Osmolytes Modulate Photoactivation of Phytochrome: Probing Protein Hydration
Abstract
1. Introduction
2. Results
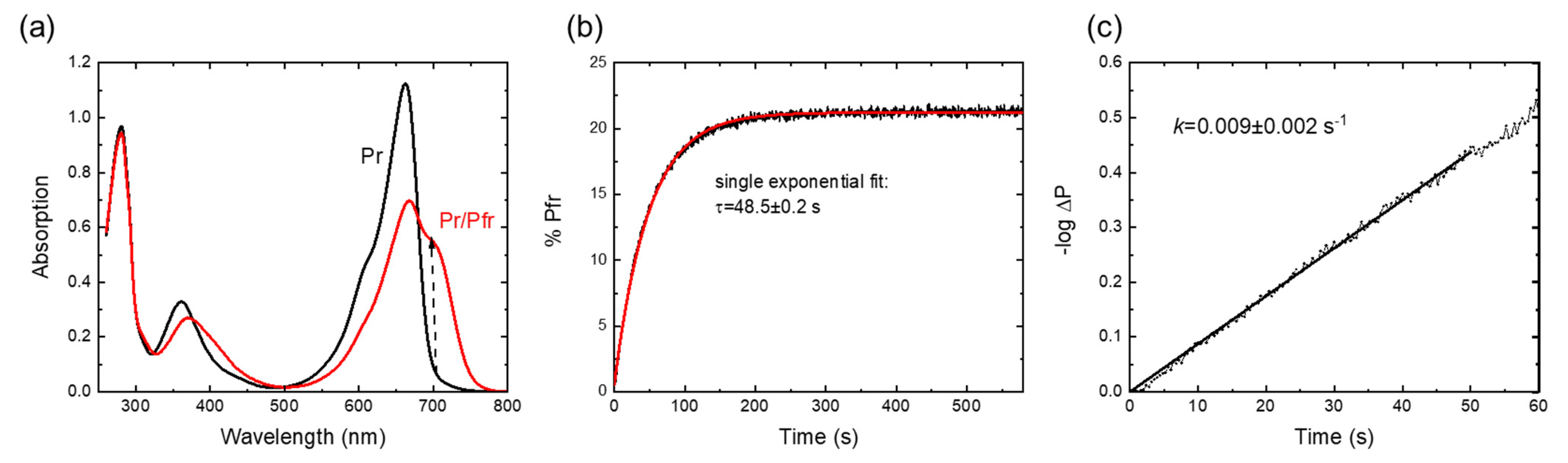
2.1. Spectroscopic Characterization and Pr→Pfr Photoconversion
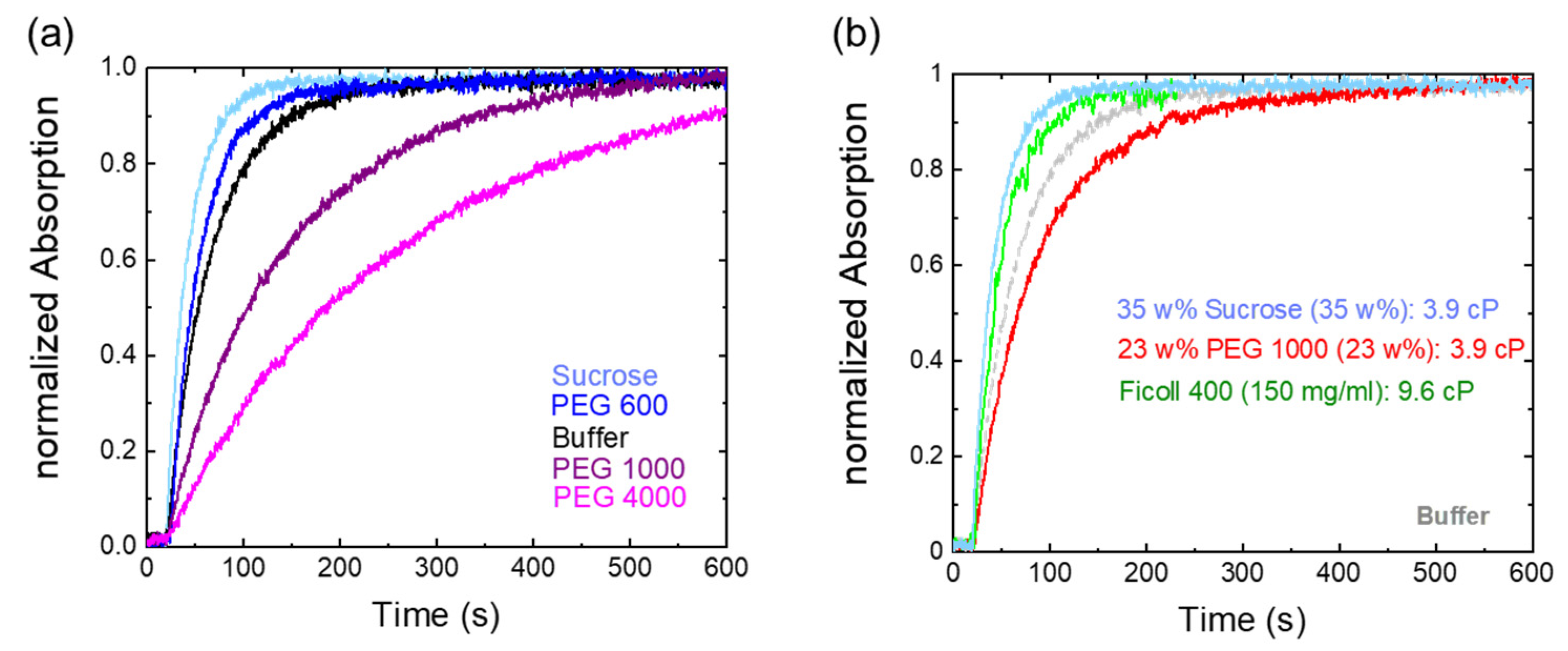
2.2. Osmolyte Effects on Pr→Pfr Photoconversion
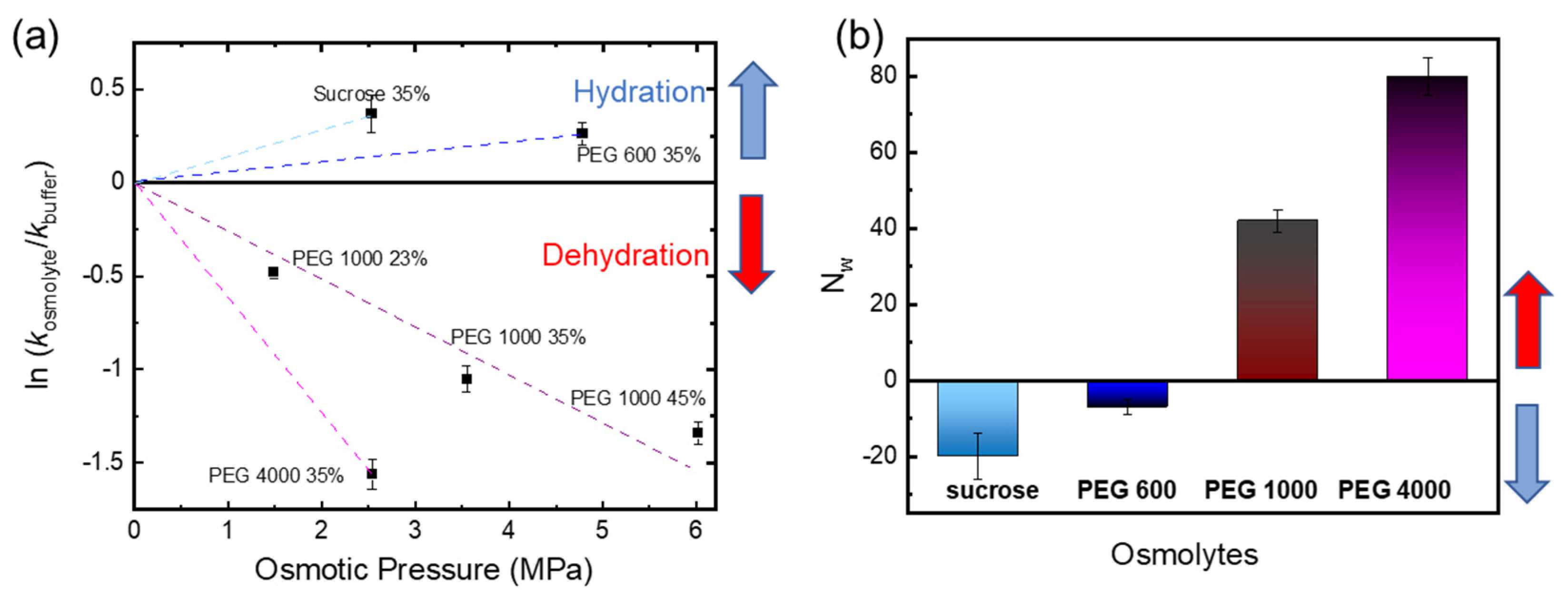
2.3. Determination of the Apparent Number of Water Molecules That Penetrate into Cph1 upon Photoconversion
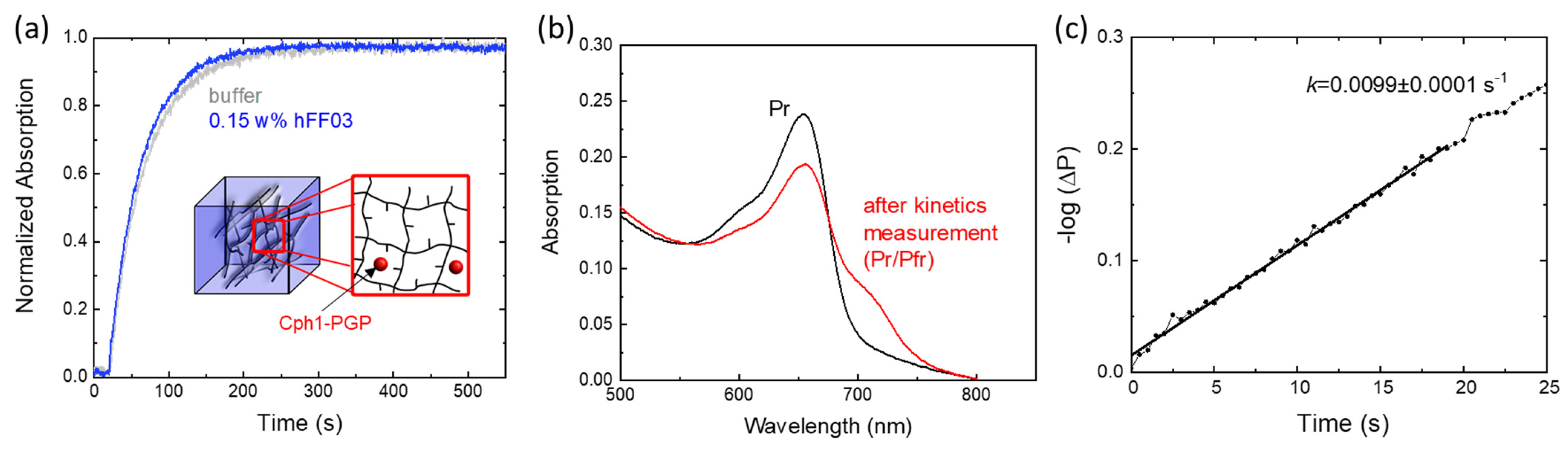
2.4. Hydrogel Effects on Pr→Pfr Photoconversion
3. Discussion
4. Materials and Methods
4.1. Phytochrome Expression and Purification
4.2. Synthesis of Peptide hFF03
4.3. Hydrogel Preparation
4.4. UV-Vis Spectroscopy and Data Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Quail, P.H. Phytochromes. Curr. Biol. 2010, 20, R504–R507. [Google Scholar] [CrossRef]
- Rockwell, N.C.; Su, Y.S.; Lagarias, J.C. Phytochrome structure and signaling mechanisms. Annu. Rev. Plant Biol. 2006, 57, 837–858. [Google Scholar] [CrossRef] [PubMed]
- Hughes, J.; Lamparter, T. Prokaryotes and Phytochrome. The Connection to Chromophores and Signaling1. Plant Physiol. 1999, 121, 1059–1068. [Google Scholar] [CrossRef] [PubMed]
- Hughes, J.; Lamparter, T.; Mittmann, F.; Hartmann, E.; Gärtner, W.; Wilde, A.; Börner, T. A prokaryotic phytochrome. Nature 1997, 386, 663. [Google Scholar] [CrossRef] [PubMed]
- Lamparter, T.; Esteban, B.; Hughes, J. Phytochrome Cph1 from the cyanobacterium Synechocystis PCC6803. Purification, assembly, and quaternary structure. Eur. J. Biochem. 2001, 268, 4720–4730. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Stensitzki, T.; Lang, C.; Hughes, J.; Mroginski, M.A.; Heyne, K. Ultrafast protein response in the Pfr state of Cph1 phytochrome. Photochem. Photobiol. Sci. 2023, 22, 919–930. [Google Scholar] [CrossRef] [PubMed]
- Velazquez Escobar, F.; Kneip, C.; Michael, N.; Hildebrandt, T.; Tavraz, N.; Gärtner, W.; Hughes, J.; Friedrich, T.; Scheerer, P.; Mroginski, M.A.; et al. The Lumi-R Intermediates of Prototypical Phytochromes. J. Phys. Chem. B 2020, 124, 4044–4055. [Google Scholar] [CrossRef]
- Rockwell, N.C.; Lagarias, J.C. A brief history of phytochromes. Chemphyschem 2010, 11, 1172–1180. [Google Scholar] [CrossRef]
- Takala, H.; Bjorling, A.; Berntsson, O.; Lehtivuori, H.; Niebling, S.; Hoernke, M.; Kosheleva, I.; Henning, R.; Menzel, A.; Ihalainen, J.A.; et al. Signal amplification and transduction in phytochrome photosensors. Nature 2014, 509, 245–248. [Google Scholar] [CrossRef]
- van Thor, J.J.; Borucki, B.; Crielaard, W.; Otto, H.; Lamparter, T.; Hughes, J.; Hellingwerf, K.J.; Heyn, M.P. Light-induced proton release and proton uptake reactions in the cyanobacterial phytochrome Cph1. Biochemistry 2001, 40, 11460–11471. [Google Scholar] [CrossRef]
- Yeh, K.C.; Wu, S.H.; Murphy, J.T.; Lagarias, J.C. A cyanobacterial phytochrome two-component light sensory system. Science 1997, 277, 1505–1508. [Google Scholar] [CrossRef] [PubMed]
- Borucki, B.; von Stetten, D.; Seibeck, S.; Lamparter, T.; Michael, N.; Mroginski, M.A.; Otto, H.; Murgida, D.H.; Heyn, M.P.; Hildebrandt, P. Light-induced proton release of phytochrome is coupled to the transient deprotonation of the tetrapyrrole chromophore. J. Biol. Chem. 2005, 280, 34358–34364. [Google Scholar] [CrossRef] [PubMed]
- Escobar, F.V.; Lang, C.; Takiden, A.; Schneider, C.; Balke, J.; Hughes, J.; Alexiev, U.; Hildebrandt, P.; Mroginski, M.A. Protonation-Dependent Structural Heterogeneity in the Chromophore Binding Site of Cyanobacterial Phytochrome Cph1. J. Phys. Chem. B 2017, 121, 47–57. [Google Scholar] [CrossRef]
- Sadeghi, M.; Balke, J.; Schneider, C.; Nagano, S.; Stellmacher, J.; Lochnit, G.; Lang, C.; Weise, C.; Hughes, J.; Alexiev, U. Transient Deprotonation of the Chromophore Affects Protein Dynamics Proximal and Distal to the Linear Tetrapyrrole Chromophore in Phytochrome Cph1. Biochemistry 2020, 59, 1051–1062. [Google Scholar] [CrossRef] [PubMed]
- Velazquez Escobar, F.; Hildebrandt, T.; Utesch, T.; Schmitt, F.J.; Seuffert, I.; Michael, N.; Schulz, C.; Mroginski, M.A.; Friedrich, T.; Hildebrandt, P. Structural Parameters Controlling the Fluorescence Properties of Phytochromes. Biochemistry 2014, 53, 20–29. [Google Scholar] [CrossRef]
- Bjorling, A.; Berntsson, O.; Lehtivuori, H.; Takala, H.; Hughes, A.J.; Panman, M.; Hoernke, M.; Niebling, S.; Henry, L.; Henning, R.; et al. Structural photoactivation of a full-length bacterial phytochrome. Sci. Adv. 2016, 2, e1600920. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi, M.; Balke, J.; Rafaluk-Mohr, T.; Alexiev, U. Long-Distance Protonation-Conformation Coupling in Phytochrome Species. Molecules 2022, 27, 8395. [Google Scholar] [CrossRef]
- Lehtivuori, H.; Rumfeldt, J.; Mustalahti, S.; Kurkinen, S.; Takala, H. Conserved histidine and tyrosine determine spectral responses through the water network in Deinococcus radiodurans phytochrome. Photochem. Photobiol. Sci. 2022, 21, 1975–1989. [Google Scholar] [CrossRef]
- Bellissent-Funel, M.C.; Hassanali, A.; Havenith, M.; Henchman, R.; Pohl, P.; Sterpone, F.; van der Spoel, D.; Xu, Y.; Garcia, A.E. Water Determines the Structure and Dynamics of Proteins. Chem. Rev. 2016, 116, 7673–7697. [Google Scholar] [CrossRef]
- Sass, H.J.; Büldt, G.; Gessenich, R.; Hehn, D.; Neff, D.; Schlesinger, R.; Berendzen, J.; Ormos, P. Structural alterations for proton translocation in the M state of wild-type bacteriorhodopsin. Nature 2000, 406, 649–653. [Google Scholar] [CrossRef]
- Alexiev, U.; Mollaaghababa, R.; Khorana, H.G.; Heyn, M.P. Evidence for long range allosteric interactions between the extracellular and cytoplasmic parts of bacteriorhodopsin from the mutant R82A and its second site revertant R82A/G231C. J. Biol. Chem. 2000, 275, 13431–13440. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Heyne, K.; Herbst, J.; Dominguez-Herradon, B.; Alexiev, U.; Diller, R. Reaction control in bacteriorhodopsin: Impact of arg82 and asp85 on the fast retinal isomerization, studied in the second site revertant arg82ala/gly231cys and various purple and blue forms of bacteriorhodopsin. J. Phys. Chem. B 2000, 104, 6053–6058. [Google Scholar] [CrossRef]
- Greenhalgh, D.A.; Subramaniam, S.; Alexiev, U.; Otto, H.; Heyn, M.P.; Khorana, H.G. Effect of Introducing Different Carboxylate-Containing Side-Chains at Position-85 on Chromophore Formation and Proton Transport in Bacteriorhodopsin. J. Biol. Chem. 1992, 267, 25734–25738. [Google Scholar] [CrossRef]
- Wolf, A.; Dragelj, J.; Wonneberg, J.; Stellmacher, J.; Balke, J.; Woelke, A.L.; Hodoscek, M.; Knapp, E.W.; Alexiev, U. The redox-coupled proton-channel opening in cytochrome c oxidase. Chem. Sci. 2020, 11, 3804–3811. [Google Scholar] [CrossRef]
- Chawla, U.; Perera, S.M.D.C.; Fried, S.D.E.; Eitel, A.R.; Mertz, B.; Weerasinghe, N.; Pitman, M.C.; Struts, A.V.; Brown, M.F. Activation of the G-Protein-Coupled Receptor Rhodopsin by Water. Angew. Chem. Int. Ed. 2021, 60, 2288–2295. [Google Scholar] [CrossRef] [PubMed]
- Losi, A.; Gartner, W. Solving Blue Light Riddles: New Lessons from Flavin-binding LOV Photoreceptors. Photochem. Photobiol. 2017, 93, 141–158. [Google Scholar] [CrossRef]
- Pennacchietti, F.; Abbruzzetti, S.; Losi, A.; Mandalari, C.; Bedotti, R.; Viappiani, C.; Zanacchi, F.C.; Diaspro, A.; Gartner, W. The dark recovery rate in the photocycle of the bacterial photoreceptor YtvA is affected by the cellular environment and by hydration. PLoS ONE 2014, 9, e107489. [Google Scholar] [CrossRef]
- Iwata, T.; Nozaki, D.; Yamamoto, A.; Koyama, T.; Nishina, Y.; Shiga, K.; Tokutomi, S.; Unno, M.; Kandori, H. Hydrogen Bonding Environment of the N3-H Group of Flavin Mononucleotide in the Light Oxygen Voltage Domains of Phototropins. Biochemistry 2017, 56, 3099–3108. [Google Scholar] [CrossRef]
- Chan, R.H.; Bogomolni, R.A. Structural water cluster as a possible proton acceptor in the adduct decay reaction of oat phototropin 1 LOV2 domain. J. Phys. Chem. B 2012, 116, 10609–10616. [Google Scholar] [CrossRef]
- Tobin, E.M.; Briggs, W.R.; Brown, P.K. The role of hydration in the phototransformation of phytochrome. Photochem. Photobiol. 1973, 18, 497–503. [Google Scholar] [CrossRef]
- Vertucci, C.W.; Vertucci, F.A.; Leopold, A.C. Water content and the conversion of phytochrome regulation of lettuce dormancy. Plant Physiol. 1987, 84, 887–890. [Google Scholar] [CrossRef] [PubMed]
- Kendrick, R.E.; Spruit, C.J. Phototransformations of phytochrome. Photochem. Photobiol. 1977, 26, 201–214. [Google Scholar] [CrossRef] [PubMed]
- von Horsten, S.; Straß, S.; Hellwig, N.; Gruth, V.; Klasen, R.; Mielcarek, A.; Linne, U.; Morgner, N.; Essen, L.-O. Mapping light-driven conformational changes within the photosensory module of plant phytochrome B. Sci. Rep. 2016, 6, 34366. [Google Scholar] [CrossRef]
- Song, C.; Lang, C.; Kopycki, J.; Hughes, J.; Matysik, J. NMR chemical shift pattern changed by ammonium sulfate precipitation in cyanobacterial phytochrome Cph1. Front. Mol. Biosci. 2015, 2, 42. [Google Scholar] [CrossRef] [PubMed]
- Altmayer, S.; Kohler, L.; Bielytskyi, P.; Gartner, W.; Matysik, J.; Wiebeler, C.; Song, C. Light- and pH-dependent structural changes in cyanobacteriochrome AnPixJg2. Photochem. Photobiol. Sci. 2022, 21, 447–469. [Google Scholar] [CrossRef]
- Parsegian, V.A.; Rand, R.P.; Rau, D.C. [3] Macromolecules and water: Probing with osmotic stress. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1995; Volume 259, pp. 43–94. [Google Scholar]
- Reid, C.; Rand, R.P. Probing protein hydration and conformational states in solution. Biophys. J. 1997, 72, 1022–1030. [Google Scholar] [CrossRef]
- Kuznetsova, I.M.; Turoverov, K.K.; Uversky, V.N. What macromolecular crowding can do to a protein. Int. J. Mol. Sci. 2014, 15, 23090–23140. [Google Scholar] [CrossRef]
- Ellis, R.J. Macromolecular crowding: Obvious but underappreciated. Trends Biochem. Sci. 2001, 26, 597–604. [Google Scholar] [CrossRef]
- Schmid, S.; Hugel, T. Controlling protein function by fine-tuning conformational flexibility. eLife 2020, 9, e57180. [Google Scholar] [CrossRef]
- Nagano, S.; Sadeghi, M.; Balke, J.; Fleck, M.; Heckmann, N.; Psakis, G.; Alexiev, U. Improved fluorescent phytochromes for in situ imaging. Sci. Rep. 2022, 12, 5587. [Google Scholar] [CrossRef]
- Pratt, L.H. Photochemistry of high molecular weight phytochrome in vitro. Photochem. Photobiol. 1975, 22, 33–36. [Google Scholar] [CrossRef]
- Mailliet, J. Structural Characterisation of Cyanobacterial Phytochrome Cph1. Ph.D. Dissertation, Universitätsbibliothek, Gießen, Germany, 2011. [Google Scholar]
- Emig, R.; Hoess, P.; Cai, H.; Kohl, P.; Peyronnet, R.; Weber, W.; Hörner, M. Benchmarking of Cph1 Mutants and DrBphP for Light-Responsive Phytochrome-Based Hydrogels with Reversibly Adjustable Mechanical Properties. Adv. Biol. 2022, 6, 2000337. [Google Scholar] [CrossRef] [PubMed]
- Hellmund, K.S.; von Lospichl, B.; Böttcher, C.; Ludwig, K.; Keiderling, U.; Noirez, L.; Weiß, A.; Mikolajczak, D.J.; Gradzielski, M.; Koksch, B. Functionalized peptide hydrogels as tunable extracellular matrix mimics for biological applications. Pept. Sci. 2021, 113, e24201. [Google Scholar] [CrossRef]
- Dargaville, B.L.; Hutmacher, D.W. Water as the often neglected medium at the interface between materials and biology. Nat. Commun. 2022, 13, 4222. [Google Scholar] [CrossRef]
- Sineshchekov, V.A. Extreme dehydration of plant tissues irreversibly converts the major and variable phyA′ into the minor and conserved phyA″. J. Photochem. Photobiol. B 2006, 85, 85–91. [Google Scholar] [CrossRef]
- Essen, L.O.; Mailliet, J.; Hughes, J. The structure of a complete phytochrome sensory module in the Pr ground state. Proc. Natl. Acad. Sci. USA 2008, 105, 14709–14714. [Google Scholar] [CrossRef]
- Lehtivuori, H.; Bhattacharya, S.; Angenent-Mari, N.M.; Satyshur, K.A.; Forest, K.T. Removal of Chromophore-Proximal Polar Atoms Decreases Water Content and Increases Fluorescence in a Near Infrared Phytofluor. Front. Mol. Biosci. 2015, 2, 65. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, Y. Rational Design of Smart Hydrogels for Biomedical Applications. Front. Chem. 2020, 8, 615665. [Google Scholar] [CrossRef]
- Andrushchenko, V.V.; Vogel, H.J.; Prenner, E.J. Optimization of the hydrochloric acid concentration used for trifluoroacetate removal from synthetic peptides. J. Pept. Sci. 2007, 13, 37–43. [Google Scholar] [CrossRef] [PubMed]

| Solvent | k (s−1) |
|---|---|
| Pure buffer | 0.010 ± 0.003 |
| Sucrose 35% | 0.014 ± 0.004 |
| PEG 600 35% | 0.013 ± 0.003 |
| PEG 1000 23% | 0.0062 ± 0.0004 |
| PEG 1000 35% | 0.0035 ± 0.0002 |
| PEG 1000 45% | 0.0026 ± 0.0001 |
| PEG 4000 35% | 0.0021 ± 0.0001 |
| Ficoll 400 (150 mg/mL) | 0.013 ± 0.001 |
| Osmolyte | ΔNw | ΔV° (L mol−1) | w (kJ mol−1) |
|---|---|---|---|
| Sucrose 35% | −20 ± 6 | −0.30 ± 0.08 | 0.8 ± 0.3 |
| PEG 600 35% | −7 ± 2 | −0.10 ± 0.02 | 0.5 ± 0.2 |
| PEG 1000 23% | 44 ± 3 | 0.80 ± 0.05 | −1.20 ± 0.08 |
| PEG 1000 35% | 40 ± 3 | 0.70 ± 0.05 | −1.9 ± 0.2 |
| PEG 4000 35% | 80 ± 5 | 1.50 ± 0.08 | −3.8 ± 0.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balke, J.; Díaz Gutiérrez, P.; Rafaluk-Mohr, T.; Proksch, J.; Koksch, B.; Alexiev, U. Osmolytes Modulate Photoactivation of Phytochrome: Probing Protein Hydration. Molecules 2023, 28, 6121. https://doi.org/10.3390/molecules28166121
Balke J, Díaz Gutiérrez P, Rafaluk-Mohr T, Proksch J, Koksch B, Alexiev U. Osmolytes Modulate Photoactivation of Phytochrome: Probing Protein Hydration. Molecules. 2023; 28(16):6121. https://doi.org/10.3390/molecules28166121
Chicago/Turabian StyleBalke, Jens, Paula Díaz Gutiérrez, Timm Rafaluk-Mohr, Jonas Proksch, Beate Koksch, and Ulrike Alexiev. 2023. "Osmolytes Modulate Photoactivation of Phytochrome: Probing Protein Hydration" Molecules 28, no. 16: 6121. https://doi.org/10.3390/molecules28166121
APA StyleBalke, J., Díaz Gutiérrez, P., Rafaluk-Mohr, T., Proksch, J., Koksch, B., & Alexiev, U. (2023). Osmolytes Modulate Photoactivation of Phytochrome: Probing Protein Hydration. Molecules, 28(16), 6121. https://doi.org/10.3390/molecules28166121





