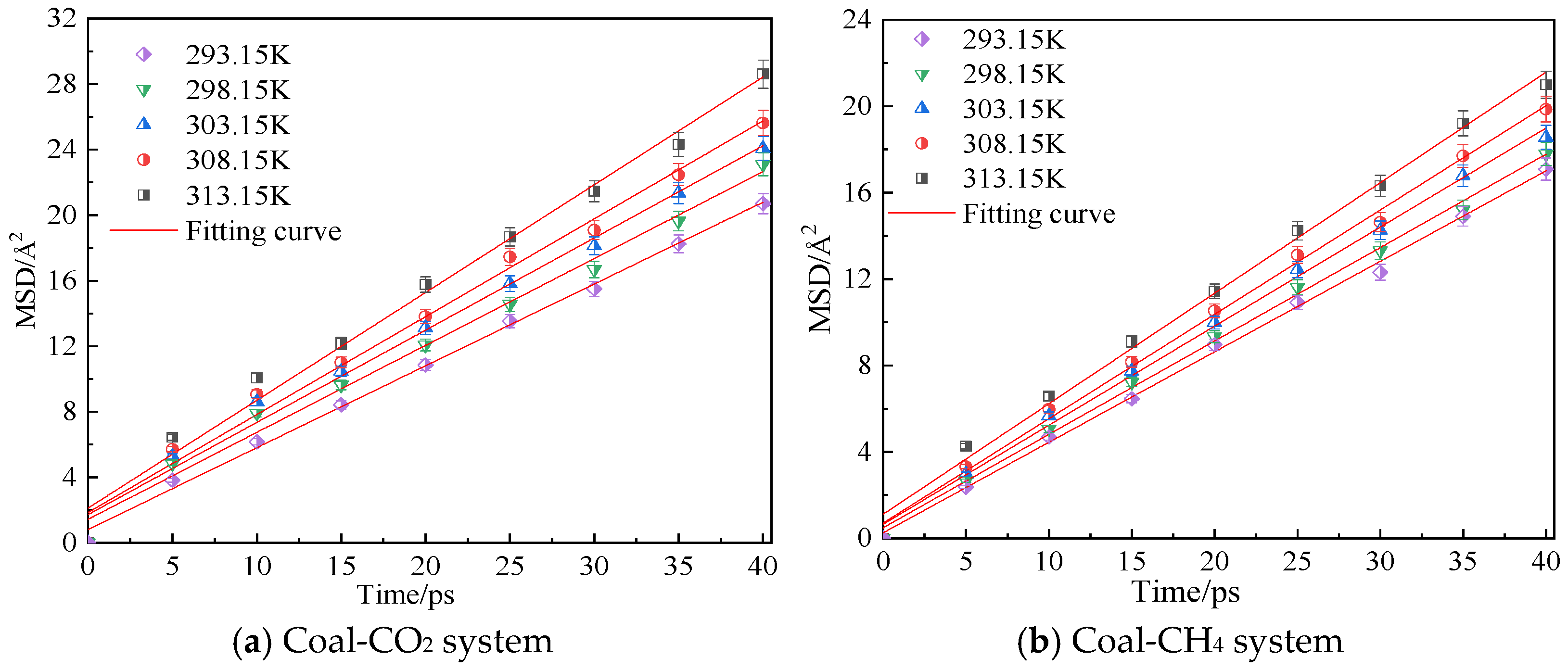
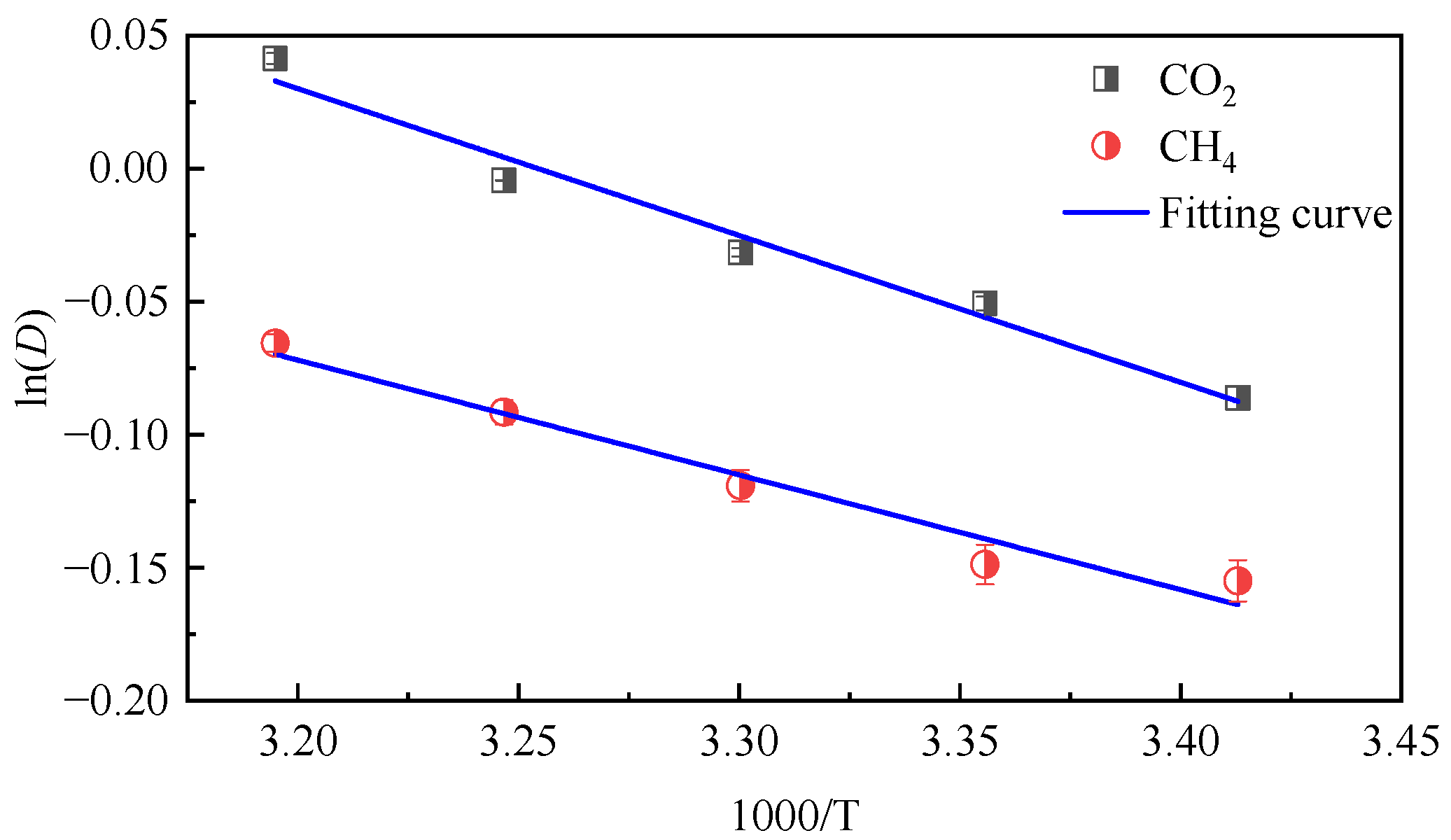
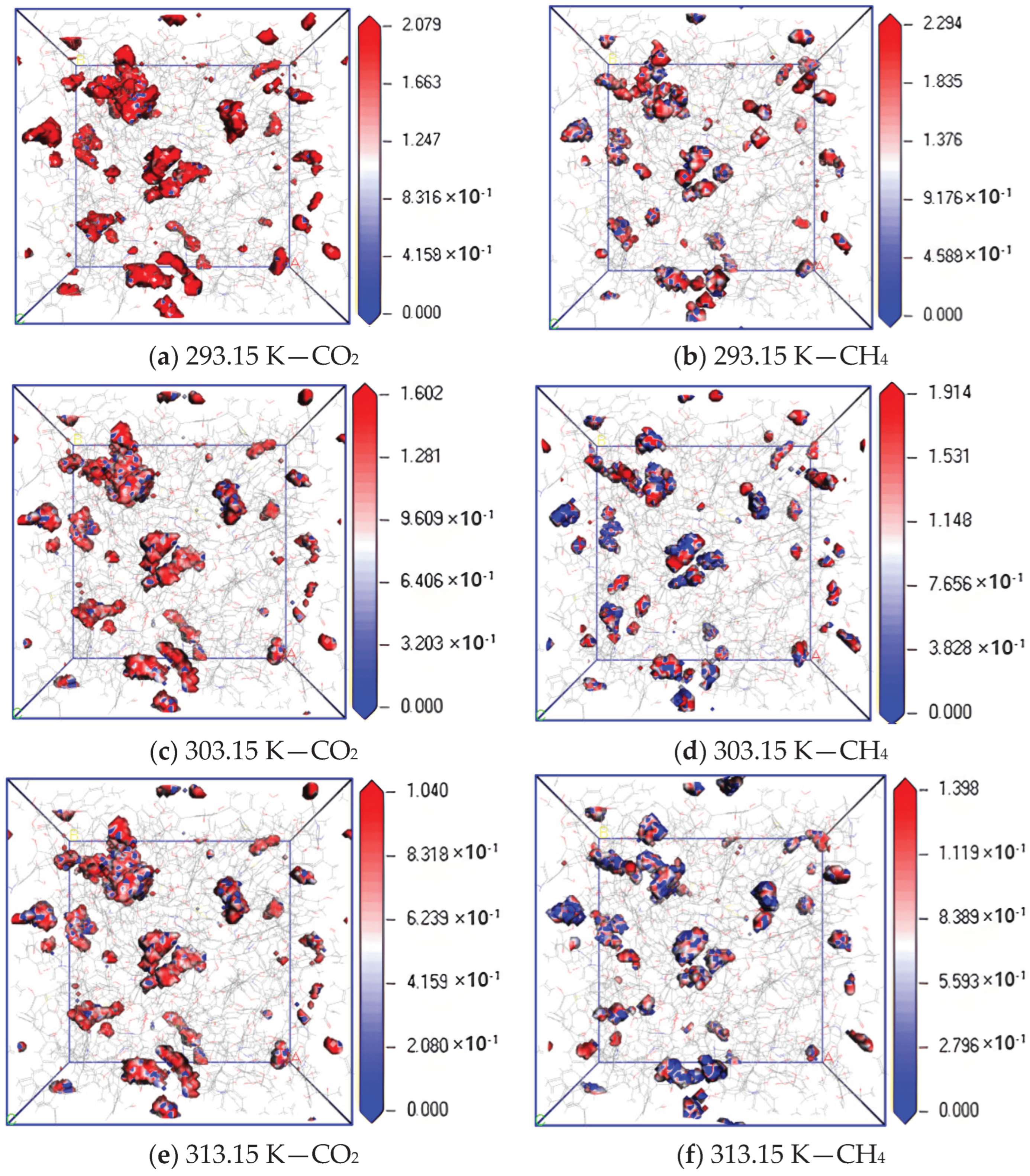
3.1. Influence of Temperature on Diffusion Performance
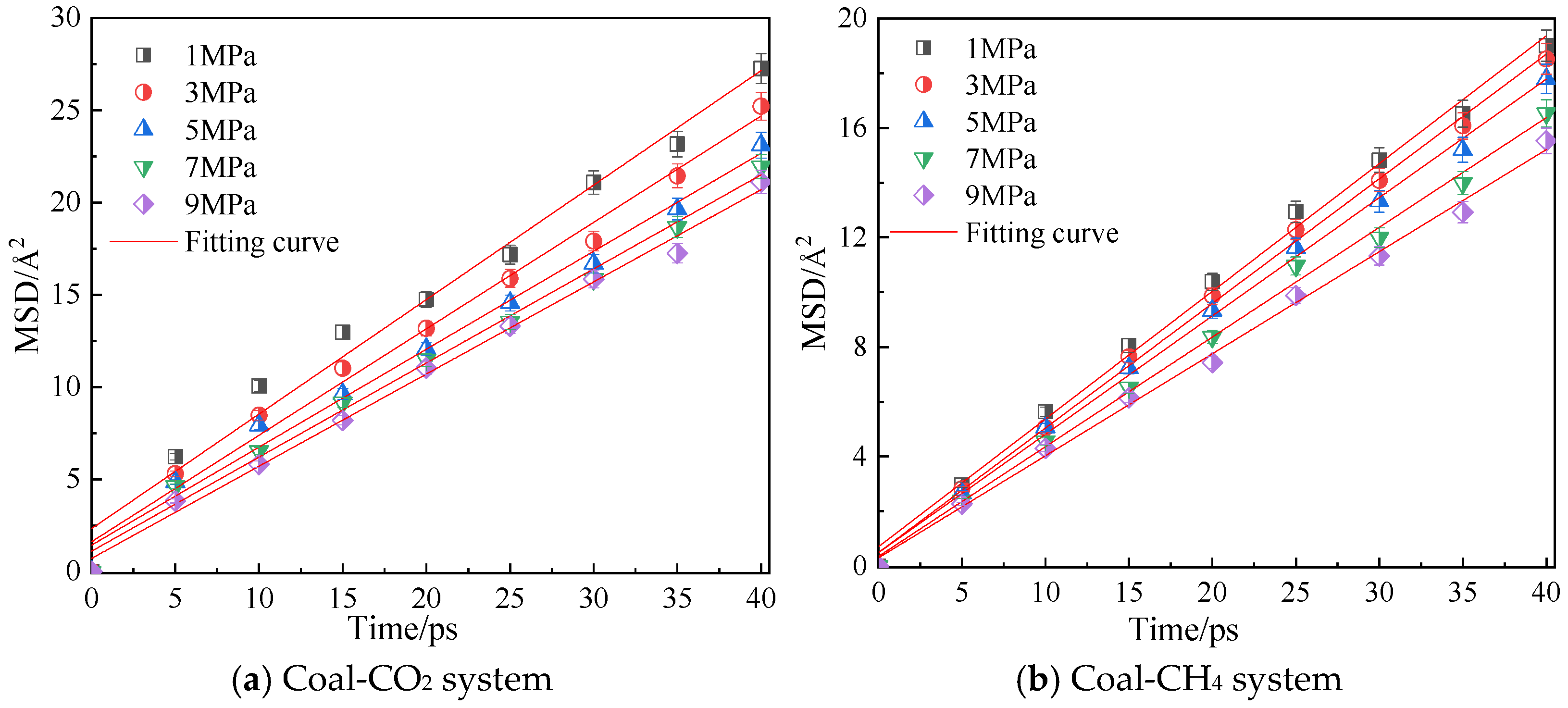
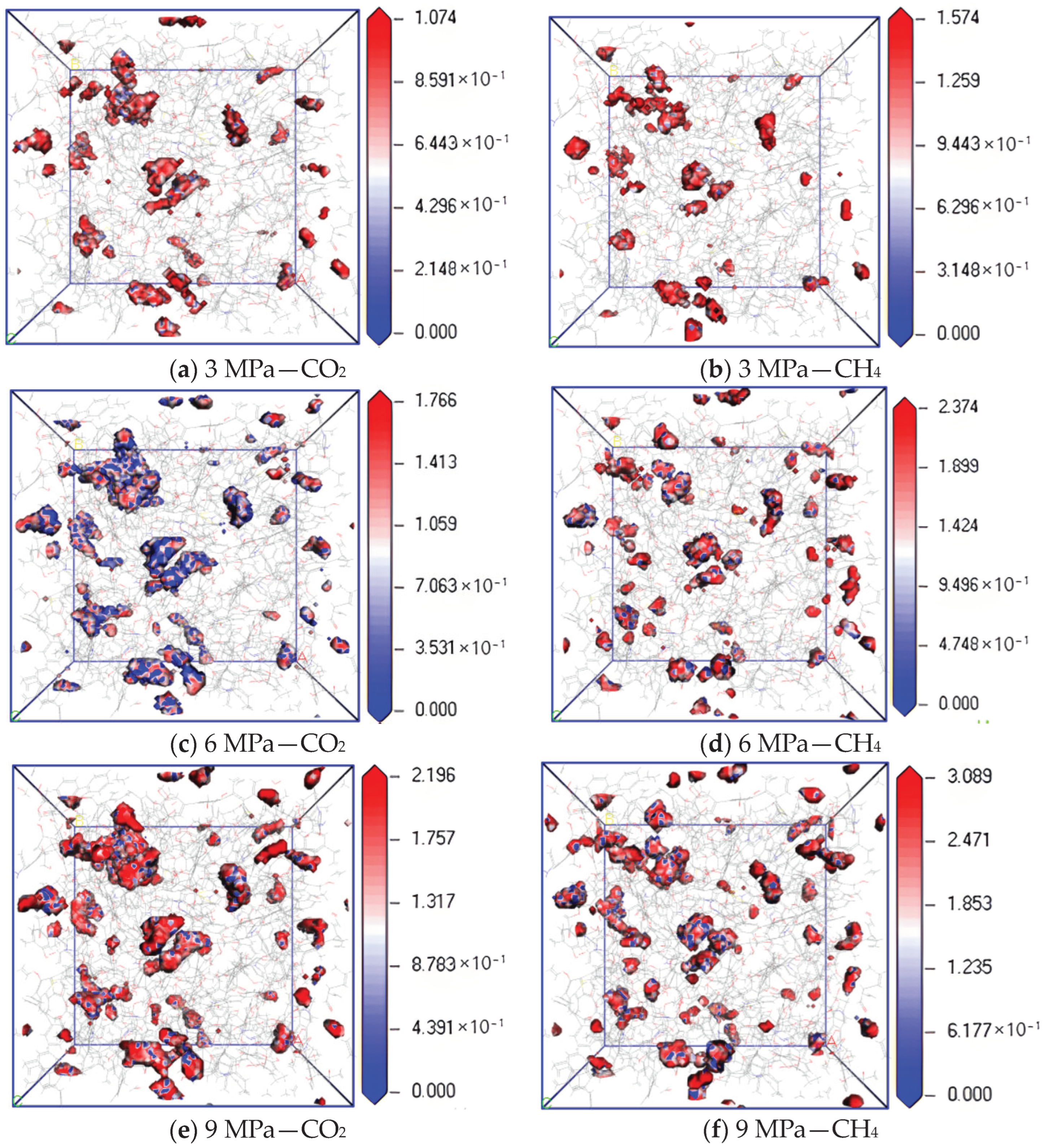
3.2. Influence of Pressure on Diffusion Performance
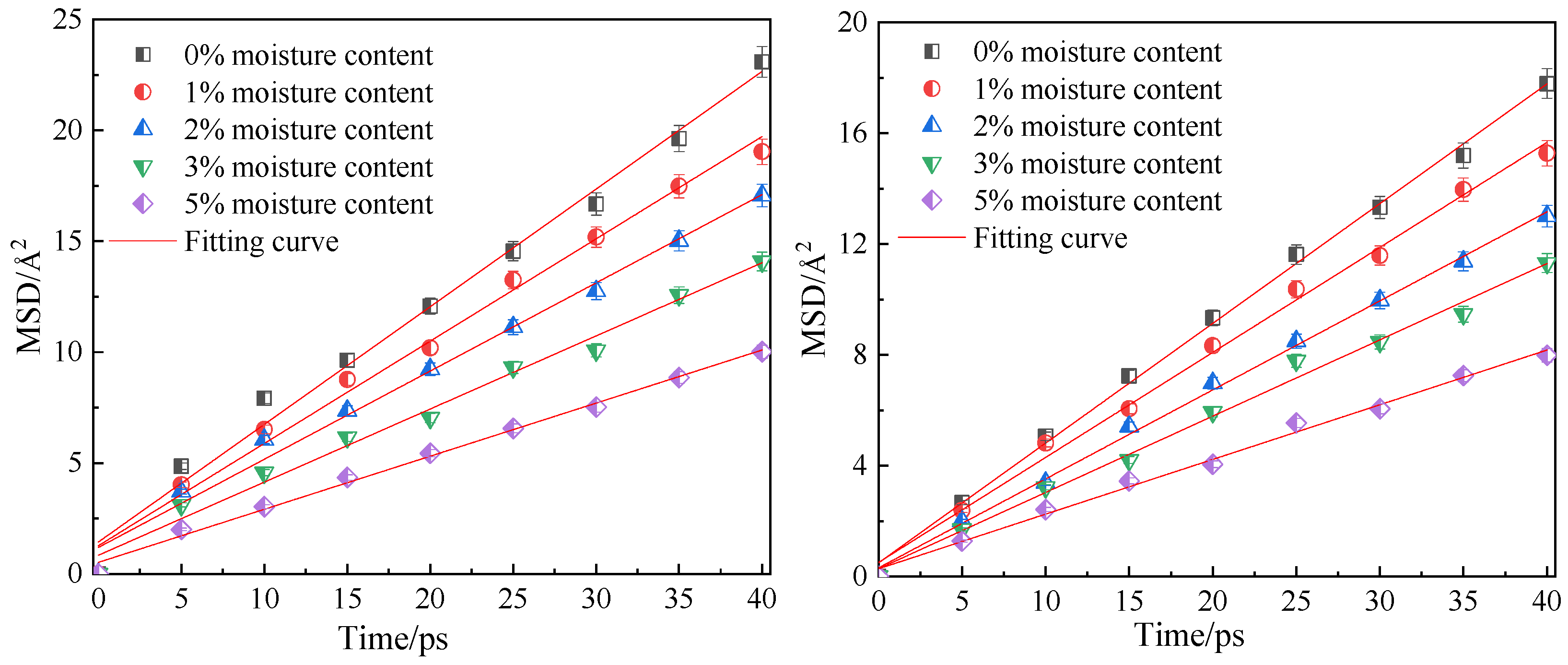
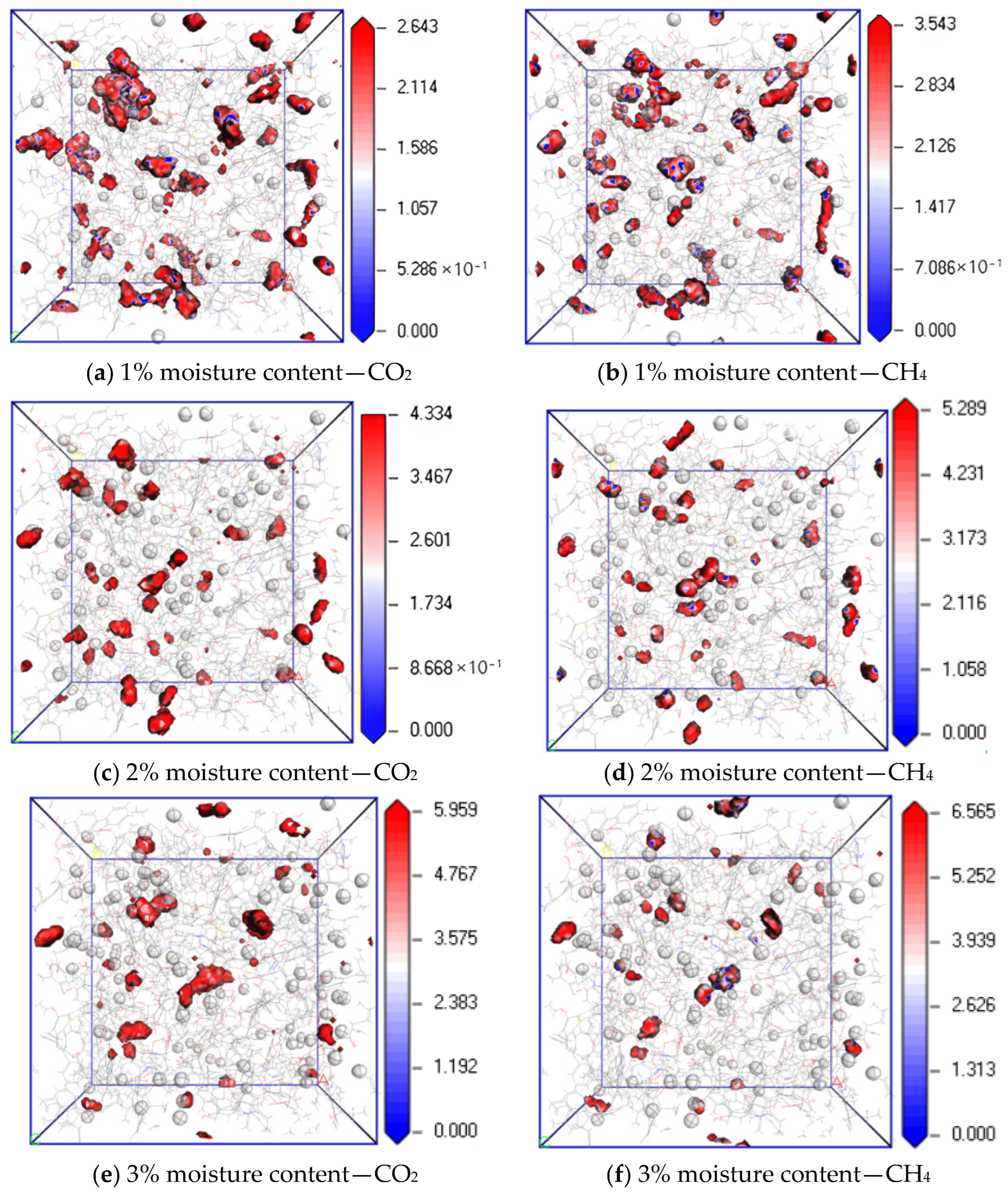
3.3. Influence of Moisture Content on Diffusion Performance
4. Conclusions
- (1)
- In the dry-mode macromolecular model, the diffusion coefficients of CO2 and CH4 gradually increase with an increase in temperature, and a high temperature is conducive to gas diffusion. Under the same conditions, the diffusion coefficient of CH4 is lower than that of CO2, and the diffusion activation energy of CO2 is 4.57 kJ·mol−1, while that of CH4 is 3.56 kJ·mol−1.
- (2)
- In the dry-mode macromolecular model, with an increase in pressure, the diffusion coefficients of CO2 and CH4 gradually decrease, and the equipotential values of CO2 and CH4 gradually increase, and a high pressure is not conducive to the diffusion of gas in the coal macromolecular model.
- (3)
- In the water-containing coal macromolecular model, with an increase in water content, the diffusion coefficients of CO2 and CH4 significantly decrease, and the equipotential values of CO2 and CH4 gradually increase, and water is not conducive to the diffusion of gas in the coal macromolecular model.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wang, D. Study on Adsorption and Diffusion Behavior of Soft and Hard Coal Gas Based on Molecular Simulation. Master’s Thesis, Henan Polytechnic University, Jiaozuo, China, January 2020. [Google Scholar]
- Zhang, K. Molecular Simulation of Methane Desorption and Diffusion during Coalbed Methane Drainage and Production. Master’s Thesis, North University of China, Taiyuan, China, 2020. [Google Scholar]
- Clarkson, C.R.; Bustin, R.M.J.F. The effect of pore structure and gas pressure upon the transport properties of coal: A laboratory and modeling study. 2. Adsorption rate modeling. Fuel 1999, 78, 1345–1362. [Google Scholar] [CrossRef]
- Li, B. Study on Adsorption-Deformation-Percolation-Diffusion Characteristics of CO2/CH4/N2 in Coal of Different Rank. Ph.D. Thesis, Liaoning Technical University, Fuxin, China, May 2022. [Google Scholar]
- Zhang, M.; Wei, C.H.; Liu, S.Y.; Liu, Z.Y.; Zhang, Z. Experiment and model study on CO2 adsorption-desorption diffusion in anthracite particles. Exp. Mech. 2021, 36, 753–761. [Google Scholar]
- Dai, X. Adsorption and Diffusion Characteristics of CH4/CO2 in Shale Minerals and Geological Risk Assessment of Shale Gas Exploitation. Master’s Thesis, Chongqing University, Chongqing, China, June 2022. [Google Scholar]
- Liu, J.; Liu, H.; Zhang, K.; Xue, S. Diffusion characteristics of CO2 and CH4 in CO2-ECBM process of low permeability coal seam. Coal Sci. Technol. 2022, 26, 1–11. [Google Scholar] [CrossRef]
- Li, K. Study on the Diffusion and Adsorption Characteristics of CO2 in Gangue Filling Materials in Mined-Out Area of Coal Mine. Master’s Thesis, China University of Mining and Technology, Xuzhou, China, June 2021. [Google Scholar]
- You, X.; He, M.; Cao, X.; Lyu, X.; Li, L. Structure and dynamics of water adsorbed on the lignite surface: Molecular dynamics simulation. Physicochem. Probl. Miner. Process. 2019, 55, 10–20. [Google Scholar]
- Hu, H.; Li, X.; Fang, Z.; Wei, N.; Li, Q. Small-molecule gas sorption and diffusion in coal: Molecular simulation. Energy 2010, 35, 2939–2944. [Google Scholar] [CrossRef]
- An, F.; Jia, H.; Feng, Y. Effect of stress, concentration and temperature on gas diffusion coefficient of coal measured through a direct method and its model application. Fuel 2022, 312, 122991. [Google Scholar] [CrossRef]
- Xu, H.; Tang, D.; Zhao, J.; Li, S.; Tao, S. A new laboratory method for accurate measurement of the methane diffusion coefficient and its influencing factors in the coal matrix. Fuel 2015, 158, 239–247. [Google Scholar] [CrossRef]
- Yu, S.; Bo, J.; Meijun, Q. Molecular Dynamic Simulation of Self- and Transport Diffusion for CO2/CH4/N2 in Low-Rank Coal Vitrinite. Fuels 2018, 32, 3085–3096. [Google Scholar] [CrossRef]
- Liu, T.; Lin, B.; Fu, X.; Gao, Y.; Kong, J.; Zhao, Y.; Song, H. Experimental study on gas diffusion dynamics in fractured coal: A better understanding of gas migration in in-situ coal seam. Energy 2020, 195, 10. [Google Scholar] [CrossRef]
- Keshavarz, A.; Sakurovs, R.; Grigore, M.; Sayyafzadeh, M. Effect of maceral composition and coal rank on gas diffusion in Australian coals. Int. J. Coal Geol. 2017, 173, 65–75. [Google Scholar] [CrossRef]
- Hu, S.B.; Li, X.C.; Wang, E.Y. Experimental and Numerical Study on Scale Effects of Gas Emission from Coal Particles. Transp. Porous Media 2016, 114, 133–147. [Google Scholar] [CrossRef]
- Hu, H.X.; Du, L.; Xing, Y.F.; Li, X.C. Detailed study on self- and multicomponent diffusion of CO2-CH4 gas mixture in coal by molecular simulation. Fuel 2017, 187, 220–228. [Google Scholar] [CrossRef]
- Yan, J.X.; Jia, B.S.; Liu, B.G.; Zhang, J.Y. Simulation Study on Molecular Adsorption of Coal in Chicheng Coal Mine. Molecules 2023, 28, 3302. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z. Microstructure Evolution of Structural Coal and Its Influence on Kinetic Characteristics of Gas Adsorption and Desorption. Ph.D. Thesis, China University of Mining and Technology, Xuzhou, China, December 2020. [Google Scholar]
- Xiang, J.H.; Zeng, F.G.; Liang, H.Z.; Sun, B.L.; Zhang, L.; Li, M.F.; Jia, J.B. Model construction of the macromolecular structure of Yanzhou Coal and its molecular simulation. J. Fuel Chem. Technol. 2011, 39, 481–488. [Google Scholar] [CrossRef]
- Li, S.; Bai, Y.; Lin, H.; Yan, M.; Long, H.; Guo, D. Effect of N2/CO2 injection pressure on methane desorption in coal rocks containing gas. Nat. Gas Ind. 2021, 41, 80–89. [Google Scholar]
- Han, J. Molecular Simulation of Gas Adsorption, Desorption and Diffusion in Water-Bearing Coal Seams. Ph.D. Thesis, Southwest Petroleum University, Chengdu, China, June 2015. [Google Scholar]
- He, P. Molecular Simulation of Prediction Models for Adsorption and Diffusion Properties of Guest Molecules in Porous Materials. Ph.D. Thesis, Beijing University of Chemical Technology, Beijing, China, June 2012. [Google Scholar]
- Zhang, B. Molecular Simulation of Adsorption and Diffusion of CH4 on Kaolinite Surface of Coal Measures. Ph.D. Thesis, Taiyuan University of Technology, Taiyuan, China, June 2018. [Google Scholar]
- Xiang, J. Structural Characteristics of Different Coal Grades and Molecular Simulation of Interactions between Coal and CH4, CO2 and H2O. Ph.D. Thesis, Taiyuan University of Technology, Taiyuan, China, June 2013. [Google Scholar]

| Gas Type | Diffusivity at Different Pressures /×10−9 m2·s−1 | ||||
|---|---|---|---|---|---|
| 1 MPa | 3 MPa | 5 MPa | 7 MPa | 9 MPa | |
| CO2 | 1.04 | 0.96 | 0.89 | 0.85 | 0.83 |
| CH4 | 0.77 | 0.75 | 0.71 | 0.67 | 0.62 |
| Gas Type | Diffusivity at Different Pressures /×10−9 m2·s−1 | ||||
|---|---|---|---|---|---|
| 1 MPa | 3 MPa | 5 MPa | 7 MPa | 9 MPa | |
| CO2 | 1.04 | 0.96 | 0.89 | 0.85 | 0.83 |
| CH4 | 0.77 | 0.75 | 0.71 | 0.67 | 0.62 |
| Gas Type | Diffusion Coefficient under Different Water Content /×10−9 m2·s−1 | ||||
|---|---|---|---|---|---|
| 0% | 1% | 2% | 3% | 5% | |
| CO2 | 0.89 | 0.76 | 0.66 | 0.55 | 0.40 |
| CH4 | 0.71 | 0.63 | 0.54 | 0.46 | 0.33 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yan, J.; Jia, B.; Liu, B.; Zhang, J. Molecular Simulation of Coal Molecular Diffusion Properties in Chicheng Coal Mine. Molecules 2023, 28, 6933. https://doi.org/10.3390/molecules28196933
Yan J, Jia B, Liu B, Zhang J. Molecular Simulation of Coal Molecular Diffusion Properties in Chicheng Coal Mine. Molecules. 2023; 28(19):6933. https://doi.org/10.3390/molecules28196933
Chicago/Turabian StyleYan, Jingxue, Baoshan Jia, Baogang Liu, and Jinyi Zhang. 2023. "Molecular Simulation of Coal Molecular Diffusion Properties in Chicheng Coal Mine" Molecules 28, no. 19: 6933. https://doi.org/10.3390/molecules28196933
APA StyleYan, J., Jia, B., Liu, B., & Zhang, J. (2023). Molecular Simulation of Coal Molecular Diffusion Properties in Chicheng Coal Mine. Molecules, 28(19), 6933. https://doi.org/10.3390/molecules28196933






